Simultaneous Application of Ammonium and Nitrate Nitrogen Enhances Phytoremediation Efficiency by Mediating Biomass and Bioavailability of Lead and Cadmium in Salix linearistipularis
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Sites and Plant Material
2.2. Experimental Design
2.3. Sampling
2.4. Pb and Cd Analysis in Plant
2.5. Pb and Cd Analysis in the Soil
2.6. Soil Chemistry Properties Analysis
2.7. Metagenomic DNA Extraction and Sequencing
2.8. Taxonomic Annotation of Heavy Metal-Resistant Reads and Microbial Function
2.9. Statistical Analysis
3. Results
3.1. Biomass and Heavy Metals Accumulation in S. linearistipularis Following Nitrogen Fertilizer Application
3.2. Pb and Cd Concentration and Transport Coefficient in Plant Tissues
3.3. Soil Chemical Properties
3.4. Speciation of Pb and Cd in Soil
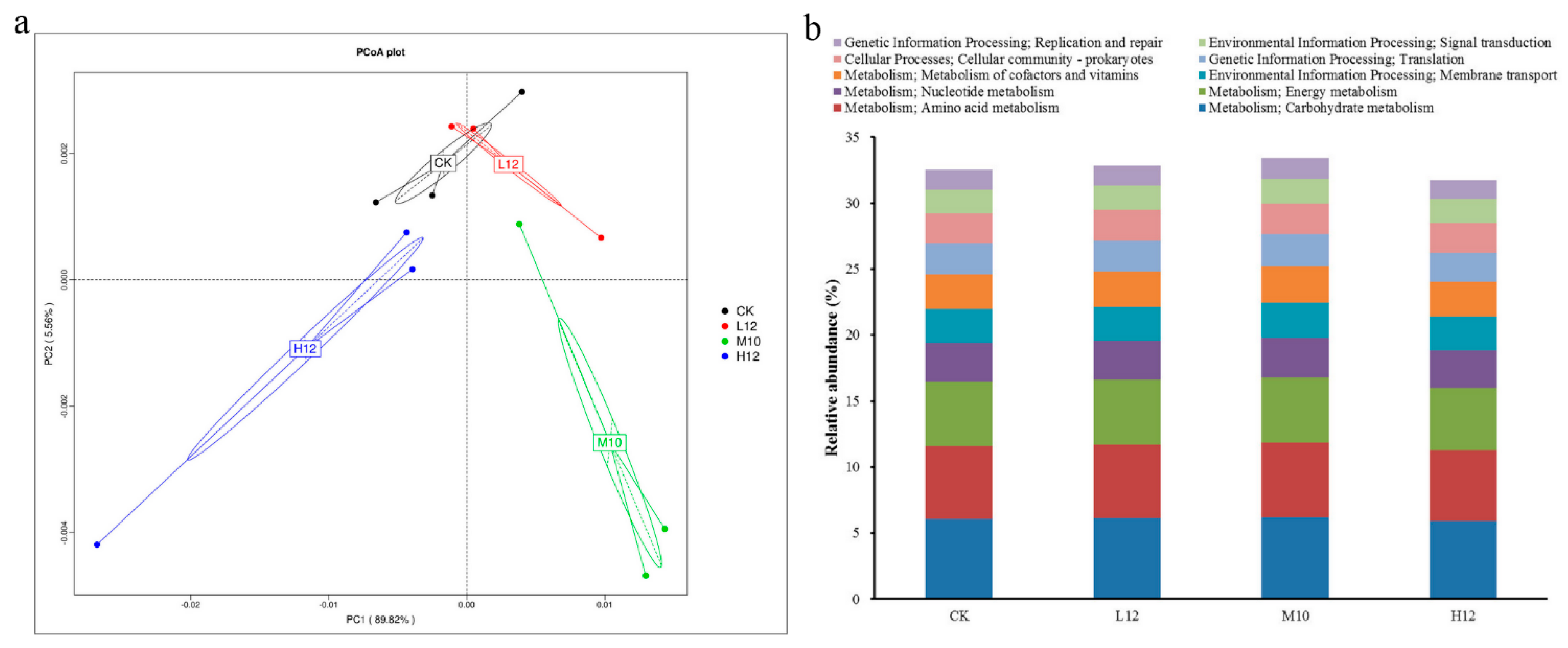
3.5. Soil Microbial Community Responses
3.6. Microbes’ Responses with Heavy Metal Resistance Genes
3.7. Microbial Metabolism
3.8. The Relationship of Microbial Community Characteristics in Relation to Soil Properties and Heavy Metal Accumulation in S. linearistipularis
4. Discussion
4.1. Enhancement of Pb and Cd Accumulation Linked to Increased Biomass and Heavy Metal Bioavailability in Soil
4.2. Changes in Microbial Community and Heavy Metal Resistance Microbes After Application of Nitrogen Fertilization
4.3. The Bioavailability of Pb and Cd Promoted by Microbial Metabolism
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yan, C.; Wang, F.; Geng, H.; Liu, H.; Pu, S.; Tian, Z.; Chen, H.; Zhou, B.; Yuan, R.; Yao, J. Integrating High-Throughput Sequencing and Metagenome Analysis to Reveal the Characteristic and Resistance Mechanism of Microbial Community in Metal Contaminated Sediments. Sci. Total Environ. 2020, 707, 136116. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.; Li, D.; Zhu, L.; Tang, X.; Wang, Y.; Mao, R.; Wu, J. Nitrate Increases Cadmium Accumulation in Sweet Sorghum for Improving Phytoextraction Efficiency Rather Than Ammonium. Front. Plant Sci. 2021, 12, 643116. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Dai, H.; Guo, B.; Noori, A.; Sun, W.; Wei, S. The Potential of Medicinal Plant Extracts in Improving the Phytoremediation Capacity of Solanum nigrum L. for heavy metal contaminated soil. Ecotoxicol. Environ. Saf. 2021, 220, 112411. [Google Scholar] [CrossRef] [PubMed]
- Gavrilescu, M. Enhancing Phytoremediation of Soils Polluted with Heavy Metals. Curr. Opin. Biotechnol. 2022, 74, 21–31. [Google Scholar] [CrossRef]
- Wang, A.; Li, X.; Hao, X.; Luo, X.; Chen, W.; Huang, Q. Ammonia Level Influences the Assembly of Dissimilatory Nitrate Reduction to Ammonia Bacterial Community in Soils under Different Heavy Metal Remediation Treatments. Sci. Total Environ. 2022, 838, 156393. [Google Scholar] [CrossRef]
- Yang, Y.; Xiong, J.; Chen, R.; Fu, G.; Chen, T.; Tao, L. Excessive Nitrate Enhances Cadmium (Cd) Uptake by up-Regulating the Expression of Osirt1 in Rice (Oryza sativa). Environ. Exp. Bot. 2016, 122, 141–149. [Google Scholar] [CrossRef]
- Bi, J.; Liu, X.; Liu, S.; Wang, Y.; Liu, M. Microstructural and Physiological Responses to Cadmium Stress under Different Nitrogen Forms in Two Contrasting Populus Clones. Environ. Exp. Bot. 2020, 169, 103897. [Google Scholar] [CrossRef]
- Yang, W.; Dai, H.; Skuza, L.; Wei, S. Enhanced Cd Phytoextraction by Solanum nigrum L. From Contaminated Soils Combined with the Application of N Fertilizers and Double Harvests. Toxics 2022, 5, 266. [Google Scholar] [CrossRef]
- Zeng, X.; Zou, D.; Wang, A.; Zhou, Y.; Liu, Y.; Li, Z.; Liu, F.; Wang, H.; Zeng, Q.; Xiao, Z. Remediation of Cadmium-Contaminated Soils Using Brassica napus: Effect of Nitrogen Fertilizers. J. Environ. Manag. 2020, 255, 109885. [Google Scholar] [CrossRef]
- Yu, F.; Li, Y.; Li, F.; Zhou, Z.; Chen, C.; Liang, X.; Li, C.; Liu, K. Nitrogen Fertilizers Promote Plant Growth and Assist in Manganese (Mn) Accumulation by Polygonum pubescens Blume Cultured in Mn Tailings Soil. Int. J. Phytoremediation 2019, 12, 1225–1233. [Google Scholar] [CrossRef]
- Ye, X.; Hu, H.; Li, H.; Xiong, Q.; Gao, H. Combined Nitrogen Fertilizer and Wheat Straw Increases the Cadmium Phytoextraction Efficiency of Tagetes patula. Ecotoxicol. Environ. Saf. 2019, 170, 210–217. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Zheng, H.; Xiang, X.; Wang, H.; Xie, M.; Liu, H.; Fang, Z.; Liu, L.; Du, S. N Fertilizers Promote Abscisic Acid-Catabolizing Bacteria to Enhance Heavy Metal Phytoremediation from Metalliferous Soils. Sci. Total Environ. 2023, 894, 164964. [Google Scholar] [CrossRef] [PubMed]
- Leite, T.d.S.; Monteiro, F.A. Nitrogen Form Regulates Cadmium Uptake and Accumulation in Tanzania Guinea Grass Used for Phytoextraction. Chemosphere 2019, 236, 124324. [Google Scholar] [CrossRef] [PubMed]
- Nakamaru, Y.M.; Matsuda, R.; Sonoda, T. Environmental Risks of Organic Fertilizer with Increased Heavy Metals (Cu and Zn) to Aquatic Ecosystems Adjacent to Farmland in the Northern Biosphere of Japan. Sci. Total Environ. 2023, 884, 163861. [Google Scholar] [CrossRef] [PubMed]
- Hou, D.; Wang, K.; Liu, T.; Wang, H.; Lin, Z.; Qian, J.; Lu, L.; Tian, S. Unique Rhizosphere Micro-Characteristics Facilitate Phytoextraction of Multiple Metals in Soil by the Hyperaccumulating Plant Sedum alfredii. Environ. Sci. Technol. 2017, 51, 5675–5684. [Google Scholar] [CrossRef]
- Islam, S.; Kormoker, T.; Idris, A.M.; Proshad, R.; Kabir, H.; Ustaoğlu, F. Plant–Microbe–Metal Interactions for Heavy Metal Bioremediation: A Review. Crop. Pasture Sci. 2021, 73, 181–201. [Google Scholar] [CrossRef]
- Niu, X.-Y.; Wang, S.-K.; Zhou, J.; Di, D.-L.; Sun, P.; Huang, D.-Z. Inoculation with Indigenous Rhizosphere Microbes Enhances Aboveground Accumulation of Lead in Salix integra Thunb. By Improving Transport Coefficients. Front. Microbiol. 2021, 12, 686812. [Google Scholar] [CrossRef]
- Barunawati, N.; Giehl, R.F.H.; Bauer, B.; von Wirén, N. The Influence of Inorganic Nitrogen Fertilizer Forms on Micronutrient Retranslocation and Accumulation in Grains of Winter Wheat. Front. Plant Sci. 2013, 4, 320. [Google Scholar] [CrossRef]
- Gu, Y.; Chen, X.; Shen, Y.; Chen, X.; He, G.; He, X.; Wang, G.; He, H.; Lv, Z. The Response of Nutrient Cycle, Microbial Community Abundance and Metabolic Function to Nitrogen Fertilizer in Rhizosphere Soil of Phellodendron chinense Schneid Seedlings. Front. Microbiol. 2023, 14, 1302775. [Google Scholar] [CrossRef]
- Niu, Y.; Zhang, M.; Bai, S.H.; Xu, Z.; Liu, Y.; Chen, F.; Guo, X.; Luo, H.; Wang, S.; Xie, J.; et al. Successive Mineral Nitrogen or Phosphorus Fertilization Alone Significantly Altered Bacterial Community Rather Than Bacterial Biomass in Plantation Soil. Appl. Microbiol. Biotechnol. 2020, 104, 7213–7224. [Google Scholar] [CrossRef]
- Lian, J.; Li, G.; Zhang, J.; Massart, S. Nitrogen Fertilization Affected Microbial Carbon Use Efficiency and Microbial Resource Limitations Via Root Exudates. Sci. Total Environ. 2024, 950, 174933. [Google Scholar] [CrossRef]
- Qasim, B.; Motelica-Heino, M.; Bourgerie, S.; Gauthier, A.; Morabito, D. Effect of Nitrate and Ammonium Fertilization on Zn, Pb, Cd Phytostabilization by Populus euramericana Dorskamp in Contaminated Technosol. Environ. Sci. Pollut. Res. 2015, 22, 18759–18771. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-F.; Li, W.-L.; Li, Q.-S.; Wang, L.-L.; He, T.; Wang, F.-P.; Xu, Z.-M. Nitrogen Fertilizer Management Affects Remobilization of the Immobilized Cadmium in Soil and Its Accumulation in Crop Tissues. Environ. Sci. Pollut. Res. 2021, 28, 31640–31652. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Wang, Q.; Chen, S.; Chen, F.; Lv, H.; Li, J.; Chen, Q.; Zhou, J.; Liang, B. Nitrate Leaching Is the Main Driving Factor of Soil Calcium and Magnesium Leaching Loss in Intensive Plastic-Shed Vegetable Production Systems. Agric. Water Manag. 2024, 293, 108708. [Google Scholar] [CrossRef]
- Zhang, F.; Jin, Q.; Peng, H.; Zhu, T. Soil Acidification in Moso Bamboo (Phyllostachys edulis) Forests and Changes of Soil Metal Ions (Cu, Pb) Concentration. Arch. Agron. Soil Sci. 2021, 67, 1799–1808. [Google Scholar] [CrossRef]
- Wang, Y.-F.; Wang, J.-F.; Xu, Z.-M.; She, S.-H.; Yang, J.-Q.; Li, Q.-S. L-Glutamic Acid Induced the Colonization of High-Efficiency Nitrogen-Fixing Strain Ac63 (Azotobacter chroococcum) in Roots of Amaranthus tricolor. Plant Soil 2020, 451, 357–370. [Google Scholar] [CrossRef]
- Feng, S.; Ren, L.; Sun, H.; Qiao, K.; Liu, S.; Zhou, A. Morphological and Physiological Responses of Two Willow Species from Different Habitats to Salt Stress. Sci. Rep. 2020, 10, 18228. [Google Scholar] [CrossRef]
- Feng, S.; Sun, H.; Ma, H.; Zhang, X.; Ma, S.; Qiao, K.; Zhou, A.; Bu, Y.; Liu, S. Sexual Differences in Physiological and Transcriptional Responses to Salinity Stress of Salix linearistipularis. Front. Plant Sci. 2020, 11, 517962. [Google Scholar] [CrossRef]
- Niu, X.; Zhou, J.; Wang, X.; Su, X.; Du, S.; Zhu, Y.; Yang, J.; Huang, D. Indigenous Bacteria Have High Potential for Promoting Salix integra Thunb. Remediation of Lead-Contaminated Soil by Adjusting Soil Properties. Front. Microbiol. 2020, 11, 924. [Google Scholar] [CrossRef]
- Rauret, G.; López-Sánchez, J.F.; Sahuquillo, A.; Rubio, R.; Davidson, C.; Ure, A.; Quevauviller, P. Improvement of the Bcr Three Step Sequential Extraction Procedure Prior to the Certification of New Sediment and Soil Reference Materials. J. Environ. Monit. 1999, 1, 57–61. [Google Scholar] [CrossRef]
- Zhang, J.; Buhe, C.; Yu, D.; Zhong, H.; Wei, Y. Ammonia Stress Reduces Antibiotic Efflux but Enriches Horizontal Gene Transfer of Antibiotic Resistance Genes in Anaerobic Digestion. Bioresour. Technol. 2020, 295, 122191. [Google Scholar] [CrossRef] [PubMed]
- Pal, C.; Bengtsson-Palme, J.; Rensing, C.; Kristiansson, E.; Larsson, D.G.J. Bacmet: Antibacterial Biocide and Metal Resistance Genes Database. Nucleic Acids Res. 2014, 42, D737–D743. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S.; Kawashima, S.; Okuno, Y.; Hattori, M. The Kegg Resource for Deciphering the Genome. Nucleic Acids Res. 2004, 32, D277–D280. [Google Scholar] [CrossRef] [PubMed]
- Huo, K.; Shangguan, X.; Xia, Y.; Shen, Z.; Chen, C. Excess Copper Inhibits the Growth of Rice Seedlings by Decreasing Uptake of Nitrate. Ecotoxicol. Environ. Saf. 2020, 190, 110105. [Google Scholar] [CrossRef]
- Xia, Z.; Xue, C.; Liu, R.; Hui, Q.; Hu, B.; Rennenberg, H. Lead Accumulation and Concomitant Reactive Oxygen Species (Ros) Scavenging in Robinia pseudoacacia Are Dependent on Nitrogen Nutrition. Plant Physiol. Biochem. 2025, 219, 109388. [Google Scholar] [CrossRef]
- Wang, S.; Lin, W.; Ye, Q.; Lv, W.; Liao, P.; Yu, J.; Mu, C.; Wu, L.; Muneer, M.A.; Zhang, Y.; et al. Effects of Different Nitrogen Fertilizer Rates on Soil Magnesium Leaching in Tea Garden. J. Soil Sci. Plant Nutr. 2024, 24, 6630–6640. [Google Scholar] [CrossRef]
- Shi, Y.; Zhang, X.; Wang, Z.; Xu, Z.; He, C.; Sheng, L.; Liu, H.; Wang, Z. Shift in Nitrogen Transformation in Peatland Soil by Nitrogen Inputs. Sci. Total Environ. 2021, 764, 142924. [Google Scholar] [CrossRef]
- Chen, W.; Peng, L.; Hu, K.; Zhang, Z.; Peng, C.; Teng, C.; Zhou, K. Spectroscopic Response of Soil Organic Matter in Mining Area to Pb/Cd Heavy Metal Interaction: A Mirror of Coherent Structural Variation. J. Hazard. Mater. 2020, 393, 122425. [Google Scholar] [CrossRef]
- Yang, W.; Dai, H.; Skuza, L.; Wei, S. Strengthening Role and the Mechanism of Optimum Nitrogen Addition in Relation to Solanum nigrum L. Cd Hyperaccumulation in Soil. Ecotoxicol. Environ. Saf. 2019, 182, 109444. [Google Scholar] [CrossRef]
- Vazquez, A.; Recalde, L.; Cabrera, A.; Groppa, M.D.; Benavides, M.P. Does Nitrogen Source Influence Cadmium Distribution in Arabidopsis Plants? Ecotoxicol. Environ. Saf. 2020, 191, 110163. [Google Scholar] [CrossRef]
- Yu, L.; Wang, X.; Li, X.; Wang, Y.; Kang, H.; Chen, G.; Fan, X.; Sha, L.; Zhou, Y.; Zeng, J. Protective Effect of Different Forms of Nitrogen Application on Cadmium-Induced Toxicity in Wheat Seedlings. Environ. Sci. Pollut. Res. 2019, 26, 13085–13094. [Google Scholar] [CrossRef]
- Wang, Y.; Bao, G. Diversity of Prokaryotic Microorganisms in Alkaline Saline Soil of the Qarhan Salt Lake Area in the Qinghai–Tibet Plateau. Sci. Rep. 2022, 12, 3365. [Google Scholar] [CrossRef]
- Ikeda, S.; Okazaki, K.; Tsurumaru, H.; Suzuki, T.; Hirafuji, M. Effects of Different Types of Additional Fertilizers on Root-Associated Microbes of Napa Cabbage Grown in an Andosol Field in Japan. Microbes Environ. 2022, 37, ME22013. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ji, R.; Zhang, H.; Christie, P.; Feng, G.; Li, X.; Gai, J. Stoichiometric Analysis of an Arable Crop–Soil–Microbe System after Repeated Fertilizer and Compost Application for 10 Years. J. Soils Sediments 2021, 21, 1466–1475. [Google Scholar] [CrossRef]
- Lin, Z.; Dou, C.; Li, Y.; Wang, H.; Niazi, N.K.; Zhang, S.; Liu, D.; Zhao, K.; Fu, W.; Li, Y.; et al. Nitrogen Fertilizer Enhances Zinc and Cadmium Uptake by Hyperaccumulator Sedum alfredii Hance. J. Soils Sediments 2020, 20, 320–329. [Google Scholar] [CrossRef]
- Kuffner, M.; Puschenreiter, M.; Wieshammer, G.; Gorfer, M.; Sessitsch, A. Rhizosphere Bacteria Affect Growth and Metal Uptake of Heavy Metal Accumulating Willows. Plant Soil 2008, 304, 35–44. [Google Scholar] [CrossRef]
- Ming, H.; Zan, S.; Liu, J.; Su, J.; Ren, K.; Lin, J.; Chen, Q.; Shi, Y.; Shi, T.; Fan, J. Promoting the Denitrification Process by Heavy Metals in Liaohe Estuary Sediment. Mar. Pollut. Bull. 2024, 203, 116408. [Google Scholar] [CrossRef]
- Tian, J.; Du, Y.; Yu, C.; Liu, W.; Zou, R.; Zhao, Y.; Zhang, T.; Jiang, Y.; Tian, Z. The Influences of Heavy Metals on Soil Microbial C, N, P Cycling and Heavy Metal Resistance under Different Fertilization Regimes. Environ. Pollut. 2025, 370, 125915. [Google Scholar] [CrossRef]
- Wu, Z.; Gao, G.; Wang, Y. Effects of Soil Properties, Heavy Metals, Pbdes on Microbial Community of E-Waste Contaminated Soil. Ecotoxicol. Environ. Saf. 2019, 180, 705–714. [Google Scholar] [CrossRef]
- Lin, Z.; Qiao, Y.; Xu, K.; Lu, L.; Shu, Q.-Y.; Tian, S. The Endophytic Fungus Serendipita Indica Reshapes Rhizosphere Soil Microbiota to Improve Salix suchowensis Growth and Phytoremediation. J. Hazard. Mater. 2025, 495, 138620. [Google Scholar] [CrossRef]
- Zhang, H.; Jiang, N.; Zhang, S.; Zhu, X.; Wang, H.; Xiu, W.; Zhao, J.; Liu, H.; Zhang, H.; Yang, D. Soil Bacterial Community Composition Is Altered More by Soil Nutrient Availability Than Ph Following Long-Term Nutrient Addition in a Temperate Steppe. Front. Microbiol. 2024, 15, 1455891. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, J.; Sapkale, V.; Shah, M.; Rajput, V.; Mehetre, G.; Agawane, S.; Kamble, S.; Dharne, M. Metagenome Sequencing to Unveil Microbial Community Composition and Prevalence of Antibiotic and Metal Resistance Genes in Hypersaline and Hyperalkaline Lonar Lake, India. Ecol. Indic. 2020, 110, 105827. [Google Scholar] [CrossRef]
- Puthusseri, R.M.; Nair, H.P.; Johny, T.K.; Bhat, S.G. Insights into the Response of Mangrove Sediment Microbiomes to Heavy Metal Pollution: Ecological Risk Assessment and Metagenomics Perspectives. J. Environ. Manag. 2021, 298, 113492. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jiang, Y.; Huang, H.; Mou, L.; Ru, J.; Zhao, J.; Xiao, S. Long-Term and High-Concentration Heavy-Metal Contamination Strongly Influences the Microbiome and Functional Genes in Yellow River Sediments. Sci. Total Environ. 2018, 637, 1400–1412. [Google Scholar] [CrossRef]
- Gong, W.-J.; Niu, Z.-F.; Wang, X.-R.; Zhao, H.-P. How the Soil Microbial Communities and Activities Respond to Long-Term Heavy Metal Contamination in Electroplating Contaminated Site. Microorganisms 2021, 9, 362. [Google Scholar] [CrossRef]
- Gan, C.-D.; Cui, S.-F.; Wu, Z.-Z.; Yang, J.-Y. Multiple Heavy Metal Distribution and Microbial Community Characteristics of Vanadium-Titanium Magnetite Tailing Profiles under Different Management Modes. J. Hazard. Mater. 2022, 429, 128032. [Google Scholar] [CrossRef]
- Zhang, J.; Mao, C.; Khan, A.; Zhao, S.; Gao, T.; Redina, M.M.; Zhang, Q.; Song, P.; Liu, P.; Li, X. Enhanced Methane Production by Using Phytoremediated Halogeton Glomeratus as Substrate Via Anaerobic Digestion. Renew. Energy 2022, 194, 28–39. [Google Scholar] [CrossRef]
- Kou, S.; Vincent, G.; Gonzalez, E.; Pitre, F.E.; Labrecque, M.; Brereton, N.J.B. The Response of a 16s Ribosomal Rna Gene Fragment Amplified Community to Lead, Zinc, Copper Pollution in a Shanghai Field Trial. Front. Microbiol. 2018, 9, 366. [Google Scholar] [CrossRef]
- Cui, J.; Zhu, R.; Wang, X.; Xu, X.; Ai, C.; He, P.; Liang, G.; Zhou, W.; Zhu, P. Effect of High Soil C/N Ratio and Nitrogen Limitation Caused by the Long-Term Combined Organic-Inorganic Fertilization on the Soil Microbial Community Structure and Its Dominated Soc Decomposition. J. Environ. Manag. 2022, 303, 114155. [Google Scholar] [CrossRef]
- Achor, S.; Aravis, C.; Heaney, N.; Odion, E.; Lin, C. Response of Organic Acid-Mobilized Heavy Metals in Soils to Biochar Application. Geoderma 2020, 378, 114628. [Google Scholar] [CrossRef]
- Wang, J.-F.; Zhu, C.-Y.; Weng, B.-S.; Mo, P.-W.; Xu, Z.-J.; Tian, P.; Cui, B.-S.; Bai, J.-H. Regulation of Heavy Metals Accumulated by Acorus calamus L. In Constructed Wetland through Different Nitrogen Forms. Chemosphere 2021, 281, 130773. [Google Scholar] [CrossRef]
- Khodamoradi, K.; Khoshgoftarmanesh, A.H.; Maibody, S.A.M.M. Root Uptake and Xylem Transport of Cadmium in Wheat and Triticale as Affected by Exogenous Amino Acids. Crop. Pasture Sci. 2017, 68, 415–420. [Google Scholar] [CrossRef]
- Kocaman, A. Combined Interactions of Amino Acids and Organic Acids in Heavy Metal Binding in Plants. Plant Signal. Behav. 2023, 18, 2064072. [Google Scholar] [CrossRef]
- Liang, Y.; Yang, C.; Sainju, U.M.; Zhang, N.; Zhao, F.; Wang, W.; Wang, J. Differential Responses of Soil Microbial N-Cycling Functional Genes to 35 Yr Applications of Chemical Fertilizer and Organic Manure in Wheat Field Soil on Loess Plateau. Agronomy 2023, 13, 1516. [Google Scholar] [CrossRef]





| Treatments | Total (N) (kg·hm−2·year−1) | Total (N) (g·pot−1·year−1) | NH4+ (g·pot−1·year−1) | NO3− (g·pot−1·year−1) | Ratio |
|---|---|---|---|---|---|
| CK | 0 | 0 | 0 | 0 | 0 |
| L10 | 60 | 0.4823 | 0.4823 | 0 | 1:0 |
| L21 | 60 | 0.4823 | 0.3215 | 0.1608 | 2:1 |
| L11 | 60 | 0.4823 | 0.24115 | 0.24115 | 1:1 |
| L12 | 60 | 0.4823 | 0.1608 | 0.3215 | 1:2 |
| L01 | 60 | 0.4823 | 0 | 0.4823 | 0:1 |
| M10 | 120 | 0.9646 | 0.9646 | 0 | 1:0 |
| M21 | 120 | 0.9646 | 0.6431 | 0.3215 | 2:1 |
| M11 | 120 | 0.9646 | 0.4823 | 0.4823 | 1:1 |
| M12 | 120 | 0.9646 | 0.3215 | 0.6431 | 1:2 |
| M01 | 120 | 0.9646 | 0 | 0.9646 | 0:1 |
| H10 | 200 | 1.6077 | 1.6077 | 0 | 1:0 |
| H21 | 200 | 1.6077 | 1.0718 | 0.5359 | 2:1 |
| H11 | 200 | 1.6077 | 0.80385 | 0.80385 | 1:1 |
| H12 | 200 | 1.6077 | 0.5359 | 1.0718 | 1:2 |
| H01 | 200 | 1.6077 | 0 | 1.6077 | 0:1 |
| Treatments | Pb Concentrations/mg·kg−1 | Cd Concentrations/mg·kg−1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Lateral Root | Taproot | Cutting | Branch | Leaf | Lateral Root | Taproot | Cutting | Branch | Leaf | |
| CK | 24.37 ± 2.1 b | 16.76 ± 1.94 b | 5.54 ± 1.44 de | 3.56 ± 0.29 bc | 9.26 ± 1.02 ab | 24 ± 4.17 de | 18.51 ± 1.78 def | 14.59 ± 1.72 bcd | 19.28 ± 1.59 ab | 46.4 ± 1.54 bc |
| L10 | 26.1 ± 5.71 b | 14.74 ± 1.4 bc | 5.61 ± 0.57 de | 4.43 ± 0.96 ab | 9.14 ± 0.93 ab | 23.71 ± 2.24 de | 14.28 ± 0.66 gh | 12.88 ± 2.34 d | 20.55 ± 2.18 a | 47.05 ± 1.59 b |
| L21 | 36.7 ± 0 a | 17.21 ± 3.59 bc | 8.55 ± 0.41 a | 1.77 ± 0.52 de | 10.81 ± 0.7 ab | 34.42 ± 0 b | 21.24 ± 4.09 cd | 8.86 ± 1.25 e | 21.14 ± 3.45 a | 47.65 ± 3.41 b |
| L11 | 39.85 ± 3.58 a | 16.05 ± 3.31 bc | 7.55 ± 0.37 ab | 3.92 ± 0.72 abc | 12.44 ± 2.58 a | 31.63 ± 2.1 bcd | 15.63 ± 1.84 fgh | 16.03 ± 3.06 abcd | 18.52 ± 2.88 ab | 46.91 ± 7.61 bc |
| L12 | 30.63 ± 1.91 b | 12.17 ± 0.54 bc | 8.19 ± 1.01 ab | 2.75 ± 0.48 cd | 9.04 ± 0.92 ab | 32.97 ± 4.78 bc | 13.17 ± 1.52 h | 15.28 ± 0.44 abcd | 19.2 ± 4.09 ab | 35.41 ± 2.61 d |
| L01 | 24.97 ± 4.9 b | 13.02 ± 1.47 bc | 3.29 ± 0.21 g | 1.43 ± 0.45 e | 9.99 ± 1.15 ab | 26.57 ± 2.49 bcde | 14.4 ± 1.95 gh | 17.34 ± 1.69 abc | 17.55 ± 2.48 b | 40.43 ± 2.03 bcd |
| M10 | 28.13 ± 1.4 b | 11.62 ± 1.71 c | 7.42 ± 2.02 ab | 2.06 ± 1.81 de | 8.3 ± 1.3 bc | 30.76 ± 6.83 bcde | 14.54 ± 1.2 fgh | 18.38 ± 1.27 a | 20.4 ± 0.86 a | 40.54 ± 4.61 bcd |
| M21 | 36.07 ± 2.62 a | 13.2 ± 2.16 c | 7.12 ± 0.27 abc | 2.7 ± 0.72 cd | 6.36 ± 0.35 c | 43.78 ± 2.44 a | 21.32 ± 0.38 cd | 18.21 ± 0.16 a | 21.45 ± 1.26 a | 36.63 ± 7.15 d |
| M11 | 33.5 ± 3.63 a | 14.36 ± 1.38 c | 7.68 ± 1.12 ab | 4.77 ± 0.51 ab | 7.73 ± 1.36 c | 33.38 ± 4.9 bc | 16.91 ± 1.35 efgh | 18.3 ± 1.82 a | 21.89 ± 2.34 ab | 45.63 ± 3.09 bc |
| M12 | 29.03 ± 7.96 b | 12.87 ± 1.68 c | 6.91 ± 0.58 bcd | 3.85 ± 0.54 abc | 5.88 ± 0.18 c | 22.41 ± 8.53 e | 16.03 ± 0.68 fgh | 14.33 ± 1.02 cd | 20.99 ± 2.01 ab | 42.07 ± 3.2 bcd |
| M01 | 25.48 ± 1 b | 15.22 ± 4.45 c | 3.13 ± 0.73 g | 3.89 ± 0.93 abc | 5.65 ± 1 c | 25.32 ± 6.86 cde | 17.66 ± 4.6 defg | 12.7 ± 1.05 d | 19.96 ± 3.08 b | 45.89 ± 5.8 bc |
| H10 | - | 20.39 ± 5.59 a | 3.64 ± 0.34 fg | 4.61 ± 0.79 ab | 7.77 ± 1.4 bc | - | 35.27 ± 1.34 a | 17.89 ± 3.91 ab | 24.97 ± 2.39 a | 56.01 ± 3.75 a |
| H21 | - | 18.28 ± 1.2 a | 4.84 ± 0.49 ef | 4.8 ± 0.69 ab | 7.68 ± 0.46 bc | - | 25.22 ± 2.71 b | 18.1 ± 1.64 a | 22.31 ± 3.66 a | 47.8 ± 3.63 b |
| H12 | - | 18.98 ± 0.48 a | 5.94 ± 0.77 cde | 4.09 ± 0.58 ab | 8.32 ± 3.12 bc | - | 23.31 ± 2.32 bc | 15.3 ± 2.54 abcd | 18.33 ± 2.51 ab | 34.18 ± 7.3 d |
| H01 | - | 20.94 ± 1.69 a | 5.16 ± 0.69 e | 4.97 ± 0.46 a | 8.23 ± 1.11 bc | - | 20.2 ± 2.38 cde | 14.54 ± 0.41 bcd | 16.59 ± 1.02 b | 38.57 ± 7.76 cd |
| P (L) | ns | * | * | * | * | ns | * | * | ns | ns |
| P (R) | * | ns | * | * | * | * | * | * | * | * |
| P (L*R) | ns | ns | * | * | ns | * | * | * | ns | * |
| Treatments | pH | SOM/g·kg−1 | CEC/cmol·Kg−1 | NH4+-N/mg·kg−1 | NO3−-N/mg·kg−1 |
|---|---|---|---|---|---|
| CK | 7.82 ± 0.01 a | 11.74 ± 0.22 de | 11.32 ± 1.39 a | 3.12 ± 0.23 de | 19.0 ± 1.00 gh |
| L10 | 7.69 ± 0.04 cd | 14.32 ± 0.49 a | 10.19 ± 0.56 b | 3.1 ± 0.12 de | 15.07 ± 0.23 h |
| L21 | 7.8 ± 0.06 b | 12.9 ± 0.04 bcd | 10.26 ± 0.74 bc | 3.12 ± 0.01 de | 16.24 ± 0.35 gh |
| L11 | 7.73 ± 0.1 bcd | 13.63 ± 0.03 abc | 10.17 ± 1.68 bc | 2.77 ± 0.08 de | 15.16 ± 0.57 h |
| L12 | 7.64 ± 0.03 d | 12.72 ± 0.34 bcd | 9.2 ± 1.31 c | 2.56 ± 0.2 e | 15.17 ± 0.85 h |
| L01 | 7.76 ± 0.04 bc | 12.63 ± 0.86 cde | 9.91 ± 1.16 c | 3.78 ± 0.29 bc | 16.77 ± 1.97 gh |
| M10 | 7.54 ± 0.04 cd | 13.93 ± 1.89 ab | 10.83 ± 1.48 b | 4.27 ± 0.44 ab | 19.1 ± 0.42 gh |
| M21 | 7.64 ± 0.1 b | 13.57 ± 0.02 abc | 8.74 ± 0.17 bc | 4.16 ± 0.15 b | 35.07 ± 1.34 d |
| M11 | 7.52 ± 0.09 bcd | 12.93 ± 1.13 bcd | 8.33 ± 0.67 bc | 4.77 ± 0.28 a | 24.23 ± 0.52 f |
| M12 | 7.54 ± 0.08 d | 12.4 ± 0.35 cde | 8.34 ± 1.19 c | 2.9 ± 0.36 de | 19.78 ± 1.46 g |
| M01 | 7.65 ± 0.05 bc | 11.83 ± 0.33 de | 8.42 ± 0.86 c | 3.31 ± 0.06 cd | 26.46 ± 0.23 ef |
| H10 | 7.49 ± 0.09 cd | 11.41 ± 0.66 e | 9.6 ± 0.23 b | 4.22 ± 0.17 ab | 49.58 ± 4.62 b |
| H21 | 7.58 ± 0.13 b | 11.41 ± 0.37 e | 9.49 ± 0.52 bc | 3.19 ± 0.31 d | 28.12 ± 3.15 e |
| H12 | 7.48 ± 0.08 d | 11.91 ± 0.41 de | 8.91 ± 0.11 c | 2.8 ± 0.12 de | 42.54 ± 1.76 c |
| H01 | 7.51 ± 0.2 bc | 12.06 ± 0.36 de | 7.72 ± 0.78 c | 4.15 ± 0.47 b | 71.47 ± 0.15 a |
| p (L) | * | * | * | * | * |
| p (R) | * | * | * | * | * |
| p (L*R) | ns | * | ns | * | * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhou, J.; Di, D.; Zhang, Y.; Gao, Z.; Niu, X.; Huang, D.; Zhu, K. Simultaneous Application of Ammonium and Nitrate Nitrogen Enhances Phytoremediation Efficiency by Mediating Biomass and Bioavailability of Lead and Cadmium in Salix linearistipularis. Forests 2026, 17, 364. https://doi.org/10.3390/f17030364
Zhou J, Di D, Zhang Y, Gao Z, Niu X, Huang D, Zhu K. Simultaneous Application of Ammonium and Nitrate Nitrogen Enhances Phytoremediation Efficiency by Mediating Biomass and Bioavailability of Lead and Cadmium in Salix linearistipularis. Forests. 2026; 17(3):364. https://doi.org/10.3390/f17030364
Chicago/Turabian StyleZhou, Jian, Dongliu Di, Yaoyao Zhang, Zhuotian Gao, Xiaoyun Niu, Dazhuang Huang, and Keye Zhu. 2026. "Simultaneous Application of Ammonium and Nitrate Nitrogen Enhances Phytoremediation Efficiency by Mediating Biomass and Bioavailability of Lead and Cadmium in Salix linearistipularis" Forests 17, no. 3: 364. https://doi.org/10.3390/f17030364
APA StyleZhou, J., Di, D., Zhang, Y., Gao, Z., Niu, X., Huang, D., & Zhu, K. (2026). Simultaneous Application of Ammonium and Nitrate Nitrogen Enhances Phytoremediation Efficiency by Mediating Biomass and Bioavailability of Lead and Cadmium in Salix linearistipularis. Forests, 17(3), 364. https://doi.org/10.3390/f17030364






