A Demographic Imbalance of Tree Populations in the Managed Part of Białowieża Forest (NE Poland): Implications for Nature-Oriented Forestry
Abstract
1. Introduction
- To what extent has forest management carried out until recently in BF influenced the current demographic status (reflected by the respective actual size distribution) of individual tree species?
- Compared with an ideal equilibrium structure, ensuring the best conservation effects, how many surplus or deficit trees exist for each species and DBH class, and what conservation measures are needed to restore the optimal structure?
2. Material and Methods
2.1. Study Area
2.2. Determination of the Actual DBH Distributions of Tree Species
2.3. Construction of Equilibrium/Sustainable Tree Size Distributions
2.4. Calculation of Surplus (Total and Reduced) and Deficit Trees of Individual Species by Diameter Class and Determination of the Area of Regeneration Units by Species and in Total
3. Results
3.1. Actual vs. Theoretical DBH Distributions
3.2. Overall Amount and Distribution of Total and Reduced Surplus Trees
3.3. Overall Amount and Distribution of Deficit Trees
3.4. Estimated Area of Regeneration Units (Regeneration Patches) Required to Reduce Species-Specific Deficits in the Smallest Diameter Class
4. Discussion
4.1. Using Equilibrium Distributions as an Indicator of the Demographic Sustainability of Tree Populations
4.2. Common Deviations Between Actual and Theoretical Tree Size Distributions: Major Causes and Implications
4.3. Measures Which Need Be Taken to Bring the Local Tree Populations Closer to This Desired State
4.4. Conserving Natural Values of BF: What Strategy to Choose?
5. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kannan, R.; James, D.A. Effects of climate change on global biodiversity: A review of key literature. Trop. Ecol. 2009, 50, 31–39. [Google Scholar]
- Wilson, E.O. Half-Earth: Our Planet’s Fight for Life; Liveright Publishing Corp: New York, NY, USA, 2016. [Google Scholar]
- IPBES. Summary for Policymakers of the Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; IPBES Secretariat: Bonn, Germany, 2019. [CrossRef]
- Sala, O.E.; Chapin, F.S.; Armesto, J.J.; Berlow, E.; Bloomfield, J.; Dirzo, R.; Huber-Sanwald, E.; Huenneke, L.F.; Jackson, R.B.; Kinzig, A.; et al. Global biodiversity scenarios for the year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef]
- Wilkening, J.L.; Magness, D.R.; Thompson, L.M.; Lynch, A.J. A brave new world: Managing for biodiversity conservation under ecosystem transformation. Land 2023, 12, 1556. [Google Scholar] [CrossRef]
- Galatowitsch, S.; Frelich, L.; Phillips-Mao, L. Regional climate change adaptation strategies for biodiversity conservation in a midcontinental region of North America. Biol. Conserv. 2009, 142, 2012–2022. [Google Scholar] [CrossRef]
- European Commision, Directorate-General for Environment. EU Biodiversity Strategy for 2030. Bringing Nature Back into Our Lives; European Commission: Brussels, Belgium, 2020.
- Pötzelsberger, E.; Bauhus, J.; Muys, B.; Wunder, S.; Bozzano, M.; Farsakoglou, A.-M.; Schuck, A.; Lindner, M.; Lapin, K. Forest Biodiversity in the Spotlight—What Drivers Change? European Forest Institute: Joensuu, Finland, 2021. [CrossRef]
- Muys, B.; Angelstam, P.; Bauhus, J.; Bouriaud, L.; Jactel, H.; Kraigher, H.; Müller, J.; Pettorelli, N.; Pötzelsberger, E.; Primmer, E.; et al. Forest Biodiversity in Europe. From Science to Policy 13; European Forest Institute: Joensuu, Finland, 2022. [CrossRef]
- Brzeziecki, B. Białowieża Forest as a biodiversity spot. Sylwan 2017, 161, 971–981, (In Polish with English Summary). [Google Scholar] [CrossRef]
- Götmark, F. Habitat management alternatives for conservation forests in the temperate zone: Review, synthesis, and implications. For. Ecol. Manag. 2013, 306, 292–307. [Google Scholar] [CrossRef]
- Ampoorter, E.; Barbaro, L.; Jactel, H.; Baeten, L.; Boberg, J.; Carnol, M.; Castagneyrol, B.; Charbonnier, Y.; Dawud, S.M.; Deconchat, M.; et al. Tree diversity is key for promoting the diversity and abundance of forest-associated taxa in Europe. Oikos 2020, 129, 133–146. [Google Scholar] [CrossRef]
- Forest Europe. The State of Europe’s Forests; Forest Europe Liason Unit: Bratislava, Slovakia, 2020.
- Dale, V.H.; Joyce, L.A.; McNulty, S.; Neilson, R.P.; Ayres, M.P.; Flannigan, M.D.; Hanson, P.J.; Irland, L.C.; Lugo, A.E.; Peterson, C.J.; et al. Climate Change and Forest Disturbances: Climate change can affect forests by altering the frequency, intensity, duration, and timing of fire, drought, introduced species, insect and pathogen outbreaks, hurricanes, windstorms, ice storms, or landslides. Bioscience 2001, 51, 723–734. [Google Scholar] [CrossRef]
- Ohse, B.; Compagnoni, A.; Farrior, C.E.; McMahon, S.M.; Salguero-Gómez, R.; Rüger, N.; Knight, T.M. Demographic synthesis for global tree species conservation. Trends Ecol. Evol. 2023, 38, 579–590. [Google Scholar] [CrossRef]
- Wassel, A.C.; Myers, J.A. Pawpaws prevent predictability: A locally dominant tree alters understory beta-diversity and community assembly. Ecosphere 2025, 16, e70115. [Google Scholar] [CrossRef]
- Palik, B.; Engstrom, R.T. Species composition. In Maintaining Biodiversity in Forest Ecosystems; Hunter, M.L., Jr., Ed.; Cambridge University Press: Cambridge, UK, 2004; pp. 65–94. [Google Scholar]
- Ellison, A.M.; Bank, M.S.; Clinton, B.D.; Colburn, E.A.; Elliott, K.; Ford, C.R.; Foster, D.R.; Kloeppel, B.D.; Knoepp, J.D.; Lovett, G.M.; et al. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Front. Ecol. Environ. 2005, 3, 479–486. [Google Scholar] [CrossRef]
- Avolio, M.L.; Forrestel, E.J.; Chang, C.C.; La Pierre, K.J.; Burghardt, K.T.; Smith, M.D. Demystifying dominant species. New Phytol. 2019, 223, 1106–1126. [Google Scholar] [CrossRef]
- Boonman, C.C.F.; Serra-Diaz, J.M.; Hoeks, S.; Guo, W.-Y.; Enquist, B.J.; Maitner, B.; Malhi, Y.; Merow, C.; Buitenwerf, R.; Svenning, J.-C. More than 17,000 tree species are at risk from rapid global change. Nat. Commun. 2024, 15, 166. [Google Scholar] [CrossRef] [PubMed]
- You, Z.; Wu, P.; Bakpa, E.P.; Zhang, L.; Ji, L.; You, S. Effect of Differential Growth Dynamics Among Dominant Species Regulates Species Diversity in Subtropical Forests: Empirical Evidence from the Mass Ratio Hypothesis. Forests 2025, 16, 1357. [Google Scholar] [CrossRef]
- Southwood, T.R.E. The number of species of insect associated with various trees. J. Anim. Ecol. 1961, 30, 1–8. [Google Scholar] [CrossRef]
- Kennedy, C.E.J.; Southwood, T.R.E. The number of species of insects associated with British trees: A re-analysis. J. Anim. Ecol. 1984, 53, 455–478. [Google Scholar] [CrossRef]
- Faliński, J.B.; Mułenko, W. Cryptogamous plants in the forest communities of Białowieża National Park. Phytocoenosis 1996, 8, 75–110. [Google Scholar]
- Brändle, M.; Brandl, R. Species richness of insects and mites on trees: Expanding Southwood. J. Anim. Ecol. 2001, 7, 491–504. [Google Scholar] [CrossRef]
- Alexander, K.; Butler, J.; Green, T. The value of different tree and shrub species to wildlife. Brit. Wildl. 2006, 18, 18–28. [Google Scholar]
- Senn-Irlet, B. Welches sind pilzreiche Holzarten? Wald Und Holz 2008, 10, 57–59. [Google Scholar]
- Gamfeldt, L.; Snäll, T.; Bagchi, R.; Jonsson, M.; Gustafsson, L.; Kjellander, P.; Ruiz-Jaen, M.C.; Fröberg, M.; Stendahl, J.; Philipson, C.D.; et al. Higher levels of multiple ecosystem services are found in forests with more tree species. Nat. Commun. 2013, 4, 1340. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Oviedo, A.; Pretzsch, H.; Ammer, C.; Andenmatten, E.; Barbati, A.; Barreiro, S.; Brang, P.; Bravo, F.; Coll, L.; Corona, P.; et al. European Mixed Forests: Definition and research perspectives. For. Syst. 2014, 23, 518–533. [Google Scholar] [CrossRef]
- Bravo-Oviedo, A.; Pretzsch, H.; del Rio, M. (Eds.) Dynamics, Silviculture and Management of Mixed Forests; Managing Forest Ecosystems; Springer: Berlin/Heilderberg, Germany, 2018; Volume 31. [Google Scholar]
- Pretzsch, H.; Poschenrieder, W.; Uhl, E.; Brazaitis, G.; Makrickiene, E.; Calama, R. Silvicultural prescriptions for mixed-species forest stands. A European review and perspective. Eur. J. For. Res. 2021, 140, 1267–1294. [Google Scholar] [CrossRef]
- Halpin, C.R.; Lorimer, C.G. A demographic approach to evaluating tree population sustainability. Forests 2017, 8, 46. [Google Scholar] [CrossRef]
- Lorimer, C.G.; Frelich, L.E. A simulation of equilibrium diameter distributions of sugar maple (Acer saccharum). Bull. Torrey Bot. Club 1984, 111, 193–199. [Google Scholar] [CrossRef]
- Muller-Landau, H.C.; Condit, R.S.; Harms, K.E.; Marks, C.O.; Thomas, S.C.; Bunyavejchewin, S.; Chuyong, G.; Co, L.; Davies, S.; Foster, R.; et al. Comparing tropical forest tree size distributions with the predictions of metabolic ecology and equilibrium models. Ecol. Lett. 2006, 9, 589–602. [Google Scholar] [CrossRef]
- Schütz, J.-P. Modelling the demographic sustainability of pure beech plenter forests in Eastern Germany. Ann. For. Sci. 2006, 63, 93–100. [Google Scholar] [CrossRef]
- Wang, X.; Hao, Z.; Zhang, J.; Lian, J.; Li, B.; Ye, J.; Yao, X. Tree size distributions in an old-growth temperate forest. Oikos 2009, 118, 25–36. [Google Scholar] [CrossRef]
- Faliński, J.B. Geobotany. In Vegetation Dynamics in Temperate Lowland Primeval Forests. Ecological Studies in Białowieża Forest; Dr W. Junk Publishers: Dordrecht, The Netherlands; Boston, MA, USA; Lancaster, UK, 1986; Volume 8, pp. 1–537. [Google Scholar]
- Okołów, C.; Karaś, M.; Bołbot, A. (Eds.) Białowieski Park Narodowy. Poznać. Zrozumieć. Zachować; Białowieża National Park. To Know. To Understand. To Maintain; BPN: Białowieża, Poland, 2009.
- Jaroszewicz, B.; Cholewińska, O.; Gutowski, J.M.; Samojlik, T.; Zimny, M.; Latałowa, M. Białowieża forest—A relic of the high naturalness of European forests. Forests 2019, 10, 849. [Google Scholar] [CrossRef]
- Faliński, J.B. Zielone Grądy i Czarne Bory Białowieży; Green Hornbeam Forests and Black Coniferous Forests of Białowieża; Inst. Wyd. Nasza Księgarnia: Warszawa, Poland, 1977. [Google Scholar]
- Faliński, J.B. Concise Geobotanical Atlas of Białowieża Forest. Phytocoen. Suppl. Cartogr. Geobot. 1994, 6, 3–12. [Google Scholar]
- Matuszkiewicz, J.M.; Tabor, J. (Eds.) Inwentaryzacja Wybranych Elementów Przyrodniczych i Kulturowych Puszczy Białowieskiej; Natural and Cultural Inventory of Białowieża Forest; IBL: Sękocin Stary, Poland, 2023. Available online: https://www.lasy.gov.pl/pl/publikacje/copy-of-gospodarka-lesna/ochrona-przyrody/inwentaryzacja-wybranych-elementow-przyrodniczych-i-kulturowych-puszczy-bialowieskiej/inwentaryzacja-wybranych-elementow-przyrodniczych-i-kulturowych-puszczy-bialowieskiej.pdf (accessed on 20 August 2025).
- Boczoń, A.; Sałachewicz, A. Climatic conditions of Białowieża Forest. In The Current State of Białowieża Forest Based on the Results of the LIFE+ Forbiosensing Project; Stereńczak, K., Ed.; Forest Research Institute: Sękocin Stary, Poland, 2022; pp. 19–35. [Google Scholar]
- Kwiatkowski, W. Vegetation landscapes of Białowieża Forest. Phytocoen. Suppl. Cartogr. Geobot. 1994, 6, 35–87. [Google Scholar]
- Paczoski, J. Lasy Białowieży; The Forests of Białowieża; PROP: Poznań, Poland, 1930. [Google Scholar]
- Matuszkiewicz, W. Zespoły Leśne Białowieskiego Parku Narodowego; Woodland communities of the Białowieża National Park; Annales UMCS. Lublin-Polonia. Supplementum VI. Sectio C; UMCS: Lublin, Poland, 1952. [Google Scholar]
- Sokołowski, A.W. Fitosocjologiczna charakterystyka zbiorowisk leśnych Białowieskiego Parku Narodowego; Phytosociological description of woodland communities of Białowieża National Park. Parki Nar. Rez. Przyr. 1993, 12, 5–190. [Google Scholar]
- Sokołowski, A.W. Lasy Puszczy Białowieskiej; Forests of Białowieża; CILP: Warszawa, Poland, 2004. [Google Scholar]
- Coomes, D.A.; Duncan, R.P.; Allen, R.B.; Truscott, J. Disturbances prevent stem size-density distributions in natural forests from following scaling relationships. Ecol. Lett. 2003, 6, 980–989. [Google Scholar] [CrossRef]
- Salk, T.T.; Frelich, L.E.; Sugita, S.; Calcote, R.; Ferrari, J.B.; Montgomery, R.A. Poor recruitment is changing the structure and species composition of an old-growth hemlock-hardwood forest. For. Ecol. Manag. 2011, 261, 1998–2006. [Google Scholar] [CrossRef]
- Kohyama, T.S.; Potts, M.D.; Kohyama, T.I.; Rahman Kassim, A.; Ashton, P.S. Demographic properties shape tree size distribution in a Malaysian rain forest. Am. Nat. 2015, 185, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Brzeziecki, B.; Pommerening, A.; Miścicki, S.; Drozdowski, S.; Żybura, H. A common lack of demographic equilibrium among tree species in Białowieża National Park (NE Poland): Evidence from long-term plots. J. Veg. Sci. 2016, 27, 460–469. [Google Scholar] [CrossRef]
- Olofsson, L.; Langvall, O.; Pommerening, A. Norway spruce (Picea abies (L.) H. Karst.) selection forests at Siljansfors in Central Sweden. Trees For. People 2023, 12, 100392. [Google Scholar] [CrossRef]
- Bolibok, L.; Brzeziecki, B. An analysis of selected allometric relationships for main tree species of the Białowieża National Park. Sylwan 2000, 6, 73–81, (In Polish with English Summary). [Google Scholar]
- BULiGL. Forest Management Plans for Białowieża, Browsk and Hajnówka Forest Districts for Years 2021–2030; BULiGL: Białystok, Poland, 2021. (In Polish)
- Brzeziecki, B.; Drozdowski, S.; Bielak, K.; Czacharowski, M.; Zajączkowski, J.; Buraczyk, W.; Gawron, L. A demographic equilibrium approach to stocking control in mixed, multiaged stands in the Białowieża Forest, Northeast Poland. For. Ecol. Manag. 2021, 481, 118694. [Google Scholar] [CrossRef]
- Seastedt, T.R.; Hobbs, R.J.; Suding, K.N. Management of novel ecosystems: Are novel approaches required? Front. Ecol. Environ. 2008, 6, 547–553. [Google Scholar] [CrossRef]
- Gonzalez, P.; Neilson, R.P.; Lenihan, J.M.; Drapek, R.J. Global patterns in the vulnerability of ecosystems to vegetation shifts due to climate change. Glob. Ecol. Biogeogr. 2010, 19, 755–768. [Google Scholar] [CrossRef]
- Milad, M.; Schaich, H.; Bürgi, M.; Konold, W. Climate change and nature conservation in Central European forests: A review of consequences, concepts and challenges. For. Ecol. Manag. 2011, 261, 829–843. [Google Scholar] [CrossRef]
- Brang, P.; Spathelf, J.; Larsen, B.; Bauhus, J.; Bončìna, A.; Chauvin, C.; Drössler, L.; García-Güemes, C.; Heiri, C.; Kerr, G.; et al. Suitability of close-to-nature silviculture for adapting temperate European forests to climate change. Forestry 2014, 87, 492–503. [Google Scholar] [CrossRef]
- Stanke, H.; Finley, A.O.; Domke, G.M.; Weed, A.S.; MacFarlane, D.W. Over half of western United States’ most abundant tree species in decline. Nat. Commun. 2021, 12, 451. [Google Scholar] [CrossRef] [PubMed]
- Więcko, E. Puszcza Białowieska; Białowieża Forest; PWN: Warszawa, Poland, 1984. [Google Scholar]
- Borecki, T.; Brzeziecki, B. Silvicultural analysis of post-Century stands in the Białowieża Forest. Sylwan 2001, 7, 19–29, (In Polish with English Summary). [Google Scholar]
- Pautasso, M.; Aas, G.; Queloz, V.; Holdenrieder, O. European ash (Fraxinus excelsior) dieback—A conservation biology challenge. Biol. Conserv. 2013, 158, 37–49. [Google Scholar] [CrossRef]
- Kowalski, T.; Bilański, P.; Kraj, W. Pathogenicity of fungi associated with ash dieback towards Fraxinus excelsior. Plant Pathol. 2017, 66, 1228–1238. [Google Scholar] [CrossRef]
- Paluch, R. The state, conditions and problems of pine natural regeneration in the Białowieża Forest. Sylwan 2004, 148, 9–21, (In Polish with English Summary). [Google Scholar]
- Paluch, R. Natural regeneration of oak in the „Białowieża Primeval Forest” Promotional Forest Complex—State, conditions and prospects. Sylwan 2005, 149, 30–41, (In Polish with English Summary). [Google Scholar]
- Matuszkiewicz, J.M. Changes in the forest associations of Poland’s Białowieża Primeval Forest in the second half of the 20th century. Czas. Geogr. 2011, 82, 69–105. [Google Scholar]
- Ducey, M.J. The Reverse-J and Beyond: Developing Practical, Effective Marking Guides. In Proceedings of the Implementing Uneven-Aged Management in New England: Is It Practical? Fox Research and Demonstration Forest, Hillsborough, NH, USA, 13 April 2006; Caroline, A., Ed.; U.N.H. Cooperative Extension: Durham, NH, USA, 2006. Available online: https://extension.unh.edu/sites/default/files/migrated_unmanaged_files/Resource002220_Rep3279.pdf (accessed on 20 August 2025).
- Jaworski, T.; Plewa, R.; Tarwacki, G.; Sućko, K.; Hilszczański, J.; Horák, J. Ecologically similar saproxylic beetles depend on diversified deadwood resources: From habitat requirements to management implications. For. Ecol. Manag. 2019, 449, 117462. [Google Scholar] [CrossRef]
- Hilszczański, J.; Jaworski, T.; Plewa, R.; Tarwacki, G.; Sućko, K.; Horák, J. Chrząszcze saproksyliczne (Saproxylic beetles). In Inwentaryzacja Wybranych Elementów Przyrodniczych i Kulturowych Puszczy Białowieskiej; Natural and Cultural Inventory of Białowieża Forest; Matuszkiewicz, J.M., Tabor, J., Eds.; IBL: Sękocin Stary, Poland, 2023; pp. 617–663. Available online: https://www.lasy.gov.pl/pl/publikacje/copy-of-gospodarka-lesna/ochrona-przyrody/inwentaryzacja-wybranych-elementow-przyrodniczych-i-kulturowych-puszczy-bialowieskiej/inwentaryzacja-wybranych-elementow-przyrodniczych-i-kulturowych-puszczy-bialowieskiej.pdf (accessed on 20 August 2025).
- Brzeziecki, B.; Drozdowski, S.; Żybura, H.; Bolibok, L.; Bielak, K.; Zajączkowski, J. Managing for naturalness alone is not an effective way to preserve all the valuable natural features of the Białowieża Forest—A reply to Jaroszewicz et al. J. Veg. Sci. 2017, 28, 223–231. [Google Scholar] [CrossRef]
- Jaroszewicz, B.; Bobiec, A.; Eycott, A.E. Lack of demographic equilibrium indicates natural, large-scale forest dynamics, not a problematic forest conservation policy—A reply to Brzeziecki et al. J. Veg. Sci. 2017, 28, 218–222. [Google Scholar] [CrossRef]
- Temperli, C.; Veblen, T.T.; Hart, S.J.; Kulakowski, D.; Tepley, A.J. Interactions among spruce beetle disturbance, climate change and forest dynamics captured by a forest landscape model. Ecosphere 2015, 6, 231. [Google Scholar] [CrossRef]
- Walters, M.B.; Roloff, G.J.; Henry, C.R.; Hartman, J.P.; Donovan, M.L.; Farinosi, E.J.; Starking, M.D. Rethinking Northern Hardwood Forest Management Paradigms with Silvicultural Systems Research: Research-Management Partnerships Ensure Relevance and Application. J. For. 2020, 118, 260–274. [Google Scholar] [CrossRef]
- Ngugi, M.R.; Neldner, V.J.; Dowling, R.M.; Li, J. Recruitment and demographic structure of floodplain tree species in the Queensland Murray-Darling basin, Australia. Ecol. Manag. Restor. 2021, 23, 64–73. [Google Scholar] [CrossRef]
- Miścicki, S. Structure and dynamics of temperate lowland natural forest in the Białowieża National Park, Poland. Forestry 2012, 85, 473–483. [Google Scholar] [CrossRef][Green Version]
- Brzeziecki, B.; Andrzejczyk, T.; Żybura, H. Natural regeneration of trees in the Białowieża Forest. Sylwan 2018, 162, 883–896, (In Polish with English Summary). [Google Scholar] [CrossRef]
- Kuijper, D.P.J.; Cromsigt, J.P.G.M.; Jędrzejewska, B.; Miścicki, S.; Churski, M.; Jędrzejewski, W.; Kweczlich, I. Bottom-up versus top-down control of tree regeneration in the Białowieża Primeval Forest, Poland. J. Ecol. 2010, 98, 888–899. [Google Scholar] [CrossRef]
- Adamowski, W. Flora naczyniowa. In Białowieski Park Narodowy. Poznać. Zrozumieć. Zachować; Okołów, C., Karaś, M., Bołbot, A., Eds.; Białowieża National Park. To Know. To Understand. To Maintain; BPN: Białowieża, Poland, 2009; pp. 60–72. [Google Scholar]
- Brzeziecki, B.; Ciurzycki, W.; Keczyński, A. Changes of herb layer vegetation during the period 1959–2016 on the permanent study plot in the compartment 319 of Białowieża National Park. Sylwan 2018, 162, 980–988, (In Polish with English Summary). [Google Scholar] [CrossRef]
- Cieśliński, S. Porosty (Lichens). In Białowieski Park Narodowy. Poznać. Zrozumieć. Zachować; Okołów, C., Karaś, M., Bołbot, A., Eds.; Białowieża National Park. To Know. To Understand. To Maintain; BPN: Białowieża, Poland, 2009; pp. 73–86. [Google Scholar]
- O’Hara, K.L. Multiaged Silviculture. Managing for Complex Forest Stand Structures; Oxford University Press: Oxford, UK; New York, NY, USA, 2014. [Google Scholar]
- Cole, D.N.; Yung, L. Beyond Naturalness: Rethinking Park and Wilderness Stewardship in an Era of Rapid Change, 1st ed.; Island Press: Washington, DC, USA, 2010. [Google Scholar]
- Wapner, P. The changing nature of nature: Environmental politics in the Anthropocene. Glob. Environ. Polit. 2014, 14, 4. [Google Scholar] [CrossRef]
- Sebek, P.; Bace, R.; Bartos, M.; Benes, J.; Chlumska, Z.; Dolezal, J.; Dvorsky, M.; Kovar, J.; Machac, O.; Mikatova, B.; et al. Does minimal intervention approach threaten the biodiversity of protected areas? A multi-taxa short-term response to intervention in temperate oak-dominated forests. For. Ecol. Manag. 2015, 358, 80–89. [Google Scholar] [CrossRef]
- Miller, K.M.; McGill, B.J.; Mitchell, B.R.; Comiskey, J.; Dieffenbach, F.W.; Matthews, E.R.; Perles, S.J.; Schmit, J.P.; Weed, A.S. Eastern national parks protect greater tree species diversity than unprotected matrix forests. For. Ecol. Manag. 2018, 414, 74–84. [Google Scholar] [CrossRef]
- Kovac, M.; Hladnik, D.; Kutnar, L. Biodiversity in (the Natura 2000) forest habitats is not static: Its conservation calls for an active management approach. J. Nat. Conserv. 2018, 43, 250–260. [Google Scholar] [CrossRef]
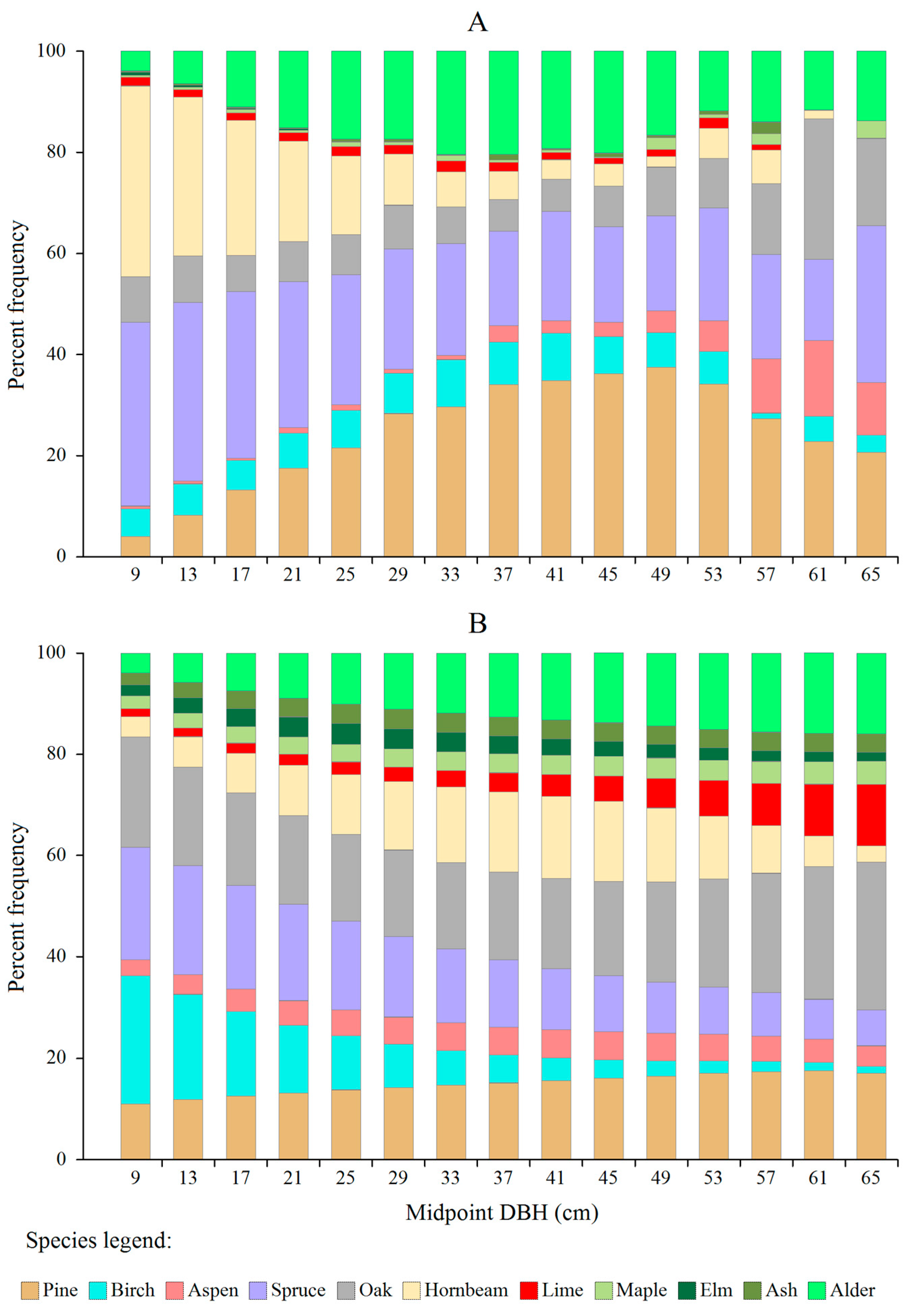
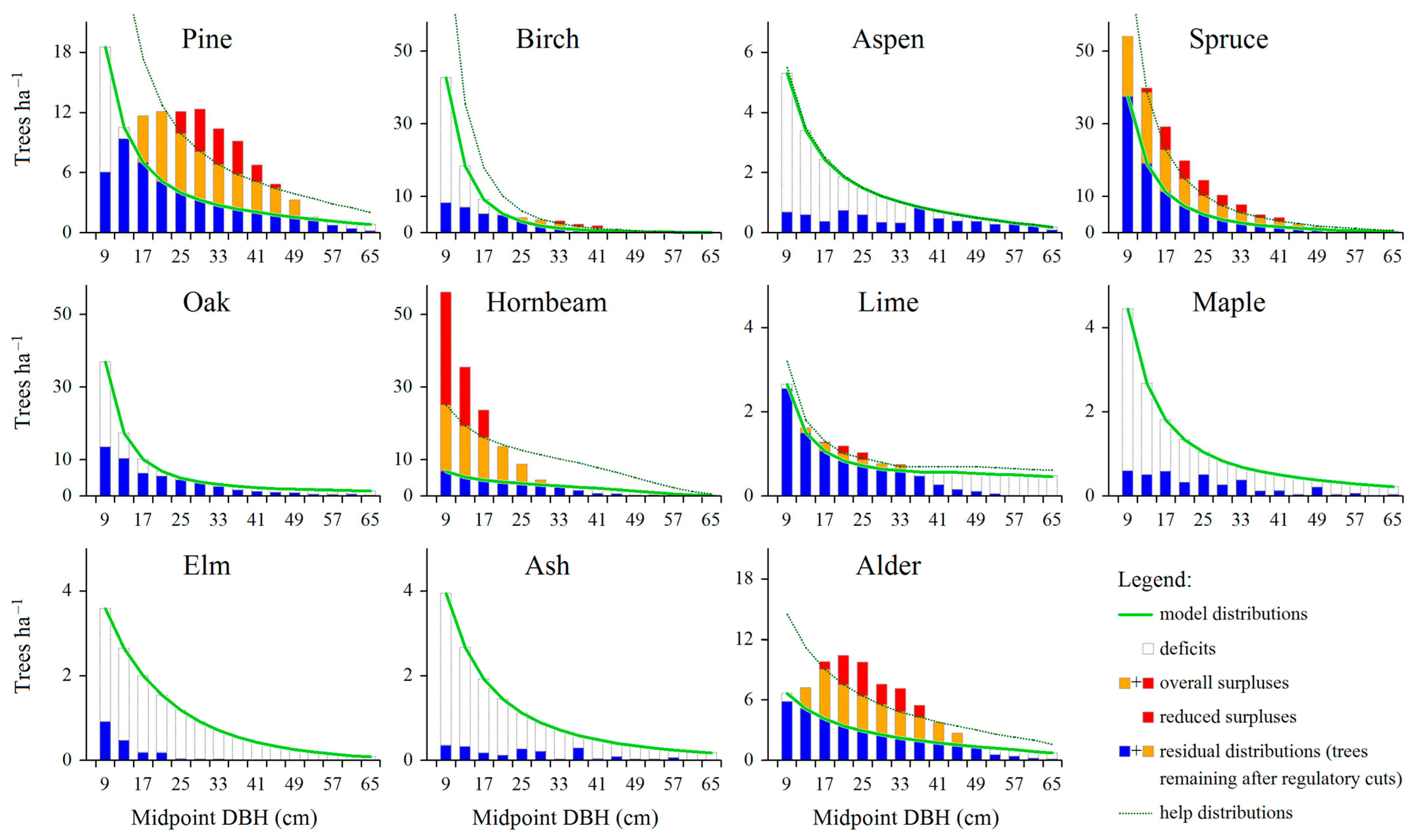
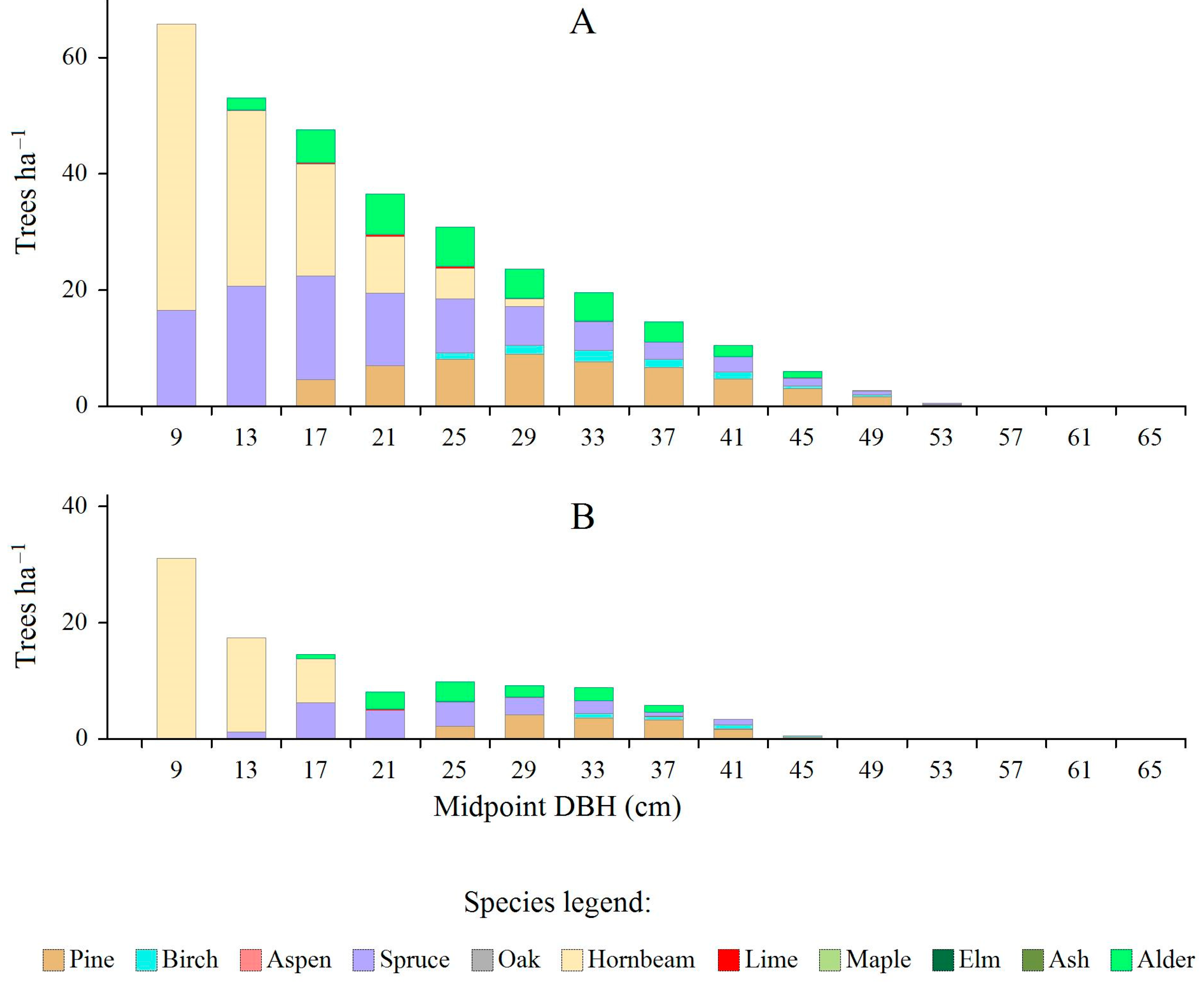


| Tree Species | Area of Growing Space for a Tree with DBH = 9 cm (GS, in m2) * | Average Number of Deficit Trees in the Smallest Diameter Class (Midpoint = 9 cm) (DEF, in Trees × ha−1) | Estimated Area of Regeneration Spots per 100 ha (RS, in ha) |
|---|---|---|---|
| Pine | 8.13 | 12.50 | 1.02 |
| Birch | 12.55 | 34.46 | 4.32 |
| Aspen | 12.55 | 4.61 | 0.58 |
| Spruce | 10.82 | 0.00 | 0.00 |
| Oak | 6.14 | 23.43 | 1.44 |
| Hornbeam | 25.24 | 0.00 | 0.00 |
| Lime | 15.98 | 0.11 | 0.02 |
| Maple | 13.29 | 3.85 | 0.51 |
| Elm | 23.10 | 2.66 | 0.62 |
| Ash | 10.75 | 3.59 | 0.39 |
| Alder | 10.14 | 0.80 | 0.08 |
| Total | 86.00 | 8.97 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brzeziecki, B.; Zajączkowski, J.; Ksepko, M. A Demographic Imbalance of Tree Populations in the Managed Part of Białowieża Forest (NE Poland): Implications for Nature-Oriented Forestry. Forests 2025, 16, 1382. https://doi.org/10.3390/f16091382
Brzeziecki B, Zajączkowski J, Ksepko M. A Demographic Imbalance of Tree Populations in the Managed Part of Białowieża Forest (NE Poland): Implications for Nature-Oriented Forestry. Forests. 2025; 16(9):1382. https://doi.org/10.3390/f16091382
Chicago/Turabian StyleBrzeziecki, Bogdan, Jacek Zajączkowski, and Marek Ksepko. 2025. "A Demographic Imbalance of Tree Populations in the Managed Part of Białowieża Forest (NE Poland): Implications for Nature-Oriented Forestry" Forests 16, no. 9: 1382. https://doi.org/10.3390/f16091382
APA StyleBrzeziecki, B., Zajączkowski, J., & Ksepko, M. (2025). A Demographic Imbalance of Tree Populations in the Managed Part of Białowieża Forest (NE Poland): Implications for Nature-Oriented Forestry. Forests, 16(9), 1382. https://doi.org/10.3390/f16091382








