Effect of Differential Growth Dynamics Among Dominant Species Regulates Species Diversity in Subtropical Forests: Empirical Evidence from the Mass Ratio Hypothesis
Abstract
1. Introduction
2. Materials and Methods
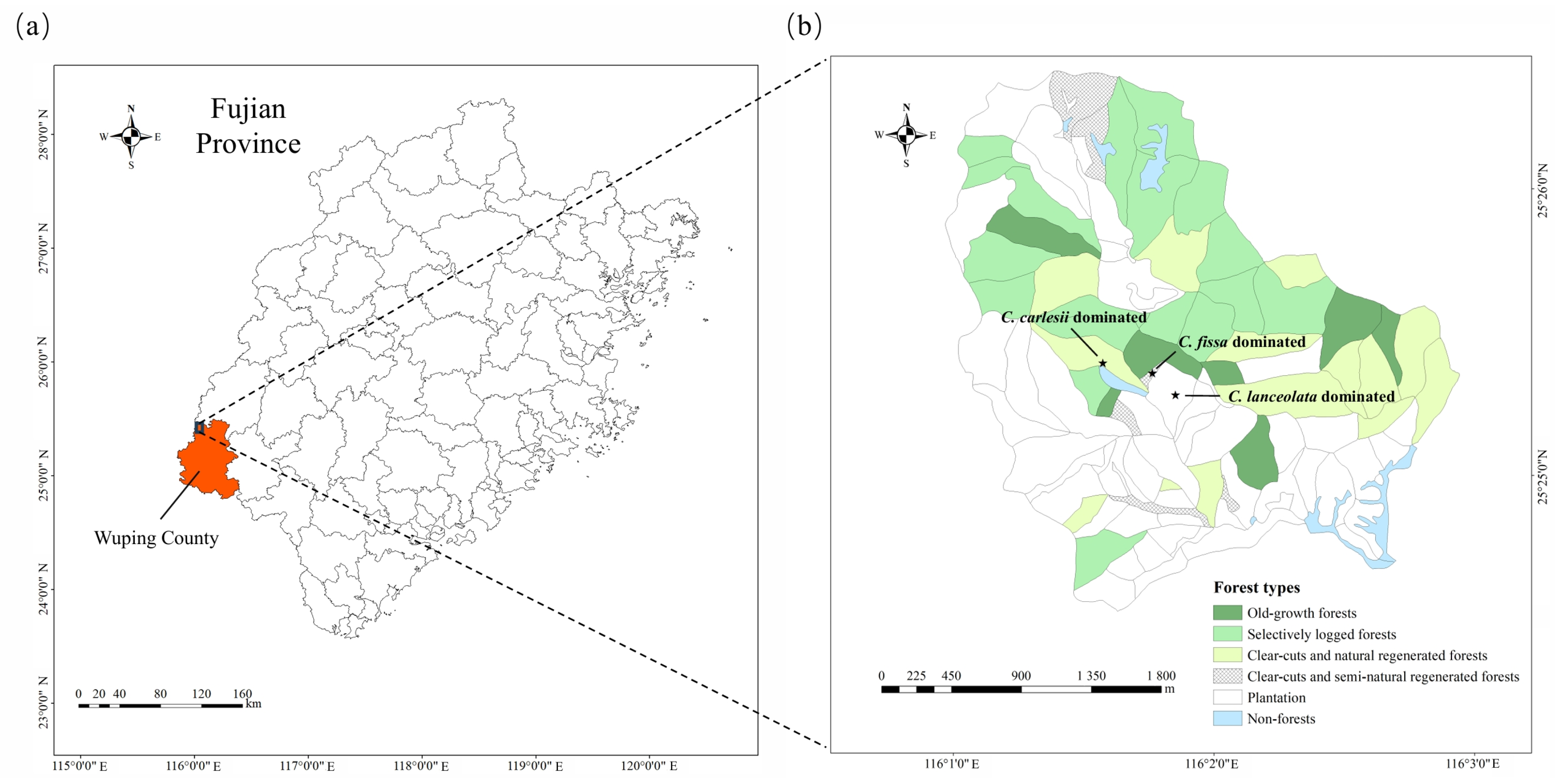
2.1. Study Area
2.2. Experiment Design and Surveys
2.3. Growth Analysis of Dominant Species
2.4. Statistical Analysis
3. Results
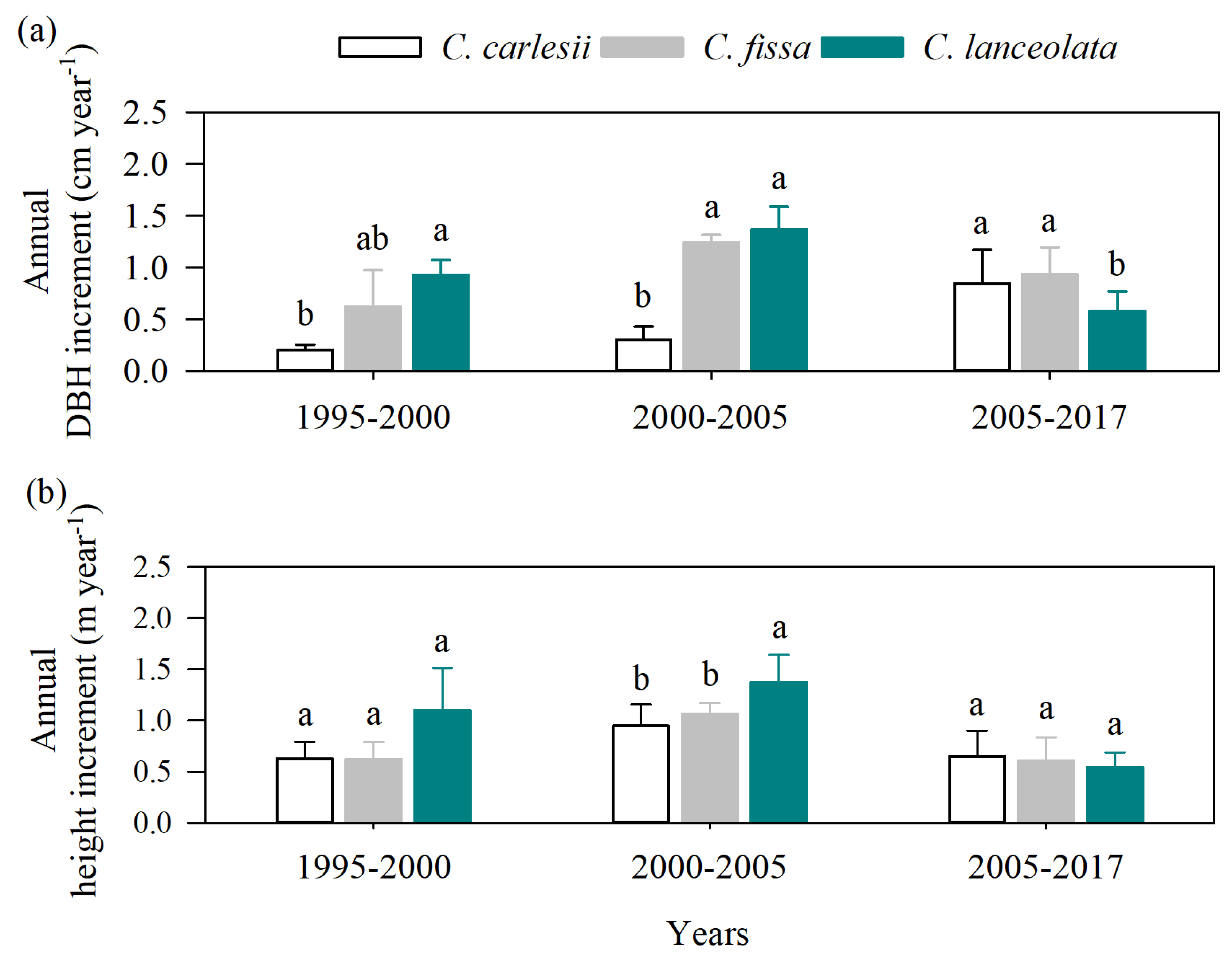
3.1. Growth Dynamics of Three Dominant Tree Species
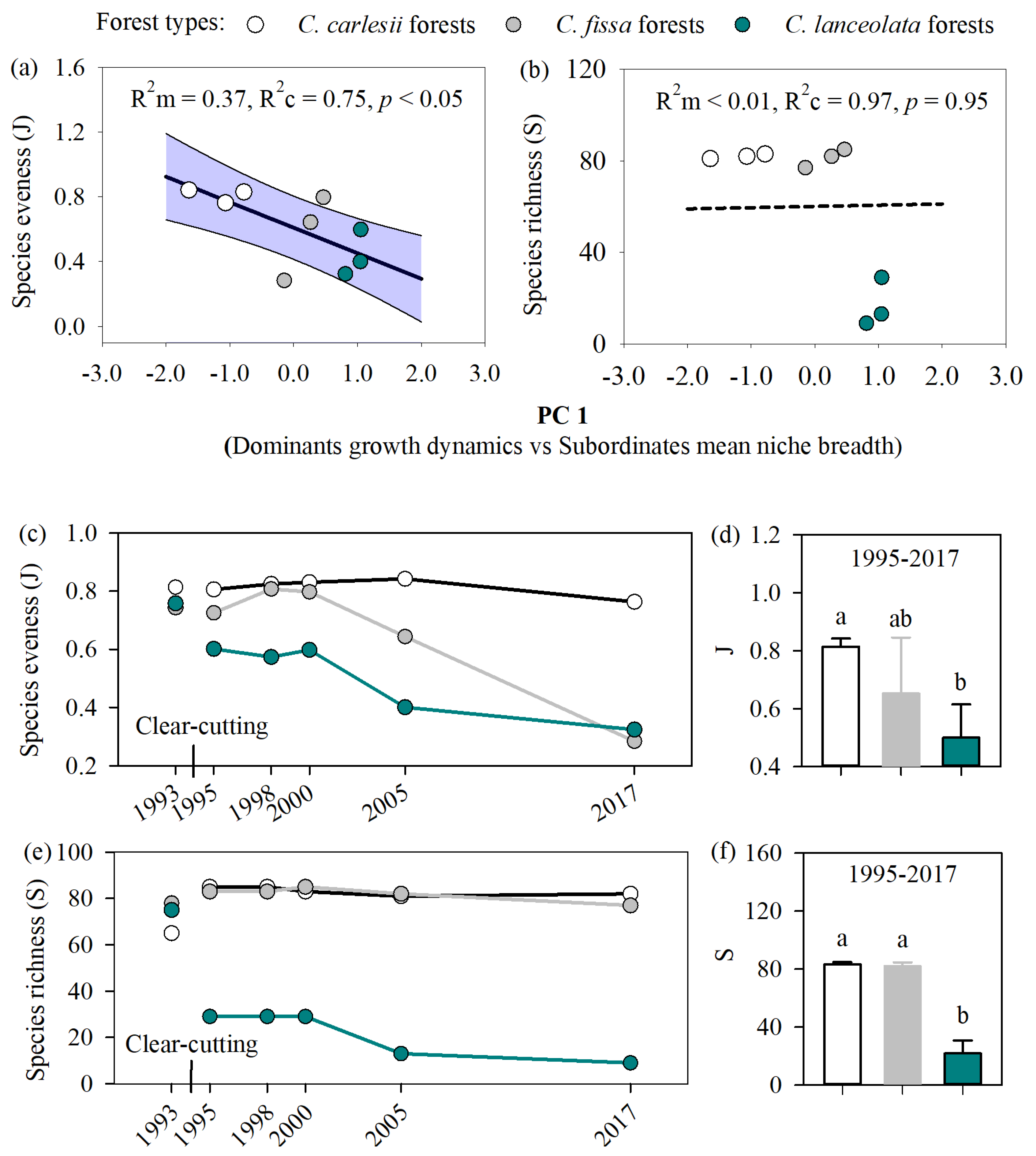
3.2. Effect of Dominant Species Growth Dynamics on Subordinate and Transient Species Niche Breadth
3.3. Relationship Between Effect of Dominant Tree Growth and Species Diversity
4. Discussion
4.1. The Differential Growth Dynamics Among Dominant Tree Species Affect the Species Evenness
4.2. Changes in Species Richness Were Unrelated to the Dominant Species Growth Dynamics
4.3. Implications for Forest Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hua, F.; Bruijnzeel, L.A.; Meli, P.; Martin, P.A.; Zhang, J.; Nakagawa, S.; Miao, X.; Wang, W.; McEvoy, C.; Peña-Arancibia, J.L.; et al. The biodiversity and ecosystem service contributions and trade-offs of forest restoration approaches. Science 2022, 376, 839–844. [Google Scholar] [CrossRef]
- Marsh, C.J.; Turner, E.C.; Blonder, B.W.; Bongalov, B.; Both, S.; Cruz, R.S.; Elias, D.M.O.; Hemprich-Bennett, D.; Jotan, P.; Kemp, V.; et al. Tropical forest clearance impacts biodiversity and function, whereas logging changes structure. Science 2025, 387, 171–175. [Google Scholar] [CrossRef]
- Conradi, T.; Kollmann, J. Species pools and environmental sorting control different aspects of plant diversity and functional trait composition in recovering grasslands. J. Ecol. 2016, 104, 1314–1325. [Google Scholar] [CrossRef]
- Harrison, S.; LaForgia, M. Seedling traits predict drought-induced mortality linked to diversity loss. Proc Natl Acad Sci USA 2019, 116, 5576–5581. [Google Scholar] [CrossRef] [PubMed]
- Chazdon, R.L.; Norden, N.; Colwell, R.K.; Chao, A. Monitoring recovery of tree diversity during tropical forest restoration: Lessons from long-term trajectories of natural regeneration. Philos. Trans. R. Soc. B Biol. Sci. 2022, 378, 20210069. [Google Scholar] [CrossRef] [PubMed]
- Wassel, A.C.; Myers, J.A. Pawpaws prevent predictability: A locally dominant tree alters understory beta-diversity and community assembly. Ecosphere 2025, 16, e70115. [Google Scholar] [CrossRef]
- Norden, N.; Angarita, H.A.; Bongers, F.; Martínez-Ramos, M.; Granzow-de la Cerda, I.; van Breugel, M.; Lebrija-Trejos, E.; Meave, J.A.; Vandermeer, J.; Williamson, G.B.; et al. Successional dynamics in Neotropical forests are as uncertain as they are predictable. Proc. Natl. Acad. Sci. USA 2015, 112, 8013–8018. [Google Scholar] [CrossRef]
- Grime, J. Benefits of Plant Diversity to Ecosystems: Immediate, Filter and Founder Effects. J. Ecol. 1998, 86, 902–910. [Google Scholar] [CrossRef]
- Kershaw, H.M.; Mallik, A.U. Predicting Plant Diversity Response to Disturbance: Applicability of the Intermediate Disturbance Hypothesis and Mass Ratio Hypothesis. Crit. Rev. Plant Sci. 2013, 32, 383–395. [Google Scholar] [CrossRef]
- Garnier, E.; Cortez, J.; Navas, M.-L.; Roumet, C.; Debussche, M.; Rard Laurent, G.; Blanchard, A.; Aubry, D.; Bellmann, A.; Neill, C.; et al. Plant functional markers capture ecosystem properties during secondary succession. Ecology 2004, 85, 2630–2637. [Google Scholar] [CrossRef]
- Vile, D.; Shipley, B.; Garnier, E. Ecosystem productivity can be predicted from potential relative growth rate and species abundance. Ecol. Lett. 2006, 9, 1061–1067. [Google Scholar] [CrossRef]
- Mokany, K.; Ash, J. Are traits measured on pot grown plants representative of those in natural communities? J. Veg. Sci. 2008, 19, 119–126. [Google Scholar] [CrossRef]
- Mulder, C.P.H.; Bazeley-White, E.; Dimitrakopoulos, P.G.; Hector, A.; Scherer-Lorenzen, M.; Schmid, B. Species evenness and productivity in experimental plant communities. Oikos 2004, 107, 50–63. [Google Scholar] [CrossRef]
- Falster, D.S.; Brännström, Å.; Dieckmann, U.; Westoby, M. Influence of four major plant traits on average height, leaf-area cover, net primary productivity, and biomass density in single-species forests: A theoretical investigation. J. Ecol. 2011, 99, 148–164. [Google Scholar] [CrossRef]
- Iida, Y.; Kohyama, T.S.; Kubo, T.; Kassim, A.R.; Poorter, L.; Sterck, F.; Potts, M.D. Tree architecture and life-history strategies across 200 co-occurring tropical tree species. Funct. Ecol. 2011, 25, 1260–1268. [Google Scholar] [CrossRef]
- Kohyama, T.S.; Takada, T.; Canham, C. One-sided competition for light promotes coexistence of forest trees that share the same adult height. J. Ecol. 2012, 100, 1501–1511. [Google Scholar] [CrossRef]
- Chen, Y.; Wright, S.J.; Muller-Landau, H.C.; Hubbell, S.P.; Wang, Y.; Yu, S. Positive effects of neighborhood complementarity on tree growth in a Neotropical forest. Ecology 2016, 97, 776–785. [Google Scholar] [CrossRef]
- Bongers, F.J.; Schmid, B.; Sun, Z.; Li, Y.; Härdtle, W.; Oheimb, G.; Li, Y.; Li, S.; Staab, M.; Ma, K.; et al. Growth–trait relationships in subtropical forest are stronger at higher diversity. J. Ecol. 2019, 108, 256–266. [Google Scholar] [CrossRef]
- Avolio, M.L.; Forrestel, E.J.; Chang, C.C.; La Pierre, K.J.; Burghardt, K.T.; Smith, M.D. Demystifying dominant species. New Phytol. 2019, 223, 1106–1126. [Google Scholar] [CrossRef]
- Campbell, B.D.; Grime, J.P.; Mackey, J.M.L. A trade-off between scale and precision in resource foraging. Oecologia 1991, 87, 532–538. [Google Scholar] [CrossRef]
- Falster, D.S.; Westoby, M. Tradeoffs between height growth rate, stem persistence and maximum height among plant species in a post-fire succession. Oikos 2005, 111, 57–66. [Google Scholar] [CrossRef]
- Koorem, K.; Moora, M. Positive association between understory species richness and a dominant shrub species (Corylus avellana) in a boreonemoral spruce forest. For. Ecol. Manag. 2010, 260, 1407–1413. [Google Scholar] [CrossRef]
- Mariotte, P.; Buttler, A.; Johnson, D.; Thébault, A.; Vandenberghe, C.; Bruun, H.H. Exclusion of root competition increases competitive abilities of subordinate plant species through root–shoot interactions. J. Veg. Sci. 2012, 23, 1148–1158. [Google Scholar] [CrossRef]
- Torrez, V.; Mergeay, J.; De Meester, L.; Honnay, O.; Helsen, K. Differential effects of dominant and subordinate plant species on the establishment success of target species in a grassland restoration experiment. Appl. Veg. Sci. 2017, 20, 363–375. [Google Scholar] [CrossRef]
- Khalil, M.I.; Gibson, D.J.; Baer, S.G.; Jones, H. Functional response of subordinate species to intraspecific trait variability within dominant species. J. Ecol. 2019, 107, 2040–2053. [Google Scholar] [CrossRef]
- Arnillas, C.A.; Borer, E.T.; Seabloom, E.W.; Alberti, J.; Baez, S.; Bakker, J.D.; Boughton, E.H.; Buckley, Y.M.; Bugalho, M.N.; Donohue, I.; et al. Opposing community assembly patterns for dominant and nondominant plant species in herbaceous ecosystems globally. Ecol. Evol. 2021, 11, 17744–17761. [Google Scholar] [CrossRef]
- Poorter, L.; Craven, D.; Jakovac, C.C.; van der Sande, M.T.; Amissah, L.; Bongers, F.; Chazdon, R.L.; Farrior, C.E.; Kambach, S.; Meave, J.A.; et al. Multidimensional tropical forest recovery. Science 2021, 374, 1370–1376. [Google Scholar] [CrossRef]
- Zhou, H.; Meng, S.; Liu, Q. Diameter Growth, Biological Rotation Age and Biomass of Chinese Fir in Burning and Clearing Site Preparations in Subtropical China. Forests 2016, 7, 177. [Google Scholar] [CrossRef]
- Zhou, H.; Meng, S.; Liu, Q. Long-term response of living forest biomass to extensive logging in subtropical China. J. For. Res. 2019, 30, 1679–1687. [Google Scholar] [CrossRef]
- Yang, Y.S.; Guo, J.; Chen, G.; Xie, J.; Gao, R.; Li, Z.; Jin, Z. Carbon and nitrogen pools in Chinese fir and evergreen broadleaved forests and changes associated with felling and burning in mid-subtropical China. For. Ecol. Manag. 2005, 216, 216–226. [Google Scholar] [CrossRef]
- Chen, H. Biomass and nutrient distribution in a Chinese-fir plantation chronosequence in Southwest Hunan, China. For. Ecol. Manag. 1998, 105, 209–216. [Google Scholar] [CrossRef]
- Lin, K.; Huang, B. Studies on β-diversity index of undergrowth plant in Chinese fir plantation. Biodivers. Sci. 2001, 9, 157–161. [Google Scholar] [CrossRef]
- Fang, Y. Species composition and diversity of evergreen broad-leaved forest of Castanopsis carlesii and C. eyrei in Wuyishan National Nature Reserve, Fujian, China. Biodivers. Sci. 2005, 13, 148. [Google Scholar] [CrossRef]
- Hu, J.; Zhou, H.; Huang, Y.; Yao, X.; Ye, S.; Yu, S. A study on plant species diversity and soil carbon and nitrogen in different Cunninghamia lanceolata stand types. Ecol. Environ. 2022, 31, 451. [Google Scholar] [CrossRef]
- Wu, Y.; Zheng, Y.; Wang, Y.; Wei, B.; Wu, C.; Shen, A.; Lu, X.; Zheng, F.; Yu, Z.; Ni, J. Relationship between the number of Cunninghamia lanceolata and community species diversity in abandoned Chinese fir forests. Acta Ecol. Sin. 2022, 42, 884–894. [Google Scholar] [CrossRef]
- Wu, Q.; Lin, H.; Chen, C.; Fan, H.; Zhang, B.; He, J.; Fang, C.; Yuan, F.; Wang, Y.; Wu, C. Ecological niche characteristics and spatial distribution patterns of woody plants in the understory of Castanopsis fissa forest after clearcutting. Chin. J. Ecol. 2025, 44, 21. [Google Scholar] [CrossRef]
- Mensah, S.; Veldtman, R.; Assogbadjo, A.E.; Glèlè Kakaï, R.; Seifert, T. Tree species diversity promotes aboveground carbon storage through functional diversity and functional dominance. Ecol. Evol. 2016, 6, 7546–7557. [Google Scholar] [CrossRef]
- Vallet, P.; Perot, T. Tree diversity effect on dominant height in temperate forest. For. Ecol. Manag. 2016, 381, 106–114. [Google Scholar] [CrossRef]
- You, S. The Effects of Different Artificial Disturbance on the Flora and Speceis Diversity of Tree Layer in Castanopsis Carlesii forest in Wupin, Fujian. Sci. Silvae Sin. 2001, 37, 106–110. [Google Scholar] [CrossRef]
- Chen, S.; You, S.; Chen, Z.; You, Z. Effect of different regeneration modes on height class and species diversity of Castanopsis carlesii community by using β-diversity. J. Plant Resour. Environ. 2013, 22, 62–69. [Google Scholar] [CrossRef]
- Wu, M.; You, S.; Yang, Y.S.; HE, Y.; Li, F. Phytocoenological Characters of the Evergreen Broad-leaf Forest Castanopsis Carlesii Form in Wuping Maobu Area, Fujian. J. Fujian Coll. For. 1994, 2, 157–162. [Google Scholar]
- You, S.; Lan, S.; Chen, S.; You, Z. Keys of Woody Plants in Fujian; China Forestry Publishing House: Beijing, China, 2013. [Google Scholar]
- Speer, J.H. Fundamentals of Tree-Ring Research; University of Arizona Press: Tucson, AZ, USA, 2010; Volume 1. [Google Scholar]
- Tong, W.; Yujun, S.; Jingjing, Q. Response of Pinus massoniana tree-ring width in the Jiangle Area of Fujian Province to climate change. J. Beijing For. Univ. 2019, 41, 30–39. [Google Scholar] [CrossRef]
- Xinguang, C.; Hongbing, H.; Yingjun, L.; Zhipeng, D.; Xiaorong, L.; Maowei, B.; Zhuangpeng, Z.; Keyan, F. Differences in the ecological resilience of planted and natural Pinus massoniana and Cunninghamia lanceolata forests in response to drought in subtropical China. Chin. J. Appl. Ecol. 2022, 32, 3531–3538. [Google Scholar] [CrossRef]
- Pretzsch, H. Forest Dynamics, Growth, and Yield: From Measurement to Model; Springer: Berlin/Heidelberg, Germany, 2009; Volume 1. [Google Scholar]
- Peet, R.K. The measurement of species diversity. Annu. Rev. Ecol. Syst. 1974, 5, 285–307. [Google Scholar] [CrossRef]
- Levins, R. Evolution in Changing Environments: Some Theoretical Explorations; Princeton University Press: Princeton, NJ, USA, 1968; Volume 1. [Google Scholar]
- Lai, J.; Zou, Y.; Zhang, S.; Zhang, X.; Mao, L.; Zhang, W.-H. glmm.hp: An R package for computing individual effect of predictors in generalized linear mixed models. J. Plant Ecol. 2022, 15, 1302–1307. [Google Scholar] [CrossRef]
- Matsuo, T.; Martinez-Ramos, M.; Bongers, F.; van der Sande, M.T.; Poorter, L. Forest structure drives changes in light heterogeneity during tropical secondary forest succession. J. Ecol. 2021, 109, 2871–2884. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, T.; Martinez-Ramos, M.; Onoda, Y.; Bongers, F.; Lohbeck, M.; Poorter, L. Light competition drives species replacement during secondary tropical forest succession. Oecologia 2024, 205, 1–11. [Google Scholar] [CrossRef]
- Sterck, F.J.; Bongers, F. Crown development in tropical rain forest trees: Patterns with tree height and light availability. J. Ecol. 2001, 89, 1–13. [Google Scholar] [CrossRef]
- Coutts, M.P. Developmental processes in tree root systems. Can. J. For. Res. 1987, 17, 761–767. [Google Scholar] [CrossRef]
- Gilliam, F.S. The Ecological Significance of the Herbaceous Layer in Temperate Forest Ecosystems. BioScience 2007, 57, 845–858. [Google Scholar] [CrossRef]
- Uriarte, M.; Swenson, N.G.; Chazdon, R.L.; Comita, L.S.; John Kress, W.; Erickson, D.; Forero-Montaña, J.; Zimmerman, J.K.; Thompson, J. Trait similarity, shared ancestry and the structure of neighbourhood interactions in a subtropical wet forest: Implications for community assembly. Ecol. Lett. 2010, 13, 1503–1514. [Google Scholar] [CrossRef]
- Liu, T.-Y.; Lin, K.-C.; Vadeboncoeur, M.A.; Chen, M.-Z.; Huang, M.-Y.; Lin, T.-C. Understorey plant community and light availability in conifer plantations and natural hardwood forests in Taiwan. Appl. Veg. Sci. 2015, 18, 591–602. [Google Scholar] [CrossRef]
- Yang, X.-D.; Yan, E.-R.; Chang, S.X.; Da, L.-J.; Wang, X.-H. Tree architecture varies with forest succession in evergreen broad-leaved forests in Eastern China. Trees 2014, 29, 43–57. [Google Scholar] [CrossRef]
- Plaga, B.N.E.; Bauhus, J.; Pretzsch, H.; Pereira, M.G.; Forrester, D.I. Influence of crown and canopy structure on light absorption, light use efficiency, and growth in mixed and pure Pseudotsuga menziesii and Fagus sylvatica forests. Eur. J. For. Res. 2023, 143, 479–491. [Google Scholar] [CrossRef]
- Sercu, B.K.; Baeten, L.; van Coillie, F.; Martel, A.; Lens, L.; Verheyen, K.; Bonte, D. How tree species identity and diversity affect light transmittance to the understory in mature temperate forests. Ecol. Evol. 2017, 7, 10861–10870. [Google Scholar] [CrossRef] [PubMed]
- Gaertner, M.; Den Breeyen, A.; Hui, C.; Richardson, D.M. Impacts of alien plant invasions on species richness in Mediterranean-type ecosystems: A meta-analysis. Prog. Phys. Geogr. Earth Environ. 2009, 33, 319–338. [Google Scholar] [CrossRef]
- Powell, K.I.; Chase, J.M.; Knight, T.M. A synthesis of plant invasion effects on biodiversity across spatial scales. Am. J. Bot. 2011, 98, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Akatov, V.V.; Akatova, T.V.; Eskina, T.G.; Sazonets, N.M.; Chefranov, S.G. Dominants in Plant Communities: The Nature of Impact on Biomass Determines the Thresholds of Impact on Local Species Richness. Biol. Bull. Rev. 2025, 15, 12–23. [Google Scholar] [CrossRef]
- Kopecký, M.; Hédl, R.; Szabó, P.; Hooftman, D. Non-random extinctions dominate plant community changes in abandoned coppices. J. Appl. Ecol. 2012, 50, 79–87. [Google Scholar] [CrossRef]
- Müllerová, J.; Hédl, R.; Szabó, P. Coppice abandonment and its implications for species diversity in forest vegetation. For. Ecol. Manag. 2015, 343, 88–100. [Google Scholar] [CrossRef]
- Pausas, J.G.; Lamont, B.B.; Paula, S.; Appezzato-da-Glória, B.; Fidelis, A. Unearthing belowground bud banks in fire-prone ecosystems. New Phytol. 2018, 217, 1435–1448. [Google Scholar] [CrossRef] [PubMed]
- De Lombaerde, E.; Baeten, L.; Verheyen, K.; Perring, M.P.; Ma, S.; Landuyt, D. Understorey removal effects on tree regeneration in temperate forests: A meta-analysis. J. Appl. Ecol. 2021, 58, 9–20. [Google Scholar] [CrossRef]
- Bell, F.W.; Newmaster, S.G. The effects of silvicultural disturbances on the diversity of seed-producing plants in the boreal mixedwood forest. Can. J. For. Res. 2002, 32, 1180–1191. [Google Scholar] [CrossRef]
- Eycott, A.E.; Watkinson, A.R.; Dolman, P.M. Ecological patterns of plant diversity in a plantation forest managed by clearfelling. J. Appl. Ecol. 2006, 43, 1160–1171. [Google Scholar] [CrossRef]
- Norden, N.; Chazdon, R.L.; Chao, A.; Jiang, Y.H.; Vilchez-Alvarado, B. Resilience of tropical rain forests: Tree community reassembly in secondary forests. Ecol. Lett. 2009, 12, 385–394. [Google Scholar] [CrossRef]
- Jakovac, C.C.; Peña-Claros, M.; Kuyper, T.W.; Bongers, F. Loss of secondary-forest resilience by land-use intensification in the Amazon. J. Ecol. 2015, 103, 67–77. [Google Scholar] [CrossRef]
- Li, W. Degradation and restoration of forest ecosystems in China. For. Ecol. Manag. 2004, 201, 33–41. [Google Scholar] [CrossRef]
- Dai, L.; Wang, Y.; Su, D.; Zhou, L.; Yu, D.; Lewis, B.J.; Qi, L. Major Forest Types and the Evolution of Sustainable Forestry in China. Environ. Manag. 2011, 48, 1066–1078. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
You, Z.; Wu, P.; Bakpa, E.P.; Zhang, L.; Ji, L.; You, S. Effect of Differential Growth Dynamics Among Dominant Species Regulates Species Diversity in Subtropical Forests: Empirical Evidence from the Mass Ratio Hypothesis. Forests 2025, 16, 1357. https://doi.org/10.3390/f16081357
You Z, Wu P, Bakpa EP, Zhang L, Ji L, You S. Effect of Differential Growth Dynamics Among Dominant Species Regulates Species Diversity in Subtropical Forests: Empirical Evidence from the Mass Ratio Hypothesis. Forests. 2025; 16(8):1357. https://doi.org/10.3390/f16081357
Chicago/Turabian StyleYou, Zhangtian, Pengfei Wu, Emily Patience Bakpa, Lifu Zhang, Lianyao Ji, and Shuisheng You. 2025. "Effect of Differential Growth Dynamics Among Dominant Species Regulates Species Diversity in Subtropical Forests: Empirical Evidence from the Mass Ratio Hypothesis" Forests 16, no. 8: 1357. https://doi.org/10.3390/f16081357
APA StyleYou, Z., Wu, P., Bakpa, E. P., Zhang, L., Ji, L., & You, S. (2025). Effect of Differential Growth Dynamics Among Dominant Species Regulates Species Diversity in Subtropical Forests: Empirical Evidence from the Mass Ratio Hypothesis. Forests, 16(8), 1357. https://doi.org/10.3390/f16081357






