Abstract
This study examines microhabitat abundance and composition on retention trees in 20 young stands in Latvia that were clear-cut between 2002 and 2004. Retention trees play a vital role in sustaining biological diversity after clear-cutting, but their mortality rate is often high due to post-harvest environmental changes. This study compares tree-related microhabitat (TreM) abundance and composition on retention trees of different dimensions, species, and vitality. The tree height and diameter were measured, and the living status and position of each tree were determined. The research questions of the study were to assess the relationships between TreMs, tree dimensions, and species, as well as to compare the occurrence of TreMs on living and dead wood. The comprehensive analysis revealed that larger tree diameters and specific tree species (Populus tremula, Fraxinus excelsior, and Salix caprea) lead to a higher abundance of TreMs. The results also highlighted the importance of preserving deadwood within forest stands as it enhances the observed abundance of TreMs. This study provides the missing information on TreMs in Latvia, thus providing data for comparison in a wider region.
1. Introduction
Clear-cuts, which are one of the most commonly used harvesting methods in the hemiboreal region [1], are intended to mimic stand-replacing disturbances. However, unlike in a stand-replacing disturbance, less dead wood remains in the stand after clearcutting. As a result of clear-cut, drastic changes in temperature and soil moisture occur, therefore influencing the survival and growth of forest biota [2]. To sustain biological diversity, legislation in most European countries requires the retention of 5–10 trees per hectare, as well as the conservation of standing and fallen deadwood [3,4]. The purpose of the retention trees is to ensure a gradual long-term input of deadwood as well as a continuous presence of living trees in the young stand [5], thus increasing the structural diversity, spatial connectivity, and scenic value of the young stands [3,6,7]. Due to substantial changes in the post-harvest environment (increased wind and drought), retention trees have high mortality [5,6,8] and often die off soon after the clear-cut [8]. This might lead to questioning their capacity to maintain species richness. Nevertheless, the presence of deadwood contributes to biodiversity as it serves as a microhabitat for specialist species from a wide spectrum of taxa such as fungi, invertebrates, vegetation, and epiphytes [9,10,11,12].
Tree-related microhabitats (TreMs) are heterogeneous substrates located on living or dead trees that are suitable for specialized species to develop and breed [13,14,15]. They promote heterogeneity of the forest stands and can be used as indicators since they may capture a large proportion of stand biodiversity [13,16] for a broad spectrum of taxonomic groups [13]. Retaining different-dimension trees can increase diversity, as they offer various types of TreMs [17]. Dendrotelms and exposed heartwood are commonly found in larger trees, while smaller trees are associated with bark loss, crown deadwood, and cracks [17].
The number and types of TreMs are affected by the management history of the stands [14]. Overall, managed forests have a lower abundance of TreMs [16,18], while the highest TreM richness and diversity are associated with old-growth [19] and unmanaged forests [14]. In intensively managed forests, a lower occurrence of TreMs is associated with the simpler stand structure and targeted felling of defected trees [20,21]. Still, harvesting often leads to modifications of retention trees such as lesions, limb breakages, intrusions, and exposure of sapwood, therefore initializing wood decay [13,20], which may result in saproxylic tree-related microhabitats [14,16].
Evidence-based retention tree selection is advised to optimize microhabitat provision in the forest stands [22] and therefore increase biodiversity. Although the number of TreM studies has increased in recent decades [23], information about microhabitats in hemiboreal forests is still scarce [19]. Accordingly, this study aims to compare TreM abundance and composition on retention trees of different dimensions, species, and living status (living tree, stump, snag, and lying dead wood) in ~20-year-old clear-cuts in Latvia. The research questions of the study were to assess the relationships between TreMs, tree dimensions, and species, as well as to compare the occurrence of TreMs on living and dead wood.
2. Materials and Methods
2.1. Studied Site and Data Collection
Between 1992 and 2022, the average annual temperature in the central region of Latvia was −2.9 ± 3.3 °C in its coldest month, February, and +18.4 ± 1.6 °C in its warmest month, July. The overall yearly mean temperature was 7.3 ± 0.7 °C. The typical annual precipitation was 641.4 ± 70.3 mm, with an average of 13.7 ± 1.2 days of precipitation per month and 12.9 ± 2.2 days during the summer months [24].
Regardless of the forest type, species composition, age, and clear-cut size, 20 young stands were selected from National Forest Inventory database to meet the only criterion that clear-cutting was performed in the period from 2002 to 2004 (Figure 1). The age of the forest stands before clear-cuts ranged from 42 to 116 years, on average 79 ± 4 (±standard error) years. The selected stands represented sites with both wet and drained soils, as well as fertility gradient ranging from oligotrophic to eutrophic. The mean size of the clear-cuts was 2.9 ± 0.4 ha, ranging from 0.9 ha to 8.2 ha.
Figure 1.
Location of the studied sites in Latvia. Forest area colored in gray.
In the selected stands, stratified selection of the retention trees was carried out, with the minimum requirement of selecting at least 5 trees per clear-cut, and the total number of retention trees per clear-cut ranged between 5 and 16. The process of selecting trees involved assessing all tree species present in the clear-cut area, with a primary focus on choosing those with larger diameters. In addition, all dead retention trees were assessed. Trees located at the edge of the area were omitted. Tree position in the forest stand was described as an individual living tree or a tree group (at least three trees in a 15 m radius). Tree height and diameter were measured, and living status (live, lying deadwood, stump, and snag) was recorded.
To ensure compatibility of the data, a uniform detection methodology by Larrieu et al. (2018) [13] was used. The methodology proposes hierarchical approach with seven TreM forms based on the same physiognomy and functional traits: cavities, tree injuries, crown deadwood, excrescences, fruiting bodies, epiphytic and epixylic structures, and exudates [13]. These forms are divided into 15 groups (woodpecker breeding cavities, rot holes, insect galleries, concavities, exposed sapwood only, exposed sapwood and hardwood, crown deadwood, twig tangles, burrs and cankers, perennial fungal fruiting bodies, ephemeral fungal fruiting bodies and slime molds, epiphytic or parasitic crypto- and phanerogams, nests, microsoil, and fresh exudates), which are then assigned to 47 types using morphological traits and their importance in biodiversity conservation as a criterion. Although primarily employed for evaluating living trees, this study adapted the method to also assess TreMs on decaying wood. The evaluation of TreMs on the retention trees was performed during a leaf-free period in autumn of 2021.
2.2. Statistical Analysis
The distribution of TreM by form was estimated. In order to evaluate the tree and stand-level properties influencing the number of TreMs, a generalized linear mixed-effect model (glmer) was fitted [25]. The model in general form was as follows:
where Xijβ is the set of tree and stand-level properties, and Oi is the random effect of the stand. First, the set of fixed effects was added to the model (forest age, tree species, height, diameter, forest type, location in the stand, and living status). The Akaike Information Criterion (AIC) [26] was employed to select the optimal combination of fixed effects. The normality and homogeneity of model residuals were assessed through diagnostic plots [27]. The explanatory variables were checked for collinearity using the variance inflation factor, where variables with a criterion exceeding five were eliminated [28]. Consequently, a set of fixed effects comprising species, diameter, and living status remained. Random effects were incorporated into an account for data dependencies, with the object number being included as the random effect (intercept). The significance of the fixed effects was evaluated using the Type II Wald chi-square test [27]. Post hoc comparisons of estimated marginal means for the significant fixed effects (categorical) were conducted using the Tukey post hoc test. Data analysis was performed using R version 4.1.3 (R Foundation for Statistical Computing, Vienna, Austria) [29] using library lme4 [25].
n = Xijβ + (Oi) + ɛij
3. Results and Discussion
In total, 10 retention tree species were evaluated: black alder (Alnus glutinosa), pedunculate oak (Quercus robur), Eurasian aspen (Populus tremula, small-leaved linden (Tilia cordata), common ash (Fraxinus excelsior), Scots elm (Ulmus glabra), Norway maple (Acer platanoides), goat willow (Salix caprea), with three being commercially the most important: silver birch (Betula pendula), Scots pine (Pinus sylvestris), and Norway spruce (Picea abies) (Figure 2). Despite commercially important species being the most prevalent tree species in the forests of Latvia [30], the majority of the retention trees were deciduous (Figure 2), which is due to the forestry preferences outlined in the legislation. This seems reasonable, as some authors have noted a general pattern of higher TreM occurrence in deciduous than coniferous species [14]. This suggests that the current legislation promotes TreM formation.
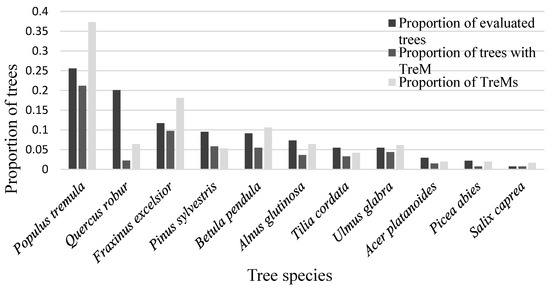
Figure 2.
Distribution of the evaluated retention trees, trees with TreMs, and sum of TreMs among different tree species.
The retention trees in the hemiboreal forests of Latvia exhibit dimensions that were in line with observations from other regions [31]. This suggests a certain degree of consistency in the size of retention trees across different geographical locations. The dimensions of the retention trees examined in this study were found to be similar to those observed in Finland [31], with the average diameter of 46.9 ± 0.8 cm (ranging from 16 to 99 cm) and the average height of 24.2 ± 0.5 m (ranging from 2.4 to 42.5 m) (Table S1) [31], yet smaller than in Germany [32] and bigger than in Estonia [33].
The abundance and richness of TreMs are associated with tree characteristics (diameter at breast height, tree species, and living status) [18,34] and with stand conditions such as topography, soils, humidity, forest management, forest type, and elevation [35,36]. Accordingly, tree living status (p < 0.01), diameter, and species (p < 0.05) were the most important factors affecting the abundance of TreMs (Figure 3). The primary driver for TreM formation was living status (Figure 3B), with deadwood having a significantly higher number of TreMs (p < 0.05), as similarly observed in other studies [14,18]. The overall survival of the retention trees was high [6], as only about 14% of the retention trees have died off since the harvest approximately 20 years ago. This survival is significantly higher compared to clear-cuts in Estonia [33]. Earlier studies conducted in the Baltic region suggest that certain tree species have a greater mortality rate for retention trees compared to others [6,8]; therefore, suitable species selection for retention trees is crucial, depending on the intended outcome. Stumps, predominantly formed by Populus tremula (68.75% of all detected stumps), were the most common form of deadwood (Figure 4). Lying dead wood, mainly composed of Fraxinus excelsior (60%), was the next most frequent type. Most of the snags were Ulmus glabra (50% of all snags). The formation of deadwood for these species was caused by the dieback [37,38,39], thus undermining their importance in the future as current deadwood decays away. In most cases, snags provide more TreMs than living trees of comparable size [14,18,40]. Snags have been found to be closely associated with TreM groups, which are defined by the presence of two key elements: woodpecker feeding holes and saproxylic fungi [14,18]. Yet, our study found that in 40% of cases, woodpecker feeding holes and 17% of saproxylic fungi were detected on stumps. These observations suggest that snags are not the exclusive substrates for these key TreM elements.
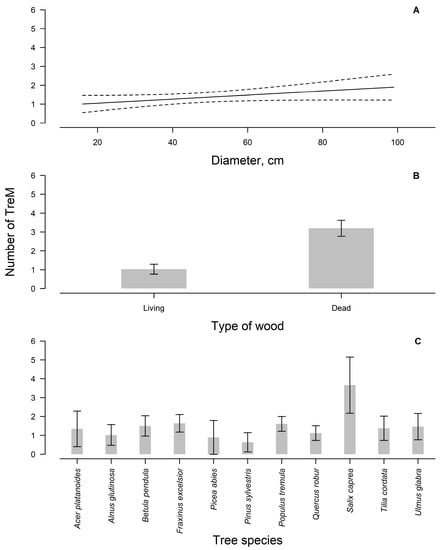
Figure 3.
The number of TreMs (±95% confidence interval) for different tree diameters (A); the number of TreM (±95% confidence interval) for dead and living deadwood (B); the number of TreM (±95% confidence interval) for different tree species (C).
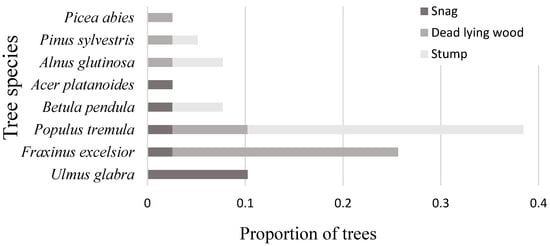
Figure 4.
The distribution of snags, dead lying wood, and stumps by tree species from total deadwood.
The decay rate, which differs widely among tree species, plays an important role in deadwood dynamics [41] and is likely a significant effect on TreM occurrence. The low wood density of fast-growing species, such as Populus tremula, influences their susceptibility to breakage, resulting in branches and crowns falling earlier than in hardwood species such as Quercus robur [37]. This is partially in line with our results, as Populus tremula was the primary producer of stumps, while dead Quercus robur were not found (Figure 4). This suggests that the retention trees of Quercus robur exhibit positive vitality following the clear-cutting process and increase their contribution to TreM formation after reaching a certain age and dimensions. In general, softwood species were the primary contributors to the production of deadwood, while hardwood species such as Tilia cordata and Quercus robur did not contribute to the formation of deadwood (Figure 4).
The tree diameter at breast height was the second strongest driver of TreM occurrence (Figure 3A), although the effect of the diameter differs by tree species [14]. Large-sized trees are especially valuable because they support all types of TreMs and increase the probability of their detection [14]. Trees with a diameter of 50–70 cm are considered to be the most valuable and are associated with TreMs such as dendrotelms, exposed heartwood, and sap run [14]. Yet, our study identified the occurrence of these TreMs on smaller trees as well (<50 cm).
Tree species play a significant role in shaping the abundance of TreMs in forest ecosystems [14,18,42], as confirmed by the findings of our study (Figure 3C). However, the formation of TreMs exhibited species-specific patterns, as consistent with observations from previous studies [17,43]. Populus tremula trees accounted for 79% of perennial polypores, while Fraxinus excelsior trees were linked to 41% of ephemeral fruiting bodies and 75% of vertebrate nests. In other studies, genera such as Abies, Alnus, Betula, Fagus, Prunus, Quercus, Sorbus, Tilia, and Ulmus have been mentioned as valuable in terms of TreMs [17]. However, our study highlighted Salix caprea, Populus tremula, and Fraxinus excelsior as the key species for facilitating the presence of TreMs (Figure 3C). Considering the impact of tree species biodiversity on the abundance and diversity of TreMs, it is crucial for forest management practices to prioritize the selection and retention of mixed tree species [17].
TreMs were detected on 168 trees (61.3% of all the evaluated trees), with 1.9 ± 0.1 (mean) and six (maximal) TreMs per tree, which is higher compared to other studies [40]. All forms of TreMs [13] were detected, with the most abundant being crown deadwood (Table S2). The abundance of this particular form of TreM is linked to the age of the trees, their wood properties, and the shape of their crowns [32]. In the evaluations of crown deadwood, it was determined that the majority of the crown deadwood consisted of dead branches (76.5%), followed by dead tops (12.2%), and the remaining 11.3% comprised broken limbs that were still present. Likewise, in previous studies, crown deadwood has been identified as the prevailing form of TreMs in terms of abundance [32,44]. Tree cavities were 19.9% of all TreMs detected, with 36.5% being insect galleries and bore holes, 39.7% being different types of rot holes, and 23.8% being woodpeckers foraging excavation and breeding cavities. The presence of rot holes has been associated with the provision of microhabitats for endangered epiphytic lichens, bryophytes [45], forest-dwelling organisms [46], and saproxylic insects [45,47]. Tree injuries and exposed wood were detected in 17.7%, from which 50% being bark loss, 16.7% trees with cracks, 6.6% trees with limb breakage, and 26.7% with stem breakage. Exudates (excretions) were less frequently observed (0.9%), deviating from the findings of other studies, where TreMs were evaluated in forest stands [46]. This might be attributed to the inclusion of different tree species in the analysis and variations in management practices.
Forest management often promotes the production of uniform stands with the removal of trees with low economic value, such as damaged trees [1,20]. Currently, conservation practices do not encompass the deliberate management of living trees to support specific TreMs, despite the recognition that the impacts of forestry can be alleviated by selecting retention trees that facilitate TreMs [4,16,48]. In order to increase the quantity of TreMs in the hemiboreal zone, it is advisable to select retention trees that have larger diameters and belong to specific species such as Populus tremula, Fraxinus excelsior, and Salix caprea (Figure 4C). By integrating TreMs alongside other biodiversity indicators, such as structural indicators, it becomes feasible to conserve a substantial portion of forest biodiversity [16].
4. Conclusions
This study contributes to the missing information regarding TreM research in Latvia [49]. Our results highlighted the importance of retention tree dimensions and species selection. Overall analysis demonstrated that bigger tree diameter and specific tree species (Populus tremula, Fraxinus excelsior, and Salix caprea) provide an increased number of TreMs; therefore, suitable species and size selection for retention trees is crucial, depending on the intended outcome. The study also emphasizes the significance of preserving deadwood within forest stands, as it enhances the observed abundance of TreMs.
The findings highlight the significance of making the selection of retention trees based on empirical evidence. This can lead to the optimization of microhabitat provision and a subsequent increase in biodiversity in young stands after clear-cutting. Although it is imperative to obtain more data to ensure the representation of additional tree species and a higher number of trees evaluated, this study focused on a specific geographic location, which may limit the generalizability of the results.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/f14101949/s1, Table S1. Descriptive statistics of evaluated trees; Table S2. Abundance of evaluated tree-related microhabitat forms and groups.
Author Contributions
Conceptualization, R.M. and D.J.; methodology, R.M.; software, R.M.; validation, R.M. and Ā.J.; formal analysis, R.M.; investigation, D.J. and L.G.-I.; resources; Ā.J.; data curation, D.J.; writing—original draft preparation, D.J.; writing—review and editing, R.M.; visualization, D.J.; supervision, R.M.; project administration, Ā.J. and Z.L.; funding acquisition, Ā.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Latvian State forests project “Effect of forestry on the forest ecosystem and related ecosystem services”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data can be obtained from the authors upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kuuluvainen, T. Forest Management and Biodiversity Conservation Based on Natural Ecosystem Dynamics in Northern Europe: The Complexity Challenge. Ambio 2009, 38, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Barahona-Segovia, R.M.; Grez, A.A.; Veloso, C. Forestry Clear-Cuts Increase Environmental Temperatures, Affecting the Ecophysiological Responses of Specialized Beetles in Fragmented Landscapes. J. Appl. Entomol. 2022, 146, 557–569. [Google Scholar] [CrossRef]
- Gustafsson, L.; Baker, S.C.; Bauhus, J.; Beese, W.J.; Brodie, A.; Kouki, J.; Lindenmayer, D.B.; Lhmus, A.; Pastur, G.M.; Messier, C.; et al. Retention Forestry to Maintain Multifunctional Forests: A World Perspective. Bioscience 2012, 62, 633–645. [Google Scholar] [CrossRef]
- Gustafsson, L.; Hannerz, M.; Koivula, M.; Shorohova, E.; Vanha-Majamaa, I.; Weslien, J. Research on Retention Forestry in Northern Europe. Ecol. Process. 2020, 9, 3. [Google Scholar] [CrossRef]
- Work, T.T.; Jacobs, J.M.; Spence, J.R.; Volney, W.J. High Levels of Green-Tree Retention Are Required to Preserve Ground Beetle Biodiversity in Boreal Mixedwood Forests. Ecol. Appl. 2010, 20, 741–751. [Google Scholar] [CrossRef]
- Rosenvald, R.; Lõhmus, A.; Kiviste, A. Preadaptation and Spatial Effects on Retention-Tree Survival in Cut Areas in Estonia. Can. J. For. Res. 2008, 38, 2616–2625. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Laurance, W.F.; Franklin, J.F. Ecology: Global Decline in Large Old Trees. Science 2012, 338, 1305–1306. [Google Scholar] [CrossRef]
- Hallinger, M.; Johansson, V.; Schmalholz, M.; Sjöberg, S.; Ranius, T. Factors Driving Tree Mortality in Retained Forest Fragments. For. Ecol. Manag. 2016, 368, 163–172. [Google Scholar] [CrossRef]
- Junninen, K.; Penttilä, R.; Martikainen, P. Fallen Retention Aspen Trees on Clear-Cuts Can Be Important Habitats for Red-Listed Polypores: A Case Study in Finland. Biodivers. Conserv. 2007, 16, 475–490. [Google Scholar] [CrossRef]
- Gustafsson, L.; Kouki, J.; Sverdrup-Thygeson, A. Tree Retention as a Conservation Measure in Clear-Cut Forests of Northern Europe: A Review of Ecological Consequences. Scand. J. For. Res. 2010, 25, 295–308. [Google Scholar] [CrossRef]
- Koivula, M. Alternative Harvesting Methods and Boreal Carabid Beetles (Coleoptera, Carabidae). For. Ecol. Manag. 2002, 167, 103–121. [Google Scholar] [CrossRef]
- Grindean, R.; Tanţău, I.; Feurdean, A. Linking Vegetation Dynamics and Stability in the Old-Growth Forests of Central Eastern Europe: Implications for Forest Conservation and Management. Biol. Conserv. 2019, 229, 160–169. [Google Scholar] [CrossRef]
- Larrieu, L.; Paillet, Y.; Winter, S.; Bütler, R.; Kraus, D.; Krumm, F.; Lachat, T.; Michel, A.K.; Regnery, B.; Vandekerkhove, K. Tree Related Microhabitats in Temperate and Mediterranean European Forests: A Hierarchical Typology for Inventory Standardization. Ecol. Indic. 2018, 84, 194–207. [Google Scholar] [CrossRef]
- Larrieu, L.; Cabanettes, A. Species, Live Status, and Diameter Are Important Tree Features for Diversity and Abundance of Tree Microhabitats in Subnatural Montane Beech-Fir Forests. Can. J. For. Res. 2012, 42, 1433–1445. [Google Scholar] [CrossRef]
- Finn, J.A. Ephemeral Resource Patches as Model Systems for Diversity-Function Experiments. Oikos 2001, 92, 363–366. [Google Scholar] [CrossRef]
- Asbeck, T.; Großmann, J.; Paillet, Y.; Winiger, N.; Bauhus, J. The Use of Tree-Related Microhabitats as Forest Biodiversity Indicators and to Guide Integrated Forest Management. Curr. For. Rep. 2021, 7, 59–68. [Google Scholar] [CrossRef]
- Courbaud, B.; Larrieu, L.; Kozak, D.; Kraus, D.; Lachat, T.; Ladet, S.; Müller, J.; Paillet, Y.; Sagheb-Talebi, K.; Schuck, A.; et al. Factors Influencing the Rate of Formation of Tree-Related Microhabitats and Implications for Biodiversity Conservation and Forest Management. J. Appl. Ecol. 2022, 59, 492–503. [Google Scholar] [CrossRef]
- Paillet, Y.; Debaive, N.; Archaux, F.; Cateau, E.; Gilg, O.; Guilbert, E. Nothing Else Matters? Tree Diameter and Living Status Have More Effects than Biogeoclimatic Context on Microhabitat Number and Occurrence: An Analysis in French Forest Reserves. PLoS ONE 2019, 14, 5. [Google Scholar] [CrossRef]
- Martin, M.; Fenton, N.J.; Morin, H. Tree-Related Microhabitats and Deadwood Dynamics Form a Diverse and Constantly Changing Mosaic of Habitats in Boreal Old-Growth Forests. Ecol. Indic. 2021, 128, 107813. [Google Scholar] [CrossRef]
- Kozák, D.; Mikoláš, M.; Svitok, M.; Bače, R.; Paillet, Y.; Larrieu, L.; Nagel, T.A.; Begovič, K.; Čada, V.; Diku, A.; et al. Profile of Tree-Related Microhabitats in European Primary Beech-Dominated Forests. For. Ecol. Manag. 2018, 429, 363–374. [Google Scholar] [CrossRef]
- Keren, S.; Diaci, J.; Motta, R.; Govedar, Z. Stand Structural Complexity of Mixed Old-Growth and Adjacent Selection Forests in the Dinaric Mountains of Bosnia and Herzegovina. For. Ecol. Manag. 2017, 400, 531–541. [Google Scholar] [CrossRef]
- Gustafsson, L.; Bauhus, J.; Asbeck, T.; Augustynczik, A.L.D.; Basile, M.; Frey, J.; Gutzat, F.; Hanewinkel, M.; Helbach, J.; Jonker, M.; et al. Retention as an Integrated Biodiversity Conservation Approach for Continuous-Cover Forestry in Europe. Ambio 2020, 49, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Paillet, Y.; Coutadeur, P.; Vuidot, A.; Archaux, F.; Gosselin, F. Strong Observer Effect on Tree Microhabitats Inventories: A Case Study in a French Lowland Forest. Ecol. Indic. 2015, 49, 14–23. [Google Scholar] [CrossRef]
- Harris, I.; Osborn, T.J.; Jones, P.; Lister, D. Version 4 of the CRU TS Monthly High-Resolution Gridded Multivariate Climate Dataset. Sci. Data 2020, 7, 109. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.M.; Walker, S.C. Fitting Linear Mixed-Effects Models Using Lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Akaike, H. A New Look at the Statistical Model Identification. IEEE Trans. Automat. Contr. 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Wald, A. Tests of Statistical Hypotheses Concerning Several Parameters When the Number of Observations Is Large. Trans. Am. Math. Soc. 1943, 54, 426. [Google Scholar] [CrossRef]
- Matisons, R.; Elferts, D.; Krišāns, O.; Schneck, V.; Gärtner, H.; Wojda, T.; Kowalczyk, J.; Jansons, Ā. Nonlinear Weather–Growth Relationships Suggest Disproportional Growth Changes of Norway Spruce in the Eastern Baltic Region. Forests 2021, 12, 661. [Google Scholar] [CrossRef]
- R Development Core Team. A Language and Environment for Statistical Computing; R Development Core Team: Vienna, Austria, 2022. [Google Scholar]
- Fescenko, A.; Nikodemus, O.; Brūmelis, G. Past and Contemporary Changes in Forest Cover and Forest Continuity in Relation to Soils (Southern Latvia). Pol. J. Ecol. 2014, 62, 625–638. [Google Scholar] [CrossRef]
- Hamalainen, A.; Hujo, M.; Heikkala, O.; Junninen, K.; Kouki, J. Retention Tree Characteristics Have Major Influence on the Post-Harvest Tree Mortality and Availability of Coarse Woody Debris in Clear-Cut Areas. For. Ecol. Manag. 2016, 369, 66–73. [Google Scholar] [CrossRef]
- Spînu, A.P.; Asbeck, T.; Bauhus, J. Combined Retention of Large Living and Dead Trees Can Improve Provision of Tree-Related Microhabitats in Central European Montane Forests. Eur. J. For. Res. 2022, 141, 1105–1120. [Google Scholar] [CrossRef]
- Rosenvald, R.; Lõhmus, P.; Rannap, R.; Remm, L.; Rosenvald, K.; Runnel, K.; Lõhmus, A. Assessing Long-Term Effectiveness of Green-Tree Retention. For. Ecol. Manag. 2019, 448, 543–548. [Google Scholar] [CrossRef]
- Kaufmann, S.; Hauck, M.; Leuschner, C. Effects of Natural Forest Dynamics on Vascular Plant, Bryophyte, and Lichen Diversity in Primeval Fagus Sylvatica Forests and Comparison with Production Forests. J. Ecol. 2018, 106, 2421–2434. [Google Scholar] [CrossRef]
- Larrieu, L.; Cabanettes, A.; Brin, A.; Bouget, C.; Deconchat, M. Tree Microhabitats at the Stand Scale in Montane Beech-Fir Forests: Practical Information for Taxa Conservation in Forestry. Eur. J. For. Res. 2014, 133, 355–367. [Google Scholar] [CrossRef]
- Asbeck, T.; Pyttel, P.; Frey, J.; Bauhus, J. Predicting Abundance and Diversity of Tree-Related Microhabitats in Central European Montane Forests from Common Forest Attributes. For. Ecol. Manag. 2019, 432, 400–408. [Google Scholar] [CrossRef]
- Basham, J.T. Stem Decay in Living Trees in Ontario’s Forests: A User’s Compendium and Guide; Minister of Supply and Services: Sault Ste. Marie, ON, Canada, 1991; ISBN 0832-7122. [Google Scholar]
- Davydenko, K.V.; Borysova, V.; Shcherbak, O.; Kryshtop, Y.; Meshkova, V. “Esche in Der Ukraine 2013–2018” Situation and Perspectives of European Ash (Fraxinus spp.) in Ukraine: Focus on Eastern Border. Balt. For. 2019, 29, 193–202. [Google Scholar] [CrossRef]
- Jürisoo, L.; Adamson, K.; Padari, A.; Drenkhan, R. Health of Elms and Dutch Elm Disease in Estonia. Eur. J. Plant Pathol. 2019, 154, 823–841. [Google Scholar] [CrossRef]
- Sever, K.; Nagel, T.A. Patterns of Tree Microhabitats across a Gradient of Managed to Old-Growth Conditions: A Case Study from Beech Dominated Forests of South-Eastern Slovenia. Acta Silvae Ligni 2020, 118, 29–40. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Cornelissen, J.H.C.; Allison, S.D.; Bauhus, J.; Eggleton, P.; Preston, C.M.; Scarff, F.; Weedon, J.T.; Wirth, C.; Zanne, A.E. Plant Traits and Wood Fates across the Globe: Rotted, Burned, or Consumed? Glob. Chang. Biol. 2009, 15, 2431–2449. [Google Scholar] [CrossRef]
- Vuidot, A.; Paillet, Y.; Archaux, F.; Gosselin, F. Influence of Tree Characteristics and Forest Management on Tree Microhabitats. Biol. Conserv. 2011, 144, 441–450. [Google Scholar] [CrossRef]
- Santopuoli, G.; di Cristofaro, M.; Kraus, D.; Schuck, A.; Lasserre, B.; Marchetti, M. Biodiversity Conservation and Wood Production in a Natura 2000 Mediterranean Forest. A Trade-off Evaluation Focused on the Occurrence of Microhabitats. IForest 2019, 12, 76–84. [Google Scholar] [CrossRef]
- Großmann, J.; Carlson, L.; Kändler, G.; Pyttel, P.; Kleinschmit, J.R.G.; Bauhus, J. Evaluating Retention Forestry 10 Years after Its Introduction in Temperate Forests Regarding the Provision of Tree-Related Microhabitats and Dead Wood. Eur. J. For. Res. 2023, 142, 1125–1147. [Google Scholar] [CrossRef]
- Fritz, Ö.; Heilmann-Clausen, J. Rot Holes Create Key Microhabitats for Epiphytic Lichens and Bryophytes on Beech (Fagus sylvatica). Biol. Conserv. 2010, 143, 1008–1016. [Google Scholar] [CrossRef]
- Basile, M.; Asbeck, T.; Jonker, M.; Knuff, A.K.; Bauhus, J. What Do Tree-Related Microhabitats Tell Us about the Abundance of Forest-Dwelling Bats, Birds, and Insects? J. Environ. Manag. 2020, 264, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Müller, J.; Bussler, H.; Gossner, M.M.; Forest, B.; Park, N. Hollow Beech Trees Identified as Keystone Structures for Saproxylic Beetles by Analyses of Functional and Phylogenetic Diversity. Anim. Conserv. 2014, 17, 154–162. [Google Scholar] [CrossRef]
- Mölder, A.; Schmidt, M.; Plieninger, T.; Meyer, P. Habitat-Tree Protection Concepts over 200 Years. Conserv. Biol. 2020, 34, 1444–1451. [Google Scholar] [CrossRef] [PubMed]
- Mason, W.L.; Diaci, J.; Carvalho, J.; Valkonen, S. Continuous Cover Forestry in Europe: Usage and the Knowledge Gaps and Challenges to Wider Adoption. For. Int. J. For. Res. 2022, 95, 1–12. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).