Assessing Tree Drought Resistance and Climate-Growth Relationships under Different Tree Age Classes in a Pinus nigra Arn. ssp. salzmannii Forest
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Field Sampling and Dendrochronological Methods
2.3. Growth Stability Components in Response to Drought Events
2.4. Data Analysis
3. Results
3.1. Characteristics of the Sampled Stands and Trees
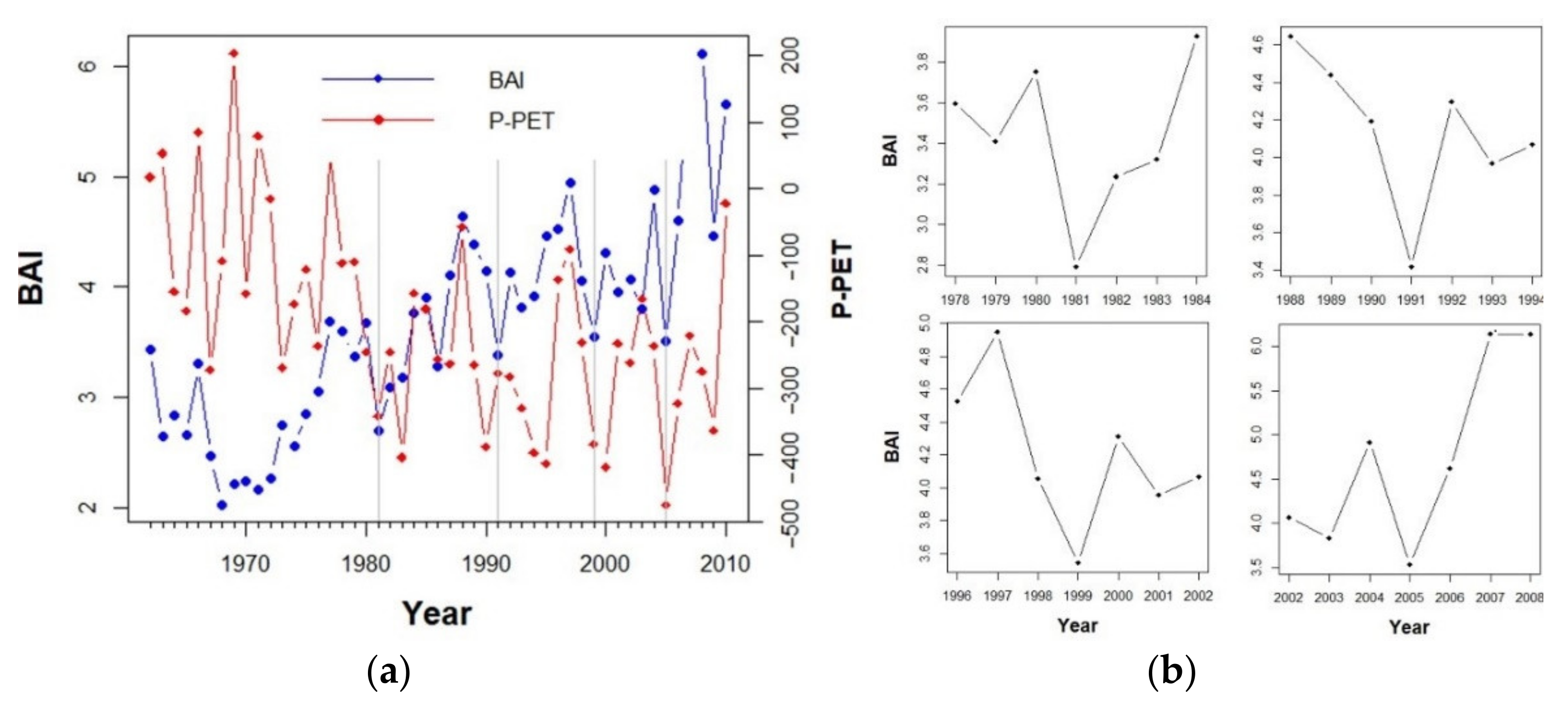
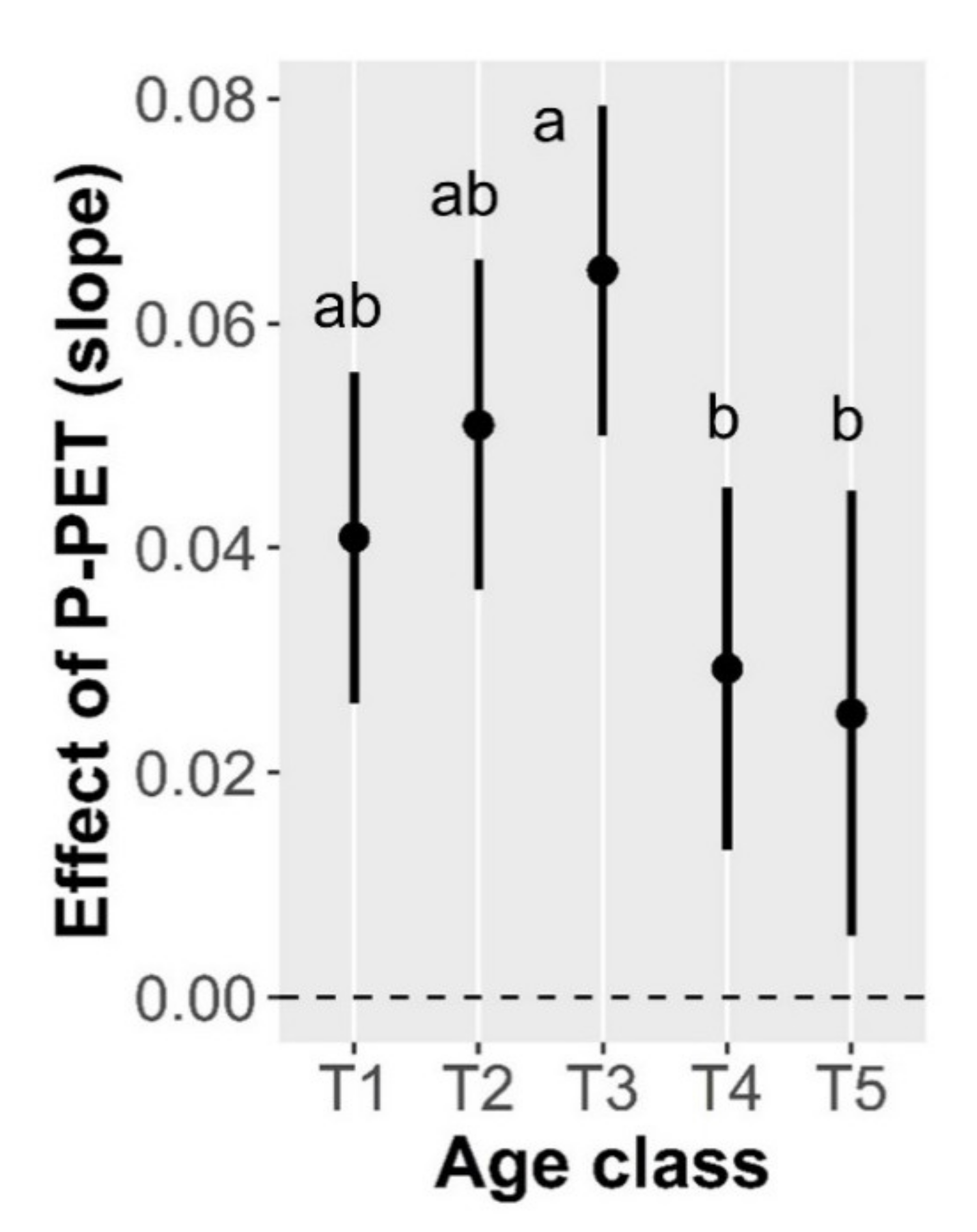
3.2. Drivers of Tree Basal Area Increment (BAI)
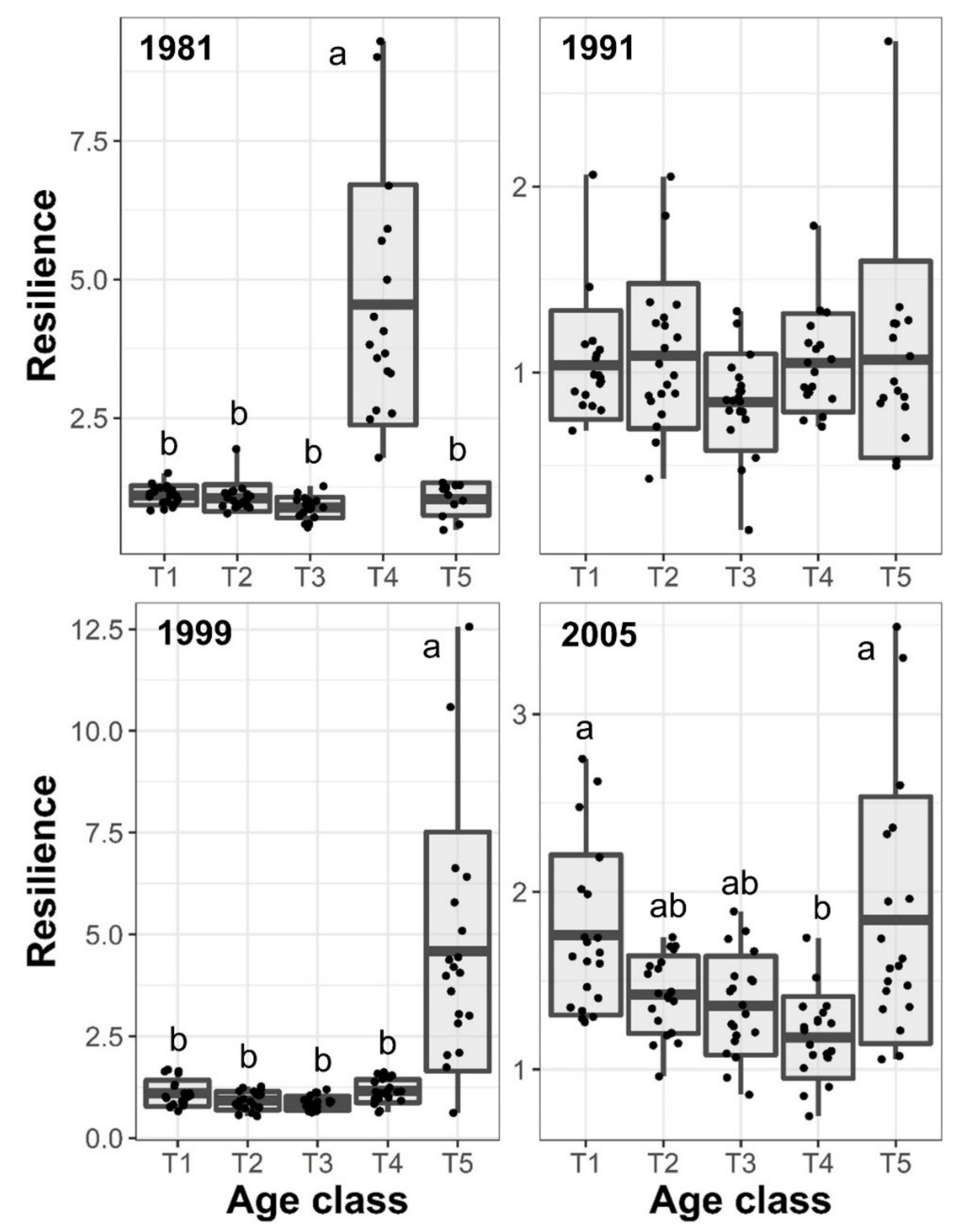
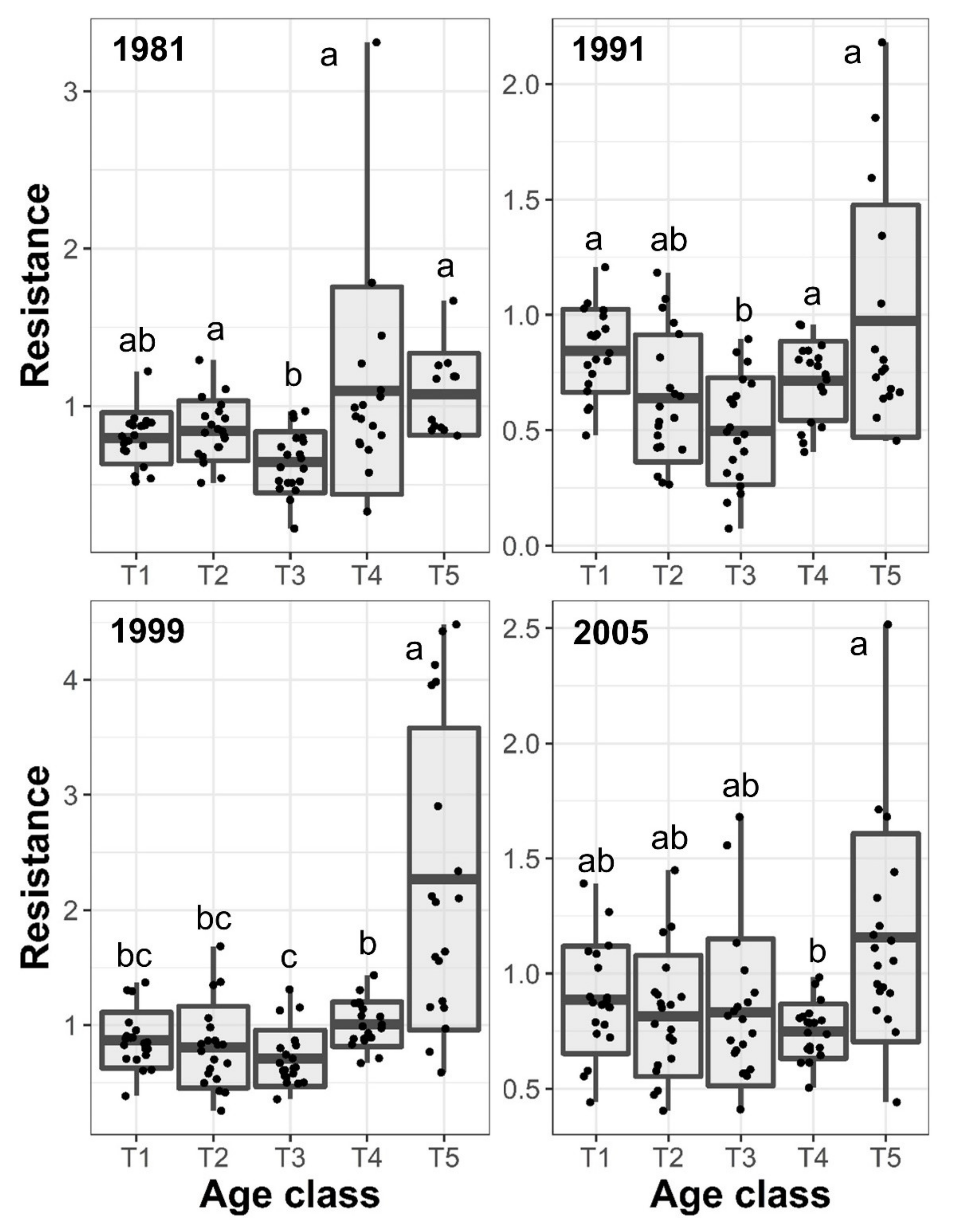
3.3. Resilience and Resistance to Drought Events
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dai, A. Increasing Drought under Global Warming in Observations and Models. Nat. Clim. Chang. 2013, 3, 52–58. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.T. A Global Overview of Drought and Heat-Induced Tree Mortality Reveals Emerging Climate Change Risks for Forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Andrews, C.M.; D’Amato, A.W.; Fraver, S.; Palik, B.; Battaglia, M.A.; Bradford, J.B. Low Stand Density Moderates Growth Declines during Hot Droughts in Semi-arid Forests. J. Appl. Ecol. 2020, 57, 1089–1102. [Google Scholar] [CrossRef]
- Bose, A.K.; Nelson, A.S.; Kane, M.; Rigling, A. Density Reduction in Loblolly Pine (Pinus Taeda L.) Stands to Increase Tree C Assimilation: An Approach with the Dual δ 13 C and δ 18 O Isotope Signatures in Needles. Ann. For. Sci. 2018, 75, 1–12. [Google Scholar] [CrossRef]
- Bottero, A.; Forrester, D.I.; Cailleret, M.; Kohnle, U.; Gessler, A.; Michel, D.; Bose, A.K.; Bauhus, J.; Bugmann, H.; Cuntz, M. Growth Resistance and Resilience of Mixed Silver Fir and Norway Spruce Forests in Central Europe: Contrasting Responses to Mild and Severe Droughts. Glob. Chang. Biol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Bose, A.K.; Gessler, A.; Bolte, A.; Bottero, A.; Buras, A.; Cailleret, M.; Camarero, J.J.; Haeni, M.; Hereş, A.; Hevia, A. Growth and Resilience Responses of Scots Pine to Extreme Droughts across Europe Depend on Predrought Growth Conditions. Glob. Chang. Biol. 2020, 26, 4521–4537. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Camarero, J.J.; Hevia, A.; Madrigal-González, J.; Linares, J.C.; Ballesteros-Canovas, J.A.; Sánchez-Miranda, A.; Alfaro-Sánchez, R.; Sangüesa-Barreda, G.; Galván, J.D. What Drives Growth of Scots Pine in Continental Mediterranean Climates: Drought, Low Temperatures or Both? Agric. For. Meteorol. 2015, 206, 151–162. [Google Scholar] [CrossRef]
- Zang, C.; Hartl-Meier, C.; Dittmar, C.; Rothe, A.; Menzel, A. Patterns of Drought Tolerance in Major European Temperate Forest Trees: Climatic Drivers and Levels of Variability. Glob. Chang. Biol. 2014, 20, 3767–3779. [Google Scholar] [CrossRef]
- Lévesque, M.; Saurer, M.; Siegwolf, R.; Eilmann, B.; Brang, P.; Bugmann, H.; Rigling, A. Drought Response of Five Conifer Species under Contrasting Water Availability Suggests High Vulnerability of Norway Spruce and European Larch. Glob. Chang. Biol. 2013, 19, 3184–3199. [Google Scholar] [CrossRef]
- Bennett, A.C.; McDowell, N.G.; Allen, C.D.; Anderson-Teixeira, K.J. Larger Trees Suffer Most during Drought in Forests Worldwide. Nat. Plants 2015, 1, 1–5. [Google Scholar] [CrossRef]
- Pretzsch, H.; Schütze, G.; Biber, P. Drought Can Favour the Growth of Small in Relation to Tall Trees in Mature Stands of Norway Spruce and European Beech. For. Ecosyst. 2018, 5, 1–19. [Google Scholar] [CrossRef]
- Colangelo, M.; Camarero, J.J.; Borghetti, M.; Gazol, A.; Gentilesca, T.; Ripullone, F. Size Matters a Lot: Drought-Affected Italian Oaks Are Smaller and Show Lower Growth Prior to Tree Death. Front. Plant Sci. 2017, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Van Mantgem, P.J.; Stephenson, N.L.; Byrne, J.C.; Daniels, L.D.; Franklin, J.F.; Fulé, P.Z.; Harmon, M.E.; Larson, A.J.; Smith, J.M.; Taylor, A.H. Widespread Increase of Tree Mortality Rates in the Western United States. Science 2009, 323, 521–524. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, A.W.; Bradford, J.B.; Fraver, S.; Palik, B.J. Effects of Thinning on Drought Vulnerability and Climate Response in North Temperate Forest Ecosystems. Ecol. Appl. 2013, 23, 1735–1742. [Google Scholar] [CrossRef]
- Forrester, D.I. Linking Forest Growth with Stand Structure: Tree Size Inequality, Tree Growth or Resource Partitioning and the Asymmetry of Competition. For. Ecol. Manag. 2019, 447, 139–157. [Google Scholar] [CrossRef]
- Sumner, G.N.; Romero, R.; Homar, V.; Ramis, C.; Alonso, S.; Zorita, E. An Estimate of the Effects of Climate Change on the Rainfall of Mediterranean Spain by the Late Twenty First Century. Clim. Dyn. 2003, 20, 789–805. [Google Scholar] [CrossRef]
- Solomon, S.; Manning, M.; Marquis, M.; Qin, D. Climate Change 2007-the Physical Science Basis: Working Group I Contribution to the Fourth Assessment Report of the IPCC; Cambridge University Press: Cambridge, UK, 2007; Volume 4, ISBN 0521705967. [Google Scholar]
- Loustau, D.; Bosc, A.; Colin, A.; Ogée, J.; Davi, H.; François, C.; Dufrêne, E.; Déqué, M.; Cloppet, E.; Arrouays, D. Modeling Climate Change Effects on the Potential Production of French Plains Forests at the Sub-Regional Level. Tree Physiol. 2005, 25, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Granier, A.; Reichstein, M.; Breda, N.; Janssens, I.A.; Falge, E.; Ciais, P.; Grünwald, T.; Aubinet, M.; Berbigier, P.; Bernhofer, C. Evidence for Soil Water Control on Carbon and Water Dynamics in European Forests during the Extremely Dry Year: 2003. Agric. For. Meteorol. 2007, 143, 123–145. [Google Scholar] [CrossRef]
- Martínez-Vilalta, J.; López, B.C.; Adell, N.; Badiella, L.; Ninyerola, M. Twentieth Century Increase of Scots Pine Radial Growth in NE Spain Shows Strong Climate Interactions. Glob. Chang. Biol. 2008, 14, 2868–2881. [Google Scholar] [CrossRef]
- Allen, C.D. Climate-Induced Forest Dieback: An Escalating Global Phenomenon. Unasylva 2009, 231, 60. [Google Scholar]
- Adams, J. Vegetation-Climate Interaction: How Plants Make the Global Environment; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009; ISBN 364200881X. [Google Scholar]
- Boisvenue, C.; Running, S.W. Impacts of Climate Change on Natural Forest Productivity–Evidence since the Middle of the 20th Century. Glob. Chang. Biol. 2006, 12, 862–882. [Google Scholar] [CrossRef]
- Andreu, L.; Gutierrez, E.; Macias, M.; Ribas, M.; Bosch, O.; Camarero, J.J. Climate Increases Regional Tree-growth Variability in Iberian Pine Forests. Glob. Chang. Biol. 2007, 13, 804–815. [Google Scholar] [CrossRef]
- Linares, J.C.; Tíscar, P.A. Buffered Climate Change Effects in a Mediterranean Pine Species: Range Limit Implications from a Tree-Ring Study. Oecologia 2011, 167, 847–859. [Google Scholar] [CrossRef] [PubMed]
- Vaganov, E.A.; Hughes, M.K.; Shashkin, A. V Growth Dynamics of Conifer Tree Rings: Images of Past and Future Environments; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2006; Volume 183, ISBN 3540312986. [Google Scholar]
- Keeling, C.D.; Chin, J.F.S.; Whorf, T.P. Increased Activity of Northern Vegetation Inferred from Atmospheric CO2 Measurements. Nature 1996, 382, 146–149. [Google Scholar] [CrossRef]
- Linares, J.C.; Tíscar, P.A. Climate Change Impacts and Vulnerability of the Southern Populations of Pinus Nigra Subsp. Salzmannii. Tree Physiol. 2010, 30, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, S.; Gracia, C.A.; Sánchez, A. Likely Effects of Climate Change on Growth of Quercus Ilex, Pinus Halepensis, Pinus Pinaster, Pinus Sylvestris and Fagus Sylvatica Forests in the Mediterranean Region. For. Ecol. Manag. 2002, 162, 23–37. [Google Scholar] [CrossRef]
- Antonova, G.F.; Cherkashin, V.P.; Stasova, V.V.; Varaksina, T.N. Daily Dynamics in Xylem Cell Radial Growth of Scots Pine (Pinus Sylvestris L.). Trees 1995, 10, 24–30. [Google Scholar] [CrossRef]
- Rossi, S.; Deslauriers, A.; Anfodillo, T.; Morin, H.; Saracino, A.; Motta, R.; Borghetti, M. Conifers in Cold Environments Synchronize Maximum Growth Rate of Tree-ring Formation with Day Length. New Phytol. 2006, 170, 301–310. [Google Scholar] [CrossRef]
- Ko Heinrichs, D.; Tardif, J.C.; Bergeron, Y. Xylem Production in Six Tree Species Growing on an Island in the Boreal Forest Region of Western Quebec, Canada. Botany 2007, 85, 518–525. [Google Scholar] [CrossRef]
- Camarero, J.J. Direct and indirect effects of the North Atlantic Oscillation on tree growth and forest decline in northeastern Spain. In Hydrological, Socioeconomic and Ecological Impacts of the North Atlantic Oscillation in the Mediterranean Region; Springer: Berlin/Heidelberg, Germany, 2011; pp. 129–152. [Google Scholar]
- Carrer, M. Individualistic and Time-Varying Tree-Ring Growth to Climate Sensitivity. PLoS ONE 2011, 6, e22813. [Google Scholar] [CrossRef]
- De Luis, M.; Gričar, J.; Čufar, K.; Raventós, J. Seasonal Dynamics of Wood Formation in Pinus Halepensis from Dry and Semi-Arid Ecosystems in Spain. IAWA J. 2007, 28, 389–404. [Google Scholar] [CrossRef]
- Martín-Benito, D.; Del Río, M.; Cañellas, I. Black Pine (Pinus Nigra Arn.) Growth Divergence along a Latitudinal Gradient in Western Mediterranean Mountains. Ann. For. Sci. 2010, 67, 401. [Google Scholar] [CrossRef]
- Martin-Benito, D.; Kint, V.; Del Rio, M.; Muys, B.; Cañellas, I. Growth Responses of West-Mediterranean Pinus Nigra to Climate Change Are Modulated by Competition and Productivity: Past Trends and Future Perspectives. For. Ecol. Manag. 2011, 262, 1030–1040. [Google Scholar] [CrossRef]
- Ferguson, I.S. Sustainable Forest Management; Oxford University Press: Australia, 1996; ISBN 0195506286. [Google Scholar]
- Entry, J.A.; Emmingham, W.H. Influence of Forest Age on Forms of Carbon in Douglas-Fir Soils in the Oregon Coast Range. Can. J. For. Res. 1998, 28, 390–395. [Google Scholar] [CrossRef]
- Mund, M.; Schulze, E. Impacts of Forest Management on the Carbon Budget of European Beech (Fagus Sylvatica) Forests. Allg. Forst Jagdztg. 2006, 177, 47. [Google Scholar]
- Jandl, R.; Lindner, M.; Vesterdal, L.; Bauwens, B.; Baritz, R.; Hagedorn, F.; Johnson, D.W.; Minkkinen, K.; Byrne, K.A. How Strongly Can Forest Management Influence Soil Carbon Sequestration? Geoderma 2007, 137, 253–268. [Google Scholar] [CrossRef]
- Hedo, J.; Lucas-Borja, M.E.; Wic, C.; Andrés-Abellán, M.; de Las Heras, J. Soil Microbiological Properties and Enzymatic Activities of Long-Term Post-Fire Recovery in Dry and Semiarid Aleppo Pine (Pinus Halepensis M.) Forest Stands. Solid Earth 2015, 6, 243–252. [Google Scholar] [CrossRef]
- Lucas-Borja, M.E.; Hedo, J.; Cerdá, A.; Candel-Pérez, D.; Viñegla, B. Unravelling the Importance of Forest Age Stand and Forest Structure Driving Microbiological Soil Properties, Enzymatic Activities and Soil Nutrients Content in Mediterranean Spanish Black Pine (Pinus Nigra Ar. Ssp. Salzmannii). Sci. Total Environ. 2016, 562, 145–154. [Google Scholar] [CrossRef]
- Lucas-Borja, M.E.; Ahrazem, O.; Candel-Pérez, D.; Moya, D.; Fonseca, T.; Tecles, E.H.; De las Heras, J.; Gómez-Gómez, L. Evaluation of Fire Recurrence Effect on Genetic Diversity in Maritime Pine (Pinus Pinaster Ait.) Stands Using Inter-Simple Sequence Repeat Profiles. Sci. Total Environ. 2016, 572, 1322–1328. [Google Scholar] [CrossRef]
- Bauhus, J.; Pare, D. Effects of Tree Species, Stand Age and Soil Type on Soil Microbial Biomass and Its Activity in a Southern Boreal Forest. Soil Biol. Biochem. 1998, 30, 1077–1089. [Google Scholar] [CrossRef]
- Allue Andrade, J.L. Phytoclimatic Atlas of Spain. In Phytoclimatic atlas of Spain. Taxonomies; Ministerio de Agricultura Pesca y Alimentación: Madrid, Spain, 1990. [Google Scholar]
- Staff, S.S. Keys to Soil Taxonomy; Department of Agriculture: Washington, DC, USA, 2014.
- Candel-Pérez, D.; Lucas-Borja, M.E.; Linares, J.C. Predicciones Del Crecimiento En Poblaciones de Pino Laricio (Pinus Nigra Arn. Ssp. Salzmannii) Bajo Diferentes Escenarios Futuros de Cambio Climático. Rev. Ecosistemas 2012, 21, 41–49. [Google Scholar] [CrossRef]
- Fritts, H.C. The Climate-Growth System. In Tree-Ring Climate; Academic Press: London, UK, 1976; pp. 207–242. ISBN 9780122684500. Available online: https://doi.org/10.1016/B978-0-12-268450-0.X5001-0 (accessed on 7 August 2021). [CrossRef]
- Holmes, R.L. Program COFECHA User’s Manual; Laboratory of Tree-Ring Research, The University of Arizona: Tucson, Arizona, 1983. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Smith, G.M. Analyzing Ecological Data; Springer: Berlin/Heidelberg, Germany, 2007; ISBN 0387459723. [Google Scholar]
- Vicente-Serrano, S.M.; Lasanta, T.; Gracia, C. Aridification Determines Changes in Forest Growth in Pinus Halepensis Forests under Semiarid Mediterranean Climate Conditions. Agric. For. Meteorol. 2010, 150, 614–628. [Google Scholar] [CrossRef]
- Schweingruber, F.H.; Eckstein, D.; Serre-Bachet, F.; Bräker, O.U. Identification, Presentation and Interpretation of Event Years and Pointer Years in Dendrochronology. Dendrochronologia 1990, 8, 9–38. [Google Scholar]
- Lloret, F.; Keeling, E.G.; Sala, A. Components of Tree Resilience: Effects of Successive Low-growth Episodes in Old Ponderosa Pine Forests. Oikos 2011, 120, 1909–1920. [Google Scholar] [CrossRef]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009; ISBN 0387874585. [Google Scholar]
- Nakagawa, S.; Schielzeth, H. A General and Simple Method for Obtaining R2 from Generalized Linear Mixed-effects Models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Pinheiro, J.; Bornkamp, B.; Glimm, E.; Bretz, F. Model-based Dose Finding under Model Uncertainty Using General Parametric Models. Stat. Med. 2014, 33, 1646–1661. [Google Scholar] [CrossRef] [PubMed]
- Breheny, P.; Burchett, W. Visualization of Regression Models Using Visreg. R J. 2017, 9, 56. [Google Scholar] [CrossRef]
- Van der Maaten-Theunissen, M.; van der Maaten, E.; Bouriaud, O. PointRes: An R Package to Analyze Pointer Years and Components of Resilience. Dendrochronologia 2015, 35, 34–38. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Camarero, J.J.; Dobbertin, M.; Fernández-Cancio, Á.; Vilà-Cabrera, A.; Manzanedo, R.D.; Zavala, M.A.; Navarro-Cerrillo, R.M. Contrasting Vulnerability and Resilience to Drought-Induced Decline of Densely Planted vs. Natural Rear-Edge Pinus Nigra Forests. For. Ecol. Manag. 2013, 310, 956–967. [Google Scholar] [CrossRef]
- González de Andrés, E.; Camarero, J.J. Disentangling Mechanisms of Drought-Induced Dieback in Pinus Nigra Arn. from Growth and Wood Isotope Patterns. Forests 2020, 11, 1339. [Google Scholar] [CrossRef]
- Savi, T.; Casolo, V.; Dal Borgo, A.; Rosner, S.; Torboli, V.; Stenni, B.; Bertoncin, P.; Martellos, S.; Pallavicini, A.; Nardini, A. Drought-Induced Dieback of Pinus Nigra: A Tale of Hydraulic Failure and Carbon Starvation. Conserv. Physiol. 2019, 7, coz012. [Google Scholar] [PubMed]
- Spinoni, J.; Vogt, J.V.; Naumann, G.; Barbosa, P.; Dosio, A. Will Drought Events Become More Frequent and Severe in Europe? Int. J. Climatol. 2018, 38, 1718–1736. [Google Scholar] [CrossRef]
- Martín-Benito, D.; Del Río, M.; Heinrich, I.; Helle, G.; Cañellas, I. Response of Climate-Growth Relationships and Water Use Efficiency to Thinning in a Pinus Nigra Afforestation. For. Ecol. Manag. 2010, 259, 967–975. [Google Scholar] [CrossRef]
- Navarro-Cerrillo, R.M.; Sánchez-Salguero, R.; Rodriguez, C.; Lazo, J.D.; Moreno-Rojas, J.M.; Palacios-Rodriguez, G.; Camarero, J.J. Is Thinning an Alternative When Trees Could Die in Response to Drought? The Case of Planted Pinus Nigra and P. Sylvestris Stands in Southern Spain. For. Ecol. Manag. 2019, 433, 313–324. [Google Scholar] [CrossRef]
- Petrucco, L.; Nardini, A.; Von Arx, G.; Saurer, M.; Cherubini, P. Isotope Signals and Anatomical Features in Tree Rings Suggest a Role for Hydraulic Strategies in Diffuse Drought-Induced Die-Back of Pinus Nigra. Tree Physiol. 2017, 37, 523–535. [Google Scholar] [PubMed]
- Magnani, F.; Mencuccini, M.; Grace, J. Age-related Decline in Stand Productivity: The Role of Structural Acclimation under Hydraulic Constraints. Plant Cell Environ. 2000, 23, 251–263. [Google Scholar] [CrossRef]
- Rukh, S.; Poschenrieder, W.; Heym, M.; Pretzsch, H. Drought Resistance of Norway Spruce (Picea Abies [L.] Karst) and European Beech (Fagus Sylvatica [L.]) in Mixed vs. Monospecific Stands and on Dry vs. Wet Sites. From Evidence at the Tree Level to Relevance at the Stand Level. Forests 2020, 11, 639. [Google Scholar] [CrossRef]
- Mencuccini, M. The Ecological Significance of Long-distance Water Transport: Short-term Regulation, Long-term Acclimation and the Hydraulic Costs of Stature across Plant Life Forms. Plant Cell Environ. 2003, 26, 163–182. [Google Scholar] [CrossRef]
- Olson, M.E.; Soriano, D.; Rosell, J.A.; Anfodillo, T.; Donoghue, M.J.; Edwards, E.J.; León-Gómez, C.; Dawson, T.; Martínez, J.J.C.; Castorena, M. Plant Height and Hydraulic Vulnerability to Drought and Cold. Proc. Natl. Acad. Sci. USA 2018, 115, 7551–7556. [Google Scholar] [CrossRef]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G. Mechanisms of Plant Survival and Mortality during Drought: Why Do Some Plants Survive While Others Succumb to Drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef]
- Choat, B.; Brodribb, T.J.; Brodersen, C.R.; Duursma, R.A.; López, R.; Medlyn, B.E. Triggers of Tree Mortality under Drought. Nature 2018, 558, 531–539. [Google Scholar] [CrossRef]
- Ryan, M.G.; Yoder, B.J. Hydraulic Limits to Tree Height and Tree Growth. Bioscience 1997, 47, 235–242. [Google Scholar] [CrossRef]
- Arend, M.; Sever, K.; Pflug, E.; Gessler, A.; Schaub, M. Seasonal Photosynthetic Response of European Beech to Severe Summer Drought: Limitation, Recovery and Post-Drought Stimulation. Agric. For. Meteorol. 2016, 220, 83–89. [Google Scholar] [CrossRef]
- Trugman, A.T.; Detto, M.; Bartlett, M.K.; Medvigy, D.; Anderegg, W.R.L.; Schwalm, C.; Schaffer, B.; Pacala, S.W. Tree Carbon Allocation Explains Forest Drought-kill and Recovery Patterns. Ecol. Lett. 2018, 21, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- Ovenden, T.S.; Perks, M.P.; Clarke, T.; Mencuccini, M.; Jump, A.S. Life after Recovery: Increased Resolution of Forest Resilience Assessment Sheds New Light on Post-drought Compensatory Growth and Recovery Dynamics. J. Ecol. 2021. [Google Scholar] [CrossRef]
- Bose, A.K.; Scherrer, D.; Camarero, J.J.; Ziche, D.; Babst, F.; Bigler, C.; Bolte, A.; Dorado-Liñán, I.; Etzold, S.; Fonti, P. Climate Sensitivity and Drought Seasonality Determine Post-Drought Growth Recovery of Quercus Petraea and Quercus Robur in Europe. Sci. Total Environ. 2021, 784, 147222. [Google Scholar] [CrossRef] [PubMed]
- Anderegg, W.R.L.; Schwalm, C.; Biondi, F.; Camarero, J.J.; Koch, G.; Litvak, M.; Ogle, K.; Shaw, J.D.; Shevliakova, E.; Williams, A.P. Pervasive Drought Legacies in Forest Ecosystems and Their Implications for Carbon Cycle Models. Science 2015, 349, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Wang, X.; Keenan, T.F.; Piao, S. Drought Timing Influences the Legacy of Tree Growth Recovery. Glob. Chang. Biol. 2018, 24, 3546–3559. [Google Scholar] [CrossRef]
- Sohn, J.A.; Saha, S.; Bauhus, J. Potential of Forest Thinning to Mitigate Drought Stress: A Meta-Analysis. For. Ecol. Manag. 2016, 380, 261–273. [Google Scholar] [CrossRef]
- Fernandes, T.J.G.; Del Campo, A.D.; Herrera, R.; Molina, A.J. Simultaneous Assessment, through Sap Flow and Stable Isotopes, of Water Use Efficiency (WUE) in Thinned Pines Shows Improvement in Growth, Tree-Climate Sensitivity and WUE, but Not in WUEi. For. Ecol. Manag. 2016, 361, 298–308. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Sangüesa-Barreda, G.; Oliva, J.; Vicente-Serrano, S.M. To Die or Not to Die: Early Warnings of Tree Dieback in Response to a Severe Drought. J. Ecol. 2015, 103, 44–57. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Navarro-Cerrillo, R.M.; Camarero, J.J.; Fernández-Cancio, Á. Selective Drought-Induced Decline of Pine Species in Southeastern Spain. Clim. Change 2012, 113, 767–785. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Linares, J.C.; Camarero, J.J.; Madrigal-González, J.; Hevia, A.; Sánchez-Miranda, Á.; Ballesteros-Cánovas, J.A.; Alfaro-Sánchez, R.; García-Cervigón, A.I.; Bigler, C. Disentangling the Effects of Competition and Climate on Individual Tree Growth: A Retrospective and Dynamic Approach in Scots Pine. For. Ecol. Manag. 2015, 358, 12–25. [Google Scholar] [CrossRef]

| Fixed Effect | F-Value | p-Value |
|---|---|---|
| Size | 398.14 | < 0.0001 |
| Age-class | 5.63 | 0.0939 |
| Water balance | 122.35 | < 0.0001 |
| Temperature | 29.70 | < 0.0001 |
| Stand basal area | 0.02 | 0.8993 |
| Stand density | 0.15 | 0.7251 |
| Age-class × Water balance | 3.88 | 0.0038 |
| 1981 | 1991 | 1999 | 2005 | |||||
|---|---|---|---|---|---|---|---|---|
| Fixed Effect | F | p | F | p | F | p | F | p |
| Resilience | ||||||||
| Age class | 96.52 | 0.0003 | 2.45 | 0.2419 | 79.41 | 0.0001 | 12.98 | 0.0075 |
| Tree size | 2.33 | 0.1306 | 0.94 | 0.3351 | 2.97 | 0.0885 | 7.37 | 0.0080 |
| Stand basal area | 5.42 | 0.1022 | 0.03 | 0.8784 | 0.03 | 0.8765 | 4.83 | 0.1153 |
| Stand density | 0.13 | 0.7444 | 1.50 | 0.3046 | 0.38 | 0.5795 | 0.17 | 0.7073 |
| Resistance | ||||||||
| Age class | 7.56 | 0.0238 | 7.92 | 0.0217 | 28.06 | 0.0013 | 5.73 | 0.0414 |
| Tree size | 0.01 | 0.9373 | 0.31 | 0.5803 | 0.60 | 0.4420 | 0.14 | 0.7130 |
| Stand basal area | 0.12 | 0.7486 | 0.60 | 0.4949 | 0.75 | 0.4488 | 0.49 | 0.5336 |
| Stand density | 0.48 | 0.5398 | 0.08 | 0.7893 | 0.21 | 0.6776 | 0.87 | 0.4198 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucas-Borja, M.E.; Bose, A.K.; Andivia, E.; Candel-Pérez, D.; Plaza-Álvarez, P.A.; Linares, J.C. Assessing Tree Drought Resistance and Climate-Growth Relationships under Different Tree Age Classes in a Pinus nigra Arn. ssp. salzmannii Forest. Forests 2021, 12, 1161. https://doi.org/10.3390/f12091161
Lucas-Borja ME, Bose AK, Andivia E, Candel-Pérez D, Plaza-Álvarez PA, Linares JC. Assessing Tree Drought Resistance and Climate-Growth Relationships under Different Tree Age Classes in a Pinus nigra Arn. ssp. salzmannii Forest. Forests. 2021; 12(9):1161. https://doi.org/10.3390/f12091161
Chicago/Turabian StyleLucas-Borja, Manuel Esteban, Arun K. Bose, Enrique Andivia, David Candel-Pérez, Pedro A. Plaza-Álvarez, and Juan C. Linares. 2021. "Assessing Tree Drought Resistance and Climate-Growth Relationships under Different Tree Age Classes in a Pinus nigra Arn. ssp. salzmannii Forest" Forests 12, no. 9: 1161. https://doi.org/10.3390/f12091161
APA StyleLucas-Borja, M. E., Bose, A. K., Andivia, E., Candel-Pérez, D., Plaza-Álvarez, P. A., & Linares, J. C. (2021). Assessing Tree Drought Resistance and Climate-Growth Relationships under Different Tree Age Classes in a Pinus nigra Arn. ssp. salzmannii Forest. Forests, 12(9), 1161. https://doi.org/10.3390/f12091161








