Abstract
Caffeine is a verified bioactive substance suitable for wood protection against pests. Unlike studies of the biocidal effects of caffeine, caffeine-wood bonds and interactions with wood polymer structures have not been studied whatsoever thus far. For this reason, caffeine (1 g/L) interactions with the main wood components (cellulose; hemicellulose; lignin and its precursors conipheryl alcohol, sinapyl alcohol, coumaryl alcohol) were analyzed in the present study. Caffeine concentrations were analyzed using UV–VIS spectrometry at wavelength 287 nm. The results confirmed caffeine variable binding with wood components in comparison to controls (pure caffeine). Cellulose and sinapyl alcohol did not interact with caffeine. Caffeine was bonded with the rest of the wood components in an increasing rank: conipheryl alcohol = lignin < hemicellulose < coumaryl alcohol. These results have a significant role in the protection of wood depending on its chemical composition and the wood species.
1. Introduction
Wood is one of the most important natural materials in the construction industry. From a chemical point of view, wood consists of lignin (L), cellulose (C), hemicelluloses (HC), and various additives and mineral substances. Cellulose and lignin are the two most widespread biopolymers in the world. Lignin is a polyphenolic amorphous substance with a high molecular weight. It appears in the largest amount in the secondary cell wall of plant cells. Its main function is to strengthen cellulose molecules within the cell walls [1]. The basic building blocks of lignin are phenylpropane derivatives (phenylpropanoids), which we refer to as lignin precursors: p-coumaryl alcohol (CuA), coniferyl alcohol (CoA) and sinapyl alcohol (SA). These hydroxycinnamic alcohols are linked to three-dimensional structures by ether bonds or bonds between two carbons [2]. Lignin is covalently bound to polysaccharides. They create intercellular fibers and strengthen cellulose molecules within the cell walls [3].
The cellulose structure is formed by unbranched chains of about 500 D-glucose units forming microfibrils determining the direction of growth of the plant cells. It is the main building block of plant primary cell walls and, together with lignin and hemicelluloses, it builds secondary cell walls [4]. Hemicelluloses are linear polysaccharides (various pentose and hexose) with short side chains and a smaller degree of crystalline content than cellulose. They fill the spaces between the cellulose and enable the binding of hydrophobic lignin [1].
The proportion of the individual components differs for coniferous and deciduous trees, as well as for individual tree species and specimens in relation to their age, position, etc. The chemical composition of wood and wood materials affects their ability to interact with water and other chemicals such as various pesticides, dyes, varnishes, and adhesives [5].
However, wood building materials and grown wood are often attacked by wood-destroying pests. Modern wood-preserving substances are supposed to be environmentally friendly and safe in terms of health. A suitable alternative to the previously used chemicals can be found in certain natural wood or plant extracts, oils, or individual substances with biocidal effects produced by plants for their own protection against pests [6,7,8,9,10].
Caffeine (C) (1,3,7-trimethyl-3,7-dihydro-1H-purin-2,6-dion) appears to be a suitable candidate for wood protection against biological attacks [11,12,13,14,15,16,17,18,19,20,21]. This methylxanthine (Figure 1) is produced by coffee tree or the plants such as legumes and certain types of vegetables. It was also tested as an active substance against termites [11]. Other authors [12,13,14,15,16] have analyzed he effectiveness of tea extracts or pure caffeine against fungi or molds in vitro studies. Pure caffeine was also applied to wood materials under various concentrations and treatment conditions [17,18,19]. Caffeine in various mixtures has already been tested [9,20,21].
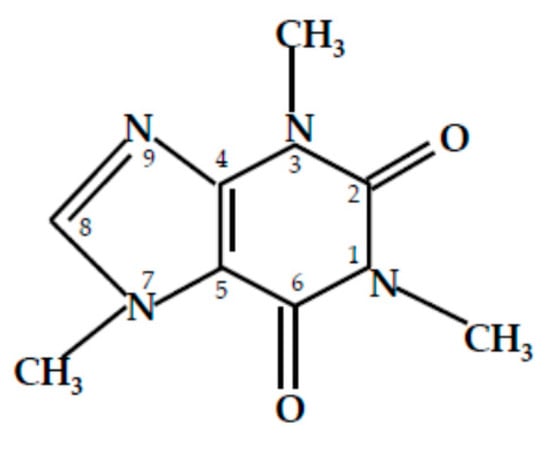
Figure 1.
Scheme of caffeine.
However, there are no studies about the chemical interactions of caffeine and individual primary wood structures, with the exception of caffeine as drug-coated cellulose wrapping [22]. For this reason, in this study, we separately analyzed caffeine interactions with basic wood components such as cellulose; hemicellulose; and lignin and its precursors conipheryl alcohol, sinapyl alcohol, and coumaryl alcohol separately as aquatic solutions in the present study.
2. Materials and Methods
2.1. Chemicals
Caffeine, conipheryl alcohol, sinapyl alcohol, and coumaryl alcohol were purchased from Sigma-Aldrich, Ltd. (Prague, Czech Republic). Cellulose and hemicellulose were purchased from P-Lab, Ltd. (Prague, Czech Republic). Microcrystalic cellulose was used (particle size = 0.02–0.1 mm). The hemicellulose was composed of galactomannan and xylan at a 1:1 ratio. Lignin with low sulfonate content was purchased from Sigma-Aldrich, Ltd. (Saint Louis, MO, USA). All of the substances were in powder the form (>99% purity). Deionized water was used as a solvent for the preparation of aquatic solutions or suspensions.
2.2. Experiment Procedure
Water stock solution of caffeine (1 g/L) was prepared for the recent experiments. The caffeine concentration was selected on basis of previous toxicity data with molds and fungi [14,15]. Glass bottles (30 mL volume) were fulfilled by 20 mL of caffeine solution with 20 mg of the appropriate biopolymer or alcohol in powder form was added. Pure caffeine solution (1 g/L) and pure component mixtures (1 g/L) were prepared as controls for analytical measurements. The caffeine and wood components were mixed before exposition in a thermostat. They were prepared as pure soluble solutions for alcohol precursors of lignin and lignin and hemicellulose. Suspension was used for cellulose. Their suspension was filtrated. Triplicates were prepared for each caffeine-polymer suspension or caffeine or alcohol solution and for all of the control samples. The bottles were stored in a thermostat at a temperature of 20 ± 2 °C in the dark. The samples were analyzed after 72 h of exposure.
2.3. Chemical Analyses
The suspension of cellulose (with or without caffeine) was filtered to obtain 5 mL of the filtrate. Determination of all solutions with or without caffeine (5 mL) was performed using UV–VIS spectrometry (spectrophotometer Biochrom Libra S22) at the optimal wavelength for the determination of caffeine (Figure 1). A 1 g/L caffeine concentration was used for this experiment [14,15]. A linear calibration curve was then generated with caffeine concentrations ranging from 1 to 10 mg/L as we only prepared 25 mL of caffeine solution (max. 25 mg of caffeine per test bottle), and we accepted any caffeine affinity to the individual wood components (see Figure 2 in the Results and Discussion chapter).
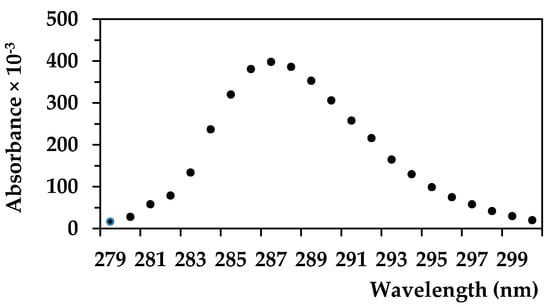
Figure 2.
Absorbances of caffeine at selected wavelengths (nm), n = 3.
2.4. Statistical Analyses
The absorbances in the presence/absence of caffeine in solutions of the individual wood components at the optimal caffeine wavelength were used to calculate caffeine concentrations in all of the samples. The caffeine absorbances in the mixtures (except for the caffeine absorbance in the pure caffeine solution) were calculated according to Formula (1):
where () is the absorbance of caffeine in the caffeine solution mixture with any wood component, xb is the absorbance of caffeine mixture with any pure wood component, and xa is the absorbance of the same pure wood component solution.
The absorbances of caffeine-component mixtures were then compared to the absorbance values of pure caffeine solution and the absorbance values were used for recalculation of the caffeine concentrations (mg/L) in Excel program. The caffeine concentrations were compared to the appropriate control (without polymers) using multivariate ANOVA, Dunnett Multiple Comparison Test considering p < 0.01 as significant (GrapPad InStat, version 3.06; GraphPad Software Inc., San Diego, CA, USA, 2003).
3. Results and Discussion
The key substance in this study is caffeine. Its behavior and properties in the aqueous environment have been well documented in the past, e.g., [23,24]. A calibration curve for caffeine dissolved in water has been constructed many times with different peaks [25,26,27] to select the most suitable wavelength for determining caffeine concentration in the range 270–290 nm.
Figure 2 and Figure 3 were produced in order to determine the caffeine concentration in our study. Based on the curve in the Figure 2, the wavelength of 287 nm was used as the optimal wavelength for measuring of caffeine absorbance. A calibration curve was then made for the selected and known caffeine –concentrations (Figure 3). The caffeine concentrations of up to 10 mg/L were analyzed. The caffeine concentrations were then calculated on the basis of absorbance data of mixtures of wood components with caffeine and by comparing the absorbance value of pure caffeine solution (Figure 4, Table 1).
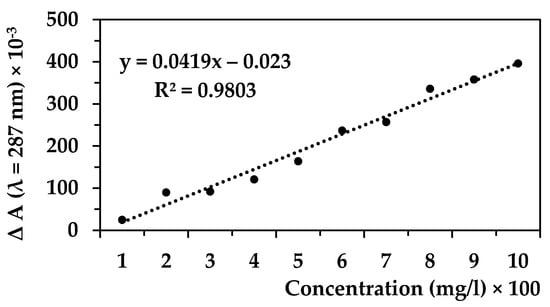
Figure 3.
Calibration curve of caffeine in the range 1–10 mg/L (wavelength 287 nm).
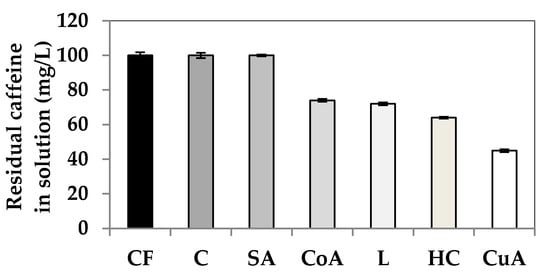
Figure 4.
Residual levels of pure solution of caffeine (CF) (1 g/L) or in the aquatic mixtures of caffeine (1 g of caffeine and 1 g of each wood component·L−1) with the selected components of wood: cellulose—C, sinapyl alcohol—SA, conipheryl alcohol—CoA, lignin—L, hemicellulose—HC, coumaryl alcohol—CuA. The results were statistically determined using ANOVA, Dunnett Multiple Comparison Test; see Tables S1–S3.

Table 1.
Absorbances of tested samples at λ = 287 nm: caffeine (CF), cellulose (C), cellulose + caffeine suspension (C + CF), lignin (L), lignin + caffeine suspension (L + CF), conipheryl alcohol (Co-A), conipheryl alcohol + caffeine solution (Co-A + CF), sinapyl alcohol (S-A), sinapyl alcohol + caffeine (S-A + CF), coumaryl alcohol (Cu-A), coumaryl alcohol + caffeine solution (Cu-A + CF). Primary data (n = 3), mean values, and their standard deviations (SD).
Each of the tested materials has its own optimal wavelength. The possibility of the same optimal wavelength for caffeine and other chemicals was verified (see Table S1). The results show that all of the studied wood components have an optimal wavelength different from the value of 287 nm. Cellulose is not soluble in water and for this reason, its optimal wavelength was not determined. The different optimal wavelengths of all of the studied chemicals enable the use of our preferred approach to calculate the caffeine concentrations in caffeine-component mixtures.
The results of the chemical analyses suggest (Table 1, Figure 4) that the ability of caffeine to interact with the tested wood components is different. There was no statistically significant difference in the residual caffeine concentration in the solution of cellulose and SA solution compared to the pure caffeine solution. This indicates no or low sorption of caffeine on these components. Cellulose is not soluble in water under normal conditions. Its solubility is possible in the other solvents, concentrated mineral acids or solutions of their salts or organic ammonium bases [28,29]. In some previous studies, caffeine leaching from treated wood samples was confirmed, so it is possible that this is due to the weak hydrogen bond between caffeine and cellulose, which makes up 50–58% of the weight of wood. The same results were found in a study by [30] where the authors did not confirm the binding of caffeine to cellulose using the FTIR method for pine wood.
Hemicelluloses are usually composed of various carbohydrate units-hexose and pentose. Composition of hemicellulose is variable among individual tree species [31]. Compared to cellulose, hemicelluloses are more soluble in water and better degrade into simple sugars. The smaller sugar units of hemicellulose or individual pentose or hexose can probably bond better to caffeine than to cellulose. The authors of one study described that the molecular dynamics simulations and the NMR experiments showed that sugars have an affinity for caffeine molecules [23]. The binding in caffeine–sugar complexes occurs by face-to-face stacking of the hydrophobic triad of protons of the pyranose rings against the caffeine face, rather than by hydrogen bonding [23]. Lignin fills the space between cellulose and hemicellulose, interconnects them, and ensures the strength and rigidity of the resulting structure in plant tissues [30]. In the case of phenolic lignin and its monomers, the ability to interact with caffeine differed significantly. The amount of residual caffeine in the solution decreased in the following order: SA, CoA = lignin, CuA. It is evident that the most effective interactions and bonds occur in the simplest precursor (CuA), which in its structure contains only hydroxyls with one carbonyl group of CoA to SA containing 2 carbonyl groups.
This is probably not a coincidence. Lignin is a mix of all these precursors in various ratios, but lignin is mostly represented by CoA (up to 95%), which has a sorption very similar to lignin (see Figure 3). For this reason, we can assume variable caffeine successful biocidal potential for different woods. The results of the paper [30] confirm our results on the binding interactions of caffeine with lignin. According to their study, caffeine may interact with the methylene group derived from the aromatic rings of the guaiacyl group of lignin according to their study. However, coumaryl alcohol without any guaiacyl group created the most intensive interactions with caffeine (Figure 3) in contrary to sinapyl alcohol with two guaiacyl groups in its structure in this study. One possible explanation is that conipheryl alcohol and sinapyl alcohol may create hydrogen bonds with caffeine due to the presence of -OCH3 groups in their molecules, but coumaryl alcohol cannot. Caffeine–coumaryl alcohol interactions are such probably based on the other kind of bonds or interactions. Generally, coumaryl alcohol has an elongation effect on the production of lignin chains [32]. Quantum mechanical calculations have determined the coumaryl alcohol monomer to be more reactive than the other two common lignin monomers, sinapyl and coniferyl alcohols [32]. The other study describes the possibility of coumaryl alcohol creating bonds with other organic compounds after its esterification [33]. Such an action mechanism likely occurs probably in the present.
Another interesting aspect may be the effect of the application of caffeine as a preservative against wood-destroying fungi. White rot is caused by fungi that break down cellulose and hemicellulose, as well as lignin [34]. Some species of white rot fungi preferably destroy only lignin [35], but some species degrade all components simultaneously [36]. The question is whether the high affinity of caffeine to lignin compared to HC and C could cause higher white-rot fungi sensitivity. It was demonstrated that caffeine differently affects fungi causing brown and white rot in wood. White-rot fungus species Trametes versicolor (Linnae ex Freis) was less sensitive to caffeine than the other three species representing fungi causing brown-rot [15]. In the experiment, the fungi were exposed to the same caffeine concentration on agar without the presence of cellulose or lignin (1 g/L). The results of [15] indicate a relatively high significance of caffeine–hemicellulose interactions for fungal sensitivity of brown-rot fungi, as they consume only cellulose and hemicellulose. Similar results were described in the study [17], in which the authors treated pine wood with caffeine in various concentrations. Species T. versicolor was again less sensitive then the other three model brown-rot fungi. We can assume that hemicelluloses are better digested and accessible to fungi than cellulose, and therefore, they break it down first. If properly correct, hemicellulose and its rapid degradation could be the key to the susceptibility of fungi to caffeine-containing fungicides. Caffeine bound to lignin is thus probably less available to fungi and may therefore be less sensitive to it [15]. However, there has yet to be more detailed research in this area. It is necessary to research various kinds of woods and fungi.
The biodegradation of wood by wood-destroying fungi will be influenced not only by the kind of rot or the sensitivity of some of the individual species, but also by the composition of the wood mass. This statement is based on the study b [19], where a different sorption of caffeine solution was found in 7 woody plants (beech, red oak, walnut, sapelli, English oak, pine, spruce) as well as different levels of biodegradability against wood-destroying fungi and molds.
4. Conclusions
The interactions of caffeine and basic wood components-cellulose, hemicellulose, lignin, and its 3 alcohol precursors (coumaryl alcohol, conipheryl alcohol, sinapyl alcohol) in aqueous solutions were investigated under defined conditions in the present study. In general, it can be concluded that the bond of caffeine to the selected wood components depends on their solubility in water and their chemical structure. No interactions of caffeine with cellulose were observed. The simpler molecules containing only hydroxyl groups (coumaryl alcohol) or hexose and pentose (hemicellulose) bonded better to caffeine molecules than to more complexed polymers (lignin, cellulose). We also observed the variable intensity of interaction among the alcohol precursors of lignin. Sinapyl alcohol was inert to caffeine, contrary to the high affinity of coumaryl alcohol. Conipheryl alcohol had interactions with caffeine very similar to the interactions that caffeine had with lignin. This may be just explained by the 95% presence of conipheryl alcohol in lignin. Coumaryl alcohol demonstrated the highest level of interactions with caffeine.
However, each tree species is composed of these components in different percentages, and other chemicals such as pectin, tannins, or dyes can also affect the binding interactions. Our research is therefore important for practical use, because after it was applied to various wood-based materials, caffeine had various effects on the same pasts, which has also been demonstrated in prior studies.
These results are the first steps to towards better understanding wood-caffeine interactions, but they can also lead to the other extensive research focused on developing new natural wood protective agents. Many natural substances have biocidal effects and understanding the mechanisms of binding these substances to wood would enable and accelerate the production and application of organic products in practice, both for the protection of logs and wood materials and possibly the protection of growing trees from pests and their premature felling.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/f12050533/s1. Table S1. The optimal wavelength (nm) of aquatic solutions (caffeine = C, hemicellulose = HC, lignine = L, conipheryl alcohol = CoA, sinapyl alcohol = SA, coumaryl alcohol—CuA) or cellulose suspension (C). Table S2. Data of Dunnett Multiple Comparisons Test (Anova)—comparison of the control (CF - caffeine solution) with other samples containing a mix of caffeine and any of the other components (HC—hemicellulose, L—lignin, CuA—coumaryl alcohol, CoA—conipheryl alcohol, C—cellulose, SA—sinapyl alcohol. Table S3. Data of Tukey-Kramer Multiple Comparisons Test (Anova)—comparison of main wood components lignin (L), cellulose (C), and hemicellulose (HC). Table S4. Data of Tukey-Kramer Multiple Comparisons Test (Anova)—comparison of lignin (L) with its the precursors (CuA—coumaryl alcohol, CoA—conipheryl alcohol, SA—sinapyl alcohol).
Author Contributions
Conceptualization, K.K.; methodology, K.K.; investigation, K.K. and K.Ď.; resources, K.K.; writing—original draft preparation, K.K. and K.Ď.; writing—review and editing, K.K. and J.N. and K.Ď.; supervision, K.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Czech Science Foundation, grant number 19-02067S.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data supporting reported results can be found at authors.
Acknowledgments
This research has been supported by the Czech Science Foundation under the Project No. 19-02067S.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Carpita, N.C.; Giheaut, D.M. Structural models of primary cell walls in flowering plants: Consistency of molecular structure with the physical properties of the wall during growth. Plant J. 1993, 3, 1–30. [Google Scholar] [CrossRef]
- Boerjan, W.; Ralph, J.; Bauche, M. Lignin Biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.G.; Pascal, F.; Capucine, D.; Denilson da Silva, P.; Xuan-mi, M. Assessingthe impact of woody and agricultural biomass variability on its behaviour in torrefaction through Principal Component Analysis. Biomass Bioenergy 2020, 134. [Google Scholar]
- Gibsen, L.J. The hierarchical structure and mechanics of plant materials. J. R. Soc. Interface 2012, 9, 2749–2766. [Google Scholar] [CrossRef] [PubMed]
- Pánek, M.; Reinprecht, L.; Hulla, M. Ten essential Oils for Beech Wood Protection–Efficacy Against Wood-destroying Fungi and Moulds, and Effect on Wood Discoloration. BioResources 2014, 9, 5588–5603. [Google Scholar] [CrossRef]
- Islam, M.S.; Hamdan, S.; Hasan, M.; Ahmed, A.S.; Rahman, M.R. Effect of coupling Reactions on the mechanical and biological properties of tropical wood polymers composites (WPC). Int. Biodeterior. Biodegrad. 2012, 72, 108–113. [Google Scholar] [CrossRef]
- Singh, T.; Singh, A.P. A review on natural products as wood protectant. Wood Sci. Technol. 2012, 46, 851–870. [Google Scholar] [CrossRef]
- Lee, S.H.; Ashaari, Z.; Lum, W.C.; Abdul Halip, J.; Ang, A.F.; Ta, L.P.; Chin, K.L.; Md Tahir, P. Thermal treatment of wood using vegetable oils: A review. Constr. Build. Mater. 2018, 181, 408–419. [Google Scholar] [CrossRef]
- Broda, M.; Mazela, B.; Frankowski, M. Durability of wood treated with aatmos and caffeine-towards the long-term carbon storage. Maderas-Cienc. Tecnol. 2018, 20, 455–468. [Google Scholar]
- Broda, M. Natural compounds for Wood protection against Fungi–A Review. Molecules 2020, 25, 3538. [Google Scholar] [CrossRef]
- Testolin, R.C.; Tischer, V.; Lima, A.O.; Cotelle, S.; Férard, J.F.; Radetski, C.M. Aquatic ecotoxicity assessment of a new natural formicide. Environ. Sci. Pollut. Res. Int. 2020, 19, 2186–2194. [Google Scholar] [CrossRef]
- Arora, D.S.; Ohlan, D. In vitro studies on antifungal activity of tea (Camellia sinensis) and coffee (Coffea arabica) against wood-rotting fungi. J. Basic Microbiol. 1997, 37, 159–165. [Google Scholar] [CrossRef]
- Zhang, H.; Qi, L.; Zhang, Z. Antifungal activity of caffeine against fungal pathogens of tea plant. J. Nanjing. Agric. Univ. 2020, 2, 63–67. [Google Scholar]
- Kobetičová, K.; Kočí, V.; Petříková, M.; Šimůnková, K.; Černý, R. Growth effectivity of molds in contact with methylxanthines. MATEC Web Conf. 2019, 282, 02058. [Google Scholar] [CrossRef]
- Kobetičová, K.; Nábělková, J.; Ďurišová, K.; Šimůnková, K.; Černý, R. Antifungal Activity of Methylxanthines in Relation to their Properties. Bioresources 2020, 15, 8110–8120. [Google Scholar]
- Barbero-López, A.; Monzó-Beltrán, J.; Virjamo, V.; Akkanen, J.; Haapala, A. Revalorization of coffee silver skin as a potential feedstock for antifungal chemicals in wood preservation. Int. Biodeter. Biodegr. 2020, 152, 105011. [Google Scholar] [CrossRef]
- Kwasniewska-Sip, P.; Cofta, G.; Nowak, P.B. Resistance of fungal growth on Scots pine treated with caffeine. Int. Biodeterior. Biodegrad. 2018, 132, 178–184. [Google Scholar] [CrossRef]
- Kwasniewska-Sip, P.; Bartkowiak, M.; Cofta, G.; Nowak, P.B. Resistance of Scots Pine (Pinus sylvestris L.) after Treatment with Caffeine and Thermal Modification against Aspergillus niger. Bioresources 2019, 14, 1890–1898. [Google Scholar]
- Kobetičová, K.; Böhm, M.; Černý, R. Mutual interactions of fungi and molds on woods treated with a caffeine solution: A preliminary study. AIP Conf. Proc. 2020, 2275, 020010. [Google Scholar]
- Ratajczak, I.; Wozniak, M.; Kwasniewska-Sip, P.; Szentner, K.; Cofta, G.; Mazela, B. Chemical characterization of wood treated with a formulation based on propolis, caffeine and organosilanes. Eur. J. Wood Wood Prod. 2018, 76, 775–781. [Google Scholar] [CrossRef]
- Pánek, M.; Šimůnková, K.; Novák, D.; Dvořák, O.; Schönfelder, O.; Šedivka, P.; Kobetičová, K. Caffeine and TiO2 Nanoparticles Treatment of Spruce and Beech Wood for Increasing Transparent Coating Resistance against UV-Radiation and Mould Attacks. Coatings 2020, 10, 1141. [Google Scholar] [CrossRef]
- Furtado, L.M.; Hilamatu, K.C.P.; Balaji, K.; Ando, R.A.; Petri, D.F.S. Miscibility and Sustained release of drug from cellulose acetate butyrate/caffeine films. J. Drug Deliv. Sci. Technol. 2020, 55, 101472. [Google Scholar] [CrossRef]
- Tavagnacco, L.; Engström, O.; Schnupf, U.; Saboungi, M.-L.; Himmel, M.; Widmalm, G.; Cesàro, A.; Brady, J.W. Caffeine and Sugars Interact in Aqueous Solutions: A Simulation and NMR Study. J. Phys. Chem. B 2020, 116, 11701–11711. [Google Scholar] [CrossRef] [PubMed]
- Oanca, G.; Nadejde, C.; Creanga, D. Caffeine-Solvent Interaction Studied by UV Spectrometry and Molecular Modelling. Rom. J. Biophys. 2014, 24, 11–23. [Google Scholar]
- Kaplan, E.; Holmes, J.H.; Sapeika, N. Caffeine content of tea and coffee. S. Afr. Med. J. 1974, 48, 510–511. [Google Scholar] [PubMed]
- Aurnaud, M.J. The pharmacology of caffeine. Prog. Drug 1987, 31, 273–313. [Google Scholar]
- Wanyika, H.N.; Gatebe, E.G.; Gitu, L.M.; Ngumba, E.K.; Maritim, C.W. Determination of caffeine content of tea and instant coffee brands found in the Kenyan market. Afr. J. Food Sci. 2020, 4, 353–358. [Google Scholar]
- Novotný, V. Chemie Dřeva a Pomocné Papírenské Přípravky pro 2.Ročník SPŠ Papírenských, 1st ed.; Státní Nakladatelství Technické Literatury: Prague, Czech Republic, 1974; p. 102. [Google Scholar]
- Hasík, P. Reologie Gelových a Solových Soustav Přírodních Polymerů a Jejich Modifikantů. Master’s Thesis, Tomas Bata Univerity in Zlín, Zlín, Czech Republic, 2007. [Google Scholar]
- Kwasniewska-Sip, P.; Wozniak, M.; Jankowski, W.; Ratajczak, I. Chemical changes of Wood Treated with Caffeine. Materials 2021, 14, 497. [Google Scholar] [CrossRef] [PubMed]
- Rowell, R.M. Handbook of wood Chemistry and Wood Composites; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Harman-Ware, A.E.; Happs, R.M.; Davison, B.H.; Mark, E.D. The effect of coumaryl alcohol incorporation on the structure and composition of lignin dehydrogenation polymers. Biotechnol. Biofuels 2017, 10, 281. [Google Scholar] [CrossRef]
- Schoch, G.; Goepfert, S.; Morant, M.; Hehn, A.; Meyer, D.; Ullmann, P.; Werck-Reichhart, D. CYP98A3 from Arabidopsis thaliana Is a 3′-Hydroxylase of Phenolic Esters, a Missing Link in the Phenylpropanoid Pathway. J. Biol. Chem. 2001, 276, 36566–36574. [Google Scholar] [CrossRef]
- Goodell, B.; Qian, Y.; Jellison, J. Fungal Decay of Wood: Soft Rot-Brown Rot-White Rot. ACS Symp. Ser. 2008, 982, 9–31. [Google Scholar]
- Schwarze, F.W.; Engels, J.; Claus, M. Fungal Strategies of Wood Decay in Trees, 1st ed.; Springer: Berlin/Heidelberg/Freiburg, Germany, 2000; p. 185. [Google Scholar]
- Martínez, A.T.; Speranza, M.; Ruiz-Dueňas, F.J.; Ferreira, P.; Camarero, S.; Guillén, F.; Martinéz, M.; Gutiérrez, A.; del Rio, J.D. Biodegradation of lignocellulosics: Microbial, chemical, and enzymatic aspects of the fungal attack of lignin. Int. Microbiol. 2005, 8, 195–204. [Google Scholar] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).