Interaction of Biochar Type and Rhizobia Inoculation Increases the Growth and Biological Nitrogen Fixation of Robinia pseudoacacia Seedlings
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Seed Sowing
2.2. Seedling Culture
2.3. Statistical Analyses
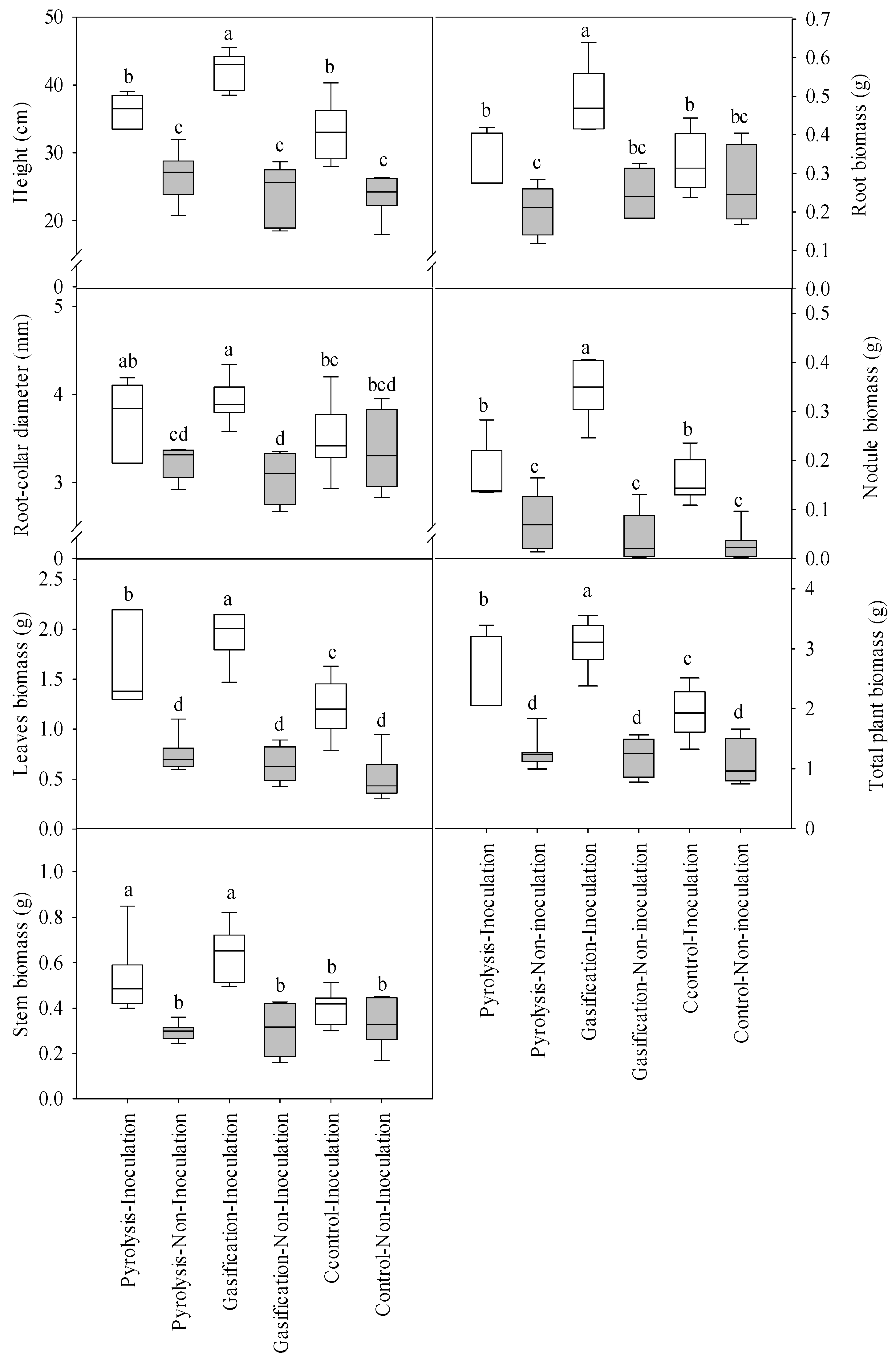
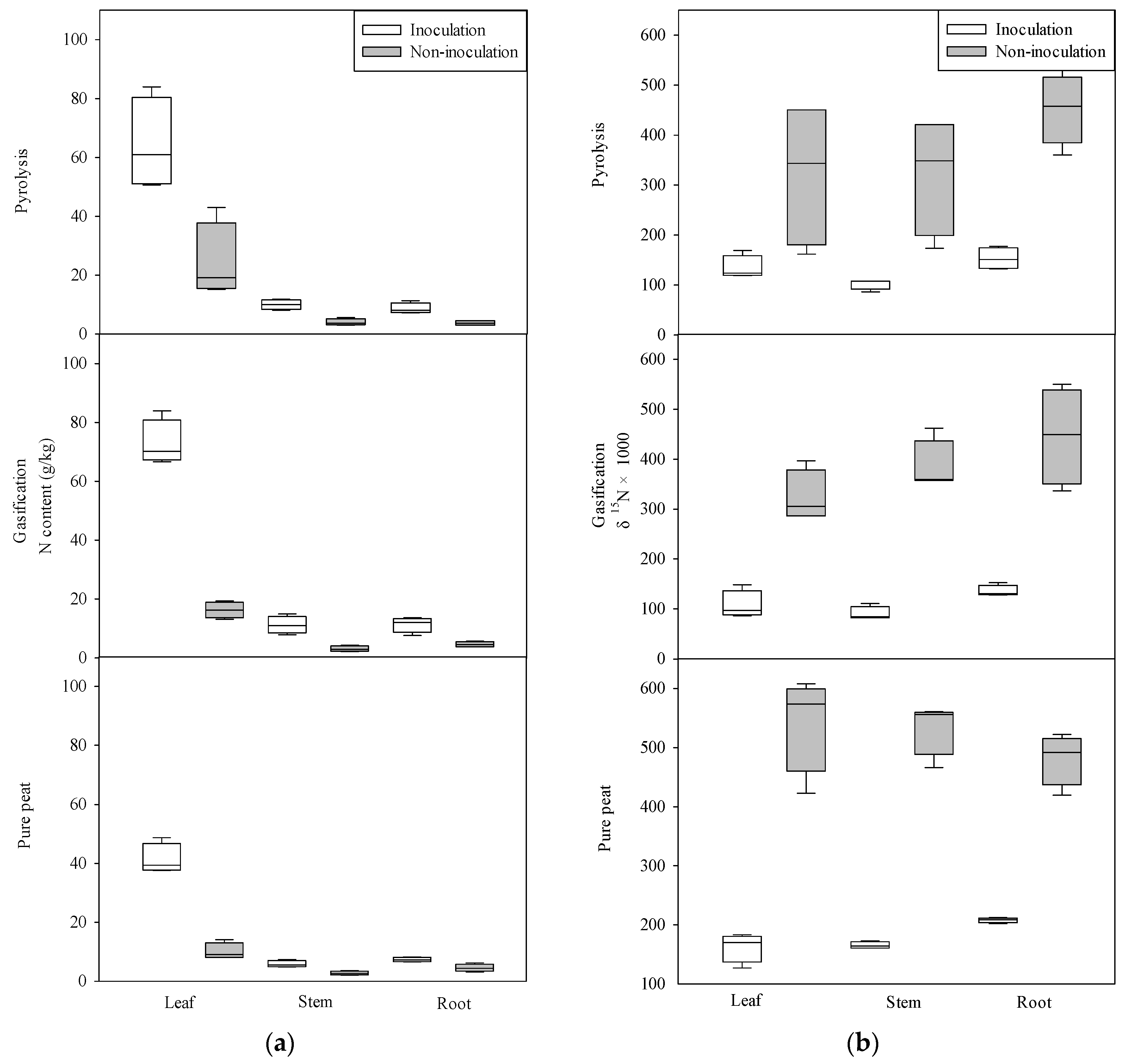
3. Results
3.1. Plant Growth
3.2. Nitrogen Absorption
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Griscom, B.W.; Adams, J.; Ellis, P.W.; Houghton, R.A.; Lomax, G.; Miteva, D.A.; Schlesinger, W.H.; Shoch, D.; Siikamäki, J.V.; Smith, P.; et al. Natural climate solutions. Proc. Natl. Acad. Sci. USA 2017, 114, 11645–11650. [Google Scholar] [CrossRef] [PubMed]
- Wolf, C.; Ripple, W.J.; Betts, M.G.; Levi, T.; Peres, C.A. Eating plants and planting forests for the climate. Glob. Chang. Biol. 2019, 25, 3995. [Google Scholar] [CrossRef] [PubMed]
- Stanturf, J.A.; Palik, B.; Dumroese, R.K. Contemporary forest restoration: A review emphasizing function. Forest. Ecol. Manag. 2014, 331, 292–323. [Google Scholar] [CrossRef]
- Bernal, B.; Murray, L.T.; Pearson, T.R.H. Global carbon dioxide removal rates from forest landscape restoration activities. Carbon Balance Manag. 2018, 13, 22. [Google Scholar] [CrossRef]
- Pawson, S.M.; Brin, A.; Brockerhoff, E.G.; Lamb, D.; Payn, T.W.; Paquette, A.; Parrotta, J.A. Plantation forests, climate change and biodiversity. Biodivers. Conserv. 2013, 22, 1203–1227. [Google Scholar] [CrossRef]
- Parrotta, J.A.; Wildburger, C.; Mansourian, S. Understanding Relationships between Biodiversity, Carbon, Forests and People: The Key to Achieving REDD+ Objectives; A Global Assessment Report. Prepared by the Global Forest Expert Panel on Biodiversity, Forest Management, and REDD+; International Union of Forest Research Organizations (IUFRO World Series): Vienna, Austria, 2012; Volume 31, 161p. [Google Scholar]
- Liang, J.J.; Crowther, T.W.; Picard, N.; Wiser, S.; Zhou, M.; Alberti, G.; Schulze, E.D.; McGuire, D.A.; Bozzato, F.; Pretzsch, H.; et al. Positive biodiversity-productivity relationship predominant in global forests. Science 2016, 354, 196. [Google Scholar] [CrossRef]
- Seidl, R.; Thom, D.; Kautz, M.; Martin-Benito, D.; Peltoniemi, M.; Vacchiano, G.; Wild, J.; Ascoli, D.; Petr, M.; Honkaniemi, J.; et al. Forest disturbances under climate change. Nat. Clim. Chang. 2017, 7, 395–402. [Google Scholar] [CrossRef]
- Sun, Q.Y.; Dumroese, R.K.; Liu, Y. Container volume and subirrigation schedule influence Quercus variabilis seedling growth and nutrient status in the nursery and field. Scand. J. Forest Res. 2018, 33, 560–567. [Google Scholar] [CrossRef]
- Haase, D.L.; Davis, A.S. Developing and supporting quality nursery facilities and staff are necessary to meet global forest and landscape restoration needs. Reforesta 2017, 4, 69–93. [Google Scholar] [CrossRef]
- Muller, J.J.; Nagel, L.M.; Palik, B.J. Forest adaptation strategies aimed at climate change: Assessing the performance of future climate-adapted tree species in a northern Minnesota pine ecosystem. Forest. Ecol. Manag. 2019, 451, 1–12. [Google Scholar] [CrossRef]
- Oliet, J.A.; Jacobs, D.F. Restoring forests: Advances in techniques and theory. New Forest. 2012, 43, 535–541. [Google Scholar] [CrossRef]
- Ferrari, A.E.; Wall, L.G. Nodulation and growth of black locust (Robinia pseudoacacia) on a desurfaced soil inoculated with a local Rhizobium isolate. Biol. Fert. Soils 2007, 43, 471–477. [Google Scholar] [CrossRef]
- Mengual, C.M.; Roldán, A.; Caravaca, F.; Schoebitz, M. Advantages of inoculation with immobilised rhizobacteria versus amendment with olive-mill waste in the afforestation of a semiarid area with Pinus halepensis Mill. Ecol. Eng. 2014, 73, 1–8. [Google Scholar] [CrossRef]
- Mengual, C.; Schoebitz, M.; Azcón, R.; Roldán, A. Microbial inoculants and organic amendment improves plant establishment and soil rehabilitation under semiarid conditions. J. Environ. Manag. 2014, 134, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.T.; Asghar, H.N.; Saleem, M.; Khan, M.Y.; Zahir, Z.A. Synergistic effect of rhizobia and biochar on growth and physiology of maize. Agron. J. 2015, 107, 2327–2334. [Google Scholar] [CrossRef]
- Herrera, M.A.; Salamanca, C.P.; Barea, J.M. Inoculation of woody legumes with selected arbuscular mycorrhizal fungi and rhizobia to recover desertified Mediterranean ecosystems. Appl. Environ. Microb. 1993, 59, 129–133. [Google Scholar] [CrossRef]
- Lal, B.; Khanna, S. Long term field study shows increased biomass production in tree legumes inoculated with Rhizobium. Plant Soil 1996, 184, 111–116. [Google Scholar] [CrossRef]
- Thrall, P.H.; Millsom, D.A.; Jeavons, A.C.; Waayers, M.; Harvey, G.R.; Bagnall, D.J.; Brockwell, J. Seed inoculation with effective root-nodule bacteria enhances revegetation success. J. Appl. Ecol. 2005, 42, 740–751. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Jacobs, D.F.; Davis, A.S. Inoculating Acacia koa with Bradyrhizobium and applying fertilizer in the nursery: Effects on nodule formation and seedling growth. HortScience 2009, 44, 443–446. [Google Scholar] [CrossRef]
- Zhang, P.; Dumroese, R.K.; Pinto, J.R. Organic or inorganic nitrogen and rhizobia inoculation provide synergistic growth response of a leguminous forb and tree. Front. Plant Sci. 2019, 10, 1308. [Google Scholar] [CrossRef]
- Diabate, M.; Munive, A.; de Faria, S.M.; Ba, A.; Dreyfus, B.; Galiana, A. Occurrence of nodulation in unexplored leguminous trees native to the West African tropical rainforest and inoculation response of native species useful in reforestation. New Phytol. 2005, 166, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, P.; Tan, T.K. Variations in the responses of Acacia mangium to inoculation with different strains of Bradyrhizobium sp. under nursery conditions. Symbiosis 2006, 41, 31–37. [Google Scholar]
- Dumroese, R.K.; Landis, T.D.; Pinto, J.R.; Haase, D.L.; Wilkinson, K.M.; Davis, A.S. Meeting forest restoration challenges: Using the Target Plant Concept. Reforesta 2016, 1, 37–52. [Google Scholar] [CrossRef]
- Dekak, A.; Chabi, R.; Menasria, T.; Benhizia, Y. Phenotypic characterization of rhizobia nodulating legumes Genista microcephala and Argyrolobium uniflorum growing under arid conditions. J. Adv. Res. 2018, 14, 35–42. [Google Scholar] [CrossRef]
- Karthikeyan, A. Effect of arbuscular mycorrhizal fungi and rhizobium on photosynthetic activity and growth response in Acacia auriculiflormis seedlings under elevated CO2. J. Trop. For. Sci. 2019, 31, 398–403. [Google Scholar] [CrossRef]
- Kirsten, S.O.; Richard, B.T. Effects of mycorrhizal colonization on biomass production and nitrogen fixation of black locust (Robinia pseudoacacia) seedlings grown under elevated atmospheric carbon dioxide. New Phytol. 1999, 142, 133–140. [Google Scholar] [CrossRef]
- Lehmann, J.; Gaunt, J.; Rondon, M. Bio-char sequestration in terrestrial ecosystems-a review. Mitig. Adapt. Strat. Glob. Chang. 2006, 11, 403–427. [Google Scholar] [CrossRef]
- Page-Dumroese, D.S.; Coleman, M.; Jones, G.; Venn, T.; Dumroese, R.K.; Anderson, N.; Chung, W.; Loeffler, D.; Archuleta, J.; Kimsey, M.; et al. Portable in-woods pyrolysis: Using forest biomass to reduce forest fuels, increase soil productivity, and sequester carbon. In Proceedings of the 2009 North American Biochar Conference, Boulder, CO, USA, 9–12 August 2009. [Google Scholar]
- Xiang, Y.Z.; Deng, Q.; Duan, H.L.; Guo, Y. Effects of biochar application on root traits: A meta-analysis. Glob. Chang. Biol. Bioenergy 2017, 9, 1563–1572. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Page-Dumroese, D.S.; Pinto, J.R. Biochar potential to enhance forest resilience, seedling quality, and nursery efficiency. Tree Plant. Notes 2020, 63, 61–69. [Google Scholar]
- Heiskanen, J.; Tammeorg, P.; Dumroese, R.K. Growth of Norway spruce seedlings after transplanting into silty soil amended with biochar: A bioassay in a growth chamber. J. For. Sci. 2013, 59, 125–129. [Google Scholar] [CrossRef]
- Lehmann, J.; Rillig, M.C.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowley, D. Biochar effects on soil biota-a review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar] [CrossRef]
- Ishii, T.; Kadoya, K. Effects of charcoal as a soil conditioner on citrus growth and vesicular-arbuscular mycorrhizal development. J. Jpn. Soc. Hortic. Sci. 1994, 63, 529–535. [Google Scholar] [CrossRef]
- Rondon, M.A.; Lehmann, J.; Ramírez, J.; Hurtado, M. Biological nitrogen fixation by common beans (Phaseolus vulgaris L.) increases with bio-char additions. Biol. Fert. Soils 2007, 43, 699–708. [Google Scholar] [CrossRef]
- Güereña, D.T.; Lehmann, J.; Thies, J.E.; Enders, A.; Karanja, N.; Neufeldt, H. Partitioning the contributions of biochar properties to enhanced biological nitrogen fixation in common bean (Phaseolus vulgaris L.). Biol. Fert. Soils 2015, 51, 479–491. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Rose, T.; Herridge, D.; Kimber, S.; Rust, J.; Cowie, A.; Morris, S. Enhanced biological N2 fixation and yield of faba bean (Vicia faba L.) in an acid soil following biochar addition: Dissection of causal mechanisms. Plant Soil 2015, 395, 7–20. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Heiskanen, J.; Englund, K.; Tervahauta, A. Pelleted biochar: Chemical and physical properties show potential use as a substrate in container nurseries. Biomass Bioenergy 2011, 35, 2018–2027. [Google Scholar] [CrossRef]
- Steiner, C.; Harttung, T. Biochar as a growing media additive and peat substitute. Solid Earth 2014, 5, 995–999. [Google Scholar] [CrossRef]
- Conversa, G.; Bonasia, A.; Lazzizera, C.; Elia, A. Influence of biochar, mycorrhizal inoculation, and fertilizer rate on growth and flowering of Pelargonium (Pelargonium zonale L.) plants. Front. Plant Sci. 2015, 6, 429. [Google Scholar] [CrossRef]
- Méndez, A.; Paz-Ferreiro, J.; Gil, E.; Gascó, G. The effect of paper sludge and biochar addition on brown peat and coir based growing media properties. Sci. Hortic-Amsterdam. 2015, 193, 225–230. [Google Scholar] [CrossRef]
- Di Lonardo, S.; Baronti, S.; Vaccari, F.P.; Albanese, L.; Battista, P.; Miglietta, F.; Bacci, L. Biochar-based nursery substrates: The effect of peat substitution on reduced salinity. Urban For. Urban Gree. 2017, 23, 27–34. [Google Scholar] [CrossRef]
- Matt, C.P.; Keyes, C.R.; Dumroese, R.K. Biochar effects on the nursery propagation of 4 northern rocky mountain native plant species. Native Plants J. 2018, 19, 14–26. [Google Scholar] [CrossRef]
- Anderson, N.; Jones, J.G.; Page-Dumroese, D.; McCollum, D.; Baker, S.; Loeffler, D.; Chung, W. A comparison of producer gas, biochar, and activated carbon from two distributed scale thermochemical conversion systems used to process forest biomass. Energies 2013, 6, 164–183. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Kumutha, K.; Krishnan, P.S. Influence of biochar and Azospirillum application on the growth of maize. Madras. Agric. J. 2011, 98, 158–164. [Google Scholar]
- Hansen, V.; Müller-Stöver, D.; Munkholm, L.J.; Peltre, C.; Petersen, C.; Hauggaard-Nielsen, H.; Jensen, L.S. The effect of straw and wood gasification biochar on carbon sequestration, selected soil fertility indicators and functional groups in soil: An incubation study. Geoderma 2016, 269, 99–107. [Google Scholar] [CrossRef]
- Hansen, V.; Hauggaard-Nielsen, H.; Petersen, C.T.; Mikkelsen, T.N.; Müller-Stöver, D. Effects of gasification biochar on plant-available water capacity and plant growth in two contrasting soil types. Soil Till. Res. 2016, 161, 1–9. [Google Scholar] [CrossRef]
- Cho, M.S.; Meng, L.; Song, J.H.; Han, S.H.; Bae, K.; Park, B.B. The effects of biochars on the growth of Zelkova serrata seedlings in a containerized seedling production system. For. Sci. Tec. 2017, 13, 25–30. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Pinto, J.R.; Heiskanen, J.; Tervahauta, A.; McBurney, K.G.; Page-Dumroese, D.S.; Englund, K. Biochar can be a suitable replacement for sphagnum peat in nursery production of Pinus ponderosa seedlings. Forests 2018, 9, 232. [Google Scholar] [CrossRef]
- Sarauer, J.L.; Coleman, M.D. Biochar as a growing media component for containerized production of Douglas-fir. Can. J. For. Res. 2018, 48, 581–588. [Google Scholar] [CrossRef]
- Ge, X.G.; Yang, Z.Y.; Zhou, B.Z.; Cao, Y.H.; Xiao, W.F.; Wang, X.M.; Li, M.H. Biochar fertilization significantly increases nutrient levels in plants and soil but has no effect on biomass of Pinus massoniana (Lamb.) and Cunninghamia lanceolata (Lamb.) Hook. saplings during the first growing season. Forests 2019, 10, 612. [Google Scholar] [CrossRef]
- Yusif, S.A.; Muhammad, I.; Hayatu, N.G.; Sauwa, M.M.; Tafinta, I.Y.; Mohammed, M.A.; Lukman, S.A.; Abubakar, G.A.; Hussain, A.M. Effects of biochar and rhizobium inoculation on nodulation and growth of groundnut in Sokoto State, Nigeria. J. Appl. Life Sci. Int. 2016, 9, 1–9. [Google Scholar] [CrossRef]
- Olson, D.F., Jr.; Karrfalt, R.P. Robinia L. Black locust. In The Woody Plant Seed Manual; Agriculture Handbook 727; Bonner, F., Karrfalt, R.P., Eds.; USDA Forest Service: Washington, DC, USA, 2008. [Google Scholar]
- Keresztesi, B. The Black Locust. Unasylva 1980, 32, 23–33. [Google Scholar]
- Roberts, D.R.; Zimmerman, R.W.; Stringer, J.W.; Carpenter, S.B. The effects of combined nitrogen on growth, nodulation, and nitrogen fixation of black locust seedlings. Can. J. For. Res. 1983, 13, 1251–1254. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Kenar, J.A.; Thompson, A.R.; Peterson, S.C. Comparison of biochars derived from wood pellets and pelletized wheat straw as replacements for peat in potting substrates. Ind. Crop. Prod. 2013, 51, 437–443. [Google Scholar] [CrossRef]
- Dumroese, R.K.; Montville, M.E.; Pinto, J.R. Using container weights to determine irrigation needs: A simple method. Nativ. Plants J. 2015, 16, 67–71. [Google Scholar] [CrossRef]
- Hardarson, G.; Danso, S.K.A. Methods for measuring biological nitrogen fixation in grain legumes. Plant Soil 1993, 152, 19–23. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Wirth, S.; Behrendt, U.; Abd_Allah, E.F.; Berg, G. Biochar treatment resulted in a combined effect on soybean growth promotion and a shift in plant growth promoting rhizobacteria. Front. Microbiol. 2016, 7, 209. [Google Scholar] [CrossRef]
- Nelson, N.O.; Agudelo, S.C.; Yuan, W.Q.; Gan, J. Nitrogen and phosphorus availability in biochar-amended soils. Soil Sci. 2011, 176, 218–226. [Google Scholar] [CrossRef]
- Brewer, C.E. Biochar Characterization and Engineering. Graduate Thesis and Dissertations, Iowa State University, Ames, IA, USA, 2012. [Google Scholar]
- Zwart, D.C.; Kim, S.H. Biochar amendment increases resistance to stem lesions caused by Phytophthora spp. in tree seedlings. HortScience 2012, 47, 1736–1740. [Google Scholar] [CrossRef]
- Mehboob, I.; Zahir, Z.A.; Arshad, M.; Tanveer, A.; Farooq-e-Azam. Growth promoting activities of different Rhizobium spp. in wheat. Pak. J. Bot. 2011, 43, 1643–1650. [Google Scholar]
- Boddey, R.M.; Urquiaga, S.; Neves, M.C.P.; Suhet, A.R.; Peres, J.R. Quantification of the contribution of N2 fixation to field-grown grain legumes-a strategy for the practical application of the 15N isotope dilution technique. Soil Biol. Biochem. 1990, 22, 649–655. [Google Scholar] [CrossRef]
- Billah, M.M.; Ahmad, W.; Ali, M. Biochar particle size and rhizobia strains effect on the uptake and efficiency of nitrogen in lentils. J. Plant Nutri. 2019, 42, 1709–1725. [Google Scholar] [CrossRef]
- Iijima, M.; Yamane, K.; Izumi, Y.; Daimon, H.; Motonaga, T. Continuous application of biochar inoculated with root nodule bacteria to subsoil enhances yield of soybean by the nodulation control using crack fertilization technique. Plant Prod. Sci. 2015, 18, 197–208. [Google Scholar] [CrossRef]
- Budi, S.W.; Setyaningsih, L. Arbuscular mycorrhizal fungi and biochar improved early growth of neem (Melia azedarach Linn.) seedling under greenhouse conditions. J. Manajemen Hutan Tropika 2013, 19, 103–110. [Google Scholar] [CrossRef][Green Version]
- Yang, X.; Tsibart, A.; Nam, H.; Hure, J.; El-Naggar, A.; Tackc, F.M.G.; Wang, C.H.; Lee, Y.H.; Tsang, D.C.W.; Ok, Y.S. Effect of gasification biochar application on soil quality: Trace metal behavior, microbial community, and soil dissolved organic matter. J. Hazard. Mater. 2019, 365, 684–694. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, A.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agr. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
| Biochar Type | pH | Density (g/mL) | Pore Volume (cm3/g) | C (%) | N (%) |
|---|---|---|---|---|---|
| Pyrolysis | 7.1 | 0.53 | 0.09 | 76.4 | 0.56 |
| Gasification | 10.2 | 0.17 | 0.016 | 91.5 | 0.89 |
| Independent Variables | Height (cm) | Root-Collar Diameter (mm) | Biomass (g) | ||||
|---|---|---|---|---|---|---|---|
| Leaves | Stem | Root | Nodule | Total | |||
| Biochar type (B) | 0.001 | 0.933 | <0.0001 | 0.043 | 0.001 | <0.0001 | <0.0001 |
| Inoculation (I) | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| B × I | 0.001 | 0.038 | 0.011 | 0.004 | 0.004 | <0.0001 | 0.001 |
| Independent Variables | Nitrogen Content (g/kg) | δ15N × 1000 | ||||
|---|---|---|---|---|---|---|
| Leaves | Stem | Root | Leaves | Stem | Root | |
| Biochar types (B) | 0.001 | 0.003 | 0.030 | 0.003 | <0.0001 | 0.004 z |
| Inoculation (I) | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| B × I | 0.041 | 0.018 | 0.058 | 0.040 | 0.057 | 0.064 z |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Q.; Liu, Y.; Liu, H.; Dumroese, R.K. Interaction of Biochar Type and Rhizobia Inoculation Increases the Growth and Biological Nitrogen Fixation of Robinia pseudoacacia Seedlings. Forests 2020, 11, 711. https://doi.org/10.3390/f11060711
Sun Q, Liu Y, Liu H, Dumroese RK. Interaction of Biochar Type and Rhizobia Inoculation Increases the Growth and Biological Nitrogen Fixation of Robinia pseudoacacia Seedlings. Forests. 2020; 11(6):711. https://doi.org/10.3390/f11060711
Chicago/Turabian StyleSun, Qiaoyu, Yong Liu, Hongbin Liu, and R. Kasten Dumroese. 2020. "Interaction of Biochar Type and Rhizobia Inoculation Increases the Growth and Biological Nitrogen Fixation of Robinia pseudoacacia Seedlings" Forests 11, no. 6: 711. https://doi.org/10.3390/f11060711
APA StyleSun, Q., Liu, Y., Liu, H., & Dumroese, R. K. (2020). Interaction of Biochar Type and Rhizobia Inoculation Increases the Growth and Biological Nitrogen Fixation of Robinia pseudoacacia Seedlings. Forests, 11(6), 711. https://doi.org/10.3390/f11060711





