Responses and Differences in Tolerance to Water Shortage under Climatic Dryness Conditions in Seedlings from Quercus spp. and Andalusian Q. ilex Populations
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Plant Material, Growth Conditions, and Drought Treatment
2.3. Perlite Water Content and Matric Potential
2.4. Damage Symptoms and Seedling Mortality
2.5. Leaf Water Status
2.6. Quantum Yield of Photosystem II, Net Photosynthesis Rate, and Stomatal Conductance
2.7. Photosynthetic Pigment and Anthocyanin Analyses
2.8. Total Sugar, Phenolic, and Amino Acid Analysis
2.9. Statistical Analyses
3. Results
3.1. Perlite Water Content and Matric Potential
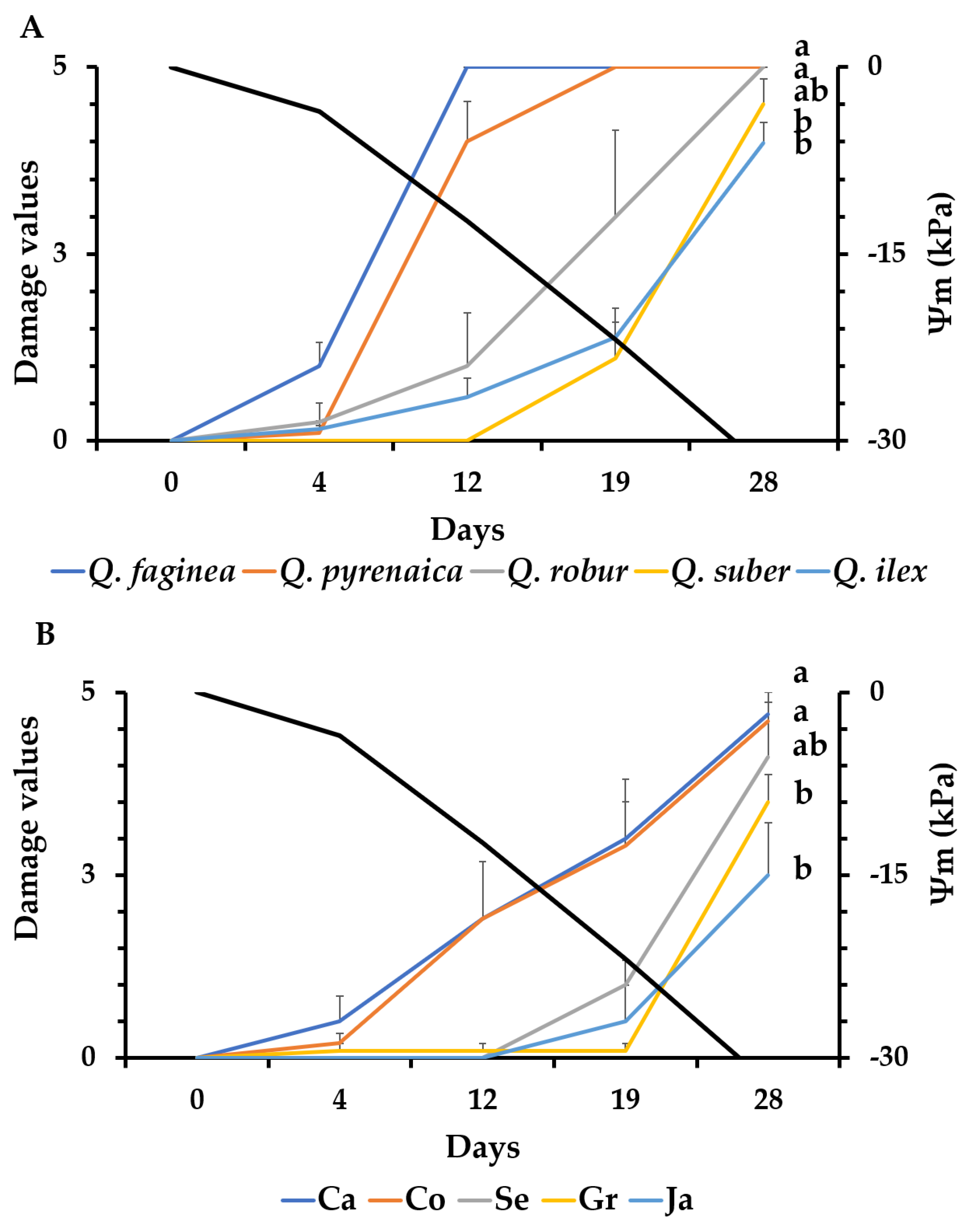
3.2. Evaluation of Damage Symptoms and Seedling Mortality
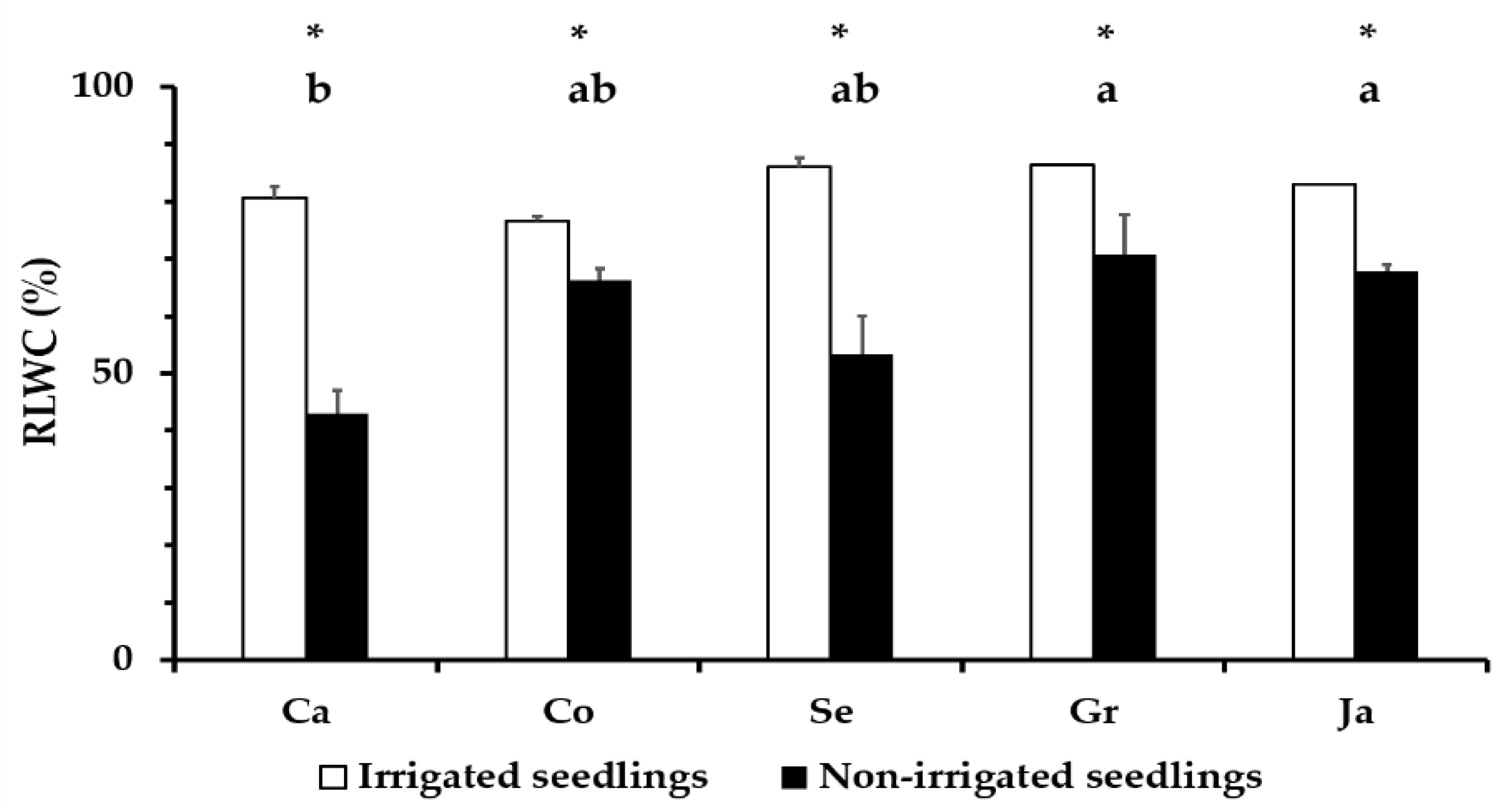
3.3. Relative Water Content in Q. ilex Leaves
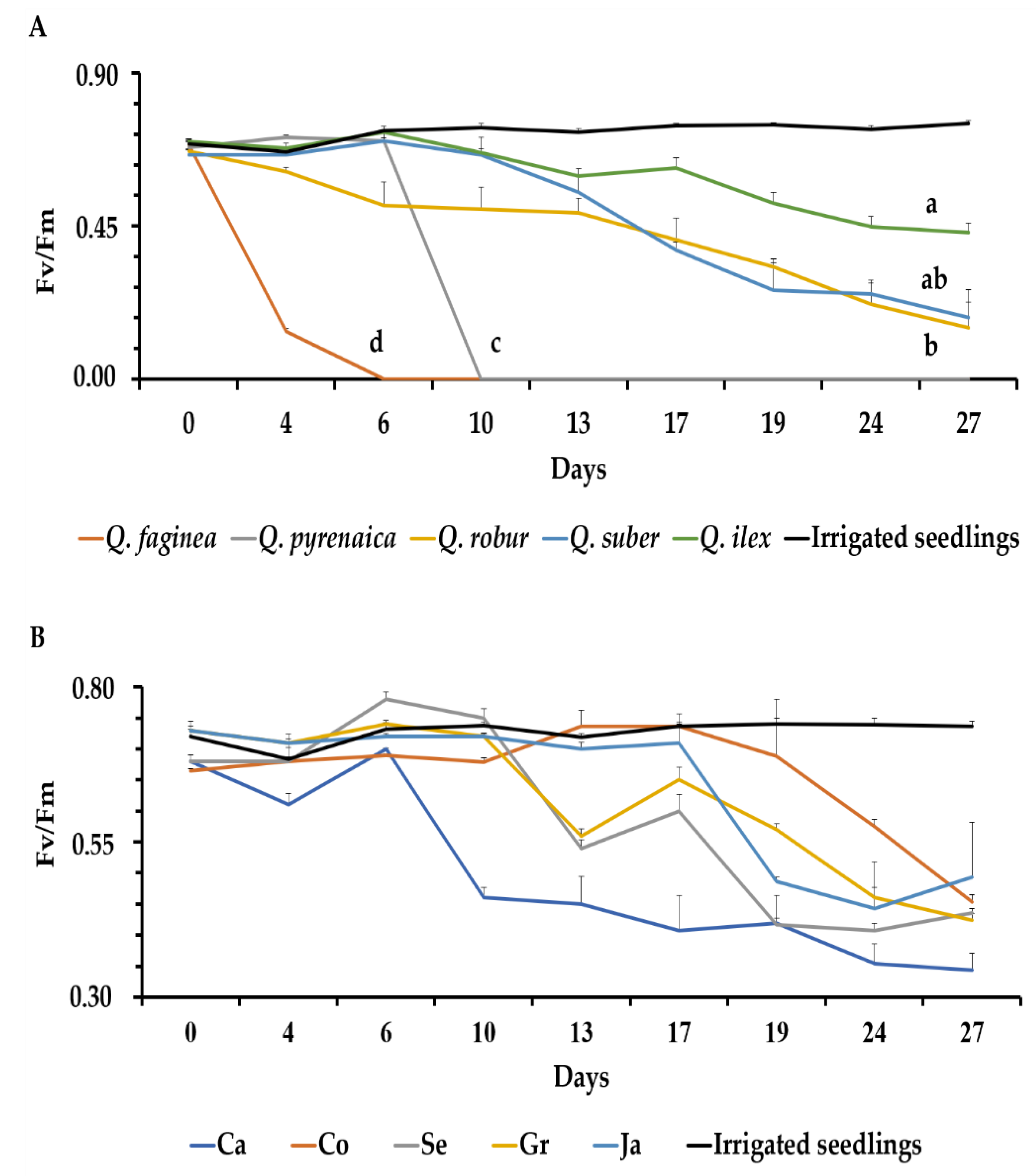
3.4. Quantum Yield of Photosystem II (Fv/Fm)
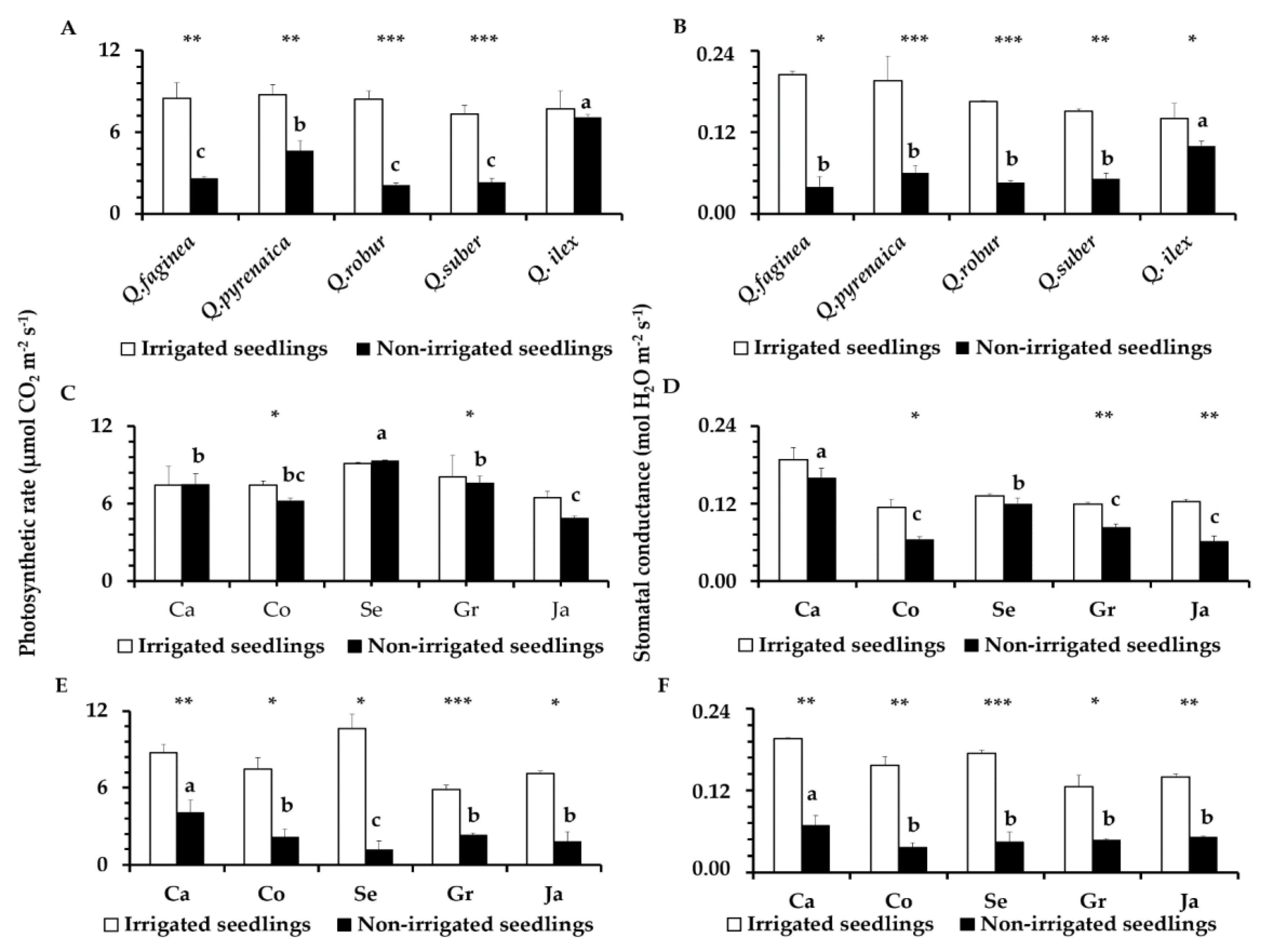
3.5. Leaf Photosynthesis Parameters
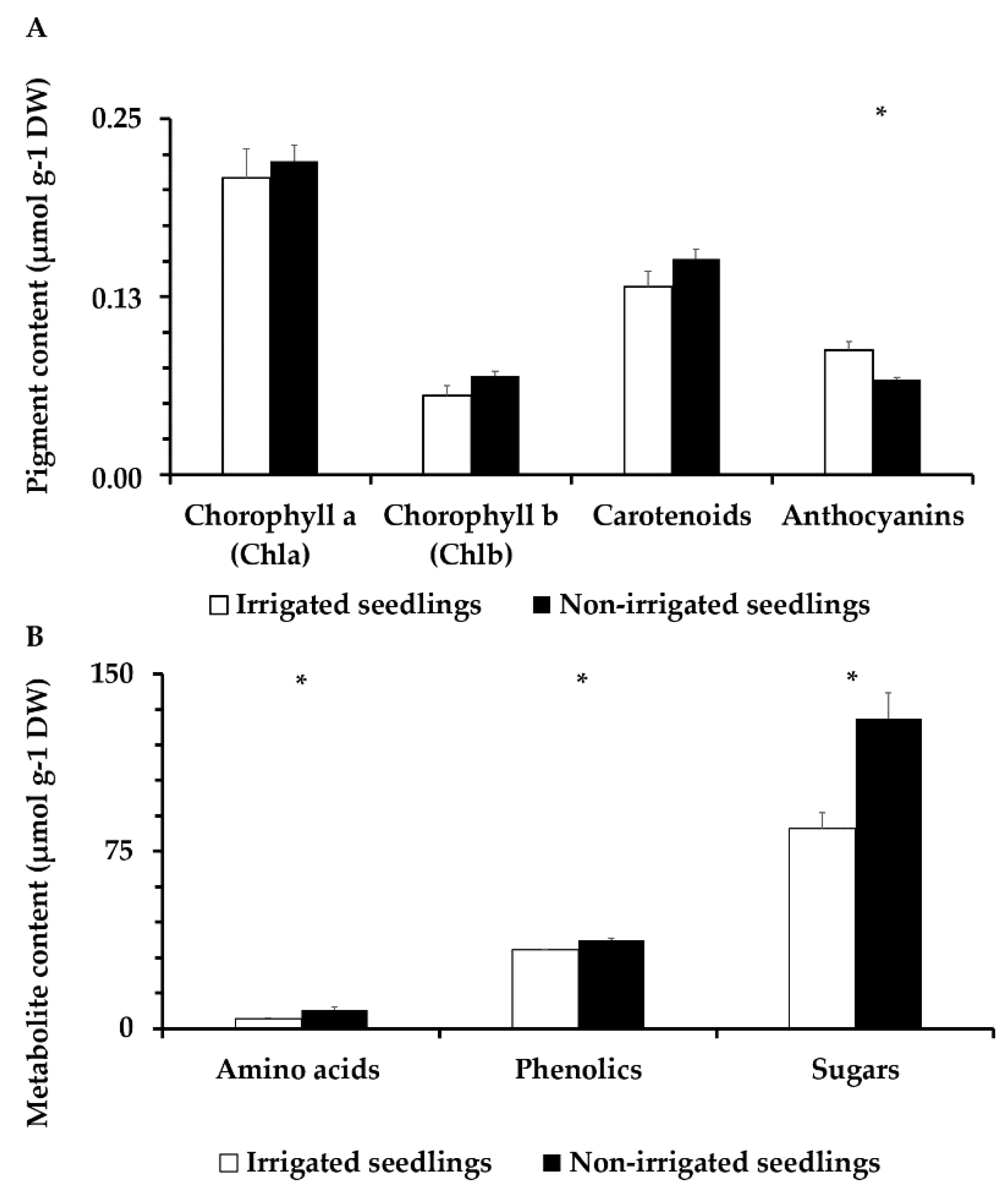
3.6. Pigment Content and Anthocyanins Analysis
3.7. Amino Acid, Total Phenolic Compound, and Sugar Content Analysis
4. Discussion
4.1. Drought Visual Symptoms and Mortality Rate as Indicators of Drought Tolerance
4.2. Leaf Water Status in Droughted Q. ilex Seedlings as a Physiological Parameter of Tolerance Differences
4.3. Photosynthesis Parameters in Droughted Quercus Seedlings and Tolerance Differences
4.4. Leaf Chemical Composition in Se Q. ilex Seedlings
4.5. Management Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Franco, A.J. Quercus L. In Flora Ibériabout; Castroviejo, S., Laínz, M., González, G.L., Montserrat, P., Muñoz-Garmendia, F., Paiva, J., Villar, L., Eds.; Real Jardín Botánico CSIC: Madrid, Spain, 1990; Volume 2, pp. 15–36. [Google Scholar]
- Nixon, K.C. Global and neotropical distribution and diversity of oak (genus Quercus) and oak forests. In Ecology and Conservation of Neotropical Montane Oak Forests; Springer: Heidelberg/Berlin, Germany, 2006; pp. 3–13. [Google Scholar]
- Moreno, G.; Pulido, F.J. The functioning, management and persistence of dehesas. In Agroforestry in Europe 6; Springer: Dordrecht, The Netherlands, 2009; pp. 127–160. [Google Scholar]
- Natalini, F.; Alejano, R.; Vázquez-Piqué, J.; Cañellas, I.; Gea-Izquierdo, G. The role of climate change in the widespread mortality of holm oak in open woodlands of Southwestern Spain. Dendrochronologia 2016, 38, 51–60. [Google Scholar] [CrossRef]
- Plieninger, T. Constructed and degraded? Origin and development of the Spanish dehesa landscape, with a case study on two municipalities. Erde 2007, 138, 25–46. [Google Scholar]
- Surová, D.; Ravera, F.; Guiomar, N.; Martínez Sastre, R.; Pinto-Correia, T. Contributions of iberiansilvo-pastoral landscapes to the well-being of contemporary society. Rangel. Ecol. Manag. 2018, 71, 560–570. [Google Scholar]
- Ruiz-Gómez, F.J.; Pérez-de-Luque, A.; Navarro-Cerrillo, R.M. The involvement of Phytophthora root rot and drought stress in Holm oak decline: From ecophysiology to microbiome influence. Curr. For. Rep. 2019, 5, 251–256. [Google Scholar] [CrossRef]
- Echevarría-Zomeño, S.; Ariza, D.; Jorge, I.; Lenz, C.; Del Campo, A.; Jorrín-Novo, J.V.; Navarro, R.M. Changes in the protein profile of Quercus ilex leaves in response to drought stress and recovery. J. Plant Physiol. 2009, 166, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Valero-Galván, J.; González-Fernández, R.; Navarro-Cerrillo, R.M.; Gil-Pelegrín, E.; Jorrín-Novo, J.V. Physiological and proteomic analyses of drought stress response in Holm oak provenances. J. Proteome Res. 2013, 12, 5110–5123. [Google Scholar] [CrossRef]
- Gil-Pelegrín, E.; Peguero-Pina, J.J.; Sancho-Knapik, D. Oaks and people: a long journey together. In Oaks Physiological Ecology; Exploring the Functional Diversity of Genus Quercus L.; Springer: Cham, Germany, 2017; pp. 1–11. [Google Scholar]
- Leiva, M.J.; Fernández-Alés, R. Variability in seedling water status during drought within a Quercus ilex subsp. ballota population, and its relation to seedling morphology. For. Ecol. Manag. 1998, 111, 147–156. [Google Scholar] [CrossRef]
- Villar-Salvador, P.; Planelles, R.; Oliet, J.; Peñuelas-Rubira, J.L.; Jacobs, D.F.; González, M. Drought tolerance and transplanting performance of holm oak (Quercus ilex) seedlings after drought hardening in the nursery. Tree Physiol. 2004, 24, 1147–1155. [Google Scholar] [CrossRef]
- Navarro-Cerrillo, R.M.; Fragueiro, B.; Ceaceros, C.; del Campo, A.; de Prado, R. Establishment of Quercus ilex L. subsp. ballota [Desf.] Samp. using different weed control strategies in southern Spain. Ecol. Eng. 2005, 25, 332–342. [Google Scholar] [CrossRef]
- Forner, A.; Valladares, F.; Aranda, I. Mediterranean trees coping with severe drought: Avoidance might not be safe. Environ. Exp. Bot. 2018, 155, 529–540. [Google Scholar] [CrossRef]
- Lloret, F.; Siscart, D.; Dalmases, C. Canopy recovery after drought dieback in holm-oak Mediterranean forests of Catalonia (NE Spain). Glob. Chang. Biol. 2004, 10, 2092–2099. [Google Scholar] [CrossRef]
- Giorgi, F.; Lionello, P. Climate change projections for the Mediterranean region. Glob. Planet. Chang. 2008, 63, 90–104. [Google Scholar] [CrossRef]
- Collins, W.J.; Fry, M.M.; Yu, H.; Fuglestvedt, J.S.; Shindell, D.T.; West, J.J. Global and regional temperature-change potentials for near-term climate forcers. Atmos. Chem. Phys. 2013, 13, 2471–2485. [Google Scholar] [CrossRef]
- Peng, C.; Ma, Z.; Lei, X.; Zhu, Q.; Chen, H.; Wang, W.; Liu, S.; Weizhong, L.; Fang, X.; Zhou, X. A drought-induced pervasive increase in tree mortality across Canada’s boreal forests. Nat. Clim. Chang. 2012, 1, 467–471. [Google Scholar] [CrossRef]
- David, T.S.; Henriques, M.O.; Kurz-Besson, C.; Nunes, J.; Valente, F.; Vaz, M.; Pereira, J.S.; Siegwolf, R.; Chaves, M.M.; Gazarini, L.C.; et al. Water-use strategies in two co-occurring Mediterranean evergreen oaks: Surviving the summer drought. Tree Physiol. 2007, 27, 793–803. [Google Scholar] [CrossRef]
- Früchtenicht, E.; Neumann, L.; Klein, N.; Bonal, D.; Brüggemann, W. Response of Quercus robur and two potential climate change winners—Quercus pubescens and Quercus ilex—To two years summer drought in a semi-controlled competition study: I—Tree water status. Environ. Exp. Bot. 2018, 152, 107–117. [Google Scholar] [CrossRef]
- Hamerlynck, E.P.; Knapp, A.K. Early season cuticular conductance and gas exchange in two oaks near the western edge of their range. Trees 1996, 10, 403–409. [Google Scholar] [CrossRef]
- Vilagrosa, A.; Chirino, E.; Peguero-Pina, J.J.; Barigah, T.S.; Cochard, H.E.; Gil-Pelegrin, E. Xylem cavitation and embolism in plants living in water-limited ecosystems. In Plant Responses to Drought Stress; Springer: Berlin/Heidelberg, Germany, 2012; pp. 63–109. [Google Scholar]
- Vander Mijnsbrugge, K.; Bischoff, A.; Smith, B. A question of origin: Where and how to collect seed for ecological restoration. Basic Appl. Ecol. 2010, 11, 300–311. [Google Scholar] [CrossRef]
- North, M.P.; Stevens, J.T.; Greene, D.F.; Coppoletta, M.; Knapp, E.E.; Latimer, A.M.; Restaino, C.M.; Tompkins, R.E.; Welch, K.R.; York, R.A.; et al. Tamm Review: Reforestation for resilience in dry western US forests. For. Ecol. Manag. 2019, 432, 209–224. [Google Scholar] [CrossRef]
- Quero, J.L.; Villar, R.; Marañón, T.; Zamora, R.; Poorter, L. Seed-mass effects in four Mediterranean Quercus species (Fagaceae) growing in contrasting light environments. Am. J. Bot. 2007, 94, 1795–1803. [Google Scholar] [CrossRef]
- Villar-Salvador, P.; Heredia, N.; Millard, P. Remobilization of acorn nitrogen for seedling growth in holm oak (Quercus ilex), cultivated with contrasting nutrient availability. Tree Physiol. 2010, 30, 257–263. [Google Scholar] [CrossRef] [PubMed]
- García-Cebrián, F.; Esteso-Martínez, J.; Gil-Pelegrín, E. Influence of cotyledon removal on early seedling growth in Quercus robur L. Ann. For. Sci. 2003, 60, 69–73. [Google Scholar] [CrossRef]
- Arend, M.; Kuster, T.; Günthardt-Goerg, M.S.; Dobbertin, M. Provenance-specific growth responses to drought and air warming in three European oak species (Quercus robur, Q. petraea and Q. pubescens). Tree Physiol. 2011, 31, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Cerrillo, R.M.; Gómez, F.J.; Cabrera-Puerto, R.J.; Sánchez-Cuesta, R.; Rodriguez, G.P.; Pérez, J.L. Growth and physiological sapling responses of eleven Quercus ilex ecotypes under identical environmental conditions. For. Ecol. Manag. 2018, 415, 58–69. [Google Scholar] [CrossRef]
- Ramirez-Valiente, J.A.; Lorenzo, Z.; Soto, A.; Valladares, F.; Gil, L.; Aranda, I. Elucidating the role of genetic drift and natural selection in cork oak differentiation regarding drought tolerance. Mol. Ecol. 2009, 18, 3803–3815. [Google Scholar] [CrossRef]
- Rey, M.D.; Castillejo, M.A.; Sánchez-Lucas, R.; Guerrero-Sanchez, V.M.; López-Hidalgo, C.; Romero-Rodríguez, C.; Valero-Galván, J.V.; Sghaier-Hammami, B.; Simova-Stilova, L.; Echevarría-Zomeño, S.; et al. Proteomics, Holm oak (Quercus ilex L.) and other recalcitrant and orphan forest tree species: How do they see each other? Int. J. Mol. Sci. 2019, 20, 692. [Google Scholar] [CrossRef]
- Fernández i Marti, A.; Romero-Rodríguez, C.; Navarro-Cerrillo, R.; Abril, N.; Jorrín-Novo, J.V.; Dodd, R. Population genetic diversity of Quercus ilex subsp. ballota (Desf.) Samp. reveals divergence in recent and evolutionary migration rates in the Spanish dehesas. Forests 2018, 9, 337. [Google Scholar]
- Simova-Stoilova, L.P.; Romero-Rodríguez, M.C.; Sánchez-Lucas, R.; Navarro-Cerrillo, R.M.; Medina-Aunon, J.A.; Jorrín-Novo, J.V. 2-DE proteomics analysis of drought treated seedlings of Quercus ilex supports a root active strategy for metabolic adaptation in response to water shortage. Front. Plant Sci. 2015, 6, 627. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants Without Soil, 2nd ed.; California Agricultural Experiment Station: Berkeley, CA, USA, 1950; p. 32. [Google Scholar]
- Bilger, U.; Schreiber, U.; Bock, M. Determination of the quantum efficiency of photosystem II and of non-photochemical quenching of chlorophyll. Oecologia 1995, 102, 425–432. [Google Scholar] [CrossRef]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The fluorescence transient as a tool to characterize and screen photosynthetic samples. In Probing Photosynthesis: Mechanisms, Regulation and Adaptation; Taylor and Francis: London, UK, 2000; pp. 445–483. [Google Scholar]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1953, 31, 426–428. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Gillespie, K.M. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Starcher, B. A ninhydrin-based assay to quantitate the total protein content of tissue samples. Anal. Biochem. 2001, 292, 25–129. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, D.; Veste, M.; Badorreck, A.; Freese, D. Evaluation of fast growing tree water use under different soil moisture regimes using wick lysimeters. iForest-Biogeosci. For. 2013, 6, 190. [Google Scholar] [CrossRef]
- Jensen, C.R.; Mogensen, V.O.; Poulsen, H.H.; Henson, I.E.; Aagot, S.; Hansen, E.; Ali, M.; Wollenweber, B. Soil water matric potential rather than water content determines drought responses in field-grown lupin (Lupinus angustifolius). Funct. Plant Biol. 1998, 25, 353–363. [Google Scholar] [CrossRef]
- Aliniaeifard, S.; Rezaei-Nejad, A.; Seifi-Kalhor, M.; Shahlaei, A.; Aliniaeifard, A. Comparison of soil and perlite (with nutrient solution supply) growing media for cultivation of lemon verbena (Lippia citriodora var. ‘Verbena’). Med. Aromat. Plant Sci. Biotechnol. 2010, 4, 30–33. [Google Scholar]
- Tala, S.; Al-Ajlouni, M.G.; Ayad, J.Y.; Othman, Y.A.; Hilaire, R.S. Performance of six different soilless green roof substrates for the Mediterranean region. Sci. Total Environ. 2020, 730, 139182. [Google Scholar]
- Maloupa, E.; Mitsios, P.F.; Martinez, S.B. Study of substrate use in Gerbera soilless culture grown in plastic greenhouses. Acta Hort. 1992, 323, 139–144. [Google Scholar] [CrossRef]
- Sudhakar, P.; Latha, P.; Reddy, P.V. Phenotyping Crop plants for Physiological and Biochemical Traits; Academic Press: London, UK, 2016. [Google Scholar]
- Hanson, A.D.; Nelsen, C.E.; Everson, E.H. Evaluation of free proline accumulation as an index of drought resistance using two contrasting barley cultivars. Crop. Sci. 1977, 17, 720–726. [Google Scholar] [CrossRef]
- Syros, T.; Yupsanis, T.; Omirou, M.; Economou, A. Photosynthetic response and peroxidases in relation to water and nutrient deficiency in gerbera. Environ. Exp. Bot. 2004, 52, 23–31. [Google Scholar] [CrossRef]
- Al-Shammari, A.M.A.; Abood, M.A.; Hamdi, G.J. Perlite affects some plant indicators and reduces water deficit in tomato. Int. J. Veg. Sci. 2018, 24, 490–500. [Google Scholar] [CrossRef]
- Arbona, V.; Iglesias, D.J.; Jacas, J.; Primo-Millo, E.; Talón, M.; Gómez-Cadenas, A. Hydrogel substrate amendment alleviates drought effects on young citrus plants. Plant Soil 2005, 270, 73–82. [Google Scholar] [CrossRef]
- Manzi, M.; Lado, J.; Rodrigo, M.J.; Zacarías, L.; Arbona, V.; Gómez-Cadenas, A. Root ABA accumulation in long-term water-stressed plants is sustained by hormone transport from aerial organs. Plant Cell Physiol. 2015, 56, 2457–2466. [Google Scholar] [CrossRef] [PubMed]
- Samartzidis, C.; Awada, T.; Maloupa, E.; Radoglou, K.; Constantinidou, H.I. Rose productivity and physiological responses to different substrates for soil-less culture. Sci. Hortic. 2005, 106, 203–212. [Google Scholar] [CrossRef]
- Jones, H.G. Monitoring plant and soil water status: Established and novel methods revisited and their relevance to studies of drought tolerance. J. Exp. Bot. 2007, 58, 119–130. [Google Scholar] [CrossRef]
- Gil-Pelegrín, E.; Saz, M.Á.; Cuadrat, J.M.; Peguero-Pina, J.J.; Sancho-Knapik, D. Oaks under Mediterranean-type climates: Functional response to summer aridity. In Oaks Physiological Ecology. Exploring the Functional Diversity of Genus Quercus L.; Springer: New York, NY, USA, 2017; pp. 137–193. [Google Scholar]
- Corcuera, L.; Camarero, J.J.; Gil-Pelegrín, E. Functional groups in Quercus species derived from the analysis of pressure-volume curves. Trees 2002, 16, 465–472. [Google Scholar] [CrossRef]
- Peguero-Pina, J.J.; Sancho-Knapik, D.; Barrón, E.; Camarero, J.J.; Vilagrosa, A.; Gil-Pelegrín, E. Morphological and physiological divergences within Quercus ilex support the existence of different ecotypes depending on climatic dryness. Ann. Bot. 2014, 114, 301–313. [Google Scholar] [CrossRef]
- Savé, R.; Castell, C.; Terradas, J. Gas exchange and water relations. In Ecology of Mediterranean Evergreen Oak Forests; Springer: Berlin, Germany, 1999; pp. 135–146. [Google Scholar]
- Sardans, J.; Peñuelas, J. Plant-soil interactions in Mediterranean forest and shrublands: Impacts of climatic change. Plant Soil 2013, 365, 1–33. [Google Scholar] [CrossRef]
- Gómez, J.M. Importance of microhabitat and acorn burial on Quercus ilex early recruitment: Non-additive effects on multiple demographic processes. Plant Ecol. 2004, 172, 287–297. [Google Scholar]
- Peguero-Piña, J.J.; Sancho-Knapik, D.; Morales, F.; Flexas, J.; Gil-Pelegrín, E. Differential photosynthetic performance and photoprotection mechanisms of three Mediterranean evergreen oaks under severe drought stress. Funct. Plant Biol. 2009, 36, 453–462. [Google Scholar] [CrossRef]
- Sancho-Knapik, D.; Mendoza-Herrer, Ó.; Gil-Pelegrín, E.; Peguero-Pina, J.J. Chl fluorescence parameters and leaf reflectance indices allow monitoring changes in the physiological status of Quercus ilex L. Under progressive water deficit. Forests 2018, 9, 400. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R.; Rosenqvist, E. Applications of chlorophyll fluorescence can improve crop production strategies: An examination of future possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Valiente, J.A.; López, R.; Hipp, A.L.; Aranda, I. Correlated evolution of morphology, gas exchange, growth rates and hydraulics as a response to precipitation and temperature regimes in oaks (Quercus). New Phytol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Méthy, M.; Damesin, C.; Rambal, S. Drought and photosystem II activity in two Mediterranean oaks. Ann. For. Sci. 1996, 53, 255–262. [Google Scholar] [CrossRef]
- Vilagrosa, A.; Morales, F.; Abadía, A.; Bellot, J.; Cochard, H.; Gil-Pelegrín, E. Are symplast tolerance to intense drought conditions and xylem vulnerability to cavitation coordinated? An integrated analysis of photosynthetic, hydraulic and leaf level processes in two Mediterranean drought-resistant species. Environ. Exp. Bot. 2010, 69, 233–242. [Google Scholar] [CrossRef]
- Fernández-Marín, B.; Hernández, A.; Garcia-Plazaola, J.I.; Esteban, R.; Míguez, F.; Artetxe, U.; Gómez-Sagasti, M.T. Photoprotective Strategies of Mediterranean Plants in Relation to Morphological Traits and Natural Environmental Pressure: A Meta-Analytical Approach. Front. Plant Sci. 2017, 8, 1051. [Google Scholar] [CrossRef]
- Ramírez-Valiente, J.A.; Aranda, I.; Sanchéz-Gómez, D.; Rodríguez-Calcerrada, J.; Valladares, F.; Robson, T.M. Increased root investment can explain the higher survival of seedlings of ‘mesic’ Quercus suber than ‘xeric’ Quercus ilex in sandy soils during a summer drought. Tree Physiol. 2019, 39, 64–75. [Google Scholar]
- Quero, J.L.; Villar, R.; Marañón, T.; Zamora, R. Interactions of drought and shade effects on seedlings of four Quercus species: Physiological and structural leaf responses. New Phytol. 2006, 170, 819–834. [Google Scholar] [CrossRef]
- Merilo, E.; Jõesaar, I.; Brosché, M.; Kollist, H. To open or to close: Species-specific stomatal responses to simultaneously applied opposing environmental factors. New Phytol. 2014, 202, 499–508. [Google Scholar] [CrossRef]
- Martin, N.; Roussel, A.; Rodriguez-Calcerrada, J.; Ruiz, J.T.; de Jalon, L.G.; Moreno, M.; Ourcival, J.M.; Simioni, G.; Cochard, H.; Limousin, J.M. Plasticity in leaf hydraulic traits and water relations in Mediterranean evergreen Quercus ilex subjected to long-term rainfall exclusion. Geophys. Res. Abstr. 2019, 21, 1. [Google Scholar]
- Acherar, M.; Ramblal, S. Comparative water relations of four mediterranean oak species. In Quercus ilex L. Ecosystems: Function, Dynamics and Management; Springer: Dordrecht, The Netherlands, 1992; pp. 177–184. [Google Scholar]
- Vaz, M.; Pereira, J.S.; Gazarini, L.C.; David, T.S.; David, J.S.; Rodrigues, A.; Maroco, J.; Chaves, M.M. Drought-induced photosynthetic inhibition and autumn recovery in two Mediterranean oak species (Quercus ilex and Quercus suber). Tree Physiol. 2010, 30, 946–956. [Google Scholar] [CrossRef] [PubMed]
- Barbeta, A.; Peñuelas, J. Sequence of plant responses to droughts of different timescales: Lessons from holm oak (Quercus ilex) forests. Plant. Ecol. Divers. 2016, 9, 321–338. [Google Scholar] [CrossRef]
- Epron, D.; Dreyer, E. Effects of severe dehydration on leaf photosynthesis in Quercus petraea (Matt.) Liebl.: Photosystem II efficiency, photochemical and non-photochemical fluorescence quenching and electrolyte leakage. Tree Physiol. 1992, 10, 273–284. [Google Scholar] [CrossRef]
- Gallé, A.; Haldimann, P.; Feller, U. Photosynthetic performance and water relations in young pubescent oak (Quercus pubescens) trees during drought stress and recovery. New Phytol. 2007, 174, 799–810. [Google Scholar] [CrossRef]
- Spyropoulos, C.G.; Mavrommatis, M. Effect of water stress on pigment formation in Quercus species. J. Exp. Bot. 1978, 29, 473–477. [Google Scholar] [CrossRef]
- Rivas-Ubach, A.; Gargallo-Garriga, A.; Sardans, J.; Oravec, M.; Mateu-Castell, L.; Pérez-Trujillo, M.; Parella, T.; Ogaya, R.; Urban, O.; Peñuelas, J. Drought enhances folivory by shifting foliar metabolomes in Quercus ilex trees. New Phytol. 2014, 202, 874–885. [Google Scholar] [CrossRef]
- Rodríguez-Calcerrada, J.; Rodrigues, A.M.; Perdiguero, P.; António, C.; Atkin, O.K.; Li, M.; Collada, C.; Gil, L. A molecular approach to drought-induced reduction in leaf CO2 exchange in drought-resistant Quercus ilex. Physiol. Plant. 2018, 162, 394–408. [Google Scholar]
- Simova-Stoilova, L.P.; López-Hidalgo, C.; Sanchez-Lucas, R.; Valero-Galvan, J.; Romero-Rodríguez, C.; Jorrín-Novo, J.V. Holm oak proteomic response to water limitation at seedling establishment stage reveals specific changes in different plant parts as well as interaction between roots and cotyledons. Plant Sci. 2018, 276, 1–13. [Google Scholar] [CrossRef]
- Maguire, A.J.; Kobe, R.K. Drought and shade deplete nonstructural carbohydrate reserves in seedlings of five temperate tree species. Ecol. Evol. 2015, 5, 5711–5721. [Google Scholar] [CrossRef]
- López-Hidalgo, C.; Guerrero-Sánchez, V.M.; Gómez-Gálvez, I.; Sánchez-Lucas, R.; Castillejo, M.A.; Maldonado-Alconada, A.M.; Valledor, L.; Jorrín-Novo, J.V. A multi-omics analysis pipeline for the metabolic pathway reconstruction in the orphan species Quercus ilex. Front. Plant. Sci. 2018, 9, 935. [Google Scholar] [CrossRef] [PubMed]
- Hughes, N.M.; Reinhardt, K.; Feild, T.S.; Gerardi, A.R.; Smith, W.K. Association between winter anthocyanin production and drought stress in angiosperm evergreen species. J. Exp. Bot. 2010, 61, 1699–1709. [Google Scholar] [CrossRef] [PubMed]
| Species | Location | MASL (m) | Tmax (°C) | Tmin (°C) | P (mm) |
|---|---|---|---|---|---|
| Quercus robur | ES01 | 488 | 24.8 | 2.2 | 1375 |
| Quercus faginea | ES10 | 1032 | 27.6 | −0.1 | 577 |
| Quercus pyrenaica | ES08 | 823 | 24.5 | −1.8 | 792 |
| Quercus suber | ES01 | 456 | 33.7 | 2.4 | 824 |
| Quercus ilex | ES11 | 506 | 34.5 | 2.6 | 635 |
| Quercus ilex (Jaen) | 38°17′ N, 2°36′ W | 643 | 23.1 | 4.4 | 795 |
| Quercus ilex (Granada) | 36°57′ N, 3°54′ W | 489 | 24.7 | 11.5 | 489 |
| Quercus ilex (Cordoba) | 38°22′ N, 4°54′ W | 618 | 26.8 | 8.1 | 613 |
| Quercus ilex (Cadiz) | 36°45′ N, 5°27′ W | 649 | 24.9 | 9.8 | 1264 |
| Quercus ilex (Seville) | 37°52′ N, 6°05′ W | 482 | 26.4 | 9.5 | 722 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
San-Eufrasio, B.; Sánchez-Lucas, R.; López-Hidalgo, C.; Guerrero-Sánchez, V.M.; Castillejo, M.Á.; Maldonado-Alconada, A.M.; Jorrín-Novo, J.V.; Rey, M.-D. Responses and Differences in Tolerance to Water Shortage under Climatic Dryness Conditions in Seedlings from Quercus spp. and Andalusian Q. ilex Populations. Forests 2020, 11, 707. https://doi.org/10.3390/f11060707
San-Eufrasio B, Sánchez-Lucas R, López-Hidalgo C, Guerrero-Sánchez VM, Castillejo MÁ, Maldonado-Alconada AM, Jorrín-Novo JV, Rey M-D. Responses and Differences in Tolerance to Water Shortage under Climatic Dryness Conditions in Seedlings from Quercus spp. and Andalusian Q. ilex Populations. Forests. 2020; 11(6):707. https://doi.org/10.3390/f11060707
Chicago/Turabian StyleSan-Eufrasio, Bonoso, Rosa Sánchez-Lucas, Cristina López-Hidalgo, Victor M. Guerrero-Sánchez, María Ángeles Castillejo, Ana María Maldonado-Alconada, Jesús V. Jorrín-Novo, and María-Dolores Rey. 2020. "Responses and Differences in Tolerance to Water Shortage under Climatic Dryness Conditions in Seedlings from Quercus spp. and Andalusian Q. ilex Populations" Forests 11, no. 6: 707. https://doi.org/10.3390/f11060707
APA StyleSan-Eufrasio, B., Sánchez-Lucas, R., López-Hidalgo, C., Guerrero-Sánchez, V. M., Castillejo, M. Á., Maldonado-Alconada, A. M., Jorrín-Novo, J. V., & Rey, M.-D. (2020). Responses and Differences in Tolerance to Water Shortage under Climatic Dryness Conditions in Seedlings from Quercus spp. and Andalusian Q. ilex Populations. Forests, 11(6), 707. https://doi.org/10.3390/f11060707





