Differential Metabolite Accumulation in Different Tissues of Gleditsia sinensis under Water Stress and Rehydration Conditions
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Materials and Growing Conditions
2.3. Measurement of Antioxidants and Malondialdehyde
2.4. Analysis of Photosynthesis Indexes of Plants
2.5. GC-MS Analysis of Primary Metabolites
2.6. LC-MS Targeted Analysis of Phenolic Metabolites
2.7. Multivariate Analysis
3. Results
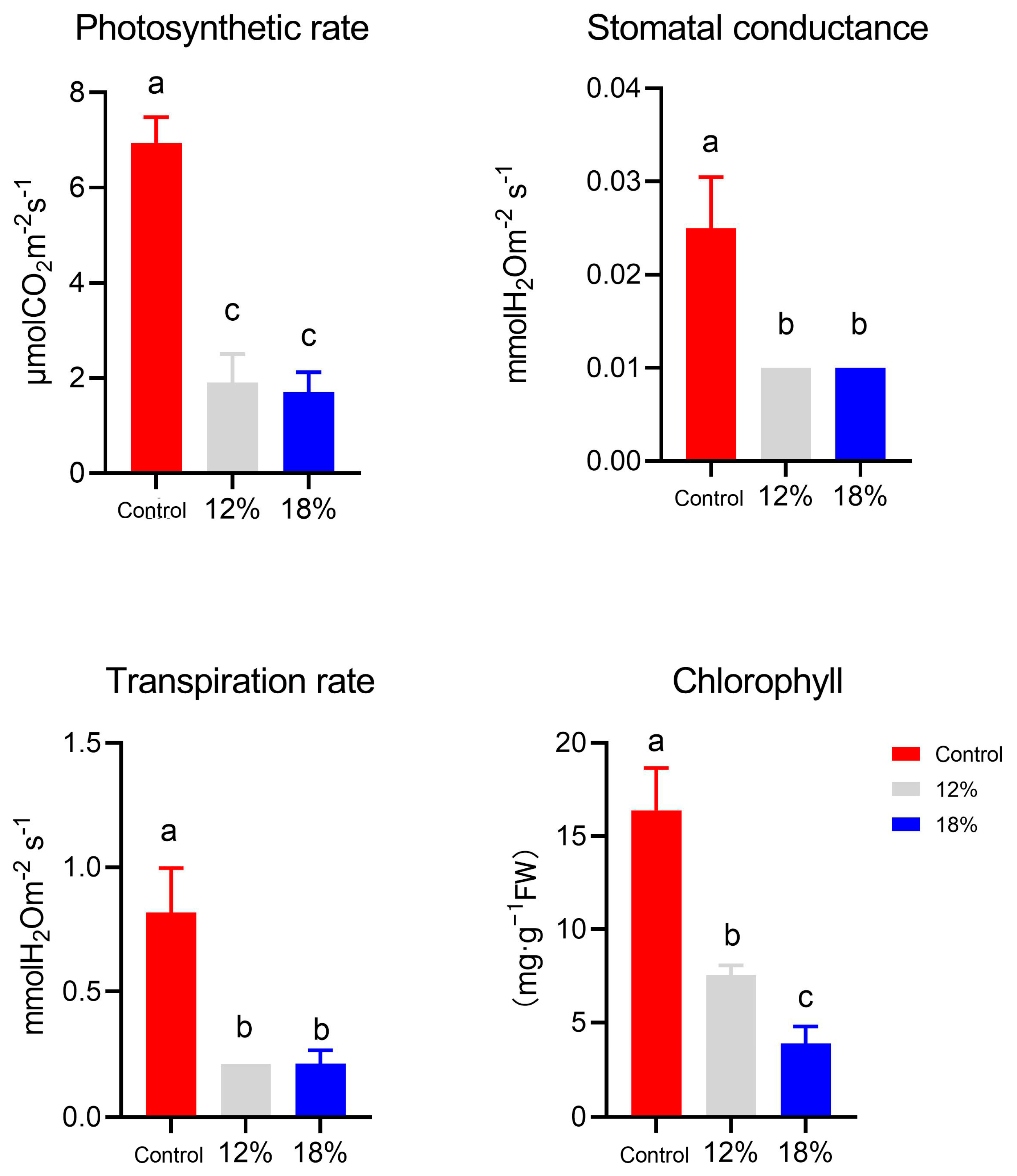
3.1. Physiological Damage under Drought Conditions
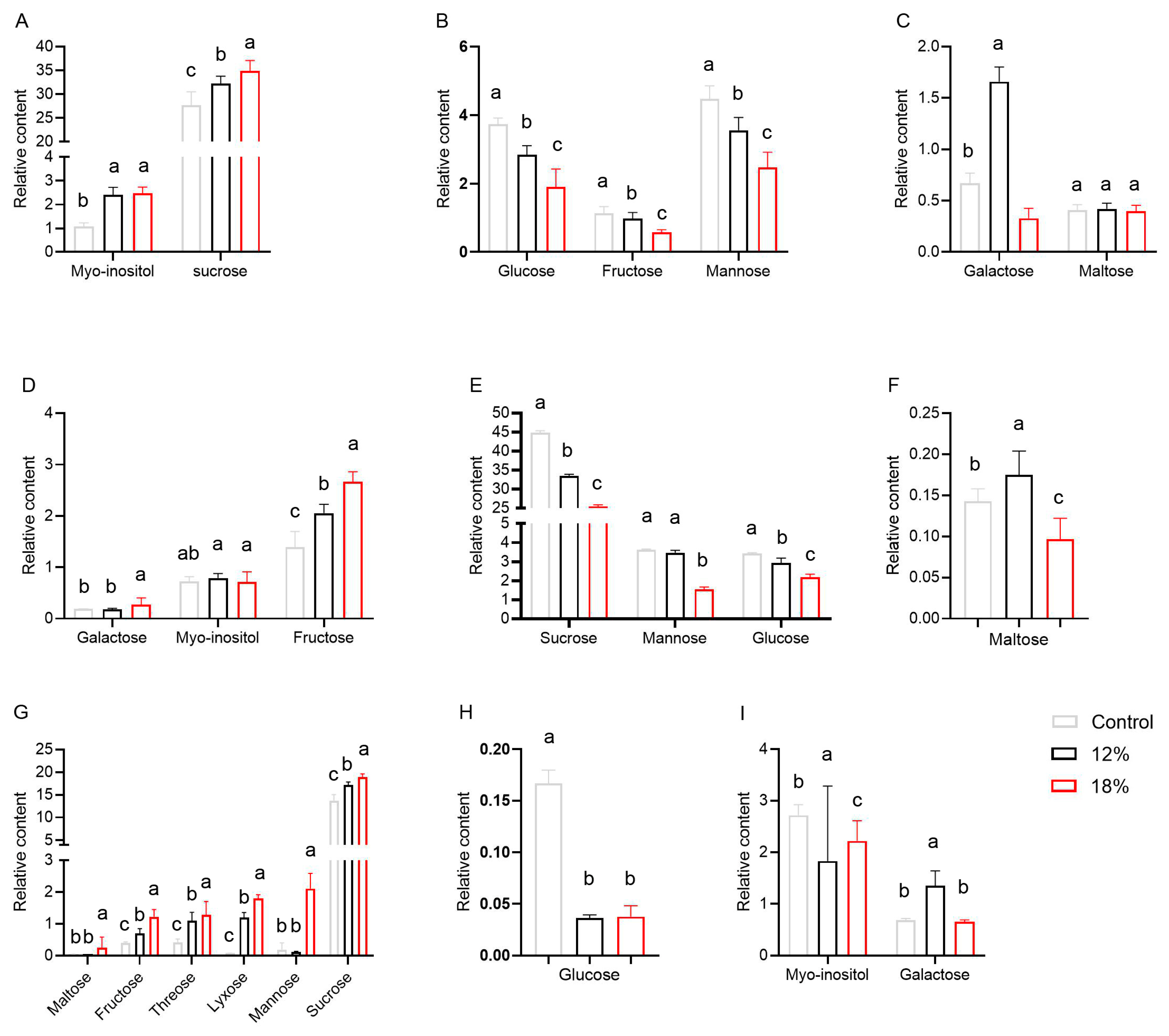
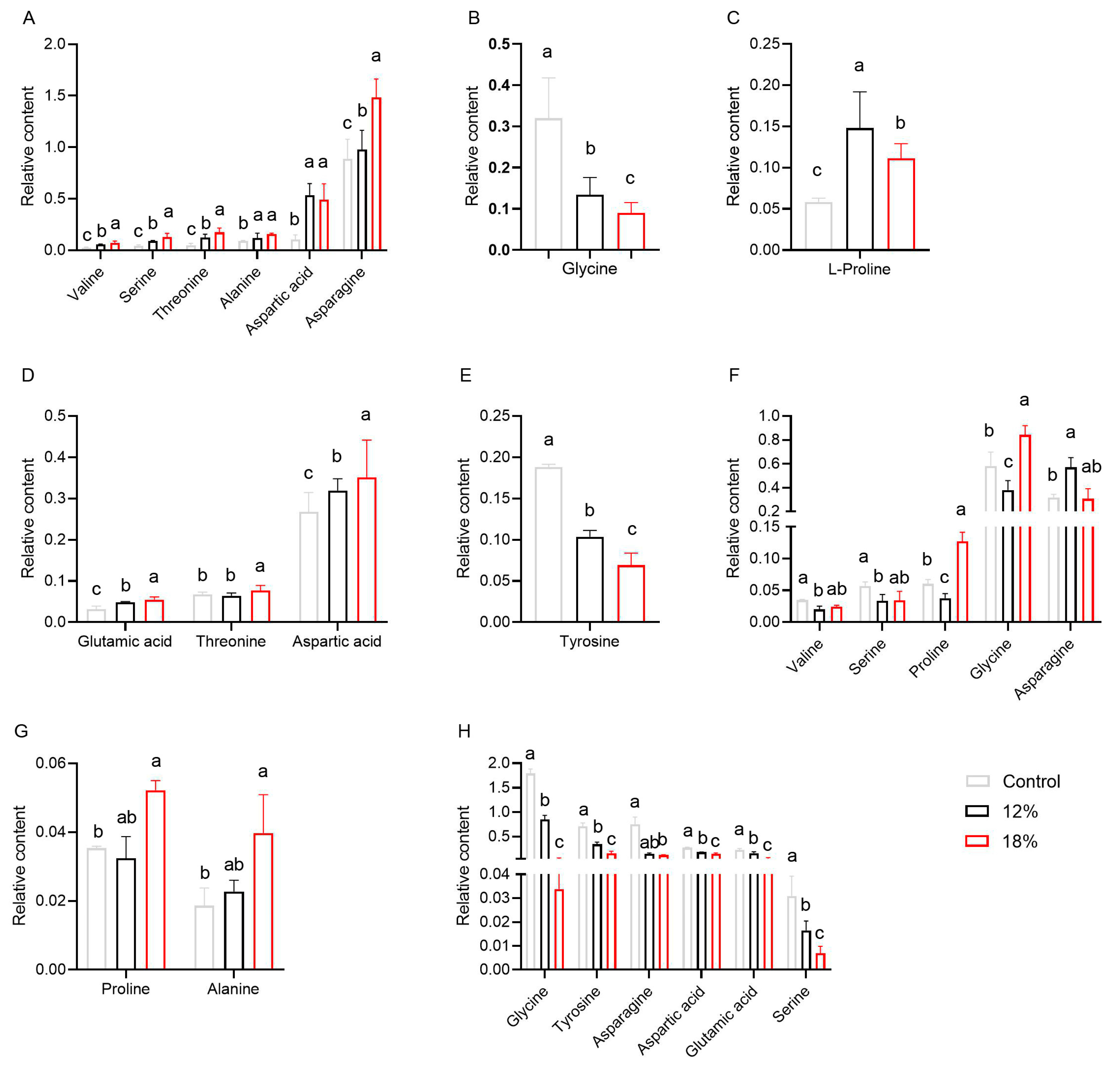
3.2. Metabolic Profile Analysis after Control and Drought Stress Treatments
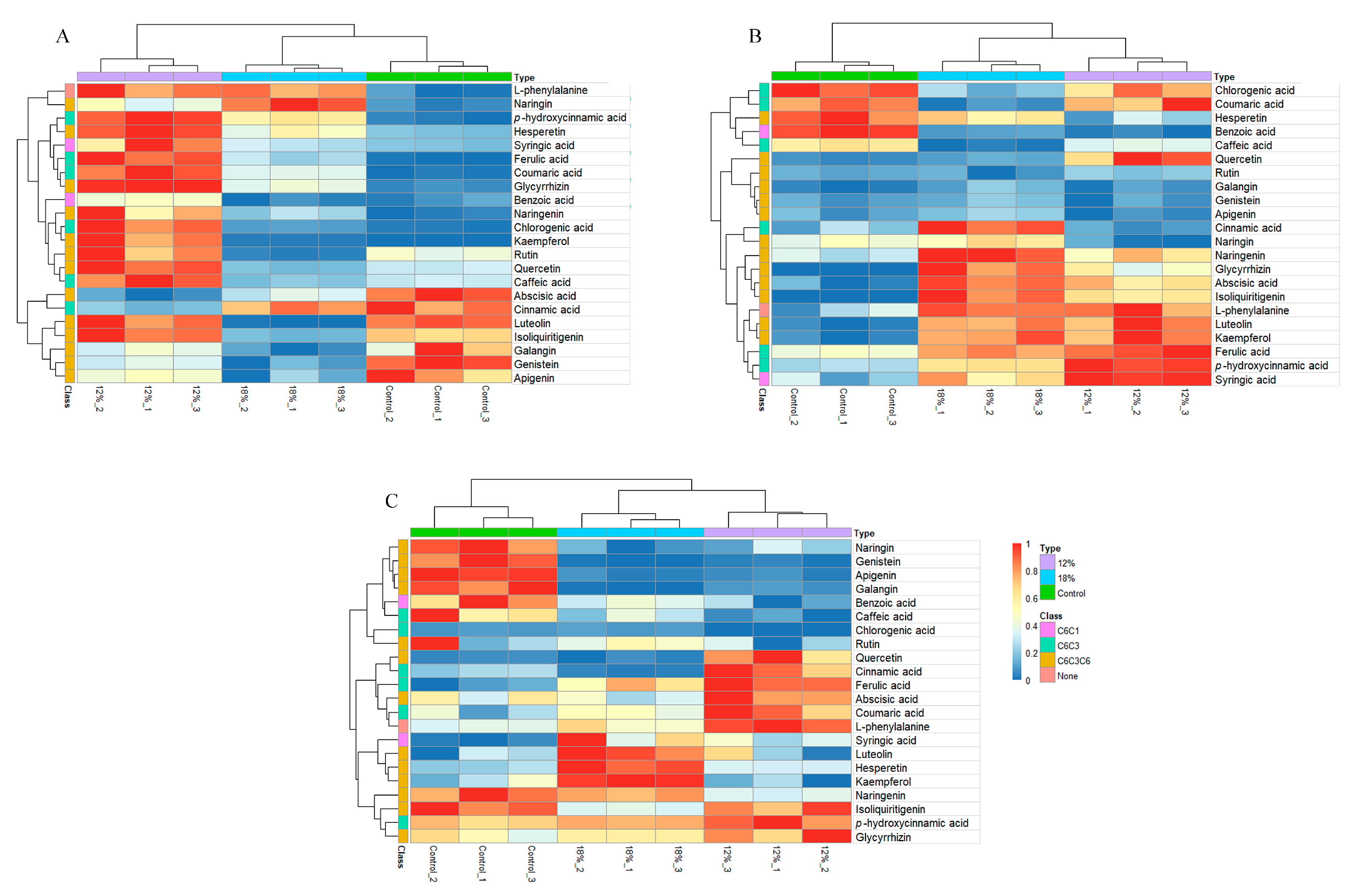
3.3. Identification of Polyphenols in G. sinensis Adapted to Drought
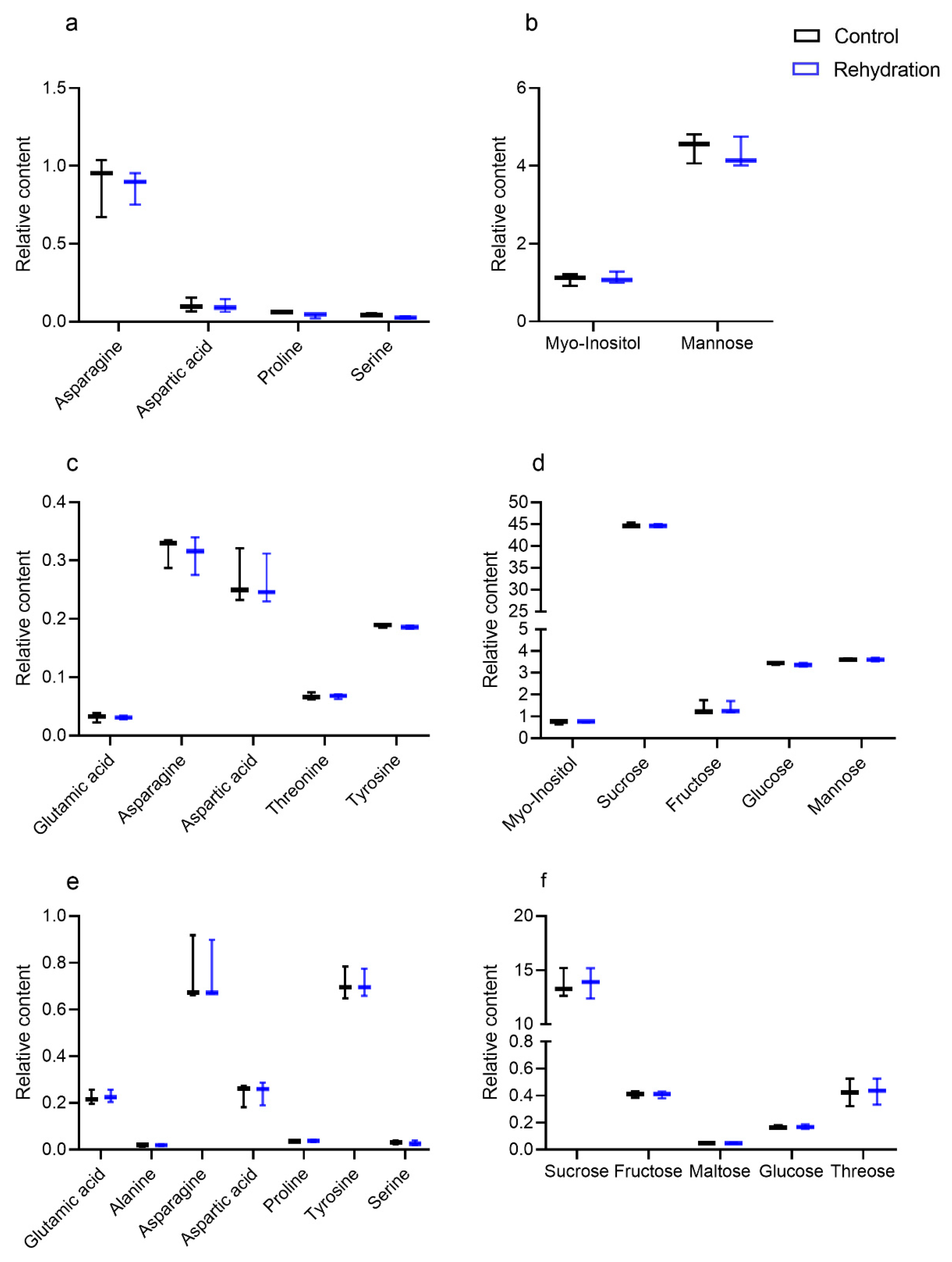
3.4. Phenotype, Physiological Parameters and Metabolites under Rehydration
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Soussana, F.N.T.J.F.; Howden, S.M. Crop and pasture response to climate change. Proc. Natl. Acad. Sci. USA 2007, 104, 19686–19690. [Google Scholar]
- Selmar, D.; Kleinwachter, M. Influencing the product quality by deliberately applying drought stress during the cultivation of medicinal plants. Ind. Crop. Prod. 2013, 42, 558–566. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant Biol. 2018, 18, 258. [Google Scholar] [CrossRef]
- Swarcewicz, B.; Sawikowska, A.; Marczak, L.; Luczak, M.; Ciesiolka, D.; Krystkowiak, K.; Kuczynska, A.; Pislewska-Bednarek, M.; Krajewski, P.; Stobiecki, M. Effect of drought stress on metabolite contents in barley recombinant inbred line population revealed by untargeted GC-MS profiling. Acta Physiol. Plant. 2017, 39, 158. [Google Scholar] [CrossRef]
- Meena, K.K.; Sorty, A.M.; Bitla, U.M.; Choudhary, K.; Gupta, P.; Pareek, A.; Singh, D.P.; Prabha, R.; Sahu, P.K.; Gupta, V.K.; et al. Abiotic stress responses and microbe-mediated mitigation in plants: The omics strategies. Front. Plant Sci. 2017, 8, 172. [Google Scholar] [CrossRef]
- You, J.; Zhang, Y.J.; Liu, A.L.; Li, D.H.; Wang, X.; Dossa, K.; Zhou, R.; Yu, J.Y.; Zhang, Y.X.; Wang, L.H.; et al. Transcriptomic and metabolomic profiling of drought-tolerant and susceptible sesame genotypes in response to drought stress. BMC Plant Biol. 2019, 19, 267. [Google Scholar] [CrossRef]
- Ma, D.Y.; Sun, D.X.; Wang, C.Y.; Li, Y.G.; Guo, T.C. Expression of flavonoid biosynthesis genes and accumulation of flavonoid in wheat leaves in response to drought stress. Plant Physiol. Biochem. 2014, 80, 60–66. [Google Scholar] [CrossRef]
- Tian, S.; Nakamura, K.; Kayahara, H. Analysis of phenolic compounds in white rice, brown rice, and germinated brown rice. J. Agric. Food Chem. 2004, 52, 4808–4813. [Google Scholar] [CrossRef]
- Torras-Claveria, L.; Jauregui, O.; Codina, C.; Tiburcio, A.F.; Bastida, J.; Viladomat, F. Analysis of phenolic compounds by high-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry in senescent and water-stressed tobacco. Plant Sci. 2012, 182, 71–78. [Google Scholar] [CrossRef]
- Nakabayashi, R.; Yonekura-Sakakibara, K.; Urano, K.; Suzuki, M.; Yamada, Y.; Nishizawa, T.; Matsuda, F.; Kojima, M.; Sakakibara, H.; Shinozaki, K.; et al. Enhancement of oxidative and drought tolerance in Arabidopsis by overaccumulation of antioxidant flavonoids. Plant J. 2014, 77, 367–379. [Google Scholar] [CrossRef]
- Quan, N.; La, A.; Do, K.; Phung, T.; Nguyen, T.; Truong, M.; Luong, M.; Do, B.; Pham, H.; Abdelnaser, E. Involvement of secondary metabolites in response to drought stress of rice (Oryza sativa L.). Agriculture 2016, 6, 23. [Google Scholar] [CrossRef]
- Espadas, J.L.; Castano, E.; Marina, M.L.; Rodriguez, L.C.; Plaza, M. Phenolic compounds increase their concentration in Carica papaya leaves under drought stress. Acta Physiol. Plant. 2019, 41, 180. [Google Scholar] [CrossRef]
- Tang, W.J.; Hazebroek, J.; Zhong, C.; Harp, T.; Vlahakis, C.; Baumhover, B.; Asiago, V. Effect of genetics, environment, and phenotype on the metabolome of Maize Hybrids Using GC/MS and LC/MS. J. Agric. Food Chem. 2017, 65, 5215–5225. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Zhang, Z.H.; Zu, Y.G.; Efferth, T.; Tang, Z.H. The combined effects of ethylene and MeJA on metabolic profiling of phenolic compounds in Catharanthus roseus revealed by metabolomics analysis. Front. Physiol. 2016, 7, 217. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Abozeid, A.; Zu, Y.G.; Tang, Z.H. The integration of GC-MS and LC-MS to assay the metabolomics profiling in Panax ginseng and Panax quinquefolius reveals a tissue- and species-specific connectivity of primary metabolites and ginsenosides accumulation. J. Pharm. Biomed. Anal. 2017, 135, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Razavizadeh, R.; Komatsu, S. Changes in essential oil and physiological parameters of callus and seedlings of Carum copticum L. under in vitro drought stress. J. Food Meas. Charact. 2018, 12, 1581–1592. [Google Scholar] [CrossRef]
- Corrales, A.R.; Carrillo, L.; Lasierra, P.; Nebauer, S.G.; Dominguez-Figueroa, J.; Renau-Morata, B.; Pollmann, S.; Granell, A.; Molina, R.V.; Vicente-Carbajosa, J.; et al. Multifaceted role of cycling DOF factor 3 (CDF3) in the regulation of flowering time and abiotic stress responses in Arabidopsis. Plant Cell Environ. 2017, 40, 748–764. [Google Scholar] [CrossRef]
- Singh, R.; Gupta, P.; Khan, F.; Singh, S.K.; T. Mishra, S.; Kumar, A.; Dhawan, S.S.; Shirke, P.A. Modulations in primary and secondary metabolic pathways and adjustment in physiological behaviour of Withania somnifera under drought stress. Plant Sci. 2018, 272, 42–54. [Google Scholar] [CrossRef]
- Liu, T.Y.; Chen, M.X.; Zhang, Y.J.; Zhu, F.Y.; Liu, Y.G.; Tian, Y.; Fernie, A.R.; Ye, N.H.; Zhang, J.H. Comparative metabolite profiling of two switchgrass ecotypes reveals differences in drought stress responses and rhizosheath weight. Planta 2019, 250, 1355–1369. [Google Scholar] [CrossRef]
- Liu, Y.T.; Xu, W.; Lei, F.H.; Li, P.F.; Jiang, J.X. Comparison and characterization of galactomannan at different developmental stages of Gleditsia sinensis Lam. Carbohydr. Polym. 2019, 223, 115127. [Google Scholar] [CrossRef]
- Wu, C.; Yang, X.; Feng, L.; Wang, F.; Yin, Y. Identification of key leaf color-associated genes in Gleditsia sinensis using bioinformatics, Horticulture. Environ. Biotechnol. 2019, 60, 711–720. [Google Scholar]
- Kuwahara, Y.; Nakajima, D.; Shinpo, S.; Nakamura, M.; Kawano, N.; Kawahara, N.; Yamazaki, M.; Saito, K.; Suzuki, H.; Hirakawa, H. Identification of potential genes involved in triterpenoid saponins biosynthesis in Gleditsia sinensis by transcriptome and metabolome analyses. J. Nat. Med. 2019, 73, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Zhang, C.Z.; Zhan, L.; Cheng, L.B.; Lu, D.B.; Wang, X.D.; Xu, H.L.; Wang, S.X.; Wu, D.; Ruan, L.G. Anticancer effects of Gleditsia sinensis extract in rats transplanted with hepatocellular carcinoma cells. Oncol. Res. 2019, 27, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Seo, C.S.; Lim, H.S.; Ha, H.; Jin, S.E.; Shin, H.K. Quantitative analysis and anti-inflammatory effects of Gleditsia sinensis thorns in RAW 264.7 macrophages and HaCaT keratinocytes. Mol. Med. Rep. 2015, 12, 4773–4781. [Google Scholar] [CrossRef]
- Zhang, J.P.; Tian, X.H.; Yang, Y.X.; Liu, Q.X.; Wang, Q.; Chen, L.P.; Li, H.L.; Zhang, W.D. Gleditsia species: An ethnomedical, phytochemical and pharmacological review. J. Ethnopharmacol. 2016, 178, 155–171. [Google Scholar] [CrossRef]
- Meunier, J.D.; Barboni, D.; Anwar-ul-Haq, M.; Levard, C.; Chaurand, P.; Vidal, V.; Grauby, O.; Huc, R.; Laffont-Schwob, I.; Rabier, J.; et al. Effect of phytoliths for mitigating water stress in durum wheat. New Phytol. 2017, 215, 229–239. [Google Scholar] [CrossRef]
- Zhu, B.; Xu, Q.W.; Zou, Y.G.; Ma, S.M.; Zhang, X.D.; Xie, X.Y.; Wang, L.C. Effect of potassium deficiency on growth, antioxidants, ionome and metabolism in rapeseed under drought stress. Plant Growth Regul. 2019, 90, 1–12. [Google Scholar] [CrossRef]
- Kang, J.; Zhao, W.; Zhu, X. Silicon improves photosynthesis and strengthens enzyme activities in the C3 succulent xerophyte Zygophyllum xanthoxylum under drought stress. J. Plant Physiol. 2016, 199, 76–86. [Google Scholar] [CrossRef]
- Cuadros-Inostroza, A.; Caldana, C.; Redestig, H.; Kusano, M.; Lisec, J.; Pena-Cortes, H.; Willmitzer, L.; Hannah, M.A. TargetSearch—A Bioconductor package for the efficient preprocessing of GC-MS metabolite profiling data. BMC Bioinform. 2009, 10, 428. [Google Scholar] [CrossRef]
- Jian, H.L.; Cristhian, C.; Zhang, W.M.; Jiang, J.X. Influence of dehulling pretreatment on physicochemical properties of Gleditsia sinensis Lam. gum. Food Hydrocoll. 2011, 25, 1337–1343. [Google Scholar] [CrossRef]
- Abedi, T.; Pakniyat, H. Antioxidant enzyme changes in response to drought stress in ten cultivars of Oilseed Rape (Brassica napus L.). Czech J. Genet. Plant Breed. 2010, 46, 27–34. [Google Scholar] [CrossRef]
- Ma, Q.; Yue, L.J.; Zhang, J.L.; Wu, G.Q.; Bao, A.K.; Wang, S.M. Sodium chloride improves photosynthesis and water status in the succulent xerophyte Zygophyllum xanthoxylum. Tree Physiol. 2012, 32, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 39. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Jiang, J.G. Osmotic adjustment and plant adaptation to environmental changes related to drought and salinity. Environ. Rev. 2010, 18, 309–319. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B.S. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic Stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef]
- Kasote, D.M.; Katyare, S.S.; Hegde, M.V.; Bae, H. Significance of antioxidant potential of plants and its relevance to therapeutic applications. Int. J. Biol. Sci. 2015, 11, 982–991. [Google Scholar] [CrossRef]
- Deng, Y.X.; Lu, S.F. Biosynthesis and regulation of phenylpropanoids in plants. Crit. Rev. Plant Sci. 2017, 36, 257–290. [Google Scholar] [CrossRef]
- Heras, R.M.; Quifer-Rada, P.; Andres, A.; Lamuela-Raventos, R. Polyphenolic profile of persimmon leaves by high resolution mass spectrometry (LC-ESI-LTQ-Orbitrap-MS). J. Funct. Foods 2016, 23, 370–377. [Google Scholar] [CrossRef]
- Ma, Y.Y.; Kosinska-Cagnazzo, A.; Kerr, W.L.; Amarowicz, R.; Swanson, R.B.; Pegg, R.B. Separation and characterization of phenolic compounds from dry-blanched peanut skins by liquid chromatography-electrospray ionization mass spectrometry. J. Chromatogr. A 2014, 1356, 64–81. [Google Scholar] [CrossRef]
- Li, X.P.; Xu, Q.Q.; Liao, W.B.; Ma, Z.J.; Xu, X.T.; Wang, M.; Ren, P.J.; Niu, L.J.; Jin, X.; Zhu, Y.C. Hydrogen peroxide is involved in abscisic acid-induced adventitious rooting in cucumber (Cucumis sativus L.) under drought stress. J. Plant Biol. 2016, 59, 536–548. [Google Scholar] [CrossRef]

| Treatment | Fresh Weight (g) | Length (cm) | |||
|---|---|---|---|---|---|
| Root | Stem | Leaf | Root | Stem | |
| Control | 1.19 ± 0.08a | 0.77 ± 0.04a | 2.23 ± 0.24a | 23.05 ± 5.32a | 24.00 ± 2.96a |
| 12% PEG | 0.93 ± 0.10b | 0.46 ± 0.07b | 1.81 ± 0.21b | 21.62 ± 2.97b | 15.96 ± 1.85b |
| 18% PEG | 0.79 ± 0.07c | 0.44 ± 0.05c | 1.15 ± 0.40c | 17.33 ± 3.78c | 14.38 ± 2.93c |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Kang, R.; Liu, Y.; Wu, K.-X.; Yan, X.; Song, Y.; Pan, L.-B.; Tang, Z.-H. Differential Metabolite Accumulation in Different Tissues of Gleditsia sinensis under Water Stress and Rehydration Conditions. Forests 2020, 11, 542. https://doi.org/10.3390/f11050542
Liu J, Kang R, Liu Y, Wu K-X, Yan X, Song Y, Pan L-B, Tang Z-H. Differential Metabolite Accumulation in Different Tissues of Gleditsia sinensis under Water Stress and Rehydration Conditions. Forests. 2020; 11(5):542. https://doi.org/10.3390/f11050542
Chicago/Turabian StyleLiu, Jia, Rui Kang, Yang Liu, Ke-Xin Wu, Xue Yan, Ying Song, Li-Ben Pan, and Zhong-Hua Tang. 2020. "Differential Metabolite Accumulation in Different Tissues of Gleditsia sinensis under Water Stress and Rehydration Conditions" Forests 11, no. 5: 542. https://doi.org/10.3390/f11050542
APA StyleLiu, J., Kang, R., Liu, Y., Wu, K.-X., Yan, X., Song, Y., Pan, L.-B., & Tang, Z.-H. (2020). Differential Metabolite Accumulation in Different Tissues of Gleditsia sinensis under Water Stress and Rehydration Conditions. Forests, 11(5), 542. https://doi.org/10.3390/f11050542






