Stand Structural Characteristics Are the Most Practical Biodiversity Indicators for Forest Management Planning in Europe
Abstract
1. Introduction
- (1)
- Biodiversity indicators in European forest research
- (2)
- Integrating biodiversity indicators into forest management
- (3)
- Practical data collection
- (4)
- Literature gaps and implications with forest management planning
2. Materials and Methods
2.1. Literature Review
- The research study was performed in Europe;
- Published in a peer-reviewed scientific journal;
- Written in English;
- The scale of research was stand or/and landscape;
- The focus was on forest biodiversity assessment;
- Biodiversity indicators and methods for data collection were clearly reported and extractable.
2.2. Data Extraction
2.3. Evaluation of Practicality
- Cost-effectiveness, i.e., what were the costs per hectare in Euros? How much workforce is required?
- Ease (simplicity) of application, i.e., are these indicators simple to use by forest managers with different backgrounds and can they identify the indicators (e.g., recognize the plant, animal species, or forest structural variables), and collect the data?
- Time-effectiveness, i.e., what is the time required for data gathering and assessment?
3. Biodiversity Indicators in European Forest Research
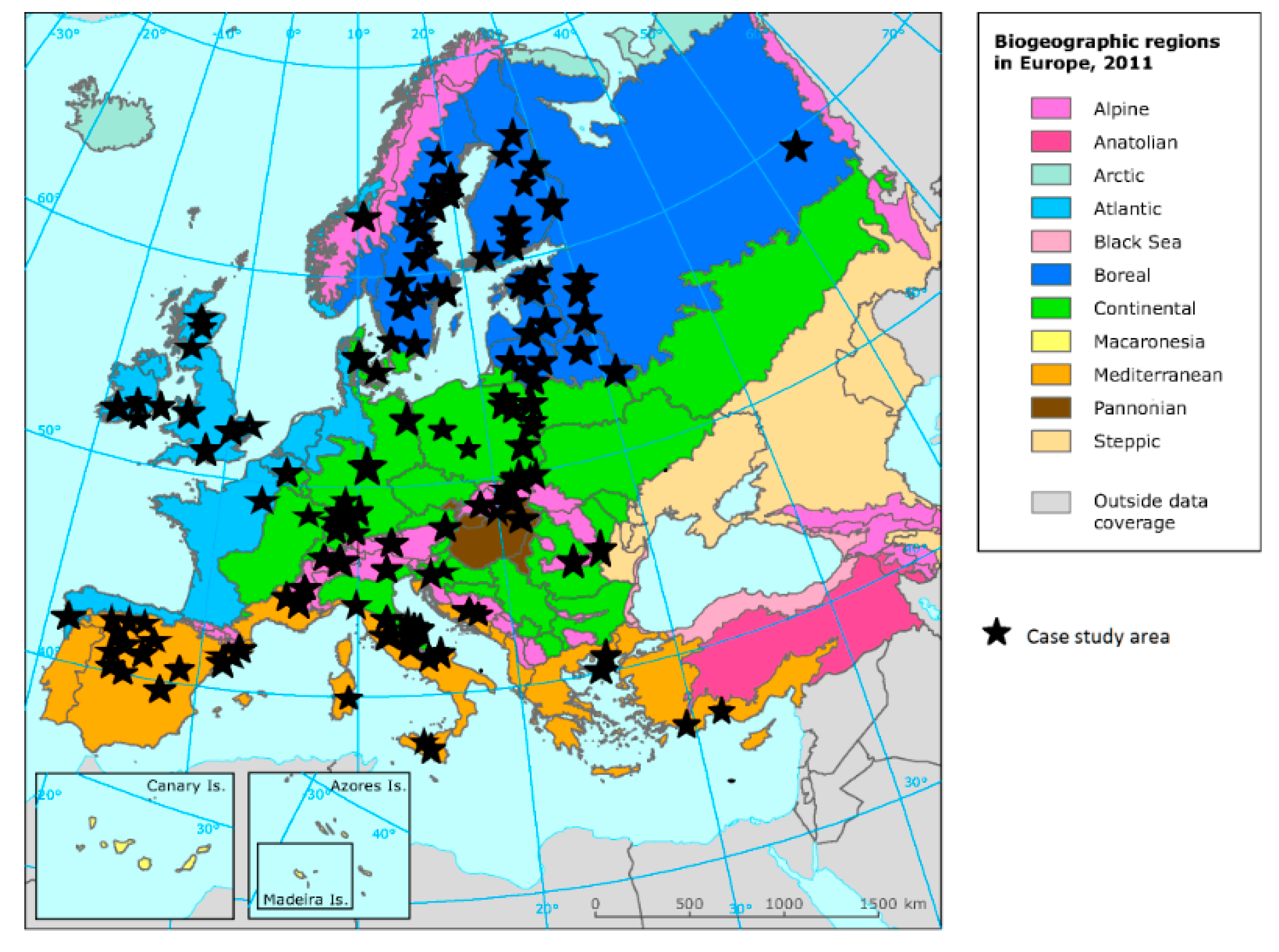
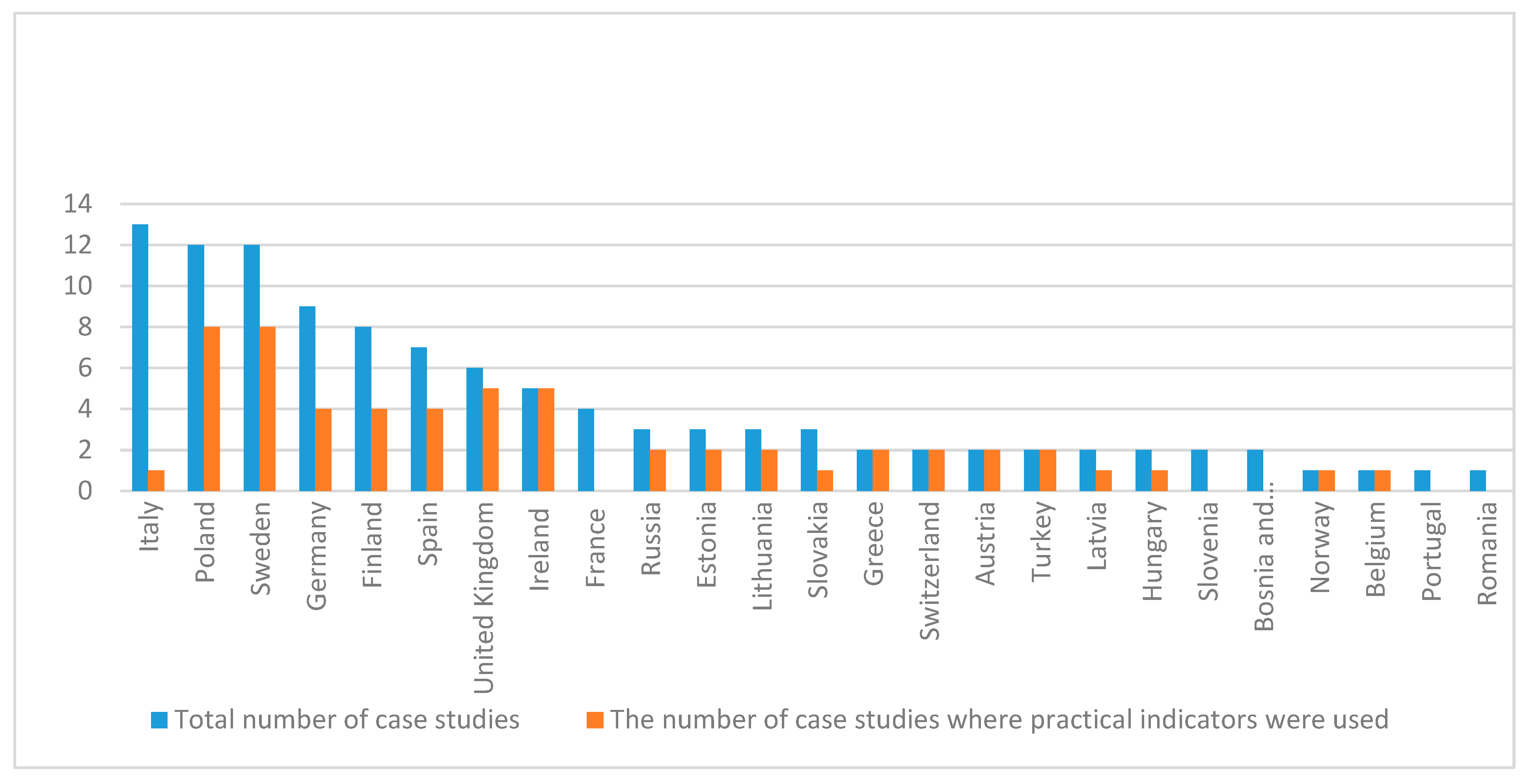
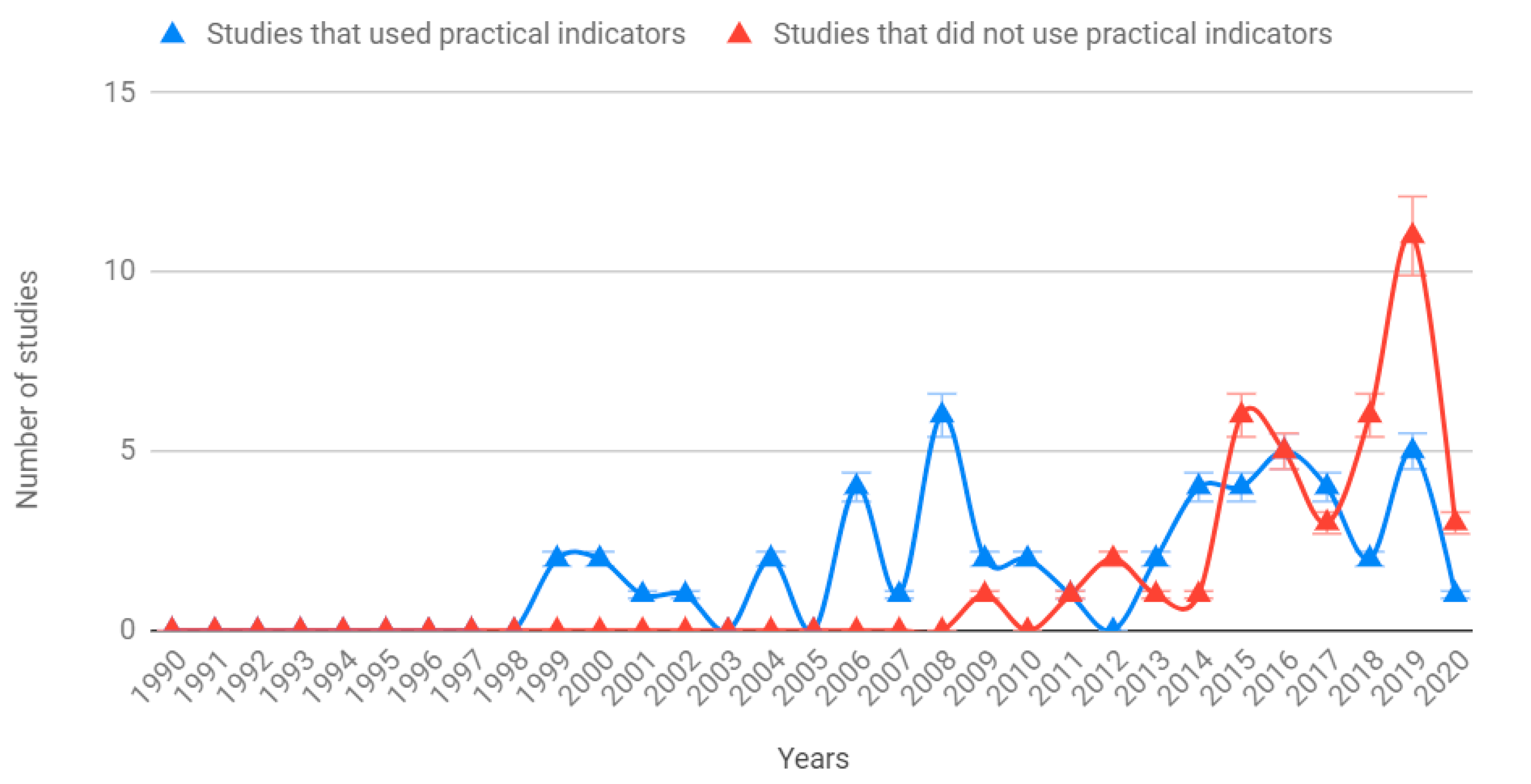
3.1. Geographic Distribution of Case Studies
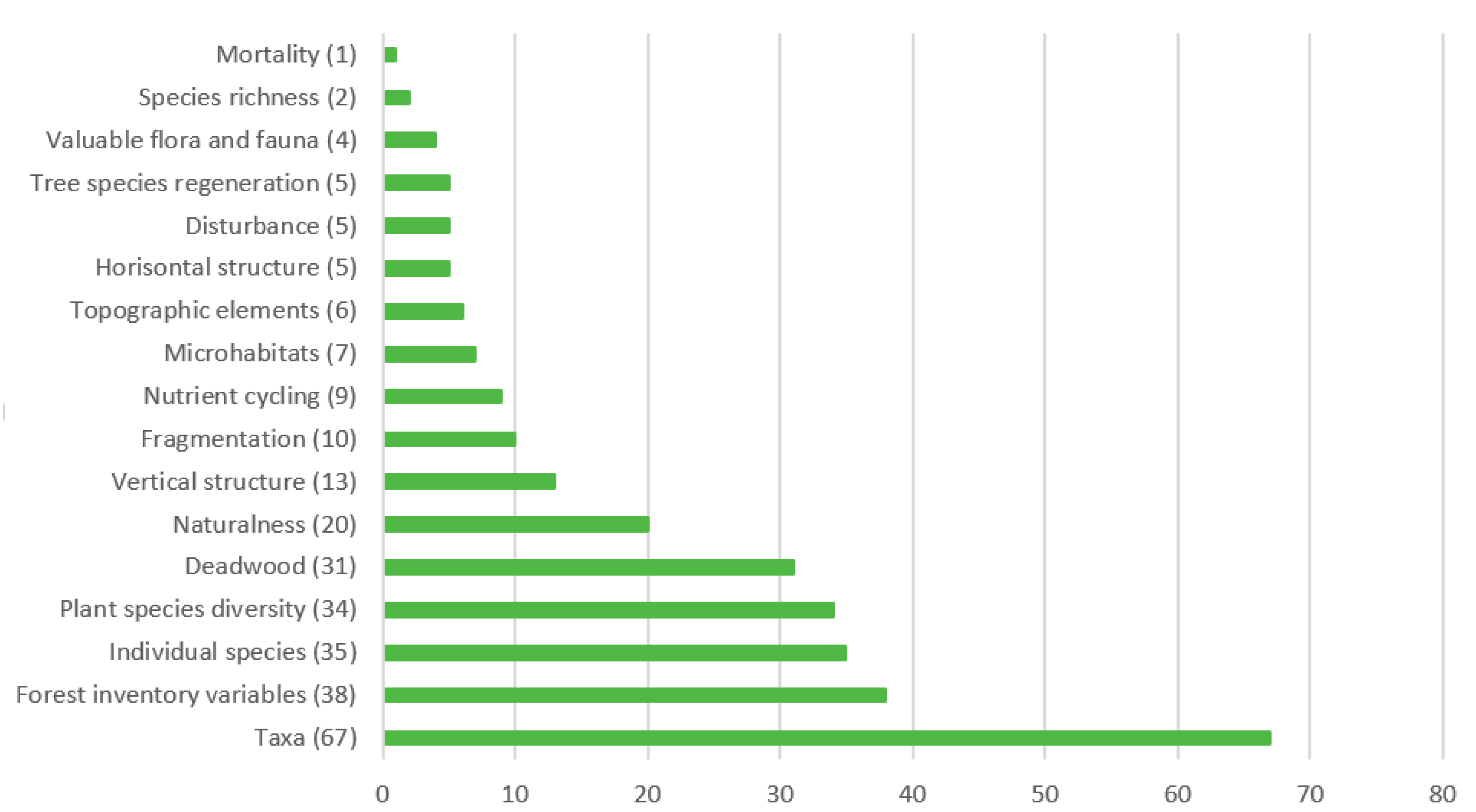
3.2. Forest Biodiversity Indicators Used in Studies
4. Integrating Biodiversity Indicators into Forest Management
4.1. The Practicality of Forest Biodiversity Indicators
4.1.1. Forest Species Composition
4.1.2. Forest Structure
4.1.3. Forest Ecosystem Functioning
4.1.4. Indicator and Umbrella Species
4.1.5. Correlation and Surrogacy
5. Practical Data Collection
5.1. Sampling Methods
5.2. National Forest Inventories (NFIs)
5.3. Flora and Fauna Atlases
5.4. Remote Sensing
5.5. Camera Traps
5.6. Smartphone Applications
6. Literature Gaps and Implications for Forest Management Planning
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kapos, V.; Iremonger, S. Achieving global and regional perspectives on forest biodiversity and conservation. In Assessment of Biodiversity for Improved Forest Planning; Bachman, P., Kohl, M., Paivinen, R., Eds.; Springer: Dordrecht, The Netherlands, 1998; pp. 3–14. [Google Scholar]
- Coote, L.; Dietzsch, A.C.; Wilson, M.W.; Graham, C.T.; Fuller, L.; Walsh, A.T.; Irwin, S.; Kelly, D.L.; Mitchell, F.J.; Kelly, T.C.; et al. Testing indicators of biodiversity for plantation forests. Ecol. Ind. 2013, 32, 107–115. [Google Scholar] [CrossRef]
- Ozanne, C.H.P.; Anhuf, D.; Boulter, S.L.; Keller, M.; Kitching, R.L.; Körner, C.; Meinzer, F.C.; Mitchell, A.W.; Nakashizuka, T.; Silva Dias, P.L.; et al. Biodiversity meets the atmosphere: a global view of forest canopies. Science 2003, 301, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Hedblom, M.; Emilsson, T.; Nielsen, A.B. The role of forest stand structure as biodiversity indicator. For. Ecol. Manage. 2014, 330, 82–93. [Google Scholar] [CrossRef]
- CBD, Convention on Biological Diversity 2000. Available online: https://www.cbd.int/convention/guide/?id=web (accessed on 28 January 2020).
- Hunter, M.L., Jr. Wildlife, forests, and forestry: Principles of managing forests for biological Diversity; Prentice-Hall: Englewood Cliffs, NJ, USA, 1990; p. 370. [Google Scholar]
- Winter, S.; McRoberts, R.E.; Bertini, R.; Chirici, G.; Bastrup-Birk, A.; Rondeux, J.; Brändli, U.B.; Marchetti, M. The Need for Harmonized Estimates of Forest biodiversity Indicators. In National Forest Inventories: Contributions to Forest Biodiversity Assessments; Springer: Dordrecht, The Netherlands, 2011. [Google Scholar]
- FAO. The State of World’s Forests. 2018. Available online: http://www.fao.org/state-of-forests/en/ (accessed on 27 July 2019).
- Forest Europe. State of Europe’s forests 2015. In Proceedings of the Ministerial Conference on the Protection of Forests in Europe, Madrid, Spain, 20–1 October 2015. [Google Scholar]
- Peterken, G. Natural Woodland: Ecology and Conservation in Northern Temperate Regions; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Anonymous. Decision No 1600/2002/EC of the European parliament and of the council of 22 July 2002 laying down the Sixth Community Environment Action Programme. Off. J. Eur. Communities 2002, 10. [Google Scholar]
- Angelstam, P.; Dönz-Breuss, M. Measuring Forest Biodiversity at the Stand Scale: An Evaluation of Indicators in European Forest History Gradients. Ecol. Bull. 2004, 51, 305–332. [Google Scholar]
- Leadley, P.; Pereira, H.M.; Alkemade, R.; Fernandez-Manjarres, J.F.; Proenca, V.; Scharlemann, J.P.W.; Walpole, M.J. Biodiversity scenarios: projections of 21st century change in biodiversity and associated ecosystem services. Tech. Ser. 50 2010, 132. [Google Scholar]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15.4, 365–377. [Google Scholar] [CrossRef]
- MEA-Millennium Ecosystem Assessment. 2005. Available online: https://millenniumassessment.org/en/Framework.html (accessed on 1 May 2019).
- Proença, V.M.; Pereira, H.M.; Guilherme, J.; Vicente, J. Plant and bird diversity in natural forests and in native and exotic plantations in NW Portugal. Acta Oecol. 2010, 36, 219–226. [Google Scholar] [CrossRef]
- Anonymous. Criteria and Indicators for the Conservation and Sustainable Management of Temperate and Boreal Forests; The Montreal process; Canadian Forest Service: Quebec, Canada, 1995.
- Ferris-Kaan, R.; Peace, A.J.; Humphrey, J.W. Assessing structural diversity in managed forests. In Assessment of Biodiversity for Improved Forest Planning. European Forest Institute Proceedings No. 18; Bachmann, P., Kohl, M., Paivinen, R., Eds.; Springer: Dordrecht, The Netherlands, 1998; pp. 331–342. [Google Scholar]
- Heywood, V.H.; Baste, I. Introduction. In Global Biodiversity Assessment. United Nations Environment Programme; Heywood, V.H., Ed.; Cambridge University Press: Cambridge, UK, 1995; pp. 1–2. [Google Scholar]
- Ferris, R.; Peace, A.J.; Newton, A.C. Macrofungal communities of lowland Scots pine (Pinus sylvestris L.) and Norway spruce (Picea abies (L.) Karsten.) plantations in England: relationships with site factors and stand structure. For. Ecol. Manage. 2000, 131, 255–267. [Google Scholar] [CrossRef]
- Noss, R.F. Indicators for monitoring biodiversity: A hierarchical approach. Conserv. Biol. 1990, 4, 355–364. [Google Scholar] [CrossRef]
- Morelli, F. Indicator species for avian biodiversity hotspots: Combination of specialists and generalists is necessary in less natural environments. J. Nat. Cons. 2015, 27, 54–62. [Google Scholar] [CrossRef]
- Boutin, S.; Haughland, D.L.; Schieck, J.; Herbers, J.; Bayne, E. A new approach to forest biodiversity monitoring in Canada. For. Ecol. Manage. 2009, 258, S168–S175. [Google Scholar] [CrossRef]
- Ferris, R.; Humphrey, J.W. A review of potential biodiversity indicators for application in British forests. Forestry 1999, 72, 313–328. [Google Scholar] [CrossRef]
- Smith, G.F.; Gittings, T.; Wilson, M.; French, L.; Oxbrough, A.; O’Donoghue, S.; Iremonger, S. Identifying practical indicators of biodiversity for stand-level management of plantation forests. Biodiv. Cons. 2008, 17, 991–1015. [Google Scholar] [CrossRef]
- Noss, R.F. Assessing and monitoring forest biodiversity: A suggested framework and indicators. For. Ecol. Manage. 1999, 115, 135–146. [Google Scholar] [CrossRef]
- Lindenmayer, D.B.; Margules, C.R.; Botkin, D.B. Indicators of biodiversity for ecologically sustainable forest management. Cons. Bio. 2000, 14, 941–950. [Google Scholar] [CrossRef]
- Failing, L.; Gregory, R. Ten common mistakes in designing biodiversity indicators for forest policy. J. Environ. Manage. 2003, 68, 121–132. [Google Scholar] [CrossRef]
- Whitman, A.A.; Hagan, J.M. Final Report to the National Commission on Science For sustainable Forestry: Biodiversity Indicators for Sustainable Forestry; National Center for Science and the Environment: Washington, DC, USA, 2003; 48p. [Google Scholar]
- Heink, U.; Kowarik, I. What are indicators? On the definition of indicators in ecology and environmental planning. Ecol. Ind. 2010, 10, 584–593. [Google Scholar] [CrossRef]
- Gao, T.; Nielsen, A.B.; Hedblom, M. Reviewing the strength of evidence of biodiversity indicators for forest ecosystems in Europe. Ecol. Ind. 2015, 57, 420–434. [Google Scholar] [CrossRef]
- Ezquerro, M.; Pardos, M.; Diaz-Balteiro, L. Operational research techniques used for addressing biodiversity objectives into forest management: an overview. Forests 2016, 7, 229. [Google Scholar] [CrossRef]
- Lexer, M.J.; Lexer, W.; Hasenauer, H. The use of forest models for biodiversity assessment at the stand level. For. Syst. 2000, 9, 297–316. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Watson, M. Guidance on Conducting a Systematic Literature Review. J. Plann. Edu. Rese. 2017, 39, 1–20. [Google Scholar] [CrossRef]
- Parisi, F.; Pioli, S.; Lombardi, F.; Fravolini, G.; Marchetti, M.; Tognetti, R. Linking deadwood traits with saproxylic invertebrates and fungi in European forests—A review. iForest 2018, 11, 423–436. [Google Scholar] [CrossRef]
- Winter, S.; Fischer, H.S.; Fischer, A. Relative quantitative reference approach for naturalness assessments of forests. For. Ecol. Manage. 2010, 259, 1624–1632. [Google Scholar] [CrossRef]
- Bani, A.; Pioli, S.; Ventura, M.; Panzacchi, P.; Borruso, L.; Tognetti, R.; Tonon, G.; Brusetti, L. The role of microbial community in the decomposition of leaf litter and deadwood. Appl. Soil Ecol. 2018, 126, 75–84. [Google Scholar] [CrossRef]
- Parisi, F.; Di Febbraro, M.; Lombardi, F.; Biscaccianti, A.B.; Campanaro, A.; Tognetti, R.; Marchetti, M. Relationships between stand structural attributes and saproxylic beetle abundance in a Mediterranean broadleaved mixed forest. For. Ecol. Manage. 2019, 432, 957–966. [Google Scholar] [CrossRef]
- Müller, M.; Brand, R. Assessing biodiversity by remote sensing in mountainous terrain: The potential of LiDAR to predict forest beetle assemblages. J. Appli. Ecol. 2009, 46, 897–905. [Google Scholar] [CrossRef]
- Pesonen, A.; Kangas, A.; Maltamo, M.; Packalen, P. Effects of auxiliary data source and inventory unit size on the efficiency of sample-based coarse woody debris inventory. For. Ecol. Manage. 2010, 259, 1890–1899. [Google Scholar] [CrossRef]
- Standovár, T.; Szmorad, F.; Kovács, B.; Kelemen, K.; Plattner, M.; Roth, T.; Pataki, Z. A novel forest state assessment methodology to support conservation and forest management planning. Commun. Ecol. 2016, 17, 167–177. [Google Scholar] [CrossRef]
- Kangas, A.; Rasinmäk, J.; Eyvindson, K.; Chambers, P. A Mobile Phone Application for the Collection of Opinion Data for Forest Planning Purposes. Envir. Manage. 2015, 55, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Güthlin, D.; Storch, I.; Küchenhoff, H. Toward Reliable Estimates of Abundance: Comparing Index Methods to Assess the Abundance of a Mammalian Predator. PLoS ONE 2014, 9, e94537. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, I.; Mert, A.; Ozkan, U.Y.; Aksan, S.; Unal, Y. Predicting bird species richness and micro-habitat diversity using satellite data. For. Ecol. Manage. 2018, 424, 483–493. [Google Scholar] [CrossRef]
- Mullen, K.; O’Halloran, J.; Breen, J.; Giller, P.; Pithon, J.; Kelly, T. Distribution and composition of carabid beetle (Coleoptera, Carabidae) communities across the plantation forest cycle—Implications for management. For. Ecol. Manage. 2008, 256, 624–632. [Google Scholar] [CrossRef]
- Simonson, S.E.; Opler, P.A.; Stohlgren, T.J.; Chong, G.W. Rapid assessment of butterfly diversity in a montane landscape. Biodiv. Conserv. 2001, 10, 1369–1386. [Google Scholar] [CrossRef]
- Sauberer, N.; Zulka, K.P.; Abensperg-Traun, M.; Berg, H.M.; Bieringer, G.; Milasowszky, N.; Moser, D.; Plutzar, C.; Pollheimer, M.; Storch, C.; et al. Surrogate taxa for biodiversity in agricultural landscapes of eastern Austria. Biol. Conserv. 2004, 117, 181–190. [Google Scholar] [CrossRef]
- Bräuniger, C.; Knapp, S.; Kühn, I.; Klotz, S. Testing taxonomic and landscape surrogates for biodiversity in an urban setting. Landsc. Urban Plan. 2010, 97, 283–295. [Google Scholar] [CrossRef]
- Ratcliffe, P.R. Biodiversity in Britain’s Forests; Forestry Authority: Edinburgh, Scotland, 1993.
- Arsenault, A.; Bradfield, G.E. Structural-compositional variation in three age-classes of temperate rainforests in southern coastal British Columbia. Can. J. Bot. 1995, 73, 54–64. [Google Scholar] [CrossRef]
- Humphrey, J.W.; Hawes, C.; Peace, A.J. Relationships between insect diversity and habitat characteristics in plantation forests. For. Ecol. Manage. 1999, 113, 11–21. [Google Scholar] [CrossRef]
- Michel, A.K.; Winter, S. Tree microhabitat structures as indicators of biodiversity in Douglas-fir forests of different stand ages and management histories in the Pacific Northwest. USA. For. Ecol. Manage. 2009, 257, 1453–1464. [Google Scholar] [CrossRef]
- Chirici, G.; McRoberts, R.E.; Winter, S.; Bertini, R.; Brändli, U.B.; Asensio, I.A.; Bastrup-Birk, A.; Rondeux, J.; Barsoum, N.; Marchetti, M. National forest inventory contributions to forest biodiversity monitoring. For. Sci. 2012, 58, 257–268. [Google Scholar] [CrossRef]
- Juutinen, A.; Mönkkönen, M. Testing alternative indicators for biodiversity conservation in old-growth boreal forests: ecology and economics. Ecol. Econom. 2004, 50, 35–48. [Google Scholar] [CrossRef]
- Thingstad, P.G.; Daverdin, M.; Speed, J.D.M. Modelling Capercaillie Tetrao urogallus lek sites distribution and their limiting factors in a central Norwegian managed forest landscape. Ornis Norv. 2018, 41, 1–12. [Google Scholar] [CrossRef]
- Uliczka, H.; Angelstam, P.; Roberge, J.M. Indicator species and biodiversity monitoring systems for non-industrial private forest owners: is there a communication problem? Ecol. Bull. 2004, 51, 379–384. [Google Scholar]
- Naumov, V.; Manton, M.; Elbakidze, M.; Rendenieks, Z.; Priednieks, J.; Uhlianets, S.; Angelstam, P. How to reconcile wood production and biodiversity conservation? The Pan-European boreal forest history gradient as an “experiment”. J. Envir. Manage. 2018, 218, 1–13. [Google Scholar] [CrossRef]
- Thers, H.; Brunbjerg, A.K.; Læssøe, T.; Ejrnæs, R.; Bøcher, P.K.; Svenning, J.C. Lidar-derived variables as a proxy for fungal species richness and composition in temperate Northern Europe. Rem. Sens. Envir. 2017, 200, 102–113. [Google Scholar] [CrossRef]
- Kirk, P.M.; Cannon, P.F.; Minter, D.W.; Stalpers, J.A. Dictionary of the Fungi, 10th ed.; CAB Internation Wallingford: Wallingford, UK, 2008; p. 771. [Google Scholar]
- Ambrosio, E.; Mariotti, M.G.; Zotti, M.; Cecchi, G.; Di Piazza, S.; Feest, A. Measuring macrofungal biodiversity quality using two different survey approaches: A case study in broadleaf Mediterranean forests. Ecol. Ind. 2018, 85, 1210–1230. [Google Scholar] [CrossRef]
- Barsoum, N.; Bruce, C.; Forster, J.; Ji, Y.Q.; Yu, D.W. The devil is in the detail: Metabarcoding of arthropods provides a sensitive measure of biodiversity response to forest stand composition compared with surrogate measures of biodiversity. Ecol. Ind. 2019, 101, 313–323. [Google Scholar] [CrossRef]
- Torras, O.; Saura, S. Effects of silvicultural treatments on forest biodiversity indicators in the Mediterranean. For. Ecol. Manage. 2008, 255, 3322–3330. [Google Scholar] [CrossRef]
- Hodge, S.J.; Peterken, G.F. Deadwood in British forests: priorities and a strategy. Forestry 1998, 71, 99–112. [Google Scholar] [CrossRef]
- Fridman, J.; Walheim, M. Amount, structure, and dynamics of dead wood on managed forestland in Sweden. For. Ecol. Manage. 2000, 131, 23–36. [Google Scholar] [CrossRef]
- Martín-Queller, E.; Gil-Tena, A.; Saura, S. Species richness of woody plants in the landscapes of Central Spain: the role of management disturbances, environment and non-stationarity. J. Veg. Sci. 2011, 2, 238–250. [Google Scholar] [CrossRef]
- Schindler, S.; von Wehrden, H.; Poirazidis, K.; Wrbka, T.; Kati, V. Multiscale performance of landscape metrics as indicators of species richness of plants, insects and vertebrates. Ecol. Ind. 2013, 31, 41–48. [Google Scholar] [CrossRef]
- Romero-Calcerrada, R.; Luque, S. Habitat quality assessment using Weights-of-Evidence based GIS modelling: The case of Picoides tridactylus as species indicator of the biodiversity value of the Finnish forest. Ecol. Modell. 2006, 196, 62–76. [Google Scholar] [CrossRef]
- Day, K.R.; Carthy, J. Changes in carabid beetle communities accompanying a rotation of Sitka spruce. Agric. Ecosys. Environ. 1988, 24, 407–415. [Google Scholar] [CrossRef]
- Thiele, H.U. Carabid Beetles in their Environments: A Study on Habitat Selection by Adaptations in Physiology and Behaviour; Springer: Berlin, Germany, 1997; p. 369. [Google Scholar]
- Butterfield, J. Carabid community succession during the forestry cycle in conifer plantations. Ecography 1997, 20, 614–625. [Google Scholar] [CrossRef]
- Vanbergen, A.J.; Woodcock, B.A.; Watt, A.D. Effect of land-use heterogeneity on carabid communities at the landscape scale. Ecography 2005, 28, 3–16. [Google Scholar] [CrossRef]
- Gregory, R.D.; Vořišek, P.; Noble, D.G.; Van Strien, A.; Klvaňová, A.; Eaton, M.; Burfield, I.J. The generation and use of bird population indicators in Europe. Bird Conserv. Internat. 2008, 18, 223–244. [Google Scholar] [CrossRef]
- Carrillo-Rubio, E.; Kery, M.; Morreale, S.J. Use of multispecies occupancy models to evaluate the response of bird communities to forest degradation associated with logging. Conserv. Bio. 2014, 28, 1034–1044. [Google Scholar] [CrossRef]
- Czeszczewik, D.; Zub, K.; Stanski, T.; Sahel, M.; Kapusta, A.; Walankiewicz, W. Effects of forest management on bird assemblages in the Bialowieza Forest, Poland. iForest 2014, 8, 377–385. Available online: http://www.sisef.it/iforest/contents/?id=ifor1212-007 (accessed on 9 January 2019). [CrossRef]
- Basile, M.; Valerio, F.; Balestrieri, R.; Posillico, M.; Bucci, R.; Altea, T.; Matteucci, G. Patchiness of forest landscape can predict species distribution better than abundance: the case of a forest-dwelling passerine, the short-toed treecreeper, in central Italy. PeerJ 2016, 4, e2398. [Google Scholar] [CrossRef] [PubMed]
- Feest, A. The Utility of the Streamlining European Biodiversity Indicators 2010 (SEBI 2010). Ecol. Ind. 2013, 28, 16–21. [Google Scholar] [CrossRef]
- Mosimann, P.; Naef-Daenzer, B.; Blattner, M. Die Zusammensetzung der Avifauna in typischen Waldgesellschaften der Schweiz. D. Ornitholog. Beobachter 1987, 84, 275–299. [Google Scholar]
- Müller-Buser, M. Die Avifauna der Bergföhrenwälder des Schweizerischen Nationalparks im Ofenpassgebiet. D. Ornitholog. Beobachter 2002, 99, 1–18. [Google Scholar]
- Bujoczek, M.; Rybicka, J.; Bujoczek, L. Effects of disturbances in a subalpine forest on its structural indicators and bird diversity. Ecol. Ind. 2020, 112, 106126. [Google Scholar] [CrossRef]
- Angelstam, P.; Dönz-Breuss, M.; Roberge, J.M. Targets and tools for the maintenance of forest biodiversity: An introduction. Ecol. Bull. 2004, 51, 11–24. [Google Scholar]
- Lešo, P.; Kropil, R.; Kajtoch, Ł. Effects of Forest Management on Bird Assemblages in Oak-Dominated Stands of the Western Carpathians—Refuges for Rare Species. For. Ecol. Manage. 2019, 453, 117620. [Google Scholar] [CrossRef]
- Hilty, J.; Merenleder, A. Faunal indicator taxa selection for monitoring ecosystem health. Bio. Conser. 2000, 92, 185–197. [Google Scholar] [CrossRef]
- Hawskworth, D.L.; Rose, F. Qualitative scale for estimating sulphur dioxide in England and Wales using epiphytic lichens. Nature 1970, 227, 145–148. [Google Scholar]
- Block, W.M.; Brennan, L.A.; Gutiérrez, R.J. The use of guilds and guild-indicators for assessing habitat suitability. In Wildlife; Verner, J., Morrison, M.L., Ralph, C.J., Eds.; The University of Wisconsin Press: Madisonm, WI, USA, 1996; pp. 109–113. [Google Scholar]
- Furness, R.W.; Greenwood, J.J.D. (Eds.) Birds as Monitors of Environmental Change; Chapman & Hall: London, UK, 1993. [Google Scholar]
- Mikusiński, G.; Gromadzki, M.; Chylarecki, P. Woodpeckers as indicators of forest bird diversity. Conserv. Biol. 2001, 15, 208–217. [Google Scholar] [CrossRef]
- Roberge, J.-M.; Angelstam, P.; Villard, M.-A. Specialised woodpeckers and naturalness in hemiboreal forests—Deriving quantitative targets for conservation planning. Bio. Conserv. 2008, 141, 997–1012. [Google Scholar] [CrossRef]
- Roberge, J.-M.; Mikusinski, G.; Svensson, S. The white-backed woodpecker: umbrella species for forest conservation planning? Biodiv. Conserv. 2008, 17, 2479–2494. [Google Scholar] [CrossRef]
- Gittings, T.; O’Halloran, J.; Kelly, T.; Giller, P.S. The Contribution of Open Spaces to the Maintenance of Hoverfly (Diptera, Syrphidae) Biodiversity in Irish Plantation Forests. For. Ecol. Manage. 2006, 237, 290–300. [Google Scholar] [CrossRef]
- Rodriguez-Prieto, I.; Bennett, V.J.; Zollner, P.A.; Mycroft, M.; List, M.; Fernández-Juricic, E. Simulating the responses of forest bird species to multi-use recreational trails. Landsc. Urban. Plan. 2014, 127, 164–172. [Google Scholar] [CrossRef]
- Pakkala, T.; Linden, A.; Tiainen, J.; Tomppo, E.; Kouki, J. Indicators of forest biodiversity: which bird species predict high breeding bird assemblage diversity in boreal forests at multiple spatial scales? Ann. Zool. Fenn. 2014, 51, 457–476. [Google Scholar] [CrossRef]
- Vangansbeke, P.; Blondeel, H.; Landuyt, D.; De Frenne, P.; Gorissen, L.; Verheyen, K. Spatially combining wood production and recreation with biodiversity conservation. Biodiv. Conserv. 2017, 26, 3213–3239. [Google Scholar] [CrossRef]
- Fleishman, E.; Murphy, D.D.; Brussard, P.F. A new method for selection of umbrella species for conservation planning. Ecol. Appl. 2000, 10, 569–579. [Google Scholar] [CrossRef]
- Suter, W.; Graf, R.F.; Hess, R. Capercaillie (Tetrao urogallus) and Avian Biodiversity: Testing the Umbrella-Species Concept. Cons. Bio. 2002, 16, 3. [Google Scholar] [CrossRef]
- Roberge, J.-M.; Angelstam, P. Usefulness of the umbrella species concept as a conservation tool. Cons. Bio. 2004, 18, 76–85. [Google Scholar] [CrossRef]
- Mikoláš, M.; Svitok, M.; Tejkal, M.; Leitão, P.J.; Morrissey, R.C.; Svoboda, M.; Fontaine, J.B. Evaluating forest management intensity on an umbrella species: Capercaillie persistence in central Europe. For. Ecol. Manage. 2015, 354, 26–34. [Google Scholar] [CrossRef]
- Niemi, G.J.; Hanowski, J.M.; Lima, A.R.; Nicholls, T.; Weiland, N. A critical analysis on the use of indicator species in management. J. Wildl. Manage. 1997, 61, 1240–1252. [Google Scholar] [CrossRef]
- Prendergast, J.R.; Eversham, B.C. Species richness covariance in higher taxa: empirical tests of the biodiversity indicator concept. Ecography 1997, 20, 210–216. [Google Scholar] [CrossRef]
- Landres, P.B.; Verner, J.; Thomas, J.W. Ecological uses of vertebrate indicator species: A critique. Cons. Bio. 1988, 2, 316–328. [Google Scholar] [CrossRef]
- Lindenmayer, D.B. Future directions for biodiversity conservation in managed forests: Indicator species, impact studies and monitoring programs. For. Ecol. Manage. 1999, 115, 277–287. [Google Scholar] [CrossRef]
- Sabatini, F.M.; Burrascano, S.; Azzella, M.M.; Barbati, A.; De Paulis, S.; Di Santo, D.; Facionia, L.; Giuliarelli, D.; Lombardi, F.; Maggi, O.; et al. One taxon does not fit all: Herb-layer diversity and stand structural complexity are weak predictors of biodiversity in Fagus sylvatica forests. Ecol. Ind. 2016, 69, 126–137. [Google Scholar] [CrossRef]
- Larrieu, L.; Gosselin, F.; Archaux, F.; Chevalier, R.; Corriol, G.; Dauffy-Richard, E.; Deconchat, M.; Gosselin, M.; Ladet, S.; Savoie, J.-M.; et al. Cost-efficiency of cross-taxon surrogates in temperate forests. Ecol. Ind. 2018, 87, 56–65. [Google Scholar] [CrossRef]
- Jones, K.B.; Neale, A.C.; Nash, M.S. Predicting nutrient and sediment loadings to streams from landscape metrics: A multiple watershed study from the United States Mid-Atlantic Region. Lands. Ecol. 2001, 16, 301–312. [Google Scholar] [CrossRef]
- Li, H.; Wu, J. Use and misuse of landscape metrics. Lands. Ecol. 2004, 19, 389–399. [Google Scholar] [CrossRef]
- Motz, K.; Sterba, H.; Pommerening, A. Sampling measures of tree diversity. For. Ecol. Manage. 2010, 260, 1985–1996. [Google Scholar] [CrossRef]
- Karahalil, U.; Başkent, E.Z.; Sivrikaya, F.; Kılıç, B. Analyzing deadwood volume of Calabrian pine (Pinus brutia Ten.) in relation to stand and site parameters: a case study in Köprülü Canyon National Park. Environ. Monit. Assess. 2017, 189, 112. [Google Scholar] [CrossRef]
- Warren, W.G.; Olsen, P.F. A line intersect technique for assessing logging waste. For. Sci. 1964, 10, 267–276. [Google Scholar]
- Polit, J.-I.; Brown, S. Mass and nutrient content of deadwood in a central Illinois floodplain forest. Wetlands 1996, 16, 488–494. [Google Scholar] [CrossRef]
- Luque, S.; Riutta, T.; Joensuu, J.; Rautjarvi, N.; Tomppo, E. Multi-source forest inventory data for biodiversity monitoring and planning at the forest landscape level. In Monitoring and Indicators of Forest Biodiversity in Europe—From Ideas to Operationality; Marchetti, M., Ed.; EFI, European Forest Institute: Joensuu, Finland, 2004; proceedings no. 51; pp. 430–444. [Google Scholar]
- Luque, S.; Riutta, T.; Joensuu, J.; Tomppo, E. Spatial analysis and remote sensing for biodiversity monitoring and planning at the forest landscape level. In Actes Hermes-science CASSINI’04, 7eme Conference GDR SIGMA; Geomatique et Analyse Spatiale: Grenoble, France, 2004; pp. 149–155. [Google Scholar]
- Roberge, J.-M.; Lämås, T.; Lundmark, T.; Ranius, T.; Felton, A.; Nordin, A. Relative contributions of set-asides and tree retention to the long-term availability of key forest biodiversity structures at the landscape scale. J. Environ. Manage. 2015, 154, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Bottalico, F.; Chirici, G.; Giannini, R.; Mele, S.; Mura, M.; Puxeddu, M.; Travaglini, D. Modeling Mediterranean forest structure using airborne laser scanning data. Internat. J. App. Earth Observ. Geoinfo. 2017, 57, 145–153. [Google Scholar] [CrossRef]
- Treinys, R.; Mozgeris, G.; Skuja, S. Can intensified forestry be responsible for changes in habitat usage by the forest-dwelling Black Stork? Eur. J. Forest Res. 2016, 135, 1175–1186. [Google Scholar] [CrossRef]
- Suchant, R.; Braunisch, V. Multidimensional habitat modelling in forest management—A case study using capercaillie in the Black Forest, Germany. Ecol. Bull. 2004, 51, 455–469. [Google Scholar]
- Lõhmus, A.; Nellis, R.; Pullerits, M.; Leivits, M. The Potential for Long-Term Sustainability in Seminatural Forestry: A Broad Perspective Based on Woodpecker Populations. Environ. Manage. 2016, 57, 558–571. [Google Scholar] [CrossRef]
- Lindberg, E.; Roberge, J.-M.; Johansson, T.; Hjältén, J. Can Airborne Laser Scanning (ALS) and Forest Estimates Derived from Satellite Images Be Used to Predict Abundance and Species Richness of Birds and Beetles in Boreal Forest? Remote Sens. 2015, 7, 4233–4252. [Google Scholar] [CrossRef]
- Mura, M.; McRoberts, R.E.; Chirici, G.; Marchetti, M. Estimating and mapping forest structural diversity using airborne laser scanning data. Remote Sens. Environ. 2015, 170, 133–142. [Google Scholar] [CrossRef]
- Tracz, W.; Ciurzycki, W.; Zaniewski, P.; Kwaśny, Ł.; Marciszewska, K.; Mozgawa, J. Identification of Zones with High Potential for Biological Diversity on Dormant Forested Landslides. Eur. J. Forest Res. 2019, 138, 363–373. [Google Scholar] [CrossRef]
- Lefsky, M.A.; Cohen, W.B.; Parker, G.G.; Harding, D.J. Lidar remote sensing for ecosytem studies. BioScience 2002, 52, 19–30. [Google Scholar] [CrossRef]
- MacKerron, G.; Mourota, S. Happiness is greater in natural environments. Glob. Environ. Change 2013, 23, 992–1000. [Google Scholar] [CrossRef]
- Pereira, H.M.; Cooper, H.D. Towards the global monitoring of biodiversity change. Tren. Ecol. Evol. 2006, 21, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.P.; Collen, B.E.N.; Atkinson, G.; Baxter, P.W.; Bubb, P.; Illian, J.B.; Nicholson, E. The Why, What, and How of Global Biodiversity Indicators Beyond the 2010 Target. Cons. Bio. 2011, 25, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Feest, A.; Aldred, T.D.; Jedamzik, K. Biodiversity Quality: A Paradigm for Biodiversity. Ecol. Ind. 2010, 10, 1077–1082. [Google Scholar] [CrossRef]
- Hooper, D.U.; Chapin, F.S., III; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monographs 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Bobbink, R.; Hornung, M.; Roelofs, J.G.M. The effects of air-borne nitrogen pollutants on species diversity in natural and semi-natural European vegetation. J. Ecol. 1998, 86, 717–738. [Google Scholar] [CrossRef]
- Feest, A. Research to Test the Integration of the Nitrogen Critical Load Exceedence Model (EG4) into the 2010 Target by Linking it to the Butterfly Indicator (EG1); European Environment Agency: Copenhagen, Denmark, 2008; p. 34.
- Maskell, L.C.; Smart, S.M.; Bullock, J.M.; Thompson, K.; Stevens, C.J. Nitrogen deposition causes widespread loss of species richness in British Habitats. Glob. Change Biol. 2010, 16, 671–679. [Google Scholar] [CrossRef]
- Stevens, C.J.; Dise, N.B.; Mountford, J.O.; Gowing, D.J. Impact of nitrogen deposition on the species richness of grasslands. Science 2004, 303, 1876–1879. [Google Scholar] [CrossRef]
- Dirnböck, T.; Grandin, U.; Bernhardt-Römermann, M.; Beudert, B.; Canullo, R.; Forsius, M.; Grabner, M.-T.; Holmberg, M.; Kleemola, S.; Lundin, L.; et al. Forest floor vegetation response to nitrogen deposition in Europe. Glob. Change Biol. 2014, 20, 429–440. [Google Scholar] [CrossRef]
- Miina, J.; Hallikainen, V.; Härkönen, K.; Merilä, P.; Packalen, T.; Rautio, P.; Salemaa, M.; Tonteri, T.; Tolvanen, A. Incorporating a Model for Ground Lichens into Multi-Functional Forest Planning for Boreal Forests in Finland. For. Ecol. Manage. 2020, 460, 117912. [Google Scholar] [CrossRef]
- Groom, G.; Mücher, C.A.; Ihse, M.; Wrbka, T. Remote sensing in landscape ecology: experiences and perspectives in a European context. Lands. Ecol. 2006, 21, 391–408. [Google Scholar] [CrossRef]
- Schindler, S.; Poirazidis, K.; Wrbka, T. Towards a core set of landscape metrics for biodiversity assessments: A case study from Dadia National Park, Greece. Ecol. Ind. 2008, 8, 502–514. [Google Scholar] [CrossRef]
- Simila, M.; Kouki, J.; Monkkonen, M.; Sippola, A.L.; Huhta, E. Co-variation and indicators of species diversity: Can richness of forest-dwelling species be predicted in northern boreal forests? Ecol. Ind. 2006, 6, 686–700. [Google Scholar] [CrossRef]
- Nordström, E.-M.; Nieuwenhuis, M.; Başkent, E.Z.; Biber, P.; Black, K.; Borges, J.G.; Bugalho, M.N.; Corradini, G.; Felton, A. Forest decision support systems for analysis of ecosystem services provisioning at landscape scale under global climate and market change scenarios. Eur. J. Forest Res. 2019, 138, 561–581. [Google Scholar] [CrossRef]

| Practical Aspects of Indicator | Management Category | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Scale | Type of Biodiversity | Practical Indicator | Cost-efficient to Sample | Easy to Recognize | Time-efficient to Sample | Unmanaged Forest | Managed Forests | Plantations | Silvopastoral System |
| Stand | Compos. | Vascular plants [55,56} | * | * | |||||
| Stand | Compos. | Carabidae beetles [46] | * | * | * | * | * | * | |
| Stand | Compos. | Spiders [25] | * | * | |||||
| Stand | Compos. | Hoverflies [25] | * | * | |||||
| Stand | Compos. | Tree species composition (richness, abundance, and diversity) [4,63] | * | * | * | * | * | * | |
| Stand | Compos. | Shrub species composition (richness, abundance, and diversity) [63] | * | * | * | * | * | * | |
| Stand | Struct. | Deadwood [2,12,20,63,64,65] | * | * | * | * | * | ||
| Stand | Struct. | Canopy cover [2,4] | * | * | * | * | * | * | |
| Stand | Struct. | Special trees (occurrence of moss and lichen-covered, bent, damaged, hollow and forked trees) [12] | * | * | * | * | |||
| Stand | Struct. | Proximity to native forests [25] | * | * | * | * | * | * | |
| Stand | Struct. | Large trees (mature trees) [12,63] | * | * | * | * | |||
| Stand | Struct. | Old forest stands [12] | * | * | * | ||||
| Stand | Struct. | Deciduous trees [12] | * | * | * | ||||
| Stand | Functi. | Stand age [2,12,25] | * | * | * | * | |||
| Stand | Functi. | Available phosphorus (P) [25] | * | ||||||
| Stand | Functi. | Elevation [25] | * | * | * | * | |||
| Stand | Functi. | Uprooted trees [12] | * | ||||||
| Stand | Functi. | Thinning frequency [25] | * | * | * | * | |||
| Stand | Functi. | Wood-decaying bracket fungi [12] | * | * | * | ||||
| Lands. | Compos. | Birds [25,55,56,58] | * | * | * | * | * | ||
| Lands. | Compos. | Tree species richness [66] | * | * | * | ||||
| Lands. | Compos. | Shrub species richness [66] | * | * | * | ||||
| Lands. | Compos. | Valuable habitats [4] | * | * | * | * | |||
| Lands. | Struct. | Patch shape, proximity, texture, diversity, and size [67] | - | - | - | - | - | - | - |
| Impractical Aspects of Indicator | Management Category | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Scale | Type of Biodiversity | Impractical Indicator and Authors | Expensive to Sample | Hard to Recognize | Time-consuming to Sample | Unmanaged Forest | Managed Forest | Plantation | Silvopastoral System |
| Stand | Compos. | Vascular plants [57,58] | * | * | * | * | * | * | |
| Stand | Compos. | Lichens [12,57,58] | * | * | * | * | |||
| Stand | Compos. | Fungi [59] | * | * | * | * | * | ||
| Stand | Compos. | Bryophyte [25] | * | * | * | ||||
| Stand | Compos. | Invertebrate species [25] | * | * | * | ||||
| Stand | Compos. | Plant species diversity [4,54] | * | * | * | * | * | * | |
| Stand | Compos. | Herb layer [68] | * | * | * | * | * | ||
| Scale and Type of Biodiversity | Indicator (Attribute) | Practical Data Collection Method for Managed Forests | Practical Data Collection Method for Unmanaged Forests | Practical Data Collection Method for Plantations | Practical Data Collection for Silvopastoral Systems |
|---|---|---|---|---|---|
| Stand Structure | Deadwood | Smartphone app [42]; NFI [63] | Smartphone app [42]; LiDAR [41]; NFI [63]; line-intersect sampling [107] | - | - |
| Structure | Big trees | Smartphone app [43]; NFI [63] | Smartphone app [43]; NFI [63] | - | - |
| Structure | Tree density | Smartphone app [43] | Smartphone app [43] | - | - |
| Structure | Micro-habitat diversity | Satellite images [45] | Satellite images [45] | - | - |
| Structure | Biomass | LiDAR [113] | LiDAR [113] | LiDAR [113] | - |
| Structure | Height | LiDAR [113] | LiDAR [113] | LiDAR [113] | - |
| Composition | Tree species diversity | Smartphone app [43]; NFI [63] | Smartphone app [43]; NFI [63] | - | - |
| Composition | Shrub species diversity | NFI [63] | NFI [63] | - | - |
| Composition | Herbs | Smartphone app [42] | Smartphone app [42] | - | - |
| Composition | Bird species richness | Satellite images [45]; National Ornithological Society [114]; National bird atlas [89]; gamekeeper register [115] | Satellite images [45]; National Ornithological Society [114]; gamekeeper register [115] | - | - |
| Composition | Fungal species richness | LiDAR [59] | - | - | |
| Composition | Composition of forest-dwelling beetles | LiDAR [40] | LiDAR [40] | - | - |
| Function | Disturbances | Smartphone app [42] | Smartphone app [42] | - | - |
| Regeneration | Smartphone app [42] | Smartphone app [42] | - | ||
| Landscape Structure | Deadwood | LiDAR + inventory data + aerial photographs [41] | LiDAR [41] | - | - |
| Structure | Micro-habitat diversity | Satellite images [45] | Satellite images [45] | - | - |
| Structure | Height | LiDAR [113] | - | - | |
| Structure | Biomass | LiDAR [113] | - | - | |
| Composition | Tree species | - | NFI [66] | ||
| Composition | Shrub species | - | NFI [66] | ||
| Composition | Bird species richness | Satellite images [45]; National Ornithological Society [114,116]; Museum of Natural History [68]; National bird atlas [87] | Satellite images [45]; National Ornithological Society [116]; National bird atlas [87] | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ćosović, M.; Bugalho, M.N.; Thom, D.; Borges, J.G. Stand Structural Characteristics Are the Most Practical Biodiversity Indicators for Forest Management Planning in Europe. Forests 2020, 11, 343. https://doi.org/10.3390/f11030343
Ćosović M, Bugalho MN, Thom D, Borges JG. Stand Structural Characteristics Are the Most Practical Biodiversity Indicators for Forest Management Planning in Europe. Forests. 2020; 11(3):343. https://doi.org/10.3390/f11030343
Chicago/Turabian StyleĆosović, Marija, Miguel N. Bugalho, Dominik Thom, and José G. Borges. 2020. "Stand Structural Characteristics Are the Most Practical Biodiversity Indicators for Forest Management Planning in Europe" Forests 11, no. 3: 343. https://doi.org/10.3390/f11030343
APA StyleĆosović, M., Bugalho, M. N., Thom, D., & Borges, J. G. (2020). Stand Structural Characteristics Are the Most Practical Biodiversity Indicators for Forest Management Planning in Europe. Forests, 11(3), 343. https://doi.org/10.3390/f11030343






