A Four-Year, Seven-State Reforestation Trial with Eastern Hemlocks (Tsuga canadensis) Resistant to Hemlock Woolly Adelgid (Adelges tsugae)
Abstract
1. Introduction
2. Methods
2.1. Source Trees
2.2. Greenhouse Propagation
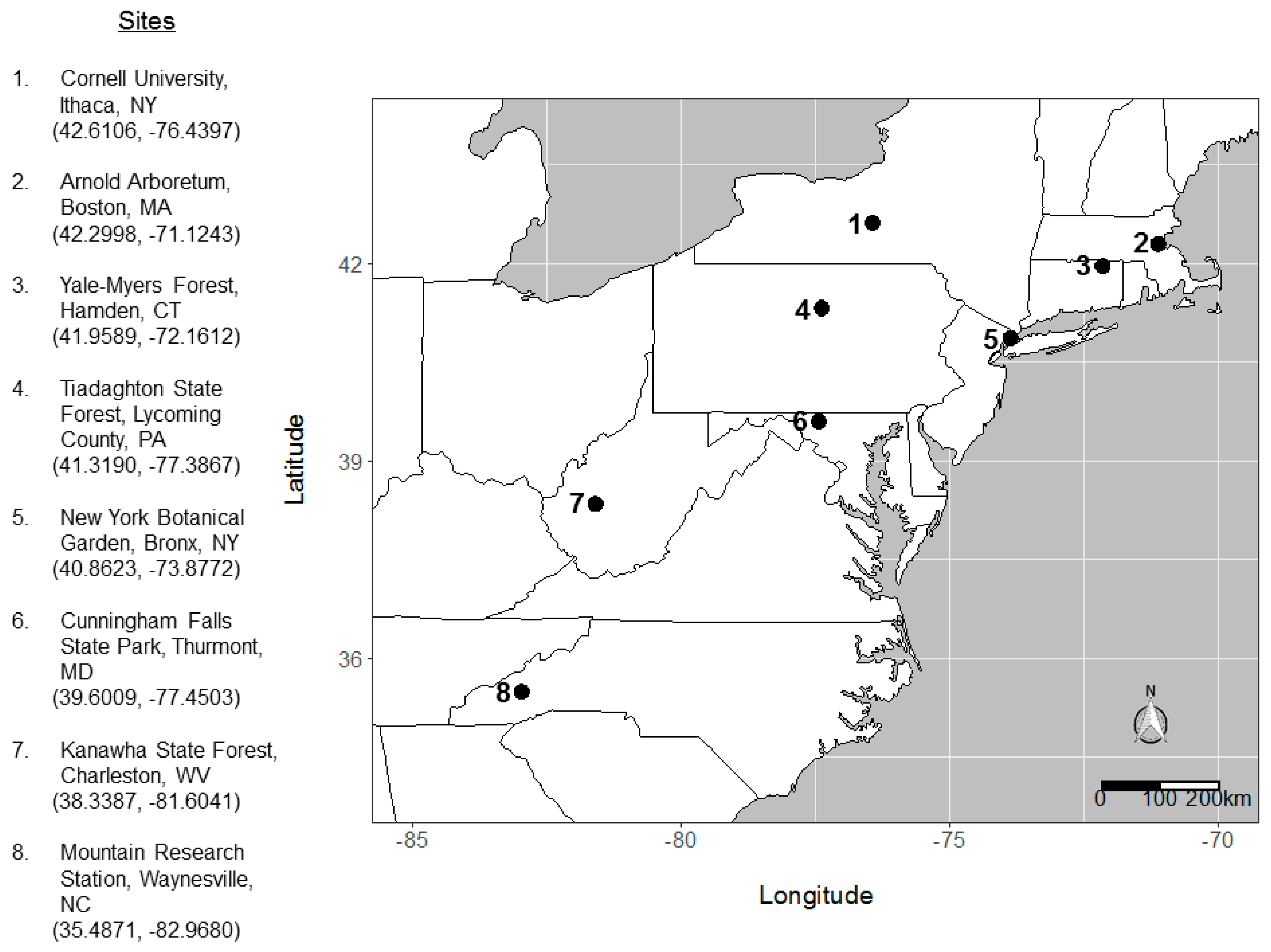
2.3. Reforestation Plot Establishment
2.4. Plot Surveys
2.5. Statistical Analysis
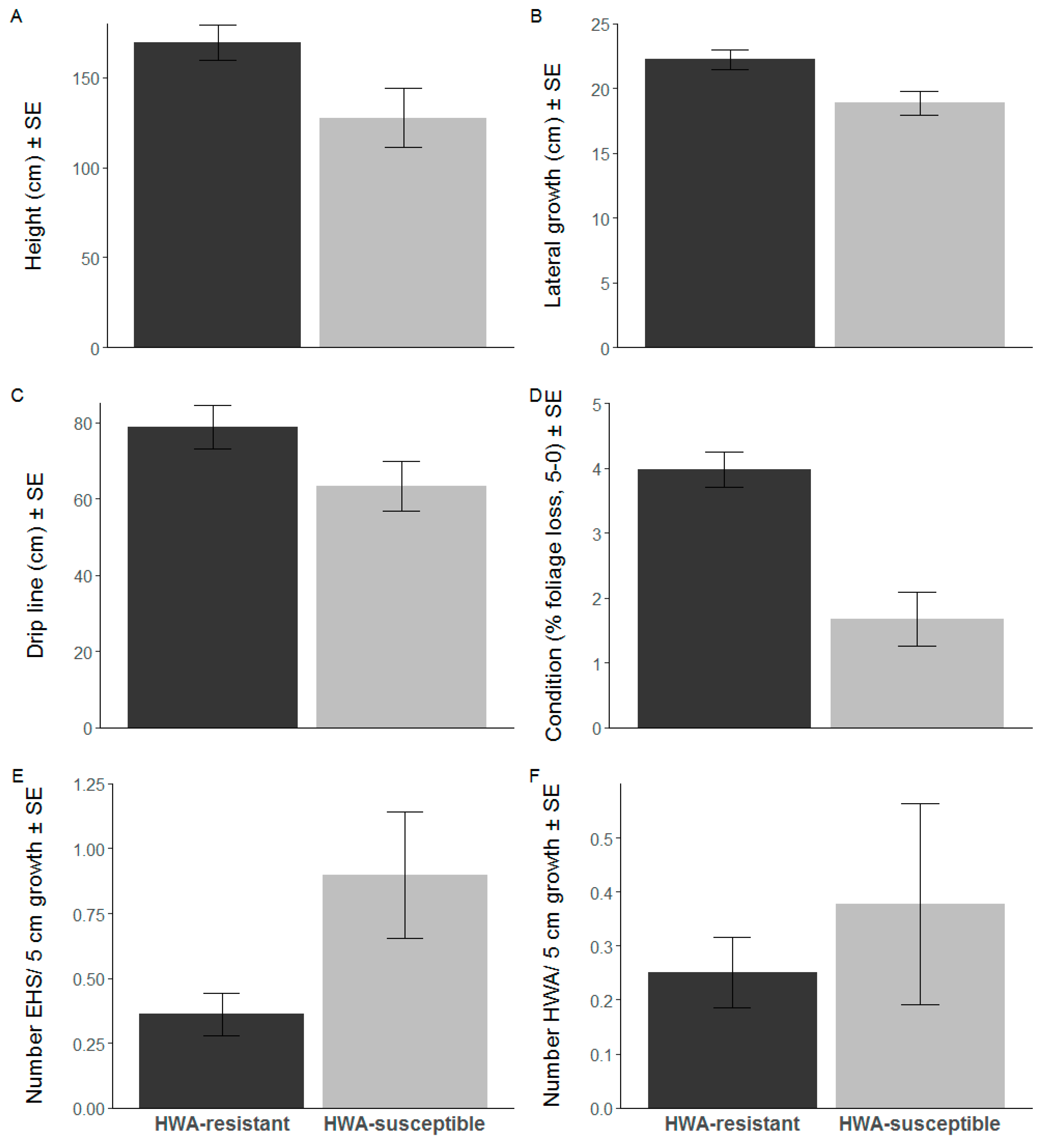
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sharma, H.; Ortiz, R. Host plant resistance to insects: An eco-friendly approach for pest management and environment conservation. J. Environ. Biol. 2002, 23, 111–136. [Google Scholar] [PubMed]
- Schlarbaum, S.E.; Hebard, F.; Spaine, P.C.; Kamalay, J.C. Three American tragedies: Chestnut blight, butternut canker, and Dutch elm disease. In Exotic Pests of Eastern Forests Conference Proceedings; Britton Kerry, O., Ed.; US Forest Service: Washington, DC, USA; Tennessee Exotic Pest Plant Council: Oak Ridge, TN, USA, 1997; pp. 45–54. [Google Scholar]
- Karnosky, D.F. Dutch elm disease: A review of the history, environmental implications, control, and research needs. Environ. Conserv. 1979, 6, 311–322. [Google Scholar] [CrossRef]
- Townsend, A.; Bentz, S.; Douglass, L. Evaluation of 19 American elm clones for tolerance to Dutch elm disease. J. Environ. Hortic. 2005, 23, 21–24. [Google Scholar]
- Knight, K.S.; Slavicek, J.M.; Kappler, R.; Pisarczyk, E.; Wiggin, B.; Menard, K. Using Dutch elm disease-tolerant elm to restore floodplains impacted by emerald ash borer. In Proceedings of the Fourth International Workshop on the Genetics of Host-Parasite Interactions in Forestry: Disease and Insect Resistance in Forest Trees, Eugene, OR, USA, 31 July–1 August 2011; Pacific Southwest Research Station: Albany, CA, USA; Forest Service: Washington, DC, USA; US Department of Agriculture: Washington, DC, USA, 2012; pp. 317–323. [Google Scholar]
- Paillet, F.L. Chestnut: History and ecology of a transformed species. J. Biogeogr. 2002, 29, 1517–1530. [Google Scholar] [CrossRef]
- Milgroom, M.G.; Cortesi, P. Biological control of chestnut blight with hypovirulence: A critical analysis. Annu. Rev. Phytopathol. 2004, 42, 311–338. [Google Scholar] [CrossRef]
- Jacobs, D.F.; Dalgleish, H.J.; Nelson, C.D. A conceptual framework for restoration of threatened plants: The effective model of american chestnut (Castanea dentata) reintroduction. New Phytol. 2013, 197, 378–393. [Google Scholar] [CrossRef]
- Cipollini, M.; Dingley, N.R.; Felch, P.; Maddox, C. Evaluation of phenotypic traits and blight-resistance in an American chestnut backcross orchard in Georgia. Glob. Ecol. Conserv. 2017, 10, 1–8. [Google Scholar] [CrossRef]
- Knight, K.S.; Haugen, L.M.; Pinchot, C.C.; Schaberg, P.G.; Slavicek, J.M. American elm (Ulmus americana) in restoration plantings: A review. In Proceedings of the American Elm Restoration Workshop 2016, Lewis Center, OH, USA, 25–27 October 2016; US Department of Agriculture: Washington, DC, USA; Forest Service: Washington, DC, USA; Northern Research Station: Newtown Square, PA, USA, 2017; pp. 133–140. [Google Scholar]
- Oten, K.L.; Merkle, S.A.; Jetton, R.M.; Smith, B.C.; Talley, M.E.; Hain, F.P. Understanding and developing resistance in hemlocks to the hemlock woolly adelgid. Southeast. Nat. 2014, 13, 147–168. [Google Scholar]
- Hanover, J.W. Physiology of tree resistance to insects. Annu. Rev. Entomol. 1975, 20, 75–95. [Google Scholar] [CrossRef]
- Showalter, D.N.; Raffa, K.F.; Sniezko, R.A.; Herms, D.A.; Liebhold, A.M.; Smith, J.A.; Bonello, P. Strategic development of tree resistance against forest pathogen and insect invasions in defense-free space. Front. Ecol. Evol. 2018, 6, 124. [Google Scholar] [CrossRef]
- Havill, N.P.; Montgomery, M.E.; Yu, G.; Shiyake, S.; Caccone, A. Mitochondrial DNA from hemlock woolly adelgid (hemiptera: Adelgidae) suggests cryptic speciation and pinpoints the source of the introduction to Eastern North America. Ann. Entomol. Soc. Am. 2006, 99, 195–203. [Google Scholar] [CrossRef]
- Ellison, A.; Orwig, D.A.; Fitzpatrick, M.; Preisser, E.L. The past, present, and future of the hemlock woolly adelgid (Adelges tsugae) and its ecological interactions with eastern hemlock (Tsuga canadensis) forests. Insects 2018, 9, 172. [Google Scholar] [CrossRef] [PubMed]
- Sumpter, K.L.; McAvoy, T.J.; Brewster, C.C.; Mayfield, A.E.; Salom, S.M. Assessing an integrated biological and chemical control strategy for managing hemlock woolly adelgid in southern Appalachian forests. For. Ecol. Manag. 2018, 411, 12–19. [Google Scholar] [CrossRef]
- Lagalante, A.F.; Montgomery, M.E.; Calvosa, F.C.; Mirzabeigi, M.N. Characterization of terpenoid volatiles from cultivars of eastern hemlock (Tsuga canadensis). J. Agric. Food Chem. 2007, 55, 10850. [Google Scholar] [CrossRef]
- Lagalante, A.F.; Montgomery, M.E. Analysis of terpenoids from hemlock (Tsuga) species by solid-phase microextraction/gas chromatography/ion-trap mass spectrometry. J. Agric. Food Chem. 2003, 51, 2115–2120. [Google Scholar] [CrossRef]
- Del Tredici, P.; Kitajima, A. Introduction and cultivation of Chinese hemlock (Tsuga chinensis) and its resistance to hemlock woolly adelgid (Adelges tsugae). Arboric. Urban For. 2004, 30, 282. [Google Scholar]
- Hoover, B.K.; Bates, R.M.; Sellmer, J.C.; Hoover, G.A. Challenging Chinese hemlock (Tsuga chinensis) with hemlock woolly adelgid (Adelges tsugae) ovisacs. Arboric. Urban For. 2009, 35, 1–4. [Google Scholar]
- Montgomery, M.E.; Bentz, S.E.; Olsen, R.T. Evaluation of hemlock (Tsuga) species and hybrids for resistance to Adelges tsugae (hemiptera: Adelgidae) using artificial infestation. J. Econ. Entomol. 2009, 102, 1247–1254. [Google Scholar] [CrossRef]
- Bentz, S.; Riedel, L.G.; Pooler, M.R.; Townsend, A. Hybridization and self-compatibility in controlled pollinations of eastern north American and Asian hemlock (Tsuga) species. J. Arboric. 2002, 28, 200–205. [Google Scholar]
- Ingwell, L.L.; Preisser, E.L. Using citizen science programs to identify host resistance in pest-invaded forests. Conserv. Biol. 2011, 25, 182–188. [Google Scholar] [CrossRef]
- McKenzie, E.A.; Elkinton, J.S.; Casagrande, R.A.; Preisser, E.L.; Mayer, M. Terpene chemistry of eastern hemlocks resistant to hemlock woolly adelgid. J. Chem. Ecol. 2014, 40, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Miller-Pierce, M.; Orwig, D.; Preisser, E. Effects of hemlock woolly adelgid and elongate hemlock scale on eastern hemlock growth and foliar chemistry. Environ. Entomol. 2010, 39, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.M.; Schaeffer, R.N.; Hickin, M.L.; Rigsby, C.M.; Sommi, A.F.; Thornber, C.S.; Orians, C.M.; Preisser, E.L. Chronic impacts of invasive herbivores on a foundational forest species: A whole-tree perspective. Ecology 2018, 99, 1783–1791. [Google Scholar] [CrossRef] [PubMed]
- Caswell, T.; Casagrande, R.; Maynard, B.; Preisser, E.L. Production and evaluation of hemlocks potentially resistant to the hemlock woolly adelgid. In Proceedings of the Fourth Symposium on Hemlock Woolly Adelgid in the Eastern United States, Hartford, CT, USA, 12–14 February 2008; Onken, B., Reardon, R., Eds.; Forest Health Technology Enterprise Team, Forest Service: Washington, DC, USA, 2008; pp. 124–134. [Google Scholar]
- Joseph, S.V.; Kristine Braman, S.; Quick, J.; Hanula, J.L. The range and response of neonicotinoids on hemlock woolly adelgid, Adelges tsugae (hemiptera: Adelgidae). J. Environ. Hortic. 2011, 29, 197–204. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 1, 1–48. [Google Scholar]
- Fox, J.; Weisberg, S. Multivar. In An R Companion to Applied Regression; SAGE Publications: Thousand Oaks, CA, USA, 2011. [Google Scholar]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin, Germany, 2016. [Google Scholar]
- Limbu, S.; Keena, M.; Whitmore, M. Hemlock woolly adelgid (hemiptera: Adelgidae): A non-native pest of hemlocks in eastern North America. J. Integr. Pest. Manag. 2018, 9, 27. [Google Scholar] [CrossRef]
- Ellison, A.M.; Barker-Plotkin, A.A.; Foster, D.R.; Orwig, D.A. Experimentally testing the role of foundation species in forests: The Harvard forest hemlock removal experiment. Methods Ecol. Evol. 2010, 1, 168–179. [Google Scholar] [CrossRef]
- Orwig, D.A.; Cobb, R.C.; D’Amato, A.W.; Kizlinski, M.L.; Foster, D.R. Multi-Year ecosystem response to hemlock woolly adelgid infestation in Southern New England forests. Can. J. For. Res. 2008, 38, 834–843. [Google Scholar] [CrossRef]
- Howe, G.A.; Jander, G. Plant immunity to insect herbivores. Annu. Rev. Plant Biol. 2008, 59, 41–66. [Google Scholar] [CrossRef]
- Mumm, R.; Hilker, M. Direct and indirect chemical defence of pine against folivorous insects. Trends Plant Sci. 2006, 11, 351–358. [Google Scholar] [CrossRef]
- McClure, M.S.; Hare, J.D. Foliar terpenoids in Tsuga species and the fecundity of scale insects. Oecologia 1984, 63, 185–193. [Google Scholar] [CrossRef]
- Kinahan, I.G.; Baranowski, A.K.; Whitney, E.R.; Savage, S.K.; Rigsby, C.M.; Shoemaker, E.E.; Orians, C.M.; Preisser, E.L. Facilitation between invasive herbivores: Hemlock woolly adelgid increases gypsy moth preference for and performance on eastern hemlock. Ecol. Entomol. 2020. [Google Scholar] [CrossRef]
- Wilson, C.M.; Vendettuoli, J.F.; Orwig, D.A.; Preisser, E.L. Impact of an invasive insect and plant defense on a native forest defoliator. Insects 2016, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Gonda-King, L.; Radville, L.; Preisser, E. False ring formation in eastern hemlock branches: Impacts of hemlock woolly adelgid and elongate hemlock scale. Environ. Entomol. 2012, 41, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Soltis, N.E.; Gómez, S.; Gonda-King, L.; Preisser, E.L.; Orians, C.M. Contrasting effects of two exotic invasive hemipterans on whole-plant resource allocation in a declining conifer. Entomol. Exp. Appl. 2015, 157, 86–97. [Google Scholar] [CrossRef]
- Ellison, A.M.; Bank, M.S.; Clinton, B.D.; Colburn, E.A.; Elliott, K.; Ford, C.R.; Foster, D.R.; Kloeppel, B.D.; Knoepp, J.D.; Lovett, G.M. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Front. Ecol. Environ. 2005, 3, 479–486. [Google Scholar] [CrossRef]
- USFS. Forest Health Protection—Hemlock Woolly Adelgid Distribution Map. Available online: http://na.fs.fed.us/fhp/hwa/maps/2011.pdf (accessed on 4 March 2020).
- Gómez, S.; Gonda-King, L.; Orians, C.; Orwig, D.; Panko, R.; Radville, L.; Soltis, N.; Thornber, C.; Preisser, E. Interactions between invasive herbivores and their long-term impact on New England hemlock forests. Biol. Invasions 2015, 17, 661–673. [Google Scholar] [CrossRef]
- Preisser, E.L.; Miller-Pierce, M.R.; Vansant, J.; Orwig, D.A. Eastern hemlock (Tsuga canadensis) regeneration in the presence of hemlock woolly adelgid (Adelges tsugae) and elongate hemlock scale (Fiorinia externa). Can. J. For. Res. 2011, 41, 2433–2439. [Google Scholar] [CrossRef]
- Ingwell, L.L.; Miller-Pierce, M.; Trotter, R.T., III; Preisser, E.L. Vegetation and invertebrate community response to eastern hemlock decline in Southern New England. Northeast. Nat. 2012, 19, 541–558. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinahan, I.G.; Grandstaff, G.; Russell, A.; Rigsby, C.M.; Casagrande, R.A.; Preisser, E.L. A Four-Year, Seven-State Reforestation Trial with Eastern Hemlocks (Tsuga canadensis) Resistant to Hemlock Woolly Adelgid (Adelges tsugae). Forests 2020, 11, 312. https://doi.org/10.3390/f11030312
Kinahan IG, Grandstaff G, Russell A, Rigsby CM, Casagrande RA, Preisser EL. A Four-Year, Seven-State Reforestation Trial with Eastern Hemlocks (Tsuga canadensis) Resistant to Hemlock Woolly Adelgid (Adelges tsugae). Forests. 2020; 11(3):312. https://doi.org/10.3390/f11030312
Chicago/Turabian StyleKinahan, Ian G., Gabrielle Grandstaff, Alana Russell, Chad M. Rigsby, Richard A. Casagrande, and Evan L. Preisser. 2020. "A Four-Year, Seven-State Reforestation Trial with Eastern Hemlocks (Tsuga canadensis) Resistant to Hemlock Woolly Adelgid (Adelges tsugae)" Forests 11, no. 3: 312. https://doi.org/10.3390/f11030312
APA StyleKinahan, I. G., Grandstaff, G., Russell, A., Rigsby, C. M., Casagrande, R. A., & Preisser, E. L. (2020). A Four-Year, Seven-State Reforestation Trial with Eastern Hemlocks (Tsuga canadensis) Resistant to Hemlock Woolly Adelgid (Adelges tsugae). Forests, 11(3), 312. https://doi.org/10.3390/f11030312




