Temperature-Dominated Driving Mechanisms of the Plant Diversity in Temperate Forests, Northeast China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
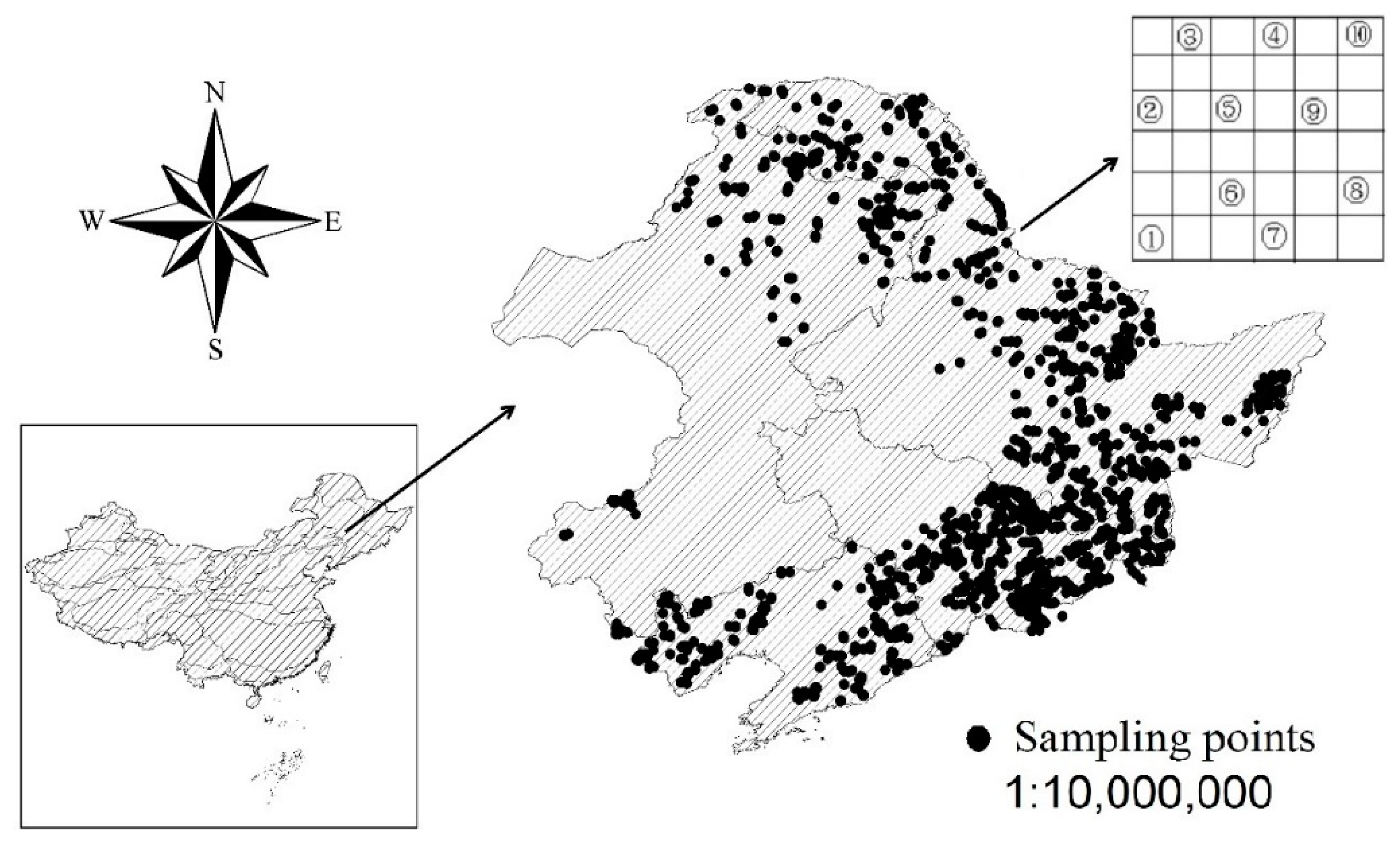
2.2. Data Investigation
2.3. Explanatory Variables
2.4. Data Analysis
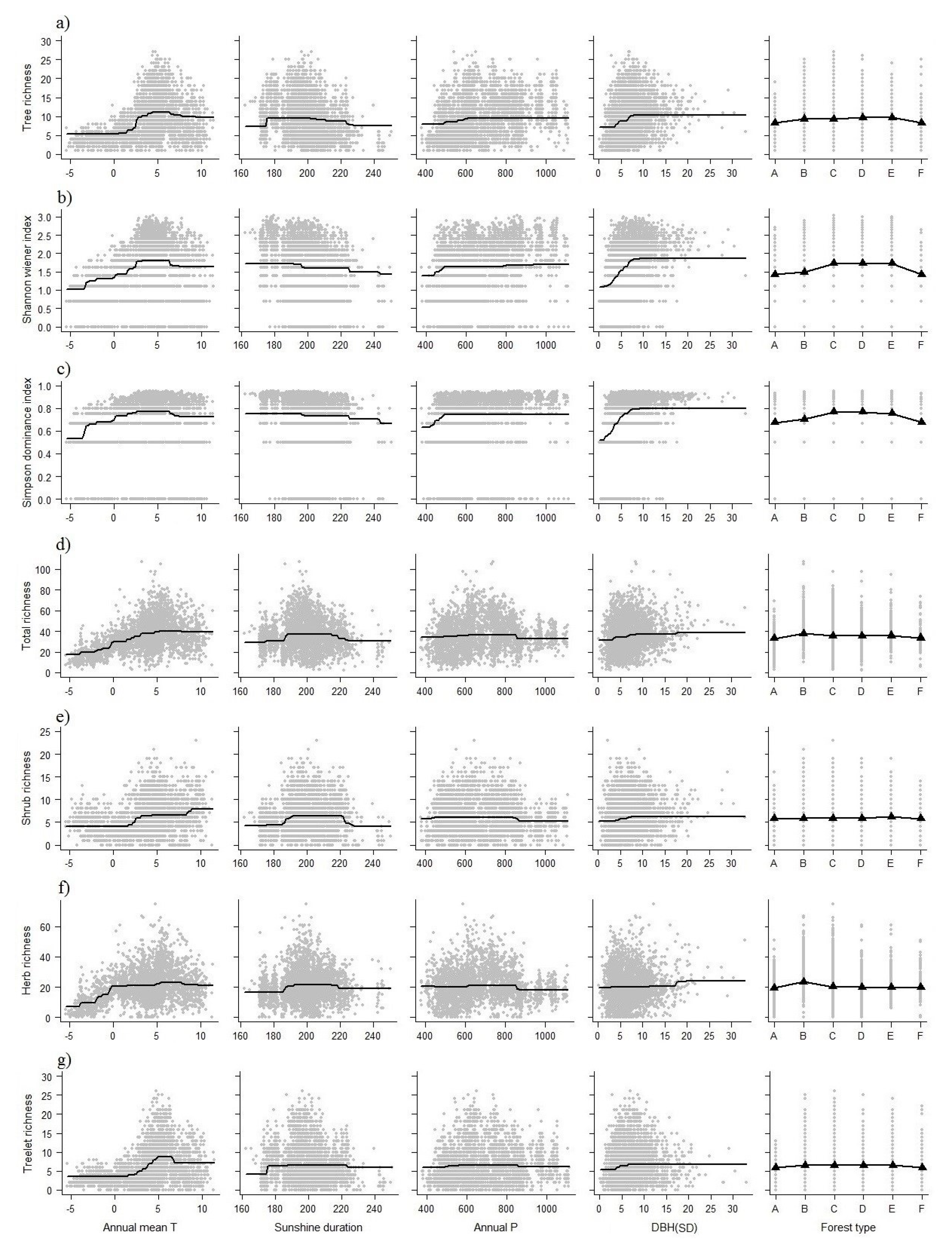
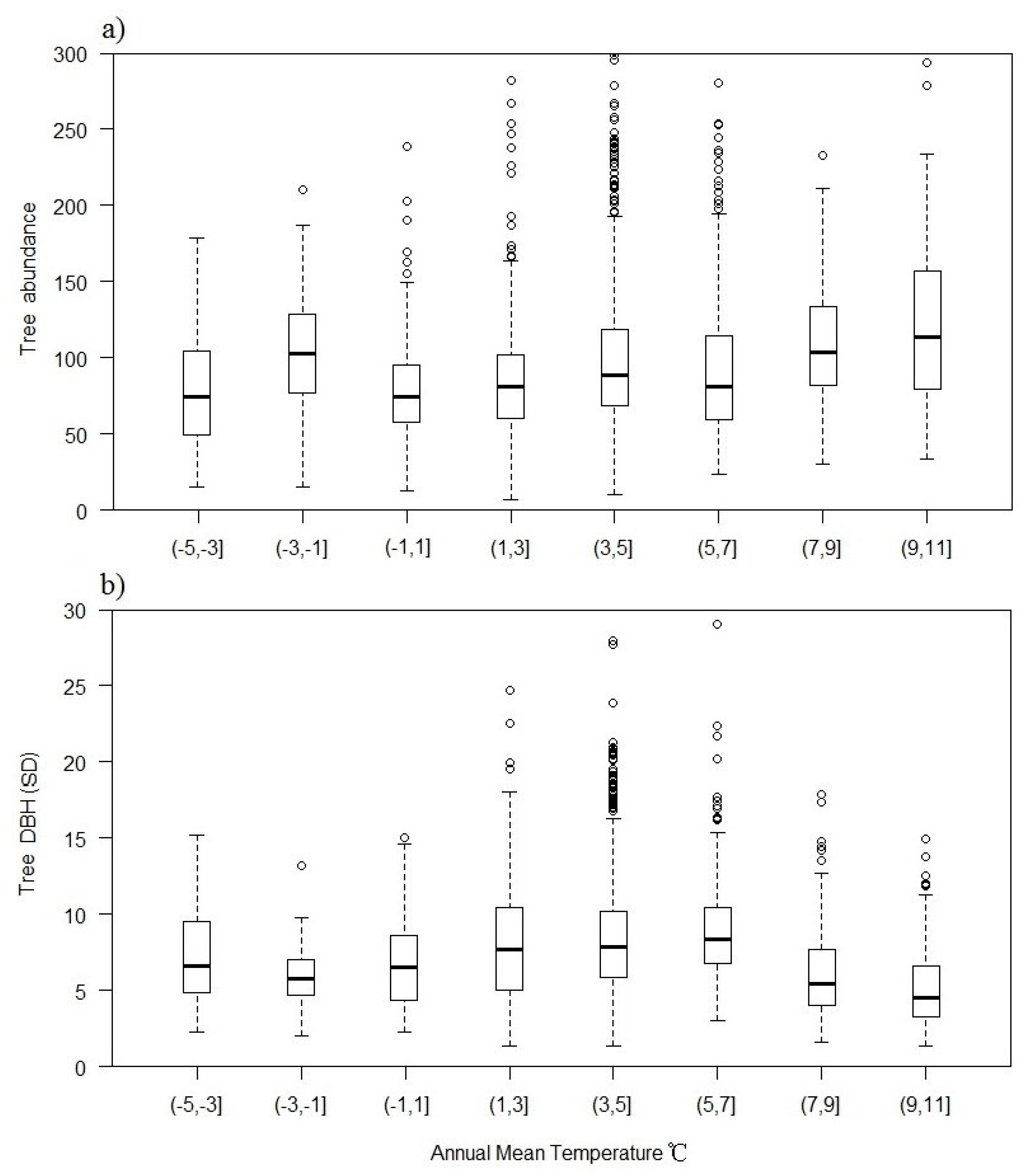
3. Results
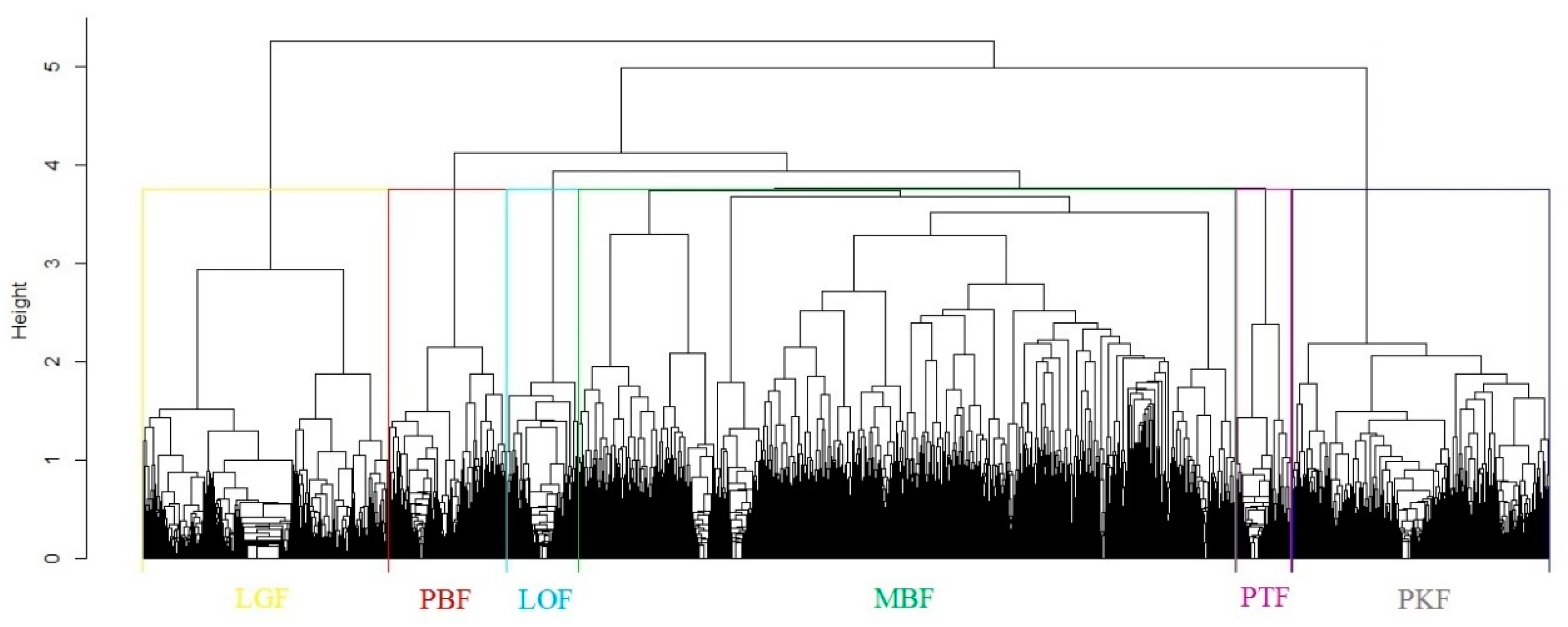
3.1. Forest Type Cluster
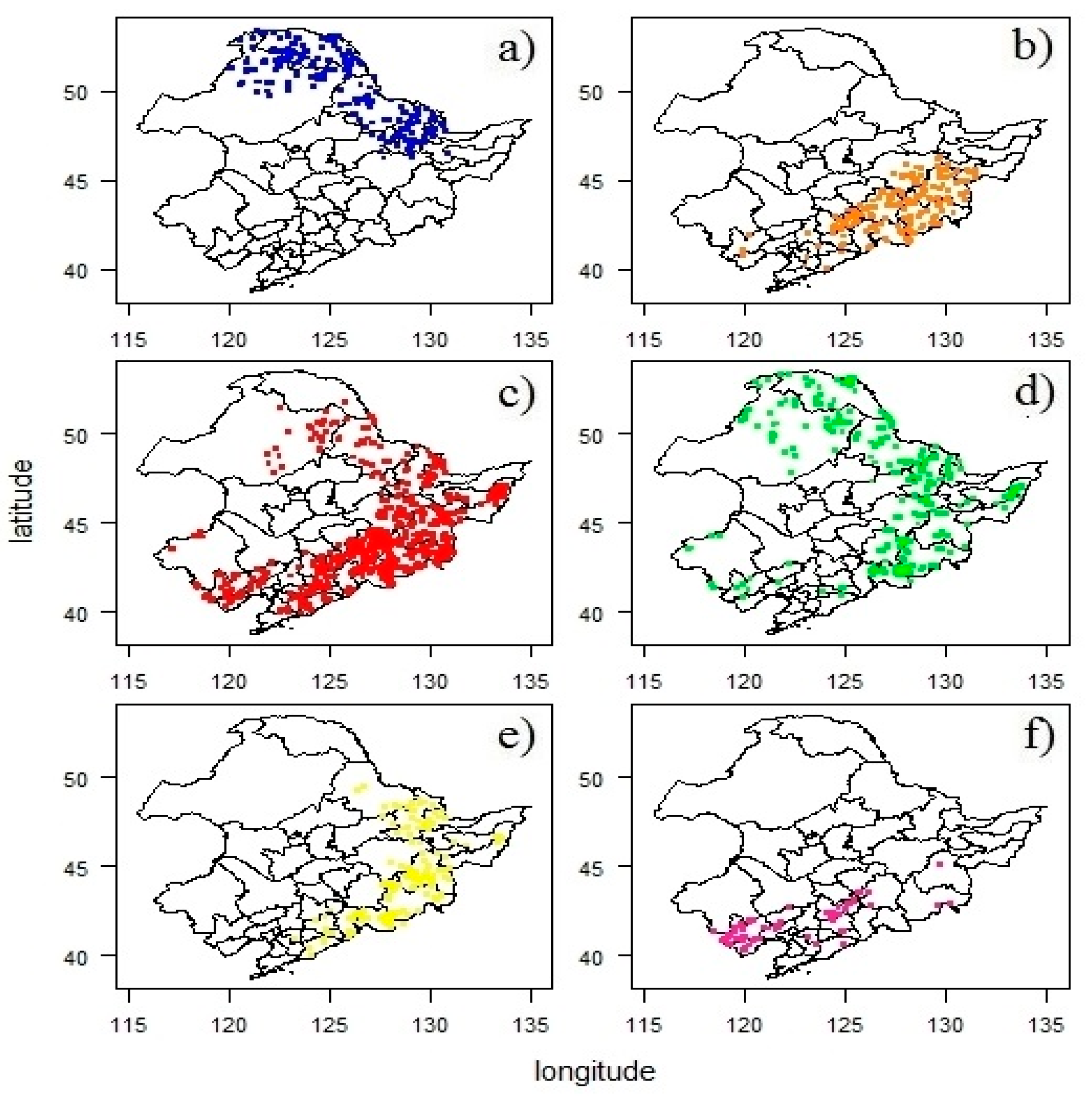
3.2. Spatial Distribution of Forest Types
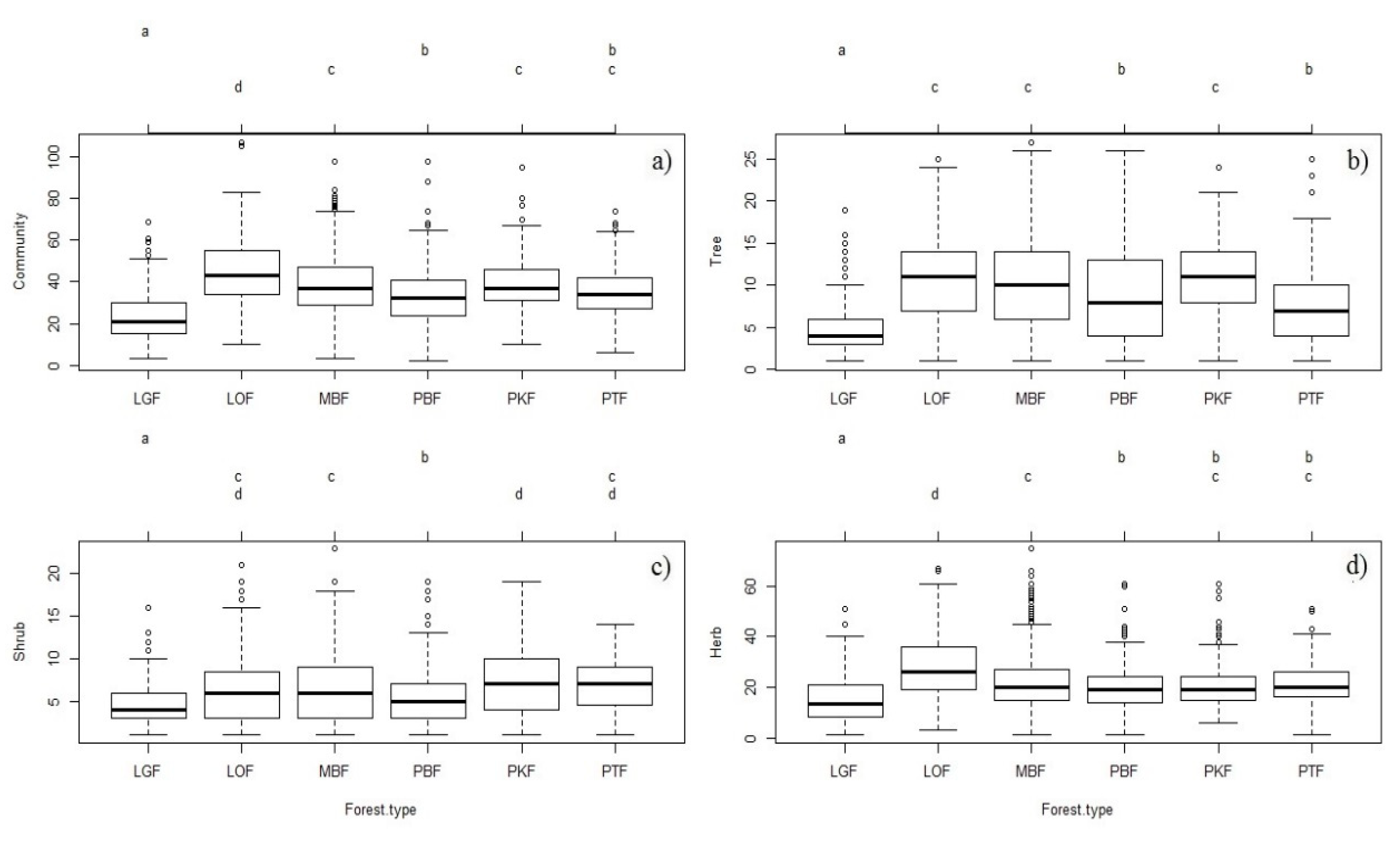
3.3. Plant Richness in Different Groups
3.4. Importance of Variables
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bormann, F.; Likens, G. An ecological study. Book reviews: Pattern and process in a forested ecosystem. Disturbance, development and the steady state based on the Hubbard brook ecosystem study. Science 1979, 205, 1369–1370. [Google Scholar]
- Shmida, A.; Wilson, M.V. Biological determinants of species-diversity. J. Biogeogr. 1985, 12, 1–20. [Google Scholar] [CrossRef]
- Gaston, K. Global patterns in biodiversity. Nature 2000, 405, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Paillet, Y.; Berges, L.; Hjalten, J.; Odor, P.; Avon, C.; Bernhardt-Roemermann, M.; Bijlsma, R.-J.; De Bruyn, L.; Fuhr, M.; Grandin, U.; et al. Biodiversity differences between managed and unmanaged forests: Meta-analysis of species richness in Europe. Conserv. Biol. 2010, 24, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Chun, J. Environmental drivers of patterns of plant diversity along a wide environmental gradient in korean temperate forests. Forests 2016, 7, 19. [Google Scholar] [CrossRef]
- Grytnes, J.; Vetaas, O. Species richness and altitude: A comparison between null models and interpolated plant species richness along the Himalayan altitudinal gradient, Nepal. Am. Nat. 2002, 159, 294–304. [Google Scholar] [CrossRef]
- Crawley, M.; Harral, J.E. Scale dependence in plant biodiversity. Science 2001, 291, 864–868. [Google Scholar] [CrossRef]
- Legendre, P.; Borcard, D.; Peres-Neto, P.R. Analyzing beta diversity: Partitioning the spatial variation of community composition data. Ecol. Monogr. 2005, 75, 435–450. [Google Scholar] [CrossRef]
- Swenson, N.; Erickson, D.; Mi, X.; Bourg, N.; Forero-Montana, J.; Ge, X.; Howe, R.; Lake, J.; Liu, X.; Ma, K.; et al. Phylogenetic and functional alpha and beta diversity in temperate and tropical tree communities. Ecology 2012, 93, S112–S125. [Google Scholar] [CrossRef]
- Macarthur, R. Patterns of species diversity. Biol. Rev. 1965, 40, 510–532. [Google Scholar] [CrossRef]
- Currie, D. Energy and large-scale patterns of animal-species and plant-species richness. Am. Nat. 1991, 137, 27–49. [Google Scholar] [CrossRef]
- Araujo, M.; Luoto, M. The importance of biotic interactions for modelling species distributions under climate change. Glob. Ecol. Biogeogr. 2007, 16, 743–753. [Google Scholar] [CrossRef]
- Kissling, W.; Rahbek, C.; Boehning-Gaese, K. Food plant diversity as broad-scale determinant of avian frugivore richness. Proc. R. Soc. B Biol. Sci. 2007, 274, 799–808. [Google Scholar] [CrossRef]
- Mayfield, M.; Levine, J. Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecol. Lett. 2010, 13, 1085–1093. [Google Scholar] [CrossRef] [PubMed]
- Meynard, C.; Lavergne, S.; Boulangeat, I.; Garraud, L.; Van Es, J.; Mouquet, N.; Thuiller, W. Disentangling the drivers of metacommunity structure across spatial scales. J. Biogeogr. 2013, 40, 1560–1571. [Google Scholar] [CrossRef] [PubMed]
- Henriques-Silva, R.; Lindo, Z.; Peres-Neto, P. A community of metacommunities: Exploring patterns in species distributions across large geographical areas. Ecology 2013, 94, 627–639. [Google Scholar] [CrossRef]
- Wang, X.; Wiegand, T.; Swenson, N.; Wolf, A.; Howe, R.; Hao, Z.; Lin, F.; Ye, J.; Yuan, Z. Mechanisms underlying local functional and phylogenetic beta diversity in two temperate forests. Ecology 2015, 96, 1062–1073. [Google Scholar] [CrossRef]
- Yuan, Z.; Wang, S.; Gazol, A.; Mellard, J.; Lin, F.; Ye, J.; Hao, Z.; Wang, X.; Loreau, M. Multiple metrics of diversity have different effects on temperate forest functioning over succession. Oecologia 2016, 182, 1175–1185. [Google Scholar] [CrossRef]
- Koenig, C.; Weigelt, P.; Kreft, H. Dissecting global turnover in vascular plants. Glob. Ecol. Biogeogr. 2017, 26, 228–242. [Google Scholar] [CrossRef]
- Bernhardt-Roemermann, M.; Baeten, L.; Craven, D.; De Frenne, P.; Hedl, R.; Lenoir, J.; Bert, D.; Brunet, J.; Chudomelova, M.; Decocq, G.; et al. Drivers of temporal changes in temperate forest plant diversity vary across spatial scales. Glob. Change Biol. 2015, 21, 3726–3737. [Google Scholar] [CrossRef]
- Putten van der, W.H. Belowground drivers of plant diversity. Science 2017, 355, 134–135. [Google Scholar] [CrossRef]
- Hawkins, B.; Field, R.; Cornell, H.; Currie, D.; Guegan, J.; Kaufman, D.; Kerr, J.; Mittelbach, G.; Oberdorff, T.; O’Brien, E.; et al. Energy, water, and broad-scale geographic patterns of species richness. Ecology 2003, 84, 3105–3117. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, H.Y.H.; Taylor, A. Multiple drivers of plant diversity in forest ecosystems. Glob. Ecol. Biogeogr. 2014, 23, 885–893. [Google Scholar] [CrossRef]
- Naud, L.; Masviken, J.; Freire, S.; Angerbjorn, A.; Dalen, L.; Dalerum, F. Altitude effects on spatial components of vascular plant diversity in a subarctic mountain tundra. Ecol. Evol. 2019, 9, 4783–4795. [Google Scholar] [CrossRef]
- O’Brien, E.; Field, R.; Whittaker, R. Climatic gradients in woody plant (tree and shrub) diversity: Water-energy dynamics, residual variation, and topography. Oikos 2000, 89, 588–600. [Google Scholar] [CrossRef]
- Wang, Z.; Fang, J.; Tang, Z.; Lin, X. Patterns, determinants and models of woody plant diversity in China. Proc. R. Soc. B Biol. Sci. 2011, 278, 2122–2132. [Google Scholar] [CrossRef]
- Golicher, D.; Cayuela, L.; Newton, A. Effects of climate change on the potential species richness of Mesoamerican forests. Biotropica 2012, 44, 284–293. [Google Scholar] [CrossRef]
- Koo, K.; Kong, W.; Nibbelink, N.; Hopkinson, C.; Lee, J. Potential effects of climate change on the distribution of cold-tolerant evergreen broadleaved woody plants in the Korean peninsula. PLoS ONE 2015, 10, e0134043. [Google Scholar] [CrossRef]
- Roux le, P.; Virtanen, R.; Heikkinen, R.; Luoto, M. Biotic interactions affect the elevational ranges of high-latitude plant species. Ecography 2012, 35, 1048–1056. [Google Scholar] [CrossRef]
- Zellweger, F.; Braunisch, V.; Morsdorf, F.; Baltensweiler, A.; Abegg, M.; Roth, T.; Bugmann, H.; Bollmann, K. Disentangling the effects of climate, topography, soil and vegetation on stand-scale species richness in temperate forests. For. Ecol. Manag. 2015, 349, 36–44. [Google Scholar] [CrossRef]
- Paudel, S.; Waeber, P.; Simard, S.; Innes, J.; Nitschke, C. Multiple factors influence plant richness and diversity in the cold and dry boreal forest of southwest Yukon, Canada. Plant Ecol. 2016, 217, 505–519. [Google Scholar] [CrossRef]
- Joshi, M.; Rawat, Y. Net primary productivity and species diversity of herbaceous vegetation in banj-oak (Quercus leucotrichophora A. Camus) forest in Kumaun Himalaya, India. J. Mt. Sci. 2011, 8, 787–793. [Google Scholar] [CrossRef]
- Kale, M.; Roy, P. Net primary productivity estimation and its relationship with tree diversity for tropical dry deciduous forests of central India. Biodivers. Conserv. 2012, 21, 1199–1214. [Google Scholar] [CrossRef]
- Cowles, J.M.; Wragg, P.D.; Wright, A.J.; Powers, J.S.; Tilman, D. Shifting grassland plant community structure drives positive interactive effects of warming and diversity on aboveground net primary productivity. Glob. Chang. Biol. 2016, 22, 741–749. [Google Scholar] [CrossRef]
- Charbonnier, F.; Roupsard, O.; le Maire, G.; Guillemot, J.; Casanoves, F.; Lacointe, A.; Vaast, P.; Allinne, C.; Audebert, L.; Cambou, A.; et al. Increased light-use efficiency sustains net primary productivity of shaded coffee plants in agroforestry system. Plant Cell Environ. 2017, 40, 1592–1608. [Google Scholar] [CrossRef]
- Zhu, Q.; Zhao, J.; Zhu, Z.; Zhang, H.; Zhang, Z.; Guo, X.; Bi, Y.; Sun, L. Remotely sensed estimation of net primary productivity (NPP) and its spatial and temporal variations in the greater Khingan mountain region, China. Sustainability 2017, 9, 1213. [Google Scholar] [CrossRef]
- Jiao, W.; Chen, Y.; Li, W.; Zhu, C.; Li, Z. Estimation of net primary productivity and its driving factors in the Ili River Valley, China. J. Arid Land 2018, 10, 781–793. [Google Scholar] [CrossRef]
- Austrheim, G.; Eriksson, O. Plant species diversity and grazing in the Scandinavian mountains - patterns and processes at different spatial scales. Ecography 2001, 24, 683–695. [Google Scholar] [CrossRef]
- Moeslund, J.; Arge, L.; Bocher, P.; Dalgaard, T.; Svenning, J. Topography as a driver of local terrestrial vascular plant diversity patterns. Nord. J. Bot. 2013, 31, 129–144. [Google Scholar] [CrossRef]
- Givnish, T. On the causes of gradients in tropical tree diversity. J. Ecol. 1999, 87, 193–210. [Google Scholar] [CrossRef]
- Zilliox, C.; Gosselin, F. Tree species diversity and abundance as indicators of understory diversity in French mountain forests: Variations of the relationship in geographical and ecological space. For. Ecol. Manag. 2014, 321, 105–116. [Google Scholar] [CrossRef]
- Rahbek, C. The elevational gradient of species richness—A uniform pattern. Ecography 1995, 18, 200–205. [Google Scholar] [CrossRef]
- Rahbek, C. The role of spatial scale and the perception of large-scale species-richness patterns. Ecol. Lett. 2005, 8, 224–239. [Google Scholar] [CrossRef]
- Otypkova, Z.; Chytry, M.; Tichy, L.; Pechanec, V.; Jongepier, J.W.; Hajek, O. Floristic diversity patterns in the White Carpathians Biosphere Reserve, Czech Republic. Biologia 2011, 66, 266–274. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, M.; Mian, R. Effects of elevation and disturbance gradients on forest diversity in the Wulingshan Nature Reserve, North China. Environ. Earth Sci. 2016, 75, 904. [Google Scholar] [CrossRef]
- Syfert, M.; Brummitt, N.; Coomes, D.; Bystriakova, N.; Smith, M. Inferring diversity patterns along an elevation gradient from stacked SDMs: A case study on Mesoamerican ferns. Environ. Earth Sci. 2018, 16. [Google Scholar] [CrossRef]
- Grime, J. Competitive exclusion in herbaceous vegetation. Nature 1973, 242, 344–347. [Google Scholar] [CrossRef]
- Connell, J. Diversity in tropical rain forests and coral reefs—High diversity of trees and corals is maintained only in a non-equilibrium state. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef]
- Leathwick, J.; Elith, J.; Francis, M.; Hastie, T.; Taylor, P. Variation in demersal fish species richness in the oceans surrounding New Zealand: An analysis using boosted regression trees. Mar. Ecol. Prog. Ser. 2006, 321, 267–281. [Google Scholar] [CrossRef]
- Mayor, S.J.; Cahill, J.F., Jr.; He, F.; Solymos, P.; Boutin, S. Regional boreal biodiversity peaks at intermediate human disturbance. Nat. Commun. 2012, 3, 1142. [Google Scholar] [CrossRef]
- Casas, C.; Ninot, J. Correlation between species composition and soil properties in the pastures of Plana de Vic (Catalonia, Spain). Acta Bot. Barc. 2003, 49, 291–310. [Google Scholar]
- Başnou, C.; Pino, J.; Šmilauer, P. Effect of grazing on grasslands in the Western Romanian Carpathians depends on the bedrock type. Preslia 2009, 81, 91–104. [Google Scholar]
- Sanaei, A.; Li, M.; Ali, A. Topography, grazing, and soil textures control over rangelands’ vegetation quantity and quality. Sci. Total Environ. 2019, 697, 1341523. [Google Scholar] [CrossRef]
- Jimenez, I.; Distler, T.; Jorgensen, P. Estimated plant richness pattern across northwest South America provides similar support for the species-energy and spatial heterogeneity hypotheses. Ecography 2009, 32, 433–448. [Google Scholar] [CrossRef]
- Jocque, M.; Field, R.; Brendonck, L.; De Meester, L. Climatic control of dispersal-ecological specialization trade-offs: A metacommunity process at the heart of the latitudinal diversity gradient? Glob. Ecol. Biogeogr. 2010, 19, 244–252. [Google Scholar] [CrossRef]
- Oberle, B.; Grace, J.; Chase, J. Beneath the veil: Plant growth form influences the strength of species richness-productivity relationships in forests. Glob. Ecol. Biogeogr. 2009, 18, 416–425. [Google Scholar] [CrossRef]
- Bartels, S.; Chen, H. Interactions between overstorey and understorey vegetation along an overstorey compositional gradient. J. Veg. Sci. 2013, 24, 543–552. [Google Scholar] [CrossRef]
- Palik, B.; Mitchell, R.; Hiers, J. Modeling silviculture after natural disturbance to sustain biodiversity in the longleaf pine (Pinus palustris) ecosystem: Balancing complexity and implementation. For. Ecol. Manag. 2002, 155, 347–356. [Google Scholar] [CrossRef]
- Johnstone, J.; Chapin, F. Fire interval effects on successional trajectory in boreal forests of northwest Canada. Ecosystems 2006, 9, 268–277. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, B.; Qian, Z. Functional diversity of Cercidiphyllum japonicum, communities in the Shennongjia Reserve, central China. J. For. Res. 2015, 26, 171–177. [Google Scholar] [CrossRef]
- Marialigeti, S.; Tinya, F.; Bidlo, A.; Odor, P. Environmental drivers of the composition and diversity of the herb layer in mixed temperate forests in Hungary. Plant Ecol. 2016, 217, 549–563. [Google Scholar] [CrossRef]
- Lehosmaa, K.; Jyvasjarvi, J.; Virtanen, R.; Ilmonen, J.; Saastamoinen, J.; Muotka, T. Anthropogenic habitat disturbance induces a major biodiversity change in habitat specialist bryophytes of boreal springs. Biol. Conserv. 2017, 215, 169–178. [Google Scholar] [CrossRef]
- Marcilio-Silva, V.; Zwiener, V.; Marques, M.C.M. Metacommunity structure, additive partitioning and environmental drivers of woody plants diversity in the Brazilian Atlantic Forest. Divers. Distrib. 2017, 23, 1110–1119. [Google Scholar] [CrossRef]
- Guo, Q.; Ricklefs, R.; Cody, M. Vascular plant diversity in eastern Asia and North America: Historical and ecological explanations. Bot. J. Linn. Soc. 1998, 128, 123–136. [Google Scholar] [CrossRef]
- Kullman, L. Palaeoecological, biogeographical and palaeoclimatological implications of early Holocene immigration of Larix sibirica Ledeb. in the Scandes mountains, Sweden. Glob. Ecol. Biogeogr. Lett. 1998, 7, 181–188. [Google Scholar] [CrossRef]
- Whittaker, R.; Field, R. Tree species richness modelling: An approach of global applicability. Oikos 2000, 89, 399–402. [Google Scholar] [CrossRef]
- Hall, A.; Miller, A.; Leggett, H.; Roxburgh, S.; Buckling, A.; Shea, K. Diversity-disturbance relationships: Frequency and intensity interact. Biol. Lett. 2012, 8, 768–771. [Google Scholar] [CrossRef]
- Kershaw, H.; Mallik, A. Predicting plant diversity response to disturbance: Applicability of the intermediate disturbance hypothesis and mass ratio hypothesis. Crit. Rev. Plant Sci. 2013, 32, 383–395. [Google Scholar] [CrossRef]
- O’Brien, E. Water-energy dynamics, climate, and prediction of woody plant species richness: An interim general model. J. Biogeogr. 1998, 25, 379–398. [Google Scholar] [CrossRef]
- Kerr, J.; Packer, L. Habitat heterogeneity as a determinant of mammal species richness in high-energy regions. Nature 1997, 385, 252–254. [Google Scholar] [CrossRef]
- Francis, A.P.; Currie, D.J. A globally consistent richness-climate relationship for angiosperms. Am. Nat. 2003, 161, 523–536. [Google Scholar] [CrossRef]
- Field, R.; Hawkins, B.A.; Cornell, H.V.; Currie, D.J.; Diniz-Filho, J.A.F.; Guegan, J.-F.; Kaufman, D.M.; Kerr, J.T.; Mittelbach, G.G.; Oberdorff, T.; et al. Spatial species-richness gradients across scales: A meta-analysis. J. Biogeogr. 2009, 36, 132–147. [Google Scholar] [CrossRef]
- Gentili, R.; Armiraglio, S.; Sgorbati, S.; Baroni, C. Geomorphological disturbance affects ecological driving forces and plant turnover along an altitudinal stress gradient on alpine slopes. Plant Ecol. 2013, 214, 571–586. [Google Scholar] [CrossRef]
- Chinese Academy of Sciences. Flora of China; Beijing Science Press: Beijing, China, 1999; Available online: http://foc.iplant.cn/ (accessed on 10 March 2018).
- Fischer, C.; Leimer, S.; Roscher, C.; Ravenek, J.; de Kroon, H.; Kreutziger, Y.; Baade, J.; Bessler, H.; Eisenhauer, N.; Weigelt, A.; et al. Plant species richness and functional groups have different effects on soil water content in a decade-long grassland experiment. J. Ecol. 2019, 107, 127–141. [Google Scholar] [CrossRef]
- Sperandii, M.; Bazzichetto, M.; Acosta, A.; Bartak, V.; Malavasi, M. Multiple drivers of plant diversity in coastal dunes: A Mediterranean experience. Sci. Total Environ. 2019, 652, 1435–1444. [Google Scholar] [CrossRef]
- China Meteorological Administration, China, 1949. Available online: http://www.cma.gov.cn/ (accessed on 30 November 2018).
- Pitman, N.C.A.; Terborgh, J.; Silman, M.R.; Nuez, P. Tree species distributions in an upper Amazonian forest. Ecology 1999, 80, 2651–2661. [Google Scholar] [CrossRef]
- Curtis, J.; McIntosh, R. An upland forest continuum in the prairie-forest border region of Wisconsin. Ecology 1951, 32, 476–496. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. Vienna, Austria. 2017. Available online: http://www.R-project.org (accessed on 2 January 2019).
- Friedman, J. Greedy function approximation: A gradient boosting machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.; Anderson, R.; Dudik, M.; Ferrier, S.; Guisan, A.; Hijmans, R.; Huettmann, F.; Leathwick, J.; Lehmann, A.; et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Ridgeway, G. Gbm: Generalized Boosted Regression Models. R package, Version 2.1.5. 2019. Available online: https://cran.r-project.org/web/packages/gbm/ (accessed on 19 March 2019).
- Friedman, J.; Meulman, J. Multiple additive regression trees with application in epidemiology. Stat. Med. 2003, 22, 1365–1381. [Google Scholar] [CrossRef]
- Hastie, T.; Tibshirani, R.; Friedman, J.H. The Elements of Statistical Learning: Data Mining, Inference and Prediction; Springer: New York, NY, USA, 2001. [Google Scholar]
- Tordoni, E.; Napolitano, R.; Maccherini, S.; Da Re, D.; Bacaro, G. Ecological drivers of plant diversity patterns in remnants coastal sand dune ecosystems along the northern Adriatic coastline. Ecol. Res. 2018, 33, 1157–1168. [Google Scholar] [CrossRef]
- Svensson, J.; Lindegarth, M.; Jonsson, P.; Pavia, H. Disturbance-diversity models: What do they really predict and how are they tested? Proc. R. Soc. B Biol. Sci. 2012, 279, 2163–2170. [Google Scholar] [CrossRef]
- Chen, S.; Jiang, G.; Ouyang, Z.; Xu, W.; Xiao, Y. Relative importance of water, energy, and heterogeneity in determining regional pteridophyte and seed plant richness in China. J. Syst. Evol. 2011, 49, 95–107. [Google Scholar] [CrossRef]
- Propastin, P.; Ibrom, A.; Knohl, A.; Erasmi, S. Effects of canopy photosynthesis saturation on the estimation of gross primary productivity from MODIS data in a tropical forest. Remote Sens. Environ. 2012, 121, 252–260. [Google Scholar] [CrossRef]
- Wright, D. Species-energy theory—An extension of species-area theory. Oikos 1983, 41, 496–506. [Google Scholar] [CrossRef]
- Gracia, M.; Montane, F.; Pique, J.; Retana, J. Overstory structure and topographic gradients determining diversity and abundance of understory shrub species in temperate forests in central Pyrenees (NE Spain). For. Ecol. Manag. 2007, 242, 391–397. [Google Scholar] [CrossRef]
- Kambiz Abrari, V.; Hamid, J.; Mohammad Reza, P.; Kambiz, E.; Alireza, M. Effect of canopy gap size and ecological factors on species diversity and beech seedlings in managed beech stands in Hyrcanian forests. J. For. Res. 2012, 23, 217–222. [Google Scholar]
- Brosofske, K.; Chen, J.; Crow, T.R. Understory vegetation and site factors: Implications for a managed Wisconsin landscape. For. Ecol. Manag. 2001, 146, 75–87. [Google Scholar] [CrossRef]
- Ferris, R.; Humphrey, J. A review of potential biodiversity indicators for application in British forests. Forestry 1999, 72, 313–328. [Google Scholar] [CrossRef]
- Tarrega, R.; Calvo, L.; Taboada, A.; Marcos, E.; Marcos, J.A. Do mature pine plantations resemble deciduous natural forests regarding understory plant diversity and canopy structure in historically modified landscapes? Eur. J. For. Res. 2011, 130, 949–957. [Google Scholar] [CrossRef]
- Shea, K.; Roxburgh, S.H.; Rauschert, E.S.J. Moving from pattern to process: Coexistence mechanisms under intermediate disturbance regimes. Ecol. Lett. 2004, 7, 491–508. [Google Scholar] [CrossRef]
- Bartels, S.; Chen, H. Is understory plant species diversity driven by resource quantity or resource heterogeneity? Ecology 2010, 91, 1931–1938. [Google Scholar] [CrossRef]
- Tinya, F.; Marialigeti, S.; Kiraly, I.; Nemeth, B.; Odor, P. The effect of light conditions on herbs, bryophytes and seedlings of temperate mixed forests in arsg, Western Hungary. Plant Ecol. 2009, 204, 69–81. [Google Scholar] [CrossRef]
- Lochhead, K.; Comeau, P. Relationships between forest structure, understory light and regeneration in complex Douglas-fir dominated stands in south-eastern British Columbia. For. Ecol. Manag. 2012, 284, 12–22. [Google Scholar] [CrossRef]
- Heithecker, T.D.; Halpern, C.B. Variation microclimate associated with dispersed-retention harvests in coniferous forests of western Washington. For. Ecol. Manag. 2006, 226, 60–71. [Google Scholar] [CrossRef]
- Barbier, S.; Gosselin, F.; Balandier, P. Influence of tree species on understory vegetation diversity and mechanisms involved—A critical review for temperate and boreal forests. For. Ecol. Manag. 2008, 254, 1–15. [Google Scholar] [CrossRef]
- Piedallu, C.; Gegout, J.-C.; Perez, V.; Lebourgeois, F. Soil water balance performs better than climatic water variables in tree species distribution modelling. Glob. Ecol. Biogeogr. 2013, 22, 470–482. [Google Scholar] [CrossRef]
- Martin-Alcon, S.; Coll, L.; Ameztegui, A. Diversifying sub-Mediterranean pinewoods with oak species in a context of assisted migration: Responses to local climate and light environment. Appl. Veg. Sci. 2016, 19, 254–266. [Google Scholar] [CrossRef]
- Hunter, A.F.; Aarssen, L.W. Plants helping plants. Bioscience 1988, 38, 34–40. [Google Scholar] [CrossRef]
- Bertness, M.D.; Callaway, R. Positive interactions in communities. Trends Ecol. Evol. 1994, 9, 191–193. [Google Scholar] [CrossRef]
- Callaway, R.M. Positive interactions among plants. Bot. Rev. 1995, 61, 306–349. [Google Scholar] [CrossRef]
- Lortie, C.J.; Brooker, R.W.; Choler, P.; Kikvidze, Z.; Michalet, R.; Pugnaire, F.I.; Callaway, R.M. Rethinking plant community theory. Oikos 2004, 107, 433–438. [Google Scholar] [CrossRef]
- Miller, T.E. Direct and indirect species interactions in an early old-field plant community. Am. Nat. 1994, 143, 1007–1025. [Google Scholar] [CrossRef]
- Callaway, R.M.; Pennings, S.C. Invasive plants versus their new and old neighbors: A mechanism for exotic invasion. Science 2000, 290, 521–523. [Google Scholar] [CrossRef]
- Martins, K.; Marques, M.; dos Santos, E.; Marques, R. Effects of soil conditions on the diversity of tropical forests across a successional gradient. For. Ecol. Manag. 2015, 349, 4–11. [Google Scholar] [CrossRef]
- Chipman, S.; Johnson, E. Understory vascular plant species diversity in the mixed wood boreal forest of western Canada. Ecol. Appl. 2002, 12, 588–601. [Google Scholar] [CrossRef]
- Halpern, C.B.; Lutz, J.A. Canopy closure exerts weak controls on understory dynamics: A 30-year study of overstory-understory interactions. Ecol. Monogr. 2013, 83, 221–237. [Google Scholar] [CrossRef]
| Forest Type | Abbr. | FT | IV | ||||
|---|---|---|---|---|---|---|---|
| Larix gmelinii forest | LGF | A | LG | BP | PS | PD | PK2 |
| 0.58 | 0.17 | 0.03 | 0.02 | 0.02 | |||
| Larix olgensis forest | LOF | B | LO | UD | QM | PK1 | BP |
| 0.65 | 0.04 | 0.04 | 0.04 | 0.03 | |||
| Mixed broadleaved forest | MBF | C | QM | TA | AM | JM | BD |
| 0.26 | 0.07 | 0.06 | 0.05 | 0.04 | |||
| Populus davidiana and Betula platyphylla forest | PBF | D | BP | PD | LG | QM | UD |
| 0.39 | 0.21 | 0.04 | 0.04 | 0.04 | |||
| Pinus koraiensis broadleaved forest | PKF | E | PK1 | AN | TA | AM | BP |
| 0.29 | 0.09 | 0.06 | 0.05 | 0.05 | |||
| Pinus tabulaeformis broadleaved forest | PTF | F | PT | QM | FR | AS | UM |
| 0.68 | 0.10 | 0.03 | 0.03 | 0.02 | |||
| Diversity | Contribution of Variables (%) | Parameters | |||||||
|---|---|---|---|---|---|---|---|---|---|
| AMT | SSD | AP | DBH (SD) | FT | EL | SL | TC | R2 | |
| Tree richness | 52.39 | 11.29 | 9.61 | 17.75 | 5.50 | 2.56 | 0.89 | 2 | 0.55 |
| Shannon wiener index | 30.37 | 6.84 | 4.80 | 32.45 | 19.64 | 4.72 | 1.17 | 2 | 0.51 |
| Simpson dominance index | 24.34 | 8.20 | 8.04 | 33.73 | 19.33 | 3.38 | 3.03 | 2 | 0.43 |
| Total richness | 53.79 | 19.03 | 10.91 | 8.16 | 5.37 | 1.47 | 1.27 | 2 | 0.49 |
| Shrub richness | 36.22 | 28.48 | 8.16 | 9.48 | 7.82 | 3.89 | 5.95 | 2 | 0.37 |
| Herb richness | 47.79 | 20.51 | 13.64 | 4.26 | 6.73 | 4.85 | 2.21 | 2 | 0.39 |
| Treelet richness | 61.05 | 10.97 | 9.12 | 6.76 | 5.18 | 5.14 | 1.78 | 2 | 0.45 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, Y.; Han, S.; Zhang, J.; Chen, Z.; Wang, W.; Feng, Y.; Jiang, Y.; Geng, S. Temperature-Dominated Driving Mechanisms of the Plant Diversity in Temperate Forests, Northeast China. Forests 2020, 11, 227. https://doi.org/10.3390/f11020227
Gu Y, Han S, Zhang J, Chen Z, Wang W, Feng Y, Jiang Y, Geng S. Temperature-Dominated Driving Mechanisms of the Plant Diversity in Temperate Forests, Northeast China. Forests. 2020; 11(2):227. https://doi.org/10.3390/f11020227
Chicago/Turabian StyleGu, Yue, Shijie Han, Junhui Zhang, Zhijie Chen, Wenjie Wang, Yue Feng, Yangao Jiang, and Shicong Geng. 2020. "Temperature-Dominated Driving Mechanisms of the Plant Diversity in Temperate Forests, Northeast China" Forests 11, no. 2: 227. https://doi.org/10.3390/f11020227
APA StyleGu, Y., Han, S., Zhang, J., Chen, Z., Wang, W., Feng, Y., Jiang, Y., & Geng, S. (2020). Temperature-Dominated Driving Mechanisms of the Plant Diversity in Temperate Forests, Northeast China. Forests, 11(2), 227. https://doi.org/10.3390/f11020227






