1. Introduction
Forests are essential to the provision of ecosystem services, products and environmental assets of a country or region, providing important incomes when globally accounting for all indicators [
1]. As a result, forests, and particularly conifer forests, have been widely exploited and transformed by different human activities, namely forestry, e.g., cultivation, management [
2], to increase some of these income sources. Furthermore, forests have been affected by land-use changes, e.g., agriculture, land development, or different natural hazards, e.g., forest fires, disturbances related to climate change, resulting in an enhanced vulnerability of many populations or even species. Therefore, the need for conservation and sustainable use of forest genetic resources has been increasingly claimed over the last decades [
3].
There is an urgent need to conserve genetic resources and to support the resilience of conifer species facing expected changes and threats. Past changes could have already affected the adaptability of the populations to the ongoing rapid climatic changes [
4], but also could have resulted in adaptive lags of some European forest tree species [
5].
Reforestation or restoration are major forestry activities, where seeds or plants are used to establish a future forest. For the last decades, there has been an increasing concern to improve resilience of future forests. A key tool to achieve this aim is the use of genetically-variable material, as this is related to future adaptation and provision of goods and services [
6]. As a consequence, great efforts are being done to define and take into account genetic features of the material deployed in forestry.
Selecting the most productive seed source for a given area has been a traditional approach by forest managers. At present, information on genetic variability and the predictions provided by climate change science enable different seed sourcing strategies to maximize the future adaptation and resilience of our forests [
7,
8,
9,
10,
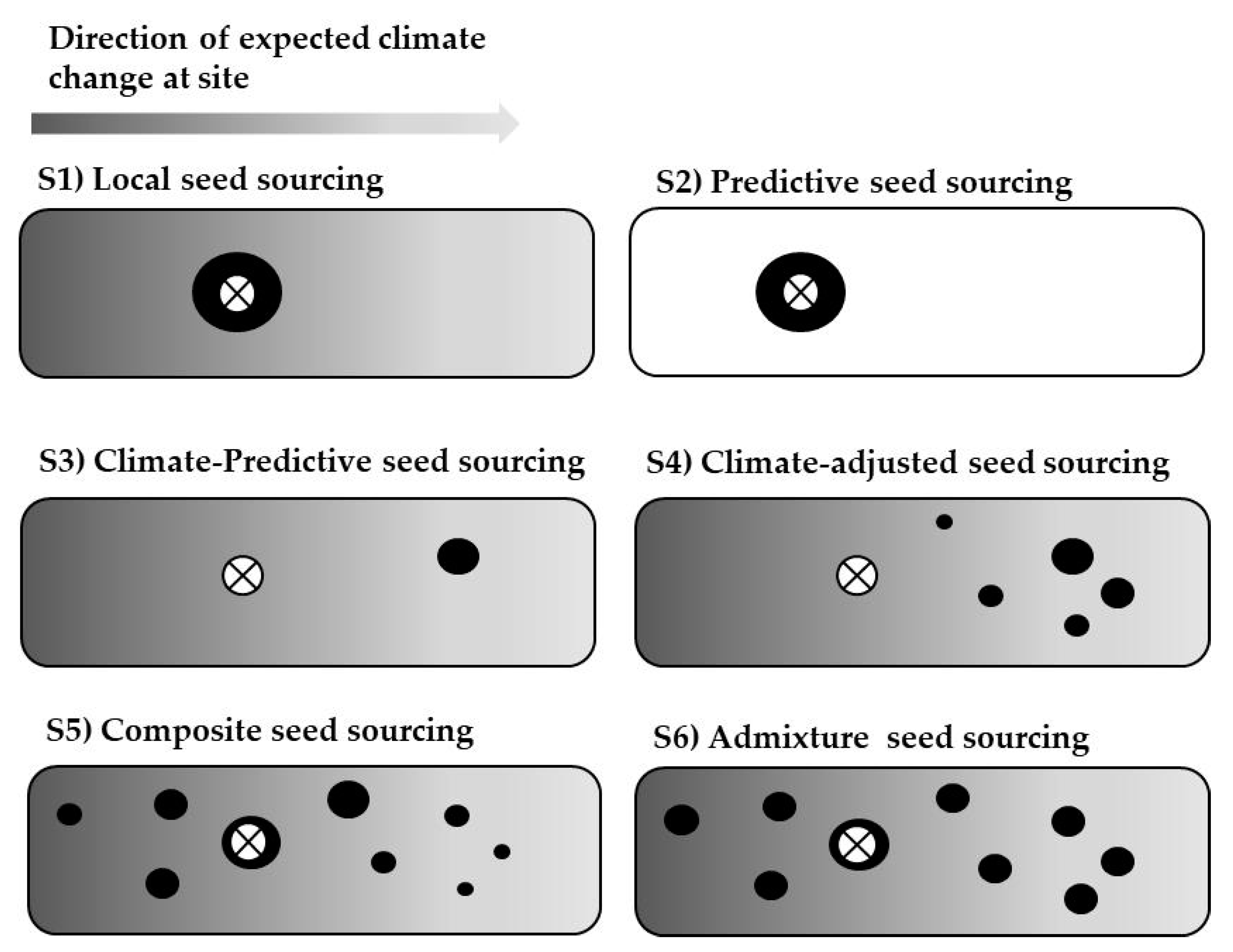
11]. Among the possible strategies, local, predicted, composite, admixture, and climate-adjusted seed sourcing have been widely promoted (see Materials and Methods for a detailed description).
These different strategies are difficult to test and compare against each other in forest conifers. Their long lifespan and wide distribution ranges impose some limitations in field tests in regards to obtaining seedlots based on the different strategies, establishing the material in common garden tests, and recording the results after a significant testing period. However, multi-site provenance tests have demonstrated their value to address many evolutionary and ecological questions related to adaptation [
12,
13], as different populations can be studied under common environmental conditions, and therefore taking advantage to simulate different seed sourcing strategies based on the results at a given time after planting.
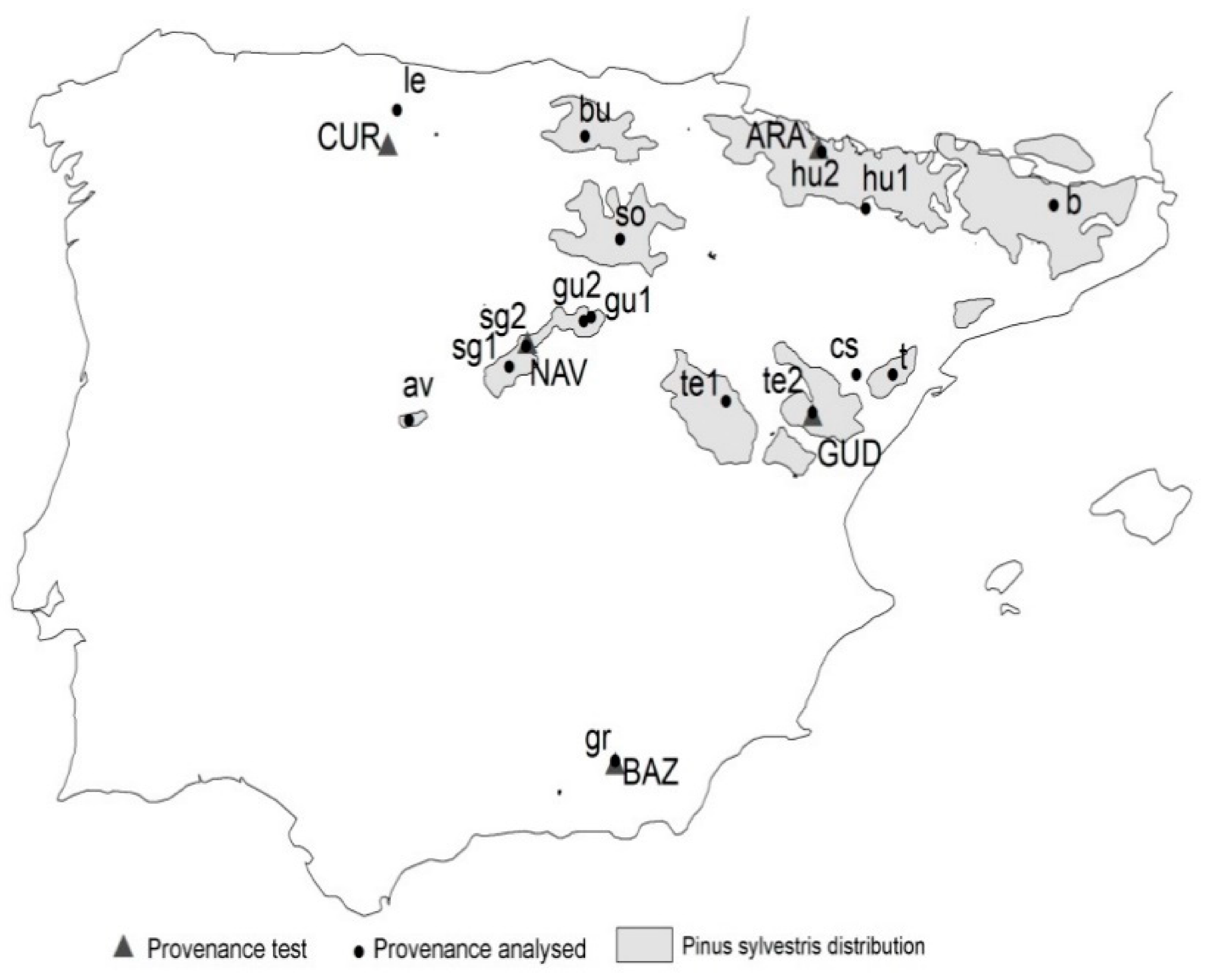
Here, we used Scots pine (Pinus sylvestris L.) provenance tests located in Spain as a study case. We departed from the hypothesis that provenance tests constitute a representative sample to analyze the performance of different populations of a given species. Then, we used a set of Scots pine provenance tests covering the natural range of the species in Spain to evaluate different seed sourcing strategies under climate change scenarios.
Scots pine in Spain is at the southern edge of its distribution [
14], and this in turn has implications for the fragmentation of the populations and genetic differentiation among populations [
15,
16,
17], but also for the genetic variation in adaptive traits [
17,
18]. The species has a low degree of domestication and remarkable phenotypic and genetic differences in important traits among populations, despite high levels of gene flow [
19]. Scots pine is a very important species in afforestation and restoration programs in Spain. It is the third species in Spain in terms of planted area, with 20,000 kg/seed/year and 1218 ha/year and with 17 regions of provenance for marketing of reproductive material [
20]. Previous studies have reported genetic variation for traits related to growth, survival and branching among the Spanish provenances and a significant genotype–environment interaction for height and diameter [
18]. Therefore, the study area provides an excellent case study to address how the species genetic variation in growth traits affects the outcome of seed sourcing strategies. In addition, it can potentially inform us of the implications for genetic conservation at the distribution limit of a widely distributed species.
The aims of this work are: (i) to analyse the levels of variation of the species for height, diameter and survival, (ii) to analyse different seed sourcing strategies (local, predicted composite, admixture and climate-adjusted) under different environments and climate change scenarios, and (iii) to propose measures for the conservation and sustainable use of the populations in Spain. Firstly, we analyzed the gain of the different strategies in traits related to adaptation (height growth and their annual components, and survival) in five different provenance tests. Secondly, we integrated this information and the existing knowledge regarding Scots pine, to analyse the strategies for the conservation of its genetic resources. We believe this case study would be useful for other widely distributed species in their distribution edge.
The main aim of this work is to experimentally test different seed sourcing strategies under the climate change high emission scenario RCP 8.5 (business as usual) in order to increase adaptation and resilience of future forests. Our conclusion is that, for Scots pine, a forest tree species in the rear-edge of distribution, the proposed methods do not provide better results than the ones traditionally used in forestry (local and predictive methods). We show that depending of the structure of variation of the species and the future climatic predictions, the theoretical expectations of the climate-adjusted, composite and admixture methods would not be met.
4. Discussion
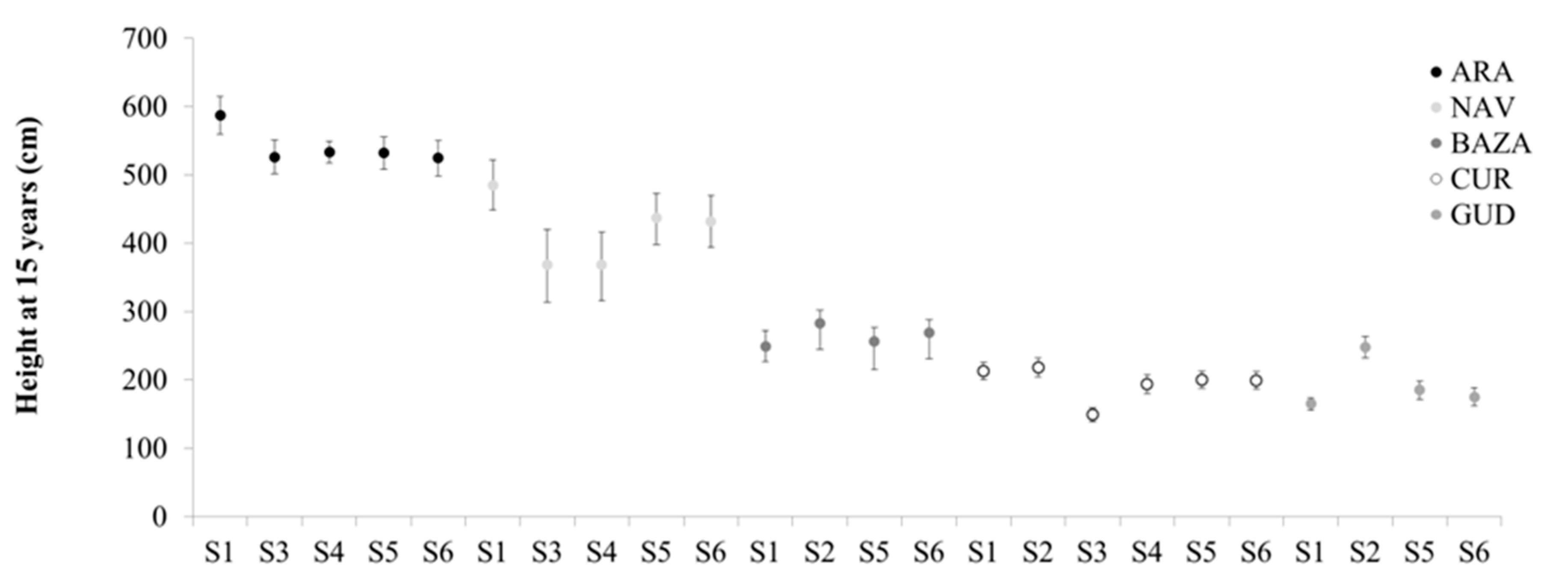
This paper analyses for the first time the outcome of different seed sourcing strategies under climate change scenarios in Scots pine by using information on provenance tests established in multi-site common environments. The age of evaluation (15 years after planting) is an adequate period for the material to acclimatise to the local conditions of each experimental site. The results also indicate the stability of the estimation across years, and the high correlation among the traits considered. These traits are important to estimate the production and future adaptation of the species, and therefore are essential in the selection of seed sources in afforestation and restoration programs.
The interpretation of the results should consider different aspects of the study, related to the type of adaptation/acclimation evaluated, namely phenotypic plasticity. In addition, the evaluation period and its relationship with the expected climatic changes in the future, the area of study and the influence of the sample sizes should be considered in the estimation of seed sourcing strategies.
Phenotypic plasticity is a process mainly related to the future response of the species to different climatic conditions when the plants are already established in common garden experiments. The results indicate the existence of a significant site effect for all the traits considered, and a significant provenance and site x provenance interaction for height and diameter, but not for survival. This is in accordance with previous results reported with the same material at age 5 [
18]. These results confirm the importance of phenotypic plasticity in growth traits. That is, the acclimation to site conditions at the species level as a whole, but also the differences in phenotypic plasticity at the provenance level [
31].
A second process of adaptation related to the survival of better adapted trees is of minor importance in this study. We need to take into consideration that mortality, like in most provenance tests, was artificially reduced by planting nursery-cultivated healthy seedlings, and therefore blurring any selection effect at very early stages of development (1–2 years). We have reduced the mortality artificially in this stage, because as in most of the provenance tests, the focus was placed on the evaluation of established seedlings during the duration of the experiment. Firstly, the selected proportion of trees, indicated by the survival in the different sites, differed among sites, with values close to 0.44 in one of the sites (GUD), and close to 0.90 in the other sites. Therefore the selection intensity was too low—compared to that observed at the seedling stage in some conifers [
32] and also in artificial selection programs of the species [
33]—to expect significant future adaptation. Secondly, natural selection will operate within populations, where standing genetic variation (intra-population genetic variation for the strategies based on one population, or a combination of among and within population genetic variation for the strategies based on the mix of different populations) should take into consideration such levels of variation [
4,
34,
35,
36] that are quite important in Scots pine [
37]. The effects of this selection would be observed in a second generation from the material established in the sites. In Scots pine, this period is around 120 years in the study area [
38], similar to the furthest climatic scenario predicted (2100). Thirdly, because there are no differences among provenances in survival (there was no bias in provenance selection due to mortality), seed sourcing strategies will not depend on these differences for the future adaptation of this species at its southern range.
While the time scale of this work comparing strategies of seed sourcing was set at 2050, it is noteworthy to warn that no provenance will be suitable for the next century if the climate change follows the high emission scenario. This situation has severe implications and negative consequences for the conservation of genetic diversity, not only for Scots pine but also for a wider set of biodiversity when losing a key component of the forest habitat.
Scots pine has a different pattern of variation at its northern edge of distribution, where a clinal pattern has been detected for neutral and adaptive traits, compared to the rear edge of distribution in Spain, where the populations are highly fragmented and genetic drift may have played an important role in the distribution of neutral and adaptive variation [
16,
17,
39]. These contrasting patterns influence seed transfer recommendations. In northern countries like Sweden and Finland, a movement of seeds from southern to northern areas has been suggested based on the future performance [
40,
41]. In Spain, however, this strategy is not recommended as local provenances are only showing the best performance in the areas where the conditions for local adaptation [
42] are met. These are ARA and NAV sites where large population sizes and extensive gene flow is detected [
21] in contrast to other marginal populations where genetic drift could have played a major role [
43,
44]. Therefore, local seed sourcing methods are superior to the other methods in only two of the sites.
Predictive seed sourcing is based on the selection of the best performing provenance (whenever it is not local), and therefore it is based on the results already available. This is the method most used based on provenance tests. By definition, this method will always provide the best results, sometimes paired with other single-source strategies like local or climate-predictive. However, there are two major drawbacks that limit its practical application. First, as it is not known a priori which provenance will be the best at each particular afforestation or restoration site, provenance tests with a comprehensive set of provenances need to be extensively replicated. Second, this is a post hoc method, for which phenotypic data collected several years after planting are needed.
Climate-based seed sourcing methods behave intermediately compared to the local/predictive seed sourcing methods. The predicted effects of climate change on the species in the Iberian peninsula include an increase in both tree growth and mortality [
45]. When considering intra-population variation, data-driven models indicate that Scots pine populations from southern Spain would have higher relative survival in northern territories (that will be warmer) under future climate scenarios than under current conditions, and Scots pine populations from the northern Iberian Range would have the largest decrease in suitable area and the lowest survival over different climatic conditions [
46]. The provenance tests reported in this study have been measured in the period 1995–2005, where the climates in the different sites have followed the trend expected in the climate change scenarios (
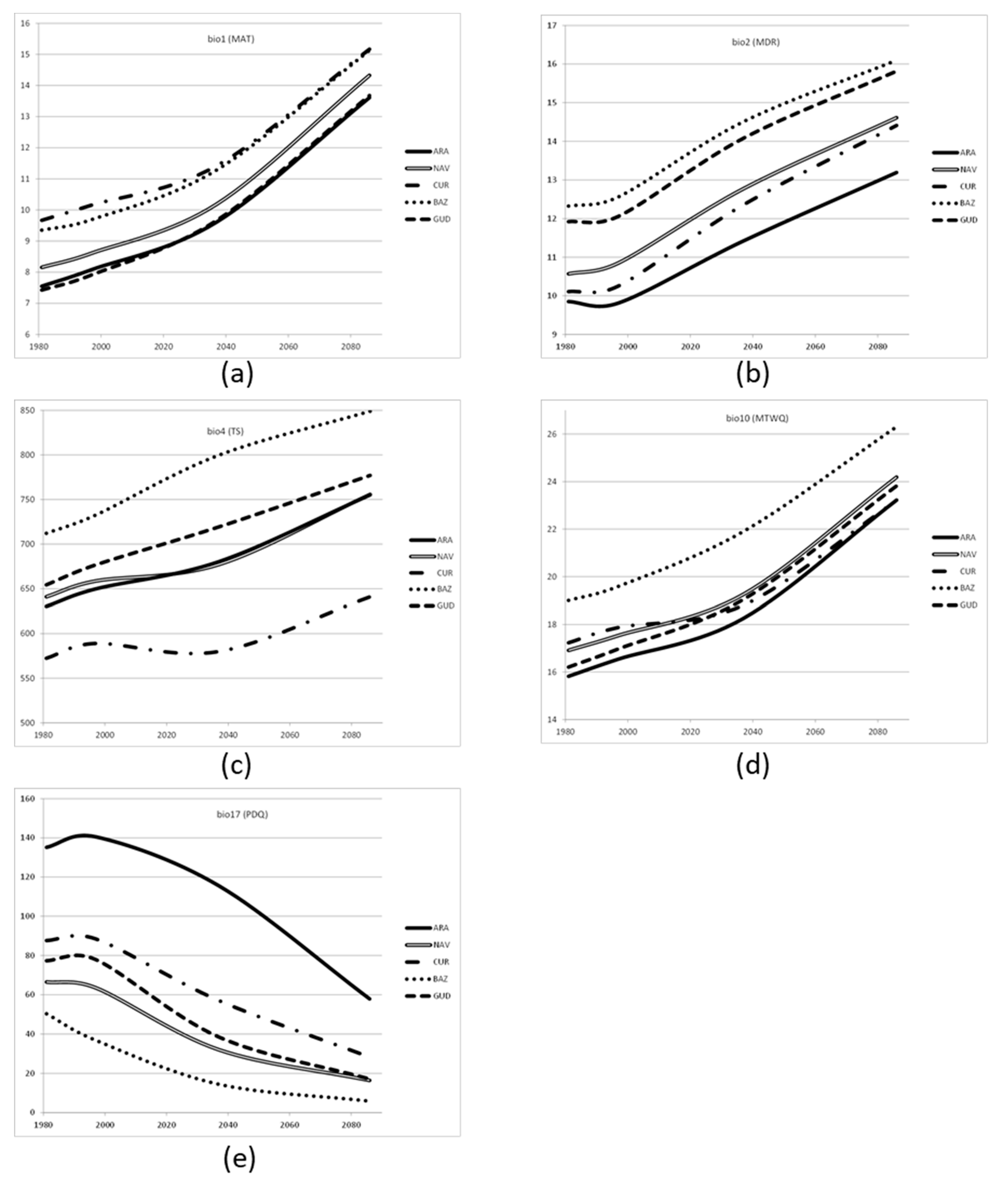
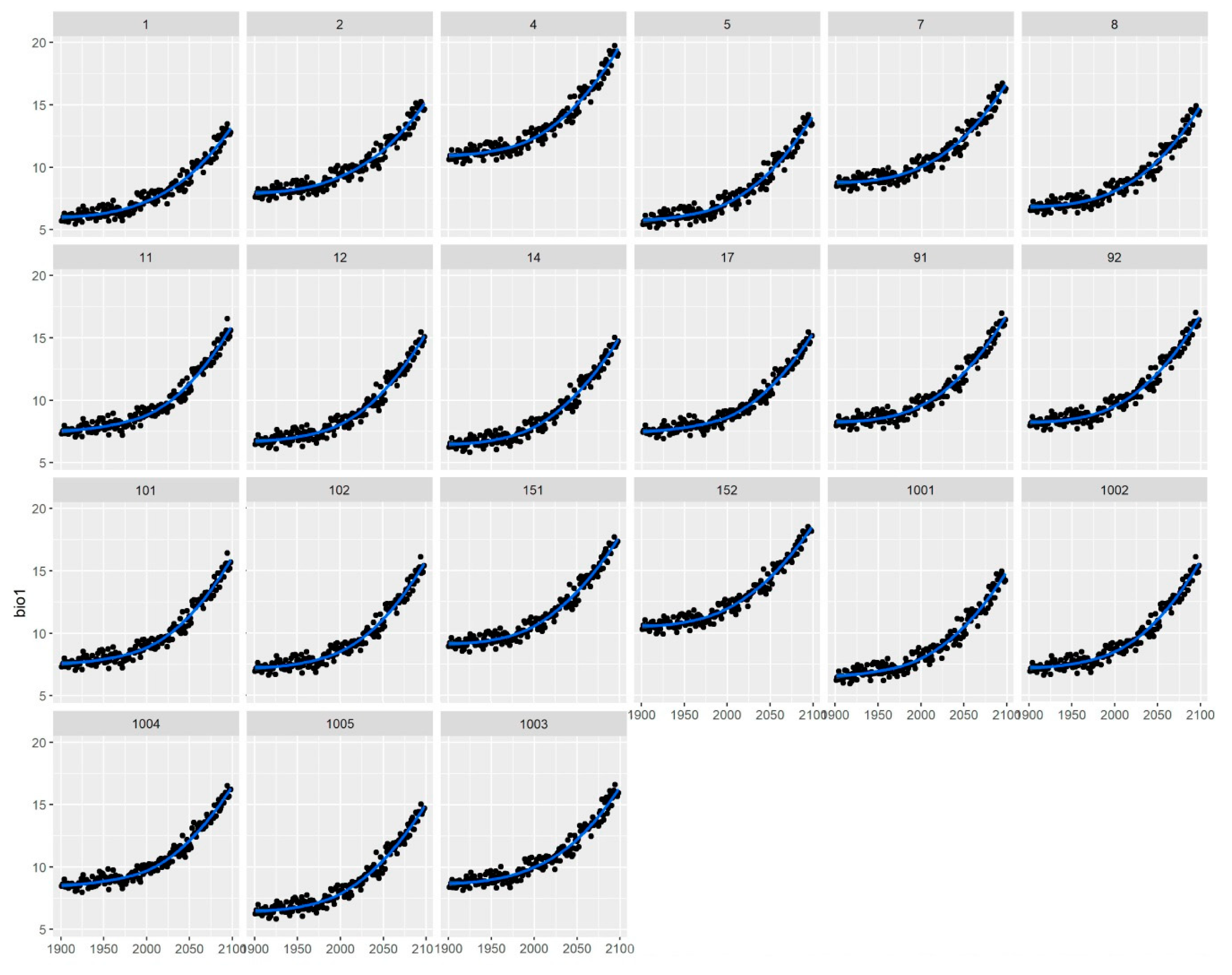
Table 1). However, these changes are still small compared to those predicted at the 2100 period, and therefore, we can expect an underestimation of the results with these two methods (Climate-predictive and Climate-adjusted).
Composite, Admixture and Climate-adjusted seed sourcing have been suggested in the context of rapid climate change [
27] but this has not been tested against other methods [
28]. This is, to our knowledge, the first attempt to test different seed sourcing strategies using experimental data in a forest tree species. We would expect a higher variance in the traits, as a proxy of the standing genetic variation. Our results suggest that the combination of different seed sources does not provide a clear advantage to the methods based on one sole population. Another aspect not considered in our study is the possibility of a combination of alleles from different populations to produce new combinations for natural selection to occur. However, it is possible that some outbreeding depression might occur as a result of these new combinations [
47,
48]. Therefore we need to take these results with caution, as the context (early estimation under the climate change scenario, non-significant variation among provenances in survival) can hinder some of the advantages of these methods as proposed by different authors [
7,
8,
9,
10,
11]. These methods can also have some risks in the conservation of the genetic resources of the species in the long term [
49], and therefore we will need to show clear advantages over traditional methods to implement them at an operational scale in conditions like the ones tested in our study.