Can Strict Protection Stop the Decline of Mangrove Ecosystems in China? From Rapid Destruction to Rampant Degradation
Abstract
1. Introduction
2. Data and Methods
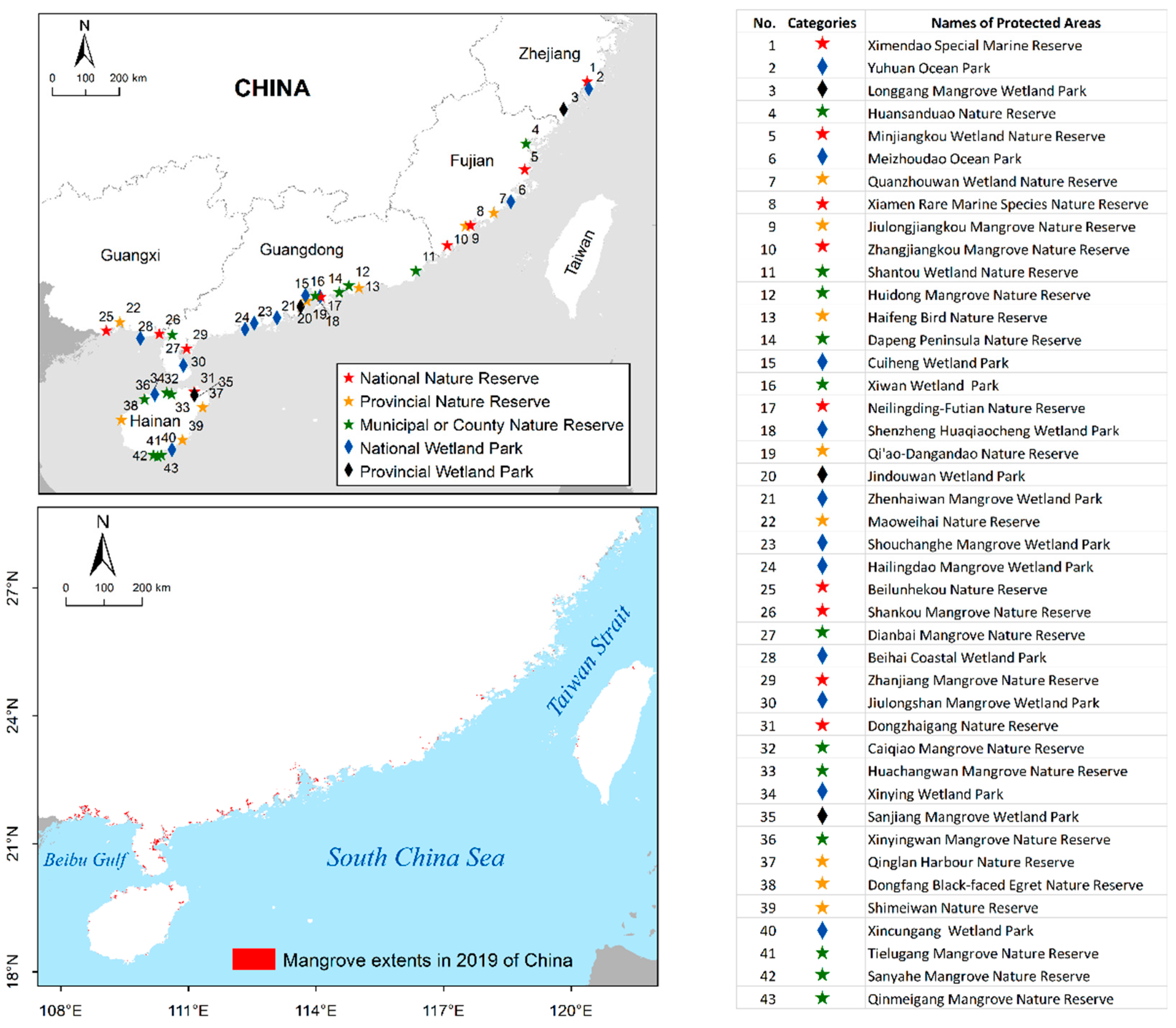
2.1. Mangrove Forests in China
2.2. Species Diversity and Population Dynamics of Mangrove Communities
2.3. Community Structure
2.4. Changes in Carbon Stock and Carbon Sequestration Rate
2.5. Landward Margins of Mangrove Forests
3. Results
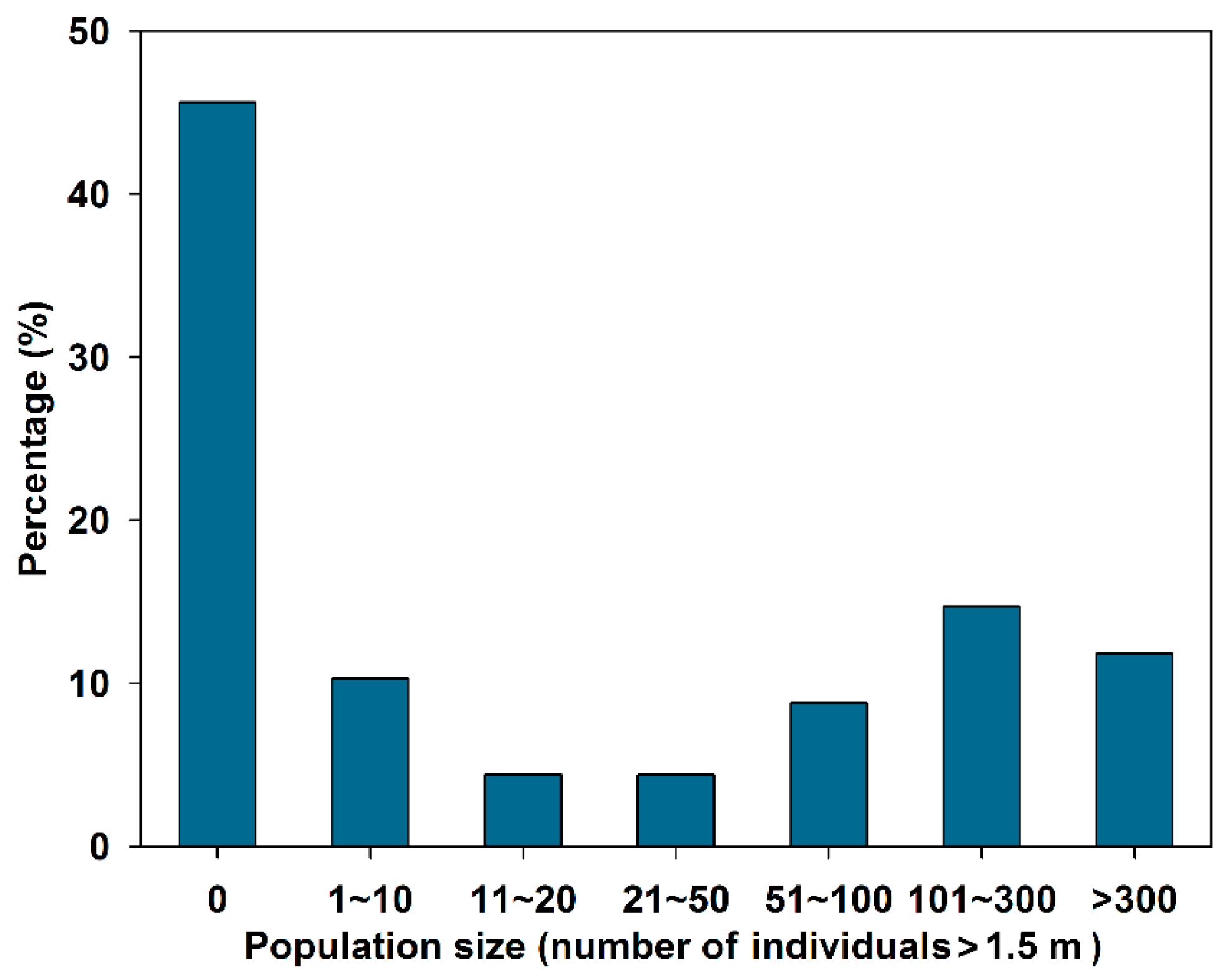
3.1. Mangrove Species Diversity and Population Dynamics
3.2. Simplification of Community Structure
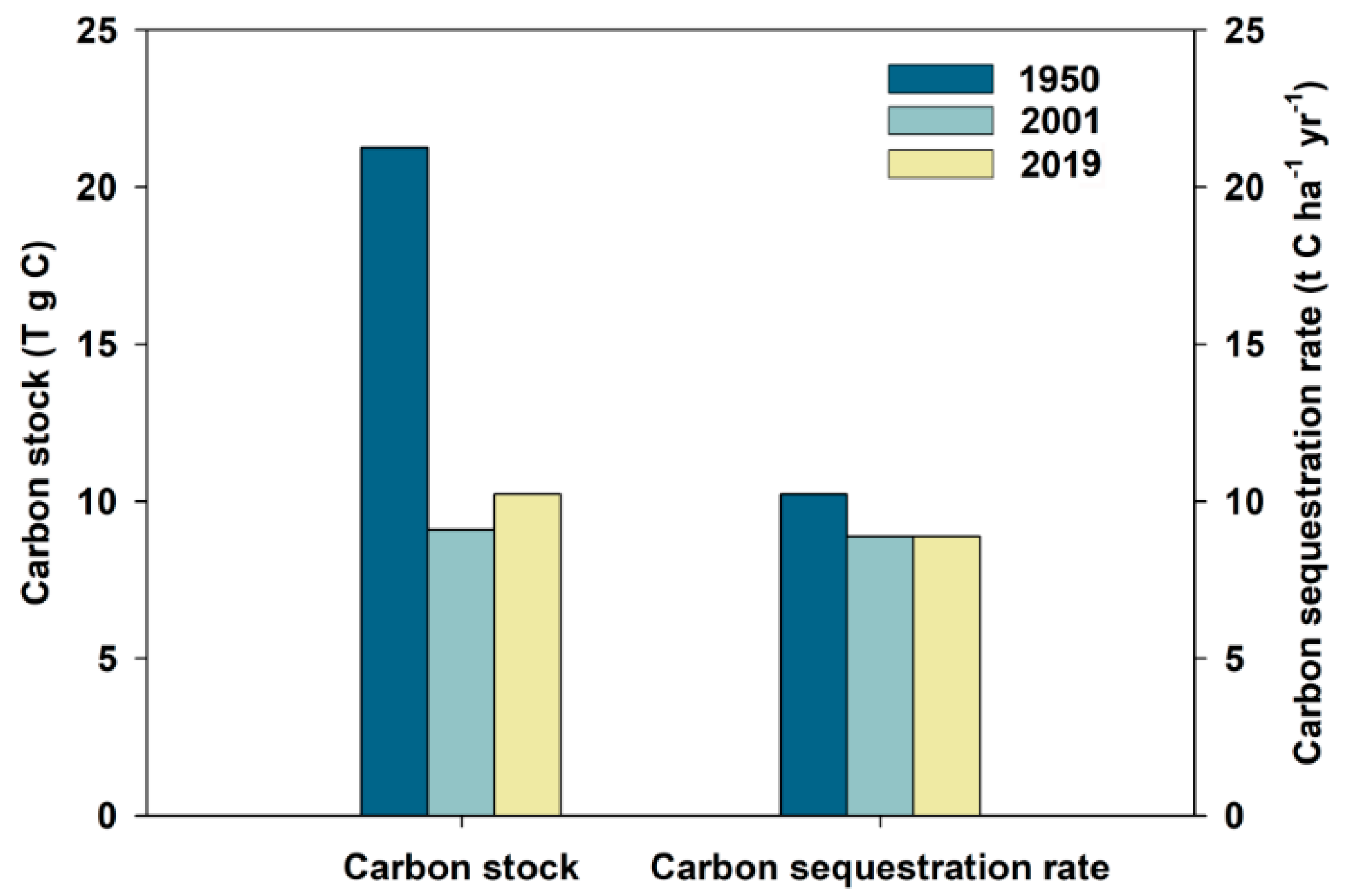
3.3. Reduced Carbon Stock and Carbon Sequestration Rate
3.4. Breakdown of Landward Forest Margins
4. Discussion
4.1. Rampant Degradation Despite Strict Protection and Increase in Mangrove Forest Area
4.2. Root Cause of Mangrove Degradation—Seawall Construction
4.3. The Effectiveness of Mangrove Conservation Actions
4.4. Conservation Priorities and Perspectives
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nordhaus, I.; Roelke, D.L.; Vaquer-Sunyer, R.; Winter, C. Coastal systems in transition: From a ‘natural’ to an ‘anthropogenically-modified’ state. Estuar. Coast. Shelf Sci. 2018, 211, 1–5. [Google Scholar] [CrossRef]
- Harfoot, M.B.J.; Tittensor, D.P.; Knight, S.; Arnell, A.P.; Blyth, S.; Brooks, S.; Butchart, S.H.M.; Hutton, J.; Jones, M.I.; Kapos, V.; et al. Present and future biodiversity risks from fossil fuel exploitation. Conserv. Lett. 2018, 11, e12448. [Google Scholar] [CrossRef]
- Carugati, L.; Gatto, B.; Rastelli, E.; Martire, M.L.; Coral, C.; Greco, S.; Danovaro, R. Impact of mangrove forests degradation on biodiversity and ecosystem functioning. Sci. Rep. 2018, 8, 13298. [Google Scholar] [CrossRef] [PubMed]
- Pimm, S.L.; Jenkins, C.N.; Abell, R.; Brooks, T.M.; Gittleman, J.L.; Joppa, L.N.; Raven, P.H.; Roberts, C.M.; Sexton, J.O. The biodiversity of species and their rates of extinction, distribution, and protection. Science 2014, 344, 1246752. [Google Scholar] [CrossRef] [PubMed]
- Aronson, J.; Alexander, S. Ecosystem restoration is now a global priority: Time to roll up our sleeves. Restor. Ecol. 2013, 21, 293–296. [Google Scholar] [CrossRef]
- Suding, K.N. Toward an era of restoration in ecology: Successes, failures, and opportunities ahead. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 465–487. [Google Scholar] [CrossRef]
- Coetzee, B.W.T. Evaluating the ecological performance of protected areas. Biodivers. Conserv. 2017, 26, 231–236. [Google Scholar] [CrossRef]
- Locke, H. Nature needs half: A necessary and hopeful new agenda for protected areas in North America and around the world. George Wright Forum 2014, 31, 359–371. [Google Scholar]
- Suman, D.O. Mangrove Management: Challenges and Guidelines. In Coastal Wetlands: An Integrated Ecosystem Approach, 2nd ed.; Perillo, G.M.E., Wolanski, E., Cahoon, D.R., Hopkinson, C.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1055–1079. [Google Scholar]
- Sidik, F.; Supriyanto, B.; Krisnawati, H.; Muttaqin, M.Z. Mangrove conservation for climate change mitigation in Indonesia. WIREs Clim. Chang. 2018, 9, e529. [Google Scholar] [CrossRef]
- Roberts, C.M.; McClean, C.J.; Veron, J.E.N.; Hawkins, J.P.; Allen, G.R.; McAllister, D.E.; Mittermeier, C.G.; Schueler, F.W.; Spalding, M.; Wells, F.; et al. Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 2002, 295, 1280–1284. [Google Scholar] [CrossRef]
- Melillo, J.M.; Lu, X.; Kicklighter, D.W.; Reilly, J.M.; Cai, Y.; Sokolov, A.P. Protected areas’ role in climate-change mitigation. Ambio 2016, 45, 133–145. [Google Scholar] [CrossRef] [PubMed]
- United Nations. Sustainable Development Goal 14—Targets, Indicators. Available online: https://sustainabledevelopment.un.org/sdg14 (accessed on 1 March 2018).
- Coates, D. Strategic Plan for Biodiversity (2011–2020) and the Aichi Biodiversity Targets. In The Wetland Book I: Structure and Function, Management, and Methods; Finlayson, C.M., Everard, M., Irvine, K., McInnes, R.J., Middleton, B.A., van Dam, A.A., Davidson, N.C., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 493–499. [Google Scholar]
- Wilson, E.O. Half-Earth: Our Planet’s Fight for Life; Liveright: New York, NY, USA, 2016. [Google Scholar]
- Lamb, D.; Erskine, P.D.; Parrotta, J.A. Restoration of degraded tropical forest landscapes. Science 2005, 310, 1628–1632. [Google Scholar] [CrossRef] [PubMed]
- Cramer, V.A.; Hobbs, R.J.; Standish, R.J. What’s new about old fields? Land abandonment and ecosystem assembly. Trends Ecol. Evol. 2008, 23, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Field, C.D. Rehabilitation of mangrove ecosystems: An overview. Mar. Pollut. Bull. 1998, 37, 383–392. [Google Scholar] [CrossRef]
- Lu, F.; Hu, H.; Sun, W.; Zhu, J.; Liu, G.; Zhou, W.; Zhang, Q.; Shi, P.; Liu, X.; Wu, X.; et al. Effects of national ecological restoration projects on carbon sequestration in China from 2001 to 2010. Proc. Natl. Acad. Sci. USA 2018, 115, 4039–4044. [Google Scholar]
- Lee, S.Y.; Primavera, J.H.; Dahdouh-Guebas, F.; McKee, K.; Bosire, J.O.; Cannicci, S.; Diele, K.; Fromard, F.; Koedam, N.; Marchand, C.; et al. Ecological role and services of tropical mangrove ecosystems: A reassessment. Glob. Ecol. Biogeogr. 2014, 23, 726–743. [Google Scholar] [CrossRef]
- Costanza, R.; de Groot, R.; Sutton, P.; van der Ploeg, S.; Anderson, S.J.; Kubiszewski, I.; Farber, S.; Turner, R.K. Changes in the global value of ecosystem services. Glob. Environ. Chang. 2014, 26, 152–158. [Google Scholar] [CrossRef]
- FAO. The World’s Mangroves 1980–2005 (FAO Forestry Paper 153); Food and Agriculture Organization of the United Nations: Rome, Italy, 2007. [Google Scholar]
- Hamilton, S.E.; Casey, D. Creation of a high spatio-temporal resolution global database of continuous mangrove forest cover for the 21st century (CGMFC-21). Glob. Ecol. Biogeogr. 2016, 25, 729–738. [Google Scholar] [CrossRef]
- Richards, D.R.; Friess, D.A. Rates and drivers of mangrove deforestation in Southeast Asia, 2000–2012. Proc. Natl. Acad. Sci. USA 2016, 113, 344–349. [Google Scholar] [CrossRef]
- Fan, H.; Wang, W. Some thematic issues for mangrove conservation in China. J. Xiamen Univ. (Nat. Sci.) 2017, 56, 323–330. [Google Scholar]
- SFA. National Mangrove Resource Inventory Report; The State Forestry Administration of the People’s Republic of China: Beijing, China, 2002.
- Wang, W.; Wang, M. The Mangroves of China; Science Press: Beijing, China, 2007. [Google Scholar]
- Wang, M.; Wang, W.; Lin, G.; Ma, W.; Fu, R. The Mangroves of Sanya; Science Press: Beijing, China, 2019. [Google Scholar]
- Li, M.S.; Lee, S.Y. Mangroves of China: A brief review. For. Ecol. Manag. 1997, 96, 241–259. [Google Scholar] [CrossRef]
- Giri, C.; Ochieng, E.; Tieszen, L.L.; Zhu, Z.; Singh, A.; Loveland, T.; Masek, J.; Duke, N. Status and distribution of mangrove forests of the world using earth observation satellite data. Glob. Ecol. Biogeogr. 2011, 20, 154–159. [Google Scholar] [CrossRef]
- Giesen, W.; Wulffraat, S.; Zieren, M.; Scholten, L. Mangrove Guidebook for Southeast Asia; Food and Agricultural Organisation and Wetlands International: Bangkok, Thailand, 2007. [Google Scholar]
- Jayanthi, M.; Thirumurthy, S.; Nagaraj, G.; Muralidhar, M.; Ravichandran, P. Spatial and temporal changes in mangrove cover across the protected and unprotected forests of India. Estuar. Coast. Shelf Sci. 2018, 213, 81–91. [Google Scholar] [CrossRef]
- Watson, J.E.M.; Iwamura, T.; Butt, N. Mapping vulnerability and conservation adaptation strategies under climate change. Nat. Clim. Chang. 2013, 3, 989–994. [Google Scholar] [CrossRef]
- Lee, C.K.F.; Duncan, C.; Owen, H.J.F.; Pettorelli, N. A new framework to assess relative ecosystem vulnerability to climate change. Conserv. Lett. 2018, 11, e12372. [Google Scholar] [CrossRef]
- Pacifici, M.; Foden, W.B.; Visconti, P.; Watson, J.E.M.; Butchart, S.H.M.; Kovacs, K.M.; Scheffers, B.R.; Hole, D.G.; Martin, T.G.; Akçakaya, H.R.; et al. Assessing species vulnerability to climate change. Nat. Clim. Chang. 2015, 5, 215–224. [Google Scholar] [CrossRef]
- Seddon, A.W.R.; Macias-Fauria, M.; Long, P.R.; Benz, D.; Willis, K.J. Sensitivity of global terrestrial ecosystems to climate variability. Nature 2016, 531, 229–232. [Google Scholar] [CrossRef]
- Bakhtiyari, M.; Lee, S.Y.; Warnken, J. Seeing the forest as well as the trees: An expert opinion approach to identifying holistic condition indicators for mangrove ecosystems. Estuar. Coast. Shelf Sci. 2019, 222, 183–194. [Google Scholar] [CrossRef]
- Erftemeijer, P.L.A. A new technique for rapid assessment of mangrove degradation: A case study of shrimp farm encroachment in Thailand. Trees 2002, 16, 204–208. [Google Scholar] [CrossRef]
- Ilman, M.; Dargusch, P.; Dart, P. A historical analysis of the drivers of loss and degradation of Indonesia’s mangroves. Land Use Policy 2016, 54, 448–459. [Google Scholar] [CrossRef]
- Lee, S.Y.; Hamilton, S.; Barbier, E.B.; Primavera, J.; Lewis III, R.R. Better restoration policies are needed to conserve mangrove ecosystems. Nat. Ecol. Evol. 2019, 3, 870–872. [Google Scholar] [CrossRef] [PubMed]
- Gilman, E.L.; Ellison, J.; Duke, N.C.; Field, C. Threats to mangroves from climate change and adaptation options: A review. Aquat. Bot. 2008, 89, 237–250. [Google Scholar] [CrossRef]
- Alongi, D.M. Mangrove Forests of Timor-Leste: Ecology, Degradation and Vulnerability to Climate Change. In Mangrove Ecosystems of Asia: Status, Challenges and Management Strategies; Faridah-Hanum, I., Latiff, A., Hakeem, K.R., Ozturk, M., Eds.; Springer: New York, NY, USA, 2014; pp. 199–212. [Google Scholar]
- Ma, Z.; Melville, D.S.; Liu, J.; Chen, Y.; Yang, H.; Ren, W.; Zhang, Z.; Piersma, T.; Li, B. Rethinking China’s new great wall. Science 2014, 346, 912–914. [Google Scholar] [CrossRef] [PubMed]
- Enwright, N.M.; Griffith, K.T.; Osland, M.J. Barriers to and opportunities for landward migration of coastal wetlands with sea-level rise. Front. Ecol. Environ. 2016, 14, 307–316. [Google Scholar] [CrossRef]
- Spencer, T.; Schuerch, M.; Nicholls, R.J.; Hinkel, J.; Lincke, D.; Vafeidis, A.T.; Reef, R.; McFadden, L.; Brown, S. Global coastal wetland change under sea-level rise and related stresses: The DIVA Wetland Change Model. Glob. Planet. Chang. 2016, 139, 15–30. [Google Scholar] [CrossRef]
- Schuerch, M.; Spencer, T.; Temmerman, S.; Kirwan, M.L.; Wolff, C.; Lincke, D.; McOwen, C.J.; Pickering, M.D.; Reef, R.; Vafeidis, A.T.; et al. Future response of global coastal wetlands to sea-level rise. Nature 2018, 561, 231–234. [Google Scholar] [CrossRef]
- Kathiresan, K. Why are mangroves degrading? Curr. Sci. 2002, 83, 1246–1249. [Google Scholar]
- Duke, N.C.; Meynecke, J.O.; Dittmann, S.; Ellison, A.M.; Anger, K.; Berger, U.; Cannicci, S.; Diele, K.; Ewel, K.C.; Field, C.D.; et al. A world without mangroves? Science 2007, 317, 41–42. [Google Scholar] [CrossRef]
- El-Juhany, L.I. Present status and degradation trends of mangrove forests on the southern Red Sea coast of Saudi Arabia. Am.-Eur. J. Agric. Environ. Sci. 2009, 6, 328–340. [Google Scholar]
- Tomlinson, P.B. The Botany of Mangroves, 2nd ed.; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- IUCN. Guidelines for Application of IUCN Red List Criteria at Regional and National Levels, Version 4.0; International Union for Conservation of Nature: Gland, Switzerland; Cambridge, UK, 2012. [Google Scholar]
- Hou, K.; He, C. Mangroves of China. Bull. Biol. 1953, 10, 365–369. [Google Scholar]
- Wang, L.; Li, Z.; Pu, Y.; Liao, W.; Zhang, Q.; Yu, K.; Zhao, H. Health evaluation of mangrove community in Dongzhaigang, Sanyahe, and Qingmeigang Mangrove Nature Reserves of Hainan province. J. Trop. Oceanogr. 2011, 30, 81–86. [Google Scholar]
- Wang, G.; Guan, D.; Peart, M.R.; Chen, Y.; Peng, Y. Ecosystem carbon stocks of mangrove forest in Yingluo Bay, Guangdong Province of South China. For. Ecol. Manag. 2013, 310, 539–546. [Google Scholar] [CrossRef]
- Liu, H.; Ren, H.; Hui, D.; Wang, W.; Liao, B.; Cao, Q. Carbon stocks and potential carbon storage in the mangrove forests of China. J. Environ. Manag. 2014, 133, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Duke, N.C.; Ball, M.C.; Ellison, J.C. Factors influencing biodiversity and distributional gradients in mangroves. Glob. Ecol. Biogeogr. Lett. 1998, 7, 27–47. [Google Scholar] [CrossRef]
- Polidoro, B.A.; Carpenter, K.E.; Collins, L.; Duke, N.C.; Ellison, A.M.; Ellison, J.C.; Farnsworth, E.J.; Fernando, E.S.; Kathiresan, K.; Koedam, N.E.; et al. The loss of species: Mangrove extinction risk and geographic areas of global concern. PLoS ONE 2010, 5, e10095. [Google Scholar] [CrossRef]
- IUCN. The IUCN Red List of Threatened Species. Version 2017-3. Available online: http://www.iucnredlist.org. (accessed on 1 May 2017).
- Daru, B.H.; Yessoufou, K.; Mankga, L.T.; Davies, T.J. A global trend towards the loss of evolutionarily unique species in mangrove ecosystems. PLoS ONE 2013, 8, e66686. [Google Scholar] [CrossRef]
- Sarker, S.K.; Reeve, R.; Paul, N.K.; Matthiopoulos, J. Modelling spatial biodiversity in the world’s largest mangrove ecosystem–The Bangladesh Sundarbans: A baseline for conservation. Divers. Distrib. 2019, 25, 729–742. [Google Scholar] [CrossRef]
- Sarker, S.K.; Matthiopoulos, J.; Mitchella, S.N.; Ahmed, Z.U.; Mamun, M.B.A.; Reeve, R. 1980s–2010s: The world’s largest mangrove ecosystem is becoming homogeneous. Biol. Conserv. 2019, 236, 79–91. [Google Scholar] [CrossRef]
- Qin, H.; Zhao, L. Evaluating the threat status of higher plants in China. Biodivers. Sci. 2017, 25, 689–695. [Google Scholar] [CrossRef]
- Benson, L.; Glass, L.; Jones, T.G.; Ravaoarinorotsihoarana, L.; Rakotomahazo, C. Mangrove carbon stocks and ecosystem cover dynamics in Southwest Madagascar and the implications for local management. Forests 2017, 8, 190. [Google Scholar] [CrossRef]
- Bosire, J.O.; Kaino, J.J.; Olagoke, A.O.; Mwihaki, L.M.; Ogendi, G.M.; Kairo, J.G.; Berger, U.; Macharia, D. Mangroves in peril: Unprecedented degradation rates of peri-urban mangroves in Kenya. Biogeosciences 2014, 11, 2623–2634. [Google Scholar] [CrossRef]
- Peltzer, D.A.; Wardle, D.A.; Allison, V.J.; Baisden, W.T.; Bardgett, R.D.; Chadwick, O.A.; Condron, L.M.; Parfitt, R.L.; Porder, S.; Richardson, S.J.; et al. Understanding ecosystem retrogression. Ecol. Monogr. 2010, 80, 509–529. [Google Scholar] [CrossRef]
- Sharma, S.; MacKenzie, R.A.; Tieng, T.; Soben, K.; Tulyasuwan, N.; Resanond, A.; Blate, G.; Litton, C.M. The impacts of degradation, deforestation and restoration on mangrove ecosystem carbon stocks across Cambodia. Sci. Total Environ. 2020, 706, 135416. [Google Scholar] [CrossRef] [PubMed]
- Vaiphasa, C.; de Boer, W.F.; Skidmore, A.K.; Panitchart, S.; Vaiphasa, T.; Bamrongrugsa, N.; Santitamnont, P. Impact of solid shrimp pond waste materials on mangrove growth and mortality: A case study from Pak Phanang, Thailand. Hydrobiologia 2007, 591, 47–57. [Google Scholar] [CrossRef]
- Pontee, N. Defining coastal squeeze: A discussion. Ocean Coast. Manag. 2013, 84, 204–207. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Cahoon, D.R.; Friess, D.A.; Guntenspergen, G.R.; Krauss, K.W.; Reef, R.; Rogers, K.; Saunders, M.L.; Sidik, F.; Swales, A.; et al. The vulnerability of Indo-Pacific mangrove forests to sea-level rise. Nature 2015, 526, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Heatherington, C.; Bishop, M.J. Spatial variation in the structure of mangrove forests with respect to seawalls. Mar. Freshw. Res. 2012, 63, 926–933. [Google Scholar] [CrossRef]
- Bartolini, F.; Cimò, F.; Fusi, M.; Dahdouh-Guebas, F.; Lopes, G.P.; Cannicci, S. The effect of sewage discharge on the ecosystem engineering activities of two East African fiddler crab species: Consequences for mangrove ecosystem functioning. Mar. Environ. Res. 2011, 71, 53–61. [Google Scholar] [CrossRef]
- Lee, S.Y. Relationship between mangrove abundance and tropical prawn production: A re-evaluation. Mar. Biol. 2004, 145, 943–949. [Google Scholar] [CrossRef]
- Nagelkerken, I. Ecological Connectivity among Tropical Coastal Ecosystems; Springer: New York, NY, USA, 2009. [Google Scholar]
- Chen, L.; Wang, W.; Zhang, Y.; Lin, G. Recent progresses in mangrove conservation, restoration and research in China. J. Plant Ecol. 2009, 2, 45–54. [Google Scholar] [CrossRef]
- Zou, F.; Yang, Q.; Dahmer, T.; Cai, J.; Zhang, W. Habitat use of waterbirds in coastal wetland on Leizhou Peninsula, China. Waterbirds 2006, 29, 459–464. [Google Scholar] [CrossRef]
- Ren, H.; Lu, H.; Shen, W.; Huang, C.; Guo, Q.; Li, Z.; Jian, S. Sonneratia apetala Buch.Ham in the mangrove ecosystems of China: An invasive species or restoration species? Ecol. Eng. 2009, 35, 1243–1248. [Google Scholar] [CrossRef]
- Peng, D.; Chen, L.; Pennings, S.C.; Zhang, Y. Using a marsh organ to predict future plant communities in a Chinese estuary invaded by an exotic grass and mangrove. Limnol. Oceanogr. 2018, 63, 2595–2605. [Google Scholar] [CrossRef]
- Fu, Y.; Chen, Y.; Zhang, Z. Temporal and spatial variation of population density of coastal cities during 1985–2010 in China. Trop. Geogr. 2014, 34, 635–642. [Google Scholar]
- Hazelden, J.; Boorman, L.A. Soils and ‘managed retreat’ in south east England. Soil Use Manag. 2001, 17, 150–154. [Google Scholar] [CrossRef]
- Sajjad, M.; Li, Y.; Tang, Z.; Cao, L.; Liu, X. Assessing hazard vulnerability, habitat conservation, and restoration for the enhancement of Mainland China’s coastal resilience. Earth’s Future 2018, 6, 326–338. [Google Scholar] [CrossRef]

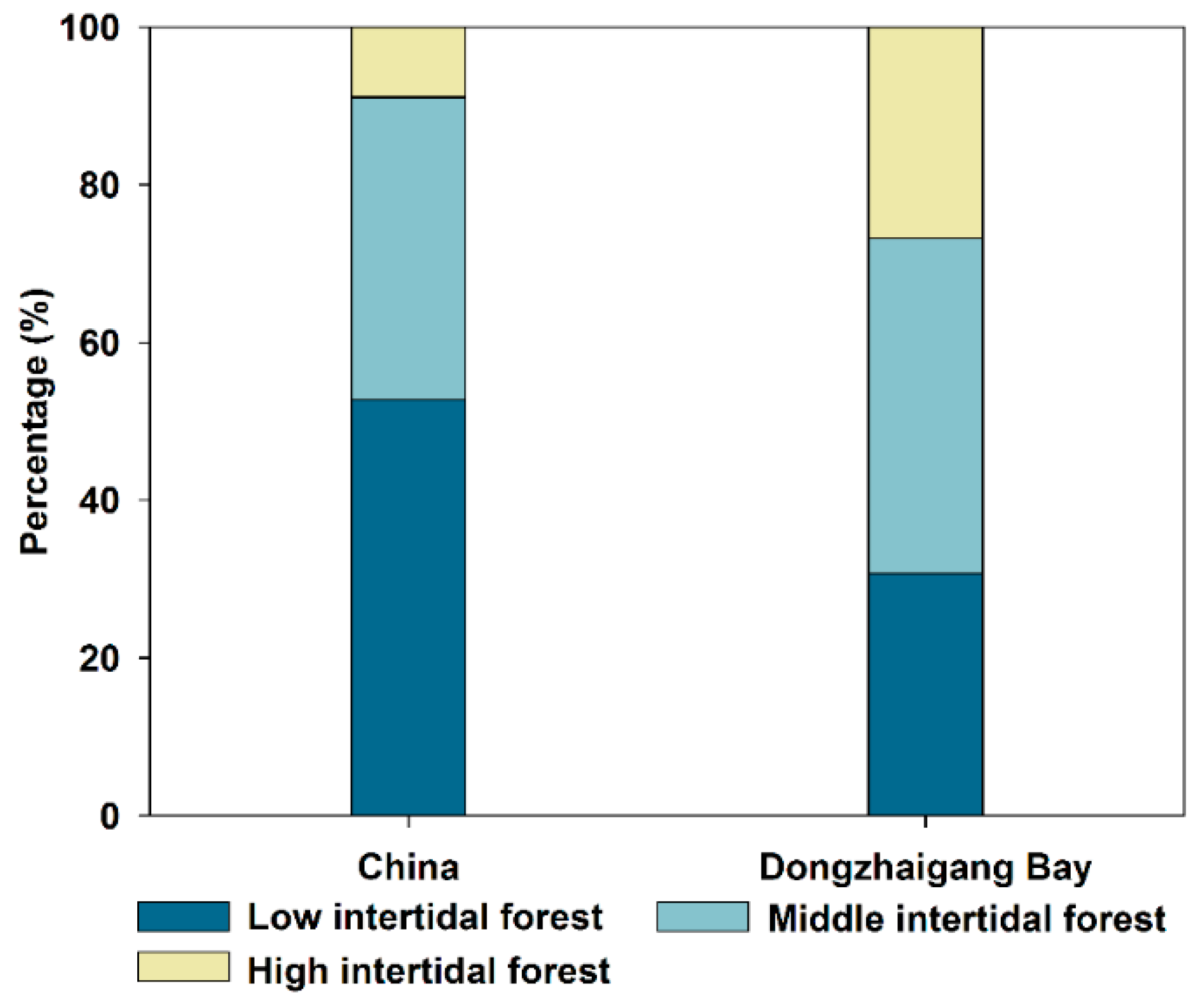

| Low Intertidal Forest | Middle Intertidal Forest | High Intertidal Forest | Total | |
|---|---|---|---|---|
| Area in 1950 (ha) | 14,973 | 20,286 | 13,041 | 48,300 |
| Area in 2001 (ha) | 11,673 | 8370 | 1982 | 22,025 |
| Area in 2019 (ha) | 13,105 | 9396 | 2226 | 24,727 * |
| NPP in 1950 (Mg·C·yr−1) | 118,889 | 183,678 | 178,596 | 481,169 |
| NPP in 2001 (Mg·C·yr−1) | 92,686 | 75,745 | 27,146 | 195,577 |
| NPP in 2019 (Mg·C·yr−1) | 104,056 | 85,078 | 30,477 | 219,611 |
| Family | Species | Intertidal Position [56] | Red List Category in China | Global Red List Category [57] | ||
|---|---|---|---|---|---|---|
| Acanthaceae | Acanthus ebracteatus | M | H | EN | LC | |
| A. ilicifolius | M | H | LC | LC | ||
| Palmae | Nypa fruticans | L | M | H | VU | LC |
| Avicenniaceae | Avicennia marina | L | LC | LC | ||
| Combretaceae | Lumnitzera littorea | M | CR | LC | ||
| L. racemosa | M | H | LC | LC | ||
| Acrostichaceae | Acrostichum aureum | H | LC | LC | ||
| A. speciosum | H | EN | LC | |||
| Euphorbiaceae | Excoecaria agallocha | M | H | LC | LC | |
| Meliaceae | Xylocarpus granatum | M | H | VU | LC | |
| Myrsinaceae | Aegiceras corniculatum | L | LC | LC | ||
| Rhizophoraceae | Bruguiera gymnorhiza | M | H | LC | LC | |
| B. sexangula | M | H | NT | LC | ||
| B. s. var. rhynchopetala | M | H | VU | NE | ||
| Ceriops tagal | M | H | LC | LC | ||
| Kandelia obovata | L | M | LC | LC | ||
| Rhizophora apiculata | M | VU | LC | |||
| R. stylosa | L | M | LC | LC | ||
| R. × lamarckii | M | H | CR | NE | ||
| Rubiaceae | Scyphiphora hydrophyllacea | H | EN | LC | ||
| Sonneratiaceae | Sonneratia alba | L | LC | LC | ||
| S. caseolaris | L | NT | LC | |||
| S. × gulngai | H | EN | NE | |||
| S. × hainanensis | H | CR | NE | |||
| S. ovata | H | CR | NT | |||
| Lythraceae | Pemphis acidula | H | EN | LC | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Fu, H.; Lee, S.Y.; Fan, H.; Wang, M. Can Strict Protection Stop the Decline of Mangrove Ecosystems in China? From Rapid Destruction to Rampant Degradation. Forests 2020, 11, 55. https://doi.org/10.3390/f11010055
Wang W, Fu H, Lee SY, Fan H, Wang M. Can Strict Protection Stop the Decline of Mangrove Ecosystems in China? From Rapid Destruction to Rampant Degradation. Forests. 2020; 11(1):55. https://doi.org/10.3390/f11010055
Chicago/Turabian StyleWang, Wenqing, Haifeng Fu, Shing Yip Lee, Hangqing Fan, and Mao Wang. 2020. "Can Strict Protection Stop the Decline of Mangrove Ecosystems in China? From Rapid Destruction to Rampant Degradation" Forests 11, no. 1: 55. https://doi.org/10.3390/f11010055
APA StyleWang, W., Fu, H., Lee, S. Y., Fan, H., & Wang, M. (2020). Can Strict Protection Stop the Decline of Mangrove Ecosystems in China? From Rapid Destruction to Rampant Degradation. Forests, 11(1), 55. https://doi.org/10.3390/f11010055




