Effects of Phosphate Solubilizing Bacteria on the Growth, Photosynthesis, and Nutrient Uptake of Camellia oleifera Abel.
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains, Plant Material, and Growth Medium
2.2. Experimental Design and Growth Condition
2.3. Plant Growth Measurement
2.4. Measurement of Gas Exchange Parameters and Relative Chlorophyll Content
2.5. Measurement of Plant Nutrient and Soil Nutrient
2.6. Data Analysis
3. Results
3.1. Plant Height and Biomass
3.2. Gas Exchange Parameters
3.3. Chlorophyll Content
3.4. Nitrogen and Phosphorus Content in Leaves
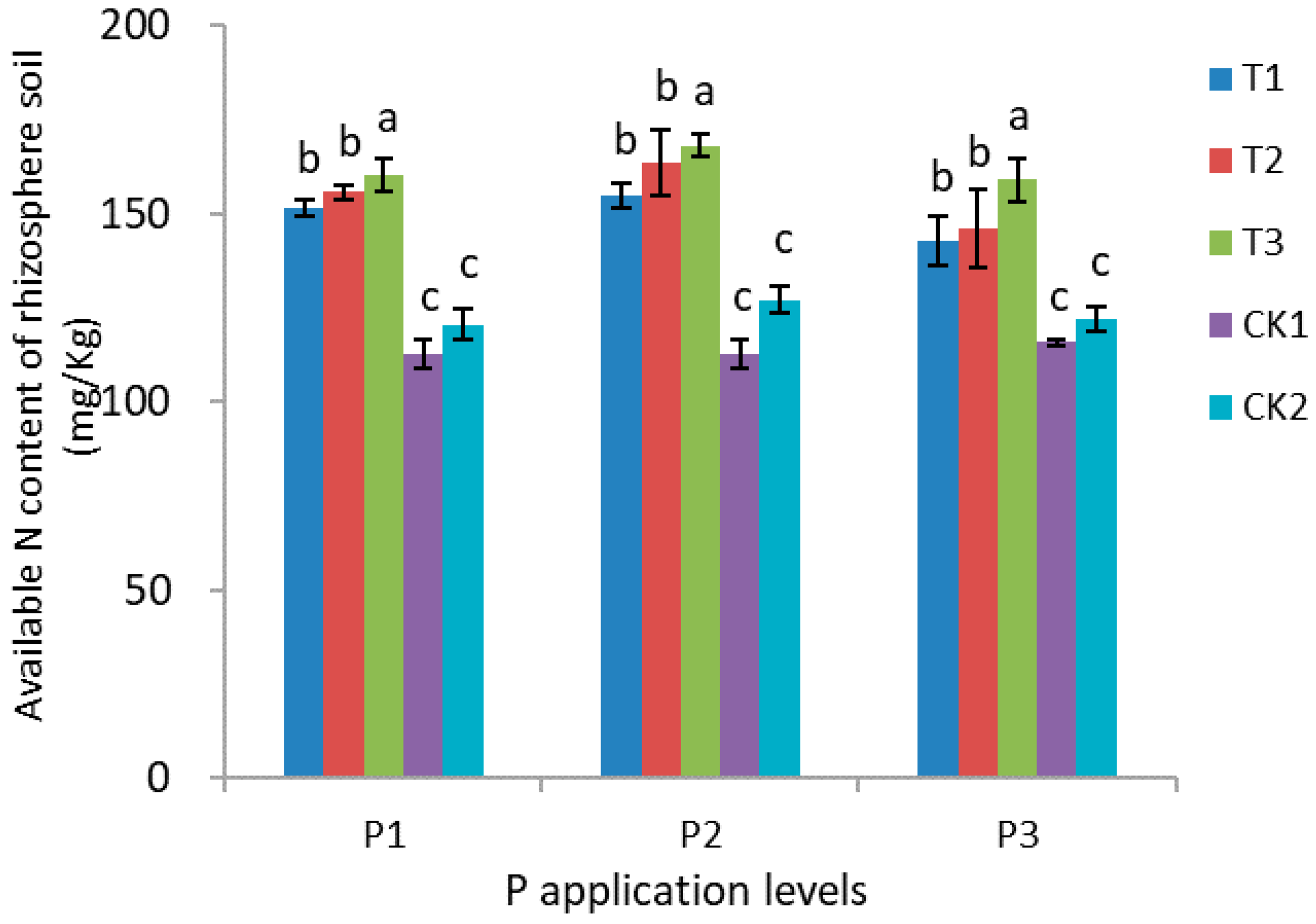
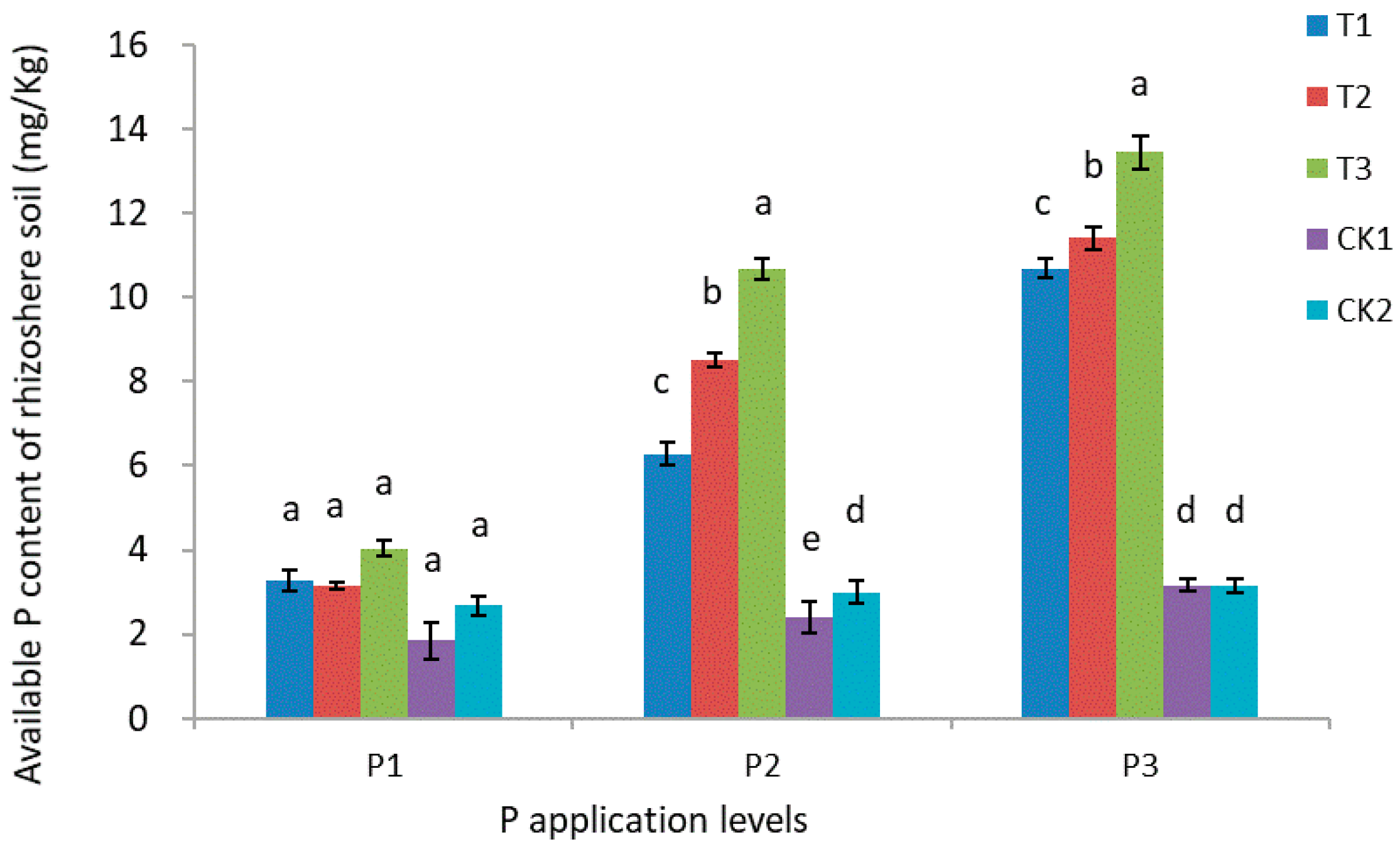
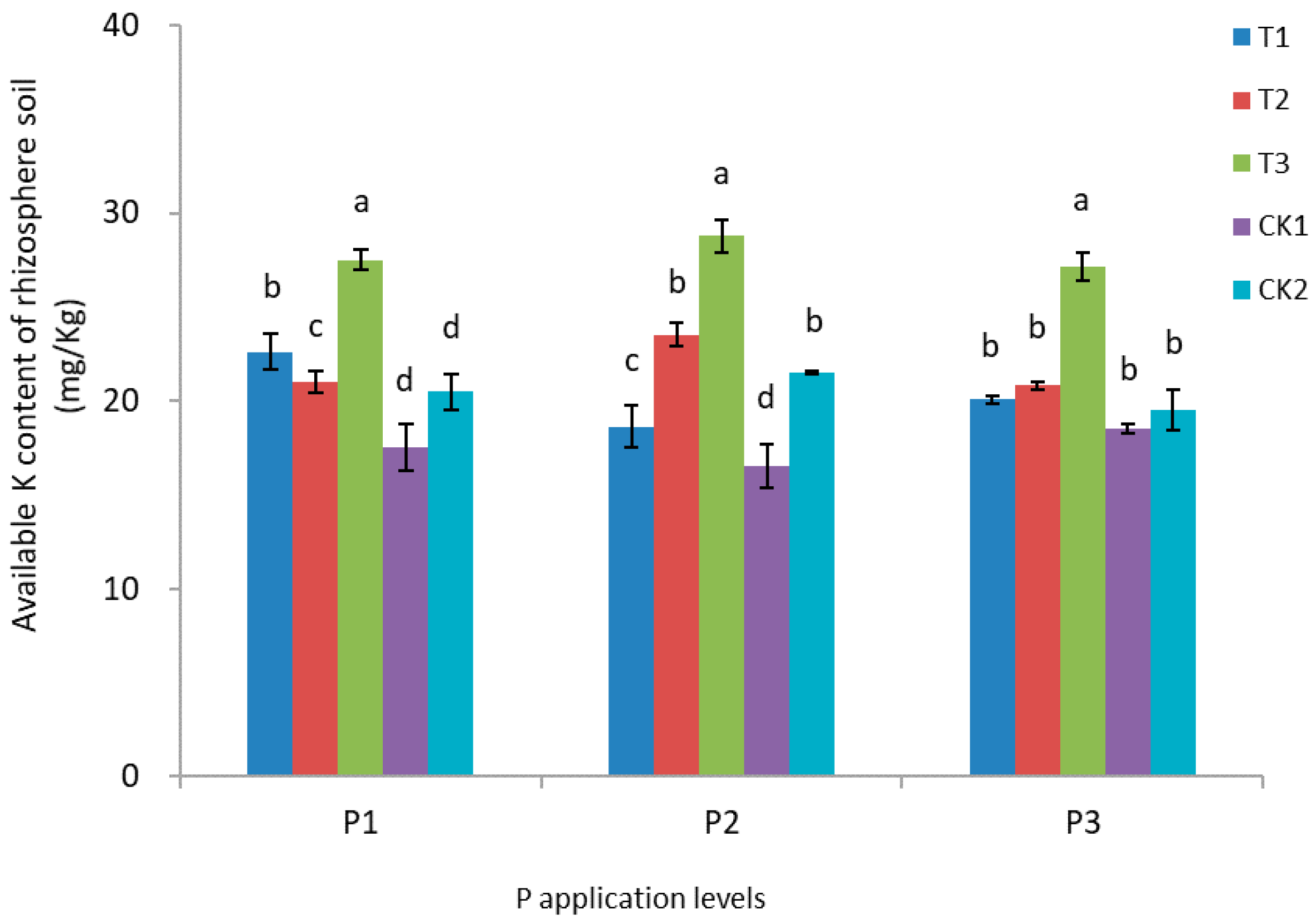
3.5. Nitrogen, Phosphorus, and Potassium Content of Rhizosphere Soil
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oteino, N.; Lally, R.D.; Kiwanuka, S.; Lloyd, A.; Ryan, D.; Germaine, K.J.; Dowling, D.N. Plant growth promotion induced by phosphate solubilizing endophytic Pseudomonas isolates. Front. Microbiol. 2015, 6, 745. [Google Scholar] [CrossRef]
- Ågren, G.I. Stoichiometry and nutrition of plant growth in natural communities. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 153–170. [Google Scholar] [CrossRef]
- Cordell, D.; Drangert, J.O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- Skinner, M.F.; Attiwill, P.M. The productivity of pine plantations in relation to previous land use. Plant Soil 1981, 61, 329–339. [Google Scholar] [CrossRef]
- Abbas, M.; Shah, J.A.; Irfan, M.; Memon, M.Y. Remobilization phosphorus in wheat cultivars under induced phosphorus deficiency. J. Plant Nutr. 2018, 41, 1–12. [Google Scholar] [CrossRef]
- Fujita, Y.; Venterink, H.O.; Van Bodegom, P.M.; Douma, J.C.; Heil, G.W.; Hölzel, N.; Jabłońska, E.; Kotowski, W.; Okruszko, T.; Pawlikowski, P.; et al. Low investment in sexual reproduction threatens plants adapted to phosphorus limitation. Nature 2013, 505, 82–86. [Google Scholar] [CrossRef]
- Gyaneshwar, P.; Kumar, G.N.; Parekh, L.J.; Poole, P.S. Role of soil microorganisms in improving p nutrition of plants. Plant Soil 2002, 245, 83–93. [Google Scholar] [CrossRef]
- Khan, M.S.; Zaidi, A.; Wani, P.A. Role of phosphate-solubilizing microorganisms in sustainable agriculture—A review. Agron. Sustain. Dev. 2007, 27, 29–43. [Google Scholar] [CrossRef]
- Coutinho, F.P.; Felix, W.P.; Yano-Melo, A.M. Solubilization of phosphates in vitro by Aspergillus spp. and Penicillium spp. Ecol. Eng. 2012, 42, 85–89. [Google Scholar] [CrossRef]
- Wang, T.; Liu, M.Q.; Li, H.X. Inoculation of phosphate-solubilizing bacteria Bacillus thuringiensis b1 increases available phosphorus and growth of peanut in acidic soil. Acta Agric. Scand. 2014, 64, 252–259. [Google Scholar]
- Collavino, M.M.; Sansberro, P.A.; Mroginski, L.A.; Aguilar, O.M. Comparison of in vitro solubilization activity of diverse phosphate-solubilizing bacteria native to acid soil and their ability to promote Phaseolus vulgaris growth. Biol. Fertil. Soils 2010, 46, 727–738. [Google Scholar] [CrossRef]
- Zak, D.; Goldhammer, T.; Cabezas, A.; Gelbrecht, J.; Gurke, R.; Wagner, C.; Reuter, H.; Augustin, J.; Klimkowska, A.; Mclnnes, R. Top soil removal reduces water pollution from phosphorus and dissolved organic matter and lowers methane emissions from rewetted peatlands. J. Appl. Ecol. 2018, 55, 311–320. [Google Scholar] [CrossRef]
- Ping, L.; Wang, K.; Zhou, C.; Xie, Y.; Yao, X.; Yin, H. Seed transcriptomics analysis in Camellia oleifera uncovers genes associated with oil content and fatty acid composition. Int. J. Mol. Sci. 2018, 19, 118. [Google Scholar]
- Lin, C.Y.; Fan, C.L. Fuel properties of biodiesel produced from Camellia oleifera Abel oil through supercritical-methanol transesterification. Fuel 2011, 90, 2240–2244. [Google Scholar] [CrossRef]
- Li, J. Soil, water and nutrient Loss in young plantation of the inter-cropped tea oil (Camellia oleifera) with different crops. Scientia Silvae Sinicae 2008, 44, 167–172, (In Chinese with English Abstract). [Google Scholar]
- Huang, W.; Liu, J.; Wang, Y.P.; Zhou, G.; Han, T.; Li, Y. Increasing phosphorus limitation along three successional forests in southern China. Plant Soil 2013, 364, 181–191. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, L.P.; Zhang, Y.; Hao, F.F.; Hu, D.N. Screening, identification and phosphate solubilizing capability of phosphate solubilizing bacteria in rhizosphere of Camellia oleifera Abel at red soil region. For. Res. 2015, 28, 409–416, (In Chinese with English Abstract). [Google Scholar]
- Huang, F.L.; Zhang, Y.; Zhang, L.P.; Wang, S.; Feng, Y.; Rong, N.H. Complete genome sequence of Bacillus megaterium JX285 isolated from Camellia oleifera rhizosphere. Comput. Biol. Chem. 2019, 79, 1–5. [Google Scholar] [CrossRef]
- Kalavrouziotis, I.K.; Koukoulakis, P.H. Soil pollution under the effect of treated municipal wastewater. Environ. Monit. Assess. 2011, 184, 6297–6305. [Google Scholar] [CrossRef]
- Bowman, R.A. A sequential extraction procedure with concentrated sulfuric acid and dilute base for soil organic phosphorus. Soil Sci. Soc. Am. J. 1989, 53, 362–366. [Google Scholar] [CrossRef]
- Wu, F.; Zhang, H.; Fang, F.; Liu, H.; Tang, M. Arbuscular mycorrhizal fungi alter nitrogen allocation in the leaves of Populus × canadensis ‘Neva’. Plant Soil 2017, 421, 477–491. [Google Scholar] [CrossRef]
- Mcgrath, R. Protein measurement by ninhydrin determination of amino acid released by alkaline hydrolysis. Anal. Biochem. 1972, 49, 95–102. [Google Scholar] [CrossRef]
- Pu, P.; Zhang, M.; Zhang, L.N. A study on temperature and time conditions of colorimetric method in measuring soil available phosphorus. Adv. Mater. Res. 2014, 838–841, 2047–2051. [Google Scholar] [CrossRef]
- Billings, G.K. The determination of potassium in oil field brines by atomic absorption spectrometry: Geological note. Clin. Infect. Dis. 1965, 13, 532–534. [Google Scholar]
- Turner, B.L.; Brenes-Arguedas, T.; Condit, R. Pervasive phosphorus limitation of tree species but not communities in tropical forests. Nature 2018, 555, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, N.; Kostic, L.; Djordjevic, A.; Nikolic, M. Phosphorus deficiency is the major limiting factor for wheat on alluvium polluted by the copper mine pyrite tailings: A black box approach. Plant Soil 2011, 339, 485–498. [Google Scholar] [CrossRef]
- Parekh, L.J.; Gyaneshwar, P.; Kumar, G.N.; Archana, G.; Poole, P.S.; Collins, M.D.; Hutson, R.A. Involvement of a phosphate starvation inducible glucose dehydrogenase in soil phosphate solubilization by Enterobacter asburiae. FEMS Microbiol. Lett. 1999, 171, 223–229. [Google Scholar]
- Bashan, Y.; Kamnev, A.A.; de-Bashan, L.E. A proposal for isolating and testing phosphate-solubilizing bacteria that enhance plant growth. Biol. Fertil. Soils 2013, 49, 1–2. [Google Scholar] [CrossRef]
- Ashmore, M.R. Assessing the future global impacts of ozone on vegetation. Plant Cell Environ. 2005, 28, 949–964. [Google Scholar] [CrossRef]
- Yu, X.; Liu, X.; Zhu, T.H.; Liu, G.H.; Mao, C. Isolation and characterization of phosphate-solubilizing bacteria from walnut and their effect on growth and phosphorus mobilization. Biol. Fertil. Soils 2011, 47, 437–446. [Google Scholar] [CrossRef]
- Wu, F.; Zhang, H.; Fang, F.; Tang, M. Nutrient allocation and photochemical responses of Populus × canadensis ‘Neva’ to nitrogen fertilization and exogenous Rhizophagus irregularis inoculation. Acta Physiol. Plant. 2018, 40, 152. [Google Scholar] [CrossRef]
- Panhwar, Q.A.; Radziah, O.; Zaharah, A.R.; Sariah, M.; Razi, I.M. Role of phosphate solubilizing bacteria on rock phosphate solubility and growth of aerobic rice. J. Environ. Biol. 2011, 32, 607–612. [Google Scholar] [PubMed]
- Wu, F.; Zhang, H.; Fang, F.; Wu, N.; Zhang, Y.; Tang, M. Effects of nitrogen and exogenous Rhizophagus irregularis on the nutrient status, photosynthesis and leaf anatomy of Populus × canadensis ‘Neva’. J. Plant Growth Regul. 2017, 36, 824–835. [Google Scholar] [CrossRef]
- Brodribb, T.J.; Field, T.S.; Jordan, G.J. Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiol. 2007, 144, 1890–1898. [Google Scholar] [CrossRef] [PubMed]
- Prodhan, M.A.; Finnegan, P.M.; Lambers, H. How does evolution in phosphorus-impoverished landscapes impact plant nitrogen and sulfur assimilation? Trends Plant Sci. 2019, 24, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Kerkhoff, A.J.; Fagan, W.F.; Elser, J.J.; Enquist, B.J. Phylogenetic and Growth Form Variation in the Scaling of Nitrogen and Phosphorus in the Seed Plants. Am. Nat. 2006, 168, E103–E122. [Google Scholar] [CrossRef]
- Diao, C.P.; Wang, Z.H.; Li, S.S.; Liu, L.; Wang, S.; Huang, N. Differences in grain nitrogen contents of high-yielding wheat cultivars and relation to NPK uptake and utilization in drylands. J. Plant Nutr. Fertil. 2018, 24, 285–295. [Google Scholar]
- Bayer, A.S.; Chow, A.W.; Morrison, J.O.; Guze, L.B. Bactericidal synergy between Penicillin or Ampicillin and aminoglycosides against antibiotic-tolerant lactobacilli. Antimicrob. Agents Chemother. 1980, 17, 359–363. [Google Scholar] [CrossRef]
- Kiratisin, P.; Apisarnthanarak, A.; Kaewdaeng, S. Synergistic activities between carbapenems and other antimicrobial agents against Acinetobacter baumannii including multidrug-resistant and extensively drug-resistant isolates. Int. J. Antimicrob. Agents 2010, 36, 243–246. [Google Scholar] [CrossRef]
- Rudresh, D.L.; Shivaprakash, M.K.; Prasad, R.D. Effect of combined application of rhizobium, phosphate solubilizing bacterium and Trichoderma spp. on growth, nutrient uptake and yield of chickpea (Cicer aritenium L.). Appl. Soil Ecol. 2005, 28, 139–146. [Google Scholar] [CrossRef]
| P Application | PSB Inoculation | Plant Height (cm) | Biomass (g) |
|---|---|---|---|
| P1 | T1 | 22.5 ± 0.9 b | 10.13 ± 0.90 c |
| T2 | 22.7 ± 1.9 b | 12.00 ± 0.60 b | |
| T3 | 26.2 ± 1.2 a | 14.27 ± 1.40 a | |
| CK1 | 15.7 ± 1.2 c | 6.67 ± 0.61 d | |
| CK2 | 17.8 ± 0.3 c | 7.00 ± 0.72 d | |
| P2 | T1 | 34.5 ± 1.0 c | 23.40 ± 1.59 c |
| T2 | 36.7 ± 1.8 b | 24.40 ± 0.72 b | |
| T3 | 37.5 ± 1.0 a | 26.97 ± 1.66 a | |
| CK1 | 16.5 ± 2.1 d | 7.67 ± 1.01 d | |
| CK2 | 18.2 ± 0.9 d | 7.97 ± 1.53 d | |
| P3 | T1 | 32.3 ± 1.5 a | 15.73 ± 0.90 b |
| T2 | 33.5 ± 0.9 a | 16.47 ± 0.99 b | |
| T3 | 34.5 ± 0.8 a | 19.13 ± 0.92 a | |
| CK1 | 16.9 ± 1.3 b | 7.24 ± 0.47 c | |
| CK2 | 17.9 ± 2.1 b | 7.50 ± 0.95 c | |
| Two-way ANOVA results | |||
| P | 0.00 ** | 0.00 ** | |
| PSB | 0.00 ** | 0.00 ** | |
| P × PSB | 0.00 ** | 0.00 ** | |
| P Application Levels | PSB Inoculation | Pn [μmol/(m2·s)] | Tr [μmol/(m2·s)] | WUE (μmol/mmol) | SPAD |
|---|---|---|---|---|---|
| P1 | T1 | 3.41 ± 0.07 c | 2.47 ± 0.07 a | 1.38 ± 0.03 b | 48.40 ± 1.64c |
| T2 | 3.64 ± 0.05 b | 2.59 ± 0.06 a | 1.40 ± 0.03 b | 51.37 ± 0.64 b | |
| T3 | 4.48 ± 0.20 a | 2.69 ± 0.09 a | 1.66 ± 0.07 a | 64.83 ± 2.70 a | |
| CK1 | 2.46 ± 0.25 d | 1.58 ± 0.09 b | 1.74 ± 0.10 a | 43.63 ± 2.21 d | |
| CK2 | 2.79 ± 0.43 d | 1.73 ± 0.03 b | 1.62 ± 0.23 a | 44.83 ± 1.72 d | |
| P2 | T1 | 4.14 ± 0.35 c | 2.20 ± 0.09 ab | 1.88 ± 0.08 a | 62.93 ± 2.40 b |
| T2 | 4.73 ± 0.40 b | 2.66 ± 0.07 a | 1.78 ± 0.14 a | 68.87 ± 1.66 a | |
| T3 | 5.69 ± 0.36 a | 2.80 ± 0.04 a | 2.03 ± 0.21 a | 71.30 ± 1.64 a | |
| CK1 | 2.93 ± 0.45 d | 1.87 ± 0.04 b | 1.56 ± 0.21 b | 47.83 ± 1.50 c | |
| CK2 | 3.07 ± 0.36 d | 1.83 ± 0.04 b | 1.68 ± 0.16 b | 48.31 ± 1.14 c | |
| P3 | T1 | 3.76 ± 0.10 c | 2.37 ± 0.03 ab | 1.58 ± 0.02 a | 52.73 ± 2.01 c |
| T2 | 3.93 ± 0.29 b | 2.42 ± 0.05 a | 1.62 ± 0.10 a | 57.87 ± 0.40 b | |
| T3 | 4.82 ± 0.20 a | 2.87 ± 0.06 a | 1.68 ± 0.04 a | 66.17 ± 2.15 a | |
| CK1 | 2.89 ± 0.23 d | 1.79 ± 0.07 b | 1.61 ± 0.10 a | 46.23 ± 1.00 d | |
| CK2 | 3.12 ± 0.16 d | 1.90 ± 0.10 b | 1.59 ± 0.02 a | 47.36 ± 0.92 d | |
| Two-way ANOVA results | |||||
| P | 0.00 ** | 0.03 * | 0.00 ** | 0.00 ** | |
| PSB | 0.01 * | 0.00 ** | 0.01 ** | 0.00 ** | |
| P × PSB | 0.17 NS | 0.00 ** | 0.17 NS | 0.00 ** | |
| P Application Levels | PSB Inoculation | Nitrogen Content (mg/kg) | Phosphorus Content (g/kg) |
|---|---|---|---|
| P1 | T1 | 1771.36 ± 86.22 b | 0.50 ± 0.03 b |
| T2 | 1924.55 ± 128.97 a | 0.56 ± 0.02 a | |
| T3 | 1989.24 ± 100.17 a | 0.55 ± 0.01 a | |
| CK1 | 1761.09 ± 102.14 b | 0.44 ± 0.02 c | |
| CK2 | 1788.55 ± 85.44 b | 0.46 ± 0.02 c | |
| P2 | T1 | 1849.29 ± 73.71 b | 0.59 ± 0.03 b |
| T2 | 2045.05 ± 50.33 a | 0.61 ± 0.02 b | |
| T3 | 2122.31 ± 101.49 a | 0.66 ± 0.01 a | |
| CK1 | 1791.32 ± 195.53 c | 0.47 ± 0.03 c | |
| CK2 | 1886.69 ± 52.92 b | 0.48 ± 0.01 c | |
| P3 | T1 | 1750.58 ± 105.83 b | 0.50 ± 0.04 a |
| T2 | 1766.99 ± 141.89 b | 0.51 ± 0.05 a | |
| T3 | 1973.33 ± 95.04 a | 0.59 ± 0.04 a | |
| CK1 | 1752.88 ± 92.92 b | 0.49 ± 0.06 a | |
| CK2 | 1831.46 ± 83.27 b | 0.51 ± 0.05 a | |
| Two-way ANOVA results | |||
| P | 0.01 * | 0.00 ** | |
| PSB | 0.00 ** | 0.00 ** | |
| P × PSB | 0.65 NS | 0.01 ** | |
| Index | P | PSB | P × PSB |
|---|---|---|---|
| Soil N content | 0.00 ** | 0.00 ** | 0.06 NS |
| Soil P content | 0.00 ** | 0.00 ** | 0.00 ** |
| Soil K content | 0.08 NS | 0.00 ** | 0.00 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, F.; Li, J.; Chen, Y.; Zhang, L.; Zhang, Y.; Wang, S.; Shi, X.; Li, L.; Liang, J. Effects of Phosphate Solubilizing Bacteria on the Growth, Photosynthesis, and Nutrient Uptake of Camellia oleifera Abel. Forests 2019, 10, 348. https://doi.org/10.3390/f10040348
Wu F, Li J, Chen Y, Zhang L, Zhang Y, Wang S, Shi X, Li L, Liang J. Effects of Phosphate Solubilizing Bacteria on the Growth, Photosynthesis, and Nutrient Uptake of Camellia oleifera Abel. Forests. 2019; 10(4):348. https://doi.org/10.3390/f10040348
Chicago/Turabian StyleWu, Fei, Jianrong Li, Yanliu Chen, Linping Zhang, Yang Zhang, Shu Wang, Xin Shi, Lei Li, and Junsheng Liang. 2019. "Effects of Phosphate Solubilizing Bacteria on the Growth, Photosynthesis, and Nutrient Uptake of Camellia oleifera Abel." Forests 10, no. 4: 348. https://doi.org/10.3390/f10040348
APA StyleWu, F., Li, J., Chen, Y., Zhang, L., Zhang, Y., Wang, S., Shi, X., Li, L., & Liang, J. (2019). Effects of Phosphate Solubilizing Bacteria on the Growth, Photosynthesis, and Nutrient Uptake of Camellia oleifera Abel. Forests, 10(4), 348. https://doi.org/10.3390/f10040348





