Altitudinal Biodiversity Gradient and Ecological Drivers for Different Lifeforms in the Baotianman Nature Reserve of the Eastern Qinling Mountains
Abstract
1. Introduction
2. Materials and Methods
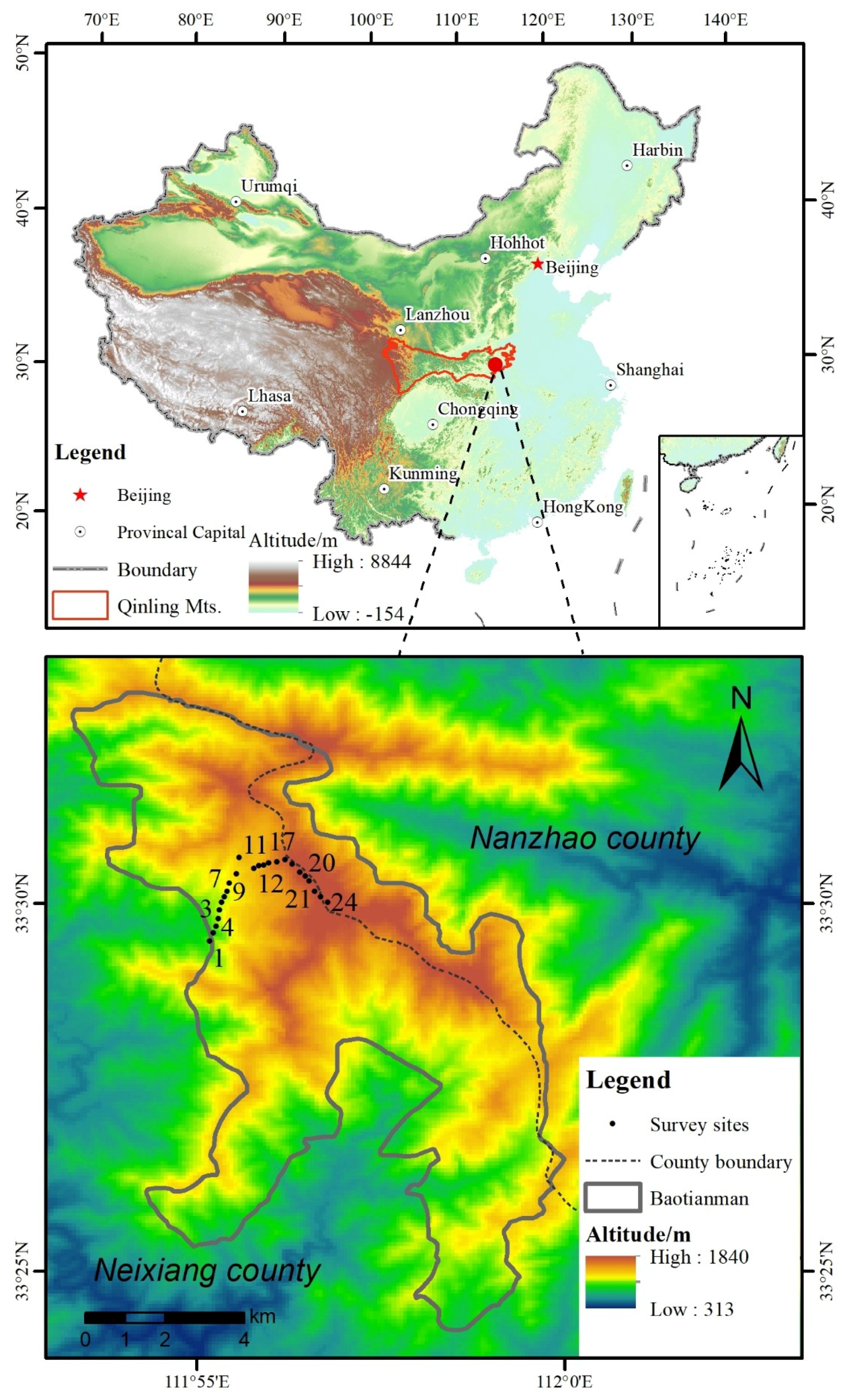
2.1. Study Area
2.2. Partitioning Community Dissimilarity
2.3. Hierarchical Clustering Based on Dissimilarity
2.4. Linear Regression
3. Results
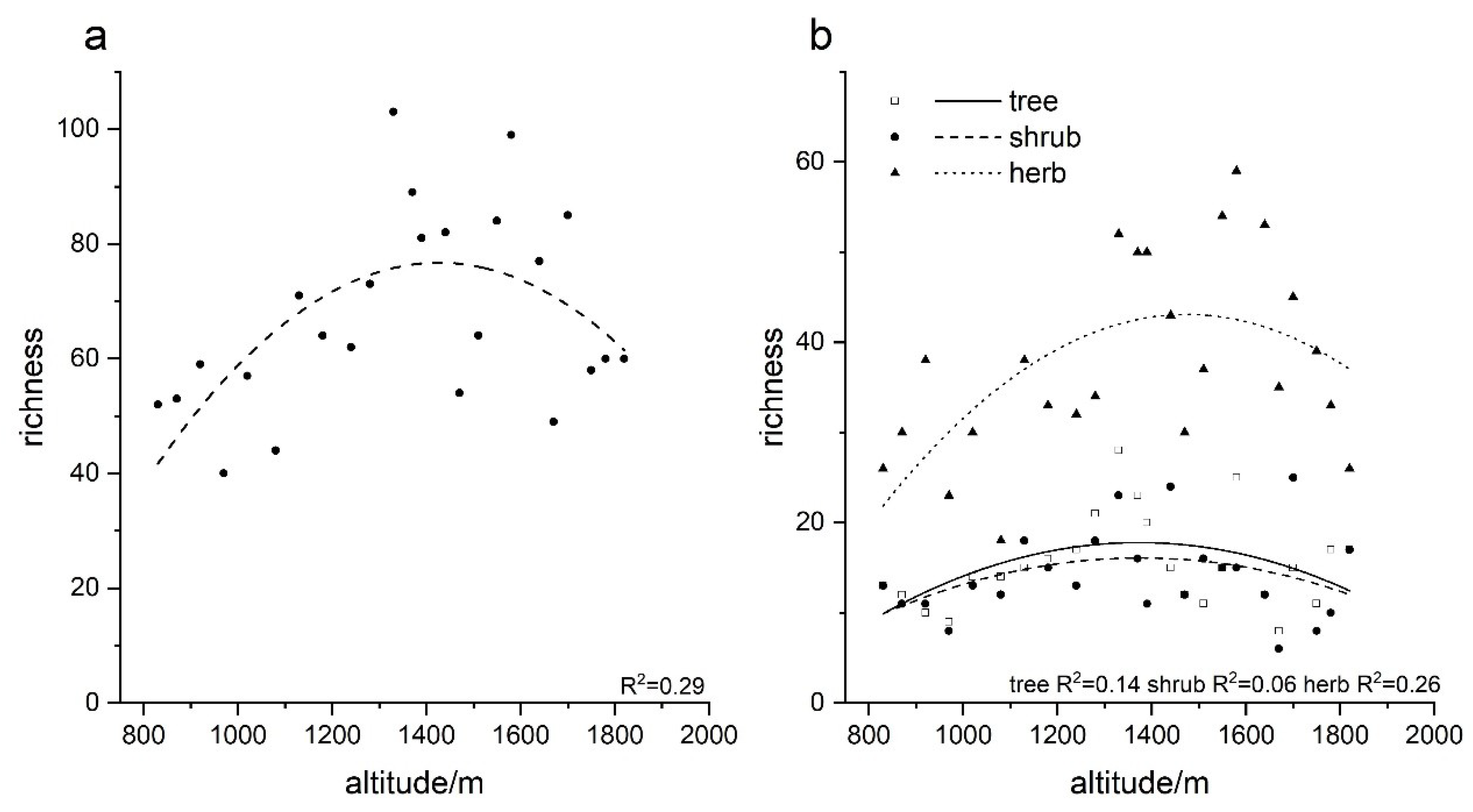
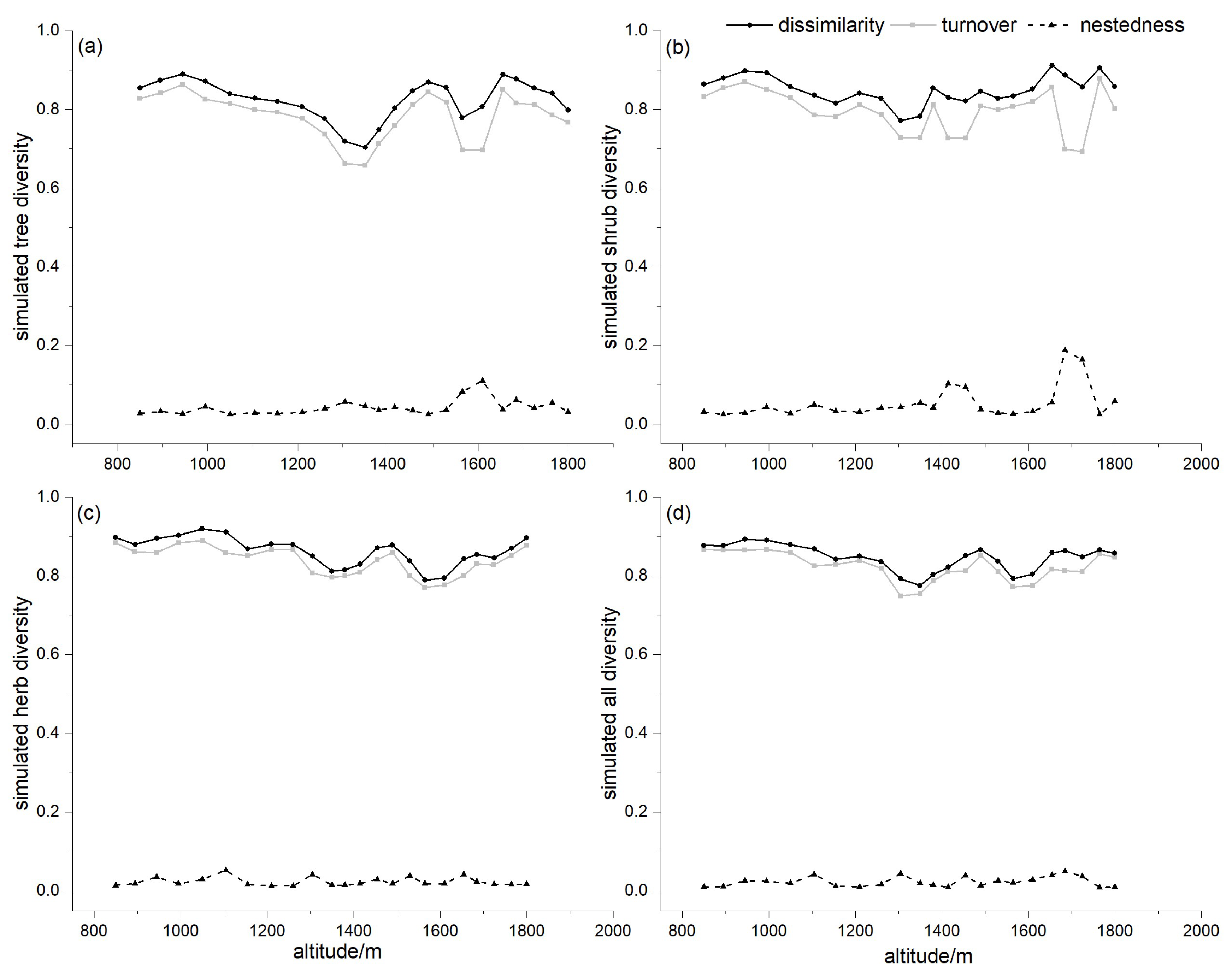
3.1. Species Composition along the Elevation Gradient
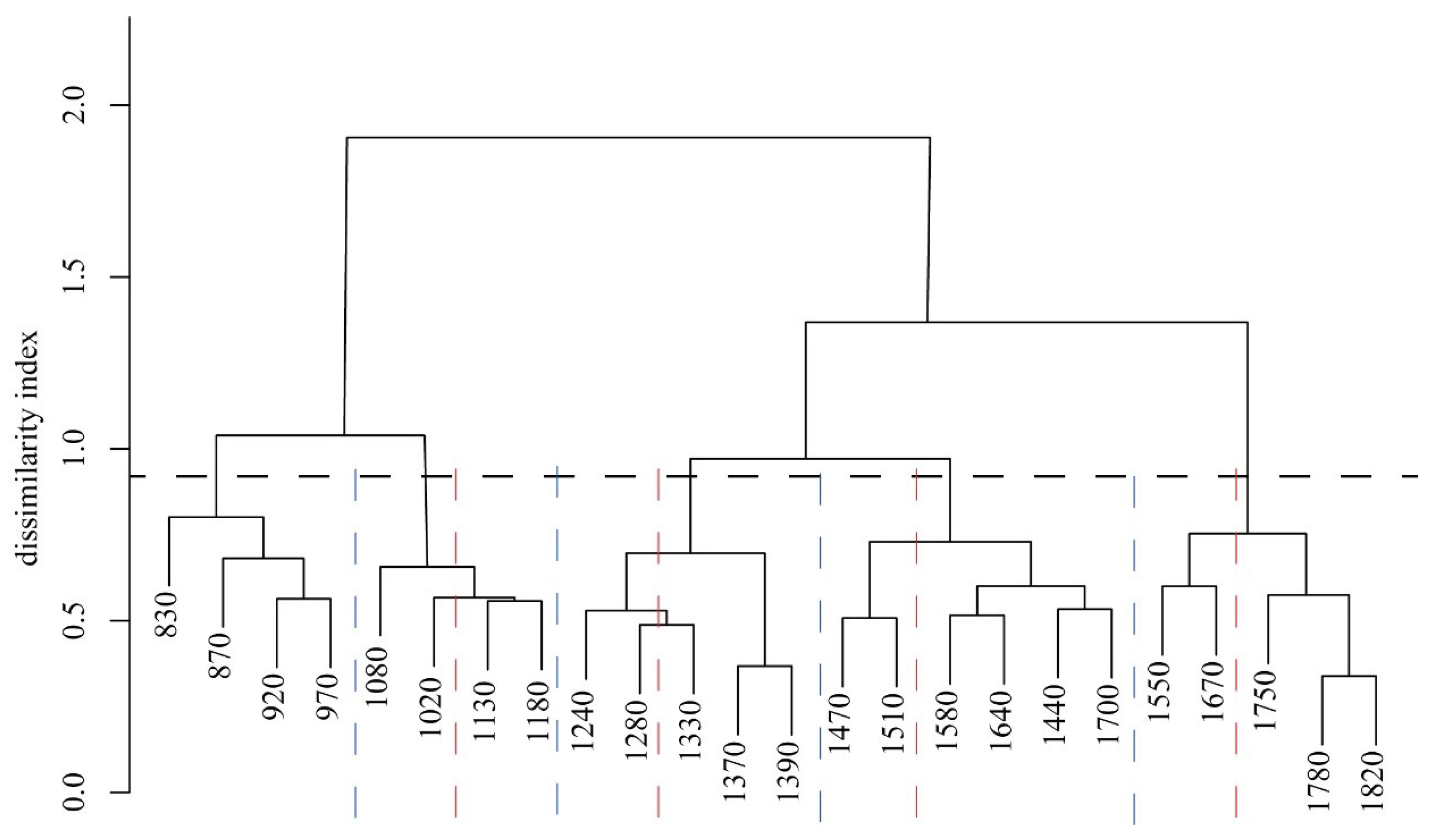
3.2. Vegetation Zonation by Pairwise Dissimilarity
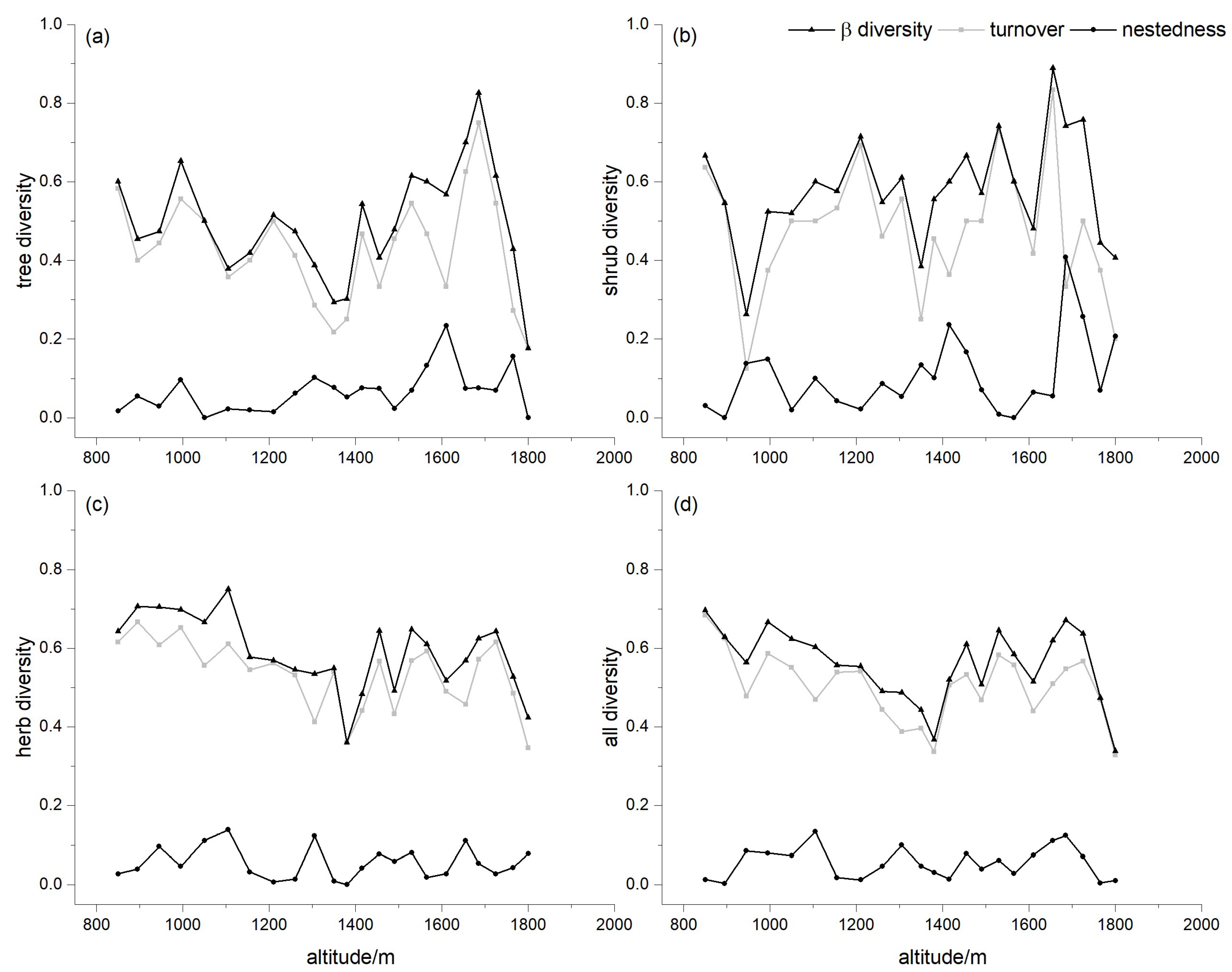
3.3. Partitioning Pairwise Multiple Sites Dissimilarity
3.4. Dissimilarity for Different Lifeforms
4. Discussion
4.1. Implications of Turnover and Nestedness for Mountain Vegetation Zonation
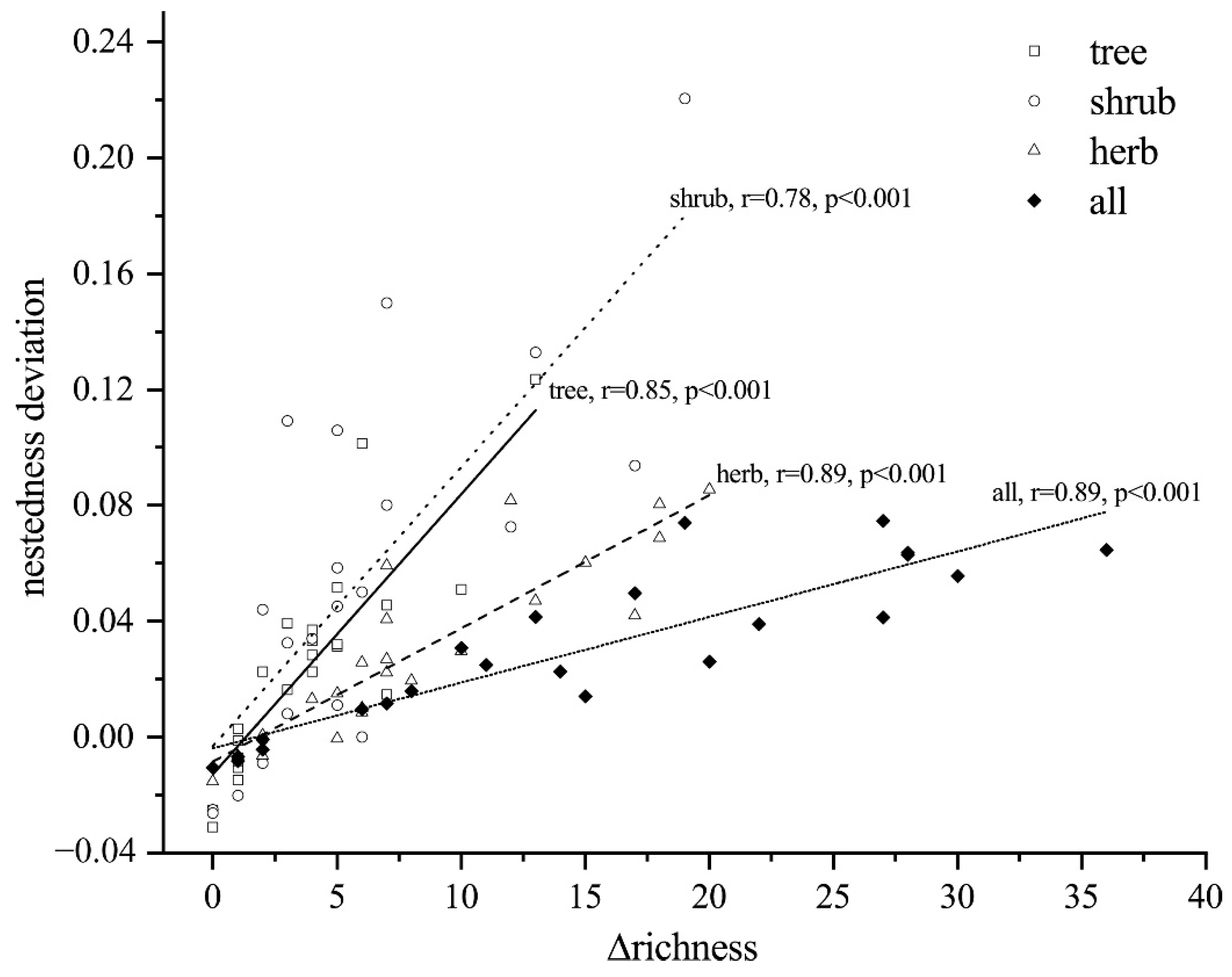
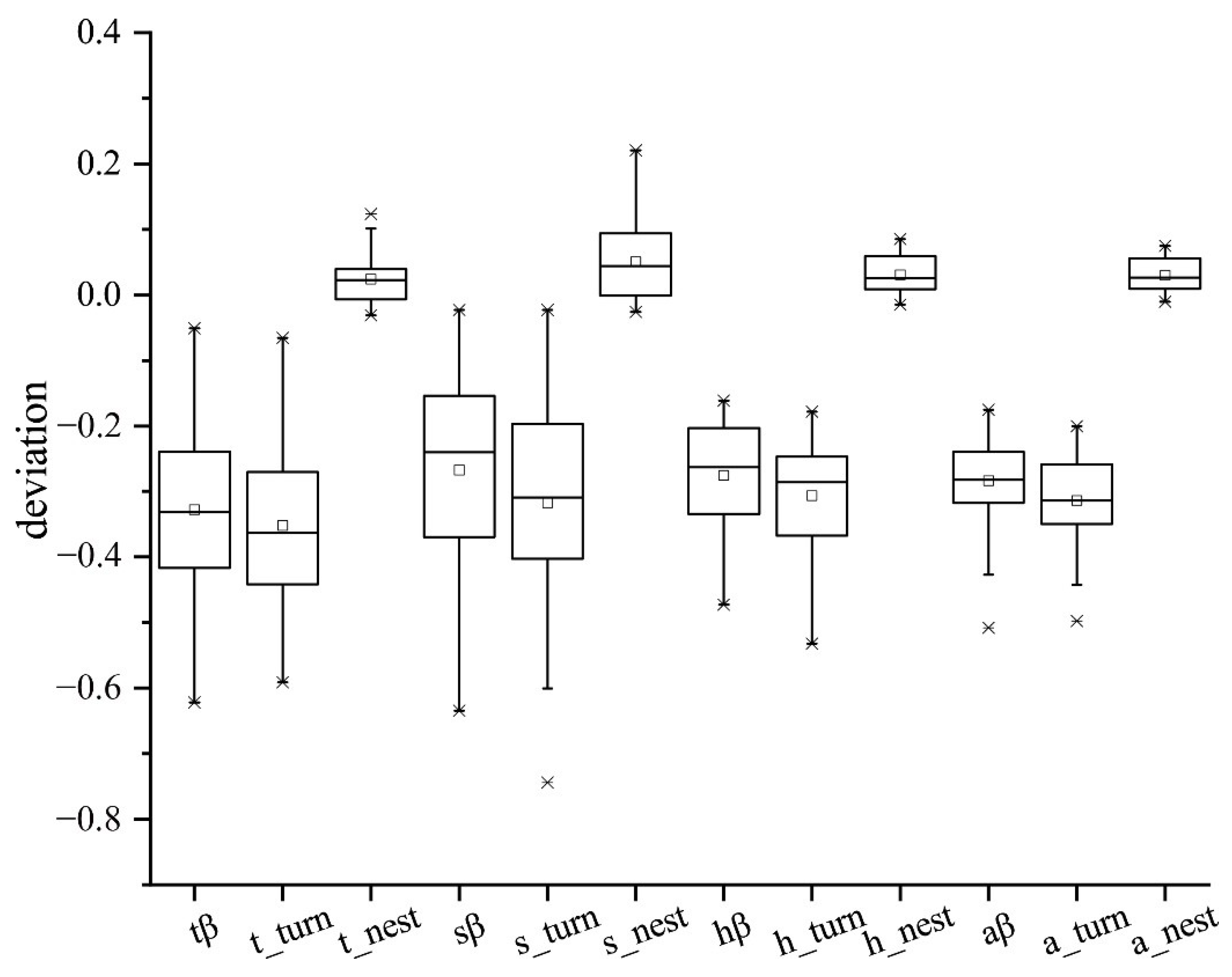
4.2. Patterns of Dissimilarity for Different Lifeforms
4.3. Ecological Drivers Underlying Turnover and Nestedness
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Whittaker, R.H. Vegetation of the Siskiyou mountains, Oregon and California. Ecol. Monogr. 1960, 30, 279–338. [Google Scholar] [CrossRef]
- Anderson, M.J.; Crist, T.O.; Chase, J.M.; Mark, V.; Inouye, B.D.; Freestone, A.L.; Sanders, N.J.; Cornell, H.V.; Comita, L.S.; Davies, K.F. Navigating the multiple meanings of β diversity: A roadmap for the practicing ecologist. Ecol. Lett. 2011, 14, 19–28. [Google Scholar] [CrossRef]
- Tuomisto, H. A diversity of beta diversities: Straightening up a concept gone awry. Part 1. Defining beta diversity as a function of alpha and gamma diversity. Ecography 2010, 33, 2–22. [Google Scholar] [CrossRef]
- Macarthur, R.; Recher, H.; Cody, M. On the Relation between Habitat Selection and Species Diversity. Am. Nat. 1966, 100, 319–332. [Google Scholar] [CrossRef]
- Thomas, O.C.; Joseph, A.V. Additive partitioning of rarefaction curves and species-area relationships: Unifying alpha-, beta- and gamma-diversity with sample size and habitat area. Ecol. Lett. 2010, 9, 923–932. [Google Scholar] [CrossRef]
- Marion, Z.H.; Fordyce, J.A.; Fitzpatrick, B.M. Pairwise beta diversity resolves an underappreciated source of confusion in calculating species turnover. Ecology 2017, 98, 933–939. [Google Scholar] [CrossRef]
- Wang, X.G.; Wiegand, T.; Anderson-Teixeira, K.J.; Bourg, N.A.; Hao, Z.Q.; Howe, R.; Jin, G.Z.; Orwig, D.A.; Spasojevic, M.J.; Wang, S.Z.; et al. Ecological drivers of spatial community dissimilarity, species replacement and species nestedness across temperate forests. Glob. Ecol. Biogeogr. 2018, 27, 581–592. [Google Scholar] [CrossRef]
- Baselga, A. The relationship between species replacement, dissimilarity derived from nestedness, and nestedness. Glob. Ecol. Biogeogr. 2012, 21, 1223–1232. [Google Scholar] [CrossRef]
- Condit, R.; Pitman, N.; Leigh, E.G.; Chave, J.; Terborgh, J.; Foster, R.B.; Núñez, P.; Aguilar, S.; Valencia, R.; Villa, G. Beta-Diversity in Tropical Forest Trees. Science 2002, 295, 666. [Google Scholar] [CrossRef]
- Hélène, M.; George, C.; Richard, C.; Stephen, H.; David, K.; Duncan, T.; Renato, V.; Green, J.L. A general framework for the distance-decay of similarity in ecological communities. Ecol. Lett. 2008, 11, 904–917. [Google Scholar] [CrossRef]
- Soininen, J.; Mcdonald, R.; Hillebrand, H.J.E. The distance decay of similarity in ecological communities. Ecography 2010, 30, 3–12. [Google Scholar] [CrossRef]
- Wang, X.; Wiegand, T.; Wolf, A.; Howe, R.; Davies, S.J.; Hao, Z. Spatial patterns of tree species richness in two temperate forests. J. Ecol. 2011, 99, 1382–1393. [Google Scholar] [CrossRef]
- Yang, Y.; Shen, Z.H.; Han, J.; Ciren, Z.Y. Plant Diversity along the Eastern and Western Slopes of Baima Snow Mountain, China. Forests 2016, 7, 89. [Google Scholar] [CrossRef]
- Catano, C.P.; Dickson, T.L.; Myers, J.A. Dispersal and neutral sampling mediate contingent effects of disturbance on plant beta-diversity: A meta-analysis. Ecol. Lett. 2017, 20, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Chase, J.M.; Myers, J.A. Disentangling the importance of ecological niches from stochastic processes across scales. Philos. Trans. R. Soc. Lond. 2011, 366, 2351–2363. [Google Scholar] [CrossRef]
- Kraft, N.J.B.; Adler, P.B.; Godoy, O.; James, E.C.; Fuller, S.; Levine, J.M. Community assembly, coexistence and the environmental filtering metaphor. Funct. Ecol. 2015, 29, 592–599. [Google Scholar] [CrossRef]
- Cavieres, L.A.; Hernandez-Fuentes, C.; Sierra-Almeida, A.; Kikvidze, Z. Facilitation among plants as an insurance policy for diversity in Alpine communities. Funct. Ecol. 2016, 30, 52–59. [Google Scholar] [CrossRef]
- Baselga, A. Partitioning the turnover and nestedness components of beta diversity. Glob. Ecol. Biogeogr. 2010, 19, 134–143. [Google Scholar] [CrossRef]
- Podani, J.; Schmera, D. A new conceptual and methodological framework for exploring and explaining pattern in presence–absence data. Oikos 2011, 120, 1625–1638. [Google Scholar] [CrossRef]
- Legendre, P. Interpreting the replacement and richness difference components of beta diversity. Glob. Ecol. Biogeogr. 2014, 23, 1324–1334. [Google Scholar] [CrossRef]
- Turtureanu, P.D.; Dengler, J. Different aspects of plant diversity show contrasting patterns in Carpathian forest openings. Plant Ecol. 2012, 213, 67–76. [Google Scholar] [CrossRef]
- Descombes, P.; Vittoz, P.; Guisan, A.; Pellissier, L. Uneven rate of plant turnover along elevation in grasslands. Alpine Bot. 2017, 127, 53–63. [Google Scholar] [CrossRef]
- Song, C. Baotianman Nature Reserve Scientific Research Collection; China Forestry Press: Beijing, China, 1994. (In Chinese) [Google Scholar]
- Chinese Flora Committee, Chinese Academy of Sciences. Flora of China; Science Press: Beijing, China, 1994. [Google Scholar]
- Baselga, A.; Orme, C.D.L. betapart: An R package for the study of beta diversity. Methods Ecol. Evol. 2012, 3, 808–812. [Google Scholar] [CrossRef]
- The R Core Team. R: A Language and Environment for Statistical Computing; The R Core Team: Vienna, Austria, 2019. [Google Scholar]
- Gotelli, N.J.; Graves, G.R. Null Models in Ecology; Smithsonian Institution: Washington, USA, 1996. [Google Scholar] [CrossRef][Green Version]
- Sanderson, J.G. Null model analysis of communities on gradients. J. Biogeogr. 2004, 31, 879–883. [Google Scholar] [CrossRef]
- Rahbek, C. The elevational gradient of species richness: A uniform pattern? Ecography 1995, 18, 200–205. [Google Scholar] [CrossRef]
- Yonghui, Y.; Baiping, Z.; Chao, Z. Geographical distribution of cripple tree forest and its importance for forest line in China (In Chinese). Progr. Geogr. 2017, 36, 491–499. [Google Scholar] [CrossRef][Green Version]
- Körner, C. Alpine Treelines: Functional Ecology of the Global High Elevation Tree Limits; Springer Science & Business Media: Basel, Switzerland, 2012. [Google Scholar]
- Jiang, Y.; Kang, M.Y.; Liu, S.; Tian, L.S.; Lei, M.D. A study on the vegetation in the east side of Helan Mountain. Plant Ecol. 2000, 149, 119–130. [Google Scholar] [CrossRef]
- Hemp, A. Continuum or zonation? Altitudinal gradients in the forest vegetation of Mt. Kilimanjaro. Plant Ecol. 2006, 184, 27–42. [Google Scholar] [CrossRef]
- Myers, J.A.; Chase, J.M.; Iván, J.; Jørgensen, P.M.; Alejandro, A.M.; Narel, P.Z.; Renate, S. Beta-diversity in temperate and tropical forests reflects dissimilar mechanisms of community assembly. Ecol. Lett. 2013, 16, 151–157. [Google Scholar] [CrossRef]
| Δrichness | β_exp | Turnover_exp | Nestedness_exp | β_obs | Turnover_obs | Nestedness_obs |
|---|---|---|---|---|---|---|
| tree | −0.28 | −0.56 ** | 0.97 *** | 0.36 | 0.01 | 0.94 *** |
| shrub | −0.01 | −0.78 *** | 0.97 *** | 0.32 | −0.28 | 0.92 *** |
| herb | 0.25 | −0.05 | 0.96 *** | 0.48 | 0.09 | 0.94 *** |
| all | −0.07 | −0.41 * | 0.98 *** | 0.49 * | 0.08 | 0.95 *** |
| Variables | C1 | C2 | CR (%) | VIF | t Value | S-W Test | Adjusted R2 | |
|---|---|---|---|---|---|---|---|---|
| W | P | |||||||
| tree turnover | 0.23 *** | 0.36 | 18.89 | 2.67 | 10.06 | 0.97 | 0.70 | 0.99 |
| shrub turnover | 0.18 *** | 0.34 | 18.18 | 2.73 | 9.57 | |||
| herb turnover | 0.61 *** | 0.61 | 32.26 | 1.55 | 22.53 | |||
| tree nestedness | 0.25 *** | 0.15 | 8.00 | 1.07 | 6.74 | |||
| shrub nestedness | 0.18 *** | 0.20 | 10.80 | 2.47 | 5.98 | |||
| herb nestedness | 0.50 *** | 0.22 | 11.88 | 1.06 | 10.02 | |||
| intercept | –0.01 | –0.75 | ||||||
| Lifeform | Turnover_obs | Nestedness_obs | Turnover_exp | Nestedness_exp |
|---|---|---|---|---|
| tree | 86.45 ± 10.68 | 13.55 ± 10.68 | 94.77 ± 2.55 | 5.22 ± 2.55 |
| shrub | 80.54 ± 17.06 | 19.46 ± 17.06 | 93.54 ± 4.88 | 6.46 ± 4.88 |
| herb | 90.81 ± 6.45 | 9.19 ± 6.45 | 97.22 ± 1.24 | 2.78 ± 1.24 |
| all | 90.70 ± 6.25 | 9.30 ± 6.25 | 97.17 ± 1.49 | 2.82 ± 1.49 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.; Wang, J.; Yu, F.; Zhang, X.; Yao, Y.; Zhang, B. Altitudinal Biodiversity Gradient and Ecological Drivers for Different Lifeforms in the Baotianman Nature Reserve of the Eastern Qinling Mountains. Forests 2019, 10, 332. https://doi.org/10.3390/f10040332
Zhao C, Wang J, Yu F, Zhang X, Yao Y, Zhang B. Altitudinal Biodiversity Gradient and Ecological Drivers for Different Lifeforms in the Baotianman Nature Reserve of the Eastern Qinling Mountains. Forests. 2019; 10(4):332. https://doi.org/10.3390/f10040332
Chicago/Turabian StyleZhao, Chao, Jing Wang, Fuqin Yu, Xinghang Zhang, Yonghui Yao, and Baiping Zhang. 2019. "Altitudinal Biodiversity Gradient and Ecological Drivers for Different Lifeforms in the Baotianman Nature Reserve of the Eastern Qinling Mountains" Forests 10, no. 4: 332. https://doi.org/10.3390/f10040332
APA StyleZhao, C., Wang, J., Yu, F., Zhang, X., Yao, Y., & Zhang, B. (2019). Altitudinal Biodiversity Gradient and Ecological Drivers for Different Lifeforms in the Baotianman Nature Reserve of the Eastern Qinling Mountains. Forests, 10(4), 332. https://doi.org/10.3390/f10040332





