Diversity and Distribution of Phytophthora Species in Protected Natural Areas in Sicily
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling and Phytophthora Isolation
2.2. Morphological Characterization of Isolates
2.3. Molecular Identification of Isolates
3. Results
3.1. Phytophthora Diversity and Distribution in Forest Stands
3.2. Phytophthora Diversity and Distribution in Rivers within PNAs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martino, A.D.; Raimondo, F.M. Biological and chorological survey of the Sicilian Flora. Webbia 1979, 34, 309–335. [Google Scholar] [CrossRef]
- Schönfelder, I.; Schönfelder, P. La Flora Mediterranea; Istituto Geografico De Agostini: Novara, Italy, 1996. [Google Scholar]
- Giardina, G.; Raimondo, F.M.; Spadaro, V. A Catalogue of Plants Growing in Sicily; Bocconea: Palermo, Italy, 2007; Volume 20, ISBN 9788879150224. [Google Scholar]
- Conti, F.; Abbate, G.; Alessandrini, A.; Blasi, C.; Bonacquisti, S.; Scassellati, E. La flora vascolare italiana: Ricchezza e originalità a livello nazionale e regionale. In Stato Delle Conoscenze Sulla Flora Vascolare D’italia; Scoppola, A., Blasi, C., Eds.; Palombi Editori: Rome, Italy, 2005; pp. 18–22. [Google Scholar]
- Rizza, C.; Scibetta, S.; Pane, A.; Maetzke, F.; La Mela Veca, D.S.; Culotta, S.; Granata, G.; La Spada, F.; Aloi, F.; Faedda, R.; et al. A new approach in the monitoring of the phytosanitary conditions of forests: The case of oak and beech stands in the Sicilian Regional Parks. Italian J. Mycol. 2016, 45, 29–46. [Google Scholar]
- Jung, T.; Horta Jung, M.; Cacciola, S.O.; Cech, T.; Bakonyi, J.; Seress, D.; Mosca, S.; Schena, L.; Seddaiu, S.; Pane, A.; et al. Multiple new cryptic pathogenic Phytophthora species from Fagaceae forests in Austria, Italy and Portugal. IMA Fungus 2017, 8, 219–244. [Google Scholar] [CrossRef]
- Erwin, D.C.; Ribeiro, O.K. Phytophthora Diseases Worldwide; American Phytopathological Society (APS Press): St. Paul, MN, USA, 1996. [Google Scholar]
- Hansen, E.M.; Reeser, P.W.; Sutton, W. Phytophthora Beyond Agriculture. Annu. Rev. Phytopathol. 2012, 50, 359–378. [Google Scholar] [CrossRef] [PubMed]
- Jung, T. Beech decline in Central Europe driven by the interaction between Phytophthora infections and climatic extremes. For. Pathol. 2009, 39, 73–94. [Google Scholar] [CrossRef]
- Jung, T.; Pérez-Sierra, A.; Durán, A.; Jung, M.H.; Balci, Y.; Scanu, B. Canker and decline diseases caused by soil- and airborne Phytophthora species in forests and woodlands. Persoonia Mol. Phylogeny Evolut. Fungi 2018, 40, 182–220. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Blaschke, H.; Neumann, P. Isolation, identification and pathogenicity of Phytophthora species from declining oak stands. Eur. J. For. Pathol. 1996, 26, 253–272. [Google Scholar] [CrossRef]
- Frisullo, S.; Lima, G.; Magnano di San Lio, G.; Camele, I.; Melissano, L.; Puglisi, I.; Pane, A.; Agosteo, G.E.; Prudente, L.; Cacciola, S.O. Phytophthora cinnamomi Involved in the Decline of Holm Oak (Quercus ilex) Stands in Southern Italy. For.Sci. 2018, 64, 290–298. [Google Scholar] [CrossRef]
- Jung, T.; Blaschke, H.; Oßwald, W. Involvement of soilborne Phytophthora species in Central European oak decline and the effect of site factors on the disease. Plant Pathol. 2000, 49, 706–718. [Google Scholar] [CrossRef]
- Jung, T.; Vettraino, A.M.; Cech, T.; Vannini, A. The impact of invasive Phytophthora species on European forests. In Phytophthora: A Global Perspective; Lamour, K., Ed.; CABI: Wallingford, UK, 2013; pp. 146–158. [Google Scholar]
- Ristaino, J.B.; Gumpertz, M.L. New frontiers in the study of dispersal and spatial analysis of epidemics caused by species in the Genus Phytophthora. Annu. Rev. Phytopathol. 2000, 38, 541–576. [Google Scholar] [CrossRef]
- Rizzo, D.M.; Garbelotto, M.; Hansen, E.M. Phytophthora ramorum: Integrative research and management of an emerging pathogen in California and Oregon forests. Annu. Rev. Phytopathol. 2005, 43, 309–335. [Google Scholar] [CrossRef]
- Balcì, Y.; Halmschlager, E. Phytophthora species in oak ecosystems in Turkey and their association with declining oak trees. Plant Pathol. 2003, 52, 694–702. [Google Scholar] [CrossRef]
- Balci, Y.; Balci, S.; Mac Donald, W.L.; Gottschalk, K.W. Pathogenicity of Phytophthora species isolated from rhizosphere soil in the eastern United States. In Proceedings of the Sudden Oak Death Third Science Symposium; Frankel, S.J., Kliejunas, J.T., Palmieri, K.M., Eds.; Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2008; pp. 225–226. [Google Scholar]
- Cooke, D.E.L.; Schena, L.; Cacciola, S.O. Tools to detect, identify and monitor Phytophthora species in natural ecosystems. J. Plant Pathol. 2007, 89, 13–28. [Google Scholar]
- Jung, T.; Stukely, M.J.C.; Hardy, G.E.S.J.; White, D.; Paap, T.; Dunstan, W.A.; Burgess, T.I. Multiple new Phytophthora species from ITS Clade 6 associated with natural ecosystems in Australia: Evolutionary and ecological implications. Persoonia Mol. Phylogeny Evolut. Fungi 2011, 26, 13–39. [Google Scholar] [CrossRef]
- Jung, T.; Chang, T.T.; Bakonyi, J.; Seress, D.; Pérez-Sierra, A.; Yang, X.; Hong, C.; Scanu, B.; Fu, C.H.; Hsueh, K.L.; et al. Diversity of Phytophthora species in natural ecosystems of Taiwan and association with disease symptoms. Plant Pathol. 2017, 66, 194–211. [Google Scholar] [CrossRef]
- Pérez-Sierra, A.; López-García, C.; León, M.; García-Jiménez, J.; Abad-Campos, P.; Jung, T. Previously unrecorded low-temperature Phytophthora species associated with Quercus decline in a Mediterranean forest in eastern Spain. For. Pathol. 2013, 43, 331–339. [Google Scholar] [CrossRef]
- Rea, A.J.; Burgess, T.I.; Hardy, G.E.S.J.; Stukely, M.J.C.; Jung, T. Two novel and potentially endemic species of Phytophthora associated with episodic dieback of Kwongan vegetation in the south-west of Western Australia. Plant Pathol. 2011, 60, 1055–1068. [Google Scholar] [CrossRef]
- Ruano-Rosa, D.; Schena, L.; Agosteo, G.E.; Magnano di San Lio, G.; Cacciola, S.O. Phytophthora oleae sp. nov. causing fruit rot of olive in southern Italy. Plant Pathol. 2018, 67, 1362–1373. [Google Scholar] [CrossRef]
- Scanu, B.; Linaldeddu, B.T.; Deidda, A.; Jung, T. Diversity of Phytophthora Species from Declining Mediterranean Maquis Vegetation, including Two New Species, Phytophthora crassamura and P. ornamentata sp. nov. PLoS ONE 2015, 10, e0143234. [Google Scholar] [CrossRef]
- Tkaczyk, M.; Nowakowska, J.A.; Oszako, T. Phytophthora species isolated from ash stands in Białowieża Forest nature reserve. Forest Pathol. 2016, 46, 660–662. [Google Scholar] [CrossRef]
- Linaldeddu, B.T.; Scanu, B.; Maddau, L.; Franceschini, A. Diplodia corticola and Phytophthora cinnamomi: The main pathogens involved in holm oak decline on Caprera Island (Italy). For. Pathol. 2014, 44, 191–200. [Google Scholar] [CrossRef]
- Scanu, B.; Hunter, G.C.; Linaldeddu, B.T.; Franceschini, A.; Maddau, L.; Jung, T.; Denman, S. A taxonomic re-evaluation reveals that Phytophthora cinnamomi and P. cinnamomi var. parvispora are separate species. For. Pathol. 2014, 44, 1–20. [Google Scholar]
- Abad, Z.G.; Abad, J.A.; Cacciola, S.O.; Pane, A.; Faedda, R.; Moralejo, E.; Pérez-Sierra, A.; Abad-Campos, P.; Alvarez-Bernaola, L.A.; Bakonyi, J.; et al. Phytophthora niederhauserii sp. nov., a polyphagous species associated with ornamentals, fruit trees and native plants in 13 countries. Mycologia 2014, 106, 431–447. [Google Scholar] [CrossRef] [PubMed]
- Brasier, C.M. The biosecurity threat to the UK and global environment from international trade in plants. Plant Pathol. 2008, 57, 792–808. [Google Scholar] [CrossRef]
- Jung, T.; Blaschke, M. Phytophthora root and collar rot of alders in Bavaria: Distribution, modes of spread and possible management strategies. Plant Pathol. 2004, 53, 197–208. [Google Scholar] [CrossRef]
- Jung, T.; Orlikowski, L.; Henricot, B.; Abad-Campos, P.; Aday, A.G.; Aguín Casal, O.; Bakonyi, J.; Cacciola, S.O.; Cech, T.; Chavarriaga, D.; et al. Widespread Phytophthora infestations in European nurseries put forest, semi-natural and horticultural ecosystems at high risk of Phytophthora diseases. For. Pathol. 2016, 46, 134–163. [Google Scholar] [CrossRef]
- Migliorini, D.; Ghelardini, L.; Tondini, E.; Luchi, N.; Santini, A. The potential of symptomless potted plants for carrying invasive soilborne plant pathogens. Divers. Distrib. 2015, 21, 1218–1229. [Google Scholar] [CrossRef]
- Pérez-Sierra, A.; Jung, T. Phytophthora in woody ornamental nurseries. In Phytophthora: A Global Perspective; Lamour, K., Ed.; CABI: Wallingford, UK, 2013; pp. 166–177. [Google Scholar]
- Prigigallo, M.I.; Mosca, S.; Cacciola, S.O.; Cooke, D.E.L.; Schena, L. Molecular analysis of Phytophthora diversity in nursery-grown ornamental and fruit plants. Plant Pathol. 2015, 64, 1308–1319. [Google Scholar] [CrossRef]
- Prigigallo, M.I.; Abdelfattah, A.; Cacciola, S.O.; Faedda, R.; Sanzani, S.M.; Cooke, D.E.L.; Schena, L. Metabarcoding analysis of Phytophthora diversity using genus-specific primers and 454 pyrosequencing. Phytopathology 2016, 106, 305–313. [Google Scholar] [CrossRef]
- Català, S.; Pérez-Sierra, A.; Abad-Campos, P. The use of genus-specific amplicon pyrosequencing to assess Phytophthora species diversity using eDNA from soil and water in northern spain. PLoS ONE 2015, 10, e0119311. [Google Scholar] [CrossRef]
- Dunstan, W.A.; Howard, K.; Stj. Hardy, G.E.; Burgess, T.I. An overview of Australia’s Phytophthora species assemblage in natural ecosystems recovered from a survey in Victoria. IMA Fungus 2016, 7, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Nagel, J.H.J.H.; Slippers, B.; Wingfield, M.J.M.J.J.; Gryzenhout, M. Multiple Phytophthora species associated with a single riparian ecosystem in South Africa. Mycologia 2015, 107, 915–925. [Google Scholar] [CrossRef]
- Reeser, P.W.; Sutton, W.; Hansen, E.M.; Remigi, P.; Adams, G.C. Phytophthora species in forest streams in Oregon and Alaska. Mycologia 2011, 103, 22–35. [Google Scholar] [CrossRef]
- Stamler, R.A.; Sanogo, S.; Goldberg, N.P.; Randall, J.J. Phytophthora species in rivers and streams of the Southwestern United sStates. Appl. Environ. Microbiol. 2016, 82, 4696–4704. [Google Scholar] [CrossRef]
- Jung, T.; Horta Jung, M.; Scanu, B.; Seress, D.; Kovács, G.M.; Maia, C.; Pérez-Sierra, A.; Chang, T.T.; Chandelier, A.; Heungens, K.; et al. Six new Phytophthora species from ITS Clade 7a including two sexually functional heterothallic hybrid species detected in natural ecosystems in Taiwan. Persoonia Mol. Phylogeny Evolut. Fungi 2017, 38, 100–135. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Tyler, B.M.; Hong, C. An expanded phylogeny for the genus Phytophthora. IMA Fungus 2017, 8, 355–384. [Google Scholar] [CrossRef]
- Cooke, D.E.; Drenth, A.; Duncan, J.M.; Wagels, G.; Brasier, C.M. A molecular phylogeny of Phytophthora and related oomycetes. Fungal Genet. Biol. 2000, 30, 17–32. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press, Inc.: San Diego, CA, USA, 1990; Volume 18, pp. 315–322. [Google Scholar]
- Safaiefarahani, B.; Mostowfizadeh-Ghalamfarsa, R.; Hardy, G.E.S.J.; Burgess, T.I. Re-evaluation of the Phytophthora cryptogea species complex and the description of a new species, Phytophthora pseudocryptogea sp. nov. Mycol. Prog. 2015, 14, 1–12. [Google Scholar] [CrossRef]
- Scibetta, S.; Schena, L.; Chimento, A.; Cacciola, S.O.; Cooke, D.E.L. A molecular method to assess Phytophthora diversity in environmental samples. J. Microbiol. Methods 2012, 88, 356–368. [Google Scholar] [CrossRef]
- Hansen, E.; Delatour, C. Phytophthora species in oak forests of north-east France. Ann. For. Sci. 1999, 56, 539–547. [Google Scholar] [CrossRef]
- Vettraino, A.M.; Barzanti, G.P.; Bianco, M.C.; Ragazzi, A.; Capretti, P.; Paoletti, E.; Luisi, N.; Anselmi, N.; Vannini, A. Occurrence of Phytophthora species in oak stands in Italy and their association with declining oak trees. Forest Pathology 2002, 32, 19–28. [Google Scholar] [CrossRef]
- Hüberli, D.; Hardy, G.E.S.J.; White, D.; Williams, N.; Burgess, T.I. Fishing for Phytophthora from Western Australia’s waterways: A distribution and diversity survey. Australasian Plant Pathol. 2013, 42, 251–260. [Google Scholar] [CrossRef]
- Oh, E.; Gryzenhout, M.; Wingfield, B.D.; Wingfield, M.J.; Burgess, T.I. Surveys of soil and water reveal a goldmine of Phytophthora diversity in South African natural ecosystems. IMA Fungus 2013, 4, 123–131. [Google Scholar] [CrossRef]
- Shrestha, S.K.; Zhou, Y.; Lamour, K. Oomycetes baited from streams in Tennessee 2010-2012. Mycologia 2013, 105, 1516–1523. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Burgess, T.I. Re-evaluation of Phytophthora citricola isolates from multiple woody hosts in Europe and North America reveals a new species, Phytophthora plurivora sp. nov. Persoonia 2009, 22, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Maseko, B.; Burgess, T.I.; Coutinho, T.A.; Wingfield, M.J. Two new Phytophthora species from South African Eucalyptus plantations. Mycol. Res. 2007, 111, 1321–1338. [Google Scholar] [CrossRef]
- Jung, T.; Hansen, E.M.; Winton, L.; Oswald, W.; Delatour, C. Three new species of Phytophthora from European oak forests. Mycol. Res. 2002, 106, 397–411. [Google Scholar] [CrossRef]
- Scanu, B.; Vannini, A.; Franceschini, A.; Vettraino, A.M.; Ginetti, B.; Moricca, S. Phytophthora spp. in Mediterranean forests. In Proceedings of the Second International Congress of Silviculture, Florence, Italy, 26–29 November 2014; Ciancio, O., Ed.; Accademia Italiana di Scienze Forestali: Florence, Italy, 2014; pp. 402–407. [Google Scholar]
- Vettraino, A.M.; Natili, G.; Anselmi, N.; Vannini, A. Recovery and pathogenicity of Phytophthora species associated with a resurgence of ink disease in Castanea sativa in Italy. Plant Pathol. 2001, 50, 90–96. [Google Scholar] [CrossRef]
- Vettraino, A.M.; Morel, O.; Perlerou, C.; Robin, C.; Diamandis, S.; Vannini, A. Occurrence and distribution of Phytophthora species in European chestnut stands, and their association with Ink Disease and crown decline. Eur. J. Plant Pathol. 2005, 111, 169. [Google Scholar] [CrossRef]
- Jung, T.; Hudler, G.W.; Jensen-Tracy, S.L.; Griffiths, H.M.; Fleischmann, F.; Osswald, W. Involvement of Phytophthora species in the decline of European beech in Europe and the USA. Mycologist 2005, 19, 159–166. [Google Scholar] [CrossRef]
- Mrázková, M.; Černý, K.; Tomšovský, M.; Strnadová, V.; Gregorová, B.; Holub, V.; Pánek, M.; Havrdová, L.; Hejná, M. Occurrence of Phytophthora multivora and Phytophthora plurivora in the Czech Republic. Plant Protect. Sci. 2013, 49, 155–164. [Google Scholar] [CrossRef]
- Orlikowski, L.B.; Ptaszek, M.; Rodziewicz, A.; Nechwatal, J.; Thinggaard, K.; Jung, T. Phytophthora root and collar rot of mature Fraxinus excelsior in forest stands in Poland and Denmark. Forest Pathol. 2011, 41, 510–519. [Google Scholar] [CrossRef]
- Jung, T.; Blaschke, H. Phytophthora root rot in declining forest trees. Phyton (Austria) 1996, 36, 95–102. [Google Scholar]
- Weiland, J.E.; Nelson, A.H.; Hudler, G.W. Aggressiveness of Phytophthora cactorum, P. citricola I, and P. plurivora from European Beech. Plant Dis. 2010, 94, 1009–1014. [Google Scholar] [CrossRef]
- Barber, P.A.; Paap, T.; Burgess, T.I.; Dunstan, W.; Hardy, G.E.S.J. A diverse range of Phytophthora species are associated with dying urban trees. Urban For. Urban Green. 2013, 12, 569–575. [Google Scholar] [CrossRef]
- Burgess, T.I.; White, D.; McDougall, K.M.; Garnas, J.; Dunstan, W.A.; Català, S.; Carnegie, A.J.; Worboys, S.; Cahill, D.; Vettraino, A.M.; et al. Distribution and diversity of Phytophthora across Australia. Pac. Conserv. Biol. 2017, 23, 150–162. [Google Scholar] [CrossRef]
- Scott, P.M.; Burgess, T.I.; Barber, P.A.; Shearer, B.L.; Stukely, M.J.C.; Hardy, G.E.S.J.; Jung, T. Phytophthora multivora sp. nov., a new species recovered from declining Eucalyptus, Banksia, Agonis and other plant species in Western Australia. Persoonia 2009, 22, 1–13. [Google Scholar] [CrossRef]
- Pane, A.; Granata, G.; Cacciola, S.O.; Puglisi, I.; Evoli, M.; Aloi, F.; La Spada, F.; Magnano di San Lio, G.; Zambounis, A. First Report of Root Rot of White Mulberry Caused by Simultaneous Infections of Phytophthora megasperma and P. multivora in Italy. Plant Dis. 2017, 101, 260. [Google Scholar] [CrossRef]
- Szabó, I.; Lakatos, F.; Sipos, G. Occurrence of soilborne Phytophthora species in declining broadleaf forests in Hungary. Eur. J. Plant Pathol. 2013, 137, 159–168. [Google Scholar] [CrossRef]
- Scarlett, K.; Daniel, R.; Shuttleworth, L.A.; Roy, B.; Bishop, T.F.A.; Guest, D.I. Phytophthora in the Gondwana Rainforests of Australia World Heritage Area. Australasian Plant Pathol. 2015, 44, 335–348. [Google Scholar] [CrossRef]
- Crous, P.W.; Wingfield, M.J.; Burgess, T.I.; Hardy, G.E.S.J.; Barber, P.A.; Alvarado, P.; Barnes, C.W.; Buchanan, P.K.; Heykoop, M.; Moreno, G.; et al. Fungal Planet description sheets. Persoonia Mol. Phylogeny Evolut. Fungi 2017, 558–624. [Google Scholar] [CrossRef]
- Puglisi, I.; De Patrizio, A.; Schena, L.; Jung, T.; Evoli, M.; Pane, A.; Van Hoa, N.; Van Tri, M.; Wright, S.; Ramstedt, M.; et al. Two previously unknown Phytophthora species associated with brown rot of Pomelo (Citrus grandis) fruits in Vietnam. PLoS ONE 2017, 12, e0172085. [Google Scholar] [CrossRef]
- Cacciola, S.O.; Pane, A.; Raudino, F.; Davino, S. First Report of Root and Crown Rot of Sage Caused by Phytophthora cryptogea in Italy. Plant Dis. 2002, 86, 1176. [Google Scholar] [CrossRef]
- Cacciola, S.O.; Chimento, A.; Pane, A.; Cooke, D.E.L.; Magnano di San Lio, G. Root and Foot Rot of Lantana Caused by Phytophthora cryptogea. Plant Dis. 2005, 89, 909. [Google Scholar] [CrossRef]
- Pane, A.; Agosteo, G.E.; Cacciola, S.O. Phytophthora species causing crown and root rot of tomato in southern Italy. EPPO Bull. 2000, 30, 251–255. [Google Scholar] [CrossRef]
- Pane, A.; Faedda, R.; Cacciola, S.O.; Rizza, C.; Scibetta, S.; Magnano di San Lio, G. Root and Basal Stem Rot of Mandevillas Caused by Phytophthora spp. in Eastern Sicily. Plant Dis. 2010, 94, 1374. [Google Scholar] [CrossRef]
- Pane, A.; Faedda, R.; Granata, G.; Puglisi, I.; Aloi, F.; La Spada, F.; Evoli, M.; Stracquadanio, C.; Cacciola, S.O. First Report of Root and Basal Stem Rot Caused by Phytophthora cryptogea and P. inundata on Dwarf Banana in Italy. Plant Dis. 2018, 102, 684. [Google Scholar] [CrossRef]
- Brasier, C.M.; Cooke, D.E.L.; Duncan, J.M.; Hansen, E.M. Multiple new phenotypic taxa from trees and riparian ecosystems in Phytophthora gonapodyides-P. megasperma ITS Clade 6, which tend to be high-temperature tolerant and either inbreeding or sterile. Mycol. Res. 2003, 107, 277–290. [Google Scholar] [CrossRef]
- Huai, W.X.; Tian, G.; Hansen, E.M.; Zhao, W.X.; Goheen, E.M.; Grünwald, N.J.; Cheng, C. Identification of Phytophthora species baited and isolated from forest soil and streams in northwestern Yunnan province, China. Forest Pathol. 2013, 43, 87–103. [Google Scholar] [CrossRef]
- Nechwatal, J.; Bakonyi, J.; Cacciola, S.O.; Cooke, D.E.L.; Jung, T.; Nagy, Z.Á.; Vannini, A.; Vettraino, A.M.; Brasier, C.M. The morphology, behaviour and molecular phylogeny of Phytophthora taxon Salixsoil and its redesignation as Phytophthora lacustris sp. nov. Plant Pathol. 2013, 62, 355–369. [Google Scholar] [CrossRef]
- Copes, W.E.; Yang, X.; Hong, C. Phytophthora Species Recovered From Irrigation Reservoirs in Mississippi and Alabama Nurseries and Pathogenicity of Three New Species. Plant Dis. 2015, 99, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.X.; Gallegly, M.E.; Richardson, P.A.; Kong, P.; Moorman, G.W.; Lea-Cox, J.D.; Ross, D.S. Phytophthora hydropathica, a new pathogen identified from irrigation water, Rhododendron catawbiense and Kalmia latifolia. Plant Pathol. 2010, 59, 913–921. [Google Scholar] [CrossRef]
- Hulvey, J.; Gobena, D.; Finley, L.; Lamour, K. Co-occurrence and genotypic distribution of Phytophthora species recovered from watersheds and plant nurseries of eastern Tennessee. Mycologia 2010, 102, 1127–1133. [Google Scholar] [CrossRef]
- Pintos, C.; Rial, C.; Aguín, O.; Ferreiroa, V.; Mansilla, J.P. First report of Phytophthora hydropathica in river water associated with riparian alder in Spain. New Dis. Rep. 2016, 33, 25. [Google Scholar]
- Vitale, S.; Luongo, L.; Galli, M.; Belisario, A. First Report of Phytophthora hydropathica Causing Wilting and Shoot Dieback on Viburnum in Italy. Plant Dis. 2014, 98, 1582. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, U.; Lundberg, L.; Sonesson, K.; Jung, T. First records of soilborne Phytophthora species in Swedish oak forests. Forest Pathol. 2003, 33, 175–179. [Google Scholar] [CrossRef]
- Jonsson, U.; Jung, T.; Sonesson, K.; Rosengren, U. Relationships between health of Quercus robur, occurrence of Phytophthora species and site conditions in southern Sweden. Plant Pathol. 2005, 54, 502–511. [Google Scholar] [CrossRef]
- Jung, T.; Cooke, D.E.L.; Blaschke, H.; Duncan, J.M.; Oßwald, W. Phytophthora quercina sp. nov., causing root rot of European oaks. Mycol. Res. 1999, 103, 785–798. [Google Scholar] [CrossRef]
- Granata, G.; Pennisi, A.M. Estese morie di platani orientali in forestazioni naturali causate da Ceratocystis fimbriata (Ell. Et. Halst.) Davidson f. platani Walter. Inf. Fitopatol. 1989, 12, 59–61. [Google Scholar]
- Cacciola, S.O.; Pane, A.; Cooke, D.E.L.; Raudino, F.; Magnano di San Lio, G. First Report of Brown Rot and Wilt of Fennel Caused by Phytophthora megasperma in Italy. Plant Dis. 2006, 90, 110. [Google Scholar] [CrossRef]
- Cacciola, S.O.; Scibetta, S.; Pane, A.; Faedda, R.; Rizza, C. Callistemon citrinus and Cistus salvifolius, Two New Hosts of Phytophthora taxon niederhauserii in Italy. Plant Dis. 2009, 93, 1075. [Google Scholar] [CrossRef] [PubMed]
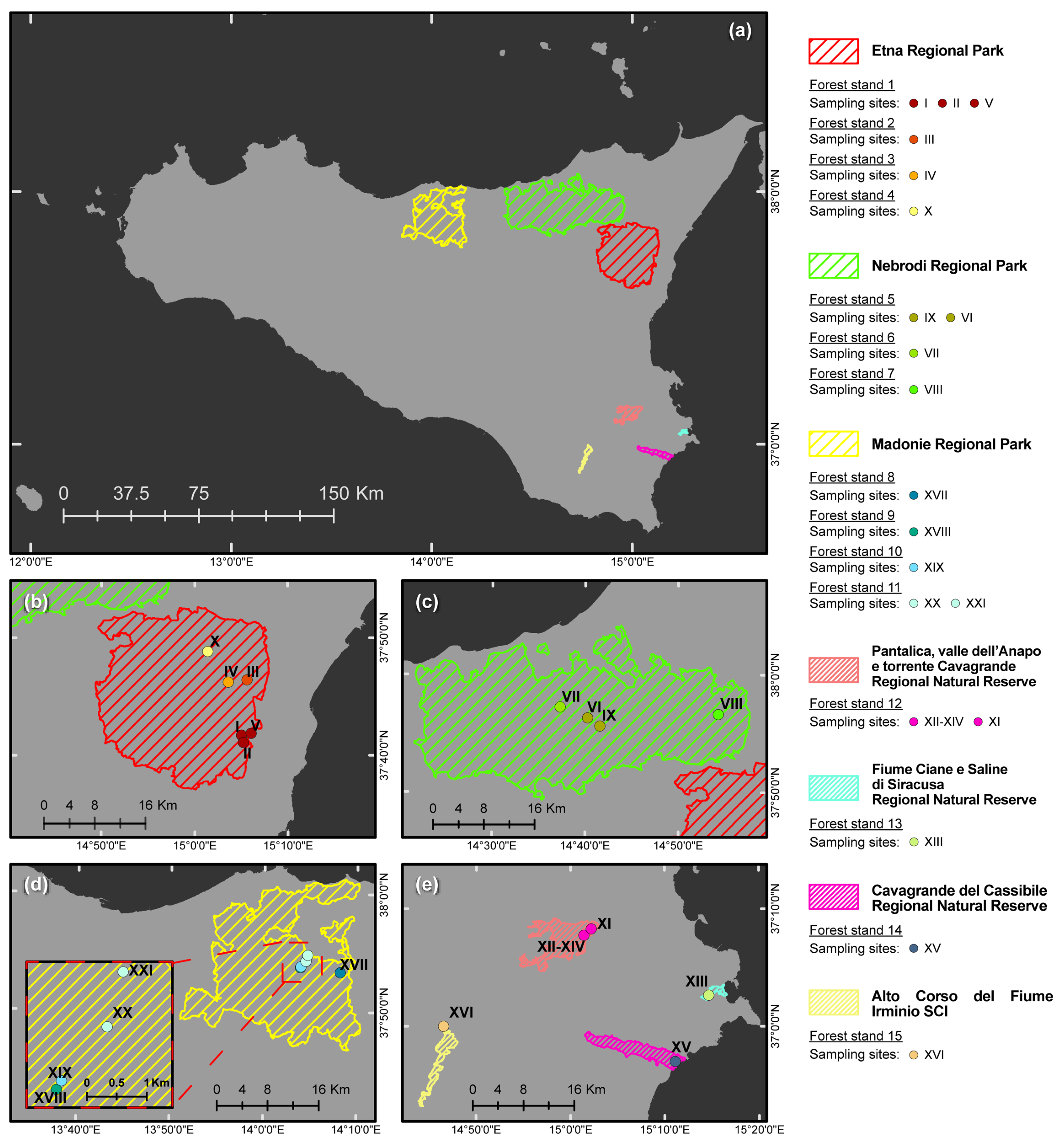
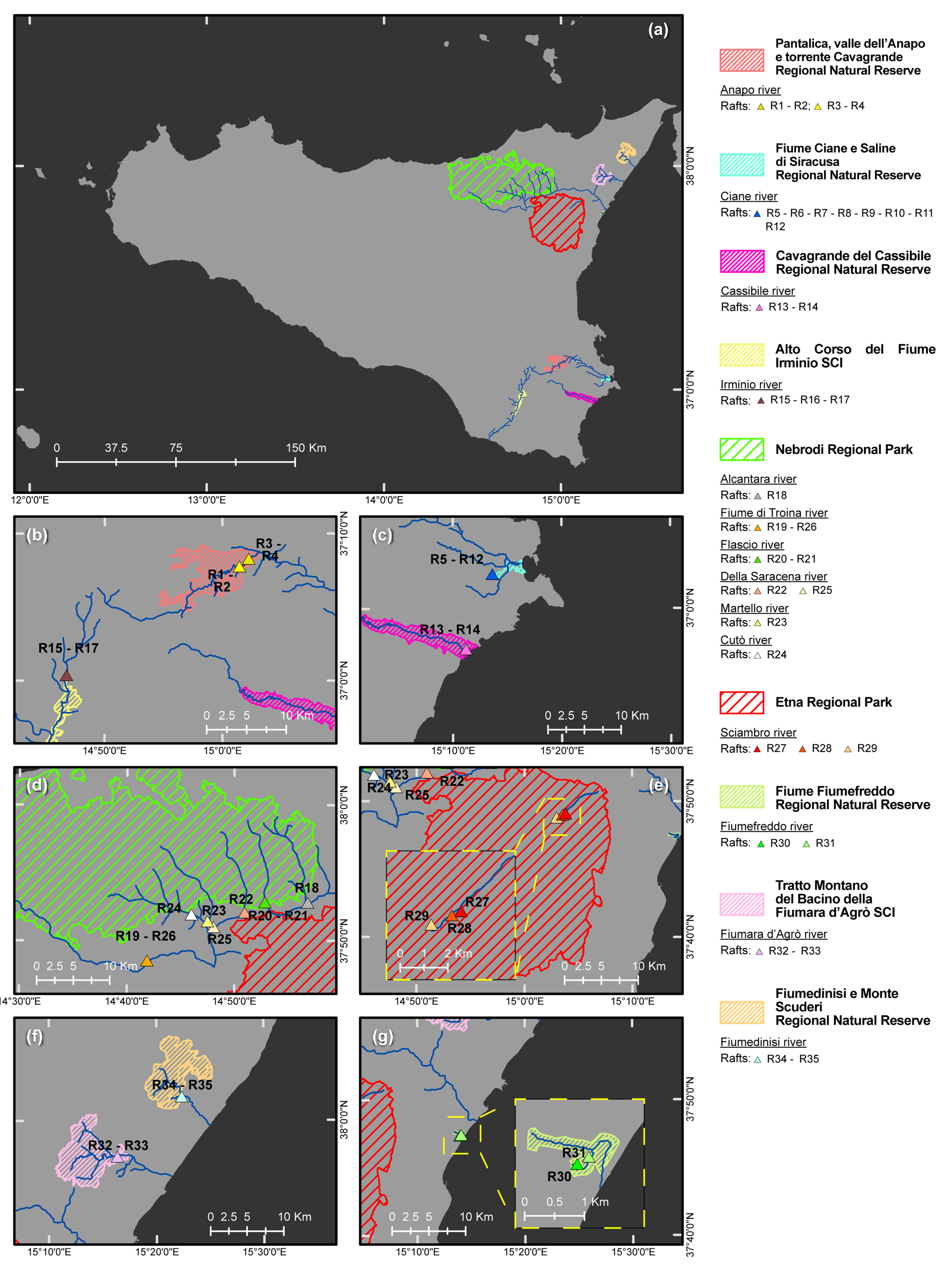

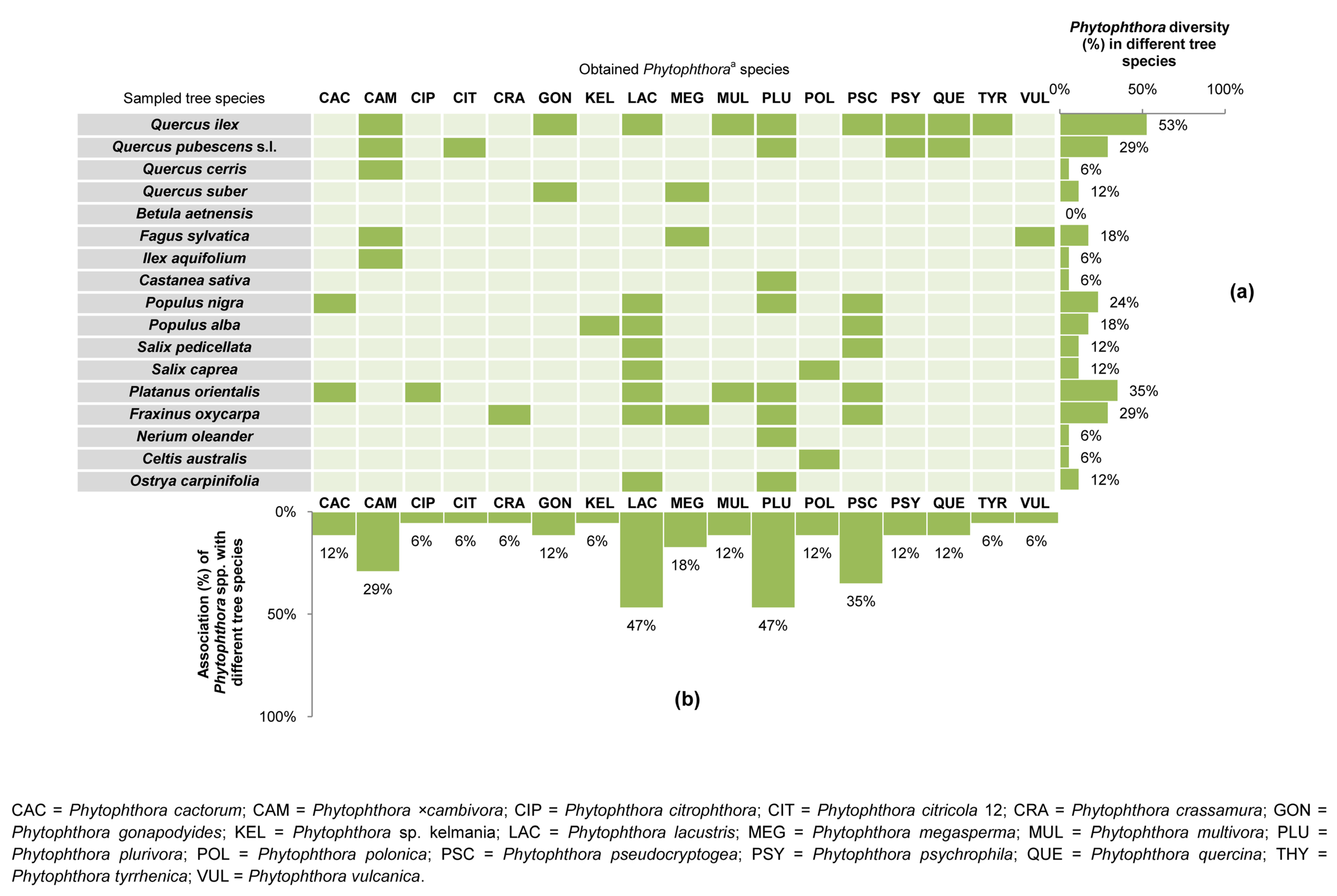
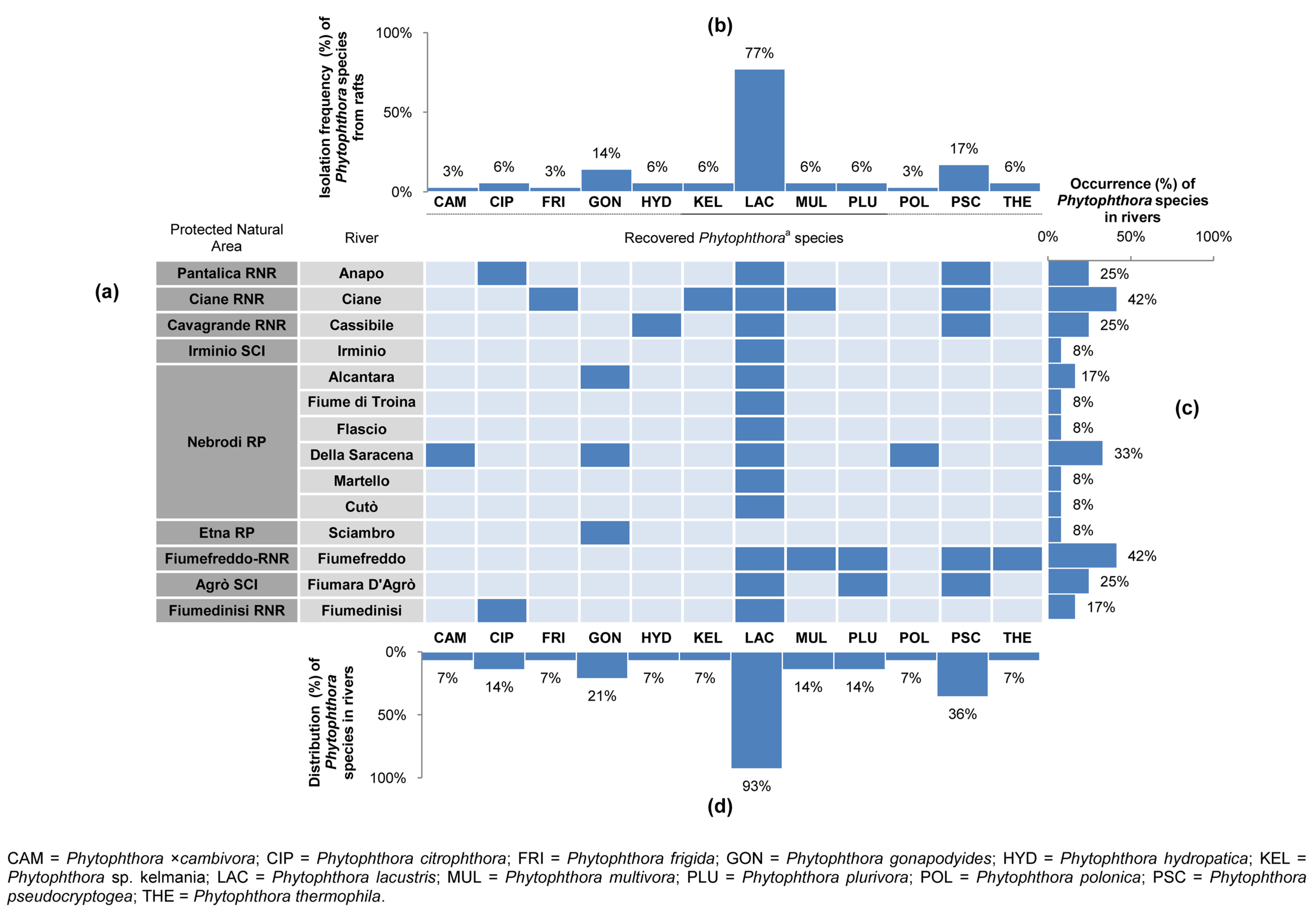
| Forest Stand (FS) No. | Protected Natural Area a | Vegetation (Natura 2000 Code, Forest Stand Type, Phytocoenosis) b,c,d | Geological Substrate | Municipality | Sampling Site No. | Geographic Coordinates (DATUM WGS84) | Altitude (m a.s.l.) | Sampled Tree Species (No. of Phytophthora-Positive Soil Samples/Sampled Trees) | Phytophthora spp. (No. of Positive Soil Samples) f |
|---|---|---|---|---|---|---|---|---|---|
| FS-1 | Etna RP | Natura 2000 CODE: 9340. Forest stand type: Meso-Mediterranean evergreen oak forest. Phytocoenosis: Teucrio siculi-Quercetum ilicis subass. Teucrietosum siculi. | Volcanic (Alkali Basalt-Na) | Zafferana Etnea (CT) | I | 37°41′44.53″ N–15°05′00.04″ E | 1030 | Quercus ilex L. (3/5) | MUL (1); QUE (2) |
| II | 37°41′05.94″ N–15°05′13.04″ E | 890 | Quercus pubescens Willd. s. l. (4/5) | PSY (4) g | |||||
| V | 37°41′53.92″ N–15°06′01.05″ E | 660 | Q. pubescens s. l. (4/5) | CAM (1); QUE (3) | |||||
| FS-2 | Etna RP | Natura 2000 CODE: 91M0. Forest stand type: Supra-Mediterranean turkey oak forest. Phytocoenosis:Vicio cassubicae-Quercetum cerridis. | Volcanic (Alkali Basalt-Na) | Sant’Alfio (CT) | III | 37°46′26.02″ N–15°05′37.23″ E | 1345 | Q. pubescens s. l. (3/6) | PSY (3) h |
| FS-3 | Etna RP | EUNIS CODE: G1.916 e. Forest stand type: Supra-Mediterranean birch forest. Phytocoenosis: Aggregation with Betula aetnensis. | Volcanic (Alkali Basalt-Na) | Sant’Alfio (CT) | IV | 37°46′14.90″ N–15°03′34.56″ E | 1667 | Betula aetnensis Raf. (0/1) | - |
| FS-4 | Etna RP | Natura 2000 CODE: 9220. Forest stand type: Supra-Mediterranean beech forest. Phytocoenosis: Epipactido meridionalis-Fagetum sylvaticae. | Volcanic (Alkali Basalt-Na) | Castiglione di Sicilia (CT) | X | 37°48′50.94″ N–15°01′24.42″ E | 1874 | Fagus sylvatica L. (1/1) | VUL (1) |
| FS-5 | Nebrodi RP | Natura 2000 CODE: 9210. Forest stand type: Supra-Mediterranean beech forest. Phytocoenosis: Anemono apenninae-Fagetum sylvaticae. | Sedimentary–M. Soro Flysh (Marly claystones and limestones, grading upward to quarzarenites) | Militello Rosmarino (ME) | VI | 37°56′22.20″ N–14°40′15.49″ E | 1450 | F. sylvatica (5/7) | CAM (4); MEG (1) i,j |
| Q. pubescens s. l. (1/1) | CAM (1) | ||||||||
| Cersarò (ME) | IX | 37°55′40.90″ N–14°41′35.48″ E | 1783 | F. sylvatica (1/3) | CAM (1) | ||||
| FS-6 | Nebrodi RP | Natura 2000 CODE: 9340. Forest stand type: Meso-Mediterranean evergreen oak forest. Phytocoenosis: Teucrio siculi-Quercetum ilicis. | Sedimentary–M. Soro Flysh (Marly claystones and limestones, grading upward to quarzarenites) | San Fratello (ME) | VII | 37°57′16.38″ N–14°37′18.34″ E | 1050 | Q. ilex (3/5) | CAM (1); GON (2); PSY (1) i |
| FS-7 | Nebrodi RP | Natura 2000 CODE: 91M0. Forest stand type: Meso-Mediterranean turkey oak forest. Phytocoenosis: Arrhenathero nebrodensis-Quercetum cerridis. | Sedimentary–M. Soro Flysh (Marly claystones and limestones, grading upward to quarzarenites) | Randazzo (CT) | VIII | 37°56′40.81″ N–14°54′17.89″ E | 1420 | F. sylvatica (1/1) | CAM (1) i |
| Quercus cerris L. (1/1) | CAM (1) i | ||||||||
| FS-8 | Nebrodi RP | Natura 2000 CODE: 9330. Forest stand type: Meso-Mediterranean cork oak forest. Phytocoenosis: Genisto aristatae-Quercetum suberis. | Sedimentary–Numidian Flysch (quarzarenites and clays) | Geraci Siculo (PA) | XVII | 37°53′22.33″ N–14° 8′10.77″ E | 710 | Quercus suber L. (2/2) | GON (2); MEG (1) |
| FS-9 | Madonie RP | Natura 2000 CODE: 9380. Forest stand type: Supra-Mediterranean holly forest. Phytocoenosis: Ilici aquifoliae-Quercetum austrotyrrhenicae. | Sedimentary–Numidian Flysh (quarzarenites and clays) | Petralia Sottana (PA) | XVIII | 37°53′46.39″ N–14°3′55.22″ E | 1390 | Ilex aquifolium L. (1/1) | CAM (1) |
| FS-10 | Madonie RP | Natura 2000 CODE: 91AA. Forest stand type: Meso-Mediterranean Quercus pubescens forest. Phytocoenosis: Qurcetum leptobalani. | Sedimentary–Numidian Flysh (quarzarenites and claystones) | Castelbuono (PA) | XIX | 37°53′51.02″ N–14°3′58.77″ E | 1412 | Q. pubescens s. l. (1/3) | CAM (1) |
| FS-11 | Madonie RP | Natura 2000 CODE: 9380. Forest stand type: Meso Mediterranean evergreen oak and holly forest. Phytocoenosis: Geranio versicoloris-Quercetum ilicis. | Sedimentary–Numidian Flysh (quarzarenites and claystones) | Castelbuono (PA) | XX | 37°54′20.46″ N–14°4′29.39″ E | 1110 | I. aquifolium (0/3) | - g |
| Q. ilex (2/4) | QUE (1); TYR (1); g | ||||||||
| XXI | 37°54′50.19″ N–14°4′40.07″ E | 850 | Castanea sativa Mill. (1/2) | PLU (1) | |||||
| FS-12 | Pantalica RNR | Natura 2000 CODE: 92C0. Forest stand type: Thermo-Mediterranean riparian plane tree forest. Phytocoenosis: Platano-Salicetum pedicellatae. | Sedimentary (algal calcarenites and calcirudites) | Sortino (SR) | XI | 37°07′48.0″ N–15°01′26.5″ E | 236 | Populusnigra L. (1/1) | PSC (1) |
| Salix pedicellata Desf. (1/1) | PSC (1) | ||||||||
| Q. ilex + Fraxinus oxycarpa Bieb., mixed sample (1/1) | PSC (1); PLU (1); LAC (1) | ||||||||
| Platanus orientalis L. (1/1) | CAC (1); PLU (1) | ||||||||
| Ostrya carpinifolia Scop. (1/1) | LAC (1); PLU (1) | ||||||||
| P. orientalis + Q. ilex, mixed sample (1/1) | LAC (1); PLU (1) | ||||||||
| XII, XIV | 37°08′19.3″ N–15°02′13.3″ E | 221 | P. nigra (1/1) | CAC (1); PLU (1) k | |||||
| Populus alba L. (1/1) | PSC (1); LAC (1); KEL (1) | ||||||||
| S.pedicellata (1/1) | PSC (1); LAC (1) | ||||||||
| Nerium oleander L. (1/1) | PLU (1) | ||||||||
| Celtis australis L. (1/1) | POL (1) | ||||||||
| Q. ilex (1/1) | PSC (1); PLU (1) | ||||||||
| P. orientalis (2/2) | CIP (1); LAC (2) | ||||||||
| FS-13 | Ciane RNR | Natura 2000 CODE: 92A0. Forest stand type: Thermo-Mediterranean riparian willow, poplar, and ash forest. Phytocoenosis: Salicetum albo–pedicellatae. | Alluvial sediments (loam and sandy limestone) | Siracusa (SR) | XIII | 37°02′40.3″ N–15°14′40.7″ E | 4 | F. oxycarpa (4/4) | CRA (1); PSC (3); LAC (2); MEG (1); PLU (2); |
| FS-14 | Cavagrande RNR | Natura 2000 CODE: 92C0. Forest stand type: Thermo-Mediterranean riparian plane tree forest. Phytocoenosis: Platano-Salicetum pedicellatae. | Alluvial sediments (loam and sandy limestone) | Siracusa (SR) | XV | 36°57′2.62″ N–15°11′8.15″ E | 8 | Salix caprea L. (2/2) | LAC (2); POL (1) |
| P. orientalis (3/3) | CAC (1); PSC (3); MUL (2); PLU (1); | ||||||||
| FS-15 | Irminio SCI | Natura 2000 CODE: 92C0. Thermo-Mediterranean riparian plane tree forest. Phytocoenosis: Platano-Salicetum pedicellatae. | Sedimentary (calcarenites and marns) | Ragusa (RG) | XVI | 37°00′1.9″ N–14°46′31.5″ E | 430 | F. oxycarpa (1/1) | PLU (1); PSC (1) |
| Q. pubescens s. l. (2/2) | CIT (1); PLU (2) | ||||||||
| P. orientalis (1/1) | PLU (1) |
| River | Protected Natural Area a | Location of Drainage Basin | Forest Vegetation in Drainage Basin (Natura 2000 Code, Forest Stand Type, Phytocoenosis) b,c,d | Geological Features of Drainage Basin | Raft No. | Municipality | Geographic Coordinates (DATUM WGS84 | Altitude (m a.s.l.) | Phytophthora spp. e |
|---|---|---|---|---|---|---|---|---|---|
| Anapo | Pantalica RNR | Northern area of eastern sector of the Hyblean plateau | Natura 2000 CODE:92C0 Forest stand types: Riparian forests. Phytocoenosis: Platano-Salicetum pedicellatae | Limestone (algal calcarenites and calcirudites) | 1 | Sortino (SR) | 37°07′48.0″ N–15°01′26.5″ E | 294 | LAC |
| 2 | Sortino (SR) | 37°07′48.0″ N–15°01′26.5″ E | 294 | LAC | |||||
| 3 | Sortino (SR) | 37°08′19.3″ N–15°02′13.3″ E | 219 | CIP, LAC, PSC | |||||
| 4 | Sortino (SR) | 37°08′19.3″ N–15°02′13.3″ E | 219 | - | |||||
| Ciane | Ciane RNR | Eastern area of eastern sector of the Hyblean plateau | Natura 2000 CODE: 92A0 Forest stand types: Riparian forests. Phytocoenosis: Salicetum albo-pedicellatae | Alluvial sediments (derived from loam and sandy limestone) | 5 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | KEL, LAC, PSC |
| 6 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | FRI, LAC | |||||
| 7 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | LAC, MUL | |||||
| 8 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | LAC, PSC | |||||
| 9 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | LAC | |||||
| 10 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | LAC | |||||
| 11 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | LAC | |||||
| 12 | Siracusa (SR) | 37°02′34.4″ N–15°13′37.5″ E | 4 | LAC | |||||
| Cassibile | CavagrandeRNR | Eastern area of western sector of the Hyblean plateau | Natura 2000 CODE: 92C0 Forest stand types: Riparian forests. Phytocoenosis: Platano-Salicetum pedicellatae | Limestone (algal calcarenites and calcirudites) | 13 | Siracusa (SR) | 36°57′2.05”N–15°11′11.22”E | 8 | HYD, LAC, PSC |
| 14 | Siracusa (SR) | 36°57′2.05”N–15°11′11.22”E | 8 | HYD, LAC | |||||
| Irminio | Irminio SCI | Northwestern area of western sector of the Hyblean plateau | Natura 2000 CODE: 92C0 Forest stand types: Riparian forests. Phytocoenosis: Platano-Salicetum pedicellatae | Limestone and claystone (calcarenites and marns) | 15 | Ragusa (RG) | 37°00′23.3″ N–14°46′45.1″ E | 400 | - |
| 16 | Ragusa (RG) | 37°00′23.3″ N–14°46′45.1″ E | 400 | - | |||||
| 17 | Ragusa (RG) | 36°57′20.7″ N–14°46′06.2″ E | 300 | LAC f | |||||
| Alcantara | Nebrodi RP | Southeastern area of Nebrodi mountains | Natura 2000 CODE:92A0 Forest stand types: Riparian forests. Phytocoenosis: Salicetum albo-purpurae | Numidian Flysch (quarzarenites and claystones) | 18 | Randazzo (CT) | 37°52′50.4″ N–14°56′49.6″ E | 718 | GON, LAC g |
| Fiume di Troina | Nebrodi RP | Southeastern area of Nebrodi mountains | Natura 2000 CODE:91AA, 91M0, 92A0 Forest stand types: Woodlands and riparian forests. Phytocoenosis: Erico-Quercetum virgilianae; Arrhenathero nebrodensis-Quercetum cerridis; Salicetum albo-purpureae. | Numidian Flysch (quarzarenites and claystones) | 19 | San Teodoro (ME) | 37°48′32.2″ N–14°41′53.1″ E | 605 | LAC h |
| 26 | San Teodoro (ME) | 37°48′32.2″ N–14°41′53.1″ E | 605 | LAC | |||||
| Flascio | Nebrodi RP | Southeastern area of Nebrodi mountains | Natura 2000 CODE:92A0 Forest stand types: Riparian forests Phytocoenosis: Salicetum albo-purpureae | Numidian Flysch (quarzarenites and claystones) | 20 | Randazzo (CT) | 37°52′51.4″ N–14°52′50.6″ E | 856 | LAC h,i |
| 21 | Randazzo (CT) | 37°52′51.4″ N–14°52′50.6″ E | 856 | LAC i | |||||
| Della Saracena | Nebrodi RP | Southeastern area of Nebrodi mountains | Natura 2000 CODE:91AA, 92A0 Forest stand types: Woodlands and riparian forests. Phytocoenosis: Erico-Quercetum virgilianae; Salicetum albo-purpureae. | Numidian Flysch (quarzarenites and claystones) | 22 | Bronte (CT) | 37°52′07.3″ N–14°50′56.2″ E | 811 | CAM, GON, LAC, POL i,j |
| 25 | Maniace (CT) | 37°51′02.6″ N–14°48′04.3″ E | 624 | GON, LAC | |||||
| Martello | Nebrodi RP | Southeastern area of Nebrodi mountains | Natura 2000 CODE:91M0, 92A0 Forest stand types: Woodlands and riparian forests. Phytocoenosis: Arrhenathero nebrodensis-Quercetum cerridis; Salicetum albo-purpurae. | Numidian Flysch (quarzarenites and claystones) | 23 | Maniace (CT) | 37°51′27.7″ N–14°47′29.8″ E | 676 | LAC |
| Cutò | Nebrodi RP | Southern area of Nebrodi mountains | Natura 2000 CODE:92A0 Forest stand types: Riparian forests Phytocoenosis: Salicetum albo-purpurae | Numidian Flysch (quarzarenites and claystones) | 24 | Maniace (CT) | 37°51′57.9″ N–14°46′00.4″ E | 708 | LAC |
| Sciambro | Etna RP | Northeastern area of Volcano Etna | Natura 2000 CODE:9530 Forest stand types: Woodland Phytocoenosis: Junipero hemisphaericae-Pinetum calabricae. | Volcanic (Alcali-Basalt-Na) | 27 | Linguaglossa (CT) | 37°46′58.9″ N–15°3′04.7″ E | 1656 | GON |
| 28 | Linguaglossa (CT) | 37°46′58.4″ N–15°3′02.5″ E | 1669 | - | |||||
| 29 | Linguaglossa (CT) | 37°46′57.0″ N–15°2′01.8″ E | 1682 | GON | |||||
| Fiumefreddo | FiumefreddoRNR | Northeastern boundary of Volcano Etna | Natura 2000 CODE:92A0 Forest stand types: Riparian Forests Phytocoenosis: Populetalia albae The following were also present: (i) citrus groves, (ii) nurseries; (iii) artificial forest of Eucalyptus and Carya cordiformis. | Alluvial sediments (derived from loam, sandy limestone, and volcanic rocks). | 30 | Fiumefreddo di Sicilia (CT) | 37°47′22.15″ N–15°13′55.63″ E | 6 | LAC, MUL, PLU, PSC, THE |
| 31 | Fiumefreddo di Sicilia (CT) | 37°47′25.98″ N–15°14′3.89″ E | 6 | LAC, PSC, THE | |||||
| Fiumara d’Agrò | Agrò SCI | Southeastern area of Peloritani mountains | Natura 2000 CODE:91AA, 91E0 Forest stand types: Woodlands and Riparian Forests. Phytocoenosis: Erico-Quercetum virgilianae; Spartio-Nerietum oleandri. | Metamorphic (Phyllites) | 32 | Limina (ME) | 37°57′22.4″ N–15°16′20.8″ E | 202 | LAC, PLU, PSC |
| 33 | Limina (ME) | 37°57′22.4″ N–15°16′20.8″ E | 202 | - | |||||
| Fiumedinisi | Fiumedinisi RNR | Southeastern area of Peloritani mountains | Natura 2000 CODE:91AA, 92A0; 92C0. Forest stand types: Woodlands and Riparian Forests. Phytocoenosis: Erico-Quercetum virgilianae; Platano-Salicetum gussonei; Salicetum albo-purpuree. | Metamorphic (mainly green shists and amphibolites) | 34 | Fiumedinisi (ME) | 38°01′47.8″ N–15°22′21.3″ E | 214 | - |
| 35 | Fiumedinisi (ME) | 38°01′47.8″ N–15°22′21.3″ E | 214 | CIP, LAC g |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, T.; La Spada, F.; Pane, A.; Aloi, F.; Evoli, M.; Horta Jung, M.; Scanu, B.; Faedda, R.; Rizza, C.; Puglisi, I.; et al. Diversity and Distribution of Phytophthora Species in Protected Natural Areas in Sicily. Forests 2019, 10, 259. https://doi.org/10.3390/f10030259
Jung T, La Spada F, Pane A, Aloi F, Evoli M, Horta Jung M, Scanu B, Faedda R, Rizza C, Puglisi I, et al. Diversity and Distribution of Phytophthora Species in Protected Natural Areas in Sicily. Forests. 2019; 10(3):259. https://doi.org/10.3390/f10030259
Chicago/Turabian StyleJung, Thomas, Federico La Spada, Antonella Pane, Francesco Aloi, Maria Evoli, Marilia Horta Jung, Bruno Scanu, Roberto Faedda, Cinzia Rizza, Ivana Puglisi, and et al. 2019. "Diversity and Distribution of Phytophthora Species in Protected Natural Areas in Sicily" Forests 10, no. 3: 259. https://doi.org/10.3390/f10030259
APA StyleJung, T., La Spada, F., Pane, A., Aloi, F., Evoli, M., Horta Jung, M., Scanu, B., Faedda, R., Rizza, C., Puglisi, I., Magnano di San Lio, G., Schena, L., & Cacciola, S. O. (2019). Diversity and Distribution of Phytophthora Species in Protected Natural Areas in Sicily. Forests, 10(3), 259. https://doi.org/10.3390/f10030259








