On the Shoulders of Giants: Continuing the Legacy of Large-Scale Ecosystem Manipulation Experiments in Puerto Rico
Abstract
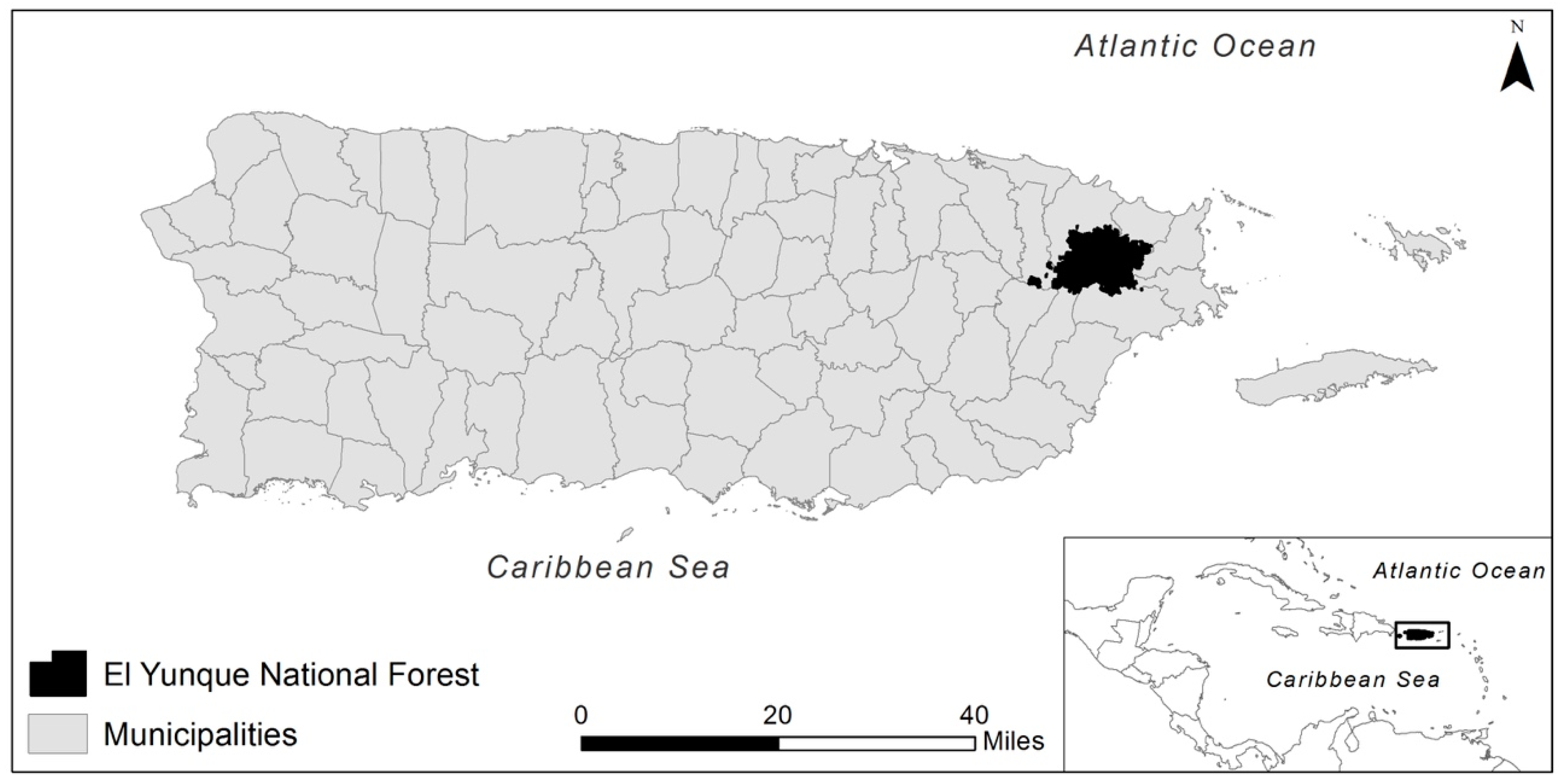
1. Introduction
2. Field Manipulation Experiments in the Luquillo Experimental Forest
2.1. Silviculture (1930s–Today)
2.2. Forest Radiation Experiment (1963–1968)
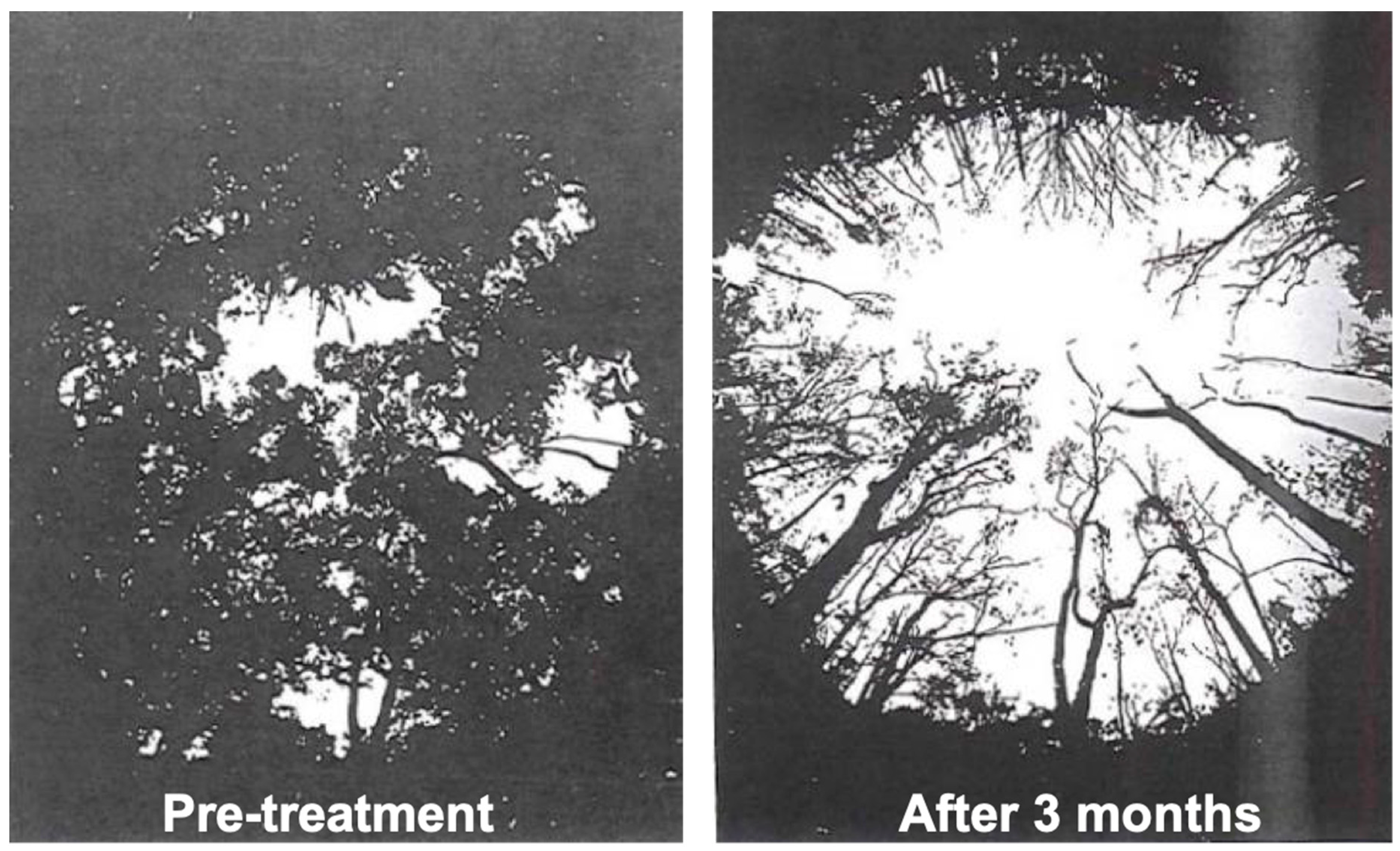
2.3. Rain Forest Herbicide Experiment (1964–1965)
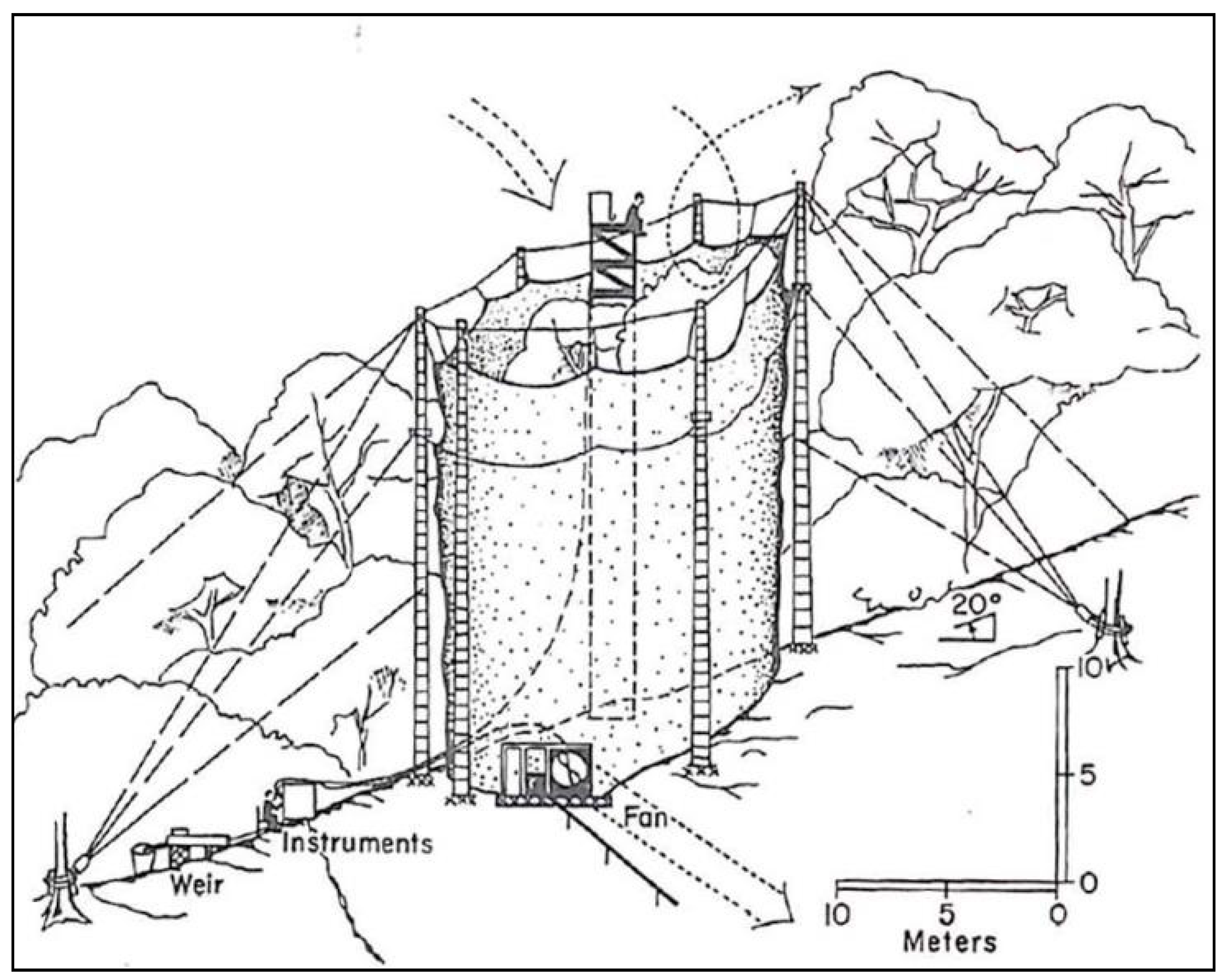
2.4. Giant Plastic Cylinder Study (1966–1967)
2.5. The GAPS Experiment (1988-Today)
2.6. Post-Hurricane Fertilization and Debris-Removal Experiment (1989)
2.7. Canopy Trimming Experiment (2002-Today)
2.8. Long-Term Nitrogen Addition in Two Forest Types (2002–Today)
2.9. Throughfall Exclusion Experiment (2008-Today)
2.10. TRACE: Tropical Responses to Altered Climate Experiment (2013-Today)
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weaver, P.L. The Luquillo Mountains: Forest Resources and Their History; United States Department of Agriculture, Forest Service International Institute of Tropical Forestry: San Juan, PR, USA, 2012.
- Harris, N.L.; Lugo, A.E.; Brown, S.; Heartsill Scalley, T. Luquillo Experimental Forest: Research History and Opportunities; U.S. Department of Agriculture: Washington, DC, USA, 2012; p. 152.
- Brown, S.; Lugo, A.E.; Silander, S.; Liegel, L. Research History and Opportunities in the Luquillo Experimental Forest; General Technical Report (GTR)-SRS-044; U.S. Dept of Agriculture, Forest Service, Southern Forest Experiment Station: New Orleans, LA, USA, 1983; p. 132.
- Quiñones, M.; Parés-Ramos, I.K.; Gould, W.A.; Gonzalez, G.; McGinley, K.; Ríos, P. El Yunque National Forest Atlas; General Technical Report; International Institute of Tropical Forestry: San Juan, PR, USA, 2018.
- Garcia-Martino, A.R.; Warner, G.S.; Scatena, F.N.; Civco, D.L. Rainfall, runoff and elevation relationships in the Luquillo Mountains of Puerto Rico. Caribb. J. Sci. 1996, 32, 413–424. [Google Scholar]
- Aronson, E.L.; McNulty, S.G. Appropriate experimental ecosystem warming methods by ecosystem, objective, and practicality. Agric. For. Meteorol. 2009, 149, 1791–1799. [Google Scholar] [CrossRef]
- Leuzinger, S.; Luo, Y.; Beier, C.; Dieleman, W.; Vicca, S.; K√∂rner, C. Do global change experiments overestimate impacts on terrestrial ecosystems? Trends Ecol. Evol. 2011, 26, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Odum, H.T. Rain forest structure and mineral cycling homeostasis. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Odum, H.T., Pigeon, R.F., Eds.; U.S. Atomic Energy Commission: Oak Ridge, TN, USA, 1970. [Google Scholar]
- Jentsch, A.; Kreyling, J.; Beierkuhnlein, C. A new generation of climate-change experiments: Events, not trends. Front. Ecol. Environ. 2007, 5, 365–374. [Google Scholar] [CrossRef]
- Kimball, B.A.; Alonso-Rodríguez, A.M.; Cavaleri, M.A.; Reed, S.C.; González, G.; Wood, T.E. Infrared heater system for warming tropical forest understory plants and soils. Ecol. Evol. 2018, 8, 1932–1944. [Google Scholar] [CrossRef] [PubMed]
- Newton, I. Isaac Newton Letter to Robert Hooke 1675. Available online: https://digitallibrary.hsp.org/index.php/Detail/objects/9792 (accessed on 27 November 2018).
- Marrero, J. A Survey of the Forest Plantations in the Caribbean National Forest. Master’s Thesis, School of Forestry and Conservation, University of Michigan, Ann Arbor, MI, USA, 1947. [Google Scholar]
- Wadsworth, F. Growth in the lower montane rain forest of Puerto Rico. Caribb. For. 1947, 8, 27–35. [Google Scholar]
- Wadsworth, F.H. Forest management in the Luquillo mountains, III. Selection of products and silvicultural policies. Caribb. For. 1952, 13, 93–142. [Google Scholar]
- Odum, H.T. The Rainforest and Man: An Introduction. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; pp. A-5–A-11. [Google Scholar]
- Odum, H.T.; Jordan, C.F. Metabolism and Evapotranspiration of the Lower Forest in a Giant Plastic Cylinder. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. I-165. [Google Scholar]
- Dowler, C.; Tschirley, F. Evaluation of herbicides applied to foliage of four tropical woody species. J. Agric. Univ. P. R. 1970, 54, 676–682. [Google Scholar]
- Dowler, C.C.; Tschirley, F.H. Effects of Herbicide on a Puerto Rican Rain Forest. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. B-315. [Google Scholar]
- Dowler, C.C.; Forestier, W.; Tschirley, F. Effect and persistence of herbicides applied to soil in Puerto Rican forests. Weed Sci. 1968, 16, 45–50. [Google Scholar]
- Odum, H.T.; Pigeon, R.F. A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde, Puerto Rico; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970.
- Scatena, F.; Larsen, M. Physical aspects of hurricane Hugo in Puerto Rico. Biotropica 1991, 23, 317–323. [Google Scholar] [CrossRef]
- Boose, E.R.; Serrano, M.I.; Foster, D.R. Landscape and regional impacts of hurricanes in Puerto Rico. Ecol. Monogr. 2004, 74, 335–352. [Google Scholar] [CrossRef]
- Beard, K.H.; Vogt, K.A.; Vogt, D.J.; Scatena, F.N.; Covich, A.P.; Sigurdardottir, R.; Siccama, T.G.; Crowl, T.A. Structural and functional responses of a subtropical forest to 10 years of hurricanes and droughts. Ecol. Monogr. 2005, 75, 345–361. [Google Scholar] [CrossRef]
- Scatena, F.; Moya, S.; Estrada, C.; Chinea, J. The first five years in the reorganization of aboveground biomass and nutrient use following Hurricane Hugo in the Bisley Experimental Watersheds, Luquillo Experimental Forest, Puerto Rico. Biotropica 1996, 28, 424–440. [Google Scholar] [CrossRef]
- Comita, L.S.; Uriarte, M.; Thompson, J.; Jonckheere, I.; Canham, C.D.; Zimmerman, J.K. Abiotic and biotic drivers of seedling survival in a hurricane-impacted tropical forest. J. Ecol. 2009, 97, 1346–1359. [Google Scholar] [CrossRef]
- Shiels, A.B.; Zimmerman, J.K.; García-Montiel, D.C.; Jonckheere, I.; Holm, J.; Horton, D.; Brokaw, N. Plant responses to simulated hurricane impacts in a subtropical wet forest, Puerto Rico. J. Ecol. 2010, 98, 659–673. [Google Scholar] [CrossRef]
- Shiels, A.B.; González, G. Understanding the key mechanisms of tropical forest responses to canopy loss and biomass deposition from experimental hurricane effects. For. Ecol. Manag. 2014, 332, 1–10. [Google Scholar] [CrossRef]
- Taylor, C.M.; Silander, S.; Waide, R.B.; Pfeiffer, W.J. Recovery of a tropical forest after gamma irradiation: A 23-year chronicle. In Tropical Forests: Management and Ecology; Springer: New York, NY, USA, 1995; pp. 258–285. ISBN 1-4612-7563-6. [Google Scholar]
- McCormick, J.F. Growth and Survival of the Sierra Palm Under Radiation Stress in Natural and Simulated Environments. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. D-193. [Google Scholar]
- Cusack, D.F.; Torn, M.S.; McDowell, W.H.; Silver, W.L. The response of heterotrophic activity and carbon cycling to nitrogen additions and warming in two tropical soils. Glob. Change Biol. 2010, 16, 2555–2572. [Google Scholar] [CrossRef]
- Wood, T.E.; Silver, W.L. Strong spatial variability in trace gasdynamics following experimental drought in a humid tropical forest. Glob. Biogeochem. Cycles 2012, 26. [Google Scholar] [CrossRef]
- Bouskill, N.J.; Chien Lim, H.; Borglin, S.; Salve, R.; Wood, T.E.; Silver, W.L.; Brodie, E.L. Pre-exposure to drought increases the resistance of tropical forest soil bacterial communities to extended drought. Int. Soc. Microb. Ecol. 2012, 7, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Brokaw, N. A Caribbean Forest Tapestry: The Multidimensional Nature of Disturbance and Response; Oxford University Press: New York, NY, USA, 2012; ISBN 0-19-533469-8. [Google Scholar]
- González, G.; Willig, M.R.; Waide, R.B. Ecological Gradient Analyses in a Tropical Landscape: Multiple Perspectives and Emerging Themes; International Institute of Tropical Forestry: San Juan, PR, USA, 2013; pp. 13–20.
- Buss, H.L.; Gould, W.A.; Larsen, M.C.; Liu, Z.; Martinuzzi, S.; Murphy, S.F.; Stallard, R.F.; Pares-Ramos, I.K.; White, A.F.; Zou, X. Water Quality and Landscape Processes of Four Watersheds in Eastern Puerto Rico; US Geological Survey: Reston, VA, USA, 2012.
- Reagan, D.P.; Waide, R.B. The Food Web of a Tropical Rain Forest; University of Chicago Press: Chicago, IL, USA, 1996; ISBN 0-226-70599-4. [Google Scholar]
- Walker, L.R. Summary of the effects of Caribbean hurricanes on vegetation. Biotropica 1991, 23, 442–447. [Google Scholar]
- Rudel, T.K.; Perez-Lugo, M.; Zichal, H. When fields revert to forest: development and spontaneous reforestation in post-war Puerto Rico. Prof. Geogr. 2000, 52, 386–397. [Google Scholar] [CrossRef]
- Koenig, N. A Comprehensive Agricultural Program for Puerto Rico; United States Department of Agriculture: Washington, DC, USA, 1953.
- Lugo, A.E. Heartsill-Scalley Research in the Luquillo Experimental Forest has advanced understanding of tropical forests and resolved management issues. In USDA Forest Service Experimental Forests and Ranges; Springer: New York, NY, USA, 2014; pp. 435–461. [Google Scholar]
- Crow, T.R. A rainforest chronicle: a 30-year record of change in structure and composition at El Verde, Puerto Rico. Biotropica 1980, 12, 42–55. [Google Scholar] [CrossRef]
- Crow, T.R.; Weaver, P.L. Tree Growth in a Moist Tropical Forest of Puerto Rico; Institute of Tropical Forestry: Río Piedras, PR, USA, 1977.
- Drew, A.P.; Boley, J.D.; Zhao, Y.; Johnston, M.H.; Wadsworth, F.H. Sixty-two years of change in subtropical wet forest structure and composition at El Verde, Puerto Rico. Interciencia 2009, 34, 34. [Google Scholar]
- Heartsill Scalley, T. Insights on Forest Structure and Composition from Long-Term Research in the Luquillo Mountains. Forests 2017, 8, 204. [Google Scholar] [CrossRef]
- Parresol, B.R. Basal area growth for 15 tropical tree species in Puerto Rico. For. Ecol. Manag. 1995, 73, 211–219. [Google Scholar] [CrossRef]
- Shugart, H.H. A Theory of Forest Dynamics. The Ecological Implications of Forest Succession Models; Springer: New York, NY, USA, 1984; ISBN 0-387-96000-7. [Google Scholar]
- Wadsworth, F.H.; Englerth, G.H. Effects of the 1956 hurricane on forests in Puerto Rico. Caribb. For. 1959, 20, 38–51. [Google Scholar]
- Shiels, A.B.; González, G.; Willig, M.R. Responses to canopy loss and debris deposition in a tropical forest ecosystem: Synthesis from an experimental manipulation simulating effects of hurricane disturbance. For. Ecol. Manag. 2014, 332, 124–133. [Google Scholar] [CrossRef]
- Zimmerman, J.K.; Hogan, J.A.; Shiels, A.B.; Bithorn, J.E.; Carmona, S.M.; Brokaw, N. Seven-year responses of trees to experimental hurricane effects in a tropical rainforest, Puerto Rico. For. Ecol. Manag. 2014, 332, 64–74. [Google Scholar] [CrossRef]
- Silver, W.L.; Brown, S.; Lugo, A.E. Effects of changes in biodiversity on ecosystem function in tropical forests. Conserv. Biol. 1996, 10, 17–24. [Google Scholar] [CrossRef]
- Silver, W.L.; Kueppers, L.M.; Lugo, A.E.; Ostertag, R.; Matzek, V. Carbon sequestration and plant community dynamics following reforestation of tropical pasture. Ecol. Appl. 2004, 14, 1115–1127. [Google Scholar] [CrossRef]
- Woodwell, G.M. Effects of ionizing radiation on terrestrial ecosystems. Science 1962, 138, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Odum, H.T.; Murphy, P.; Drewry, G.; McCormick, J.F.; Schinhan, C.; Morales, E.; McIntyre, J.A. Effects of Gamma Radiation on the Forest at El Verde. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. D-3. [Google Scholar]
- Murphy, P.G. Tree Growth at El Verde and the Effects of Ionizing Radiation. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. D-141. [Google Scholar]
- Smith, R.F. The Vegetation Structure of a Puerto Rican Forest Before and After Short-Term Gamma Radiation. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. D-103. [Google Scholar]
- Scatena, F.N. The Death of a Luquillo Giant; Annual Letter; U.S.D.A. Forest Service International Institute of Tropical Forestry: San Juan, PR, USA, 1998.
- McCormick, J.F. Direct and Indirect Effects of Gamma Radiation on Seedling Diversity and Abundance in a Tropical Forest. In A Tropical Rain Forest: A Study of Irradiation and Ecology at El Verde; Atomic Energy Commission, Division of Technical Information: Oak Ridge, TN, USA, 1970; p. D-201. [Google Scholar]
- Stellman, J.M.; Stellman, S.D.; Christian, R.; Weber, T.; Tomasallo, C. The extent and patterns of usage of Agent Orange and other herbicides in Vietnam. Nature 2003, 422, 681. [Google Scholar] [CrossRef] [PubMed]
- Young, A.L. The History of the US Department of Defense Programs for the Testing, Evaluation, and Storage of Tactical Herbicides; Office of the Under Secretary of Defense: Arlington, VA, USA, 2006; pp. 1–81.
- Brown, S.; Lugo, A.E. Tropical secondary forests. J. Trop. Ecol. 1990, 6, 1–32. [Google Scholar] [CrossRef]
- Gómez-Pompa, A.; Vazquez-Yanes, C. Studies on the secondary succession of tropical lowlands: The life cycle of secondary species. In Proceedings of the First International Congress of Ecology: Structure, Functioning and Management of Ecosystems, The Hague, The Netherlands, 8–14 September 1974. [Google Scholar]
- Baccini, A.; Goetz, S.; Walker, W.; Laporte, N.; Sun, M.; Sulla-Menashe, D.; Hackler, J.; Beck, P.; Dubayah, R.; Friedl, M. Estimated carbon dioxide emissions from tropical deforestation improved by carbon-density maps. Nat. Clim. Chang. 2012, 2, 182. [Google Scholar] [CrossRef]
- Giam, X. Global biodiversity loss from tropical deforestation. Proc. Natl. Acad. Sci. 2017, 114, 5775–5777. [Google Scholar] [CrossRef] [PubMed]
- Silver, W.; Ostertag, R.; Lugo, A. The potential for carbon sequestration through reforestation of abandoned tropical agricultural and pasture lands. Restor. Ecol. 2000, 8, 394–407. [Google Scholar] [CrossRef]
- Scatena, F.N.; Silver, W.; Siccama, T.; Johnson, A.; Sanchez, M.J. Biomass and Nutrient Content of the Bisley Experimental Watersheds, Luquillo Experimental Forest, Puerto Rico, Before and After Hurricane Hugo, 1989. Biotropica 1993, 25, 15–27. [Google Scholar] [CrossRef]
- Silver, W.L.; Scatena, F.N.; Johnson, A.H.; Siccama, T.G.; Watt, F. At What Temporal Scales Does Disturbance Affect Belowground Nutrient Pools? Biotropica 1996, 28, 441–457. [Google Scholar] [CrossRef]
- Silver, W.L.; Vogt, K.A. Fine-root dynamics following single and multiple disturbances in a subtropical wet forest ecosystem. J. Ecol. 1993, 81, 729–738. [Google Scholar] [CrossRef]
- Teh, Y.A.; Silver, W.L.; Scatena, F.N. A decade of belowground reorganization following multiple disturbances in a subtropical wet forest. Plant Soil 2009, 323, 197–212. [Google Scholar] [CrossRef]
- Busch, J.; Engelmann, J. Cost-effectiveness of reducing emissions from tropical deforestation, 2016–2050. Environ. Res. Lett. 2017, 13, 015001. [Google Scholar] [CrossRef]
- Strobl, E. The economic growth impact of hurricanes: evidence from US coastal counties. Rev. Econ. Stat. 2011, 93, 575–589. [Google Scholar] [CrossRef]
- Shiels, A.B.; Gonzalez, G.; Lodge, D.J.; Willig, M.R.; Zimmerman, J.K. Cascading effects of canopy opening and debris deposition from a large-scale hurricane experiment in a tropical rain forest. Bioscience 2015, 65, 871–881. [Google Scholar] [CrossRef]
- Zimmerman, J.; Pulliam, W.; Lodge, D.; Quinones-Orfila, V.; Fetcher, N.; Guzman-Grajales, S.; Parrotta, J.; Asbury, C.E.; Walker, L.; Waide, R. Nitrogen immobilization by decomposing woody debris and the recovery of tropical wet forest from hurricane damage. Oikos 1995, 72, 314–322. [Google Scholar] [CrossRef]
- Yang, X.; Warren, M.; Zou, X. Fertilization responses of soil litter fauna and litter quantity, quality, and turnover in low and high elevation forests of Puerto Rico. Appl. Soil Ecol. 2007, 37, 63–71. [Google Scholar] [CrossRef]
- Gonzalez, G.; Li, Y.; Zou, X. Effects of post-hurricane fertilization and debris removal on earthworm abundance and biomass in subtropical forests in Puerto Rico. In Minhocas na America Latina: Biodiversidade e Ecologia; Brown, G.G., Fragoso, C., Eds.; International Institute of Tropical Forestry: San Juan, PR, USA, 2007; pp. 99–108. [Google Scholar]
- Halleck, L.F.; Sharpe, J.M.; Zou, X.Z. Understorey fern responses to post-hurricane fertilization and debris removal in a Puerto Rican rain forest. J. Trop. Ecol. 2004, 20, 173–181. [Google Scholar] [CrossRef]
- Walker, L.R.; Zimmerman, J.K.; Lodge, D.J.; Guzman-Grajales, S. An altitudinal comparison of growth and species composition in hurricane-damaged forests in Puerto Rico. J. Ecol. 1996, 877–889. [Google Scholar] [CrossRef]
- Sanford, R.L., Jr.; Parton, W.J.; Ojima, D.S.; Lodge, D.J. Hurricane effects on soil organic matter dynamics and forest production in the Luquillo Experimental Forest, Puerto Rico: Results of simulation modeling. Biotropica 1991, 23, 364–372. [Google Scholar] [CrossRef]
- Zalamea, M.; González, G.; Ping, C.-L.; Michaelson, G. Soil organic matter dynamics under decaying wood in a subtropical wet forest: effect of tree species and decay stage. Plant Soil 2007, 296, 173–185. [Google Scholar] [CrossRef]
- González, G.; Lodge, D.J.; Richardson, B.A.; Richardson, M.J. A canopy trimming experiment in Puerto Rico: The response of litter decomposition and nutrient release to canopy opening and debris deposition in a subtropical wet forest. For. Ecol. Manag. 2014, 332, 32–46. [Google Scholar] [CrossRef]
- Silver, W.L.; Hall, S.J.; González, G. Differential effects of canopy trimming and litter deposition on litterfall and nutrient dynamics in a wet subtropical forest. For. Ecol. Manag. 2014, 332, 47–55. [Google Scholar] [CrossRef]
- Lodge, D.J.; Cantrell, S.A.; González, G. Effects of canopy opening and debris deposition on fungal connectivity, phosphorus movement between litter cohorts and mass loss. For. Ecol. Manag. 2014, 332, 11–21. [Google Scholar] [CrossRef]
- Liu, X.; Zeng, X.; Zou, X.; Lodge, D.; Stankavich, S.; González, G.; Cantrell, S. Responses of Soil Labile Organic Carbon to a Simulated Hurricane Disturbance in a Tropical Wet Forest. Forests 2018, 9, 420. [Google Scholar] [CrossRef]
- Gutiérrez del Arroyo, O.; Silver, W.L. Disentangling the long-term effects of disturbance on soil biogeochemistry in a wet tropical forest ecosystem. Glob. Chang. Biol. 2018, 24, 1673–1684. [Google Scholar] [CrossRef] [PubMed]
- Matson, P.; Lohse, K.A.; Hall, S.J. The globalization of nitrogen deposition: consequences for terrestrial ecosystems. AMBIO J. Hum. Environ. 2002, 31, 113–119. [Google Scholar] [CrossRef]
- Bobbink, R.; Hicks, K.; Galloway, J.; Spranger, T.; Alkemade, R.; Ashmore, M.; Bustamante, M.; Cinderby, S.; Davidson, E.; Dentener, F. Global assessment of nitrogen deposition effects on terrestrial plant diversity: A synthesis. Ecol. Appl. 2010, 20, 30–59. [Google Scholar] [CrossRef] [PubMed]
- Cusack, D.F.; Silver, W.; McDowell, W.H. Biological nitrogen fixation in two tropical forests: ecosystem-level patterns and effects of nitrogen fertilization. Ecosystems 2009, 12, 1299–1315. [Google Scholar] [CrossRef]
- Wood, T.E.; Detto, M.; Silver, W.L. Sensitivity of soil respiration to variability in soil moisture and temperature in a humid tropical forest. PLoS ONE 2013, 8, e80965. [Google Scholar] [CrossRef] [PubMed]
- Diffenbaugh, N.; Scherer, M. Observational and model evidence of global emergence of permanent, unprecedented heat in the 20th and 21st centuries. Clim. Chang. 2011, 107, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Mora, C.; Frazier, A.G.; Longman, R.J.; Dacks, R.S.; Walton, M.M.; Tong, E.J.; Sanchez, J.J.; Kaiser, L.R.; Stender, Y.O.; Anderson, J.M.; et al. The projected timing of climate departure from recent variability. Nature 2013, 502, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Wood, T.E.; Cavaleri, M.A.; Reed, S.C. Tropical forest carbon balance in a warmer world: a critical review spanning microbial- to ecosystem-scale processes. Biol. Rev. 2012, 87, 912–927. [Google Scholar] [CrossRef] [PubMed]
- Cavaleri, M.A.; Reed, S.C.; Smith, W.K.; Wood, T.E. Urgent need for warming experiments in tropical forests. Glob. Chang. Biol. 2015, 21, 2111–2121. [Google Scholar] [CrossRef] [PubMed]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wood, T.E.; González, G.; Silver, W.L.; Reed, S.C.; Cavaleri, M.A. On the Shoulders of Giants: Continuing the Legacy of Large-Scale Ecosystem Manipulation Experiments in Puerto Rico. Forests 2019, 10, 210. https://doi.org/10.3390/f10030210
Wood TE, González G, Silver WL, Reed SC, Cavaleri MA. On the Shoulders of Giants: Continuing the Legacy of Large-Scale Ecosystem Manipulation Experiments in Puerto Rico. Forests. 2019; 10(3):210. https://doi.org/10.3390/f10030210
Chicago/Turabian StyleWood, Tana E., Grizelle González, Whendee L. Silver, Sasha C. Reed, and Molly A. Cavaleri. 2019. "On the Shoulders of Giants: Continuing the Legacy of Large-Scale Ecosystem Manipulation Experiments in Puerto Rico" Forests 10, no. 3: 210. https://doi.org/10.3390/f10030210
APA StyleWood, T. E., González, G., Silver, W. L., Reed, S. C., & Cavaleri, M. A. (2019). On the Shoulders of Giants: Continuing the Legacy of Large-Scale Ecosystem Manipulation Experiments in Puerto Rico. Forests, 10(3), 210. https://doi.org/10.3390/f10030210





