Damage Diversity as a Metric of Structural Complexity after Forest Wind Disturbance
Abstract
1. Introduction
2. Materials and Methods
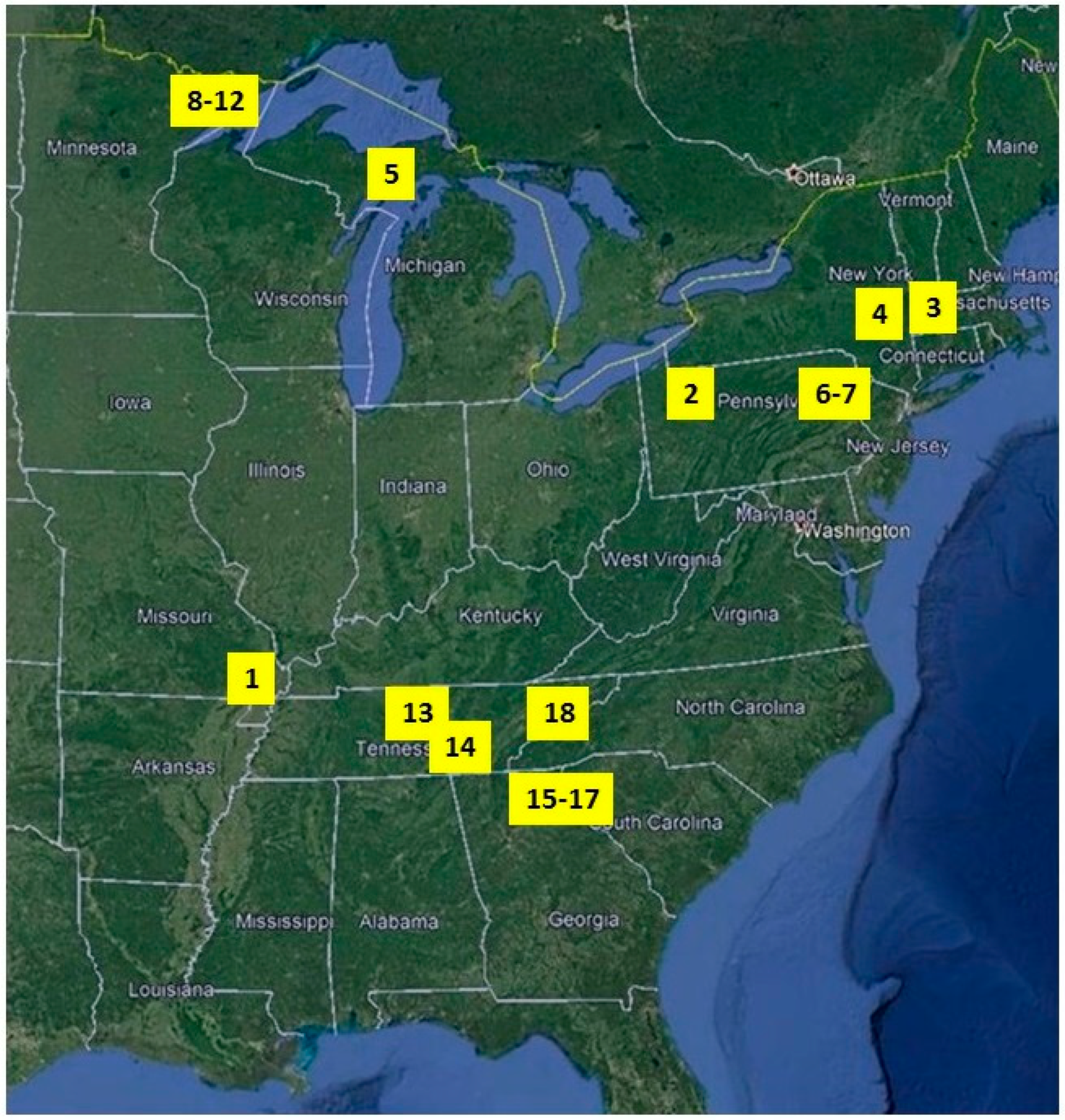
2.1. Study Sites
2.2. Methods
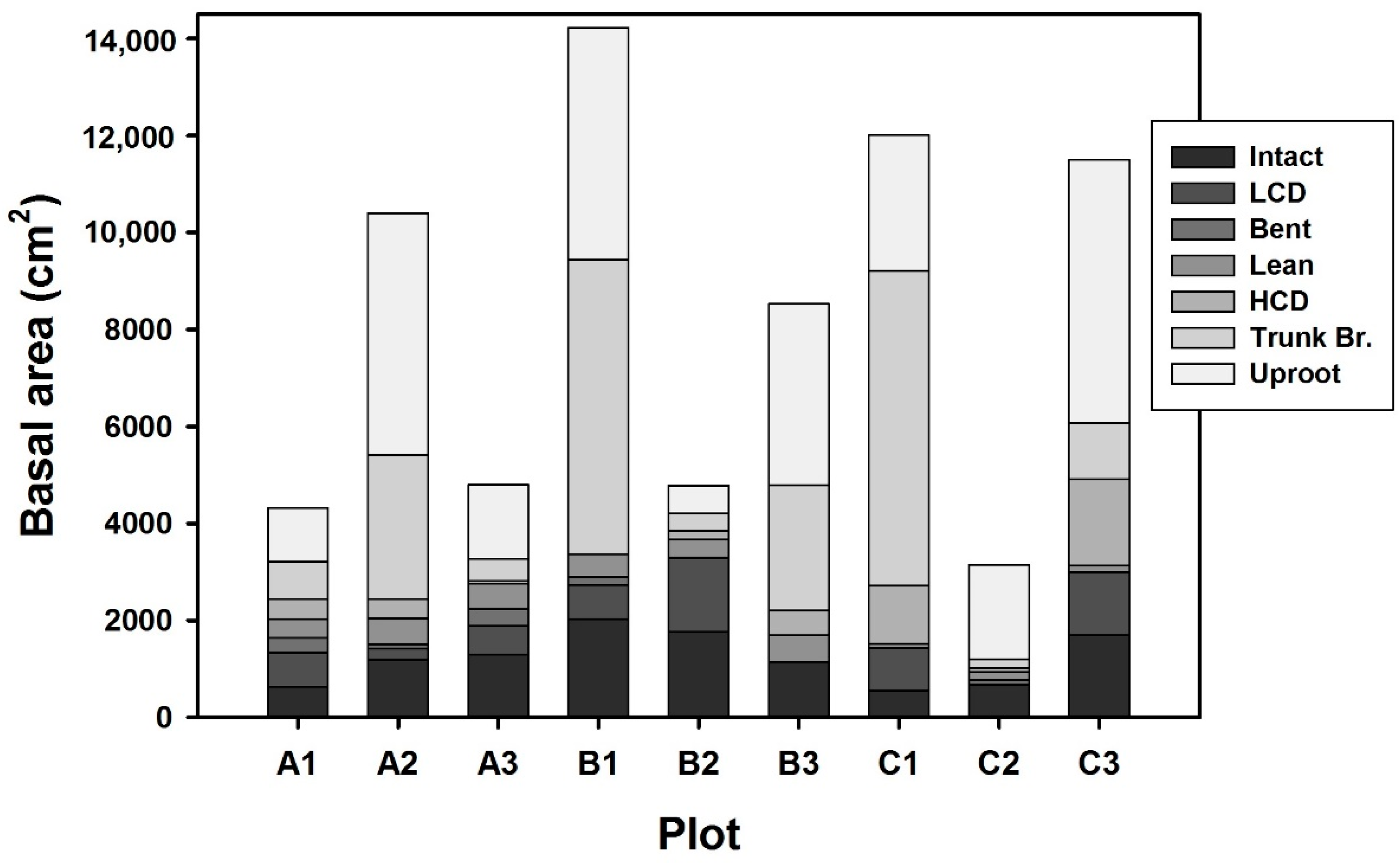
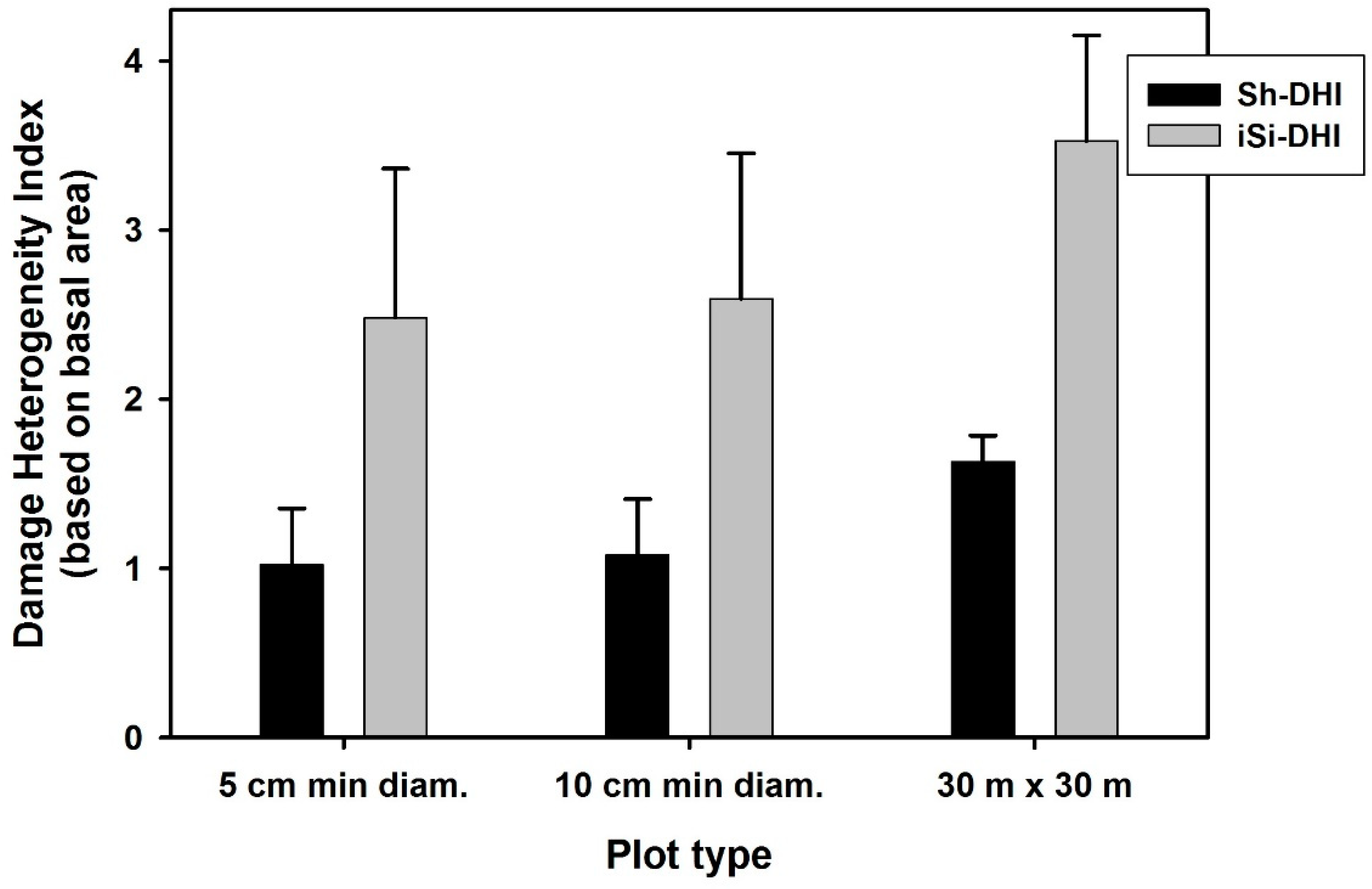
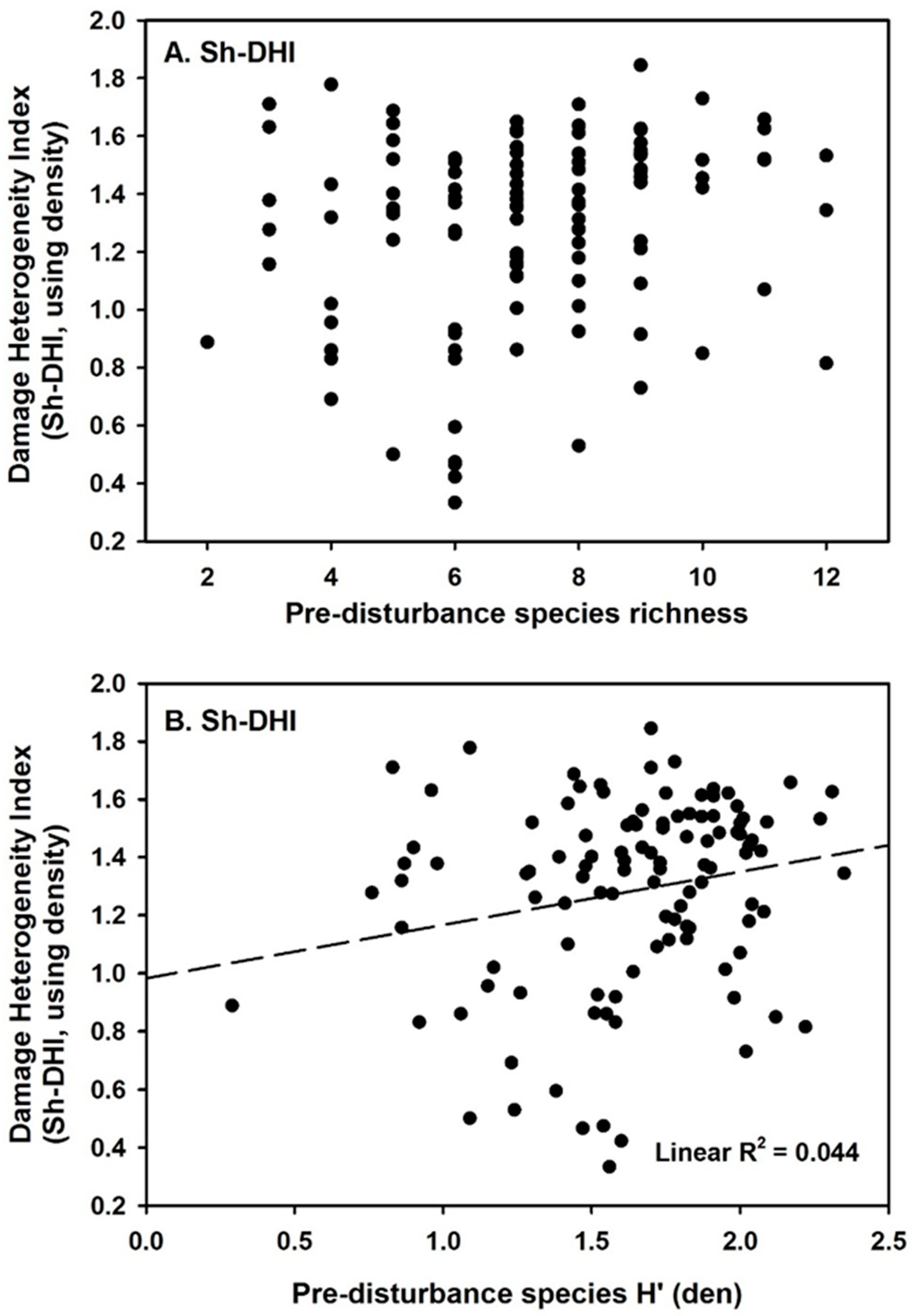
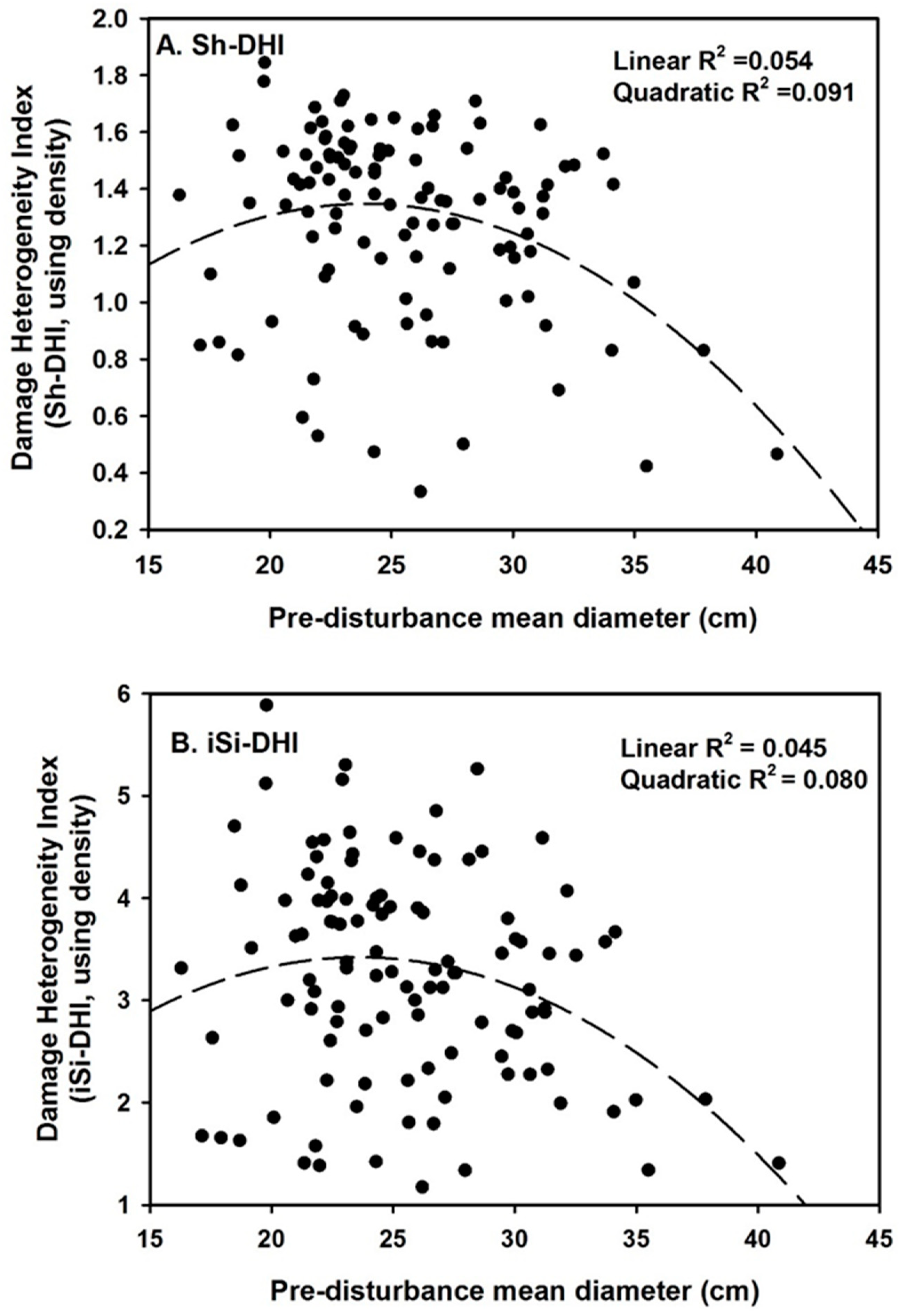
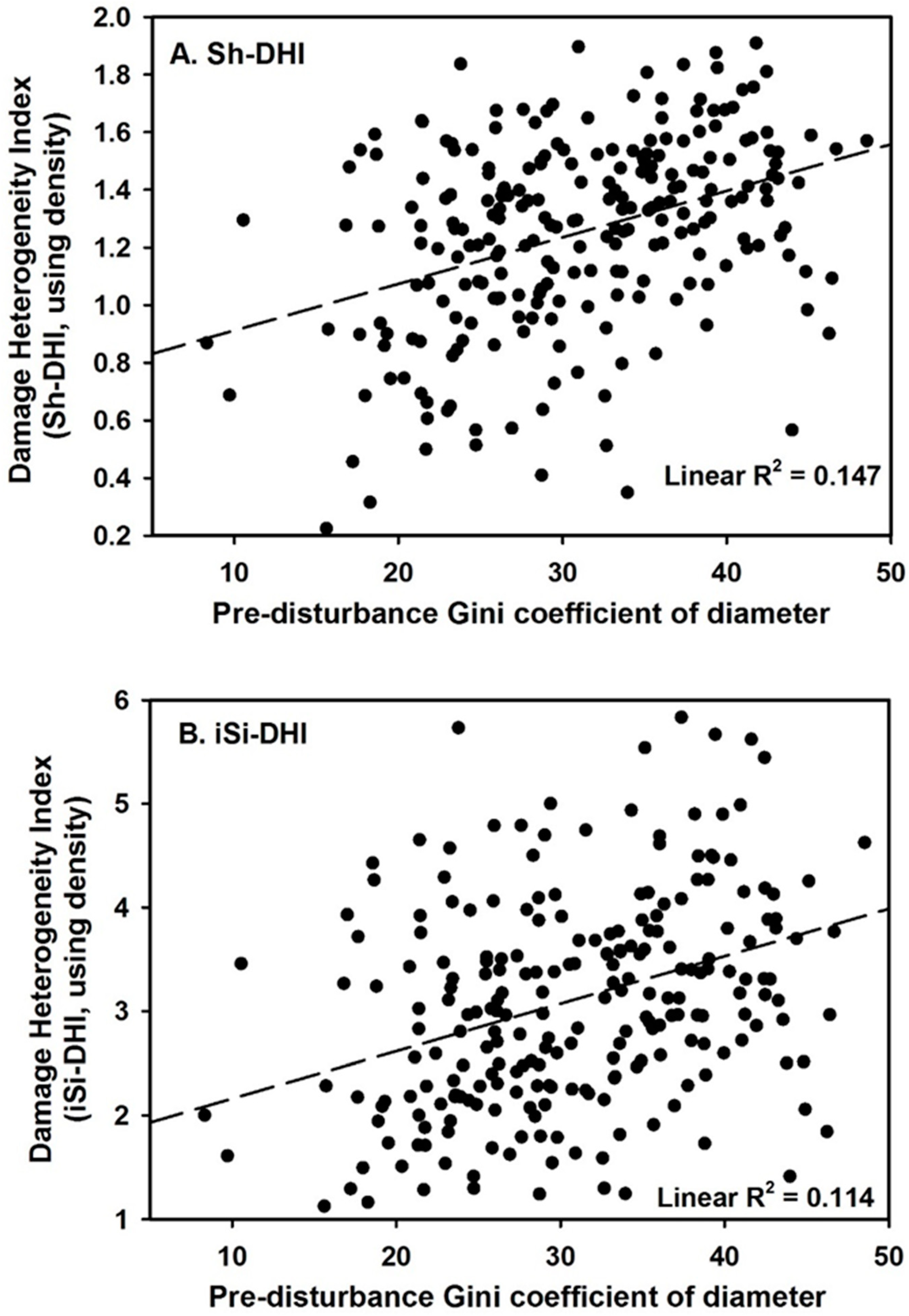
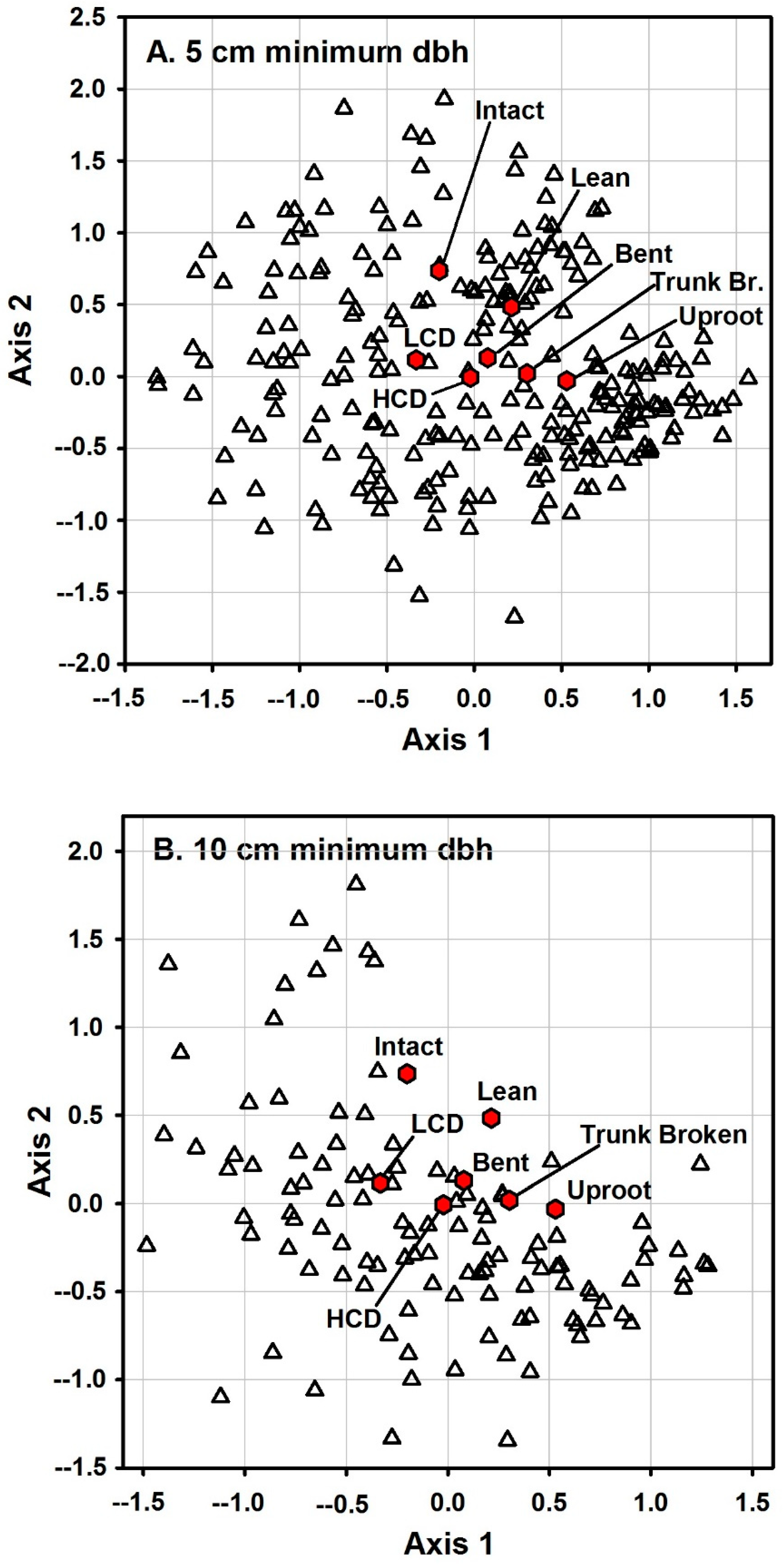
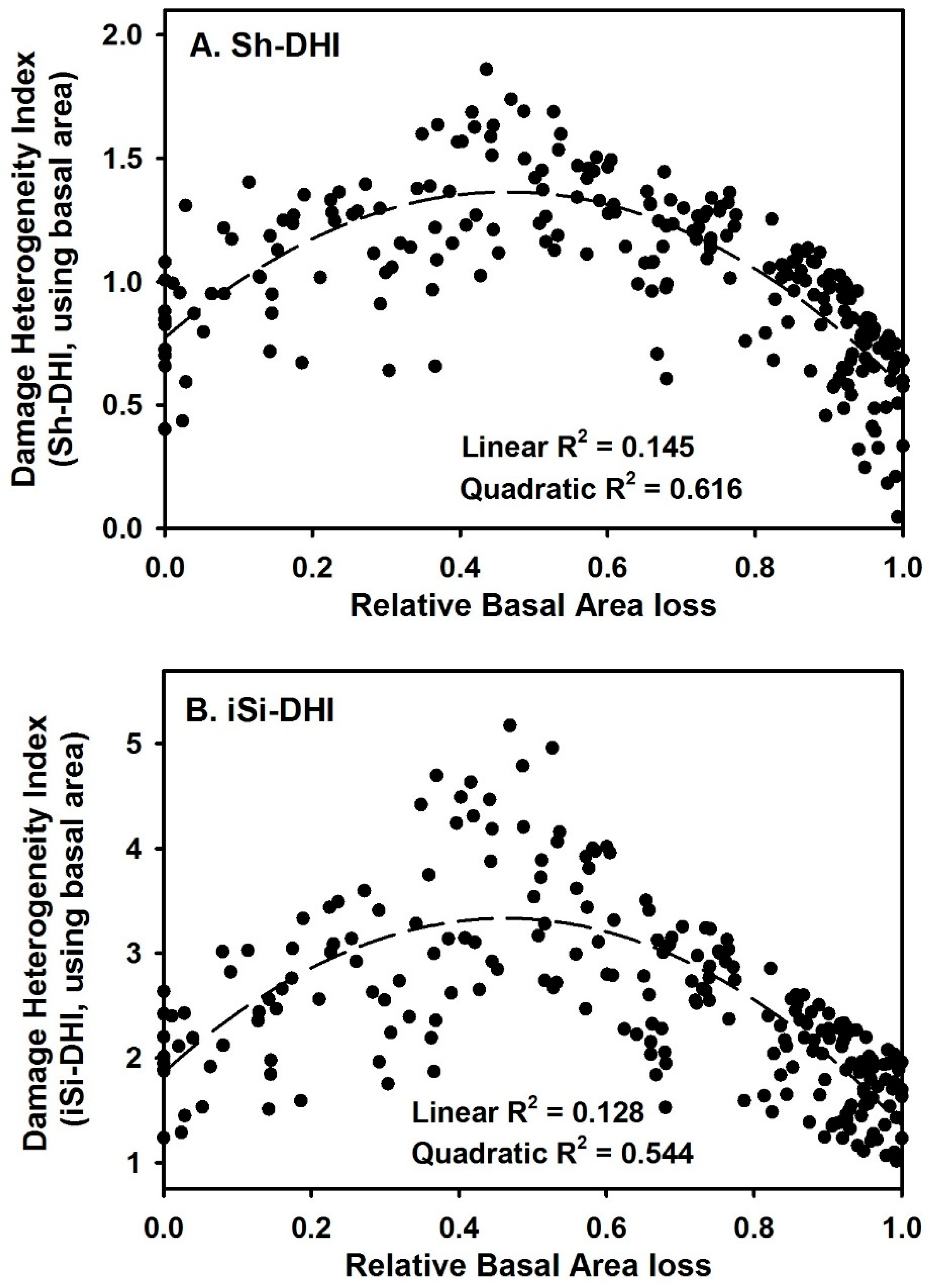
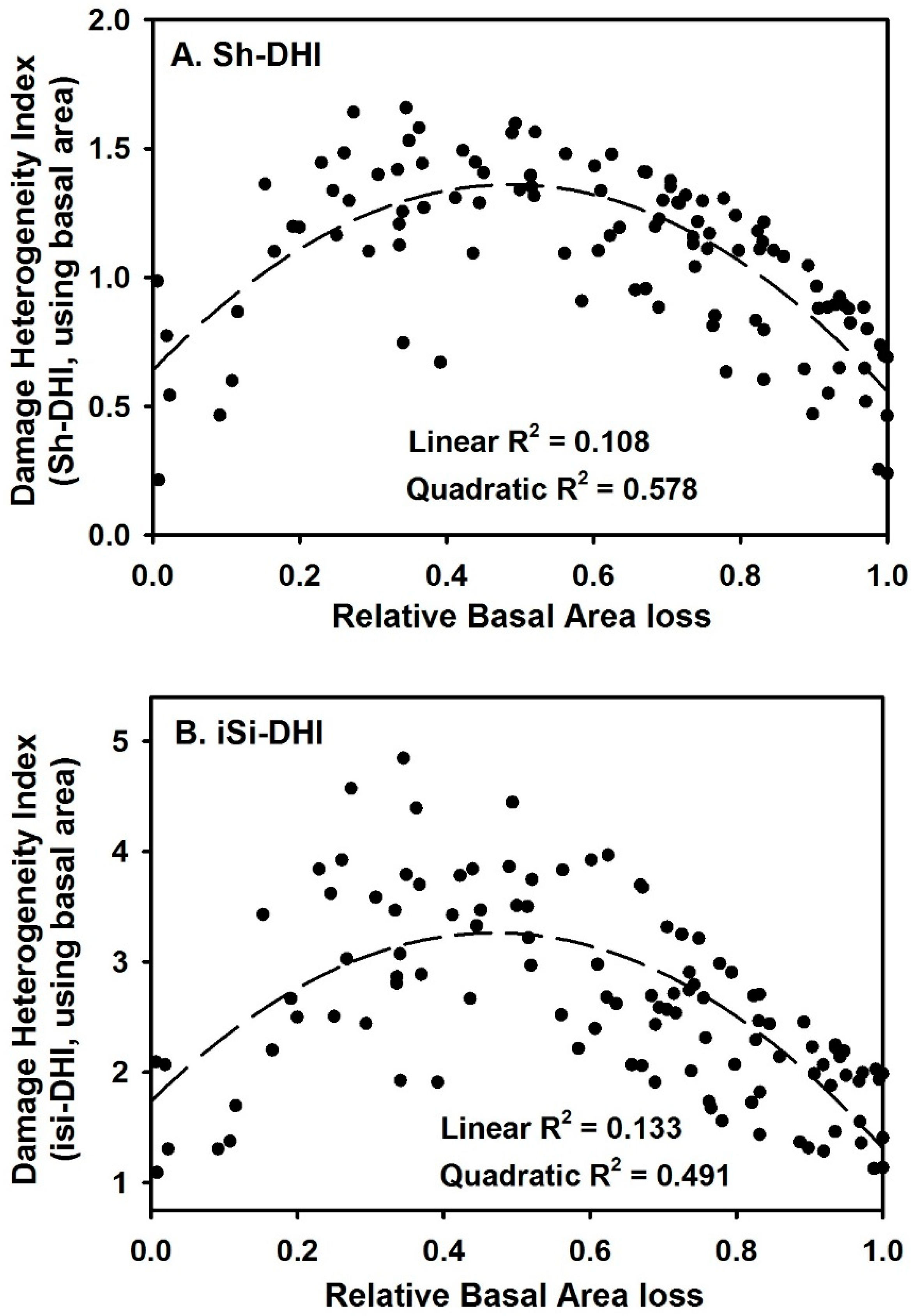
3. Results
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Swanson, M.E.; Franklin, J.F.; Beschta, R.L.; Crisafulli, C.M.; DellaSala, D.A.; Hutto, R.L.; Lindenmayer, D.B.; Swanson, F.J. The forgotten stage of forest succession: Early-successional eocsystems on forest sites. Front. Ecol. Environ. 2011, 9, 117–125. [Google Scholar] [CrossRef]
- D’Amato, A.W.; Bradford, J.B.; Fraver, S.; Palik, B.J. Forest management for mitigation and adaptation to climate change: Insights from long-term silviculture experiments. For. Ecol. Manag. 2011, 262, 803–816. [Google Scholar] [CrossRef]
- Fahey, R.T.; Alveshere, B.C.; Burton, J.I.; D’Amato, A.W.; Dickinson, Y.L.; Keeton, W.S.; Kern, C.C.; Larson, A.J.; Palik, B.J.; Puettmann, K.J.; et al. Shifting conceptions of complexity in forest management and silviculture. For. Ecol. Manag. 2018, 421, 59–71. [Google Scholar] [CrossRef]
- Zenner, E.K. Does old-growth condition imply high live-tree structural complexity? For. Ecol. Manag. 2004, 195, 243–258. [Google Scholar] [CrossRef]
- Lutz, J.A.; Larson, A.J.; Freund, J.A.; Swanson, M.E.; Bible, K.J. The importance of large-diameter trees to forest structural heterogeneity. PLoS ONE 2013, 8, e82784. [Google Scholar] [CrossRef] [PubMed]
- Kinzig, A.P.; Pacala, S.W.; Tilman, D. The Functional Consequences of Biodiversity; Princeton University Press: Princeton, NJ, USA, 2001. [Google Scholar]
- Tilman, D.; Isbell, F.; Cowles, J.M. Biodiversity and ecosystem functioning. Annu. Rev. Ecol. Syst. 2014, 45, 471–493. [Google Scholar] [CrossRef]
- Isbell, F.; Craven, D.; Connolly, J.; Loreau, M.; Schmid, B.; Beierkuhnlein, C.; Bezemer, T.M.; Bonin, C.; Bruelheide, H.; de Luca, E.; et al. Biodiversity increases the resistance fo ecosystem productivity to climate extremes. Nature 2015, 526, 574–577. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, G.E. Homage to Santa Rosalia, or why are there so many kinds of animals? Am. Nat. 1959, 93, 145–159. [Google Scholar] [CrossRef]
- Tilman, D.; Pacala, S. The maintenance of species richness in plant communities. In Species Diversity in Ecological Communities; Ricklefs, R.E., Schluter, D., Eds.; University of Chicago Press: Chicago, IL, USA, 1993; pp. 13–25. [Google Scholar]
- MacArthur, R.H. Population ecology of some warblers of northeastern coniferous forests. Ecology 1958, 39, 599–619. [Google Scholar] [CrossRef]
- Neumann, M.; Starlinger, F. The significance of different indices for stand structure and diversity in forests. For. Ecol. Manag. 2001, 145, 91–106. [Google Scholar] [CrossRef]
- McElhinny, C.; Gibbons, P.; Brack, C.; Bauhus, J. Forest and woodland stand structural complexity: Its definition and measurement. For. Ecol. Manag. 2005, 218, 1–24. [Google Scholar] [CrossRef]
- Danescu, A.; Albrecht, A.T.; Bauhus, J. Structural diversity promotes productivity of mixed, uneven-aged forests in southwestern Germany. Oecologia 2016, 182, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.R.; Knight, D.H.; Franklin, J.F. Landscape patterns and legacies resulting from large, infrequent forest disturbances. Ecosystems 1998, 1, 497–510. [Google Scholar] [CrossRef]
- Franklin, J.F.; Lindenmayer, D.B.; MacMahon, J.A.; McKee, A.; Magnuson, J.; Perry, D.A.; Waide, R.; Foster, D. Threads of continuity: Ecosystem disturbance, recovery, and the theory of biological legacies. Conserv. Pract. 2000, 1, 8–16. [Google Scholar] [CrossRef]
- Keeton, W.S.; Franklin, J.F. Do remnant old-growth trees accelerate rates of succession in mature Douglas-fir forests? Ecol. Monogr. 2005, 75, 103–118. [Google Scholar] [CrossRef]
- Franklin, J.F.; Mitchell, R.J.; Palik, B.J. Natural Disturbance and Stand Development Principles for Ecological Forestry; General Technical Report NRS-19; USDA Forest Service: Newtown Square, PA, USA, 2007.
- Bouget, C.; Duelli, P. The effects of windthrow on forest insect communities: A literature review. Boil. Conserv. 2004, 118, 281–299. [Google Scholar] [CrossRef]
- Janssen, P.; Fortin, D.; Hebert, C. Beetle diversity in a matrix of old-growth boreal forest: Influence of habitat heterogeneity at multiple scales. Ecography 2009, 32, 423–432. [Google Scholar] [CrossRef]
- Goetz, S.J.; Steinberg, D.; Betts, M.G.; Holmes, R.T.; Doran, P.J.; Dubayah, R.; Hofton, M. Lidar remote sensing variables predict breeding habitat of a Neotropical migrant bird. Ecology 2010, 91, 1569–1576. [Google Scholar] [CrossRef]
- Arnan, X.; Bosch, J.; Comas, L.; Gracia, M.; Retana, J. Habitat determinants of abundance, structure and composition of flying Hymenoptera communities in mountain old-growth forests. Insect Conserv. Divers. 2011, 4, 200–211. [Google Scholar] [CrossRef]
- Huth, N.; Possingham, H.P. Basic ecological theory can inform habitat restoration for woodland birds. J. Appl. Ecol. 2011, 48, 293–300. [Google Scholar] [CrossRef]
- Hardiman, B.S.; Gough, C.M.; Halperin, A.; Hofmeister, K.L.; Nave, L.E.; Bohrer, G.; Curtis, P.S. Maintaining high rates of carbon storage in old forests: A mechanism linking canopy structure to forest function. For. Ecol. Manag. 2013, 298, 111–119. [Google Scholar] [CrossRef]
- Gough, C.M.; Curtis, P.S.; Hardiman, B.S.; Scheuermann, C.M.; Bond-Lamberty, B. Disturbance, complexity and succession of net ecosystem production in North America’s temperate deciduous forests. Ecosphere 2016, 7, e01375. [Google Scholar] [CrossRef]
- Wang, W.; Lei, X.; Ma, Z.; Kneeshaw, D.D.; Peng, C. Positive relationship between aboveground carbon stocks and structural diversity in spruce-dominated forest stands in New Brunswick, Canada. For. Sci. 2011, 57, 506–515. [Google Scholar]
- Ford, S.E.; Keeton, W.S. Enhanced carbon storage through management for old-growth characteristics in northern hardwood-conifer forests. Ecosphere 2017, 8, e01721. [Google Scholar] [CrossRef]
- Szmyt, J.; Dobrowolska, D. Spatial diversity of forest regeneration after catastrophic wind in northeastern Poland. iForest 2016, 9, 414–421. [Google Scholar] [CrossRef]
- Fraver, S.; Dodds, K.J.; Kenefic, L.S.; Morrill, R.; Seymour, R.S.; Sypitkowski, E. Forest structure following tornado damage and salvage logging in northern Maine, USA. Can. J. For. Res. 2017, 47, 560–564. [Google Scholar] [CrossRef]
- Meigs, G.W.; Keeton, W.S. Intermediate-severity wind disturbance in mature temperate forests: Legacy structure, carbon storage, and stand dynamics. Ecol. Appl. 2018, 28, 798–815. [Google Scholar] [CrossRef]
- Kuuluvainen, T. Forest management and biodiversity conservation based on natural ecosystem dynamics in northern Europe: The complexity challenge. Ambio 2009, 38, 309–315. [Google Scholar] [CrossRef]
- Palik, B.J.; Mitchell, R.J.; Hiers, J.K. Modelling silviculture after natural disturbance to sustain biodiversity in the longleaf pine (Pinus palustris) ecosystem: balancing complexity and implementation. For. Ecol. Manag. 2002, 155, 347–356. [Google Scholar] [CrossRef]
- Jogiste, K.; Korjus, H.; Stanturf, J.A.; Frelich, L.E.; Baders, E.; Donis, J.; Jansons, A.; Kangur, A.; Koster, K.; Learman, D. Hemiboreal forest: Natural disturbances and the importance of ecosystem legacies to management. Ecosphere 2017, 8, e02503. [Google Scholar] [CrossRef]
- Donato, D.C.; Campbell, J.L.; Franklin, J.F. Multiple successional pathways and precocity in forest development: Can some forests be born complex? J. Veg. Sci. 2012, 23, 576–584. [Google Scholar] [CrossRef]
- Bace, R.; Svoboda, M.; Janda, P.; Morrissey, R.C.; Wild, J.; Clear, J.L.; Cada, V.; Donato, D.C. Legacy of pre-disturbance spatial pattern determines early structural diversity following severe disturbance in montane spruce forests. PLoS ONE 2015, 10, e0139214. [Google Scholar] [CrossRef] [PubMed]
- Franklin, J.F.; Van Pelt, R. Spatial aspects of structural complexity. J. For. 2004, 102, 22–28. [Google Scholar]
- Bachofen, H.; Zingg, A. Effectiveness of structure improvement thinning on stand structure in subalpine Norway spruce (Picea abies (L.) Karst.) stands. For. Ecol. Manag. 2001, 145, 137–149. [Google Scholar] [CrossRef]
- Schneider, E.E.; Larson, A.J. Spatial aspects of structural complexity in Sitka spruce–western hemlock forests, including evaluation of a new canopy gap delineation method. Can. J. For. Res. 2017, 47, 1033–1044. [Google Scholar] [CrossRef]
- Meigs, G.W.; Morrissey, R.C.; Bace, R.; Chaskovskyy, O.; Cada, V.; Despres, T.; Donato, D.C.; Janda, P.; Labusova, J.; Seedre, M.; et al. More ways than one: Mixed-severity disturbance regimes foster structural complexity via multiple developmental pathways. For. Ecol. Manag. 2017, 406, 410–426. [Google Scholar] [CrossRef]
- Peck, J.E.; Zenner, E.K.; Brang, P.; Zingg, A. Tree size distribution and abundance explain structural complexity differentially within stands of even-aged and uneven-aged structure types. Eur. J. For. Res. 2014, 133, 335–346. [Google Scholar] [CrossRef]
- Zenner, E.K.; Peck, J.E. Floating neighborhoods reveal contribution of individual trees to high sub stand scale heterogeneity. For. Ecol. Manag. 2018, 412, 29–40. [Google Scholar] [CrossRef]
- Beatty, S.W. Influence of microtopography and canopy species on spatial patterns of forest understory plants. Ecology 1984, 65, 1406–1419. [Google Scholar] [CrossRef]
- Peterson, C.J.; Carson, W.P.; McCarthy, B.C.; Pickett, S.T.A. Microsite variation and soil dynamics within newly created treefall pits and mounds. Oikos 1990, 58, 39–46. [Google Scholar] [CrossRef]
- Peterson, C.J.; Pickett, S.T.A. Microsite and elevational influences on forest regeneration three years after catastrophic windthrow. J. Veg. Sci. 1990, 1, 657–662. [Google Scholar] [CrossRef]
- Carlton, G.C.; Bazzaz, F.A. Regeneration of three sympatric birch species on experimental hurricane blowdown microsites. Ecol. Monogr. 1998, 68, 99–120. [Google Scholar] [CrossRef]
- Waldron, K.; Ruel, J.-C.; Gauthier, S.; de Grandpre, L.; Peterson, C.J. Effects of post-windthrow salvage logging on microsites, plant composition and regeneration. Appl. Veg. Sci. 2014, 17, 323–334. [Google Scholar] [CrossRef]
- Spicer, J.E.; Suess, K.F.; Wenzel, J.W.; Carson, W.P. Does salvage logging erase a key physical legacy of a tornado blowdown? A case study of tree tip-up mounds. Can. J. For. Res. 2018, 48, 976–982. [Google Scholar] [CrossRef]
- Plotkin, A.B.; Schoonmaker, P.; Leon, B.; Foster, D. Microtopography and ecology of pit-mound structures in second-growth versus old-growth forests. For. Ecol. Manag. 2017, 404, 14–23. [Google Scholar] [CrossRef]
- Sass, E.M.; D’Amato, A.W.; Foster, D.R.; Plotkin, A.B.; Fraver, S.; Schoonmaker, P.K.; Orwig, D.A. Long-term influence of disturbance-generated microsites on forest structural and compositional development. Can. J. For. Res. 2018, 48, 958–965. [Google Scholar] [CrossRef]
- Vodde, F.; Jogiste, K.; Engelhart, J.; Frelich, L.E.; Moser, W.K.; Sims, A.; Metslaid, M. Impact of wind-induced microsites and disturbance severity on tree regeneration patterns: Results from the first post-storm decade. For. Ecol. Manag. 2015, 348, 174–185. [Google Scholar] [CrossRef]
- Simon, A.; Gratzer, G.; Sieghardt, M. The influence of windthrow microsites on tree regeneration and establishment in an old growth mountain forest. For. Ecol. Manag. 2011, 262, 1289–1297. [Google Scholar] [CrossRef]
- Cannon, J.B.; O’Brien, J.J.; Loudermilk, E.L.; Dickinson, M.B.; Peterson, C.J. The influence of experimental wind disturbance on forest fuels and fire characteristics. For. Ecol. Manag. 2014, 330, 294–303. [Google Scholar] [CrossRef]
- Cline, S.P.; Berg, A.B.; Wight, H.M. Snag characteristics and dynamics in Douglas-fir forests, western Oregon. J. Wildl. Manag. 1980, 44, 773–786. [Google Scholar] [CrossRef]
- Tilghman, N.G. Impacts of white-tailed deer on forest regeneration in northwestern Pennsylvania USA. J. Wildl. Manag. 1989, 53, 524–532. [Google Scholar] [CrossRef]
- Ripple, W.J.; Larsen, E.J. The role of postfire coarse woody debris in aspen regeneration. West. J. Appl. For. 2001, 16, 61–64. [Google Scholar]
- De Chantal, M.; Granstrom, A. Aggregations of dead wood after wildfire act as browsing refugia for seedlings of Populus tremula and Salix Caprea. For. Ecol. Manag. 2007, 250, 3–8. [Google Scholar] [CrossRef]
- Relva, M.A.; Westerholm, C.L.; Kitzberger, T. Effects of introduced ungulates on forest understory communities in northern Patagonia are modified by timing and severity of stand mortality. Plant Ecol. 2009, 201, 11–22. [Google Scholar] [CrossRef]
- Cornett, M.W.; Reich, P.B.; Puettmann, K.J.; Frelich, L.E. Seedbed and moisture availability determine safe sites for early Thuja occidentalis (Cupressaceae) regeneration. Am. J. Bot. 2000, 87, 1807–1814. [Google Scholar] [CrossRef] [PubMed]
- Harmon, M.E.; Franklin, J.F.; Swanson, F.J.; Sollins, P.; Gregory, S.V.; Lattin, J.D.; Anderson, N.H.; Cline, S.P.; Aumen, N.G.; Sedell, J.R.; et al. Ecology of coarse woody debris in temperate ecosystems. Adv. Ecol. Res. 1986, 15, 133–302. [Google Scholar]
- Webb, S.L. Disturbance by wind in temperate-zone forests. In Ecosystems of Disturbed Ground; Walker, L.R., Ed.; Elsevier: Amsterdam, The Netherlands, 1999; pp. 187–222. [Google Scholar]
- Cooper-Ellis, S.; Foster, D.R.; Carlton, G.; Lezberg, A. Forest response to catastrophic wind: Results from an experimental hurricane. Ecology 1999, 80, 2683–2696. [Google Scholar] [CrossRef]
- Girard, F.; De Grandpre, L.; Ruel, J.-C. Partial windthrow as a driving process of forest dynamics in old-growth boreal forests. Can. J. For. Res. 2014, 44, 1165–1176. [Google Scholar] [CrossRef]
- Peterson, C.J. Damage and recovery from two different tornadoes in the same old Growth forest: A comparison of infrequent events. For. Ecol. Manag. 2000, 135, 237–252. [Google Scholar] [CrossRef]
- Vandermeer, J.; Mallona, M.A.; Boucher, D.; Yih, K.; Perfecto, I. Three years of ingrowth following catastrophic hurricane damage on the Caribbean coast of Nicaragua: Evidence in support of the direct regeneration hypothesis. J. Trop. Ecol. 1995, 11, 465–471. [Google Scholar] [CrossRef]
- Harcombe, P.A.; Leipzig, L.E.M.; Elsik, I.S. Effects of Hurricane Rita on three long-term forest study plots in east Texas, USA. Wetlands 2009, 29, 88–100. [Google Scholar] [CrossRef]
- Nagel, T.A.; Svoboda, M.; Diaci, J. Regeneration patterns after intermediate wind disturbance in an old-growth Fagus-Abies forest in southeastern Slovenia. For. Ecol. Manag. 2006, 226, 268–278. [Google Scholar] [CrossRef]
- Dodet, M.; Collet, C.; Frochot, H.; Wehrlen, L. Tree regeneration and plant species diversity responses to vegetation control following a major windthrow in mixed broadleaved stands. Eur. J. For. Res. 2011, 130, 41–53. [Google Scholar] [CrossRef]
- Peterson, C.J. Consistent influence of tree diameter and species on damage in nine Eastern North America tornado blowdowns. For. Ecol. Manag. 2007, 250, 96–108. [Google Scholar] [CrossRef]
- Peterson, C.J.; Pickett, S.T.A. Stem damage and resprouting following catastrophic windthrow in an old-growth hemlock-hardwoods forest. For. Ecol. Manag. 1991, 42, 205–217. [Google Scholar] [CrossRef]
- Peterson, C.J.; Pickett, S.T.A. Forest reorganization: A case study in an old-growth forest catastrophic blowdown. Ecology 1995, 76, 763–774. [Google Scholar] [CrossRef]
- Peterson, C.J. Within-stand variation in windthrow in southern-boreal forests of Minnesota: Is it predictable? Can. J. For. Res. 2004, 34, 365–375. [Google Scholar] [CrossRef]
- Peterson, C.J.; Carson, W.P. Generalizing forest regeneration models: The dependence of propagule availability on disturbance history and stand size. Can. J. For. Res. 1996, 26, 45–52. [Google Scholar] [CrossRef]
- Peterson, C.J.; Leach, A.D. Limited salvage logging effects on forest regeneration after moderate-severity windthrow. Ecol. Appl. 2008, 18, 407–420. [Google Scholar] [CrossRef]
- Peterson, C.J.; Rebertus, A.J. Tornado damage and initial recovery in three adjacent, lowland temperate forests in Missouri. J. Veg. Sci. 1997, 8, 559–564. [Google Scholar] [CrossRef]
- Sobhani, V.M.; Barrett, M.J.; Peterson, C.J. Robust prediction of treefall pit and mound sizes from tree size across 10 forest blowdowns in eastern North America. Ecosystems 2014, 17, 837–850. [Google Scholar] [CrossRef]
- Bjorkbom, J.C.; Larson, R.G. The Tionesta Scenic and Research Natural Areas; General Technical Report NE-31; USDA Forest Service: Washington, DC, USA, 1977.
- U.S. Department of Agriculture Forest Service. After the Blowdown: A Resource Assessment of the Boundary Waters Canoe Area Wilderness, 1999-2003; General Technical Report NRS-7; U.S. Department of Agriculture Forest Service: Washington, DC, USA, 2007.
- Pielou, E.C. Measurement of diversity in different types of biological collections. J. Theor. Boil. 1966, 13, 131–135. [Google Scholar] [CrossRef]
- Weiner, J.; Solbrig, O.T. The meaning and measurement of size hierarchies in plant populations. Oecologia 1984, 61, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Stoudhammer, C.L.; LeMay, V.M. Introduction and evaluation of possible indices of stand structural complexity. Can. J. For. Res. 2001, 31, 1105–1115. [Google Scholar] [CrossRef]
- Everham, E.M.; Brokaw, N.V.L. Forest damage and recovery from catastrophic wind. Bot. Rev. 1996, 62, 113–185. [Google Scholar] [CrossRef]
- Canham, C.D.; Papaik, M.J.; Latty, E.F. Interspecific variation in susceptibility to windthrow as a function of tree size and storm severity for northern temperate tree species. Can. J. For. Res. 2001, 31, 1–10. [Google Scholar] [CrossRef]
- Frelich, L.E.; Sugita, S.; Reich, P.B.; Davis, M.B.; Friedman, S.K. Neighborhood effects in forests: Implications for within-stand patch structure. J. Ecol. 1998, 86, 149–161. [Google Scholar] [CrossRef]
- Ulanova, N.G. The effects of windthrow on forests at different spatial scales: A review. For. Ecol. Manag. 2000, 135, 155–167. [Google Scholar] [CrossRef]
- Phillips, J.D.; Marion, D.A.; Turkington, A.V. Pedologic and geomorphic impacts of a tornado blowdown event in a mixed pine-hardwood forest. Catena 2008, 75, 278–287. [Google Scholar] [CrossRef]
- Basnet, K.; Scatena, F.N.; Likens, G.E.; Lugo, A.E. Ecological consequences of root grafting in Tabonuco (Dacryodes excelsa) trees in the Luquillo Experimental Forest, Puerto Rico. Biotropica 1993, 25, 28–35. [Google Scholar] [CrossRef]
- Bauhus, J.; Puettmann, K.; Messier, C. Silviculture for old-growth attributes. For. Ecol. Manag. 2009, 258, 525–537. [Google Scholar] [CrossRef]

| Site | Pre-Dist Species Richness | Pre-Dist. H’ (Density) | Pre-Dist 1/λ (Density) | Pre-Dist. Evenness (Density) | Severity (prop. Basal Area Lost) | Num. Trees | Pre-Dist. Diam. (cm) |
|---|---|---|---|---|---|---|---|
| Mingo *a,5 | 7.67 ± 2.34 | 1.56 ± 0.28 | 3.72 ± 1.12 | 0.78 ± 0.09 | 0.58 ± 0.23 | 192 | 17.08 ± 3.48 |
| Mingo *b,5 | 8.67 ± 1.15 | 1.80 ± 0.14 | 4.84 ± 0.88 | 0.83 ± 0.03 | 0.75 ± 0.23 | 92 | 28.58 ± 3.59 |
| Tionesta 1994 a,5 | 3.48 ± 1.46 | 1.04 ± 0.31 | 2.55 ± 0.75 | 0.82 ± 0.12 | 0.30 ± 0.28 | 643 | 27.00 ± 8.09 |
| Gould Farm a,5 | 7.50 ± 1.84 | 1.54 ± 0.30 | 3.80 ± 1.47 | 0.77 ± 0.11 | 0.85 ± 0.19 | 360 | 22.02 ± 3.21 |
| Texas Hill a,5 | 5.60 ± 1.84 | 1.23 ± 0.37 | 2.78 ± 1.07 | 0.72 ± 0.12 | 0.97 ± 0.04 | 255 | 21.84 ± 2.23 |
| Rapid River a,5 | 6.09 ± 1.97 | 1.27 ± 0.30 | 2.91 ± 0.74 | 0.72 ± 0.10 | 0.28 ± 0.21 | 573 | 15.99 ± 0.86 |
| Taylor a,5 | 4.96 ± 2.23 | 1.36 ± 0.37 | 3.46 ± 1.20 | 0.86 ± 0.09 | 0.80 ± 0.25 | 1224 | 23.73 ± 5.41 |
| Hattons a,5 | 3.88 ± 0.64 | 0.90 ± 0.21 | 2.04 ± 0.58 | 0.67 ± 0.12 | 0.73 ± 0.27 | 158 | 22.57 ± 4.61 |
| Fishhook a,5 | 3.31 ± 0.79 | 0.79 ± 0.22 | 1.93 ± 0.39 | 0.67 ± 0.16 | 0.47 ± 0.16 | 611 | 15.35 ± 1.92 |
| Med. Lake a,5 | 4.44 ± 0.73 | 0.85 ± 0.21 | 1.85 ± 0.40 | 0.58 ± 0.13 | 0.63 ± 0.14 | 554 | 11.01 ± 2.83 |
| Shirttail a,5 | 5.17 ± 1.04 | 1.32 ± 0.26 | 3.28 ± 0.82 | 0.81 ± 0.11 | 0.86 ± 0.16 | 548 | 16.26 ± 2.61 |
| Twin a,5 | 5.35 ± 1.27 | 1.17 ± 0.22 | 2.64 ± 0.73 | 0.72 ± 0.12 | 0.83 ± 0.14 | 878 | 15.19 ± 3.94 |
| ThreeMile a,10 | 3.86 ± 1.39 | 1.00 ± 0.35 | 2.48 ± 0.84 | 0.77 ± 0.14 | 0.29 ± 0.24 | 404 | 22.40 ± 3.89 |
| Natchez Trace b,5 | 12.07 ± 2.38 | 2.02 ± 0.27 | 5.87 ± 1.75 | 0.82 ± 0.06 | 0.38 ± 0.20 | 1637 | 19.07 ± 2.56 |
| Gum Road a,5 | 7.89 ± 2.21 | 1.86 ± 0.32 | 5.92 ± 1.87 | 0.91 ± 0.04 | 0.33 ± 0.26 | 674 | 23.24 ± 3.23 |
| Boggs Creek a,10 | 8.24 ± 2.07 | 1.79 ± 0.32 | 5.12 ± 1.77 | 0.86 ± 0.08 | 0.66 ± 0.29 | 894 | 25.71 ± 5.29 |
| Smokies a,10 | 7.14 ± 1.96 | 1.66 ± 0.31 | 4.66 ± 1.74 | 0.86 ± 0.09 | 0.58 ± 0.24 | 487 | 25.60 ± 3.03 |
| Martin Branch a,10 | 7.28 ± 1.32 | 1.73 ± 0.17 | 4.87 ± 0.96 | 0.88 ± 0.06 | 0.74 ± 0.24 | 383 | 26.72 ± 5.49 |
| Timpson a,10 | 7.71 ± 1.07 | 1.77 ± 0.18 | 4.91 ± 1.20 | 0.87 ± 0.06 | 0.72 ± 0.16 | 274 | 27.47 ± 3.17 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peterson, C.J. Damage Diversity as a Metric of Structural Complexity after Forest Wind Disturbance. Forests 2019, 10, 85. https://doi.org/10.3390/f10020085
Peterson CJ. Damage Diversity as a Metric of Structural Complexity after Forest Wind Disturbance. Forests. 2019; 10(2):85. https://doi.org/10.3390/f10020085
Chicago/Turabian StylePeterson, Chris J. 2019. "Damage Diversity as a Metric of Structural Complexity after Forest Wind Disturbance" Forests 10, no. 2: 85. https://doi.org/10.3390/f10020085
APA StylePeterson, C. J. (2019). Damage Diversity as a Metric of Structural Complexity after Forest Wind Disturbance. Forests, 10(2), 85. https://doi.org/10.3390/f10020085





