Climate Change Impacts on Pinus pinea L. Silvicultural System for Cone Production and Ways to Contour Those Impacts: A Review Complemented with Data from Permanent Plots
Abstract
:1. Introduction
- Cones represent an annual income for forest owners with increasing importance;
- The substitution of maritime pine as a consequence of high mortality of this species caused by the nematode (Bursaphelenchus xylophilus (Steiner et Buhrer) Nickle); and
- The plantation of umbrella pine inter-mixed within cork oak sparse stands.
- Cone production from plots established in stands whose trees were subject to numerous levels of competition and
- A wide cone production range in number and weight at tree and stand level. At some situations those were much higher than the ones presented for other countries. These differences may be explained by the favorable site conditions in the country and/or stand management practices.
2. Umbrella Pine Ecology for Cone Production
3. Umbrella Pine Nut Production and Productivity in Mediterranean Countries
4. Analysis of Portuguese Silvicultural Systems for Pine Nut Production Using Permanent Plot Data
4.1. Traditional Umbrella Pine Silvicultural Systems for Cone Production
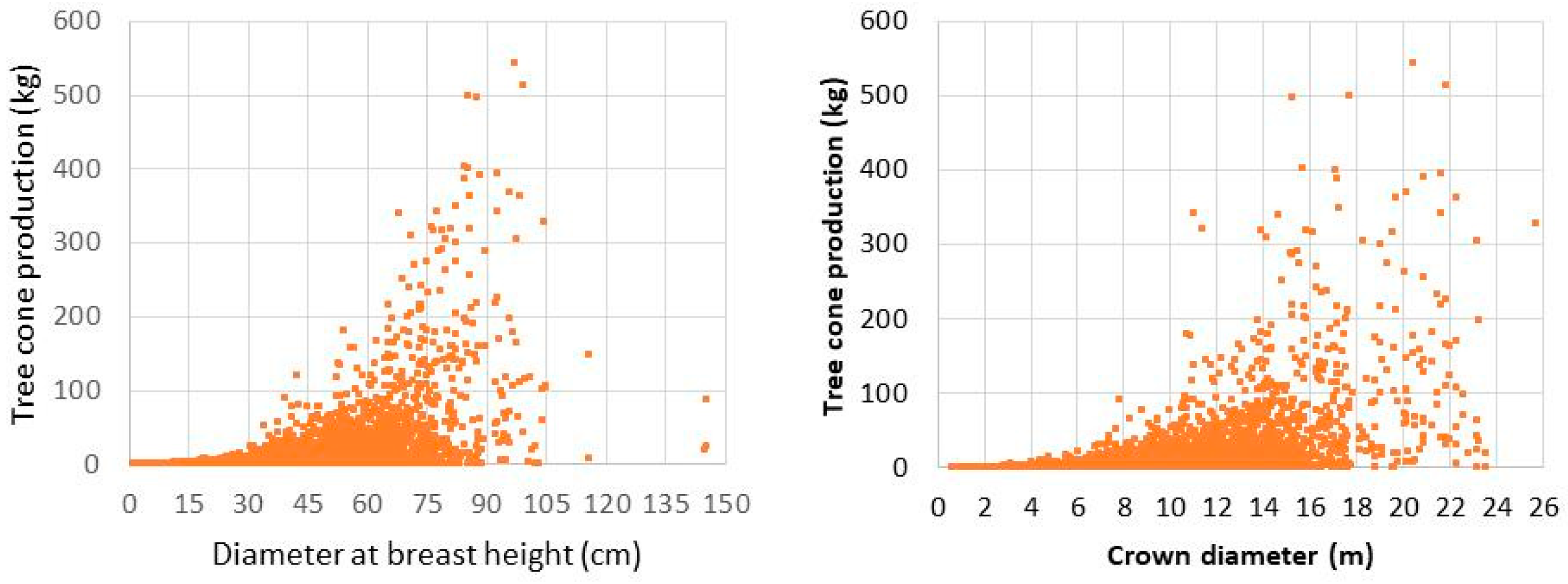
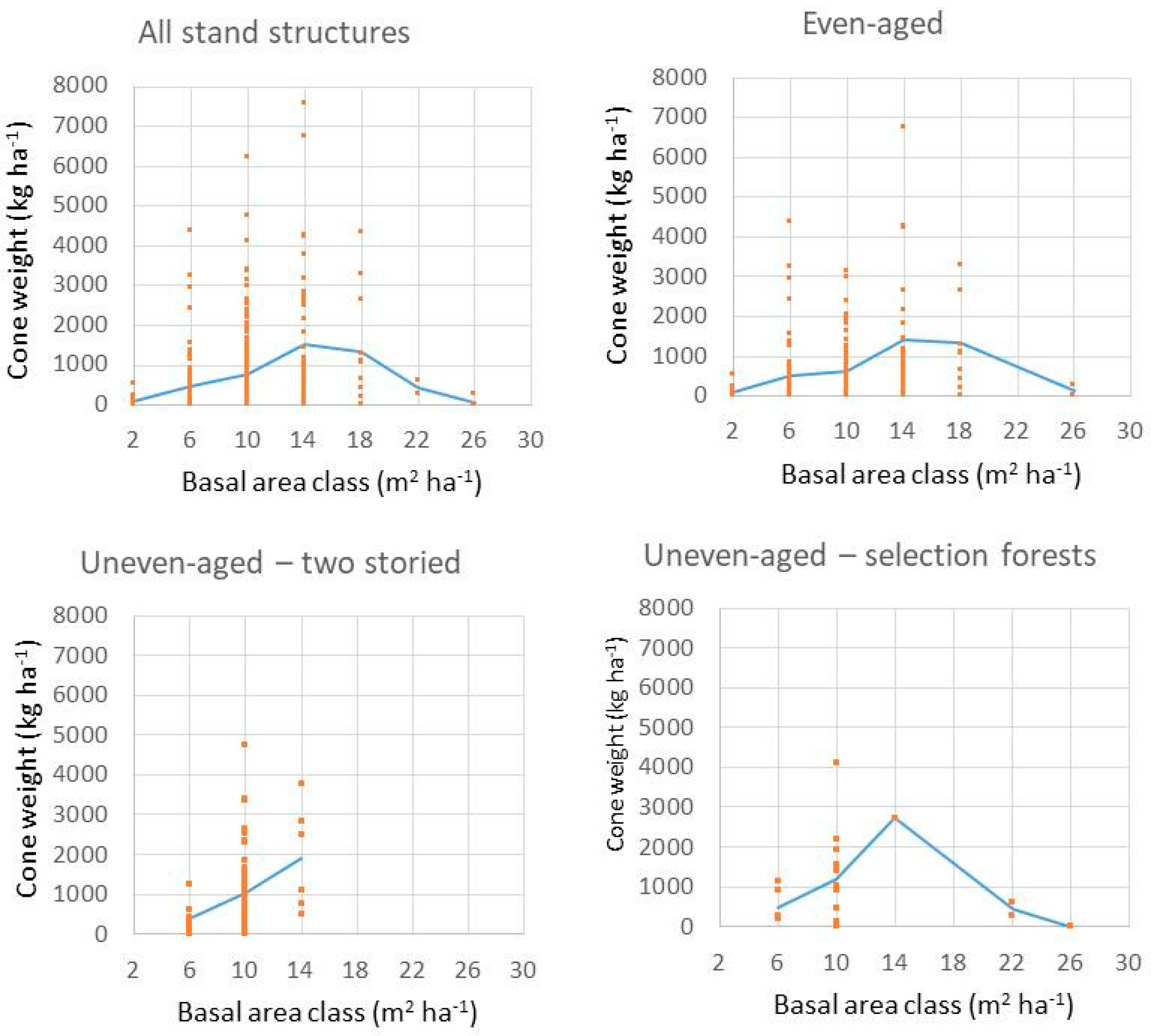
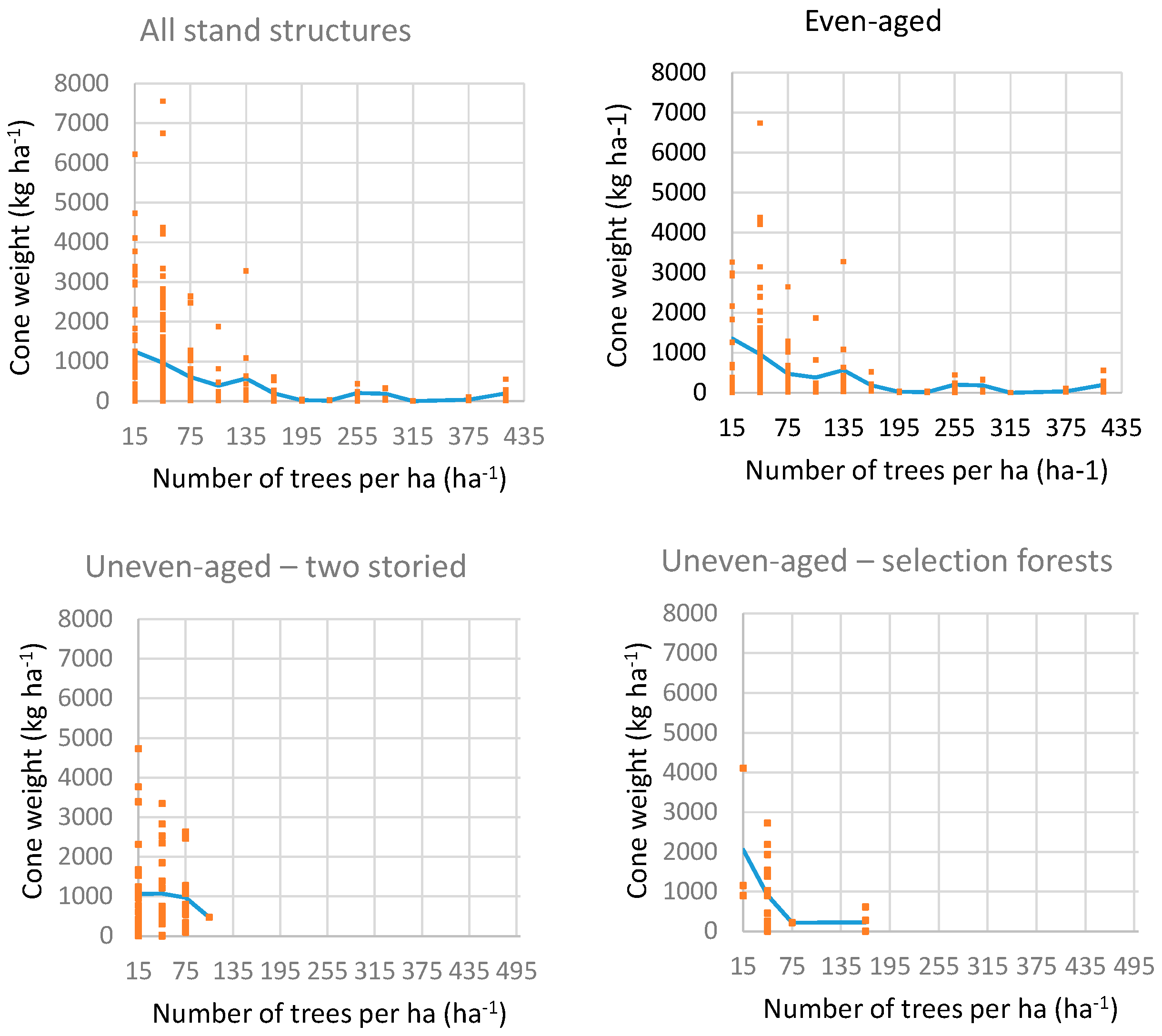
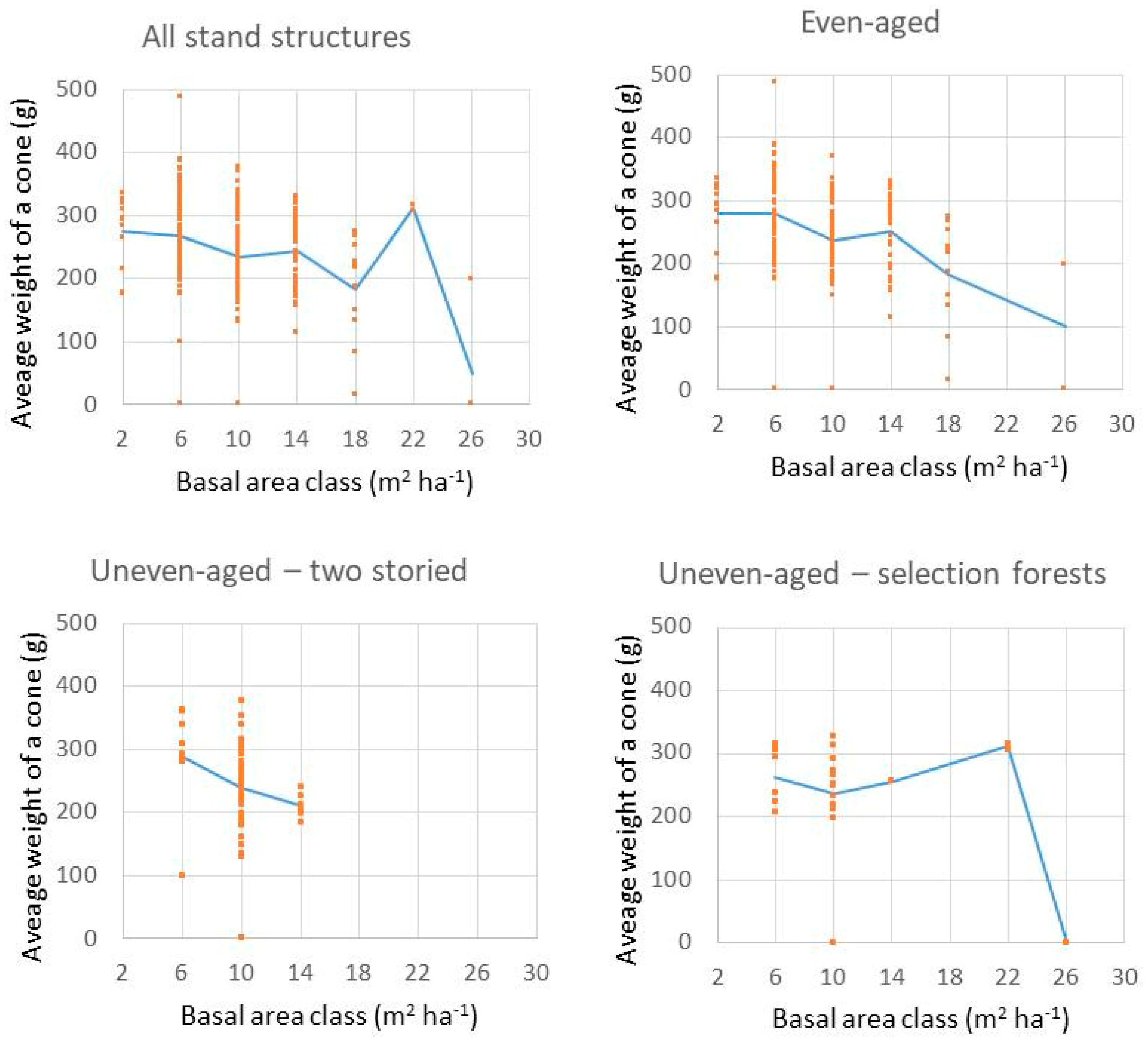
4.2. Impact of Competition on Cone Production at Portuguese Umbrella Pine Stands
4.2.1. Description of Data Used
4.2.2. Methodology
4.2.3. Results
5. Climate Change Impacts on Mediterranean Forests
5.1. Climate Changes on Mediterranean Basin
- An annual average temperature increase of 0.5 °C per decade;
- Decrease in thermal amplitude due to a more pronounced increase in minimum temperature;
- Increase in the number of extremely hot days;
- Reduction in the number of cold days;
- Increase in the frequency of heat waves;
- Total rainfall decrease in about 80% of the meteorological stations;
- Spring rainfall decrease, coinciding with major cone growth one year before harvest, and increase in autumn;
- Increase in the frequency and intensity of droughts.
5.2. Impacts of Climate Change on Mediterranean Forests
- Increase of dieback processes (growth reduction, loss of vegetative vigor, defoliation, mortality) associated with the occurrence of drought processes and higher summer temperatures [15];
- Decrease of net primary productivity of Mediterranean forests, which might become carbon sources in the second half of the 21st century [15].
6. Adapting Umbrella Pines Stand Management to Climate Change
6.1. Tree Scale
6.1.1. Photosynthetic Efficiency and Individual Resistance to Biotic and Abiotic Agents Increase
6.1.2. Promote the Change of Structures or Species
6.2. Stand Scale
6.2.1. Managing Competition between Trees
6.2.2. Increasing Inter- and Intra-Specific Diversity
6.2.3. Irrigation
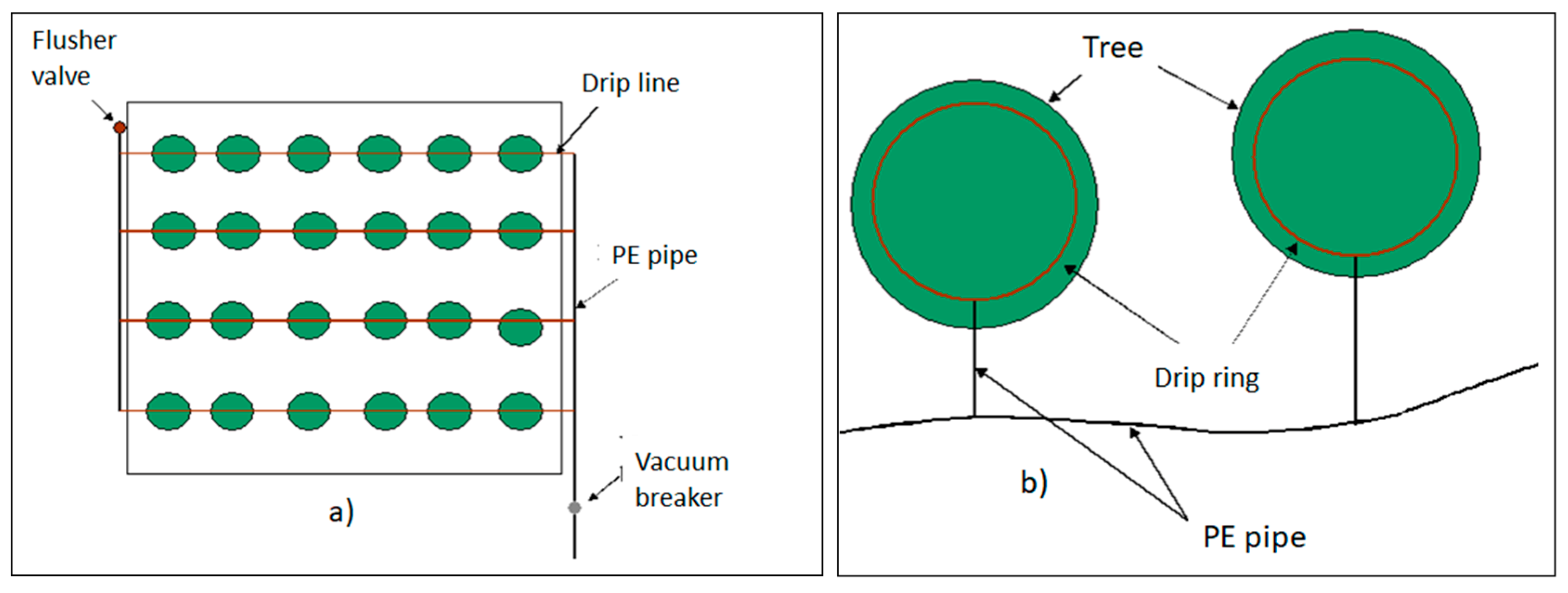
Irrigation Systems
- Plant foliage is not wetted, reducing the potential for foliar diseases, which have a particular importance in young stands;
- Since the area between rows is not irrigated, fewer weeds grow, which can reduce herbicide use;
- Water is distributed more uniformly, with typically 90% (or higher) uniformity;
- Energy costs reduction, since the system operates at lower pressure and less water is required;
- Water can be distributed on the soil surface (surface drip irrigation) or through lines in the plant root zone (subsurface drip irrigation), which will reduce damage to irrigation components by machinery and/or cattle;
- The drip irrigation system can be used to distribute water-soluble fertilizer. Fertirrigation allows fertilizer to be applied to the crop as needed during the growing season, reducing nutrient losses and leaching;
- Due to low water application rates, drip irrigation can be used on sloppy grounds without causing erosion or runoff;
- The initial cost of the system can be highly relative to other types of irrigation systems, and there can be recurrent costs if the tubing is replaced periodically;
- Emitter openings are very small, so water must be filtered to prevent plugging;
- Depending on the source water quality, chlorination or acid rising can, eventually, be a problem.
6.2.4. Fertilization
6.3. Landscape Scale
7. Discussion and Conclusions
7.1. Tree Level
7.2. Stand Level
7.3. Landscape Level
7.4. Summarizing
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mutke, S.; Calama, R.; Gonzalez-Martínez, S.C.; Montero, G.; Gordo, F.J.; Bono, M.; Gil, L. Mediterranean Stone Pine: Botany and Horticulture. In Horticultural Reviews; Janick, J., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2012; Volume 39, pp. 153–201. ISBN 978-1-118-09678-9. [Google Scholar]
- Correia, A.; Faias, S.P.; Ruiz-Penado, R.; Chianucci, F.; Cutini, A.; Fontes, L.; Manetti, M.C.; Montero, G.; Soares, P.; Tomé, M. Generalized biomass equations for Stone pine (Pinus pinea L.) across the Mediterranean basin. For. Ecol. Manag. 2018, 429, 425–436. [Google Scholar] [CrossRef]
- Mutke, S.; Gordo, J.; Gil, L. Variability of Mediterranean Stone pine cone production: Tree rings reflect growth adjustments and enhanced synchrony among sites in Iberian stone pine (Pinus pinea L.) under climate change. Agric. For. Meteorol. 2005, 132, 263–272. [Google Scholar] [CrossRef]
- Calama, R.; Gordo, J.; Madrigal, G.; Mutke, S.; Conde, M.; Montero, G.; Pardos, M. Enhanced tools for predicting annual stone pine (Pinus pinea L.) cone production at tree and forest scale in Inner Spain. For. Syst. 2016, 25, 1–17. [Google Scholar] [CrossRef]
- Calama, R.; Sánchez-González, M.; Montero, G. Management Oriented Growth Models for Multifuncional Mediterranean Forests: The Case of the Stone Pine (Pinus pinea L.). In Scientific Tools and Reserach Needs for Multifunctional Mediterranean Forest; Palahí, M., Birot, Y., Rois, M., Eds.; European Forest Institute: Joensuu, Finland, 2007; pp. 57–69. ISBN 978-952-5453-19-5. [Google Scholar]
- Calama, R.; Conde, M.; de-Dios-García, J.; Madrigal, G.; Vázquez-Piqué, J.; Gordo, F.J.; Pardos, M. Linking climate, annual growth and competition in a Mediterranean forest: Pinus pinea in the Spanish Northern Plateau. Agric. For. Meteorol. 2019, 264, 309–332. [Google Scholar] [CrossRef]
- Rodriguez, M. Climate Zones in Spain. Available online: https://prezi.com/b0om-j4tnkdv/climate-zones-in-spain/ (accessed on 24 November 2015).
- Cardoso, M.M.; Lobo, P.A. Delimitação de pisos bioclimáticos e regiões de proveniência de Pinus pinea L. em Portugal. Silva Lusit. 2001, 9, 93–108. [Google Scholar]
- Freire, J.P.A. Modelação do crescimento e da produção de pinha no pinheiro manso. Ph.D. Thesis, Universidade Técnica de Lisboa, Instituto Superior de Agronomia, Lisboa, Portugal, 2009. [Google Scholar]
- ICNF. IFN6—Áreas dos usos do solo e das espécies florestais de Portugal continental. Resultados preliminaries; Instituto da Conservação da Natureza e das Florestas: Lisboa, Portugal, 2013; p. 34.
- Campelo, F.; Nabais, C.; Freitas, H.; Gutiérrez, E. Climatic significance of tree-ring width and intra-annual density fluctuations in Pinus pinea from dry Mediterranean area in Portugal. Ann. For. Sci. 2006, 64, 229–238. [Google Scholar] [CrossRef]
- De Luis, M.; Novak, K.; Čufar, K.; Raventós, J. Size mediated climate-growth relationships in Pinus halepensis and Pinus pinea. Trees 2009, 23, 1065–1073. [Google Scholar] [CrossRef]
- Mutke, S.; Gordo, J.; Climent, J.; Gil, L. Shoot growth and phenology modelling of grafted stone pine (Pinus pinea L.) in Inner Spain. Ann. For. Sci. 2003, 60, 527–537. [Google Scholar] [CrossRef]
- Pessoa, F.; Lidon, F.; Reboredo, F. Drought effects on Portuguese Forest Cover. In Forest Context and Policies in Portugal; Reboredo, F., Ed.; Springer: Cham, Switzerland, 2014; pp. 67–96. ISBN 978-3-319-08455-8. [Google Scholar]
- García-Güemes, C.; Calama, R. La práctica de la selvicultura para la adaptación al cambio climático. In Los bosques y la biodiversidad frente al cambio climático: Impactos, Vulnerabilidad y Adaptación en España; Zavala, M.A., Ed.; Ministerio para la Transición Ecológica: Madrid, Spain, 2015; Chapter 46; p. 12. ISBN 978-84-491-0038-3. [Google Scholar]
- Richardson, D.M.; Rundel, P.W. Ecology and biogeography of Pinus: An introduction. In Ecology and Biogeography of Pinus; Richardson, D.M., Ed.; Cambridge University Press: New York, NY, USA, 1998; pp. 3–45. ISBN 978-0521789103. [Google Scholar]
- Pereira, S.; Prieto, A.; Calama, R.; Diaz-Balteiro, L. Optimal management in Pinus pinea L. stands combining silvicultural schedules for timber and cone production. Silva Fenn. 2015, 49, 1226. [Google Scholar] [CrossRef]
- Barrero, M.; Loisel, R.; Quézel, P.; Richardson, D.M.; Romane, F. Pines of the Mediterranean Basin. In Ecology and biogeography of Pinus; Richardson, D.M., Ed.; Cambridge University Press: New York, NY, USA, 1998; pp. 153–170. ISBN 978-0521789103. [Google Scholar]
- Mutke, S.; Gordo, J.; Khouja, M.L.; Fady, B. Low genetic and high environmental diversity at adaptive traits in Pinus pinea from provenance tests in France and Spain. Opt. Méditerranéennes 2013, 105, 73–79. [Google Scholar]
- Sáez-Laguna, E.; Guevera, M.A.; Díaz, L.M.; Sánchez-Gómez, D.; Collada, C.; Aranda, I.; Cervera1, M.T. Epigenetic Variability in the Genetically Uniform Forest Tree Species Pinus pinea L. PLoS ONE 2014, 9, e103145. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Gómez, D.; Velasco-Conde, T.; Cano-Martín, F.J.; Guevara, M.A.; Cervera, M.T.; Aranda, I. Inter-clonal variation in functional traits in response to drought for a genetically homogeneous Mediterranean conifer. Environ. Exp. Bot. 2011, 70, 104–109. [Google Scholar] [CrossRef]
- Viñas, R.A.; Caudullo, G.; Oliveira, S.; de Rigo, D. Pinus pinea in Europe: Distribution, habitat, usage and threats. In European Atlas of Forest Tree Species; European Commission: Brussels, Belgium, 2016; p. 204. ISBN 978-92-79-36740-3. [Google Scholar] [CrossRef]
- Mamo, N.; Mihretu, M.; Fecadu, M.; Tigabu, M.; Teketay, D. Variation in seed and germination characteristics among Juniperus procera populations in Ethiopia. For. Ecol. Manag. 2006, 225, 320–327. [Google Scholar] [CrossRef]
- Loewe, V.; Navarro-Cerrillo, R.M.; Sánchez Lucas, R.; Ruiz Gómez, F.J.; Jorrín-Novo, J. Variability studies of allochthonous stone pine (Pinus pinea L.) plantations in Chile through nut protein profiling. J Proteom. 2018, 175, 95–104. [Google Scholar] [CrossRef]
- Agrimi, M.; Ciancio, O. Le pin pignon (Pinus pinea L.); Silva Mediterranea, Comitédes questions forestières méditerranéennes; FAO: Rome, Italy, 1993; p. 173. [Google Scholar]
- Saraiva, I. O pinhão; Litexa Editora, Lda.: Lisboa, Portugal, 1997; 112p, ISBN 972-578-129-5. [Google Scholar]
- Calama, R.; Gordo, F.J.; Mutke, S.; Montero, G. An empirical ecological-type model for predicting stone pine (Pinus pinea L.) cone production in the Northern Plateau (Spain). For. Ecol. Manag. 2008, 255, 660–673. [Google Scholar] [CrossRef]
- Rodrigues, A.; Silva, G.L.; Casquilho, M.; Freire, J.; Carrasquinho, I.; Tomé, M. Linear mixed modelling of cone production for Stone Pine in Portugal. Silva Lusit. 2014, 22, 1–27. [Google Scholar]
- Gonçalves, A.C.; Afonso, A.; Pereira, D.G.; Pinheiro, A. Influence of umbrella pine (Pinus pinea L.) stand type and tree characteristics on cone production. Agrofor. Syst. 2016, 91, 1019–1030. [Google Scholar] [CrossRef]
- Ganatsas, P.; Tsakaldimi, M.; Thanos, C. Seed and cone diversity and seed germination of Pinus pinea in Strofylia Site of the Natura 2000 Network. Biodivers. Conserv. 2008, 17, 2427–2439. [Google Scholar] [CrossRef]
- Skordilis, A.; Thanos, C.A. Comparative ecophysiology of seed germination strategies in the seven pine species naturally growing in Greece. In Basic and Applied Aspects of Seed Biology; Ellis, R.H., Black, M., Murdoch, A.J., Hong, T.D., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands; Great Britain, UK, 1997; pp. 623–632. [Google Scholar]
- Escudero, A.; Perez-Garcia, F.; Luzuriaga, A.K. Effects of light, temperature and population variability on the germination of seven Spanish pines. Seed Sci. Res. 2002, 12, 261–271. [Google Scholar] [CrossRef]
- Loewe, M.V.; Delard, R.C. Effect of irrigation in growth and fruit production in stone pine (Pinus pinea L.) in Chile. In Proceedings of the XXIX International Horticultural Congress, Brisbane, Australia, 17–22 August 2014. [Google Scholar]
- Silva, J. Monitorização intensiva do crescimento anual em pinheiro manso (Pinus pinea L.) sujeito a diferentes tratamentos. Master’s Thesis, Forest Research Centre, School of Agronomy, University of Lisbon, Lisbon, Portugal, 2015. [Google Scholar]
- Valdiviesso, T.; Pimpão, M.; Trindade, C.S.; Varela, M.C. Reproductive phenology of Pinus pinea. In Mediterranean Pine Nuts from Forests and Plantations; Carrasquinho, I., Correia, A.C., Mutke, S., Eds.; CIHEAM: Zaragoza, Spain, 2017; pp. 63–68. [Google Scholar]
- Moreno-Fernández, D.; Cañellas, I.; Calama, R.; Gordo, J.; Sánchez-González, M. Thinning increases cone production of stone pine (Pinus pinea L.) stands in the Northern Plateau (Spain). Ann. For. Sci. 2013, 70, 761–768. [Google Scholar] [CrossRef]
- Carneiro, A.N.; d’Alpuim, M.S.H.; Carvalho, M.A.V. Manual Ilustrado de Enxertia do Pinheiro Manso; Estação Florestal Nacional: Oeiras, Portugal, 2007; p. 41. [Google Scholar]
- Evaristo, I.; Tenreiro, R.; Costa, R. Caracterização de Parâmetros Biométricos e de Ácidos Gordos em Pinhões de Populações Portuguesas de Pinus pinea L. Silva Lusit. 2008, 16, 1–19. [Google Scholar]
- Montero-González, G.; Candela-Plaza, J.A.; Rodríguez-Navarro, A. El Pino pinõnero (Pinus pinea L.) en Andalucía: Ecología, distribución y selvicultura; Junta de Andalucia: Sevilha, Spain, 2004; ISBN 84-95785-94-3.
- Calama, R.; Montero, G. Cone and seed production from stone pine (Pinus pinea L.) stands in Central Range (Spain). Eur. J. For. Res. 2007, 126, 23–35. [Google Scholar] [CrossRef]
- Castro-García, S.; Blanco-Rodán, G.L.; Gil-Ribes, J.A. Vibrational and operational parameters in mechanical cone harvesting of stone pine (Pinus pinea L.). Biosys. Eng. 2012, 112, 352–358. [Google Scholar] [CrossRef]
- Calama, R.; Mutke, S.; Tomé, J.; Gordo, J.; Montero, G.; Tomé, M. Modelling spatial and temporal variability in a zeroinflated variable: The case of stone pine (Pinus pinea L.) cone production. Ecol. Model. 2011, 222, 606–618. [Google Scholar] [CrossRef]
- Bonari, E.; Bagliacca, M.; Ciomei, D.; Senesi, G. Raccolta dei pinoli con machine scuotitrici. Macchine e Motori Agricoli 1980, 38, 41–46. [Google Scholar]
- Küçüker, D.M.; Baskent, E.Z. State of stone pine (Pinus pinea) forests in Turkey and their economic importance for rural development. In Options Méditerranéennes: Série A. Séminaires Méditerranéens; Carrasquinho, I., Correia, A.C., Mutke, S., Eds.; CIHEAM Options Méditerranéennes: Série A; Séminaires Méditerranéens: Zaragoza, Spain, 2017; pp. 111–117. [Google Scholar]
- Boutheina, A.; El Aouni, M.H.; Balandier, P. Influence of stand and tree attributes and silviculture on cone and seed productions in forests of Pinus pinea L. in northern Tunisia. Opt. Méditerranéennes Série A Séminaires Méditerranéens 2013, 105, 9–14. [Google Scholar]
- Freire, J.P.A.; Tomé, M.; Silva, C.S.; Telles, M.R. Silvicultural guidelines for managing P. pinea stands in a southeastern Portuguese region for cone production. In Revised Silvicultural Guidelines for Selected MPT and NWFPs. StarTree Deliverable 2.3; Sheppard, J., Mangold, L., Spiecker, H., Eds.; FP7 Project No 311919 KBBE.2012.1.2-06; European Commission: Brussels, Belgium, 2016; p. 113. [Google Scholar]
- Correia, A.V.; Oliveira, A.C. Principais espécies florestais com interesse para Portugal—Zonas de influência Mediterrânea; Estudos e informação 318; Direcção Geral das Florestas: Lisboa, Portugal, 2002; pp. 77–84. ISBN 922-8097-37-9.
- Louro, G.; Marques, H.; Salinas, F. Elementos de apoio à elaboração de projectos florestais, 2ª Edição ed; Estudos e informação 321; Direcção Geral das Florestas: Lisboa, Portugal, 2002; 126p, ISBN 972-8097-48-4.
- INRB. Condução de povoamentos de pinheiro manso e características nutricionais do pinhão; Instituto Nacional dos Recursos Biológicos, I.P.: Oeiras, Portugal, 2008; 50p, ISBN 978-989-95658-3-8.
- Yang, S.; Burkhart, H.E. Application of Height- and Diameter-Based Relative Spacing for Estimation of Stand Basal Area. For. Sci. 2018, 64, 28–32. [Google Scholar] [CrossRef]
- Carrasquinho, I.; Freire, J.A.; Rodrigues, A.; Tomé, M. Selection of Pinus pinea L. plus tree candidates for cone production. Ann. For. Sci. 2010, 67, 814. [Google Scholar] [CrossRef]
- Tomé, M.; Faias, S.P. (Eds.) State of the Art, Review of Silviculture, Models and Decision Support Tools for Multipurpose Trees (MPT) and Non-Wood Forest Products (NWFP); Deliverable 2.1 of the StarTree Project FP7 Project No 311919 KBBE.2012.1.2-06; European Commission: Brussels, Belgiu, 2014; p. 193. [Google Scholar]
- Mutke, S.; Calama, R.; Nasrallah Neaymeh, E.; Roques, A. Impact of the Dry Cone Syndrome on commercial kernel yield of stone pine cones. In Mediterranean Pine Nuts from Forests and Plantations; Carrasquinho, I., Correia, A.C., Mutke, S., Eds.; CIHEAM Options Méditerranéennes: Série A; Séminaires Méditerranéens: Zaragoza, Spain, 2017; Volume 122, pp. 154–196. [Google Scholar]
- Brebu, M.; Ucar, S.; Vasile, C.; Yanik, J. Co-pyrolysis of pine cone with synthetic polymers. Fuel 2010, 89, 1911–1918. [Google Scholar] [CrossRef]
- Ayrilmis, N.; Buyuksari, U.; Avci, E.; Koc, E. Utilization of pine (Pinus pinea L.) cone in manufacture of wood based composite. For. Ecol. Manag. 2009, 259, 65–70. [Google Scholar] [CrossRef]
- Carvalho, A.; Flannigan, M.D.; Logan, M.D.; Logan, K.A.; Gowman, L.M.; Miranda, A.M.; Borrego, C. The impact of spatial resolution on area burned and fire occurrence projections in Portugal under climate change. Clim. Chang. 2010, 98, 177–197. [Google Scholar] [CrossRef]
- Andrade, C.; Santos, J.A. Climate change projections for rainfall in Portugal. In Proceedings of the 11th International Conference of Numerical Analysis and Applied Mathematics, AIP Conference 1558, Rhodes, Greece, 21–27 September 2013. [Google Scholar] [CrossRef]
- Miranda, P.M.A.; Coelho, F.E.S.; Tomé, A.R.; Valente, M.A.; Carvalho, A.; Pires, C.; Pires, H.O.; Pires, V.C.; Ramalho, C. 20th century Portuguese Climate and Climate Scenarios. In Climate Change in Portugal: Scenarios, Impacts and Adptation Measures; Santos, F.D., Forbes, K., Moita, R., Eds.; University of Lisbon, CC-IAM: Lisbon, Portugal, 2002; pp. 23–83. [Google Scholar]
- Soares, P.; Cardoso, R.; Lima, D.; Miranda, P. Climate change signal in the Portuguese precipitation: High-resolution projections using WRF model and EURO-CORDEX multi-model ensembles. In Proceedings of the 19th EGU General Assembly (EGU2017), Vienna, Austria, 23–28 April 2017. [Google Scholar]
- Cunha, L.V.; Oliveira, R.P.; Nascimento, J.; Ribeiro, L. Impacts of climate change on water resources: A case-study for Portugal. In Proceedings of the Fourth InterCeltic Colloquium on Hydrology and Management of Water Resources, Guimarães, Portugal, July 2005. [Google Scholar]
- Mariotti, A.; Panning, Y.; Alessandri, Z. Long-term climate change in the Mediterranean region in the midst of decadal variability. Clim. Dyn. 2015, 44, 1437–1456. [Google Scholar] [CrossRef]
- Bucak, T.; Trolle, D.; Andersen, H.E.; Thodsen, H.; Erdogan, S.; Levi, E.; Filiz, N.; Beklioglu, M. Future water availability in the largest freshwater Mediterranean lake is at great risk as evidenced from simulations with the SWAT model. Sci. Total Environ. 2017, 581–582, 413–425. [Google Scholar] [CrossRef] [PubMed]
- MAFDR. Estratégias de adaptação da agricultura e das florestas às alterações climáticas; Ministério da Agricultura, Florestas e Desenvolvimento Rural: Lisboa, Portugal, 2013; 88p.
- Rosário, L. Indicadores de Desertificação para Portugal Continental; Direcção-Geral dos Recursos Florestais: Lisboa, Portugal, 2004; 59p, ISBN 972-8797-55-7.
- Pereira, J.S.; Correia, A.V.; Correia, A.C.; Ferreira, M.T.; Onofre, N.; Freitas, H.; Godinho, F. Florestas e Biodiversidade. In Alterações Climáticas em Portugal. Cenários, Impactos e Medidas de Adaptação—Projecto SIAM II; Santos, F.D., Miranda, P., Eds.; Gradiva: Lisboa, Portugal, 2006; pp. 301–343. ISBN 989-616-081-3. [Google Scholar]
- Lindner, M.; Maroschek, M.; Neitherer, S.; Kremer, A.; Barati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Lindner, M.; Calama, R. Climate change and the need for adaptation in Mediterranean forests. In Mediterranean Forests Management under New Context of Climate Change; Lucas-Borja, M.E., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2013; pp. 13–28. ISBN 978-1-62417-868-9. [Google Scholar]
- Thompson, I.; Mackey, B.; McNulty, S.; Mosseler, A. Forest Resilience, Biodiversity, and Climate Change. A Synthesis of the Biodiversity/Resilience/Stability Relationship in Forest Ecosystems; Technical Series No. 43; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2009; ISBN 92-9225-137-6. [Google Scholar]
- Innes, J.; Joyce, L.A.; Kellomaki, S.; Loumann, B.; Ogden, A.; Parrotta, J.; Thompson, I. Management for adaptation. In Adapaptation of Forests and People to Climate Change: A Global Assesment Report; IUFRO World Serires; IUFRO: Vienna, Austria, 2009; Volume 22, pp. 135–169. ISBN 978-3-901347-80-1. [Google Scholar]
- Lanner, R.M. An observation on apical dominance and the umbrella-crown of italian stone pine (Pinus pinea, Pinaceae). Econ. Bot. 1989, 43, 128–130. [Google Scholar]
- Mutke, S.; Sievänen, R.; Sievänen, E.; Nikinmaa, E.; Perttunen, J.; Gil, L. Crown architecture of grafted Stone pine (Pinus pinea L.): Shoot growth and bud differentiation. Trees 2005, 19, 15–25. [Google Scholar] [CrossRef]
- Oliveira, J.M. As Árvores: Estrutura Ciclo Vegetativo Crescimento. Apontamentos da disciplina de Biologia Florestal; Escola Superior de Tecnologia e Gestão de Viseu: Viseu, Portugal, 2007; 19p. [Google Scholar]
- Correia, A.C.; Tomé, M.; Pacheco, C.A.; Faias, S.; Dias, A.C.; Freire, J.; Carvalho, P.O.; Pereira, J.S. Biomass allometry and carbon factors for a Mediterranean pine (Pinus pinea L.) in Portugal. For. Syst. 2010, 19, 418–433. [Google Scholar] [CrossRef]
- Surový, P.; Ribeiro, N.A.; Pereira, J.S. Observations on 3-dimensional crown growth of Stone pine. Agrofor. Syst. 2011, 82, 105–110. [Google Scholar] [CrossRef]
- Sharma, R.P.; Vacek, Z.; Vacek, S. Modelling tree crown-to-bole diameter ratio for Norway spruce and European beech. Silva Fenn. 2017, 51, 1740. [Google Scholar] [CrossRef]
- Oliveras, I.; Martínez-Vilalta, J.; Jimendez-Ortiz, T.; Lledó, M.J.; Escarré, A.; Piñol, J. Hydraulic properties of Pinus halepensis, Pinus pinea and Tetraclinis articulata in a dune ecosystem of Eastern Spain. Plant Ecol. 2003, 169, 131–141. [Google Scholar] [CrossRef]
- Tyree, M.T.; Sperry, J.S. The vulnerability of xylem to cavitation and embolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 19–38. [Google Scholar] [CrossRef]
- Davis, S.D.; Ewers, F.W.; Sperry, J.S.; Portwood, K.A.; Crocker, M.C.; Adams, G.C. Shoot dieback during prolonged drought in Ceanothus (Rhamnaceae) chaparral of California: A possible case of hydraulic failure. Am. J. Bot. 2002, 89, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Koch, G.W.; Sillentt, S.C.; Jennings, G.M.; Davis, S.D. The limits to tree height. Nature 2004, 428, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Vilalta, J.; Sala, A.; Piñol, J. The hydraulic architecture of Pinaceae—A review. Plant Ecol. 2004, 171, 3–13. [Google Scholar] [CrossRef]
- Borghetti, M.; Cinnirella, S.; Cinnirella, S.; Magnani, F.; Magnani, F.; Saracino, A.; Saracino, A. Impact of long-term drought on xylem embolism and growth in Pinus halepensis Mill. Trees 1998, 12, 187–195. [Google Scholar] [CrossRef]
- Brodribb, T.J.; McAdam, S.A.M.; Jordan, G.J.; Martinsa, S.C.V. Conifer species adapt to low-rainfall climates by following one of two divergent pathways. Proc. Natl. Acad. Sci. USA 2014, 111, 14489–14493. [Google Scholar] [CrossRef] [PubMed]
- Reid, R.; Hurley, M.; Stephen, P. The Influence of Competition on Tree Growth and Form; Melbourne University: Melbourne, Australia, 2017. [Google Scholar]
- Linkevicius, E.; Kuliesis, A.; Rohle, H.; Schroder, J.; Aleinikovas, M. The impact of competition for growing space on diameter, basal area and height growth in pine trees. Balt For. 2014, 20, 301–313. [Google Scholar]
- Natalini, F.; Correia, A.C.; Vázquez-Piqué, J.; Alejano, R. Tree rings reflect growth adjustments and enhanced synchrony among sites in Iberian stone pine (Pinus pinea L.) under climate change. Ann. For. Sci. 2015, 72, 1023–1033. [Google Scholar] [CrossRef]
- West, G.G. Pinus radiata growth responses to pruning, thinning, and nitrogen fertilizer in Kaingaroa forests. N. Z. J. For. Sci. 1998, 28, 165–181. [Google Scholar]
- Kırdar, E.; Özel, H.B.; Ertekin, M. Effects of pruning on height and diameter growth at stone pine (Pinus pinea L.) afforestations. Bartın Orman Fakültesi Dergisi 2010, 12, 1–10. [Google Scholar]
- Fernández, M.P.; Basauri, J.; Madariaga, C.; Miguélez, M.M.; Olea, R.; Zubizarreta-Gerendiain, A. Effects of thinning and pruning on stem and crown characteristics of radiata pine (Pinus radiata D. Don). iForest 2017, 10, 383–390. [Google Scholar] [CrossRef]
- McDonalds, P.M.; Skinner, C.N.; Fiddler, G.O. Ponderosa pine needle length: An early indicator of release treatment effectiveness. Can. J. For. Res. 1992, 22, 761–764. [Google Scholar] [CrossRef]
- Graham, J.H.; Duda, J.J.; Brown, M.L.; Kitchen, S.G.; Emelen, J.M.; Malol, J.; Bankstalhl, E.; Krzysik, A.J.; Balbach, H.E.; Freeman, D.C. The effects of drought and disturbance on the growth and developmental instability of loblolly pine (Pinus taeda L.). Ecol. Indic. 2015, 20, 143–150. [Google Scholar] [CrossRef]
- Departament de Medi Ambier i Habitatge de Catalunya. Adapting forest management to increased drought risk in Europe. Available online: http://www.futureforest.eu/uploads/drought_guide.pdf (accessed on 18 July 2018).
- McDowell, N.G.; Pocman, W.; Allen, C.; Bresheares, D.; Cobb, N. Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Neves, M. Estudo comparativo dos mecanismos de resistência à secura de alguns porta-enxertos do género Prunus e a sua aplicação ao melhoramento. Ph.D. Thesis, Universidade do Algarve, Unidade de Ciências e Tecnologias Agrárias, Algarve, Portugal, 1997. [Google Scholar]
- USDA. Tree Nuts: World Markets and Trade. Almonds; United States Department of Agriculture, Foreign Agricultural Service: Washington, DC, USA, 2017.
- KEFRI. Tree Planting and Management Techniques under Limited Water Availability; Guideline for Farmers and Extension Agents; Kenya Forestry Research Institute: Nairobi, Kenya, 2014; p. 20. [Google Scholar]
- Kohler, M.; Nägele, G.; Sohn, S.; Bauhus, J. The impact of different thinning regimes on the drought tolerance of Norway Spruce (Picea abies). Eur. J. For. Res. 2010, 129, 1109–1118. [Google Scholar] [CrossRef]
- Briggs, R.D.; Lemin, R.C. Soil drainage class effects on early response of balsam fir to pre-commercial thinning. Soil Sci. Soc. Am. J. 1994, 58, 1231–1239. [Google Scholar] [CrossRef]
- Burns, J.; Puettmann, K.J.; Perala, D. Strip thinning and spacing increases tree growth of young black spruce. North. J. Appl. For. 1996, 13, 68–72. [Google Scholar] [CrossRef]
- Medhurst, J.L.; Beagle, C.L.; Nielson, W.A. Early-age and later-age thinning affects growth, dominance, and intraspecific competition in Eucalyptus nitens plantations. Can. J. For. Res. 2001, 31, 187–197. [Google Scholar] [CrossRef]
- Krannitz, P.G.; Duralia, T.E. Cone and seed production in Pinus ponderosa: A review. West. N. Am. Nat. 2004, 64, 208–218. [Google Scholar]
- Varmola, M.; Salminen, H. Timing and intensity of precommercial thinning in Pinus sylvestris stands. Scand. J. For. Res. 2004, 19, 142–151. [Google Scholar] [CrossRef]
- Juodvalkis, A.; Kairiukstis, L.; Vasiliauskas, R. Effects of thinning on growth of six tree species in north-temperate forests of Lithuania. Eur. J. For. Res. 2005, 124, 187–192. [Google Scholar] [CrossRef]
- Rytter, L.; Werner, M. Influence of early thinning in broadleaved stands on development of remaining stems. Scand. J. For. Res. 2007, 22, 198–210. [Google Scholar] [CrossRef]
- Niemisto, P. Effect of growing density on biomass and stem volume growth of downy birch stands on peatland in Western and Northern Finland. Silva Fenn. 2013, 47, 1–12. [Google Scholar] [CrossRef]
- Diaconu, D.; Kahle, H.P.; Spieckner, H. Tree- and Stand-Level Thinning Effects on Growth of European Beech (Fagus sylvatica L.) on a Northeast- and a Southwest-Facing Slope in Southwest Germany. Forests 2015, 6, 3256–3277. [Google Scholar] [CrossRef]
- Chung, C.-H.; Lin, C.-J.; Lin, S.-T.; Huang, C.-Y. Determining ideal timing of row thinning for a Cryptomeria japonica plantation using event history analysis. Forests 2017, 8, 77. [Google Scholar] [CrossRef]
- Mechergui, T.; Boussaidi, N.; Pardos, M. Short-term effect of early thinning on growth in stone pine in Tunisia. J. New Sci. 2017, 46, 2509–2516. [Google Scholar]
- Loewe, V.; Venegas, A.; Delard, C.; Gonzalez, M. Thinning effect in two young stone pine plantations (Pinus pinea L.) in central southern Chile. Opt. Méditerranéennes 2013, 105, 44–55. [Google Scholar]
- Gonçalves, A.C.; Pommerening, A. Spatial dynamics of cone production in Mediterranean climates: A case study of Pinus pinea L. in Portugal. For. Ecol. Manag. 2011, 266, 83–93. [Google Scholar] [CrossRef]
- Sucoff, E.; Hong, S.G. Effects of thinning on needle water potential in red pine. For. Sci. 1974, 20, 25–29. [Google Scholar] [CrossRef]
- Teskey, R.O.; Bongart, B.C.; Cregg, B.M.; Dougherty, P.M.; Hennessey, T.C. Physiology and genetics of tree growth response to moisture and temperature stress: An examination of the characteristics of loblolly pine (Pinus taeda L.). Tree Physiol. 1987, 3, 41–61. [Google Scholar] [CrossRef]
- Powers, M.D.; Pregitzer, K.S.; Palik, B.J.; Webster, C.R. Wood δ13C, δ18O and radial growth responses of residual red pine to variable retention harvesting. Tree Physiol. 2010, 30, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Mazza, G.; Amorini, E.; Cutini, A.; Manetti, M.C. The influence of thinning on rainfall interception by Pinus pinea L. in Mediterranean coastal stands (Castel Fusano-Rome). Ann. For. Sci. 2011, 68, 1323–1332. [Google Scholar] [CrossRef]
- Bréda, N.; Granier, A.; Aussenac, G. Effects of thinning on soil and tree water relations, transpiration and growth in an oak forest (Quercus petraea Matt.) Liebl.). Tree Physiol. 1995, 15, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.Y.; Concilio, A.; Oakley, B.; North, M.; Chen, J.Q. Spatial variability in microclimate in a mixed-conifer forest before and after thinning and burning treatments. For. Ecol. Manag. 2010, 259, 904–915. [Google Scholar] [CrossRef]
- Molina, A.J.; del Campo, A.D. The effects of experimental thinning on throughfall and stemflow: A contribution towards hydrology-oriented silviculture in Aleppo pine plantations. For. Ecol. Manag. 2012, 269, 206–213. [Google Scholar] [CrossRef]
- Guillemot, E.; Klein, E.K.; Davi, H.; Coubert, F. The effects of thinning intensity and tree size on the growth response to annual climate in Cedrus atlantica: A linear mixed modeling approach. Ann. For. Sci. 2015, 72, 651–663. [Google Scholar] [CrossRef]
- Linares, J.C.; Delgado-Huertas, A.; Carreira, J.A. Climatic trends and different drought adaptive capacity and vulnerability in a mixed Abies pinsapo—Pinus halepensis forest. Clim. Chang. 2011, 105, 67–90. [Google Scholar] [CrossRef]
- Sánchez-Salguero, R.; Navarro-Verrillo, R.M.; Swetnam, T.W.; Zavdala, M.A. Is drought the main decline factor at the rear edge of Europe? The case of southern Iberian pine plantations. For. Ecol. Manag. 2012, 271, 158–169. [Google Scholar] [CrossRef]
- McDowell, N.G.; Adams, H.D.; Bailey, J.D.; Hess, M.; Kob, T.E. Homeostatic maintenance of ponderosa pine gas exchange in response to stand density changes. Ecol. Appl. 2006, 16, 1164–1182. [Google Scholar] [CrossRef]
- Bladon, K.D.; Silins, U.; Landhäusser, S.M.; Messier, C.; Lieffers, V.J. Carbon isotope discrimination and water stress in trembling aspen following variable retention harvesting. Tree Physiol. 2007, 27, 1065–1071. [Google Scholar] [CrossRef]
- Ayari, A.; Khouja, M.L. Ecophysiological variables influencing Aleppo pine seed and cone production: A review. Tree Physiol. 2014, 34, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Chowanski, K.M. Developing Management Guidelines that Balance Cattle and Timber Production with Ecological Interests in the Black Hills of South Dakota. Ph.D. Thesis, South Dakota State University, Brookings, SD, USA, 2016. [Google Scholar]
- Oregon Department of Forestry, Forest Health. Drought Stress in Conifers; Oregon Department of Forestry, Forest Health: Salem, OR, USA, 2016.
- Bošeľa, M.; Konopka, B.; Seben, V.; Vladovic, J.; Tobin, B. Modelling height to diameter ratio—An opportunity to increase Norway spruce stand stability in the Western Carpathians. Ann. For. Sci. 2015, 72, 651–663. [Google Scholar] [CrossRef]
- Verkaik, I.; Espelta, J.M. Post-fire regeneration thinning, cone production, serotiny and regeneration age in Pinus halepensis. For. Ecol. Manag. 2006, 231, 155–163. [Google Scholar] [CrossRef]
- Ayari, A.; Zubizarreta, G.A.; Moya, D.; Khorchani, A.; Khaldi, A. Importance of Non-wood forests products in Tunisia. In Tunisia: Economic, Political and Social Issues; Pusatieri, M., Cannamela, J., Eds.; Nova Science Publishers: Hauppauge, NY, USA, 2012; pp. 141–153. ISBN 978-1-61942-157-8. [Google Scholar]
- Ruano, I.; Rodríguez, E.; Bravo, F. Effects of pre-commercial thinning on growth and reproduction in post fire regeneration of Pinus halepensis Mill. Ann. For. Sci. 2013, 70, 357–366. [Google Scholar] [CrossRef]
- Bravo, F.; Manguire, D.A.; González-Martínez, S.C. Factors affecting cone production in Pinus pinaster Ait.: Lack of growth-reproduction trade-offs but significant effects of climate and tree and stand characteristics. For. Syst. 2017, 26, 1–13. [Google Scholar] [CrossRef]
- Moreira, F.; Duarte, I.; Catry, F.; Acácio, A. Cork extraction as a key factor determining post-fire cork oak survival in a mountain region of southern Portugal. For. Ecol. Manag. 2007, 253, 30–37. [Google Scholar] [CrossRef]
- Catry, F.X.; Moreira, F.; Pausas, J.G.; Fernandes, P.; Rego, F.; Cardillo, E.; Curt, T. Cork oak vulnerability to fire: The role of bark harvesting, tree characteristics and abiotic factors. PLoS ONE 2012, 7, e39810. [Google Scholar] [CrossRef] [PubMed]
- Moreno, G.; Obrador, J.J.; García, A. Impact of evergreen oaks on soil fertility and crop production in intercropped dehesas. Agric. Ecosyst. Environ. 2007, 119, 270–280. [Google Scholar] [CrossRef]
- Correia, A.C.; Galla, A.; Nunes, A.; Pereira, J.S. Ecological Interactions between Cork Oak (Quercus suber L.) and Stone Pine (Pinus pinea L.): Results from a Pot Experiment. Forests 2018, 9, 534. [Google Scholar] [CrossRef]
- David, T.S.; Henriques, M.O.; Kurz-Besson, C.; Nunes, J.; Valente, F.; Vaz, M.; Pereira, J.S.; Siegwolf, R.; Chaves, M.M.; Gazarini, L.C.; et al. Water-use strategies in two co-occurring Mediterranean evergreen oaks: Surviving the summer drought. Tree Physiol. 2007, 27, 793–803. [Google Scholar] [CrossRef] [PubMed]
- David, T.S.; Pinto, C.A.; Nadezhdina, N.; Kurz-Besson, C.; Henriques, M.O.; Quilhó, T.; Cermak, J.; Chaves, M.M.; Pereira, J.S.; David, J.S. Root functioning, tree water use and hydraulic redistribution in Quercus suber trees: A modeling approach based on root sap flow. For. Ecol. Manag. 2013, 307, 136–146. [Google Scholar] [CrossRef]
- Pinto, C.A.; Nadezhdina, N.; David, J.S.; Kurz-Besson, C.; Caldeira, M.C.; Henriques, M.O.; Monteiro, F.G.; Pereira, J.S.; David, T.S. Transpiration in Quercus suber trees under shallow water table conditions: The role of soil and groundwater. Hydrol. Process. 2013. [Google Scholar] [CrossRef]
- Knoke, T.; Ammer, C.; Stimm, B.; Mosandl, R. Admixing broadleaves to coniferous tree species: A review on yield, ecological stability and economics. Eur. J. For. Res. 2008, 127, 89–101. [Google Scholar] [CrossRef]
- Jactel, H.; Nicoll, B.C.; Branco, M.; Gonzalez-Olabarria, J.R.; Grodzki, W.; Långström, B.; Moreira, F.; Netherer, D.; Orazio, C.; Piou, D.; et al. The influence of forest stand management on biotic and abiotic risks of damage. Ann. For. Sci. 2009, 66, 701. [Google Scholar] [CrossRef]
- Bono, D.; Aletà, N. Cone yield evaluation of a grafted Pinus pinea L. trial. In Mediterranean Stone Pine for Agroforestry; Mutke, S., Piqué, M., Calama, R., Eds.; Séminaires Méditerranéens: Zaragoza, Spain, 2013; Volume 105, pp. 35–41. [Google Scholar]
- Aletà, N.; Vilanova, A. Cone production of stone pine grafted onto Allepo pine. In Proceedings of the 5th International Conference on Mediterranean Pines (Medpine5), Solsona, Spain, 22–26 September 2014. [Google Scholar]
- Loewe, V. Caracterización de la variabilidad, crecimiento y producción del pino piñonero (Pinus pinea L.) en Chile en función del clima y de algunas prácticas silviculturales. PhD. Thesis, Department of Forest Engendering, Córdoba University, Córdoba, Chile, 2016. [Google Scholar]
- Loewe, M.V.; Alvarez, A.P.; Balzarini, M.; Delard, C.; Cerrillo, R. Mineral fertilization and irrigation effects on fruiting and growth in stone pine (Pinus pinea L.) crop. Fruits 2017, 72, 281–287. [Google Scholar] [CrossRef]
- Evans, R.G.; Sadler, J. Methods and technologies to improve efficiency of water use. Water Resour. Res. 2008, 44, W00E04. [Google Scholar] [CrossRef]
- Ruiz-Sanchez, M.C.; Domingo, R.; Castel, J.R. Review. Deficit irrigation in fruit trees and vines in Spain. Span. J. Agric. Res. 2010, 8, S5–S20. [Google Scholar]
- Goodwin, I.; Boland, A.-M. Scheduling deficit irrigation of fruit trees for optimizing water use efficiency. In Deficit Irrigation Practices; FAO: Rome, Italy, 2002; pp. 67–79. [Google Scholar]
- Wang, D. Deficit irrigation of peach trees to reduce water consumption. Water Resour. Manag. 2011, 6, 497–505. [Google Scholar] [CrossRef]
- Zuazo, V.H.D.; Pleguezuelo, C.R.R.; Tarifa, D.F. Impact of sustained-deficit irrigation on tree growth, mineral nutrition, fruit yield and quality of mango in Spain. Fruits 2011, 66, 257–268. [Google Scholar] [CrossRef]
- Puerto, P.; Domingo, R.; Torresb, R.; Pérez-Pastor, A.; García-Riquelme, M. Remote management of deficit irrigation in almond trees based on maximum daily trunk shrinkage. Water relations and yield. Agric. Water Manag. 2013, 126, 33–45. [Google Scholar] [CrossRef]
- Fallahi, E.; Neilsen, D.; Neilsen, G.H.; Fallahi, B.; Shafii, B. Efficient irrigation for optimum fruit quality and yield in apples. HortScience 2010, 45, 1616–1619. [Google Scholar] [CrossRef]
- Bryla, D.R.; Dickson, E.; Shenk, R.; Johnson, R.S.; Crisosto, C.H.; Trout, T.J. Influence of irrigation method and scheduling on patterns of soil and tree water status and its relation to yield and fruit quality in peach. HortScience 2005, 40, 2118–2124. [Google Scholar] [CrossRef]
- Godin, R.; Broner, I. Micro-Sprinkler Irrigation for Orchards. Colorado State University Extension Irrigation Specialist. Fact Sheet No. 4.703. 2013. Available online: http://www.webcitation.org/70xRCvJlc (accessed on 18 July 2018).
- USGA. Drip Irrigation Improves Water Management. 2017. Available online: http://www.webcitation.org/70z4WQBdB (accessed on 18 July 2018).
- Bravdo, B.; Proebsting, E.L. Use of drip irrigation in orchards. HortTechology 1993, 3, 44–49. [Google Scholar] [CrossRef]
- Sanfor, S.; Panuska, J. The Basics of Micro Irrigation. College of Agricultural & Life Sciences, University of Wisconsin-Extension Archived by WebCite®. Available online: http://www.webcitation.org/710FcYudj (accessed on 18 July 2018).
- Lamond, W.J.; Orzolik, M.D.; Harper, J.K.; Kime, L.F.; Jarrett, A.R. Drip Irrigation for Vegetable Production; Pennsylvania State University: University Park, PA, USA, 2012; Archived by WebCite®; Available online: http://www.webcitation.org/710HySBfY (accessed on 18 July 2018).
- Simonne, E.; Hochmuth, R.; Breman, J.; Lamount, W.; Treadwell, K.; Gazula, A. Drip-Irrigation Systems for Small Conventional Vegetable Farms and Organic Vegetable Farms, Bulletin HS1144; University of Florida Extension: Gaineville, FL, USA, 2015; Archived by WebCite®; Available online: http://www.webcitation.org/710JIDKRX (accessed on 18 July 2018).
- DIG. Irrigating Trees with Drip Irrigation. 2018. Archived by WebCite®. Available online: http://www.webcitation.org/710T2LlDn (accessed on 18 July 2018).
- Tuton, H. Sub-Surface Irrigation Systems. Archived by WebCite®. Available online: http://www.webcitation.org/711f1GKik (accessed on 19 July 2018).
- Calama, R.; Madrigal, G.C.J.; Montero, G. Effects of fertilization on the production of an edible forest fruit: Stone pine (Pinus pinea L.) nuts in the south-west of Andalusia. For. Syst. 2007, 16, 241–252. [Google Scholar] [CrossRef]
- Zhou, G.; Peng, S.; Liu, Y.; Wei, Q.; Han, J.; Islam, M. The physiological and nutritional responses of seven different citrus rootstock seedlings to boron deficiency. Trees 2014, 28, 295–307. [Google Scholar] [CrossRef]
- González, J.R.; Trabasobares, A.; Palahí, M.; Pukkala, T. Predicting stand damage and tree survival in burned forests in Catalonia (North-East Spain). Ann. For. Sci. 2007, 64, 733–742. [Google Scholar] [CrossRef]
- Sharma, A.; Bohn, K.K.; Jose, S.; Dwivedi, P. Even-Aged vs. Uneven-Aged Silviculture: Implications for Multifunctional Management of Southern Pine Ecosystems. Forests 2016, 7, 86. [Google Scholar] [CrossRef]
- Parr, C.L.; Andersen, A.N. Patch mosaic burning for biodiversity conservation: A critique of the pyrodiversity paradigm. Conserv. Biol. 2006, 20, 1610–1619. [Google Scholar] [CrossRef] [PubMed]
- Wardell-Johnson, G.; Burrows, N.; Li, S. Emergent patterns in the mosaic of patch burning vary with fire environment and landscape context in south-western Austalia. In Proceedings of the Bushfire Conference, Brisbane, Australia, 6–9 June 2006. In A Fire-Prone Environment: Translating Science into Practice. [Google Scholar]
- Ducan, B.W.; Schmalzer, P.A.; Breinninger, D.R.; Stolen, E.D. Comparing fuels reduction and patch mosaic fire regimes for reducing fire spread potential: A spatial modeling approach. Ecol. Model. 2015, 314, 90–99. [Google Scholar] [CrossRef]
- FAO. International Handbook on Forest. Technical Guide for the Countries of the Mediterranean Basin; FAO: Tholonet, France, 2001; 163p. [Google Scholar]
- D.R.F.C. Plano de Gestão Florestal. Mata Nacional do Urso. Mata Nacional do Pedrogão; Direcção Regional das Florestas do Centro; Autoridade Florestal Nacional; Ministério da Agricultura, Desenvolvimento Rural e das Pescas: Lisbon, Portugal, 2010; p. 174.
- Nitsch, N.; Osterburg, B.; Roggendorf, W.; Laggner, B. Cross compliance and the protection of grassland—Illustrative analyses of land use transitions between permanent grassland and arable land in German regions. Land Use Policy 1984, 29, 440–448. [Google Scholar] [CrossRef]
- Dragomir, N. Multifunctional utilization of pastures in Romania. Sci. Pap. Anim. Sci. Biotechnol. 2009, 41, 191–194. [Google Scholar]
- Nunes, A.N.; Almeida, A.C.; Coelho, A.O.A. Impacts of land use and cover type on runoff and soil erosion in a marginal area of Portugal. Appl. Geogr. 2011, 31, 687–699. [Google Scholar] [CrossRef]
- Panagos, P.; Borrelli, P.; Poesen, J.; Ballabio, C.; Lugato, E.; Meusburger, K.; Montanarella, L.; Alewell, C. The new assessment of soil loss by water erosion in Europe. Environ. Sci. Policy 2015, 54, 438–447. [Google Scholar] [CrossRef]
- Varela, R.; Górriz-Mifsud, E.; Ruiz-Mirazo, J.; López-i-Gelats, F. Payment for Targeted Grazing: Integrating Local Shepherds into Wildfire Prevention. Forests 2018, 9, 464. [Google Scholar] [CrossRef]
- Freire, J.A.; Tomé, M.; Constantino, M. Grafted stone pine stand installation optimization. In Proceedings of the 5th International Conference on Mediterranean Pines (MEDPINE5), Solsona, Spain, 22–26 September 2014. [Google Scholar]
- Pique-Nicolau, M.; del-Rio, M.; Calama, R.; Montero, G. Modelling silviculture alternatives for managing Pinus pinea L. forest in North-East Spain. For. Syst. 2011, 20, 3–30. [Google Scholar] [CrossRef]
- Manso, R.; Pukkala, T.; Pardos, M.; Miina, J.; Calama, R. Modelling Pinus pinea forest management to attain natural regeneration under present and future climatic scenarios. Can. J. For. Res. 2014, 44, 250–262. [Google Scholar] [CrossRef]
| Countries | Tree Cone Number | Tree Cone Weight (kg) | Cone Average Fresh Weight (g) | |||
|---|---|---|---|---|---|---|
| Values | References | Values | References | Values | References | |
| Portugal (overall) | 100–120 | [37] | 286 122–300 | [26,38] | ||
| Portugal (Setúbal distric) | 200–250 >2000 | [37] | 15 0.06–390 | [9] | ||
| Spain (overall) | 1–473 | [39,40,41] | 0.2–160 | [39,42] | 29–852 | [39,40] |
| Spain (Northern Plateau) | 9 | [4] | 2.4 | [4] | ||
| Spain (Central Region) | 15 | [4] | 4.6 | [4] | ||
| Italy | 1–305 | [25,43] | 344–423 | [43] | ||
| Turkey | 146,300 | [25,43,44] | 100–120 | [44] | ||
| Tunisia | 7 | [45] | 60–389 | [45] | ||
| Greece | 101.2–162.5 | [30] | ||||
| Operation | Traditional Silvicultural Systems | ||
|---|---|---|---|
| [47] | [48] | [49] | |
| Initial density | 625 Trees ha−1 | 500 till 600 Trees ha−1 | 208 till 400 Trees ha−1 |
| Pruning | Removal of branches without female flowers (without indication of periodicity) | 1/3 branches removal: Between 8 and 12 years Between 20 and 25 years Removal of branches without female flowers: Between 35 and 40 years Between 50 and 60 years | 1/3 till 2/3 branches removal: Between 5 and 6 years Between 10 and 12 years Between 20 and 25 years |
| Thinning | At 10 years till 500 Trees ha−1 At 15 years till 300 Trees ha−1 At 31 years till 100 Trees ha−1 | Between 15 and 20 years till 440 Trees ha−1 Between 20 and 25 years till 352 Trees ha−1 Between 25 and 30 years till 281 Trees ha−1 Between 35 and 40 years till 225 Trees ha−1 | Between 10 and 12 years Between 20 and 25 years No information on density after thinning |
| Final density | 100 Trees ha−1 | 225 Trees ha−1 | Between 100 and 120 Trees ha−1 |
| Mdist | 10.0 m | 6.7 m | Between 10.0 and 9.2 m |
| Revolution | At 80 years | Between 80 and 100 years | No information about the revolution |
| Stand Structure | dg | Cone Yield (kg ha−1 year−1) | GUp (m2 ha−1) | NUp (ha) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Min | Avg | Max | Min | Avg | Max | Min | Avg | Max | ||
| Even-aged | ]0,30] | 1.4 | 302.9 | 553.8 | 3.8 | 7.2 | 12.1 | 60 | 220 | 509 |
| ]30,50] | 319.2 | 1631.1 | 3335.9 | 6.1 | 12.9 | 24.7 | 39 | 96 | 215 | |
| ]50,70] | 7.1 | 2593.0 | 6349.0 | 5.6 | 11.0 | 18.4 | 18 | 44 | 88 | |
| ]70,90] | 3261.5 | 4819.7 | 7550.8 | 7.2 | 12.5 | 15.3 | 18 | 28 | 32 | |
| Uneven-aged two storied | ]30,50] | 463.9 | 1560.8 | 3161.0 | 10.0 | 13.4 | 26.9 | 60 | 102 | 297 |
| ]50,70] | 175.5 | 1365.0 | 3344.1 | 6.5 | 8.8 | 11.5 | 21 | 31 | 56 | |
| ]70,90] | 35.8 | 1501.8 | 4731.8 | 2.5 | 8.1 | 10.9 | 6 | 18 | 28 | |
| Uneven-aged selection forests | ]30,50] | 329.1 | 880.9 | 1706.5 | 12.5 | 16.3 | 20.7 | 127 | 133 | 151 |
| ]50,70] | 109.0 | 842.1 | 1538.0 | 8.8 | 10.5 | 11.6 | 35 | 42 | 50 | |
| G | Cc | N | Up | |||||
|---|---|---|---|---|---|---|---|---|
| (m2 ha−1) | (%) | (ha−1) | dg | |||||
| Up | Total | Up | Total | Up | Total | (cm) | (m) | |
| Average | 10.1 | 11.6 | 55.7 | 61.3 | 50 | 67 | 54.8 | 13.8 |
| Minimum | 6.6 | 6.6 | 25.5 | 25.9 | 18 | 21 | 36.6 | 9.4 |
| Maximum | 17.0 | 17.7 | 75.2 | 79.6 | 141 | 152 | 74.7 | 17.2 |
| Cone Weight (103 kg ha−1 year−1) | Cone Number (103 ha−1 year−1) | |||||||
|---|---|---|---|---|---|---|---|---|
| 2004-05 | 2005-06 | 2006-07 | 2007-08 | 2004-05 | 2005-06 | 2006-07 | 2007-08 | |
| Average | 2.794 | 0.937 | 1.779 | 0.264 | 8.412 | 4.318 | 6.55 | 1.267 |
| Maximum | 6.739 | 1.538 | 4.256 | 0.504 | 21.146 | 7.516 | 14.487 | 1.981 |
| Minimum | 1.247 | 0.548 | 0.54 | 0.063 | 3.597 | 1.687 | 2.423 | 0.34 |
| Carbon Sequestration (kg ha−1 year−1) | Average | ||||
|---|---|---|---|---|---|
| 2004-05 | 2005-06 | 2006-07 | 2007-08 | (kg ha−1 year−1) | |
| Average | 768.2 | 257.6 | 489.2 | 72.6 | 396.9 |
| Maximum | 1853.0 | 422.9 | 1170.2 | 138.6 | 896.2 |
| Minimum | 342.9 | 150.7 | 148.5 | 17.3 | 164.9 |
| N (Trees ha−1) | Reference | Mcw (m) | Mnc | Mwc (kg) | (kg ha−1) |
|---|---|---|---|---|---|
| 28 | [46] | 18.9 | 2136 | 500.4 | 1124.3 |
| 75 | [44] | 11.6 | 1871 | 322.4 | 604.5 |
| 78 | [174] | 11.3 | 400 | 117.8 | 582.7 |
| 100 | [17] | 10.0 | 349 | 90.4 | 422.7 |
| 120 | [49] | 9.1 | 349 | 90.4 | 277.2 |
| 150 | [175] | 8.2 | 349 | 90.4 | 243.6 |
| 225 | [48] | 6.7 | 92 | 22.4 | 196.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
A. Freire, J.; C. Rodrigues, G.; Tomé, M. Climate Change Impacts on Pinus pinea L. Silvicultural System for Cone Production and Ways to Contour Those Impacts: A Review Complemented with Data from Permanent Plots. Forests 2019, 10, 169. https://doi.org/10.3390/f10020169
A. Freire J, C. Rodrigues G, Tomé M. Climate Change Impacts on Pinus pinea L. Silvicultural System for Cone Production and Ways to Contour Those Impacts: A Review Complemented with Data from Permanent Plots. Forests. 2019; 10(2):169. https://doi.org/10.3390/f10020169
Chicago/Turabian StyleA. Freire, João, Gonçalo C. Rodrigues, and Margarida Tomé. 2019. "Climate Change Impacts on Pinus pinea L. Silvicultural System for Cone Production and Ways to Contour Those Impacts: A Review Complemented with Data from Permanent Plots" Forests 10, no. 2: 169. https://doi.org/10.3390/f10020169
APA StyleA. Freire, J., C. Rodrigues, G., & Tomé, M. (2019). Climate Change Impacts on Pinus pinea L. Silvicultural System for Cone Production and Ways to Contour Those Impacts: A Review Complemented with Data from Permanent Plots. Forests, 10(2), 169. https://doi.org/10.3390/f10020169






