Identification and Expression of NAC Transcription Factors of Vaccinium corymbosum L. in Response to Drought Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Drought Conditions
2.2. Determination of Leaf Chlorophyll Fluorescence Parameters
- (1)
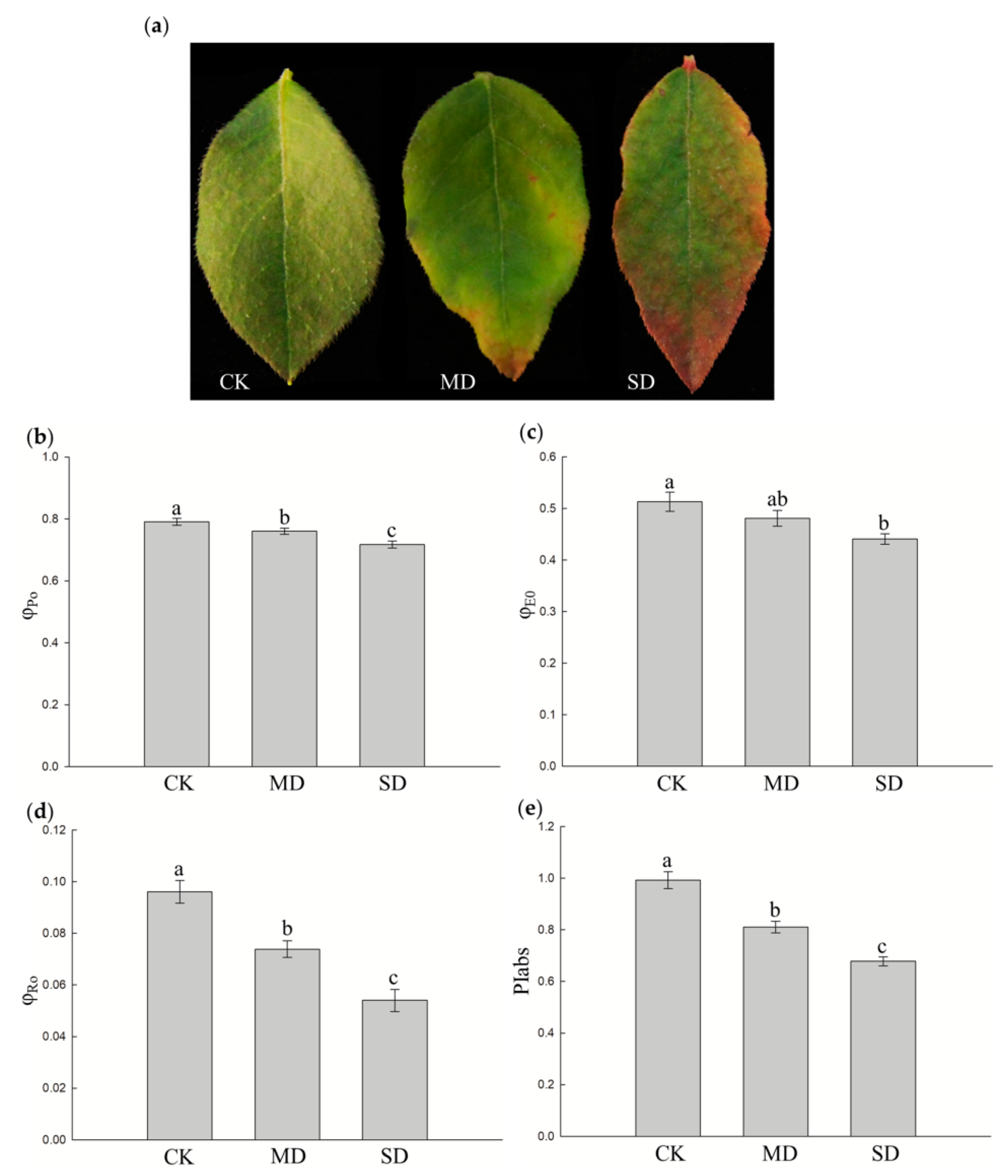
- The maximum quantum yield for primary photochemistry, φPo = Fv/Fm = (Fm − Fo)/Fm. Fm and Fo indicate maximal recorded fluorescence intensity and minimal recorded fluorescence intensity, respectively.
- (2)
- The quantum yield for electron transport, φEo = (Fv/Fm) × (1 − Vj). Vj is the relative variable fluorescence intensity at the J-step.
- (3)
- The reduction of end electron acceptors at the PSI (photosystem I) acceptor side, φRo = TRo/ABS × (1 − Vi). TRo indicates trapped energy by PSII (at t = 0), ABS indicates absorption flux of PSII (at t =0), Vi is the relative variable fluorescence intensity at the I-step.
- (4)
- The performance index on absorption basis, PIabs = (RC/ABS) × φPo/(1 − φPo) × (1 − Vj)/(1 + Vj).
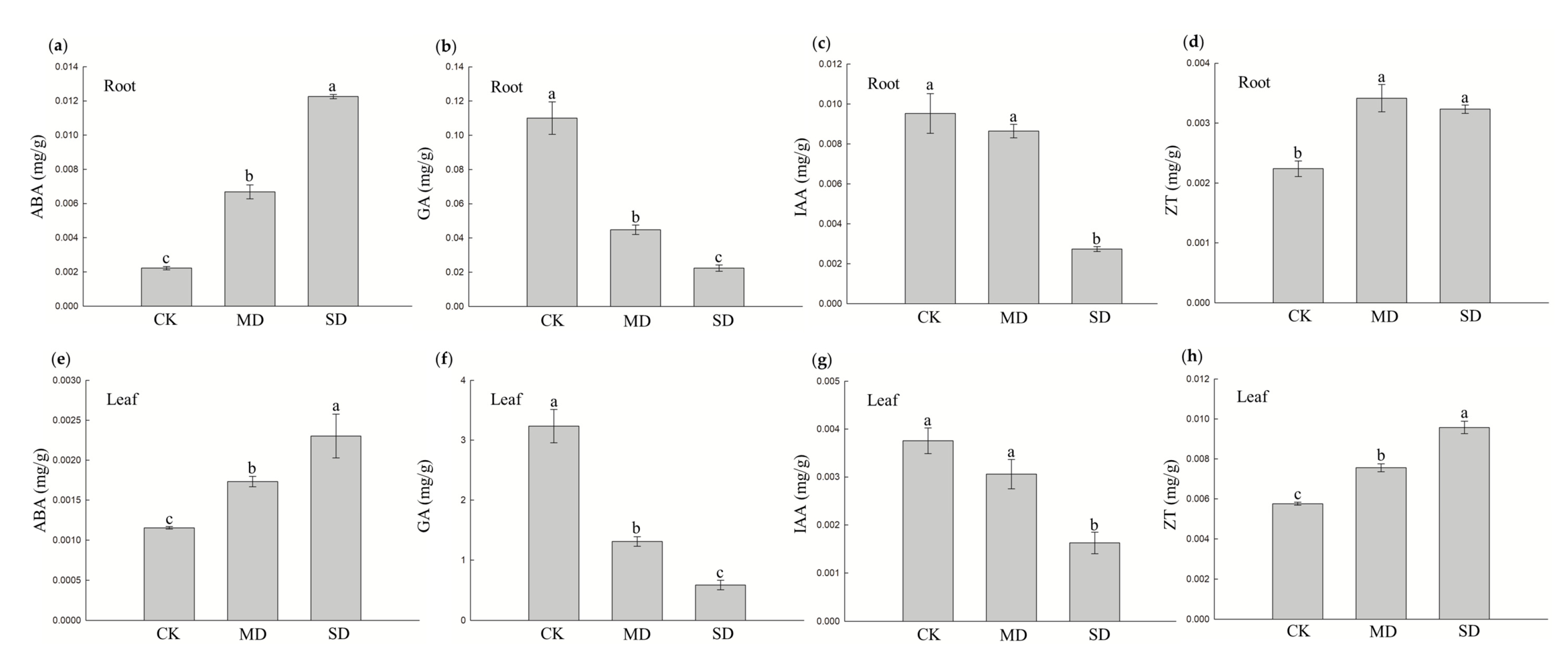
2.3. Measurement of Phytohormones
2.4. RNA Extraction and qRT-PCR Analysis of Gene Expression
2.5. Expression Characterization of NAC TFs Using RNA-seq
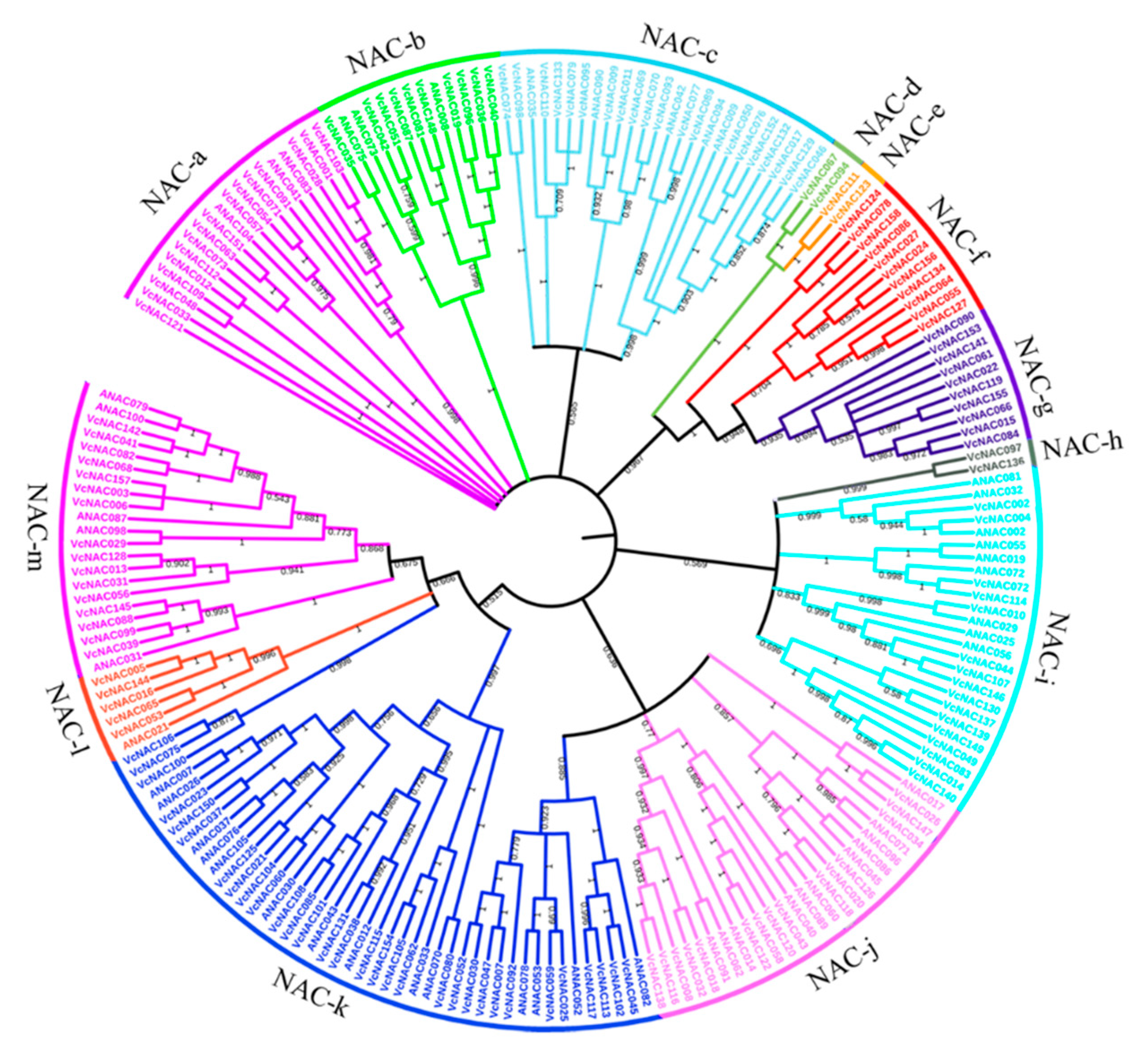
2.6. Phylogenetic Analysis of VcNAC Proteins
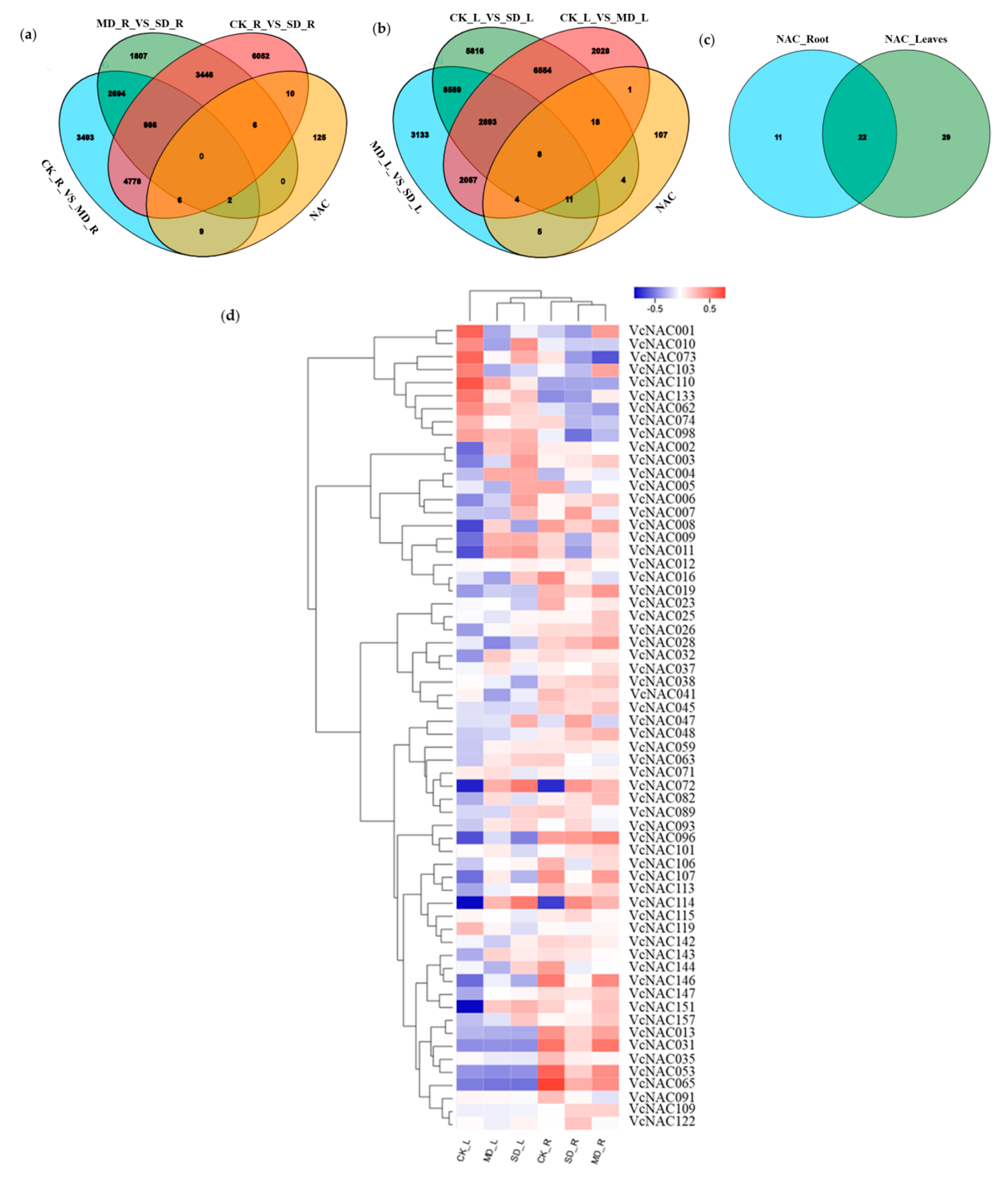
2.7. Identification and Analysis of Differently Expressed Genes
2.8. Statistical Analyses of Data
3. Results
3.1. Effect of Drought Stress on Chlorophyll a Fluorescence Parameters of Blueberry Leaves
3.2. Hormonal Changes in Blueberry Response to Drought Stress
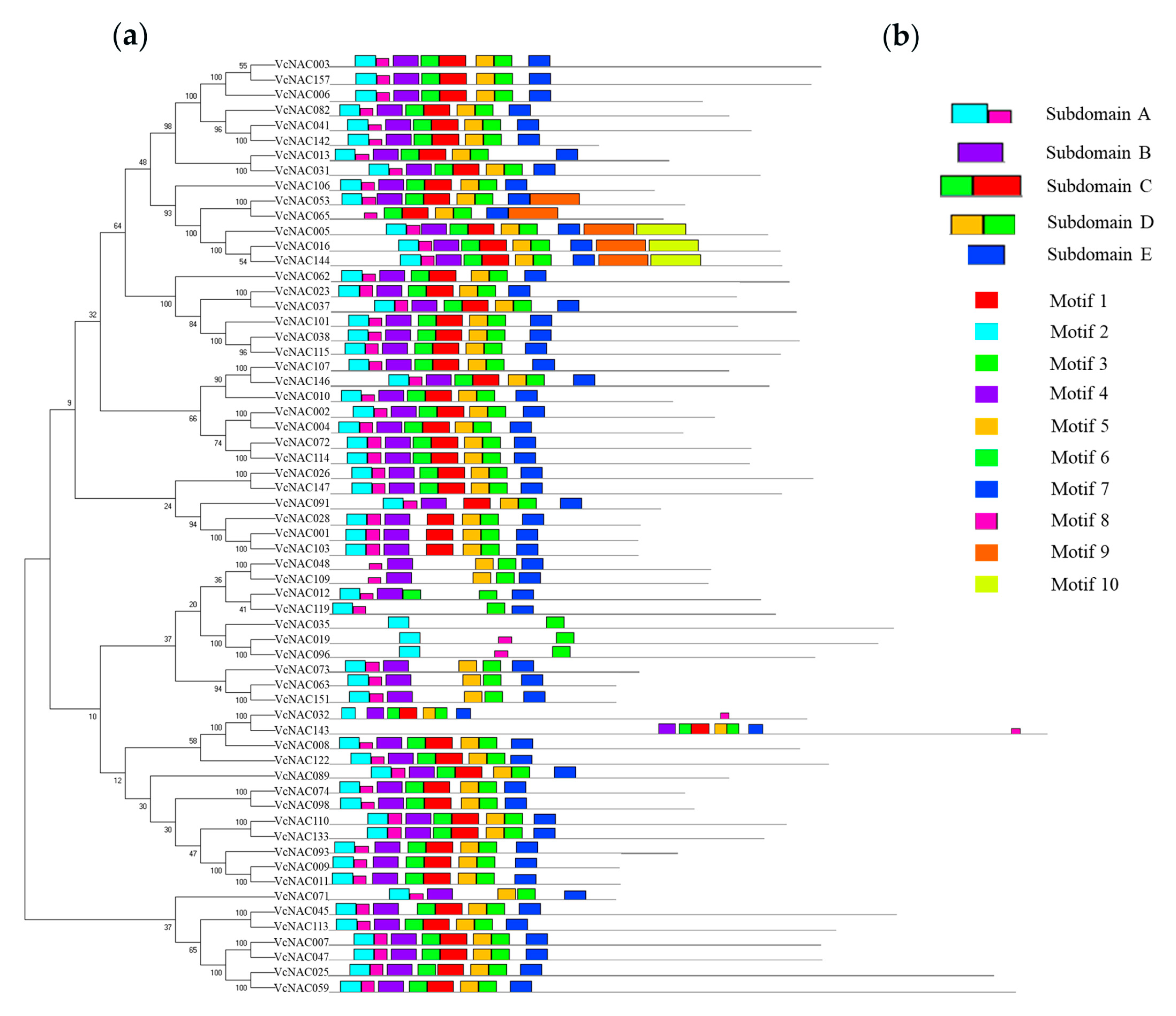
3.3. Identification and Phylogenetic Analysis of NAC TFs in Blueberry
3.4. Identification and Cluster Analysis of Differently Expressed NAC Genes under Drought Stress
3.5. Conserved Motifs and Phylogenetic Analysis of DEGs
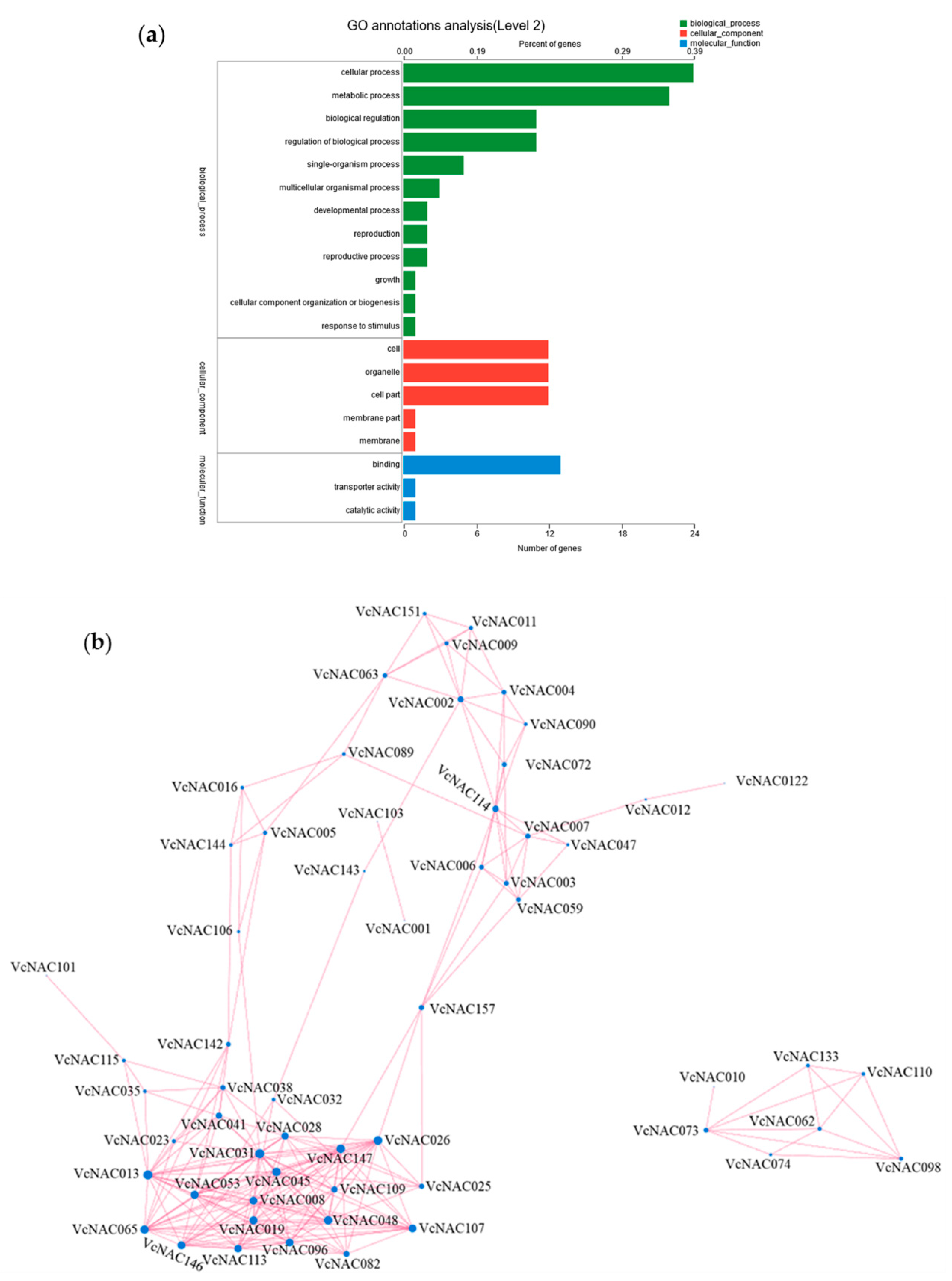
3.6. GO Annotation and Correlation Analysis of DEGs
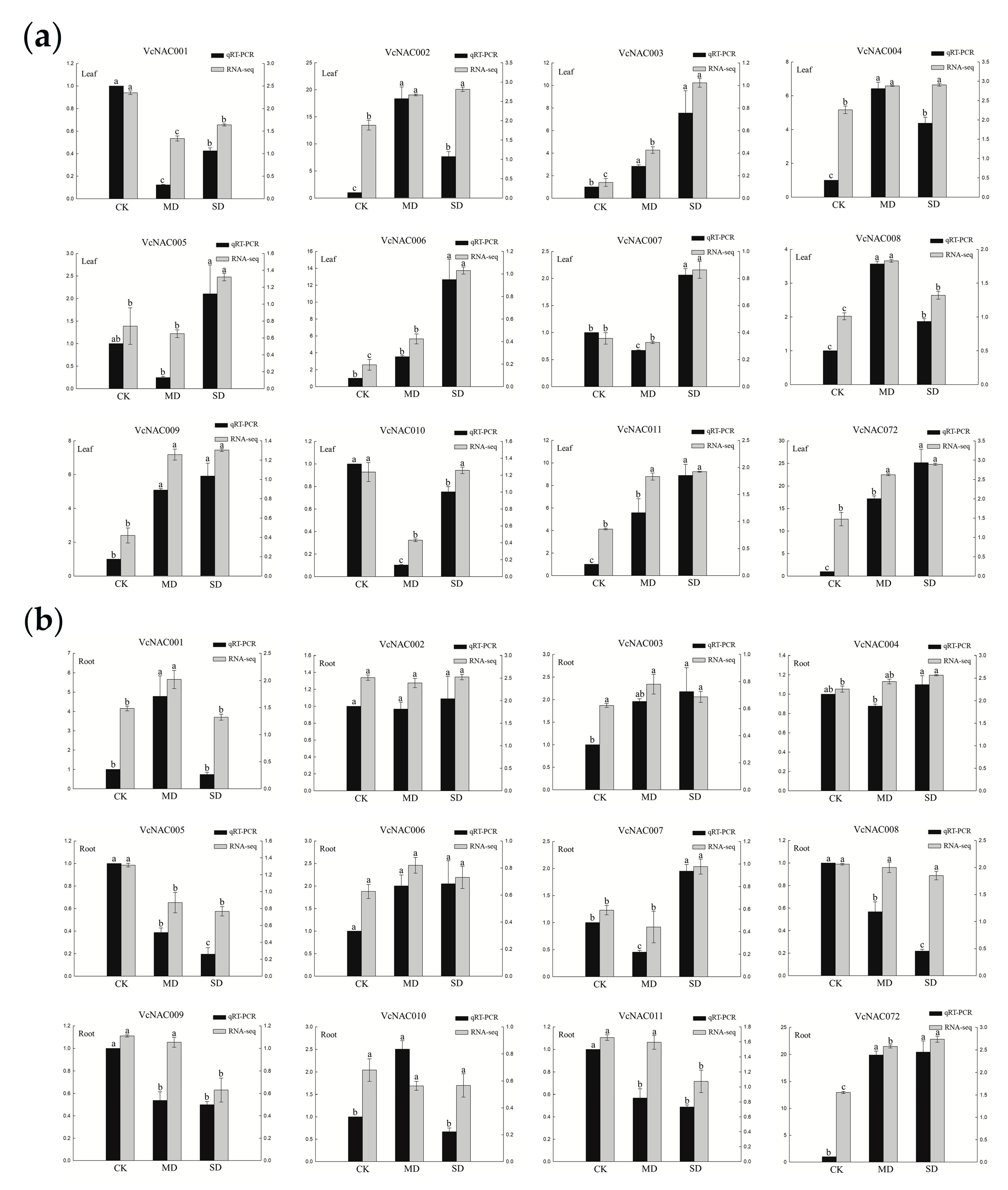
3.7. Validation of RNA-seq Data by Quantitative Real Time (qRT)-PCR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhao, M.R.; Li, J.; Zhu, L.; Chang, P.; Li, L.L.; Zhang, L.Y. Identification and Characterization of MYB-bHLH-WD40 Regulatory Complex Members Controlling Anthocyanidin Biosynthesis in Blueberry Fruits Development. Genes Basel 2019, 10, 496. [Google Scholar] [CrossRef] [PubMed]
- Link, A.; Balaguer, F.; Goel, A. Cancer chemoprevention by dietary polyphenols: Promising role for epigenetics. Biochem. Pharmacol. 2010, 80, 1771–1792. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.F.; Li, B.B.; Shi, W.J.; Gong, Z.Z.; Chen, L.; Hou, Z.X. Transcriptional Activation of Anthocyanin Biosynthesis in Developing Fruit of Blueberries (Vaccinium corymbosum L.) by Preharvest and Postharvest UV Irradiation. J. Agric. Food Chem. 2018, 66, 10931–10942. [Google Scholar] [CrossRef] [PubMed]
- Plunkett, B.J.; Espley, R.V.; Dare, A.P.; Warren, B.W.; Grierson, E.R.P.; Cordiner, S.; Turner, J.L.; Allan, A.C.; Albert, N.W.; Davies, K.M.; et al. MYBA From Blueberry (Vaccinium Section Cyanococcus) Is a Subgroup 6 Type R2R3MYB Transcription Factor That Activates Anthocyanin Production. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Root, T.L.; Price, J.T.; Hall, K.R.; Schneider, S.H.; Rosenzweig, C.; Pounds, J.A. Fingerprints of global warming on wild animals and plants. Nature 2003, 421, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.Y.; Wang, Y.; Zhao, P.X.; Xu, P.; Yu, G.H.; Zhang, L.Y.; Xiong, Y.; Xiang, C.B. AtEDT1/HDG11 regulates stomatal density and water-use efficiency via ERECTA and E2Fa. New Phytol. 2019, 223, 1478–1488. [Google Scholar] [CrossRef]
- Li, M.; Lin, L.; Zhang, Y.H.; Sui, N. ZmMYB31, a R2R3-MYB transcription factor in maize, positively regulates the expression of CBF genes and enhances resistance to chilling and oxidative stress. Mol. Biol. Rep. 2019, 46, 3937–3944. [Google Scholar] [CrossRef]
- Xie, Z.L.; Nolan, R.O.R.; Jiang, H.; Tang, B.Y.; Zhang, M.C.; Li, Z.H.; Yin, Y.H. The AP2/ERF Transcription Factor TINY Modulates Brassinosteroid-Regulated Plant Growth and Drought Responses in Arabidopsis. Plant Cell 2019, 31, 1788–1806. [Google Scholar] [CrossRef]
- Wang, S.J.; Huang, J.J.; Wang, X.D.; Dang, H.; Jiang, T.B.; Han, Y.Z. Expression Analysis of the NAC Transcription Factor Family of Populus in Response to Salt Stress. Forests 2019, 10, 688. [Google Scholar] [CrossRef]
- Ooka, H.; Satoh, K.; Doi, K.; Nagata, T.; Otomo, Y.; Murakami, K.; Matsubara, K.; Osato, N.; Kawai, J.; Carninci, P.; et al. Comprehensive analysis of NAC family genes in Oryza sativa and Arabidopsis thaliana. DNA Res. 2003, 10, 239–247. [Google Scholar] [CrossRef]
- Li, W.; Li, X.X.; Chao, J.T.; Zhang, Z.L.; Wang, W.F.; Guo, Y.F. NAC Family Transcription Factors in Tobacco and Their Potential Role in Regulating Leaf Senescence. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Liu, Z.W.; Wu, Z.J.; Li, H.; Zhuang, J. Transcriptome-Wide Identification and Expression Analysis of the NAC Gene Family in Tea Plant [Camellia sinensis (L.) O. Kuntze]. PLoS ONE 2016, 11, e166727. [Google Scholar] [CrossRef] [PubMed]
- Mitsuda, N.; Iwase, A.; Yamamoto, H.; Yoshida, M.; Seki, M.; Shinozaki, K.; Ohme-Takagi, M. NAC transcription factors, NST1 and NST3, are key regulators of the formation of secondary walls in woody tissues of Arabidopsis. Plant Cell 2007, 19, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.S.; Avci, U.; Grant, E.H.; Haigler, C.H.; Beers, E.P. XND1, a member of the NAC domain family in Arabidopsis thaliana, negatively regulates lignocellulose synthesis and programmed cell death in xylem. Plant J. 2008, 53, 425–436. [Google Scholar] [CrossRef]
- Zhang, H.H.; Cui, X.Y.; Guo, Y.X.; Luo, C.B.; Zhang, L.Y. Picea wilsonii transcription factor NAC2 enhanced plant tolerance to abiotic stress and participated in RFCP1-regulated flowering time. Plant Mol. Biol. 2018, 98, 471–493. [Google Scholar] [CrossRef]
- Liu, C.; Wang, B.M.; Li, Z.X.; Peng, Z.H.; Zhang, J.R. TsNAC1 Is a Key Transcription Factor in Abiotic Stress Resistance and Growth. Plant Physiol. 2018, 176, 742–756. [Google Scholar] [CrossRef]
- Li, S.; Wang, N.; Ji, D.D.; Zhang, W.X.; Wang, Y.; Yu, Y.C.; Zhao, S.Z.; Lyu, M.H.; You, J.J.; Zhang, Y.Y.; et al. A GmSIN1/GmNCED3s/GmRbohBs Feed-Forward Loop Acts as a Signal Amplifier That Regulates Root Growth in Soybean Exposed to Salt Stress. Plant Cell 2019, 31, 2107–2130. [Google Scholar] [CrossRef]
- Yuan, X.; Wang, H.; Cai, J.T.; Bi, Y.; Li, D.Y.; Song, F.M. Rice NAC transcription factor ONAC066 functions as a positive regulator of drought and oxidative stress response. BMC Plant Biol. 2019, 19, 278. [Google Scholar] [CrossRef]
- Kowitcharoen, L.; Wongs-Aree, C.; Setha, S.; Komkhuntod, R.; Srilaong, V.; Kondo, S. Changes in abscisic acid and antioxidant activity in sugar apples under drought conditions. Sci. Hortic. Amst. 2015, 193, 1–6. [Google Scholar] [CrossRef]
- Zhang, S.H.; Xu, X.F.; Sun, Y.M.; Zhang, J.L.; Li, C.Z. Influence of drought hardening on the resistance physiology of potato seedlings under drought stress. J. Integr. Agric. 2018, 17, 336–347. [Google Scholar] [CrossRef]
- Jiang, D.G.; Zhou, L.Y.; Chen, W.T.; Ye, N.H.; Xia, J.X.; Zhuang, C.X. Overexpression of a microRNA-targeted NAC transcription factor improves drought and salt tolerance in Rice via ABA-mediated pathways. Rice 2019, 12, 76. [Google Scholar] [CrossRef] [PubMed]
- Shahnejat-Bushehri, S.; Tarkowska, D.; Sakuraba, Y.; Balazadeh, S. Arabidopsis NAC transcription factor JUB1 regulates GA/BR metabolism and signalling. Nat. Plants 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.K.; Kjaersgaard, T.; Nielsen, M.M.; Galberg, P.; Petersen, K.; O’Shea, C.; Skriver, K. The Arabidopsis thaliana NAC transcription factor family: Structure-function relationships and determinants of ANAC019 stress signalling. Biochem. J. 2010, 426, 183–196. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.Z.; Liu, H.; Yan, G.J.; Li, G.; Turner, N.C. Roots of Lucerne Seedlings are More Resilient to a Water Deficit than Leaves or Stems. Agron. Basel 2019, 9, 123. [Google Scholar] [CrossRef]
- Zaman, N.K.; Abdullah, M.Y.; Othman, S.; Zaman, N.K. Growth and Physiological Performance of Aerobic and Lowland Rice as Affected by Water Stress at Selected Growth Stages. Rice Sci. 2018, 25, 82–93. [Google Scholar] [CrossRef]
- Gao, S.S.; Wang, Y.L.; Yu, S.; Huang, Y.Q.; Liu, H.C.; Chen, W.; He, X.Y. Effects of drought stress on growth, physiology and secondary metabolites of Two Adonis species in Northeast China. Sci Hortic. Amst. 2020, 259, 108795. [Google Scholar] [CrossRef]
- Bankaji, I.; Sleimi, N.; Vives-Peris, V.; Gomez-Cadenas, A.; Perez-Clemente, R.M. Identification and expression of the Cucurbita WRKY transcription factors in response to water deficit and salt stress. Sci. Hortic. Amst. 2019, 256, 562. [Google Scholar] [CrossRef]
- Strauss, A.J.; Kruger, G.H.J.; Strasser, R.J.; Van Heerden, P.D.R. Ranking of dark chilling tolerance in soybean genotypes probed by the chlorophyll a fluorescence transient O-J-I-P. Environ. Exp. Bot. 2006, 56, 147–157. [Google Scholar] [CrossRef]
- Durgbanshi, A.; Arbona, V.; Pozo, O.; Miersch, O.; Sancho, J.V.; Gomez-Cadenas, A. Simultaneous determination of multiple phytohormones in plant extracts by liquid chromatography-electrospray tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 8437–8442. [Google Scholar] [CrossRef]
- Wen, Y.; Su, S.C.; Ma, L.Y.; Wang, X.N. Effects of gibberellic acid on photosynthesis and endogenous hormones of Camellia oleifera Abel. in 1st and 6th leaves. J. For. Res. Jpn. 2018, 23, 309–317. [Google Scholar] [CrossRef]
- Vashisth, T.; Johnson, L.K.; Malladi, A. An efficient RNA isolation procedure and identification of reference genes for normalization of gene expression in blueberry. Plant Cell Rep. 2011, 30, 2167–2176. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Bailey, T.L.; Johnson, J.; Grant, C.E.; Noble, W.S. The MEME Suite. Nucleic Acids Res. 2015, 43, W39–W49. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wang, Y.H.; Li, B.; Tan, H.; Li, D.N.; Li, L.; Liu, X.; Han, J.C.; Meng, X.J. Comparative transcriptome analysis of genes involved in anthocyanin synthesis in blueberry. Plant Physiol. Biochem. 2018, 127, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Lin, X.; Zhang, D.; Li, Q.; Zhao, X.; Chen, S. Genome-Wide Analysis of NAC Gene Family in Betula pendula. Forests 2019, 10, 741. [Google Scholar] [CrossRef]
- Dong, H.Y.; Xu, B.; Ji, K.S. Comparative transcriptome analysis of genes involved in response to thermal stress and leaf colour change of Acer palmatum. Sci. Hortic. Amst. 2019, 255, 77–85. [Google Scholar] [CrossRef]
- Wang, L.K.; Feng, Z.X.; Wang, X.; Wang, X.W.; Zhang, X.G. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef]
- Singh, A.K.; Sharma, V.; Pal, A.K.; Acharya, V.; Ahuja, P.S. Genome-Wide Organization and Expression Profiling of the NAC Transcription Factor Family in Potato (Solanum tuberosum L.). DNA Res. 2013, 20, 403–423. [Google Scholar] [CrossRef]
- Zhu, C.C.; Shi, F.H.; Chen, Y.; Wang, M.; Zhao, Y.Q.; Geng, G.M. Transcriptome Analysis of Chinese Chestnut (Castanea mollissima Blume) in Response to Dryocosmus kuriphilus Yasumatsu Infestation. Int. J. Mol. Sci. 2019, 20, 855. [Google Scholar] [CrossRef]
- Battino, M.; Beekwilder, J.; Denoyes-Rothan, B.; Laimer, M.; McDougall, G.J.; Mezzetti, B. Bioactive compounds in berries relevant to human health. Nutr. Rev. 2009, 67 (Suppl. 1), S145–S150. [Google Scholar] [CrossRef]
- Dixon, R.A.; Xie, D.Y.; Sharma, S.B. Proanthocyanidins—A final frontier in flavonoid research? New Phytol. 2005, 165, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.F.; Kim, M.Y.; Ha, J.; Lee, S.H. Overexpression of the Soybean NAC Gene GmNAC109 Increases Lateral Root Formation and Abiotic Stress Tolerance in Transgenic Arabidopsis Plants. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Walworth, A.; Song, G.Q. The Cold-Regulated Genes of Blueberry and Their Response to Overexpression of VcDDF1 in Several Tissues. Int. J. Mol. Sci. 2018, 19, 1553. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Allakhverdiev, S.I. Recent progress in the studies of structure and function of photosystem II. J. Photochem. Photobiol. B 2011, 104, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.J.; Geng, Q.W.; Du, Y.P.; Yang, X.H.; Zhai, H. Induction of cyclic electron flow around photosystem I during heat stress in grape leaves. Plant Sci. 2017, 256, 65–71. [Google Scholar] [CrossRef]
- Sun, Y.J.; Liu, X.H.; Zhai, H.; Gao, H.Y.; Yao, Y.X.; Du, Y.P. Responses of photosystem II photochemistry and the alternative oxidase pathway to heat stress in grape leaves. Acta Physiol. Plant 2016, 38, 232. [Google Scholar] [CrossRef]
- Ruban, A.V. Nonphotochemical Chlorophyll Fluorescence Quenching: Mechanism and Effectiveness in Protecting Plants from Photodamage. Plant Physiol. 2016, 170, 1903–1916. [Google Scholar] [CrossRef]
- Zhou, R.H.; Kan, X.; Chen, J.J.; Hua, H.L.; Li, Y.; Ren, J.J.; Feng, K.; Liu, H.H.; Deng, D.X.; Yin, Z.T. Drought-induced changes in photosynthetic electron transport in maize probed by prompt fluorescence, delayed fluorescence, P700 and cyclic electron flow signals. Environ. Exp. Bot. 2019, 158, 51–62. [Google Scholar] [CrossRef]
- Zhang, H.H.; Xu, N.; Sui, X.; Zhong, H.X.; Yin, Z.P.; Li, X.; Sun, G.Y. Photosystem II Function Response to Drought Stress in Leaves of Two Alfalfa (Medicago sativa) Varieties. Int. J. Agric. Biol. 2018, 20, 1012–1020. [Google Scholar] [CrossRef]
- He, K.; Zhao, X.; Chi, X.Y.; Wang, Y.P.; Jia, C.L.; Zhang, H.P.; Zhou, G.K.; Hu, R.B. A novel Miscanthus NAC transcription factor MlNAC10 enhances drought and salinity tolerance in transgenic Arabidopsis. J. Plant Physiol. 2019, 233, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.W.; He, K.; Chi, X.Y.; Chai, G.H.; Wang, Y.P.; Jia, C.L.; Zhang, H.P.; Zhou, G.K.; Hu, R.B. Miscanthus NAC transcription factor MlNAC12 positively mediates abiotic stress tolerance in transgenic Arabidopsis. Plant Sci 2018, 277, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.D.; Seo, P.J.; Yoon, H.K.; Park, C.M. The Arabidopsis NAC Transcription Factor VNI2 Integrates Abscisic Acid Signals into Leaf Senescence via the COR/RD Genes. Plant Cell 2011, 23, 2155–2168. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.; Vankova, R.; Yamaguchi-Shinozaki, K.; Shinozaki, K.; Tran, L.S.P. Cytokinins: Metabolism and function in plant adaptation to environmental stresses. Trends Plant Sci 2012, 17, 172–179. [Google Scholar] [CrossRef]
- Helaly, M.N.; El-Hoseiny, H.; El-Sheery, N.I.; Rastogi, A.; Kalaji, H.M. Regulation and physiological role of silicon in alleviating drought stress of mango. Plant Physiol. Biochem. 2017, 118, 31–44. [Google Scholar] [CrossRef]
- Huang, H.J.; Yang, Z.Q.; Zhang, M.Y.; Li, Y.X.; Zhang, J.H.; Hou, M.Y. Effects of Water Stress on Growth, Photosynthesis, Root Activity and Endogenous Hormones of Cucumis sativus. Int. J. Agric. Biol. 2018, 20, 2579–2589. [Google Scholar] [CrossRef]
- Yang, H.; Gu, X.T.; Ding, M.Q.; Lu, W.P.; Lu, D.L. Activities of starch synthetic enzymes and contents of endogenous hormones in waxy maize grains subjected to post-silking water deficit. Sci. Rep. UK 2019, 9, 7059. [Google Scholar] [CrossRef]
- Alshareef, N.O.; Wang, J.Y.; Ali, S.; Al-Babili, S.; Tester, M.; Schmockel, S.M. Overexpression of the NAC transcription factor JUNGBRUNNEN1 (JUB1) increases salinity tolerance in tomato. Plant Physiol. Biochem. 2019, 140, 113–121. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhong, M.; He, L.; Wang, B.; Liu, Q.L.; Pan, Y.Z.; Jiang, B.B.; Zhang, L. Overexpression of a chrysanthemum transcription factor gene DgNAC1 improves drought tolerance in chrysanthemum. Plant Cell Tiss. Org. 2018, 135, 119–132. [Google Scholar] [CrossRef]
- Hu, R.B.; Qi, G.A.; Kong, Y.Z.; Kong, D.J.; Gao, Q.A.; Zhou, G.K. Comprehensive Analysis of NAC Domain Transcription Factor Gene Family in Populus trichocarpa. BMC Plant Biol. 2010, 10, 145. [Google Scholar] [CrossRef]
- Yang, X.W.; Wang, X.Y.; Ji, L.; Yi, Z.L.; Fu, C.X.; Ran, J.C.; Hu, R.B.; Zhou, G.K. Overexpression of a Miscanthus lutarioriparius NAC gene MlNAC5 confers enhanced drought and cold tolerance in Arabidopsis. Plant Cell Rep. 2015, 34, 943–958. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.S.; Oh, N.; Chung, P.J.; Kim, Y.S.; Do Choi, Y.; Kim, J.K. Overexpression of OsNAC14 Improves Drought Tolerance in Rice. Front. Plant Sci. 2018, 9, 310. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.J.; You, J.; Xie, K.B.; Xie, W.B.; Xiong, L.Z. Systematic sequence analysis and identification of tissue-specific or stress-responsive genes of NAC transcription factor family in rice. Mol. Genet. Genom. 2008, 280, 547–563. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Gao, L.; Zhang, Y.; Zhang, F.; Yang, X.; Huang, D. Genome-wide investigation of the NAC transcription factor familyin melon (Cucumis melo L.) and their expression analysis under salt stress. Plant Cell Rep. 2016, 35, 1827–1839. [Google Scholar] [CrossRef]
- Mahmood, K.; Xu, Z.H.; El-Kereamy, A.; Casaretto, J.A.; Rothstein, S.J. The Arabidopsis Transcription Factor ANAC032 Represses Anthocyanin Biosynthesis in Response to High Sucrose and Oxidative and Abiotic Stresses. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Tran, L.S.P.; Nakashima, K.; Sakuma, Y.; Simpson, S.D.; Fujita, Y.; Maruyama, K.; Fujita, M.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Isolation and functional analysis of Arabidopsis stress-inducible NAC transcription factors that bind to a drought-responsive cis-element in the early responsive to dehydration stress 1 promoter. Plant Cell 2004, 16, 2481–2498. [Google Scholar] [CrossRef]
- Li, S.; Gao, J.; Yao, L.Y.; Ren, G.D.; Zhu, X.Y.; Gao, S.; Qiu, K.; Zhou, X.; Kuai, B.K. The role of ANAC072 in the regulation of chlorophyll degradation during age- and dark-induced leaf senescence. Plant Cell Rep. 2016, 35, 1729–1741. [Google Scholar] [CrossRef]
- Jeong, J.S.; Park, Y.T.; Jung, H.; Park, S.H.; Kim, J.K. Rice NAC proteins act as homodimers and heterodimers. Plant Biotechnol. Rep. 2009, 3, 127–134. [Google Scholar] [CrossRef]
- Wang, X.; Culver, J.N. DNA binding specificity of ATAF2, a NAC domain transcription factor targeted for degradation by Tobacco mosaic virus. BMC Plant Biol. 2012, 12, 157. [Google Scholar] [CrossRef]
- Mathew, I.E.; Das, S.; Mahto, A.; Agarwal, P. Three Rice NAC Transcription Factors Heteromerize and Are Associated with Seed Size. Front. Plant Sci. 2016, 7, 1638. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, K.; Wang, A.; Sun, Y.; Yu, M.; Zhang, L. Identification and Expression of NAC Transcription Factors of Vaccinium corymbosum L. in Response to Drought Stress. Forests 2019, 10, 1088. https://doi.org/10.3390/f10121088
Liang K, Wang A, Sun Y, Yu M, Zhang L. Identification and Expression of NAC Transcription Factors of Vaccinium corymbosum L. in Response to Drought Stress. Forests. 2019; 10(12):1088. https://doi.org/10.3390/f10121088
Chicago/Turabian StyleLiang, Kehao, Aibin Wang, Yongjiang Sun, Mingxin Yu, and Lingyun Zhang. 2019. "Identification and Expression of NAC Transcription Factors of Vaccinium corymbosum L. in Response to Drought Stress" Forests 10, no. 12: 1088. https://doi.org/10.3390/f10121088
APA StyleLiang, K., Wang, A., Sun, Y., Yu, M., & Zhang, L. (2019). Identification and Expression of NAC Transcription Factors of Vaccinium corymbosum L. in Response to Drought Stress. Forests, 10(12), 1088. https://doi.org/10.3390/f10121088



