Salinity Tolerance in Fraxinus angustifolia Vahl.: Seed Emergence in Field and Germination Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. Germination in Natural Environment
2.2. Germination Trials
2.3. Statistical Analysis
3. Results
3.1. Seed Germination in Natural Environment
3.2. Seed Viability and Embryo Development
3.3. Chilling and Salinity Effect in Germination
3.4. Recovery from Salt Stress
4. Discussion
4.1. Chilling Effect
4.2. Salinity Effect in Laboratory and in the Field
4.3. Recovery
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Caudullo, G.; Houston Durrant, T. Fraxinus angustifolia in Europe: distribution, habitat, usage and threats. In European Atlas of Forest Tree Species; San-Miguel Ayanz, J., De Rigo, D., Caudullo, G., Houston Durrant, T., Mauri, A., Eds.; Publication Office of the European Union: Luxembourg, 2016; p. 97. ISBN 978-92-79-36740-3. [Google Scholar]
- Raddi, S. Fraxinus angustifolia. In Enzyklopädie der Holzgewächse Handb. und Atlas der Dendrologie; Wiley-VCH Verlag GmbH & Co KGaA: Weinheim, Germany, 2014; pp. 1–18. [Google Scholar]
- Pinheiro, J.C.; Marques, C.R.; Pinto, G.; Bouguerra, S.; Mendo, S.; Gomes, N.C.; Gonçalves, F.; Rocha-Santos, T.; Duarte, A.C.; Roembke, J.; et al. The performance of Fraxinus angustifolia as a helper for metal phytoremediation programs and its relation to the endophytic bacterial communities. Geoderma 2013, 202–203, 171–182. [Google Scholar] [CrossRef]
- Rosselli, W.; Keller, C.; Boschi, K. Phytoextraction capacity of trees growing on a metal contaminated soil. Plant Soil 2003, 256, 265–272. [Google Scholar] [CrossRef]
- Baycu, G.B.; Tolunay, D.; Özden, H.; Günebakan, S. Ecophysiological and seasonal variations in Cd, Pb, Zn, and Ni concentrations in the leaves of urban deciduous trees in Istanbul. Environ. Pollut. 2006, 143, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Semenzato, P.; Cattaneo, D.; Dainese, M. Growth prediction for five tree species in an Italian urban forest. Urban For. Urban Green. 2011, 10, 169–176. [Google Scholar] [CrossRef]
- Roloff, A.; Korn, S.; Gillner, S. The Climate-Species-Matrix to select tree species for urban habitats considering climate change. Urban For. Urban Green. 2009, 8, 295–308. [Google Scholar] [CrossRef]
- Jaeger, C.; Gessler, A.; Biller, S.; Rennenberg, H. Differences in C metabolism of ash species and provenances as a consequence of root oxygen deprivation by waterlogging. J. Exp. Bot. 2009, 60, 4335–4345. [Google Scholar] [CrossRef]
- Miyamoto, S. Site Suitability Assessment for Irrigating Urban Landscapes with Water of Elevated Salinity in the Southwest Consolidated Final Report Part I. Water Quality and Plant Salt Tolerance; TWRI TR-416; Texas A&M University, AgriLife Research Center, Water Resources Institute: El Paso, TX, USA, 2011. [Google Scholar]
- Wu, L.; Guo, X.; Hunter, K.; Zagory, E.; Waters, R.; Brown, J. Studies of salt tolerance of landscape plant species and California native grasses for recycled water irrigation. Slosson Rep. 2001, 2000–2001, 1–14. [Google Scholar]
- Xie, Y.J.; Xu, S.; Han, B.; Wu, M.Z.; Yuan, X.X.; Han, Y.; Gu, Q.; Xu, D.K.; Yang, Q.; Shen, W.B. Evidence of Arabidopsis salt acclimation induced by up-regulation of HY1 and the regulatory role of RbohD-derived reactive oxygen species synthesis. Plant J. 2011, 66, 280–292. [Google Scholar] [CrossRef]
- European Commission. Interpretation Manual of European Union Habitats—EUR28; DG Environment B.3, Nature ENV.; European Commission: Brussels, Belgium, 2013; p. 144. [Google Scholar]
- Schwartz, S.S.; Jenkins, D.G. Temporary aquatic habitats: Constraints and opportunities. Acquat. Ecol. 2000, 34, 3–8. [Google Scholar] [CrossRef]
- Viciani, D.; Lastrucci, L.; Dell’Olmo, L.; Ferretti, G.; Foggi, B. Natura 2000 habitats in Tuscany (central Italy): Synthesis of main conservation features based on a comprehensive database. Biodivers. Conserv. 2014, 23, 1551–1576. [Google Scholar] [CrossRef]
- Gardner, R.C.; Barchiesi, S.; Beltrame, C.; Finlayson, C.; Galewski, T.; Harrison, I.; Paganini, M.; Perennou, C.; Pritchard, D.; Rosenqvist, A.; et al. State of the World’s Wetlands and Their Services to People: A Compilation of Recent Analyses; Ramsar Convention Secretariat: Gland, Switzerland, 2015. [Google Scholar]
- MedWet Secretariat. Wetlands for Sustainable Development in the Mediterranean Region. A Framework for Action 2016–2030, MedWet Secretariat: Palais de la Porte Dorée, Paris, France, 2016; ISBN 978-2-9554-6210-2.
- Ramsar Convention on Wetlands. Global Wetland Outlook: State of the World’s Wetlands and their Services to People; Dudley, N., Ed.; Ramsar Convention Secretariat: Gland, Switzerland, 2018. [Google Scholar]
- Davidson, N.C. How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar. Freshw. Res. 2014, 65, 934–941. [Google Scholar]
- Jones, A.; Panagos, P.; Barcelo, S.; Bouraoui, F.; Bosco, C.; Dewitte, O.; Gardi, C.; Erhard, M.; Hervás, J.; Hiederer, R.; et al. The State of Soil in Europe; European Commission JRC-IES: Ispra, Italy, 2012; ISBN 978-92-79-22806-3. [Google Scholar]
- Tsanis, I.K.; Daliakopoulos, I.N.; Koutroulis, A.G.; Karatzas, G.P.; Varouchakis, E.; Kourgialas, N. Soil salinization. In Soil Threats in Europe; JRC Technical Report. EUR 27607; Stolte, J., Tesfai, M., Øygarden, L., Kværnø, S., Keizer, J., Verheijen, F., Panagos, P., Ballabio, C., Hessel, R., Eds.; Publications Office of the European Union: Luxembourg, 2015; pp. 104–117. [Google Scholar]
- Daliakopoulos, I.N.; Tsanis, I.K.; Koutroulis, A.; Kourgialas, N.N.; Varouchakis, A.E.; Karatzas, G.P.; Ritsema, C.J. The threat of soil salinity: A European scale review. Sci. Total Environ. 2016, 573, 727–739. [Google Scholar] [CrossRef]
- Lefebvre, G.; Redmond, L.; Germain, C.; Palazzi, E.; Terzago, S.; Willm, L.; Poulin, B. Predicting the vulnerability of seasonally-flooded wetlands to climate change across the Mediterranean Basin. Sci. Total Environ. 2019, 692, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Juffe-Bignoli, D.; Burgess, N.D.; Bingham, H.; Belle, E.M.; De Lima, M.G.; Deguignet, M.; Bertzky, B.; Milam, A.N.; Martinez-Lopez, J.; Lewis, E.; et al. Protected Planet Report 2014; UNEP-WCMC: Cambridge, UK, 2014; ISBN 978-92-807-3416-4. [Google Scholar]
- Rundel, P.W. Landscape disturbance in Mediterranean-type ecosystems: An overview. In Landscape Disturbance and Biodiversity in Mediterranean-Type Ecosystems; Rundel, P.W., Montenegro, G., Jaksic, F.M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 3–22. ISBN 978-3-662-03543-6. [Google Scholar]
- Herbert, E.R.; Boon, P.; Burgin, A.J.; Neubauer, S.C.; Franklin, R.B.; Ardón, M.; Hopfensperger, K.N.; Lamers, L.P.M.; Gell, P. A global perspective on wetland salinization: Ecological consequences of a growing threat to freshwater wetlands. Ecosphere 2015, 6, art206. [Google Scholar] [CrossRef]
- Geijzendorffer, I.R.; Beltrame, C.; Chazee, L.; Gaget, E.; Galewski, T.; Guelmami, A.; Perennou, C.; Popoff, N.; Guerra, C.A.; Leberger, R.; et al. A more effective Ramsar Convention for the conservation of Mediterranean wetlands. Front. Ecol. Evol. 2019, 7, 1–6. [Google Scholar] [CrossRef]
- Zedler, J.B. What’s new in adaptive management and restoration of coasts and estuaries? Estuaries Coasts 2017, 40, 1–21. [Google Scholar] [CrossRef]
- Hoepffner, N.; Dowell, M.D.; Edwards, M.; Fonda-Umano, S.; Green, D.R.; Greenaway, B.; Hansen, B.; Heinze, C.; Leppänen, J.-M.; Lipiatou, E.; et al. Marine and Coastal Dimension of Climate Change in Europe Marine and Coastal Dimension of Climate Change in Europe; EUR 22554 EN; Hoepffner, N., Ed.; Institute for Environment and Sustainability: Ispra, Italy, 2006; ISBN 9279037471. [Google Scholar]
- IPCC. Summary for policymakers. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Field, C.B., Barros, V.R., Dokken, D.J., Mach, K.J., Mastrandrea, M.D., Bilir, T.E., Chatterjee, M., Ebi, K.L., Estrada, Y.O., Genova, R.C., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- Amores, M.J.; Verones, F.; Raptis, C.; Juraske, R.; Pfister, S.; Stoessel, F.; Antón, A.; Castells, F.; Hellweg, S. Biodiversity impacts from salinity increase in a coastal wetland. Environ. Sci. Technol. 2013, 47, 6384–6392. [Google Scholar] [CrossRef] [PubMed]
- EEA Natura. Network Viewer. 2000. Available online: http://natura2000.eea.europa.eu/# (accessed on 10 August 2019).
- Levin, N.; Elron, E.; Gasith, A. Decline of wetland ecosystems in the coastal plain of Israel during the 20th century: Implications for wetland conservation and management. Landsc. Urban Plan. 2009, 92, 220–232. [Google Scholar] [CrossRef]
- Cosentino, B.; Colleges, W.S. Dispersal and wetland fragmentation. In The Wetland Book: I: Structure and Function, Management, and Methods; Finlayson, C.M., Everard, M., Irvine, K., McInnes, R.J., Middleton, B.A., van Dam, A.A., Davidson, N.C., Eds.; Springer: Dordrecht, The Netherlands, 2018; pp. 105–111. ISBN 9789048196593. [Google Scholar]
- Ghelardini, L.; Migliorini, D.; Santini, A.; Pepori, A.L.; Maresi, G.; Vai, N.; Montuschi, C.; Carrari, E.; Feducci, M.; Capretti, P.; et al. From the Alps to the Apennines: possible spread of ash dieback in Mediterranean areas. In Dieback of European Ash (Fraxinus spp.): Consequences and Guidelines for Sustainable Management; The Report on European Cooperation in Science & Technology (COST) Action FP1103 FRAXBACK; Vasaitis, R., Enderle, R., Eds.; SLU Service/Repro: Uppsala, Sweden, 2017; pp. 140–149. ISBN 978-91-576-8696-1. [Google Scholar]
- Ortega, M.; Velasco, J.; Millán, A.; Guerrero, C. An ecological integrity index for littoral wetlands in agricultural catchments of semiarid mediterranean regions. Environ. Manag. 2004, 33, 412–430. [Google Scholar] [CrossRef]
- Schuerch, M.; Spencer, T.; Temmerman, S.; Kirwan, M.L.; Wolff, C.; Lincke, D.; McOwen, C.J.; Pickering, M.D.; Reef, R.; Vafeidis, A.T.; et al. Future response of global coastal wetlands to sea-level rise. Nature 2018, 561, 231–234. [Google Scholar] [CrossRef]
- Fini, A.; Guidi, L.; Giordano, C.; Baratto, M.C.; Ferrini, F.; Brunetti, C.; Calamai, L.; Tattini, M. Salinity stress constrains photosynthesis in Fraxinus ornus more when growing in partial shading than in full sunlight: Consequences for the antioxidant defence system. Ann. Bot. 2014, 114, 525–538. [Google Scholar] [CrossRef][Green Version]
- Hoeppner, S.S.; Shaffer, G.P.; Perkins, T.E. Through droughts and hurricanes: Tree mortality, forest structure, and biomass production in a coastal swamp targeted for restoration in the Mississippi River Deltaic Plain. Forest 2008, 256, 937–948. [Google Scholar] [CrossRef]
- Conner, W.H.; Mcleod, K.W.; Mccarron, J.K. Flooding and salinity effects on growth and survival of four common. Wetl. Ecol. Manag. 1997, 5, 99–109. [Google Scholar] [CrossRef]
- Bauhus, J.; Forrester, D.I.; Pretzsch, H.; Felton, A.; Pyttel, P.; Benneter, A. Silvicultural Options for Mixed-Species Stands. In Mixed-Species Forests; Pretzsch, H., Forrester, D.I., Bauhus, J., Eds.; Springer-Verlag GmbH: Berlin, Germany, 2017; pp. 433–501. ISBN 978-3-662-54551-5. [Google Scholar]
- Fady, B.; Aravanopoulos, F.A.; Alizoti, P.; Mátyás, C.; von Wühlisch, G.; Westergren, M.; Belletti, P.; Cvjetkovic, B.; Ducci, F.; Huber, G.; et al. Forest Ecology and Management Evolution-based approach needed for the conservation and silviculture of peripheral forest tree populations. For. Ecol. Manag. 2016, 375, 66–75. [Google Scholar] [CrossRef]
- Barducci, A.; Guzzi, D.; Marcoionni, P.; Pippi, I.; Raddi, S. PROBA contribution to wetland monitoring in the coastal zone of San Rossore Natural Park. In Proceedings of the European Space Agency, (Special Publication) ESA SP, Montreux, Switzerland, 23–27 April 2007. [Google Scholar]
- Bertacchi, A.; Lombardi, T.; Tomei, P.E. Le aree umide salmastre della Tenuta di San Rossore (PI): Zonazione e successione delle specie vegetali in relazione alla salinità del suolo. INTER NOS 2007, 1, 1–14. [Google Scholar]
- Sani, A.; Tomei, P.E. La vegetazione psammofila del litorale di San Rossore (Toscana settentrionale) e la sua importanza conservazionistica. Parlatorea 2006, 8, 99–119. [Google Scholar]
- Bertacchi, A. Dune habitats of the Migliarino—San Rossore—Massaciuccoli Regional Park (Tuscany-Italy). J. Maps 2017, 13, 322–331. [Google Scholar] [CrossRef]
- Rhoades, J.D.; Chanduvi, F.; Lesch, S. Soil salinity assessment. In Methods and Interpretation of Electrical Conductivity Measurements; FAO Irrigation and Darinage: Rome, Italy, 1999; ISBN 92-5-104281-0. [Google Scholar]
- ISO. Soil Quality—Determination of pH; ISO 10390; ISO: Geneva, Switzerland, 2005. [Google Scholar]
- RStudio Team. RStudio: Integrated Development for R. RStudio, Inc.: Boston, MA, USA; Available online: http://www.rstudio.com/ (accessed on 1 February 2019).
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality (complete sample). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Royston, P. Remark AS R94: A Remark on Algorithm AS 181: The W-test for Normality. J. R. Stat. Soc. Ser. C (Appl. Stat.) 1995, 44, 547–551. [Google Scholar] [CrossRef]
- Wilcoxon, F. Individual comparisons by ranking methods. Biom. Bull. 1945, 1, 80–83. [Google Scholar] [CrossRef]
- Canty, A.; Ripley, B.; Bootstrap Functions. Package ‘boot’ R—Project. Available online: https://cran.r-project.org/web/packages/boot/boot.pdf (accessed on 23 October 2019).
- Canty, A.J. Resampling Methods in R: The boot Package. Newsl. R. Proj. 2002, 2, 2–7. [Google Scholar]
- ISTA. ISTA Working Sheets on Tetrazolium Testing 2003, 1st ed.; Including Supplements 2011; Testing, I.S., Ed.; ISTA: Bassersdorf, Switzerland, 2011; Volume 2, ISBN 3906549402. [Google Scholar]
- ISTA. International Rules for Seed Testing.; International Seed Testing Association: Bassersdorf, Switzerland, 2017; ISBN 2310-3655. [Google Scholar]
- Magini, E. Forest seed handling, equipment and procedures. Unasylva 1962, 64, 1–22. [Google Scholar]
- Bewley, J.D.; Bradford, K.J.; Hilhorst, H.W.M.; Nonogaki, H. Seeds: Physiology of Development, GERMINATION and dormancy, 3rd ed.; Springer: New York, NY, USA; Berlin/Heidelberg, Germany; Dordrecht, The Netherlands; London, UK, 2013; ISBN 9781461446934. [Google Scholar]
- Klein, J.P.; van Houwelingen, H.C.; Ibrahim, J.G.; Scheike, T.H. Handbook of Survival Analysis; CRC Press: Boca Raton, FL, USA; Taylor and Francis Group: Abingdon, UK, 2016; ISBN 9781466555679. [Google Scholar]
- Muenchow, G. Ecological use of failure time analysis. Ecology 1986, 67, 246–250. [Google Scholar] [CrossRef]
- 60. Kalbfleish, J.D.; Prentice, R.L. The Statistical Analysis Failure Time Data, 2nd ed.Kalbfleish, J.D., Prentice, R.L., Eds.; John Wiley & Sons: New York, NY, USA, 2011; ISBN 9781118031230. [Google Scholar]
- McNair, J.N.; Sunkara, A.; Frobish, D. How to analyse seed germination data using statistical time-to-event analysis: Non-parametric and semi-parametric methods. Seed Sci. Res. 2012, 22, 77–95. [Google Scholar] [CrossRef]
- Kaplan, E.L.; Meier, P. Nonparametric estimation from incomplete samples. J. ASA 1958, 53, 457–481. [Google Scholar]
- Nelson, W. Hazard plotting for incomplete failure data. J. Qual. Technol. 1969, 1, 27–52. [Google Scholar] [CrossRef]
- Nelson, W. Theory and applications of hazard plotting for censored failure data. Technometrics 1972, 14, 945–966. [Google Scholar] [CrossRef]
- Aalen, O. Nonparametric inference for a family of counting processes. Ann. Stat. 1978, 6, 701–726. [Google Scholar] [CrossRef]
- Zhou, M. Empirical Likelihood Method in Survival Analysis. Zhou, M., Ed.; Chapman and Hall, CRC: London, UK; New York, NY, USA, 2015; ISBN 9781466554931. [Google Scholar]
- Thomas, D.R.; Grunkemeier, G.L. Confidence interval estimation of survival probabilities for censored data. J. Am. Stat. Assoc. 1975, 70, 865–871. [Google Scholar] [CrossRef]
- Strobl, R.; Verbeke, T. Confidence Intervals for the Kaplan-Meier Estimator. Package ‘km.ci’ for RStudio. Available online: https://cran.r-project.org/web/packages/km.ci/km.ci.pdf (accessed on 23 October 2019).
- Allison, P.D. When Can You Safely Ignore Multicollinearity? Available online: https://statisticalhorizons.com/multicollinearity (accessed on 15 October 2019).
- Gerbing, D.W. R Data Analysis without Programming. Group, R.T.& F.: New York, NY, USA; London, UK, 2014; ISBN 9781317931669. [Google Scholar]
- Cox, D.R. Regression models and life-tables. J. R. Stat. Soc. Ser. B 1972, 34, 187–220. [Google Scholar] [CrossRef]
- Therneau, T.M.; Survival Analysis. RStudio Package ‘survival’. Available online: https://cran.r-project.org/web/packages/survival/survival.pdf (accessed on 23 October 2019).
- Grambsch, P.M.; Therneau, T.M. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 1994, 81, 515–526. [Google Scholar] [CrossRef]
- Hess, K.R. Graphical methods for assessing violations of the proportional hazards assumption in Cox regression. Stat. Med. 1995, 14, 1707–1723. [Google Scholar] [CrossRef]
- Ripley, B.; Venables, B.; Bates, D.M.; Hornik, K.; Gebhardt, A.; Firth, D. Package ‘MASS.’. Available online: https://cran.r-project.org/web/packages/MASS/MASS.pdf (accessed on 23 October 2019).
- Chen, B.E.; Jiang, W.; Tu, D. A hierarchical Bayes model for biomarker subset effects in clinical trials. Comput. Stat. Data Anal. 2014, 71, 324–334. [Google Scholar] [CrossRef]
- Fang, T.; Mackillop, W.; Jiang, W.; Hildesheim, A.; Wacholder, S.; Chen, B.E. A Bayesian method for risk window estimation with application to HPV vaccine trial. Comput. Stat. Data Anal. 2017, 112, 53–62. [Google Scholar] [CrossRef]
- Baskin, C.C.; Baskin, J.M. Seeds–Ecology, Biogeography, and, Evolution of Dormancy and Germination, 2nd ed.; Academic Press: San Diego, CA, USA, 2014; ISBN 978-0-12-416677-6. [Google Scholar]
- Piotto, B.; Di Noi, A. Seed Propagation of Mediterranean TREES and shrubs. APAT, Ed.; IGER SRL: Roma, Italy, 2003; ISBN 88-448-0081-0. [Google Scholar]
- Carta, A.; Bedini, G.; Mu, J.V. Comparative seed dormancy and germination of eight annual species of ephemeral wetland vegetation in a Mediterranean climate. Plant Biol. 2013, 214, 339–349. [Google Scholar] [CrossRef]
- Carta, A.; Hanson, S.; Müller, J.V. Plant regeneration from seeds responds to phylogenetic relatedness and local adaptation in Mediterranean Romulea (Iridaceae) species. Ecol. Evol. 2016, 6, 4166–4178. [Google Scholar] [CrossRef]
- Raquin, C.; Jung-Muller, B.; Dufour, J.; Frascaria-Lacoste, N. Rapid seedling obtaining from European ash species Fraxinus excelsior (L.) and Fraxinus angustifolia (Vahl.). Ann. For. Sci. 2002, 59, 219–224. [Google Scholar]
- Lombardo, G.; Scialabba, A.; Schicchi, R. Seed cryopreservation of Fraxinus angustifolia Vahl. Afr. J. Biotechnol. 2013, 12, 1930–1936. [Google Scholar]
- French, D.; Meilan, R. Germination trials for Asian and North American ash species. Tree Plant Notes 2013, 56, 27–34. [Google Scholar]
- Piotto, B.; Piccini, C. Influence of pretreatment and temperature on the germination of Fraxinus angustifolia seeds. Seed Sci. Technol. 1998, 26, 799–812. [Google Scholar]
- Cabra-Rivas, I.; Castro-Díez, P. Potential germination success of exotic and native trees coexisting in central Spain riparian forests. Int. J. Ecol. 2016, 2016, 7614683. [Google Scholar] [CrossRef]
- Gérard, P.R.; Fernández-Manjarrés, J.F.; Bertolino, P.; Dufour, J.; Raquin, C.; Frascaria-Lacoste, N. New insights in the recognition of the European ash species Fraxinus excelsior L. and Fraxinus angustifolia Vahl as useful tools for forest management. Ann. For. Sci. 2006, 63, 733–738. [Google Scholar] [CrossRef][Green Version]
- Tilki, F.; Çiçek, E. Effects of stratification, temperature and storage on germination in three provenances of Fraxinus angustifolia subsp. oxycarpa seeds. Turk. J. Agric. For. 2005, 29, 323–330. [Google Scholar]
- He, H.; De Souza Vidigal, D.; Basten Snoek, L.; Schnabel, S.; Nijveen, H.; Hilhorst, H.; Bentsink, L. Interaction between parental environment and genotype affects plant and seed performance in Arabidopsis. J. Exp. Bot. 2014, 65, 6603–6615. [Google Scholar] [CrossRef] [PubMed]
- Née, G.; Xiang, Y.; Soppe, W.J.J. The release of dormancy, a wake-up call for seeds to germinate. Curr. Opin. Plant Biol. 2017, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tian, C.; Wang, L. Cold stratification pretreatment improves salinity tolerance in two wheat varieties during germination. Seed Sci. Technol. 2018, 46, 87–92. [Google Scholar] [CrossRef]
- Meng, H.; Wang, X.; Tong, S.; Lu, X.; Hao, M. Seed germination environments of Typha latifolia and Phragmites australis in wetland restoration. Ecol. Eng. 2016, 96, 194–199. [Google Scholar]
- Bungard, R.A.; McNeil, D.; Morton, J.D. Effects of chilling, light and nitrogen-containing compounds on germination, rate of germination and seed imbibition of Clematis vitalba L. Ann. Bot. 1997, 79, 643–650. [Google Scholar] [CrossRef]
- Chen, S.; Chou, S.; Tsai, C.; Hsu, W.; Baskin, C.C.; Baskin, J.M.; Chien, C.; Kuo-Huang, L. Plant physiology and biochemistry effects of moist cold stratification on germination, plant growth regulators, metabolites and embryo ultrastructure in seeds of Acer morrisonense (Sapindaceae). Plant Physiol. Biochem. 2015, 94, 165–173. [Google Scholar] [CrossRef]
- Miransari, M.; Smith, D.L. Plant hormones and seed germination. Environ. Exp. Bot. 2014, 99, 110–121. [Google Scholar] [CrossRef]
- Gai, Y.P.; Li, X.Z.; Ji, X.L.; Wu, C.A.; Yang, G.D.; Zheng, C. Chilling stress accelerates degradation of seed storage protein and photosynthetic protein during cotton seed germination. J. Agron. Crop Sci. 2008, 194, 278–288. [Google Scholar] [CrossRef]
- Einali, A.L.I.R.; Sadeghipour, H.R. Alleviation of dormancy in walnut kernels by moist chilling is independent from storage protein mobilization. Tree Physiol. 2007, 27, 519–525. [Google Scholar] [CrossRef]
- Peel, M.D.; Waldron, B.L.; Jensen, K.B.; Chatterton, N.J.; Horton, H.; Dudley, L.M. Screening for salinity tolerance in alfalfa: A repeatable method. Crop Sci. 2004, 44, 2049–2053. [Google Scholar] [CrossRef]
- Rajjou, L.; Duval, M.; Gallardo, K.; Catusse, J.; Bally, J.; Job, C.; Job, D. Seed germination and vigor. Annu. Rev. Plant Biol. 2012, 63, 507–533. [Google Scholar] [CrossRef] [PubMed]
- Zedler, J.B. Wetlands at your service: Reducing impacts of agriculture at the watershed scale. Front. Ecol. Environ. 2003, 1, 65–72. [Google Scholar] [CrossRef]
- Shumway, S.W.; Bertness, M. Salt stress limitation of seedling recruitment in a salt marsh plant community. Oecologia 1992, 92, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Ungar, I.A. Halophyte seed germination. Bot. Rev. 1978, 44, 233–264. [Google Scholar] [CrossRef]
- Grigore, M.-N. Romanian Salt Tolerant Plants: Taxonomy and Ecology; TEHNOPRESS: Iasi, Romania, 2012; ISBN 978-973-702-923-2. [Google Scholar]
- González, M.B. Adaptation of halophytes to different habitats. In Seed Dormancy and Germination; IntechOpen: London, UK, 2019; pp. 1–23. [Google Scholar]
- Grigore, M.-N.; Toma, C. Anatomical Adaptations of Halophytes; Springer International Publishing: Cham, Switzerland, 2017; ISBN 978-3-319-66480-4. [Google Scholar]
- Liang, W.; Ma, X.; Wan, P.; Liu, L. Plant salt-tolerance mechanism: A review. Biochem. Biophys. Res. Commun. 2018, 495, 286–291. [Google Scholar] [CrossRef]
- Läuchli, A.; Grattan, S.R. Plant growth and development under salinity stress. In Advances in Molecular Breeding Toward Drought and Salt Tolerant Crops; Jenks, M.A., Hasegawa, P.M., Jain, S.M., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 1–32. [Google Scholar]
- Polle, A.; Rennenberg, H. Physiological responses to abiotic and biotic stress in forest trees. Forests 2019, 10, 711. [Google Scholar] [CrossRef]
- Kozlowski, T.T. Responses of woody plants to flooding and salinity. Tree Physiol. Monogr. 1997, 1, 1–29. [Google Scholar] [CrossRef]
- Kozlowski, T.T. Responses of woody plants to human-induced environmental stresses: Issues, problems, and strategies for alleviating stress. CRC Crit. Rev. Plant Sci. 2000, 19, 91–170. [Google Scholar] [CrossRef]
- Gotelli, N.J.; Ellison, A.M. A Primer of Ecological Statistics; Sinauer: Sunderland, MA, USA, 2013; ISBN1 1605350648. ISBN2 9781605350646. [Google Scholar]
- Esechie, H.A.; Al-Saidi, A.; Al-Khanjari, S. Effect of sodium chloride salinity on seedling emergence in chickpea. J. Agron. Crop Sci. 2002, 188, 155–160. [Google Scholar] [CrossRef]
- Sosa, L.; Llanes, A.; Reinoso, H.; Reginato, M.; Luna, V. Osmotic and specific ion effexts on the germination of Prosopis strobulifera. Ann. Bot. 2005, 96, 261–267. [Google Scholar] [CrossRef]
- Hong, L.; Ping, C.; ZhaoHui, Z.; ShouWen, S.; PiJun, L.; BaoQing, W. Seeds germination of three common afforestating trees under salt and drought stress in Xinjiang. Acta Bot. Boreali-Occidentalia Sin. 2011, 31, 1466–1473. [Google Scholar]
- Zhang, Z.; Yu, F. Effects of Salt Stress on Seed Germination of Four Ornamental Non-halophyte Effects of Salt Stress on Seed Germination of Four Ornamental Non- halophyte Species. Int. J. Agric. Biol. 2019, 21, 47–53. [Google Scholar]
- Tonon, G.; Kevers, C.; Faivre-Rampant, O.; Graziani, M.; Gaspar, T. Effect of NaCl and mannitol iso-osmotic stresses on proline and free polyamine levels in embryogenic Fraxinus angustifolia callus. J. Plant Physiol. 2004, 161, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Llanes, A.; Andrade, A.; Masciarelli, O.; Alemano, S.; Luna, V. Drought and salinity alter endogenous hormonal profiles at the seed germination phase. Seed Sci. Res. 2016, 26, 1–13. [Google Scholar] [CrossRef]
- Macovei, A.; Pagano, A.; Leonetti, P.; Carbonera, D.; Balestrazzi, A.; Araújo, S.S. Systems biology and genome-wide approaches to unveil the molecular players involved in the pre-germinative metabolism: Implications on seed technology traits. Plant Cell Rep. 2017, 36, 669–688. [Google Scholar] [CrossRef]
- Shen, X.; Wang, Z.; Song, X.; Xu, J.; Jiang, C.; Zhao, Y.; Ma, C.; Zhang, H. Transcriptomic profiling revealed an important role of cell wall remodeling and ethylene signaling pathway during salt acclimation in Arabidopsis. Plant Mol. Biol. 2014, 86, 303–317. [Google Scholar] [CrossRef]
- Sivritepe, N.; Sivritepe, H.O.; Eris, A. The effects of NaCl priming on salt tolerance in melon seedlings grown under saline conditions. Sci. Hortic. (Amsterdam) 2003, 97, 229–237. [Google Scholar] [CrossRef]
- Bajji, M.; Kinet, J.; Lutts, S. Osmotic and ionic effects of NaCl on germination, early seedling growth, and ion content of Atriplex halimus (Chenopodiaceae). Can. J. Bot. 2002, 80, 297–304. [Google Scholar] [CrossRef]
- Ievinsh, G. Biological basis of biological diversity: Physiological adaptations of plants to heterogeneous habitats along a sea coast. Acta Univ. Latv. 2006, 710, 53–79. [Google Scholar]
- Zhu, J.K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef]
- Zhu, J.K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
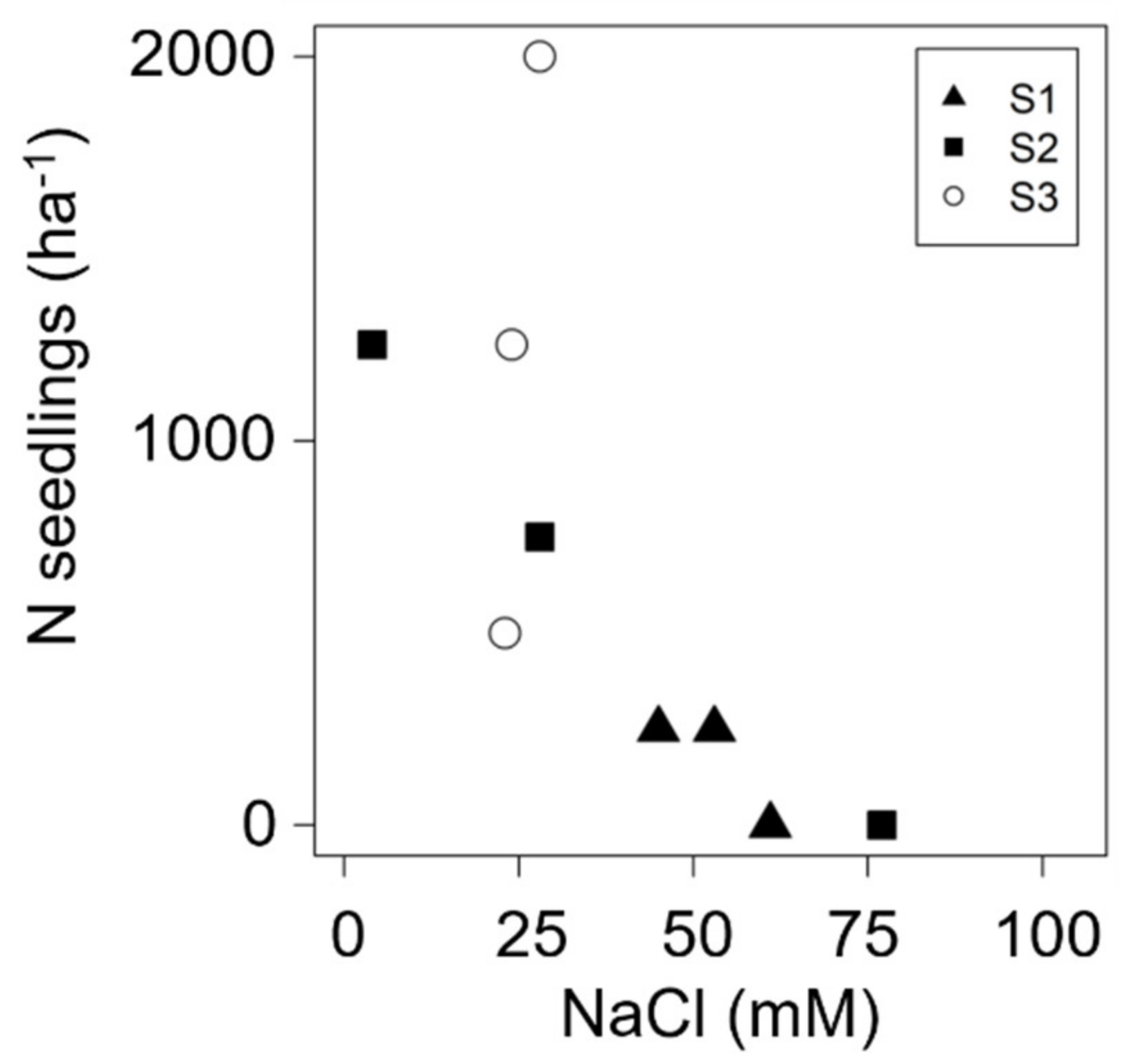
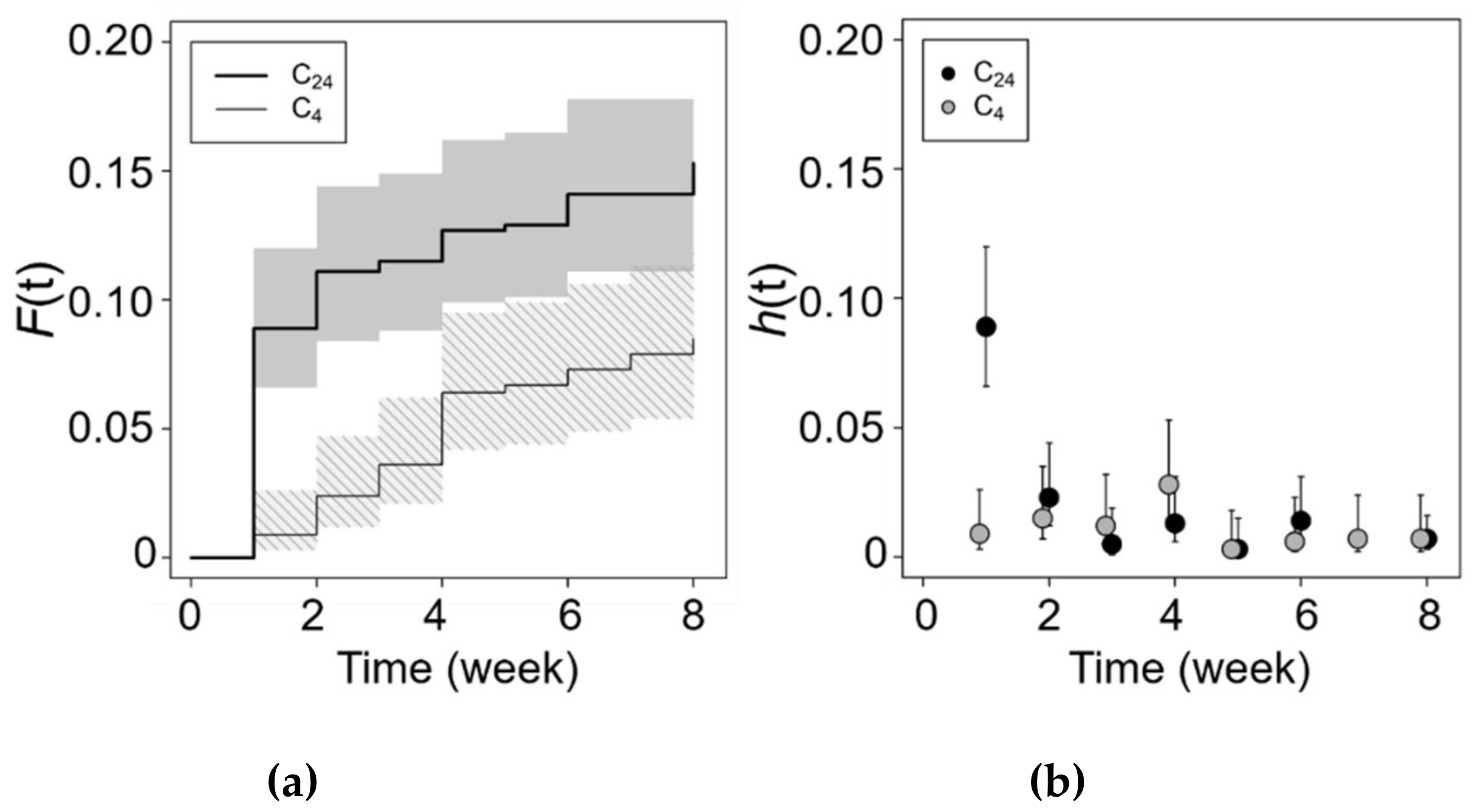
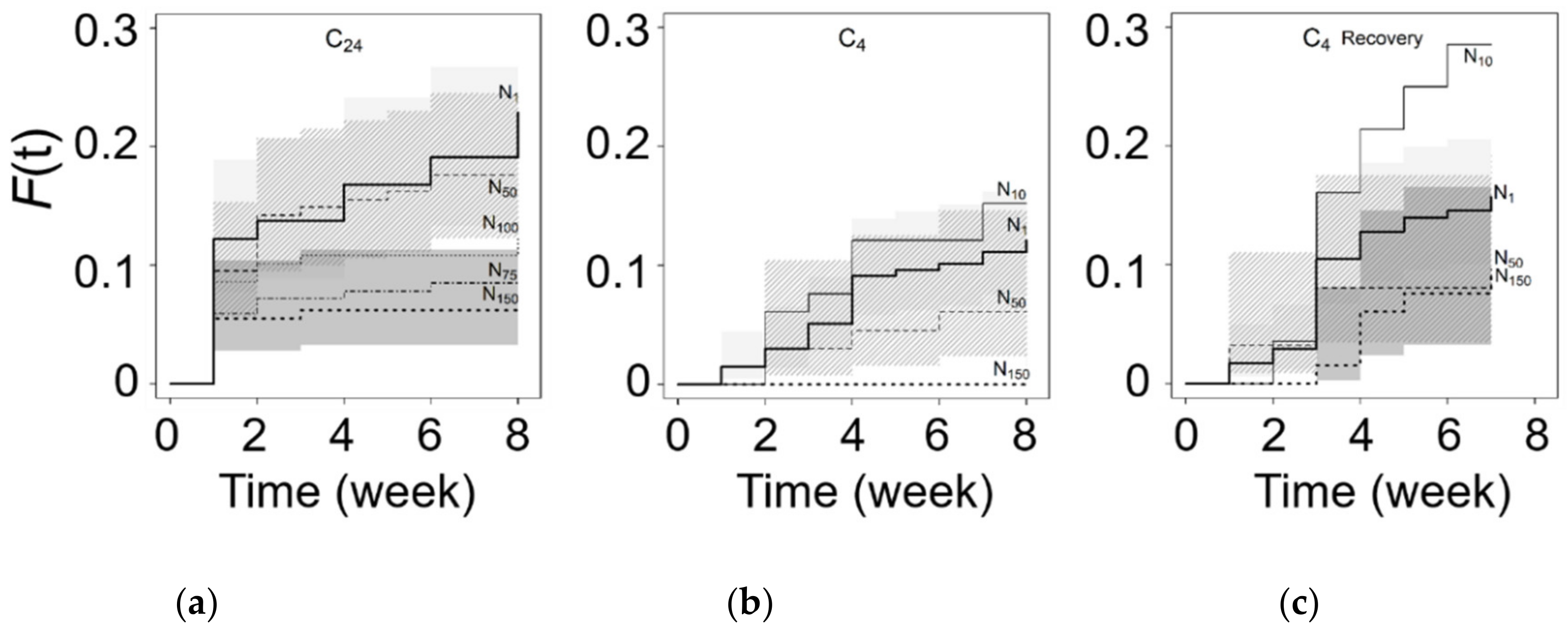

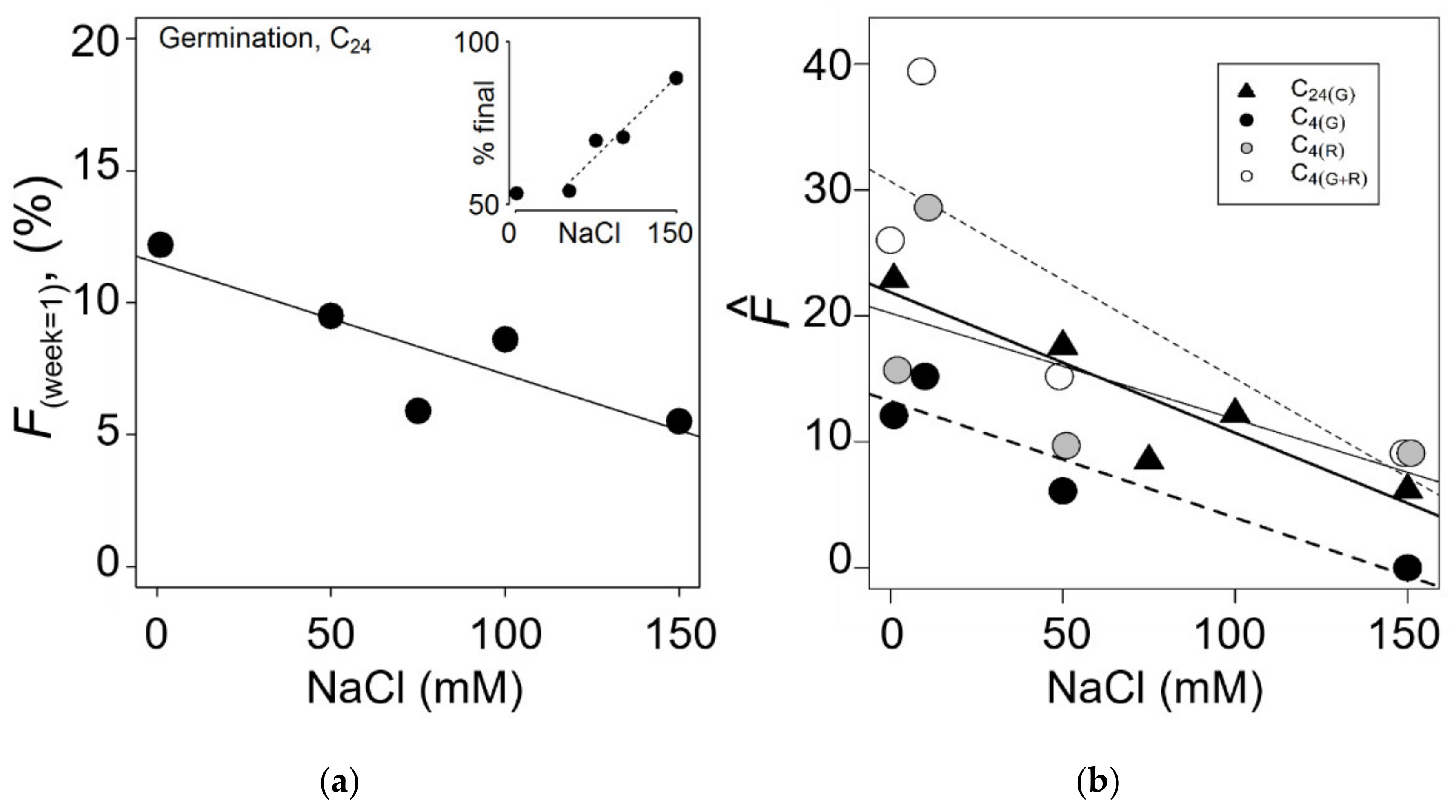
| Site | (m) | N | Minimun–Maximum | Mean (SE) | 95% CI | W | ECe | % | |
|---|---|---|---|---|---|---|---|---|---|
| soil (mM NaCl) | |||||||||
| S1 | 890 | 30 | 32–95 | 55 (2.6) | a1 | 50–61 | 0.345 | 3.1–10.2 | 96 |
| S2 | 1210 | 30 | 3–102 | 38 (5.6) | b | 28–50 | 0.008 | 4.1–11.2 | 45 |
| S3 | 1650 | 30 | 18–41 | 27 (0.8) | b | 25–29 | 0.009 | 2.7–4.4 | 7 |
| Total | 1250 | 90 | 3–102 | 40 (2.4) | 35–45 | 0.001 | 2.7–11.2 | 48 | |
| seedlings (ha−1) | |||||||||
| S1 | 3 | 0–500 | 167 (96) | b1 | 0–333 | 0.001 | |||
| S2 | 3 | 0–1500 | 667 (226) | ab | 167–1000 | 0.415 | |||
| S3 | 3 | 0–2500 | 1250 (307) | a | 583–1750 | 0.751 | |||
| Total | 9 | 0–2500 | 694 (168) | 389–1028 | 0.010 | ||||
| Trial | Zi | PH p | Wald p 1 | ′bhm′ p | Cut off Values (95% CI) |
|---|---|---|---|---|---|
| Complete dataset | |||||
| G | nacl | 0.930 | 0.016 | 0.011 | 1–50 (1-1) – (50-50) |
| chilling | 0.004 | 0.011 | 0.008 | ||
| interaction | 0.526 | 0.155 | 0.218 | ||
| 1st week excluded | |||||
| G | nacl | 0.559 | 0.030 | 0.022 | 1–50 (1-1) – (50-50) |
| chilling | 0.383 | 0.830 | 0.585 | ||
| interaction | 0.904 | 0.639 | 0.644 | ||
| C24 | |||||
| G | nacl | 0.014 | 0.000 | 0.000 | 1–75 (1-1) – (75-75) |
| C4 | |||||
| G | nacl | 0.683 | 0.009 | 0.005 | 1–50 (1-1) – (50-50) |
| R | nacl | 0.104 | 0.057 | 0.021 | 10–50 (1-10) – (50-150) |
| G + R | nacl | 0.010 | 0.001 | 0.000 | 1–50 (1-1) – (10-150) |
| Model 1 | df | LLV 2 | AIC | BIC | LRT p-Value |
|---|---|---|---|---|---|
| Complete dataset (Germination) | |||||
| nacl + chilling + nacl × chilling1a | 3 | −906 a | 1818 3 | 1833 3 | 0.001, 0.011, 0.092 |
| nacl + chilling | 2 | −907 a | 1819 4 | 18294 | 0.000, 0.000 |
| Nacl | 1 | −916 b | 1833 | 1838 | 0.000 |
| Chilling | 1 | −923 d | 1848 | 1853 | 0.047 |
| nacl × chilling | 1 | −922 c | 1846 | 1850 | 0.015 |
| None | 0 | −925 e | 1850 | 1850 | |
| 1st week excluded (Germination) | |||||
| nacl + chilling + nacl × chilling1b | 3 | −473 a | 952 3 | 967 3 | 0.003, 0.830, 0.626 |
| nacl + chilling | 2 | −473 a | 951 4 | 960 4 | 0.000, 0.605 |
| nacl | 1 | −473 a | 949 | 954 | 0.000 |
| chilling | 1 | −489 c | 981 | 986 | 0.034 |
| Nacl × chilling | 1 | −479 b | 959 | 964 | 0.000 |
| none | 0 | −492 d | 983 | 983 | |
| C24 (Germination). | |||||
| nacl | 1 | −609 a | 1220 | 1225 | 0.000 |
| none | 0 | −618 b | 1236 | 1236 | |
| C4 (Germination) | |||||
| nacl | 1 | −218 a | 437 | 442 | 0.000 |
| none | 0 | −215 b | 451 | 451 | |
| C4 (Recovery). | |||||
| nacl | 1 | −317 a | 635.1 | 637.4 | 0.037 |
| none | 0 | −319 b | 637.4 | 640.1 | |
| C4 (Germination + Recovery) | |||||
| Nacl | 1 | −559 a | 1120 | 1125 | 0.000 |
| none | 0 | −567 b | 1134 | 1134 | |
| chilling | nacl | (CI Range) | (CI Range) | RR | |
|---|---|---|---|---|---|
| Germination | |||||
| C24 | 1 | 22.9 (16.5–30.8) | 25.0 (17.1–35.9) | a | 1 |
| 50 | 17.6 (12.3–24.5) | 18.7 (12.5–27.5) | ab | 0.8 | |
| 75 | 8.5 (5.0–14.0) | 8.7 (5.0–14.9) | c | 0.4 | |
| 100 | 12.2 (7.8–18.7) | 12.6 (7.7–20.4) | bc | 0.5 | |
| 150 | 6.2 (3.3–11.3) | 6.2 (3.2–19.1) | c | 0.3 | |
| 1+50+150 | 15.3 (12.2–19.0) | 16.1 (12.5–20.6) | |||
| C4 | 1 | 12.1 (8.3–17.4) | 12.8 (8.5–19.0) | a | 1 |
| 10 | 15.2 (8.4–25.7) | 16.0 (8.5–29.4) | a | 1.3 | |
| 50 | 6.1 (2.4–14.6) | 6.2 (2.4–15.7) | a | 0.5 | |
| 150 | 0.0 | 0.0. | b | 0 | |
| 1+50+150 | 8.5 (5.9–12.0) | 8.8 (6.1–12.7) | |||
| Recovery | |||||
| C4 | 1 | 15.7 (11.0–21.9) | 16.7 (11.3–24.3) | b | 1 |
| 10 | 28.6 (18.4–41.5) | 32.2 (19.1–52.3) | a | 1.8 | |
| 50 | 9.7 (4.5–19.6) | 10.0 (4.5–21.6) | b | 0.6 | |
| 150 | 9.1 (4.2–18.5) | 9.4 (4.2–20.3) | b | 0.6 | |
| 1+50+150 | 13.0 (9.7–17.3) | 13.4 (9.9–18.7) | |||
| Germination + Recovery | |||||
| C4 | 1 | 26.0 (20.4–32.6) | 29.6 (22.2–38.9) | ab | 1 |
| 10 | 39.4 (28.5–51.5) | 48.0 (31.7–70.4) | a | 1.5 | |
| 50 | 15.2 (8.4–25.7) | 16.2 (8.6–29.5) | bc | 0.6 | |
| 150 | 9.1 (4.2–18.4) | 9.4 (4.2–20.3) | c | 0.3 | |
| 1+50+150 | 20.4 (16.4–25.1) | 22.5 (17.6–28.6) | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raddi, S.; Mariotti, B.; Martini, S.; Pierguidi, A. Salinity Tolerance in Fraxinus angustifolia Vahl.: Seed Emergence in Field and Germination Trials. Forests 2019, 10, 940. https://doi.org/10.3390/f10110940
Raddi S, Mariotti B, Martini S, Pierguidi A. Salinity Tolerance in Fraxinus angustifolia Vahl.: Seed Emergence in Field and Germination Trials. Forests. 2019; 10(11):940. https://doi.org/10.3390/f10110940
Chicago/Turabian StyleRaddi, Sabrina, Barbara Mariotti, Sofia Martini, and Alberto Pierguidi. 2019. "Salinity Tolerance in Fraxinus angustifolia Vahl.: Seed Emergence in Field and Germination Trials" Forests 10, no. 11: 940. https://doi.org/10.3390/f10110940
APA StyleRaddi, S., Mariotti, B., Martini, S., & Pierguidi, A. (2019). Salinity Tolerance in Fraxinus angustifolia Vahl.: Seed Emergence in Field and Germination Trials. Forests, 10(11), 940. https://doi.org/10.3390/f10110940





