Vulnerability of Conifer Regeneration to Spruce Budworm Outbreaks in the Eastern Canadian Boreal Forest
Abstract
:1. Introduction
- (i)
- Balsam fir will be more affected by SBW than black spruce due to the phenological synchrony of balsam fir with SBW.
- (ii)
- Taller seedlings will have a higher level of defoliation due to the sheltering effect provided to smaller seedlings.
- (iii)
- Defoliation will be more intense as the distance between seedling and residual forest increases because the refugee effect of the mature stand is increasingly limited with distance.
2. Materials and Methods
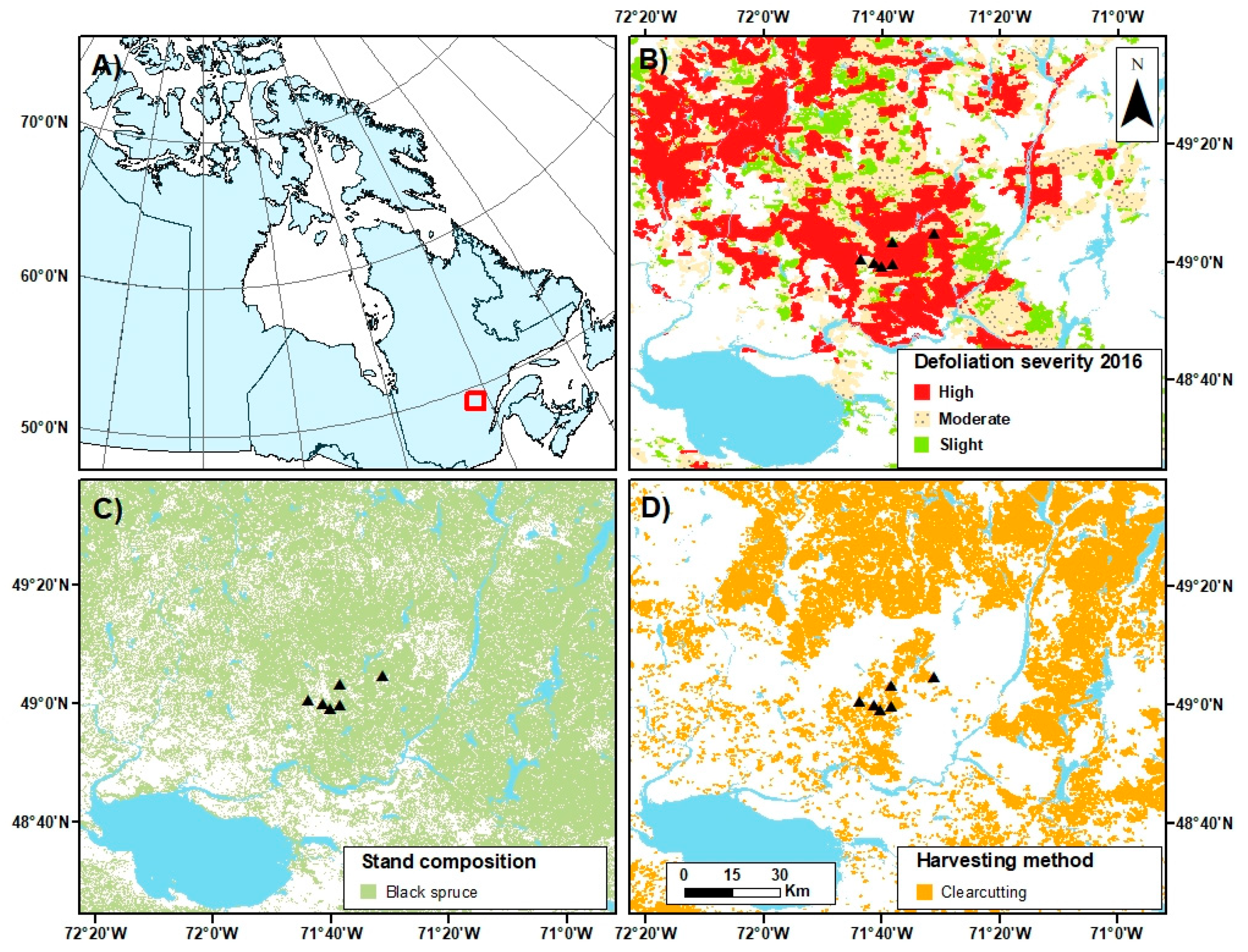
2.1. Study Area
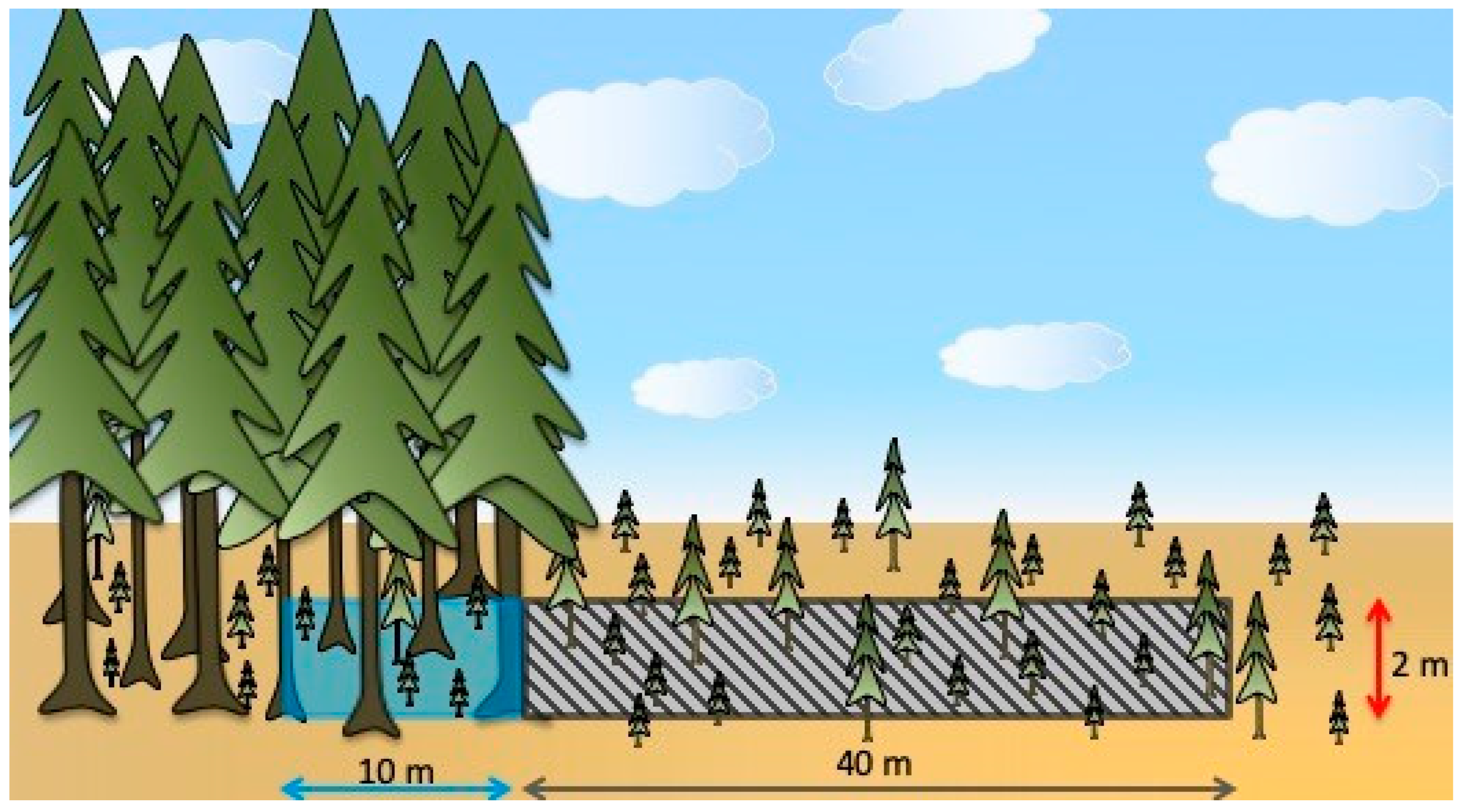
2.2. Site Selection and Experimental Design
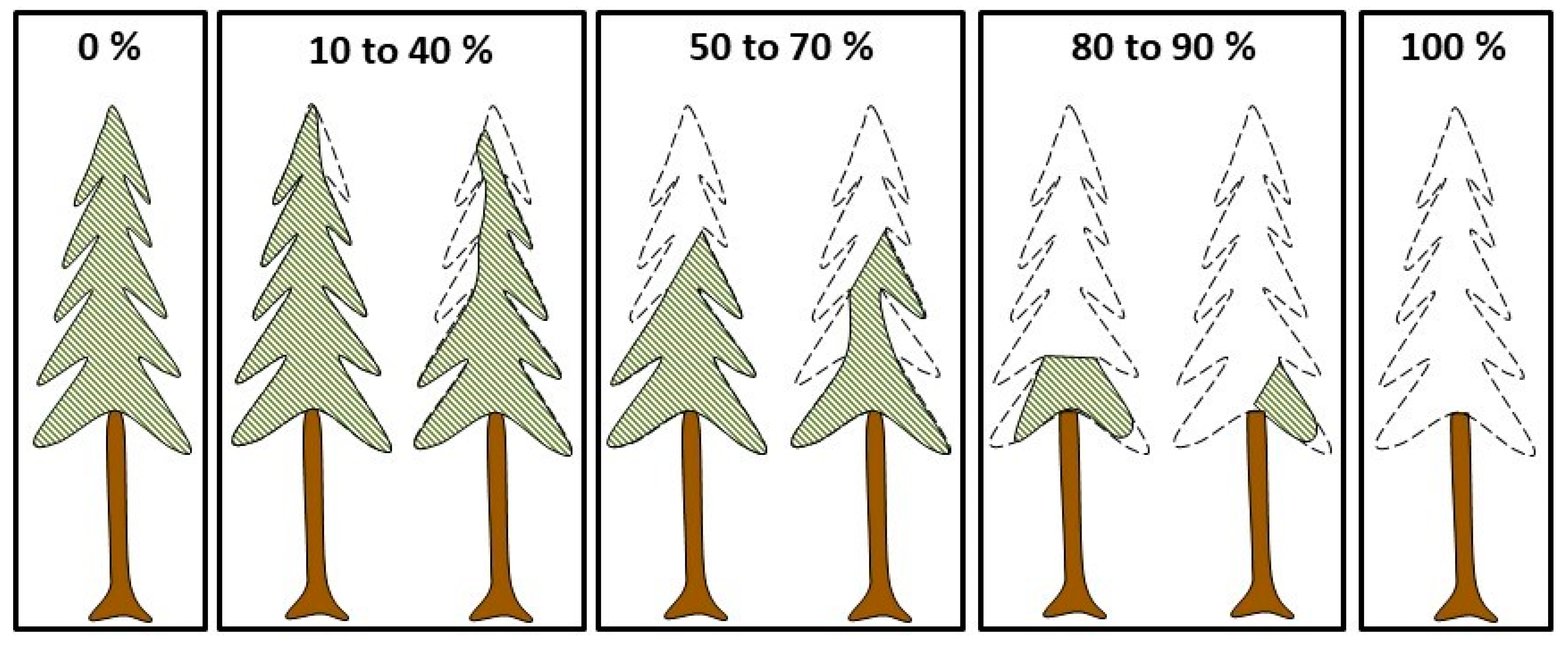
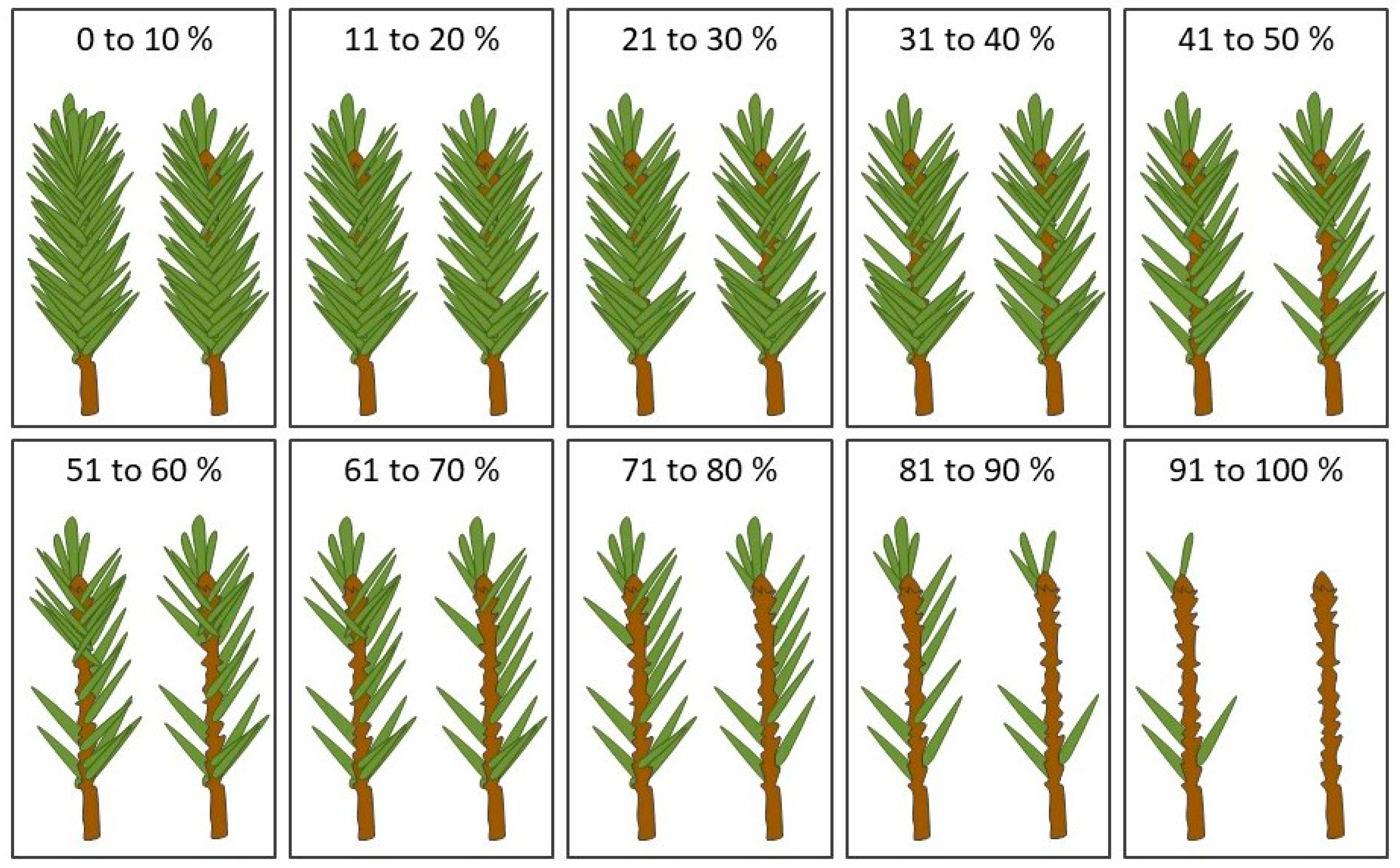
2.3. Measurements and Data Compilation
2.4. Data Analysis
3. Results
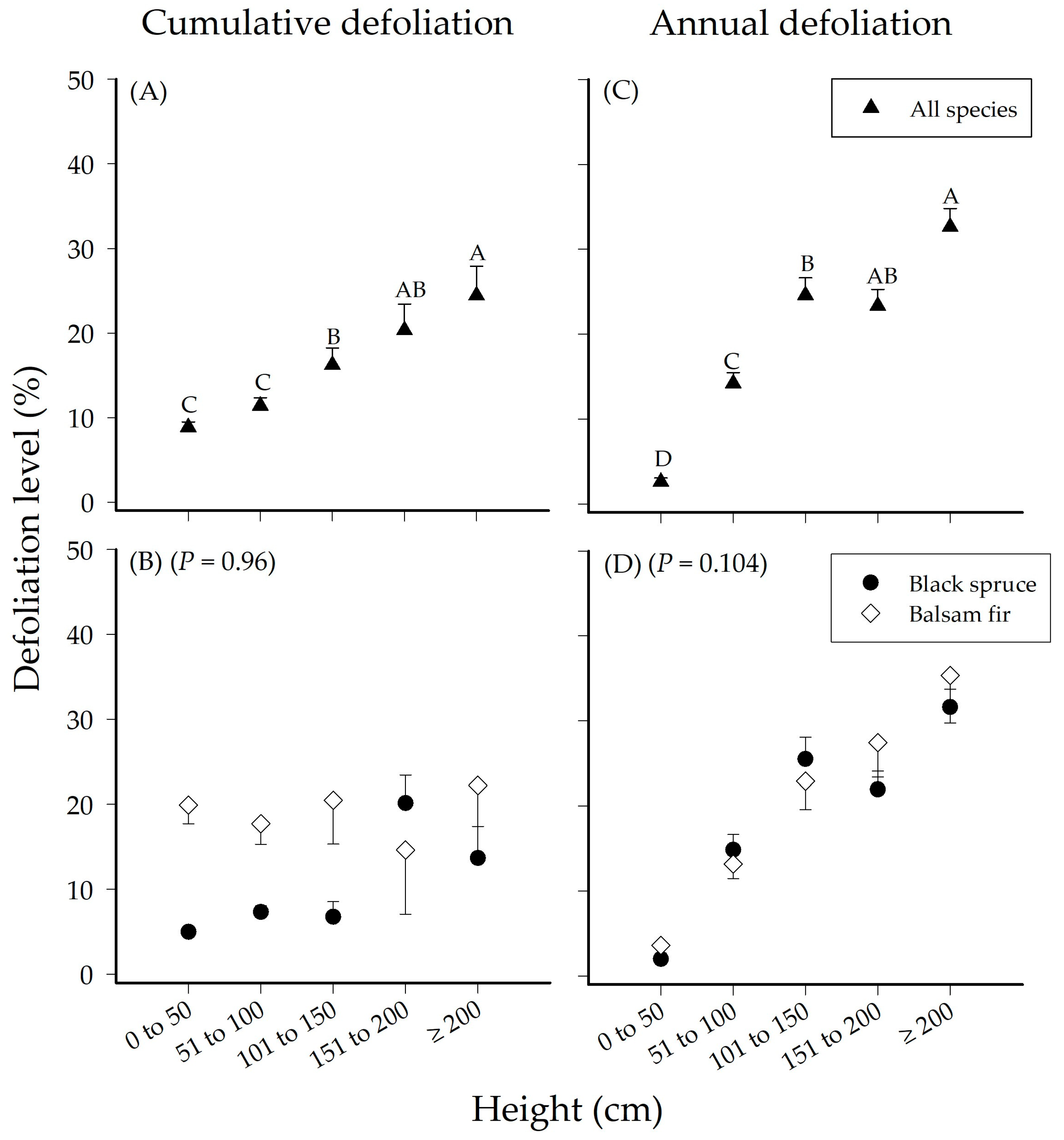
3.1. Effect of Height on Seedling Defoliation
3.2. Effect of Distance on Seedling Defoliation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Seidl, R.; Thom, D.; Kautz, M.; Martin-Benito, D.; Peltoniemi, M.; Vacchiano, G.; Wild, J.; Ascoli, D.; Petr, M.; Honkaniemi, J.; et al. Forest disturbances under climate change. Nat. Clim. Chang. 2017, 7, 395–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montoro Girona, M.; Navarro, L.; Morin, H. A secret hidden in the sediments: Lepidoptera scales. Front. Ecol. Evol. 2018, 6, 1–5. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Morin, H.; Lussier, J.-M.; Ruel, J.-C. Post-cutting mortality following experimental silvicultural treatments in unmanaged boreal forest stands. Front. Forests Glob. Chang. 2019, 2, 1–16. [Google Scholar] [CrossRef]
- Navarro, L.; Harvey, A.-É.; Ali, A.; Bergeron, Y.; Morin, H. A Holocene landscape dynamic multiproxy reconstruction: How do interactions between fire and insect outbreaks shape an ecosysten over long time scales? PLoS ONE 2018, 15, e0204316. [Google Scholar] [CrossRef]
- Waldron, K.; Ruel, J.-C.; Gauthier, S. The effects of site characteristics on the landscape-level windthrow regime in the North Shore region of Quebec, Canada. Forestry 2013, 86, 159–171. [Google Scholar] [CrossRef]
- De Grandpré, L.; Waldron, K.; Bouchard, M.; Gauthier, S.; Beaudet, M.; Ruel, J.-C.; Hébert, C.; Kneeshaw, D. Incorporating insect and wind disturbances in a natural disturbance-based management framework for the boreal forest. Forests 2018, 9, 471. [Google Scholar] [CrossRef]
- Ulanova, N.G. The effects of windthrow on forests at different spatial scales: A review. For. Ecol. Manag. 2000, 135, 155–167. [Google Scholar] [CrossRef]
- Boulanger, Y.; Gray, D.R.; Cooke, B.J.; De Grandpre, L. Model-specification uncertainty in future forest pest outbreak. Glob. Chang. Biol. 2016, 22, 1595–1607. [Google Scholar] [CrossRef] [PubMed]
- Alfaro, R.I.; Taylor, S.; Brown, R.G.; Clowater, J.S. Susceptibility of northern British Columbia forest to spruce budworm defoliation. For. Ecol. Manag. 2011, 145, 181–190. [Google Scholar] [CrossRef]
- MacKinnon, W. The Influence of Surrounding Forest Landscape and Stand Conditions on Stand Susceptibility and Vulnerablity to Spruce Budworm in New Brunswick. Ph.D. Thesis, University of New Brunswich, Fredericton, NB, Canada, 1992; p. 20. [Google Scholar]
- Jetté, J.-P.; Vaillancourt, M.-A.; Leduc, A.; Gauthier, S. Les enjeux écologiques de l’aménagement forestier. In Aménagement Écosystémique en Forêt Boréale; Presses de l’Université du Québec: Quebec City, QC, Canada, 2008; pp. 1–10. [Google Scholar]
- Montoro Girona, M. À la Recherche de L’aménagement Durable en Forêt Boréale: Croissance, Mortalité et Régénération des Pessières Noires Soumises à Différents Systèmes Sylvicoles. Ph.D. Thesis, Université du Québec à Chicoutimi, Chicoutimi, QC, Canada, 2017; p. 287. [Google Scholar] [CrossRef]
- National Forestry. National Forest and Forest Management Statistics. (Last Update 23 February 2017). Available online: http://nfdp.ccfm.org/index_e.php (accessed on 14 November 2018).
- Gauthier, S.; Bernier, P.; Burton, P.J.; Edwards, J.; Isaac, K.; Isabel, N.; Jayen, K.; Le Goff, H.; Nelson, E.A. Climate change vulnerability and adaptation in the managed Canadian boreal forest. Environ. Rev. 2014, 22, 256–285. [Google Scholar] [CrossRef]
- Ressources Naturelles Canada. La recherche au centre de foresteries des Laurentides de Ressources naturelles Canada « La tordeuse des bourgeons de l’épinette ». In Spruce Budworm; Ressources Naturelles Canada: Ottawa, ON, Canada, 2012; pp. 1–16. [Google Scholar]
- Ressources Naturelles Canada. Tordeuse des Bourgeons de L’épinette (Fiche D’information). (Last Update 15 August 2016). Available online: http://www.rncan.gc.ca/forets/feux-insectes-perturbations/principaux-insectes/13404 (accessed on 18 February 2018).
- Morin, H.; Laprise, D.; Simard, A.A.; Amouch, A. Régime des épidémies de la tordeuse des bourgeons de l’épinette dans l’Est de l’Amérique du Nord. In Aménagement Écosystémique en Forêt Boréale; Gauthier, S., Vaillancourt, M.-A., Leduc, A., De Grandpré, L., Kneeshaw, D., Morin, H., Drapeau, P., Bergeron, Y., Eds.; Les Presses de l’Université du Québec: Quebec City, QC, Canada, 2008; pp. 165–192. [Google Scholar]
- Logan, J.A.; Régnière, J.; Powell, J.A. Assessing the impacts of global warming on forest pest dynamics. Front. Ecol. Environ. 2003, 1, 130–137. [Google Scholar] [CrossRef]
- Navarro, L.; Morin, H.; Bergeron, Y.; Girona, M.M. Changes in spatiotemporal patterns of 20th century spruce budworm outbreaks in eastern canadian boreal forests. Front. Plant Sci. 2018, 9, 1905–1920. [Google Scholar] [CrossRef] [PubMed]
- Blais, J. Trends in the frequency, extent, and severity of spruce budworm outbreaks in eastern Canada. Can. J. For. Res. 1983, 13, 539–547. [Google Scholar] [CrossRef]
- Rowe, J.S.; Scotter, G.W. Fire in the boreal forest. Quat. Res. 1973, 3, 444–464. [Google Scholar] [CrossRef]
- Nie, Z.; MacLean, D.; Taylor, A.R. Forest overstory composition and seedling heigh influence defoliation of understory regeneration by spruce budworm. For. Ecol. Manag. 2018, 409, 353–360. [Google Scholar] [CrossRef]
- Nie, Z.; Maclean, D.A.; Taylor, A.R. Disentangling variables that influence growth response of balsam fir regeneration during a spruce budworm outbreak. For. Ecol. Manag. 2019, 433, 13–23. [Google Scholar] [CrossRef]
- MacKinnon, W.; MacLean, D. Effects of surrounding forest and site conditions on growth reduction of balsam fir and spruce caused by spruce budworm defoliation. Can. J. For. Res. 2004, 34, 2351–2362. [Google Scholar] [CrossRef]
- Hennigar, C.R.; MacLean, D.; Quiring, D.T.; Kershaw, J.A.J. Diffenrences in spruce budworm defoliation among balsam fir and white, red, and black spruce. For. Sci. 2008, 54, 158–166. [Google Scholar]
- Nealis, V.G.; Régnière, J. Insect-host relationships influencing disturbance by the spruce budworm in a boreal mixedwood forest. Can. J. For. Res. 2004, 34, 1870–1882. [Google Scholar] [CrossRef]
- Blais, J. Some relationships of the spruce budworm, Choristoneura fuminerana (CLEM.) to black spruce, Picea mariana (MOENCH) Voss. For. Chron. 1957, 33, 364–372. [Google Scholar] [CrossRef]
- Pureswaran, D.S.; De Grandpré, L.; Paré, D.; Taylor, A.; Barette, M.; Morin, H.; Régnière, J.; Kneeshaw, D.D. Climate-induces changes in host tree-insect phenology may drive ecological strate-shift in boreal forests. Ecology 2015, 96, 1480–1491. [Google Scholar] [CrossRef]
- Fuentealba, A.; Bauce, É. Carry-over effects of host nutritional quality on performance of spruce budworm progeny. Bull. Entomol. Res. 2012, 102, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Bauce, É.; Crépin, M.; Carisey, N. Spruce budworm growth, development and food utilization on young and old balsam fir trees. Oecologia 1994, 97, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Cotton-Gagnon, A.; Simard, M.; De Grandpré, L.; Kneeshaw, D. Salvage logging during spruce budworm outbreaks increases defoliation of black spruce regeneration. For. Ecol. Manag. 2018, 430, 421–430. [Google Scholar] [CrossRef]
- Bognounou, F.; De Grandpré, L.; Pureswaran, D.S.; Kneeshaw, D. Temporal variation in plant neighborhood effetcs on the defoliation of primary and secondary hosts by an insect pest. Ecosphere 2017, 8, 1–15. [Google Scholar] [CrossRef]
- Maclean, D.; Piene, H. Spatial and temporal patterns of balsam fir mortality in spaced and unspaced stands caused by spruce budworm defoliation. Can. J. For. Res. 1995, 25, 902–911. [Google Scholar] [CrossRef]
- Piene, H. Spruce budworm defoliation and growth loss in young balsam fir: Defoliation in spaced and unspaced stands and individual tree survival. Can. J. For. Res. 1989, 19, 1211–1217. [Google Scholar] [CrossRef]
- Sainte-Marie, G.B.; Kneeshaw, D.D.; MacLean, D.A.; Hennigar, C.R. Estimating forest vulnerability to the next spruce budworm outbreak: Will past silvicultural efforts pay dividends? Can. J. For. Res. 2015, 45, 314–324. [Google Scholar] [CrossRef]
- Baskerville, G. Spruce Budworm: The Answer Is Forest Management: Or Is It. For. Chron. 1975, 51, 157–160. [Google Scholar] [CrossRef]
- MacLean, D. Forest management strategies to reduce spruce budworm damage in the Fundy Model Forest. For. Chron. 1996, 72, 399–405. [Google Scholar] [CrossRef] [Green Version]
- Ruel, J.C.; Huot, M. Impacts de la tordeuse des bourgeons de l’épinette (Choristoneura fumiferana (Clem.)) sur la régénération des sapinières après la coupe à blanc. For. Chron. 1993, 69, 163–172. [Google Scholar] [CrossRef]
- Krause, C.; Morin, H.; Plourde, P.Y. Juvenile growth of black spruce (Picea mariana [Mill.] BSP) stands established during endemic and epidemic attacks by spruce budworm (Choristoneura fumiferana [Clemens]) in the boreal forest of Quebec, Canada. For. Chron. 2009, 85, 267–276. [Google Scholar] [CrossRef]
- Piene, H.; Maclean, D.A.; Landry, M. Spruce budworm defoliation and growth loss in young balsam fir: Relationships between volume growth and foliage weight in spaced and unspaced, defoliated and protected stands. For. Ecol. Manag. 2003, 179, 37–53. [Google Scholar] [CrossRef]
- Ministère des Ressources Naturelles. Plan D’aménagement Forestier Intégré Tactique: Unité D’aménagement 024-51; Secteur des Opération Régionales, Ed.; Ministère des Ressouces Naturelles: Quebec City, QC, Canada, 2013; p. 360.
- Rossi, S.; Morin, H.; Deslauriers, A.; Plourde, P.Y. Predicting xylem phenology in black spruce under climate warming. Glob. Chang. Biol. 2011, 17, 614–625. [Google Scholar] [CrossRef]
- MDDELCC. Normales Climatiques du Québec 1981–2010: Péribonka. (Last Update 2018). Available online: http://www.mddelcc.gouv.qc.ca/climat/normales/sommaire.asp?cle=7065960 (accessed on 13 April 2018).
- Gouvernement du Canada. Canadian Climate Normals 1981–2010 Station Data. (Last Update 11 January 2018). Available online: http://climate.weather.gc.ca/climate_normals/results_1981_2010_e.html?searchType=stnProv&lstProvince=QC&txtCentralLatMin=0&txtCentralLatSec=0&txtCentralLongMin=0&txtCentralLongSec=0&stnID=5889&dispBack=0 (accessed on 13 April 2018).
- Robitaille, A.; Saucier, J.P. Paysages du Québec méridional; Les Publications du Québec: Québec, QC, Canada, 1998; p. 213.
- Raymond, R.; Mailloux, A.; Dubé, A. Pédologie de la Région du Lac-Saint-Jean; Ministère de l’Agriculture et de la Colonisation: l’Alimentation, QC, Canada, 1965; p. 159.
- Gouvernement du Québec. I.G.O.—Données Écoforestières. (Last Update 30 November 2017). Available online: https://geoegl.msp.gouv.qc.ca/igo/mffpecofor/ (accessed on 27 March 2018).
- MFFP. Aires Infestées par la Tordeuse des Bourgeons de L’épinette au Québec en 2016; Ministère des Forêts, de la Faune et des Parcs: l’Alimentation, QC, Canada, 2016; p. 20.
- Fenton, N.; Imbeau, L.; Work, T.; Jacobs, J.; Bescond, H.; Drapeau, P.; Bergeron, Y. Lessons learned from 12 years of ecological research on partial cuts in black spruce forests of northwestern Quebec. For. Chron. 2013, 89, 350–359. [Google Scholar] [CrossRef]
- Hunter, M.L. Wildlife, Forests, and Forestry: Principales of Managing Forests for Biological Diversity; Prentice-Hall: Englewood Cliffs, NJ, USA, 1990; p. 370. [Google Scholar]
- MacLean, D.; Erdle, T. Development of relationships between spruce budworm defoliation and forest stand increment in New Brunswick. In Environmental Influences on Tree and Stand Increment; Solomon, D., Brann, T., Eds.; Maine Agricultural Experiment Station: Durham, NH, USA, 1985; pp. 106–115. [Google Scholar]
- Anderson, M.J.; Gorley, R.; Clarke, K. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; Primer-E Ltd.: Plymouth, UK, 2008; p. 218. [Google Scholar]
- Clarke, K.; Gorley, R.; Somerfield, P.; Warwick, R. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 3rd ed.; Primer-E Ltd.: Plymouth, UK, 2014; p. 262. [Google Scholar]
- Triola, M.; Triola, M. Biostatistique Pour Les Sciences de la Vie et de la Santé; Pearson: Montreuil, France, 2012; p. 367. [Google Scholar]
- Gauthier, S.; Vaillancourt, M.-A.; Leduc, A.; Kneeshaw, D.; Drapeau, P.; De Grandpré, L.; Claveau, Y.; Paré, D. Ecosystem Management in the Boreal Forest; Presses de l’Université du Québec: Quebec City, QC, Canada, 2009; p. 570. [Google Scholar]
- Prévost, M.; Raymond, P.; Lussier, J.-M. Regeneration dynamics after patch cutting and scarification in yellow birch—Conifer stands. Can. J. For. Res. 2010, 40, 357–369. [Google Scholar] [CrossRef]
- Fuentealba, A.; Bauce, É. Interspecific variation in resistance of two host tree species to spruce budworm. Acta Oecol. 2016, 70, 10–20. [Google Scholar] [CrossRef]
- Nealis, V.G. Comparative ecology of conifer-feeding spruce budworms (Lepidoptera: Tortricidae). Can. Entomol. 2016, 148, S33–S57. [Google Scholar] [CrossRef]
- Chabot, M.; Boulet, B.; Morneau, L. La tordeuse des bourgeons de l’épinette. In Le Guide Sylvicole du Québec: Les Fondements Biologiques de la Sylviculture. Tome 1; Les Publications du Québec: Québec, QC, Canada, 2013; pp. 400–413. [Google Scholar]
- Fuentealba, A.; Pureswaran, D.S.; Bauce, É.; Despland, E. How does synchrony with host plant affect the performance of an outbreaking insect defoliator? Oecologia 2017, 184, 847–857. [Google Scholar] [CrossRef]
- Bronson, D.R.; Gower, S.T.; Tanner, M.; Van Herk, I. Effect of ecosystem warming on boreal black spruce bud burst and shoot growth. Glob. Chang. Biol. 2009, 15, 1534–1543. [Google Scholar] [CrossRef]
- Cotton-Gagnon, A. Effets de Facteurs Naturels et de la Coupe de Récupération sur la Défoliation de la Régénération Préétablie Pendant une Épidémie de la Tordeuse des Bourgons de L’épinette. Master’s Thesis, Université Laval, Quebec City, QC, Canada, 2017; p. 50. [Google Scholar]
- Swaine, J.M. The relation of insect activities to forest development as exemplified in the forest of eastern North America. For. Chron. 1933, 9, 9–32. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Lussier, J.-M.; Morin, H.; Thiffault, N. Conifer regeneration after experimental shelterwood and seed-tree treatments in boreal forests: Finding sylvicultural alternatives. Front. Plant Sci. 2018, 9, 1145–1159. [Google Scholar] [CrossRef] [PubMed]
- Rosenvald, R.; Lõhmus, A. For what, when, and where is green-tree retention better than clear-cutting? A review of the biodiversity aspects. For. Ecol. Manag. 2008, 255, 1–15. [Google Scholar] [CrossRef]
- Fischer, J.; Lindenmayer, D. Landscape modification and habitat fragmentation: A synthesis. Glob. Ecol. Biogeogr. 2007, 16, 265–280. [Google Scholar] [CrossRef]
- Puettmann, K.J.; Wilson, S.M.; Baker, S.C.; Donoso, P.J.; Drössler, L.; Amente, G.; Harvey, B.D.; Knoke, T.; Lu, Y.; Nocentini, S.; et al. Silvicultural alternatives to conventional even-aged forest management—What limits global adoption? For. Ecosyst. 2015, 2, 1–16. [Google Scholar] [CrossRef]
- Groot, A.; Lussier, J.-M.; Mitchell, A.K.; MacIsaac, D.A. A silvicutural systems perspective on changing Canadian forestry practices. For. Chron. 2005, 81, 50–56. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Morin, H.; Lussier, J.-M.; Walsh, D. Radial growth response of black spruce stands ten years after experimental shelterwoods and seed-tree cuttings in boreal forest. Forests 2016, 7, 240. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Rossi, S.; Lussier, J.-M.; Walsh, D.; Morin, H. Understanding tree growth responses after partial cuttings: A new approach. PLoS ONE 2017, 12, e0172653. [Google Scholar] [CrossRef]
- Zhang, X.; Lei, Y.; Ma, Z.; Kneeshaw, D.; Peng, C. Insect-induced tree mortality of boreal forests in eastern Canada under a changing climate. Ecol. Evol. 2014, 4, 2384–2394. [Google Scholar] [CrossRef] [PubMed]
- Blais, J.R. The vulnerability of balsam fir to spruce budworm attack in northwestern Ontario, with special reference to the physiological age of tree. For. Chron. 1958, 34, 405–422. [Google Scholar] [CrossRef]
- Bergeron, Y.; Leduc, A.; Morin, H.; Joyal, C. Balsam fir mortality following the last spruce budworm outbreak in northwestern Quebec. Can. J. For. Res. 1995, 25, 1375–1384. [Google Scholar] [CrossRef]
- Johnstone, J.F.; Allen, C.D.; Franklin, J.F.; Frelich, L.E.; Harvey, B.J.; Higuera, P.E.; Mack, M.C.; Meentemeyer, R.K.; Metz, M.R.; Perry, G.L.W.; et al. Changing disturbance regimes, ecological memory, and forest resilience. Front. Ecol. Environ. 2016, 14, 369–378. [Google Scholar] [CrossRef]
- Bouchard, M.; Kneeshaw, D.; Messier, C. Forest dynamics following spruce budworm outbreaks in the nothern and southern mixedwoods of central Quebec. Can. J. For. Res. 2007, 37, 763–772. [Google Scholar] [CrossRef]
- Taylor, S.L.; MacLean, D.A. Legacy of insect defoliators: Increased wind-related mortality two decades after a spruce budworm outbreak. For. Sci. 2008, 55, 256–267. [Google Scholar]

| Plot | Harvesting Date | Species | Number of Seedlings | Distance Classes | Height Classes (cm) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R.S. | N | I | D | 0 to 50 | 51 to 100 | 101 to 150 | 151 to 200 | ≥200 | ||||
| A | 2003 | Black spruce | 50 | 17 | 22 | 11 | 0 | 39 | 8 | 3 | 0 | 0 |
| Balsam fir | 150 | 21 | 65 | 42 | 22 | 75 | 44 | 18 | 4 | 9 | ||
| Total | 200 | 38 | 87 | 53 | 22 | 114 | 52 | 21 | 4 | 9 | ||
| B | 2008 | Black spruce | 76 | 29 | 18 | 19 | 10 | 31 | 22 | 10 | 7 | 6 |
| Balsam fir | 1 | 1 | 0 | 9 | 0 | 0 | 0 | 0 | 1 | 0 | ||
| Total | 77 | 30 | 18 | 19 | 10 | 31 | 22 | 10 | 8 | 6 | ||
| C | 1994 | Black spruce | 39 | 1 | 3 | 15 | 20 | 25 | 10 | 3 | 1 | 0 |
| Balsam fir | 29 | 8 | 14 | 3 | 4 | 2 | 12 | 5 | 5 | 5 | ||
| Total | 68 | 9 | 17 | 18 | 24 | 27 | 22 | 8 | 6 | 5 | ||
| D | 2000 | Black spruce | 32 | 6 | 12 | 10 | 4 | 2 | 5 | 8 | 5 | 12 |
| Balsam fir | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Total | 32 | 6 | 12 | 10 | 4 | 2 | 5 | 8 | 5 | 12 | ||
| E | 1999 | Black spruce | 40 | 14 | 8 | 11 | 7 | 7 | 17 | 5 | 2 | 9 |
| Balsam fir | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Total | 40 | 14 | 8 | 11 | 7 | 7 | 17 | 5 | 2 | 9 | ||
| F | 1999 | Black spruce | 87 | 6 | 28 | 22 | 31 | 18 | 29 | 14 | 17 | 9 |
| Balsam fir | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | ||
| Total | 88 | 7 | 28 | 22 | 31 | 18 | 29 | 14 | 18 | 9 | ||
| Factors | DF | SS | MS | Pseudo-F | p(MC) | |
|---|---|---|---|---|---|---|
| Cumulative Defoliation | Height | 4 | 11061 | 2765.1 | 14.553 | 0.001 |
| Species | 1 | 8720.2 | 8720.2 | 45.895 | 0.001 | |
| Height × Species | 4 | 1538.2 | 384.6 | 2.024 | 0.96 | |
| Residual | 495 | 94052 | 190 | |||
| Annual Defoliation | Height | 4 | 1.50 × 105 | 37559 | 26.434 | 0.001 |
| Species | 1 | 333.4 | 333.4 | 0.235 | 0.849 | |
| Height × Species | 4 | 9654 | 2413.5 | 1.6987 | 0.104 | |
| Residual | 496 | 7.05 × 105 | 1420.8 |
| Factors | DF | SS | MS | Pseudo-F | p(MC) | |
|---|---|---|---|---|---|---|
| Cumulative Defoliation | Distance | 3 | 1586.8 | 528.95 | 2.42 | 0.054 |
| Species | 1 | 15624 | 15624 | 71.52 | 0.001 | |
| Distance × Species | 3 | 2140.4 | 713.46 | 3.27 | 0.022 | |
| Residual | 497 | 1.09 × 105 | 218.45 | |||
| Annual Defoliation | Distance | 3 | 5992.3 | 1997.4 | 1.16 | 0.288 |
| Species | 1 | 997.9 | 997.86 | 0.58 | 0.573 | |
| Distance × Species | 3 | 10544 | 3514.5 | 2.04 | 0.049 | |
| Residual | 498 | 8.57 × 105 | 1721.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lavoie, J.; Montoro Girona, M.; Morin, H. Vulnerability of Conifer Regeneration to Spruce Budworm Outbreaks in the Eastern Canadian Boreal Forest. Forests 2019, 10, 850. https://doi.org/10.3390/f10100850
Lavoie J, Montoro Girona M, Morin H. Vulnerability of Conifer Regeneration to Spruce Budworm Outbreaks in the Eastern Canadian Boreal Forest. Forests. 2019; 10(10):850. https://doi.org/10.3390/f10100850
Chicago/Turabian StyleLavoie, Janie, Miguel Montoro Girona, and Hubert Morin. 2019. "Vulnerability of Conifer Regeneration to Spruce Budworm Outbreaks in the Eastern Canadian Boreal Forest" Forests 10, no. 10: 850. https://doi.org/10.3390/f10100850
APA StyleLavoie, J., Montoro Girona, M., & Morin, H. (2019). Vulnerability of Conifer Regeneration to Spruce Budworm Outbreaks in the Eastern Canadian Boreal Forest. Forests, 10(10), 850. https://doi.org/10.3390/f10100850






