1. Introduction
Acute myocardial infarction (AMI) in adults younger than 45 years is an increasingly recognized entity with distinct epidemiological and clinical features, driven by a complex interplay of traditional factors (smoking, dyslipidemia, obesity, insulin resistance) and non-traditional determinants such as chronic inflammation, psychosocial stress, sleep disorders, recreational drug use, and genetic susceptibility. This trend affects individuals in their most productive decades and is associated with substantial long-term risks of recurrent ischemia, arrhythmias, adverse remodeling, and impaired quality of life, which are only partially captured via traditional, episodic clinic-based follow-up [
1,
2,
3].
Digital health technologies offer new opportunities to address these gaps through the continuous remote monitoring of physiological and behavioral parameters. Contemporary wearable systems integrate photoplethysmography, accelerometry, and oximetry to derive heart rate, activity, sleep, and surrogate measures of autonomic function, enabling a high–temporal resolution assessment of post-AMI recovery in real-world settings. In parallel, advances in bio-integrated flexible electronics—such as conformal hemodynamic patches, electrospun polymeric fibers, and flexible graphene-based transducers—are pushing the frontier toward skin-like, mechanically compliant devices capable of the real-time monitoring of blood pressure, flow, and bioelectrical or biochemical signals for cardiovascular healthcare. Together with artificial intelligence algorithms, these technologies support precision cardiology paradigms that couple multi-parametric sensing with personalized risk stratification and dynamic therapy adjustment [
4,
5].
Within this evolving landscape, medical-grade wearable devices like the SiDLY Care Pro occupy a pragmatic position between research-grade flexible bioelectronics and routine clinical practice. The SiDLY system provides the validated, non-invasive monitoring of heart rate, oxygen saturation, and physical activity through a wrist-worn multisensor platform, complemented with fall detection, SOS alerting, geolocation, and encrypted cloud-based telemonitoring. Unlike emerging bio-integrated flexible electronics, which prioritize intimate skin conformity and advanced hemodynamic or biochemical sensing, SiDLY emphasizes usability, regulatory compliance, and integration into telemedicine workflows, making it suitable for heterogeneous young AMI populations with variable digital literacy and lifestyles [
5,
6,
7].
The present study, as summarised in
Table 1, enhances existing knowledge by (i) implementing SiDLY-enabled continuous monitoring among young AMI survivors within real-world clinical pathways, (ii) combining objective biosignals with validated patient-reported outcome measures (IPAQ, DASS-21, SF-36) to obtain a multidimensional view of physical, psychological, and behavioral recovery, and (iii) positioning this clinically deployable wearable within the broader context of bio-integrated flexible electronics, highlighting how scalable telemonitoring today can complement and prepare the ground for the future integration of more sophisticated flexible platforms in cardiovascular secondary prevention. While prior studies and meta-analyses show that wearable-supported cardiac rehabilitation increases daily steps, improves exercise capacity, and reduces rehospitalizations in broad coronary artery disease populations [
8,
9,
10], evidence remains scarce for young AMI survivors under 50 years of age, who display distinct recovery trajectories and a non-negligible burden of recurrent events despite milder angiographic disease and lower short-term mortality compared with older patients [
11,
12,
13].
2. Materials and Methods
The remote monitoring of patients with early myocardial infarction using wearable devices represents a progressive trend in healthcare, reflecting the increased integration of wearable technologies for continuous, real-time health assessment. This approach is particularly relevant in an era where telemedicine and remote patient management are pivotal for chronic disease care and the risk mitigation of acute cardiac events. Wearable devices enable continuous data collection, optimizing patient management and reducing the risk of post-infarction complications. The present work aligns with the evolving landscape of personalized medicine, where technology-driven solutions enhance both treatment effectiveness and patient engagement.
This study specifically targets a younger cohort with early myocardial infarction—an important choice, given their extended post-event life expectancy and heightened risk for developing long-term complications. Personalized monitoring in this group, addressing both physiological and psychological aspects, is critical for proactive management and improved long-term outcomes. The primary aim of this study was to evaluate the utility of wearable devices for the remote monitoring of motor activity in patients following early myocardial infarction.
2.1. Inclusion Criteria
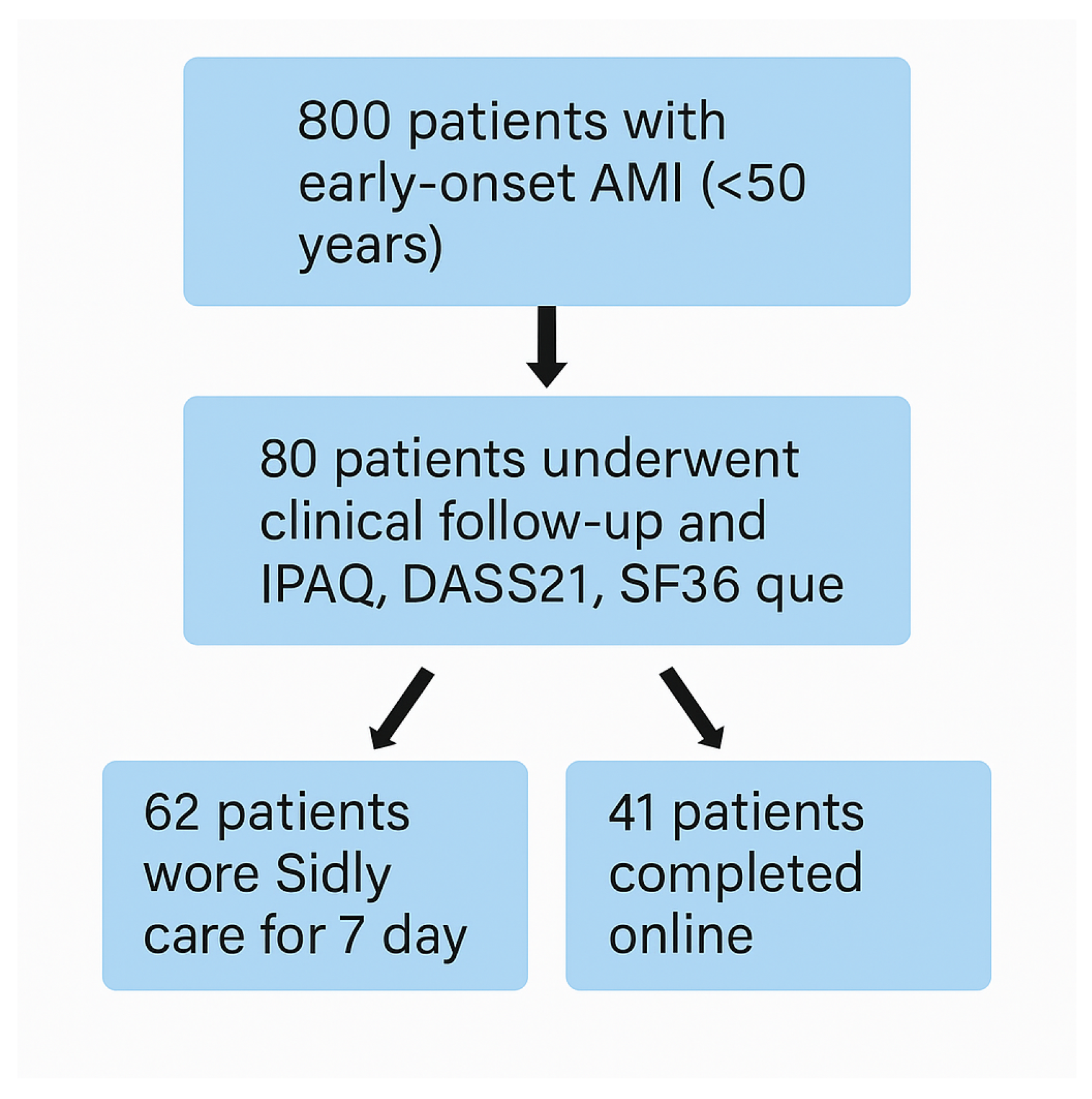
Patients were randomly selected from a large institutional database comprising approximately 800 individuals who had experienced an acute myocardial infarction (AMI) before the age of 50. This registry includes patients with confirmed ST-elevation myocardial infarction (STEMI) and non–ST-elevation myocardial infarction (NSTEMI), documented according to current ESC and ACC/AHA diagnostic criteria, integrating biochemical, electrocardiographic, and imaging-based evidence. For the purposes of this study, stringent inclusion criteria were applied to ensure the homogeneity of the analytical cohort and relevance to the research objectives. Eligible participants met the following conditions:
Age at the time of the AMI index of less than 50 years, ensuring that the sample reflects the population with premature myocardial infarction.
A minimum of 12 months elapsed since the index event, permitting the stabilization of acute-phase physiological alterations and enabling a reliable assessment of long-term recovery patterns.
Evidence of myocardial infarction documented through troponin elevation, ischemic symptoms, and compatible ECG or imaging findings.
Clinical stability at the time of enrollment, defined as the absence of acute coronary syndromes, decompensated heart failure, or significant arrhythmias in the preceding 3 months.
An ability to provide informed consent and a willingness to participate in remote monitoring through wearable technology.
From the total eligible pool, 80 participants were randomly selected, following a stratified approach to balance sex distribution, index MI type, and treatment modality (PCI vs. conservative). Among these, 62 individuals were equipped with a Sidly Care Pro, Sidly, Poland wearable device, which enabled the continuous monitoring of physiological and behavioral parameters relevant to post-AMI recovery. A schematic representation of the recruitment process and allocation strategy is provided in
Figure 1.
Figure 1 ensured a representative, clinically stable, and methodologically coherent sample suitable for downstream analyses involving biosignal acquisition, behavioral profiling, and patient-reported outcomes.
The cohort’s male predominance reflects the study’s deliberate focus on early-onset AMI in young males, for whom preliminary data from regional registries and prior AI-ECG analyses demonstrate a markedly higher incidence compared to females, often linked to aggressive atherosclerosis and autonomic imbalances.
2.2. Clinical and Instrumental Evaluation
After written informed consent was obtained in accordance with the Declaration of Helsinki and institutional ethical board approval, all enrolled patients underwent a comprehensive and standardized clinical assessment. This multidimensional evaluation was designed to capture cardiovascular status, the comorbidity burden, pharmacological regimens, and potential opportunities for therapeutic optimization. A detailed medical history was collected, including the following: 1. Demographic information and cardiovascular risk profile; 2. Characteristics of the index AMI (STEMI/NSTEMI, culprit vessel, revascularization strategy, complications); 3. Comorbidities such as hypertension, diabetes mellitus, dyslipidemia, chronic kidney disease, and obstructive sleep apnea; 4. Family history of premature coronary artery disease; 5. Lifestyle factors (smoking, alcohol intake, physical activity habits); 6. Pharmacological therapies, with an assessment of adherence and potential drug–drug interactions. A comprehensive physical examination was performed, evaluating vital signs, cardiovascular auscultation, evidence of heart failure (NYHA class, peripheral edema, jugular venous pressure), and anthropometric parameters (weight, BMI, waist circumference). Each participant underwent a standard 12-lead electrocardiogram, analyzed for residual ischemic changes, Q-waves, conduction abnormalities, ventricular hypertrophy, and arrhythmic patterns. An additional non-invasive hemodynamic evaluation included heart rate, blood pressure measurement following ESC guidelines, and an assessment of autonomic tone through resting heart rate variability when feasible. Venous blood samples were collected in the fasting state to obtain the following: 1. Lipid profile (total cholesterol, LDL-C, HDL-C, triglycerides); 2. Glycemic parameters (fasting glucose, HbA1c); 3. Renal and hepatic function tests; 4. Inflammatory markers when indicated (hs-CRP); 5. Cardiac biomarkers if clinically appropriate. Laboratory results were used to guide therapeutic optimization, with tailored adjustments in lipid-lowering agents, antihypertensives, glucose-lowering medications, or antiplatelet therapy where necessary.
2.3. Tele-Monitoring System Application
At the conclusion of the clinical visit, patients were provided with instructions and fitted with the Sidly wearable device for a seven-day monitoring period. The device records electrical signals from cardiac contractions, converting them into digital heart rate (bpm) readouts. The selection criteria ensured the exclusion of confounding factors such as pre-existing cardiac disease, pharmacological interventions, or lifestyle modifiers (e.g., smoking, caffeine, recent vigorous exercise). Participants received comprehensive instructions to avoid strenuous activity, caffeine, or food intake at least two hours prior to measurement.
The Sidly device was worn on the non-dominant wrist, ensuring good electrode–skin contact and minimal motion artifacts. Device placement and baseline signal stability were verified by trained personnel before the measurement period was started.
This device facilitates remote health monitoring via an online platform directly connected to a Telecenter, enabling real-time health status surveillance and individualized safety thresholds for vital sign alerts. The parameters monitored included heart rate, oxygen saturation, step count, positional changes (syncope risk), and medication timing. All sensors were calibrated prior to the experimental phase to ensure cross-device fidelity.
2.4. Online Questionnaire
After completing seven days of telemonitoring, patients completed an online questionnaire with three sections. The first collected demographic data (age range, gender, educational attainment). The second section assessed computer literacy, including skills in email use, online transactions, information retrieval, social media, and video conferencing. The third section evaluated attitudes toward telemonitoring (e.g., preference for integration with a smartwatch/phone, desire for manual vs. automated updates, the importance of data access, and preferences for direct or clinician-mediated feedback).
2.5. IPAQ
Participants completed the International Physical Activity Questionnaire (IPAQ) to assess physical activity over the preceding week. The IPAQ quantifies time spent in vigorous, moderate, and walking activities, as well as sedentary behavior, considering only activities of at least a 10 min duration.
2.6. SF36
The Short Form Health Survey 36 (SF-36) questionnaire was administered to quantify patient health status and quality of life. The SF-36 measures eight health domains using 36 items, with responses scored on Likert scales. The final item assesses perceived health changes over the past year.
2.7. DASS21
To evaluate psychological status, the 21-item Depression, Anxiety, and Stress Scale (DASS-21) was used, assessing symptoms with seven items for each domain.
2.8. Heart Rate Data Analysis
Heart rate data were acquired automatically every 15 min via the Sidly device and stored securely. Data were collected in three time windows: long (24 readings/6 h), medium (12 readings/3 h), and short (6 readings/1.5 h), capturing both acute and circadian variations. After transfer to the secure server, data underwent the following:
Artifact removal for sensor loss, motion, and extreme values (<40 or >180 bpm);
Normalization by z-score for interindividual comparison;
Segmentation into time windows;
Imputation of isolated missing data via linear interpolation (up to 2 consecutive samples).
Manual and automated annotations defined periods of rest, tachycardia, or bradycardia.
2.9. Dynamic Time Warping Barycenter Averaging (DBA)
The Dynamic Time Warping Barycenter Averaging (DBA) algorithm was applied to compute average heart rate patterns from time series of variable length/alignment. Series within each group were aligned and averaged using DTW, iteratively updated until convergence, to obtain a barycentric profile representative of the group.
2.10. Computational Workflow
The analysis comprised three main stages:
- 1.
Clustering based on DTW distance to group time series by similarity.
- 2.
DBA computation within each cluster to create barycentric patterns.
- 3.
Cluster and barycenter validation by silhouette score, variance, and visual assessment (including PCA-based projection for pattern visualization).
PCA projections facilitated a visualization of cluster separability and pattern structure. These analyses identified diverse dynamic profiles such as rest–tachycardia–rest and sustained tachycardia.
2.11. Statistical Analysis
Group differences were assessed with Chi-squared, ANOVA, or t-tests, using Bonferroni correction for multiple comparisons. The variance in barycenter patterns and group membership was related to physiological and subjective variables, providing context for the time-series findings.
3. Results
3.1. Results of the Online Questionnaire
Given the paramount importance of patient confidence in the use of Sidly remote monitoring devices, we developed an online questionnaire to evaluate satisfaction and perceived security associated with device wear. Robust engagement with device features is essential not only for facilitating the timely detection of health deterioration in home settings but also for ensuring effective communication with remote clinical teams.
The cohort predominantly comprised individuals aged 41–50 years (56.1%), males (85.4%), and those with a middle school education (39%). Digital literacy varied: 36.6% demonstrated excellent competency in email communications, with substantial proficiency in online news reading (43.9%) and moderate abilities in banking apps (31.7%), recreational gaming (26.8%), television applications (36.6%), video calling/conferencing (31.7%), social media (36.6%), and messaging systems (36.6%).
Nearly half the respondents (48.8%) regarded remote monitoring as essential for both personal health and broader healthcare-system efficacy. Preferences were pronounced for smartwatch-connected systems (73.2%) and those featuring automatic updates (87.8%). A majority favored direct connectivity with specialist telehealth teams (68.3%) and full reimbursement via the national health system (90.2%). The value of the continuous home monitoring of vital parameters was underscored by 61%, with over half desiring health feedback directly from the device (53.7%;
Figure 2 and
Figure 3) and a clear preference for clinician-generated feedback over AI-based analysis (90.2%).
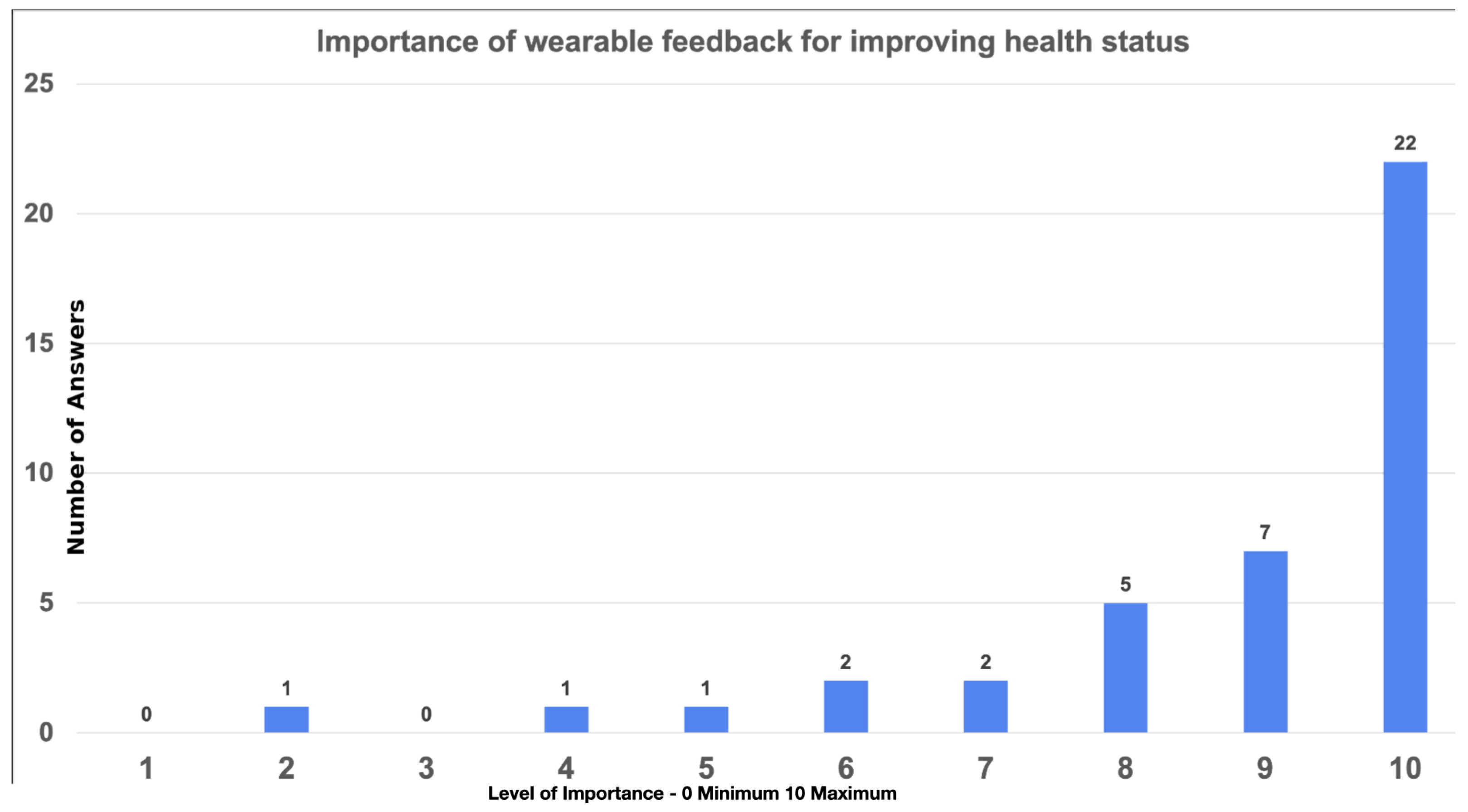
The distribution of responses to the questionnaire was highly skewed toward the maximum score (
Figure 2), with 61% of participants selecting 10 and very few responses below 8. A chi-square goodness-of-fit test was performed to evaluate whether the observed frequencies across the 10 response categories (1–10) were compatible with a uniform distribution under the null hypothesis of no preference. Using the observed counts (0, 0, 0, 0, 1, 2, 1, 5, 7, 25) and an expected count of 4.1 for each category (total
), the test yielded
with 9 degrees of freedom (df = 10 − 1), corresponding to a
p-value
, which indicates a statistically significant departure from uniformity and confirms that the responses were overwhelmingly concentrated at the upper end of the scale.
For
Figure 3, the distribution of responses to this questionnaire item was again markedly skewed toward the highest score, with 53.7% of participants selecting 10 and only a few responses in the lower range. Using the observed counts across the 10 response categories (1–10), namely (0, 1, 0, 1, 1, 2, 2, 5, 7, 22) for a total of
respondents, we tested the null hypothesis of a uniform distribution of answers by applying a chi-square goodness-of-fit test, with an expected frequency of 4.1 per category. The resulting statistic was
with 9 degrees of freedom, corresponding to a
p-value
, indicating a highly significant deviation from uniformity and confirming that responses were strongly concentrated at the upper end of the scale.
3.2. Results of the IPAQ Questionnaire
Patient responses were analyzed to determine the time spent on physical activities during the past seven days. The initial section of the survey addressed occupational activity, revealing that, while most participants were employed outside the home, the predominant group engaged neither in vigorous nor moderate work-related physical activity, with walking during work hours typically lasting less than ten minutes.
An assessment of commuting patterns showed that, over the preceding week, the majority relied on cars for daily travel, averaging approximately two hours per day in the vehicle. Active modalities such as cycling and walking were uncommon; most participants reported no use of bicycles and limited walking for their journeys.
Regarding domestic activity, patients performed minimal physical tasks in or around the home, including chores, gardening, repairs, and family care. The largest subgroup did not undertake vigorous activities (such as heavy lifting, chopping wood, or shoveling snow) or moderate activities (such as light lifting, cleaning windows, or polishing) in the prior week.
Recreational and leisure activity levels were similarly low: most patients did not walk for at least ten minutes or participate in vigorous or moderate exercise or sport during their free time within the last seven days.
The final section of the questionnaire focused on sedentary behavior, capturing time spent seated at work, home, during training courses, or during leisure activities (such as desk work, reading, socializing, or watching television). On both weekdays and weekends, most participants spent approximately two hours each day in sedentary positions.
Demographic profiling identified the largest groups as unemployed, predominantly male, and chiefly holding a middle school diploma.
3.3. Results of the SF36 Questionnaire
The majority of patients considered their health to be good and described their health as about the same as the previous year. The largest portion considered themselves partially limited in performing demanding activities like running, lifting heavy objects, or engaging in strenuous sports. In contrast, they did not consider themselves limited in activities requiring moderate physical effort (like carrying grocery bags, climbing one floor of stairs), bending or kneeling, walking one kilometer, 100 m, or a few hundred meters, taking a bath, or dressing themselves. However, a perception of partial limitation in climbing a few flights of stairs emerged. In the last 4 weeks, due to physical health, the majority of patients did not reduce the time spent at work, did not perform less than they would have liked, and did not perceive greater difficulty in doing their work or other activities but had to limit some types of work. The predominant group did not reduce their time at work due to emotional health but did perform less due to it, feeling a drop in concentration. In contrast, their physical and emotional health did not interfere with normal social activities with family and friends. The perception of physical pain was mild and did not interfere with their usual work. Over part of the time in the last 4 weeks, the largest number of patients felt “lively and bright,” “very agitated,” “calm and serene,” “full of energy,” “discouraged and sad”, almost never down or without energy, tired for a long time, and equally divided between “almost always” happy and happy “some of the time”.
The majority believed their health and emotional state hardly interfered with social activities and also stated that they enjoyed excellent health. The patients examined did not have a clear perception of being more likely to get sick than others, nor could they say whether their health was the same as others’ or whether there was a possibility of worsening.
3.4. Results of the DASS21 Questionnaire
The DASS21 questionnaire required patients to self-assess symptom frequency over the preceding week, rating each item from zero (never) to three (very often). Most respondents reported rarely experiencing difficulty relaxing and equally infrequent complaints of dry mouth. The largest proportion indicated never feeling unable to experience positive emotions, nor did they report difficulties with breathing (such as an increased respiratory rate or dyspnea at rest) or initiating activities.
Excessive reactions to specific situations were uncommon, with responses divided between “never” and “sometimes.” Tremors, significant nervousness, panic episodes, or overwhelming feelings of inadequacy were also notably absent among most patients.
Virtually no participants endorsed hopelessness about the future, while symptoms of stress and difficulty relaxing were rare. The prevailing majority did not report depression, persistent discouragement, intolerance of obstacles, feelings related to imminent panic, a widespread lack of enthusiasm, or worthlessness.
Feelings of irritability were infrequent, and very few patients noted palpitations or tachycardic sensations in the absence of physical exertion. Nearly all respondents ruled out unprovoked fear or a sense that life was meaningless.
3.5. Results Regarding the Application of the Sidly Telemonitoring System
One of the major applications of artificial intelligence is certainly in the field of telemedicine and "smart-home technologies." Thanks to remote patient monitoring and management platforms, it is possible to personalize treatment planning, define the correct medication dosage, and identify patients at risk of adverse events, anticipating potential readmissions. The concept of the “Internet of Things” (IoT) is also increasingly developed: it is based on the use of various electronic devices with embedded sensors in everyday objects, which are then associated with online platforms. The sensor records specific data related to the patient’s health status and transmits them to the remote device via specific connectors. By leveraging IoT, medical staff can monitor numerous vital parameters, recognizing early signs of potential health deterioration. The device chosen for our study was the Sidly watch, with the goal of assessing the usefulness of wearable devices in monitoring motor activity among patients with early myocardial infarction. We examined a total of 80 patients, randomly selected from a database of patients with early myocardial infarction. All patients underwent a follow-up visit, but only 66 agreed to use the device for 7 days. The device enables the remote monitoring of heart rate, oxygen saturation, and the number of steps taken each day; it also signals sudden position changes that may indicate syncopal episodes, as well as the exact time to take medication. The maximum and minimum frequency is depicted in
Figure 4. The measured heart rate is depicted in
Figure 5. The average total number of steps over 24 h was between 2000 and 4000 steps, the daytime steps were between 1200 and 2500, and the total steps in a week were between 10,000 and 20,000 (
Figure 6). The average oxygen saturation was consistently around 99%, with minimum values between 70 percent and 78.
3.6. Results on Time-Series Clustering
This section reports the results of the heart rate analysis performed using a multi-scale window extraction strategy. By partitioning the raw heart rate time series into short-, medium-, and long-range windows, we capture variability and structural patterns across distinct temporal resolutions. This approach enables a robust characterization of heart rate dynamics, simultaneously accounting for rapid fluctuations and longer-term trends while mitigating the impact of missing values through interpolation.
Figure 7 presents representative heart rate patterns identified at each temporal scale.
Short-range windows emphasize rapid variability and transient dynamics across clusters, reflecting the system’s sensitivity to brief physiological events or artifacts. Each window comprises up to six measurements, with no missing values permitted, and clustering yields 13 DBA k-means prototypes. Several clusters are strongly associated with tachycardic activity. In particular, Cluster 10 captures transitions from resting states to tachycardia, characterized by a pronounced rise in heart rate marking the onset of activity. Cluster 7 uniquely represents recovery dynamics, with heart rate declining from tachycardic levels back to rest. Cluster 5 corresponds to prolonged and severe tachycardia, with sustained heart rates exceeding 120 bpm for extended periods, in some cases lasting up to two hours. Conversely, Clusters 11 and 6 are predominantly linked to bradycardic or sleep-related patterns, exhibiting lower and more stable heart rate values indicative of reduced physiological demand. Together, these results demonstrate the ability of short-range clustering to disentangle physiologically meaningful states within long-term heart rate recordings.
Medium-range windows capture intermediate temporal structures, providing insight into ongoing physiological fluctuations and moderate trends that are not discernible at shorter scales. Each window includes up to 12 measurements, allowing for up to one missing value imputed via interpolation. Clustering at this scale yields six representative patterns, reflecting sustained yet dynamic heart rate behavior over intermediate durations.
Long-range windows reveal broad temporal dynamics and support the identification of persistent shifts or gradual modulations in heart rate. These windows comprise up to 24 measurements, with up to two missing values interpolated, and produce four distinct clusters. Clusters 3 and 4 represent outlier patterns: one describes a complete cycle from rest to tachycardia and back to rest, while the other captures a unidirectional transition from rest to tachycardia. In contrast, Clusters 1 and 2 differ primarily in their mean heart rate levels, with Cluster 1 centered around approximately 60 bpm and Cluster 2 around 75 bpm.
Overall, clustering across multiple temporal scales enables a nuanced analysis of heart rate dynamics, facilitating the discrimination between brief, intermediate, and persistent physiological changes. This multi-scale perspective provides a powerful framework for the physiological interpretation and clinically meaningful segmentation of long-term heart rate data.
To further assess the robustness of the proposed clustering framework across heterogeneous physiological signals, we extended the analysis to multiple data modalities, including pedometer-derived activity counts, oxygen saturation () levels, and heart rate measurements. This multi-sensor perspective enables the identification of complementary behavioral and physiological patterns, providing integrated insight into daily activity profiles, cardiorespiratory status, and transitions between functional states.
Figure 8 summarizes the clustering outcomes for each modality, highlighting characteristic groupings and temporal transitions reflected in step activity, oxygen saturation dynamics, and heart rate variability. For consistency across signals, medium-range windows were adopted, and the number of clusters was fixed at six. Within this setting, we systematically compared three variants of time-series k-means—Euclidean k-means, soft-DTW k-means, and DBA k-means—by varying the underlying distance metric while holding all other parameters constant. This design allowed us to evaluate the impact of shape-based versus alignment-aware similarity measures on clustering performance in physiological time series.
Following this comparative analysis, DBA k-means was selected as the primary clustering strategy for subsequent experiments. This choice reflects DBA’s ability to capture temporal dynamics through the computation of barycenters under the dynamic time warping (DTW) metric, thereby accommodating temporal shifts and non-linear distortions commonly observed in physiological signals. In contrast to Euclidean-based k-means, DBA k-means yields clusters that are more temporally coherent and physiologically interpretable, making it particularly well suited for multi-modal time-series analysis.
Finally,
Figure 9 presents an in-depth analysis of long-range heart rate patterns, revealing sustained temporal dynamics that extend across extended time intervals. By examining these patterns, we capture slow physiological modulations and trends that are often overlooked in short or medium-term analyses. This comprehensive long-range perspective enables the identification of persistent states such as prolonged tachycardia, bradycardia, or recovery phases, which have critical implications for understanding autonomic regulation and cardiovascular health. Through the clustering of these extended windows, distinct phenotypic behaviors emerge, highlighting the significance of the temporal scale in heart rate variability analysis and its potential clinical applications. This approach facilitates an improved characterization of chronic conditions and dynamic physiological responses over hours or longer periods.
Beyond descriptive visualization, the clustering results provide quantitative and qualitative evidence that wearable-derived heart rate data contain structured and clinically interpretable temporal patterns. Across all temporal scales, clusters were not arbitrarily distributed but consistently organized around physiologically meaningful regimes, including resting states, activity-induced tachycardia, recovery phases, and sustained autonomic activation. The recurrence of these patterns across individuals and window lengths supports the internal consistency of the analytical framework and indicates that the extracted clusters reflect underlying cardiac dynamics, rather than noise or random variability. Notably, the identification of low-populated clusters capturing extreme or transitional heart rate behaviors demonstrates the sensitivity of the approach to atypical yet clinically relevant events that would likely be missed through conventional averaging or threshold-based monitoring. Importantly, the clustering framework aims not to diagnose pathological conditions but to characterize recovery-related physiological variability in a post-myocardial infarction population. The results show that continuous wearable monitoring, when combined with time-series analysis, can stratify patients according to dynamic heart rate behaviors over daily life. This stratification provides a richer representation of post-infarction physiology than isolated summary metrics such as the mean heart rate or the total step count, thereby supporting the conclusion that wearable devices can yield clinically meaningful information when interpreted through appropriate analytical models.
4. Discussion
This study introduces a multi-scale, unsupervised analytical framework for characterizing post-myocardial infarction (MI) physiology using longitudinal data acquired from wearable sensors. By decomposing heart rate dynamics across short-, medium-, and long-range temporal windows, the proposed clustering strategy reveals latent autonomic patterns that are not accessible through conventional summary statistics or threshold-based monitoring. This approach enables a form of digital phenotyping, in which physiologically meaningful states emerge directly from a data-driven temporal structure.
At short temporal scales, clustering isolated rapid fluctuations and transient autonomic responses. Distinct clusters captured transitions from rest to tachycardia, recovery trajectories indicative of parasympathetic reactivation, prolonged episodes of sustained tachycardia, and bradycardic or sleep-associated states characterized by autonomic quiescence. The ability to discriminate between activation, recovery, and sustained stress-related patterns highlights the sensitivity of short-range clustering to rapid autonomic regulation and transient perturbations.
Medium-range windows provided an intermediate temporal resolution, capturing autonomic modulations that extend beyond brief fluctuations but do not yet reflect fully sustained physiological states. These clusters revealed diurnal oscillations, stabilization phases during recovery, and activity-associated variability, offering insight into the consistency and adaptability of autonomic control during daily life. Importantly, such patterns are often obscured in short-term analyses and diluted in long-term averages.
At longer temporal scales, clustering identified stable physiological phenotypes persisting over several hours. These included complete cycles from rest to tachycardia and back, unidirectional transitions toward sustained tachycardia, and clusters differentiated primarily by baseline heart rate levels. This long-range phenotyping exposes heterogeneity in resting autonomic tone and recovery profiles among young post-MI patients, potentially reflecting differences in autonomic balance, circadian regulation, or cardiovascular conditioning.
From a clinical standpoint, the results demonstrate that wearable-derived data are not merely descriptive but actionable when contextualized temporally. For example, long-range clusters characterized by a persistently elevated baseline heart rate or sustained tachycardia may indicate delayed autonomic recovery or suboptimal rehabilitation engagement, whereas clusters dominated by stable low heart rate patterns are consistent with adequate recovery and parasympathetic reactivation. The ability to distinguish these phenotypes using unsupervised analysis supports the conclusion that wearable monitoring can complement traditional follow-up by revealing latent recovery trajectories that are otherwise inaccessible during intermittent clinical visits.
Extending the clustering framework to pedometer-derived activity counts and oxygen saturation further demonstrated its multimodal applicability. Recurrent patterns linked activity intensity to predictable heart rate responses, while sporadic desaturation clusters emerged as distinct outliers requiring clinical interpretation. Comparative evaluation of Euclidean, soft-DTW, and DBA k-means confirmed that alignment-aware clustering—specifically DBA k-means—more effectively captures the intrinsic temporal variability of physiological signals, reinforcing the importance of time-warp–tolerant methods for wearable-derived data.
From a clinical perspective, early MI—particularly in younger individuals—poses a paradox: patients often regain functional independence yet remain exposed to long-term cardiovascular risk. Traditional follow-up strategies, based on intermittent assessments, are poorly suited to detect subtle physiological deviations or maladaptive recovery trajectories. The findings of this study support wearable-based remote monitoring as a feasible and clinically meaningful complement to standard post-MI care.
Patient acceptance of digital monitoring was high, with a strong preference for smartwatch-based systems and a clear recognition of the importance of reimbursement by national health systems to ensure equitable adoption. Notably, participants overwhelmingly favored personalized feedback delivered by healthcare professionals, underscoring that effective digital health solutions must augment—not replace—clinical relationships. This hybrid model aligns closely with current paradigms in digital medicine, which emphasize human-in-the-loop systems for trust, adherence, and safety.
Objective monitoring revealed low overall physical activity levels, stable oxygen saturation, and predominantly normal heart rate profiles punctuated by rare bradycardic episodes. These findings are consistent with known post-MI recovery patterns, in which reduced activity and subtle autonomic dysregulation may persist despite subjective well-being. Continuous monitoring enables the detection of asymptomatic but potentially relevant events, such as episodic bradycardia or prolonged tachycardia, which may otherwise go unnoticed yet warrant clinical attention.
Importantly, the clustering-based framework moves beyond simple alerting systems by contextualizing physiological signals within temporal patterns. For example, sustained tachycardia clusters may identify individuals requiring intensified rehabilitation, medication adjustment, or closer follow-up, while long-range phenotypes characterized by an elevated baseline heart rate may signal chronic sympathetic predominance and increased long-term risk. Similarly, multimodal patterns linking activity and cardiorespiratory responses can inform personalized rehabilitation intensity and pacing.
More broadly, this approach supports the development of scalable, data-driven secondary prevention strategies. By enabling an automated yet interpretable characterization of physiological states, multi-scale clustering can underpin early warning systems, adaptive rehabilitation programs, and machine-learning–assisted clinical decision support. Such tools are particularly relevant for younger post-MI populations, who are often underrepresented in cardiovascular research despite their extended lifetime risk horizon.
In summary, this work demonstrates how wearable sensing, combined with advanced time-series analytics, can bridge the gap between continuous data acquisition and clinically actionable insight. By aligning methodological innovation with patient-centered design and healthcare system integration, this framework contributes to the evolving landscape of digital cardiovascular medicine and highlights the potential of multi-scale digital phenotyping to support personalized, long-term post-MI care.
To make the clinical translation explicit, clusters can be mapped to predefined actions within a telemonitoring-supported rehabilitation pathway. Short-range clusters capturing minute-to-hour dynamics support real-time safety and workload control: sustained tachycardia patterns (e.g., heart rate above 120 beats per minute for 90–120 min or repeated rest-to-tachycardia transitions within 24 h) would automatically trigger a nurse triage alert on the monitoring dashboard, followed by telephone contact within 24 h to assess symptoms, adherence to exercise prescription, hydration, and medication intake, with escalation to a cardiologist for pharmacological review (for example, beta-blocker titration) or a temporary reduction in training intensity if the pattern persisted for at least three days. Abrupt heart rate variability or frequent transitions not aligned with scheduled exercise sessions would instead prompt targeted lifestyle counseling delivered remotely, focusing on pacing, warm-up and cool-down routines, and stress management, whereas stable low–heart rate clusters compatible with sleep or deep rest predominantly occurring during night-time windows would be documented as physiological recovery without triggering active intervention, providing reassurance to both clinicians and patients.
Medium-range clusters summarizing day-level behavior over several days inform adherence and behavioral monitoring, while long-range clusters extending over weeks enable risk stratification and pathway modulation. Clusters characterized by low step counts and a blunted heart rate response during daytime hours for at least five consecutive days would lead to a nurse-led motivational interview to reinforce rehabilitation goals and explore barriers to activity such as fatigue, anxiety, or musculoskeletal limitations, whereas clusters showing excessive activity combined with an elevated mean heart rate would prompt an adjustment of the exercise prescription and education on safe exertion thresholds. Persistent assignment to sedentary or high resting heart rate clusters over two to three weeks would trigger reclassification to a “sub-optimal recovery” pathway with prioritized in-person assessment and, where appropriate, additional imaging or functional testing, while progressive migration from sedentary to more active clusters with a stable heart rate would support the continuation of the standard rehabilitation pathway with routine follow-up only.
In sum, our findings affirm remote monitoring’s utility as an adjunct to conventional post-MI management. When supported through appropriate clinical oversight and human interaction, wearable technologies can facilitate improved recovery trajectories, reduced hospitalizations, and the emergence of a more proactive, responsive, and sustainable model of cardiovascular care.
While the study is exploratory in nature, the convergence of multiple data sources—objective wearable measurements, validated questionnaires, and consistent clustering behavior across temporal scales—provides convergent evidence supporting the study’s conclusions. The consistency of observed patterns across heart rate, activity, and oxygen saturation further reinforces the robustness of the findings. Although larger cohorts and longer monitoring periods are required for predictive validation, the present results are sufficient to demonstrate feasibility, interpretability, and clinical relevance of wearable-based multi-scale monitoring among young post-myocardial infarction patients.
5. Limitations
Although wearable devices such as Sidly have provided good accuracy for basic monitoring (heart rate, oxygen saturation, and motor activity), some parameters, such as detecting postural changes in cases of syncope episodes, may be influenced by errors or false positives. It is important to improve the sensitivity of the sensors to minimize such limitations, perform a rigorous validation of wearable devices in real clinical scenarios, and, if necessary, integrate data from the devices with other forms of monitoring or medical consultations. Moreover, the economic sustainability and reimbursement of the device via the national healthcare system were seen as essential for wider technology dissemination. Cost and accessibility are determining factors for the continuation of long-term monitoring and for expanding its use. An additional limitation of the study is the small sample size of patients monitored. The small sample size may compromise the generalizability of the results and increase the risk of biases, as limited samples may not adequately represent the general population. This could lead to unreliable conclusions or an overestimation or underestimation of the effectiveness of the monitored devices. Expanding the sample in future studies will provide more robust and meaningful results, improving the validity and reliability of the conclusions.
A critical aspect that requires attention is the limited duration of monitoring (only 7 days), which, while allowing for immediate and useful data collection to assess the initial treatment response, limits the ability to analyze the long-term effects of wearable devices on post-myocardial infarction recovery and recurrence prevention. Monitoring patients over a more extended period would allow the observation of how the consistency of device use may affect vital parameters over time and whether adopting wearable technologies can indeed reduce the risk of future cardiovascular events.
The heterogeneity of the sample represents another limitation, as the lack of detailed stratification based on relevant factors such as the severity of the cardiac event, comorbidities (e.g., diabetes, hypertension), and the level of physical activity before the infarction can introduce significant biases into the results. Patients with different severities of infarction or pre-existing health conditions may respond very differently to wearable device use, making it more challenging to draw universal conclusions about the effectiveness of remote monitoring. In the future, it would be useful to stratify data for these factors in order to better understand how remote monitoring adapts to the specific needs of different patient types.
These limitations must be considered when planning the future large-scale implementation of remote monitoring through wearable devices. Overcoming the limited study duration, addressing sample heterogeneity, and improving measurement precision are crucial steps to ensure that wearable devices can be truly useful in clinical practice to monitor early myocardial infarction patients effectively and safely over the long term.
6. Future Developments
Future developments of this study will aim to expand the sample and extend the follow-up period to collect long-term data that can provide a more comprehensive view of the effectiveness of remote monitoring and recurrence prevention among young patients with early myocardial infarction. Additionally, we plan to include an economic analysis to assess the sustainability of the proposed model, aiming to analyze not only clinical benefits but also the costs associated with large-scale implementation. The economic component is crucial to evaluate the practical feasibility and potential impact of remote monitoring in the healthcare context. Finally, another step may include a direct comparison between different wearable devices to strengthen technological recommendations and select the most effective and cost-efficient solutions to integrate and expand the results obtained so far.
7. Conclusions
The results of this study confirm that wearable devices like Sidly offer significant benefits in monitoring motor activity and vital parameters in patients with early myocardial infarction. Remote monitoring, combined with continuous and personalized feedback, can facilitate recovery and reduce the risk of complications. However, it is necessary to improve the precision of the data (especially regarding acute event detection) and ensure that the healthcare system adequately supports the adoption of these technologies.
Additionally, the active participation of patients, which proved to be crucial in our study, suggests that technology should be integrated into a context that includes both self-management by the patient and continuous support from healthcare professionals. Only through an integrated approach, combining technological innovation and effective communication between the patient and the medical team, will it be possible to maximize the effectiveness of remote monitoring for patients with early myocardial infarction.
Future developments may include the improvement of wearable devices, integration with other healthcare technologies, and a deeper evaluation of the long-term impact of these devices on physical recovery and quality of life for patients.
What emerged from our study could have a significant impact on the applicability of such devices in clinical practice. We consider it essential to promote the implementation of a remote monitoring protocol by integrating wearable devices such as Sidly into the post-infarction follow-up routine. Healthcare professionals could define personalized protocols for each patient, using real-time data to intervene promptly in the case of vital parameter anomalies or during physical activity. The creation of automatic alerts for critical events, such as reduced oxygen saturation or bradycardia episodes, could enhance treatment effectiveness.
At the same time, we believe that patient education and awareness are critical: active patient participation was a crucial component of our study. To maximize the benefits of remote monitoring, it is essential that patients receive proper training on the correct use of devices and the interpretation of the data collected. Targeted education sessions could improve self-management and patient awareness of their health status. This would enable the definition of individualized therapeutic paths: data collected via wearable devices could be used to further personalize post-infarction treatment. For example, a patient showing signs of low physical activity or an irregular heart rate could benefit from a more targeted cardiovascular rehabilitation program. In this context, remote monitoring would allow a dynamic adaptation of therapies.
However, economic sustainability and the reformulation of healthcare policies represent the biggest challenges. An important next step for the widespread adoption of technologies like Sidly is the support of the healthcare system. Policies should include incentives for healthcare professionals to adopt remote monitoring devices and ensure financial coverage for patients. Economic sustainability is crucial to make it an available and accessible tool on a large scale. Finally, we recommend that wearable device manufacturers invest in technological improvements, focusing on sensor accuracy and refining the ability to detect acute events. Additionally, further clinical studies should explore the long-term impact of remote monitoring, assessing not only the clinical benefits but also its effect on quality of life, reduction in relapses, and adherence to therapy.
In conclusion, the introduction of remote monitoring devices in the management of myocardial infarction could represent a breakthrough in treatment personalization and complication prevention. However, the real success of this approach will depend on the synergistic integration of technology, healthcare professionals, and patients.