On the Determination of Magnesium Degradation Rates under Physiological Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Specimen Preparation and Sterilization
2.2.1. Mg-2Ag Alloy
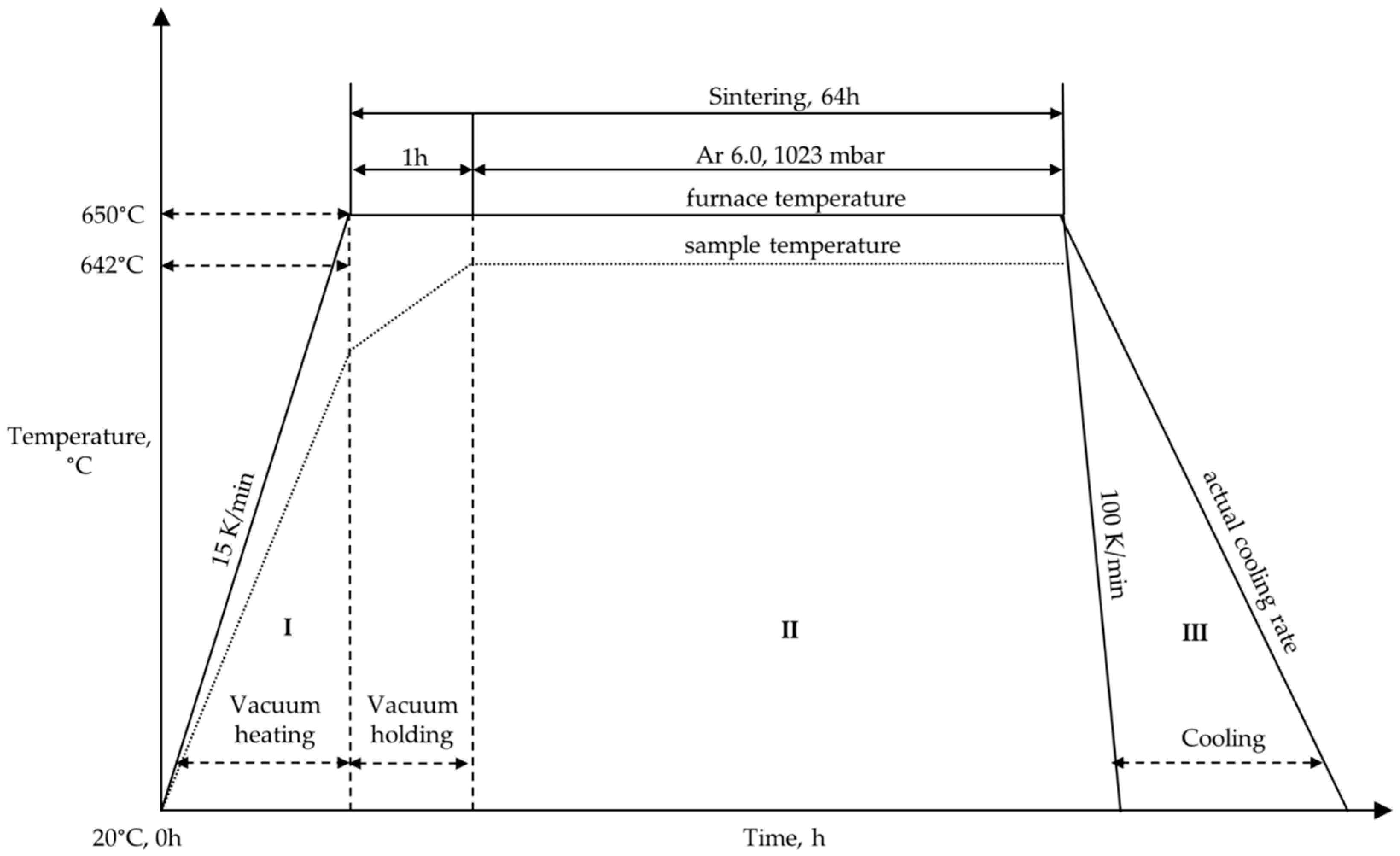
2.2.2. Mg-0.3Ca Alloy
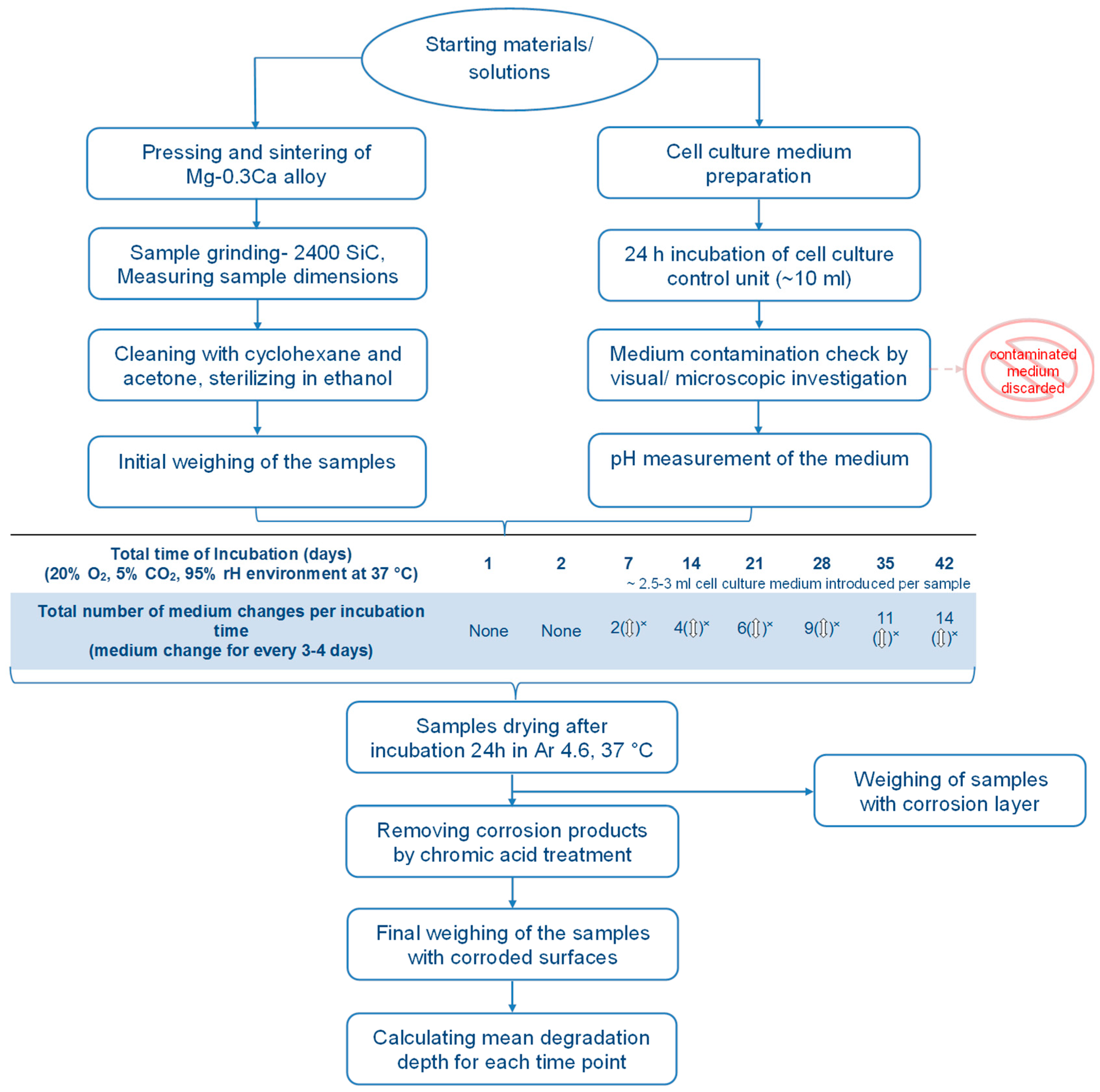
2.3. Immersion Test
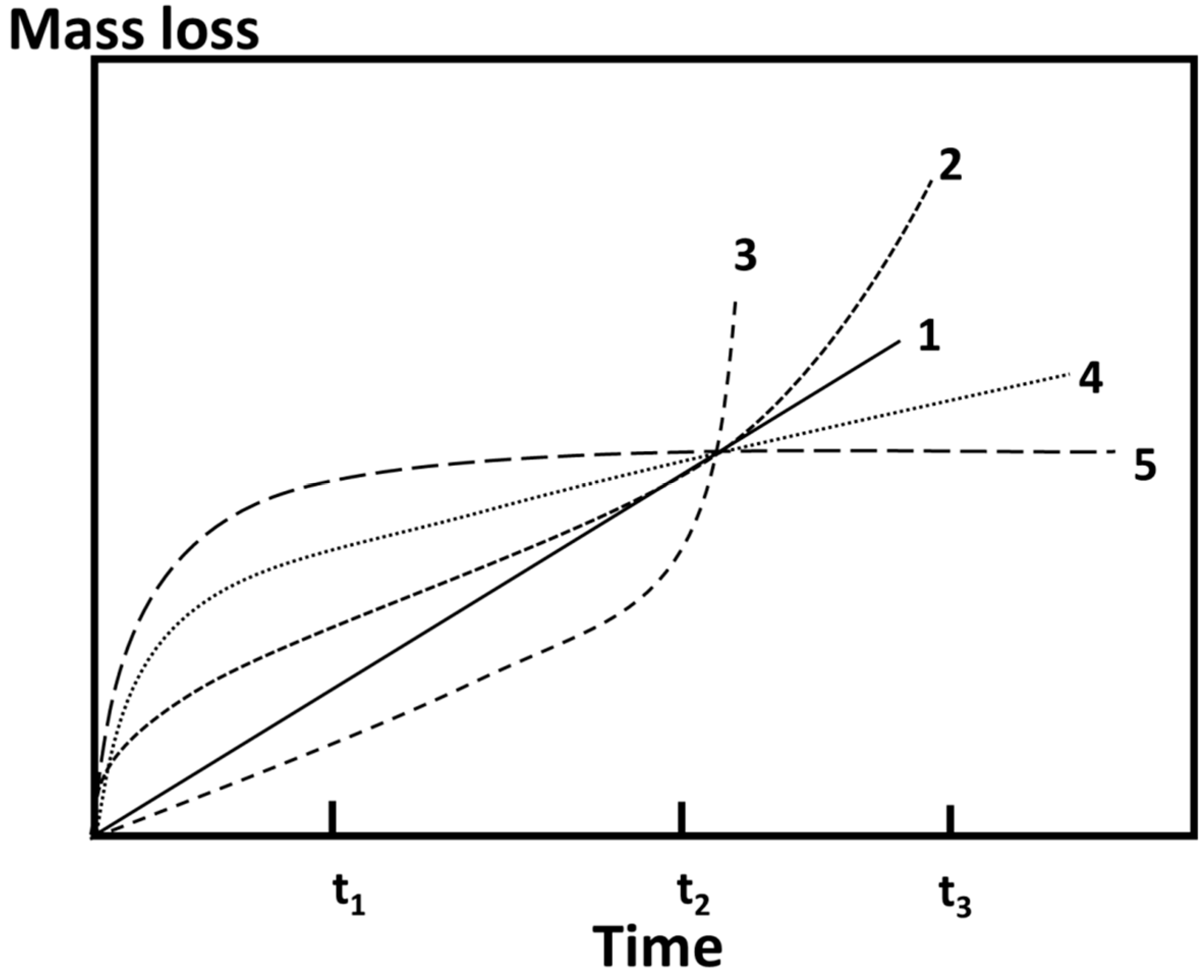
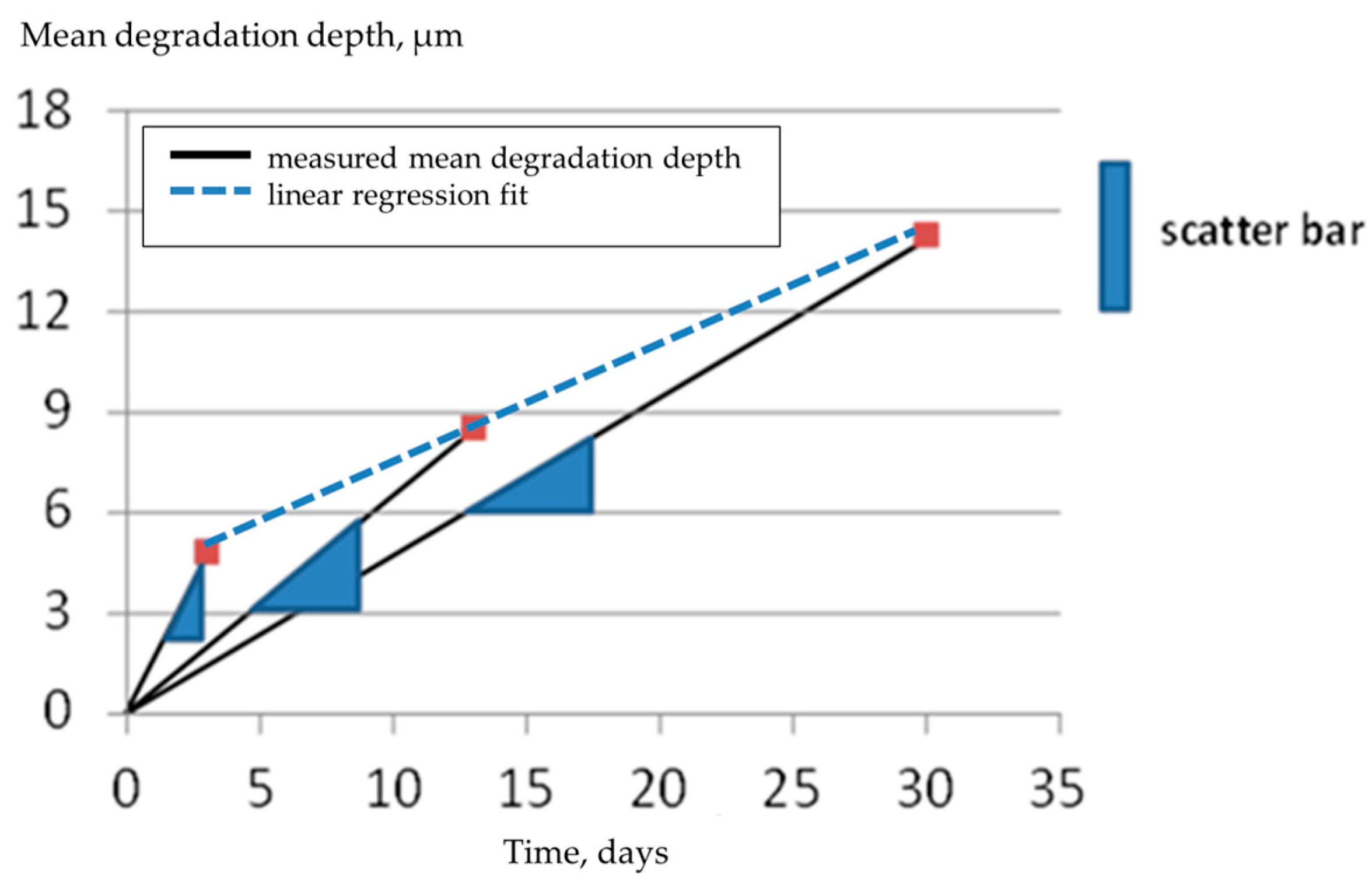
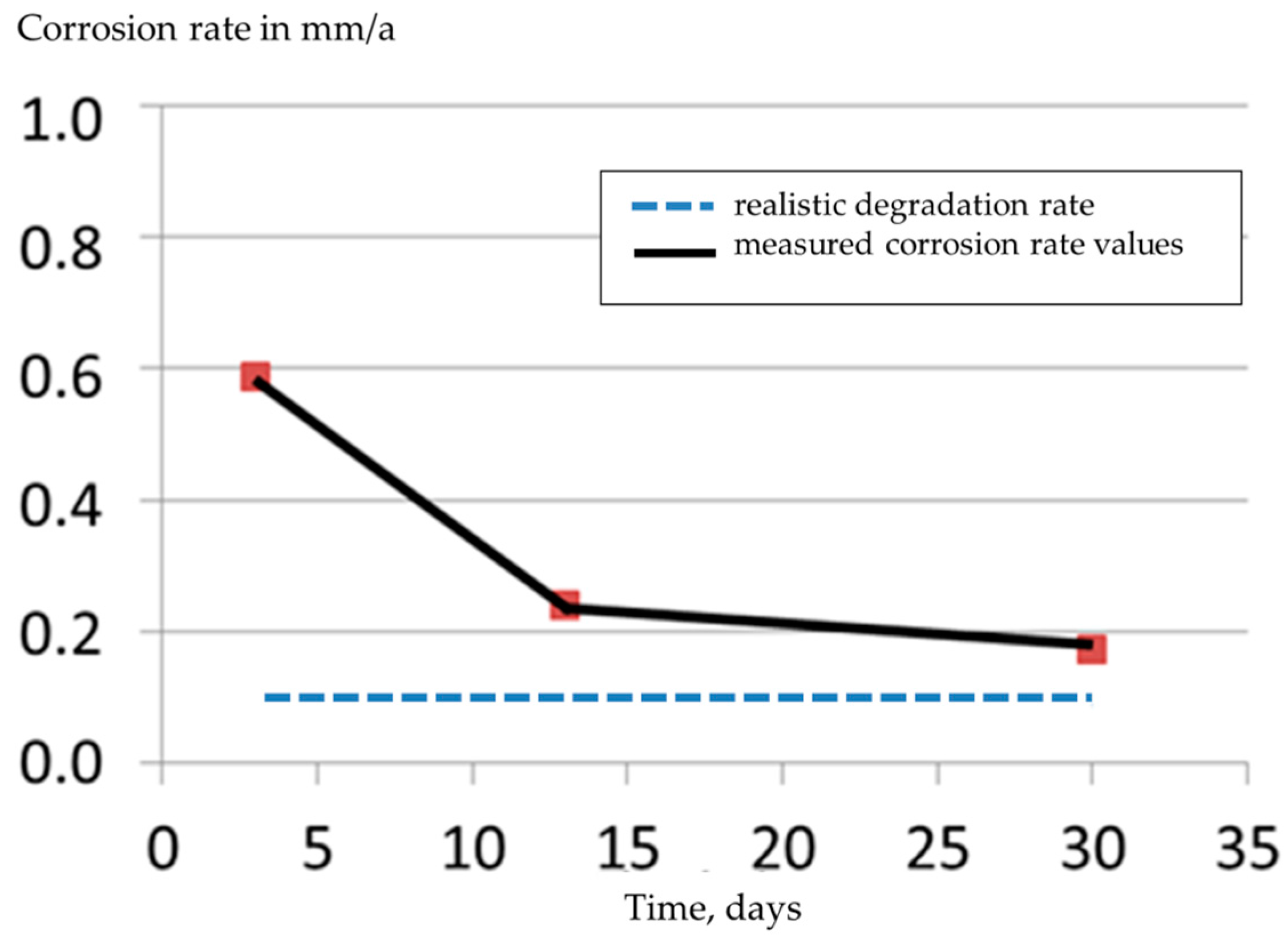
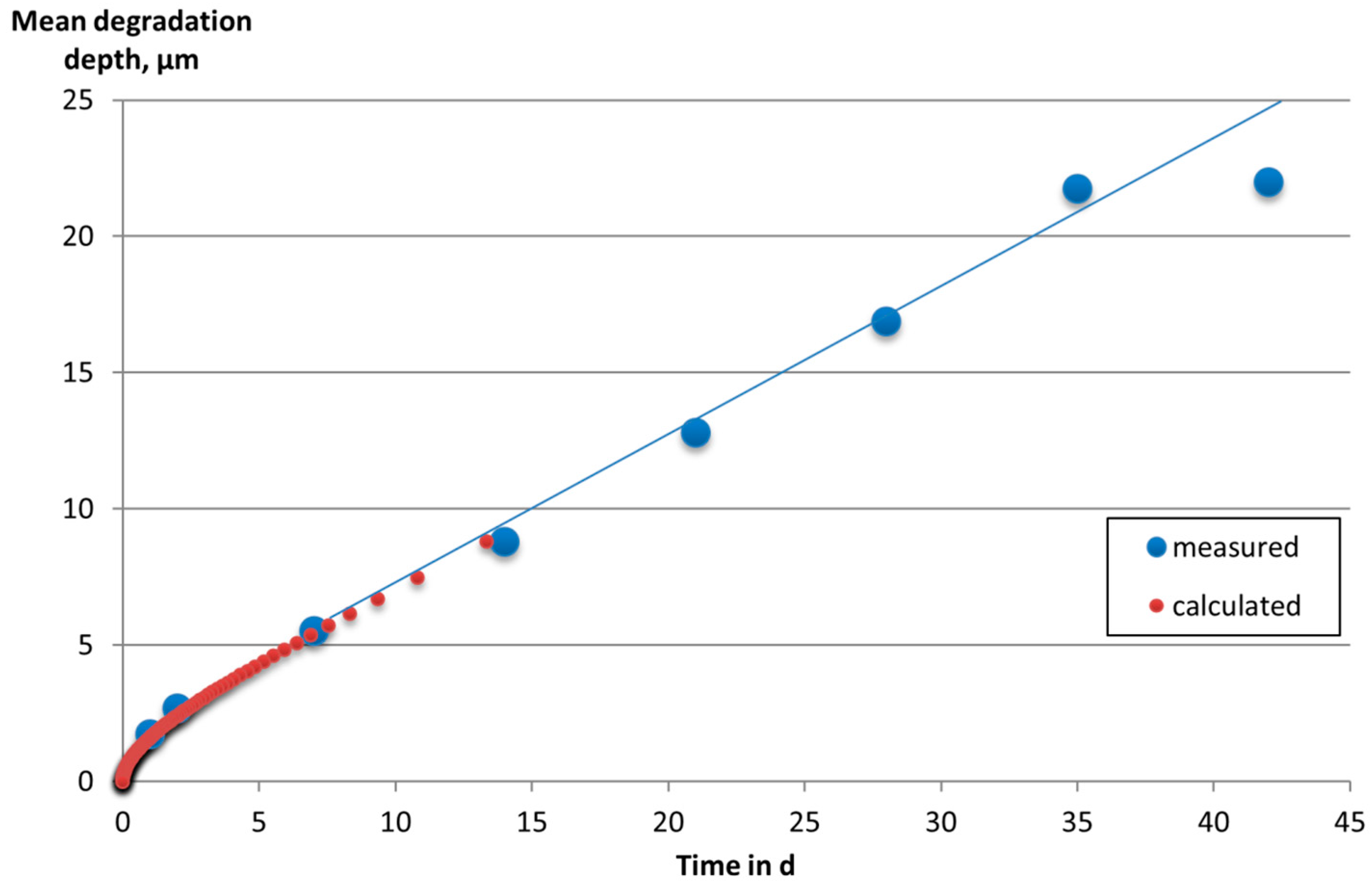
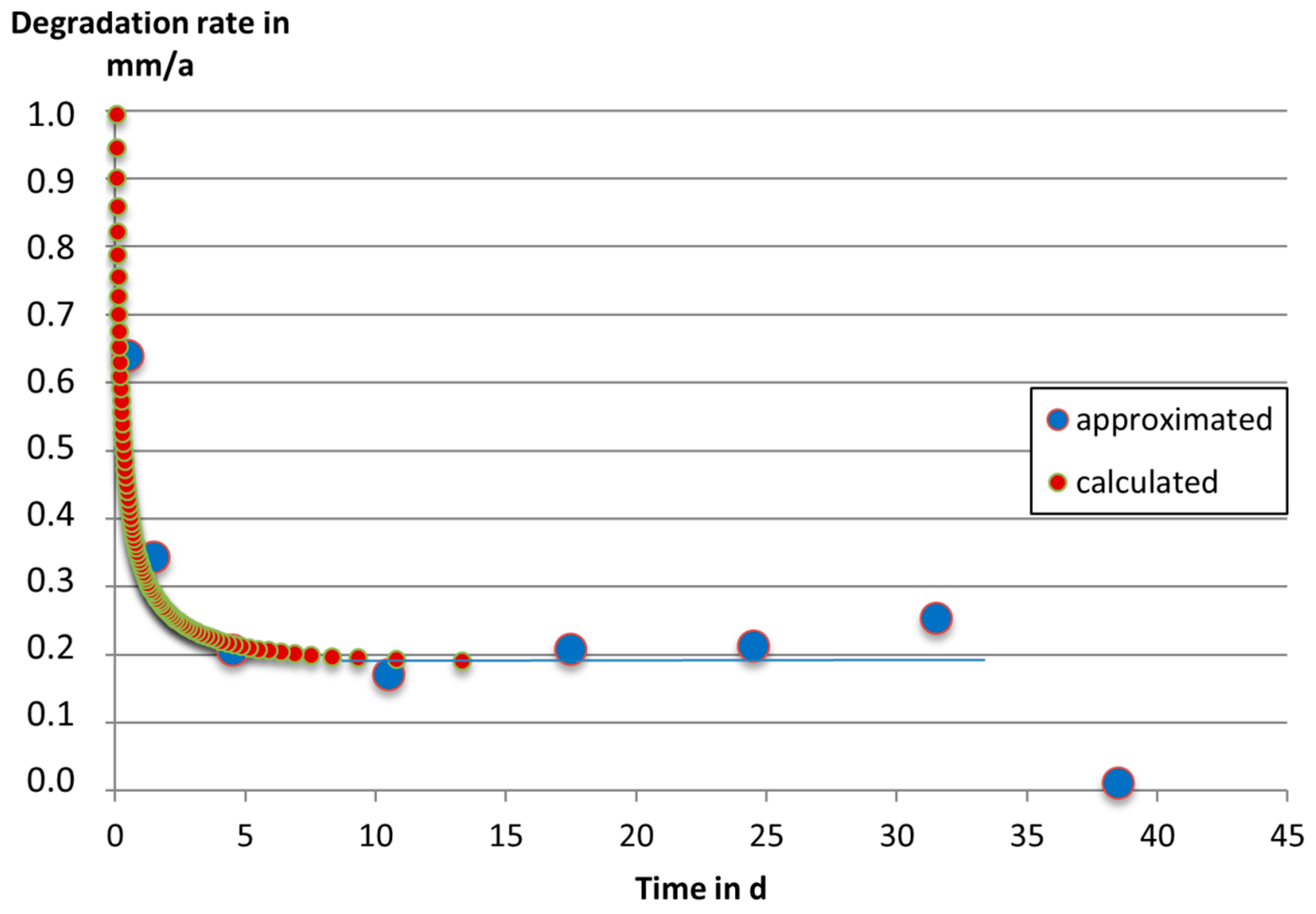
3. Results and Discussion
4. Conclusions
- Higher probability in determining the realistic corrosion rate of the alloy.
- The amount of samples needed for this model is highly reduced compared to statistical approaches (at least n = 6 per time point).
- With increasing incubation time, the risk of medium contamination gets higher. If the contamination occurs for a certain time point during immersion test, the measurement can be easily repeated from the start until the required time point without disturbing the existing setup.
- The reduction of the amount of samples makes it possible to experiment with more alloys at the same time.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, N.; Zheng, Y. Novel Magnesium Alloys Developed for Biomedical Application: A Review. J. Mater. Sci. Technol. 2013, 29, 489–502. [Google Scholar] [CrossRef]
- Expert group on Vitamins. Safer upper levels for Vitamins and Minerals; Food Standards Agency: London, UK, 2003. [Google Scholar]
- He, G.; Wu, Y.; Zhang, Y.; Zhu, Y.; Liu, Y.; Li, N.; Li, M.; Zheng, G.; He, B.; Yin, Q.; et al. Addition of Zn to the ternary Mg–Ca–Sr alloys significantly improves their antibacterial properties. J. Mater. Chem. B 2015, 3, 6676–6689. [Google Scholar] [CrossRef] [PubMed]
- Lock, J.Y.; Wyatt, E.; Upadhyayula, S.; Whall, A.; Nunez, V.; Vullev, V.I.; Liu, H. Degradation and antibacterial properties of magnesium alloys in artificial urine for potential resorbable ureteral stent applications. J. Biomed. Mater. Res. Part A 2014, 102, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Jang, T.-S.; Jung, H.-D.; Kim, S.-M.; Kim, H.-E.; Koh, Y.-H.; Song, J. Poly(ether imide)-silica hybrid coatings for tunable corrosion behavior and improved biocompatibility of magnesium implants. Biomed. Mater. 2016, 11, 035003. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.X.; Cui, F.Z.; Yin, Q.S.; Zhang, Y.; Zhang, T.; Wang, X.M. Characterization and Degradation study of Calcium Phosphate Coating on Magnesium Alloy. Bone Implant. IEEE Trans. Plasma Sci. 2009, 37, 1161–1168. [Google Scholar] [CrossRef]
- Kirkland, N.T.; Birbilis, N.; Staiger, M.P. Assessing the corrosion of biodegradable magnesium implants: A critical review of current methodologies and their limitations. Acta Biomater. 2012, 8, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.H.M.; Luthringer, B.J.C.; Feyerabend, F.; Willumeit, R. Mg and Mg alloys: How comparable are in vitro and in vivo corrosion rates? A review. Acta Biomater. 2015, 13, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Jung, O.; Smeets, R.; Porchetta, D.; Kopp, A.; Ptock, C.; Müller, U.; Heiland, M.; Schwade, M.; Behr, B.; Kröger, N.; et al. Optimized in vitro procedure for assessing the cytocompatibility of magnesium-based biomaterials. Acta Biomater. 2015, 23, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Witte, F.; Xi, T.; Zheng, Y.; Yang, K.; Yang, Y.; Zhao, D.; Meng, J.; Li, Y.; Li, W.; et al. Recommendation for modifying current cytotoxicity testing standards for biodegradable magnesium-based materials. Acta Biomater. 2015, 21, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Standard Guide for Laboratory Immersion Corrosion Testing of Metals; ASTM International: West Conshohocken, PA, USA, 2012.
- Feyerabend, F. 8—In Vitro Analysis of Magnesium Corrosion in Orthopaedic Biomaterials. In Biomaterials for Bone Regeneration; Dubruel, P., Vlierberghe, S.V., Eds.; Woodhead Publishing: Cambridge, UK, 2014; pp. 225–269. [Google Scholar]
- Gu, X.; Zheng, Y.; Cheng, Y.; Zhong, S.; Xi, T. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, N.T.; Lespagnol, J.; Birbilis, N.; Staiger, M.P. A survey of bio-corrosion rates of magnesium alloys. Corros. Sci. 2010, 52, 287–291. [Google Scholar] [CrossRef]
- Xin, Y.; Hu, T.; Chu, P.K. In vitro studies of biomedical magnesium alloys in a simulated physiological environment: A review. Acta Biomater. 2011, 7, 1452–1459. [Google Scholar] [CrossRef] [PubMed]
- Agha, N.A.; Feyerabend, F.; Mihailova, B.; Heidrich, S.; Bismayer, U.; Willumeit-Romer, R. Magnesium degradation influenced by buffering salts in concentrations typical of in vitro and in vivo models. Mater. Sci. Eng. C 2016, 58, 817–825. [Google Scholar] [CrossRef] [PubMed]
- Marco, I.; Feyerabend, F.; Willumeit-Romer, R.; Van der Biest, O. Degradation testing of Mg alloys in Dulbecco's modified eagle medium: Influence of medium sterilization. Mater. Sci. Eng. C 2016, 62, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Wolff, M.; Ebel, T.; Dahms, M. Sintering of Magnesium. Adv. Eng. Mater. 2010, 12, 829–836. [Google Scholar] [CrossRef]
- Ning, C.; Zhou, L.; Zhu, Y.; Li, Y.; Yu, P.; Wang, S.; He, T.; Li, W.; Tan, G.; Wang, Y.; et al. Influence of Surrounding Cations on the Surface Degradation of Magnesium Alloy. Implants under a Compressive Pressure. Langmuir 2015, 31, 13561–13570. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.C.; Liu, C.L.; Zhang, X.M.; Tang, G.Y.; Tian, X.B.; Chu, P.K. Corrosion behavior of biomedical AZ91 magnesium alloy in simulated body fluids. J. Mater. Res. 2007, 22, 2004–2011. [Google Scholar] [CrossRef]
| Material | Particle Size/Type | Manufacturer |
|---|---|---|
| Pure Mg powder | Spherical powder <45 µm | Société pour la Fabrication du Magnésium, Martigny, Switzerland |
| Master alloy powder (MAP) Mg-10Ca | Spherical powder 45–63 µm | Zentrum für Funktionswerkstoffe gemeinnützige GmbH, Clausthal, Germany and HZG |
| Pure Mg | Ingot | Xinxiang Jiuli Magnesium Co., Ltd, Xinxiang, China |
| Pure silver | granules | ESG Edelmetall-Handel GmbH. & Co. KG, Rheinstetten, Germany |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nidadavolu, E.P.S.; Feyerabend, F.; Ebel, T.; Willumeit-Römer, R.; Dahms, M. On the Determination of Magnesium Degradation Rates under Physiological Conditions. Materials 2016, 9, 627. https://doi.org/10.3390/ma9080627
Nidadavolu EPS, Feyerabend F, Ebel T, Willumeit-Römer R, Dahms M. On the Determination of Magnesium Degradation Rates under Physiological Conditions. Materials. 2016; 9(8):627. https://doi.org/10.3390/ma9080627
Chicago/Turabian StyleNidadavolu, Eshwara Phani Shubhakar, Frank Feyerabend, Thomas Ebel, Regine Willumeit-Römer, and Michael Dahms. 2016. "On the Determination of Magnesium Degradation Rates under Physiological Conditions" Materials 9, no. 8: 627. https://doi.org/10.3390/ma9080627
APA StyleNidadavolu, E. P. S., Feyerabend, F., Ebel, T., Willumeit-Römer, R., & Dahms, M. (2016). On the Determination of Magnesium Degradation Rates under Physiological Conditions. Materials, 9(8), 627. https://doi.org/10.3390/ma9080627







