Crack Healing Performance of PVA-Coated Granules Made of Cement, CSA, and Na2CO3 in the Cement Matrix
Abstract
:1. Introduction
Self-Healing through Granulation and Film Coating
2. Experiment Procedure
2.1. Granulation/Coating Materials
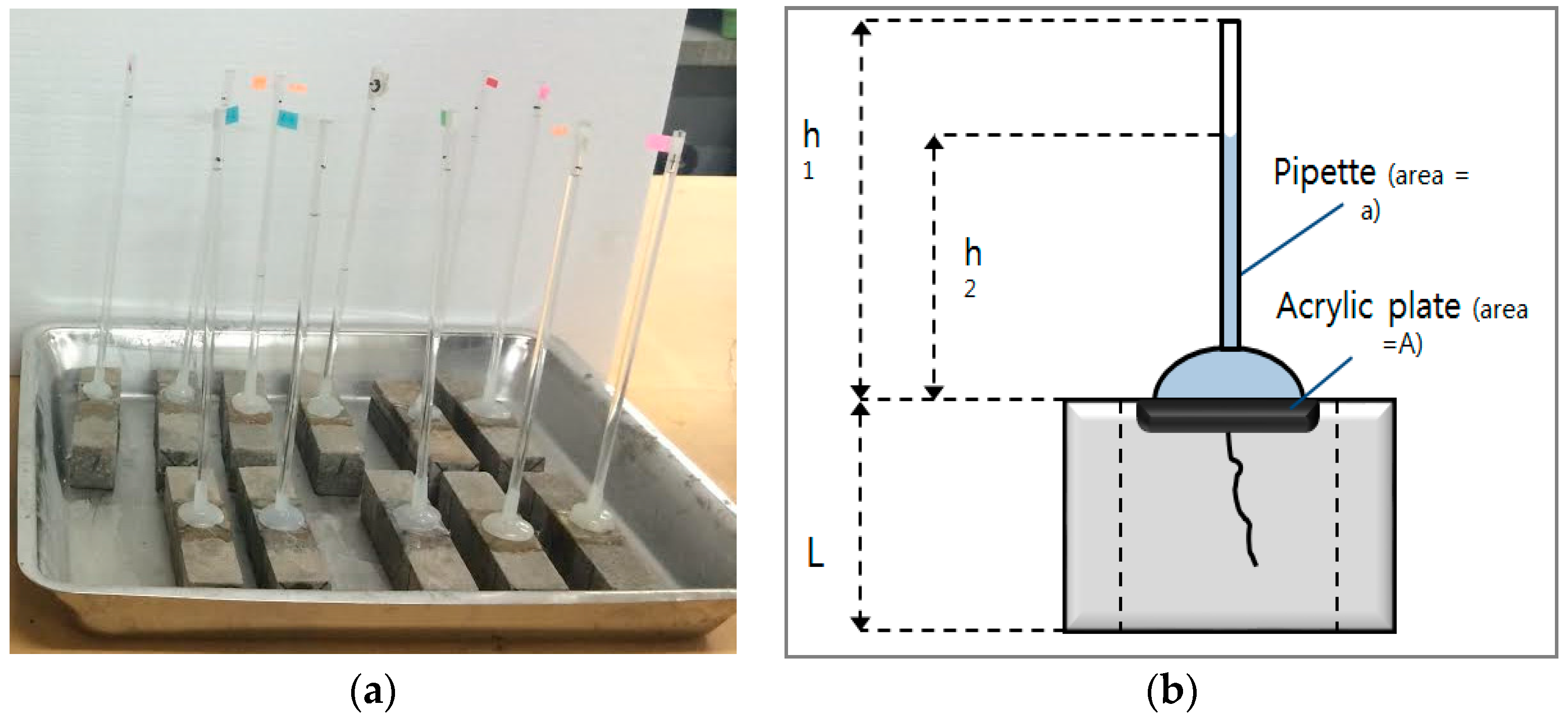
2.2. Preparation of Specimens for Evaluating the Healing Efficiency
2.3. Evaluation of the Healing Efficiency
3. Results and Discussion
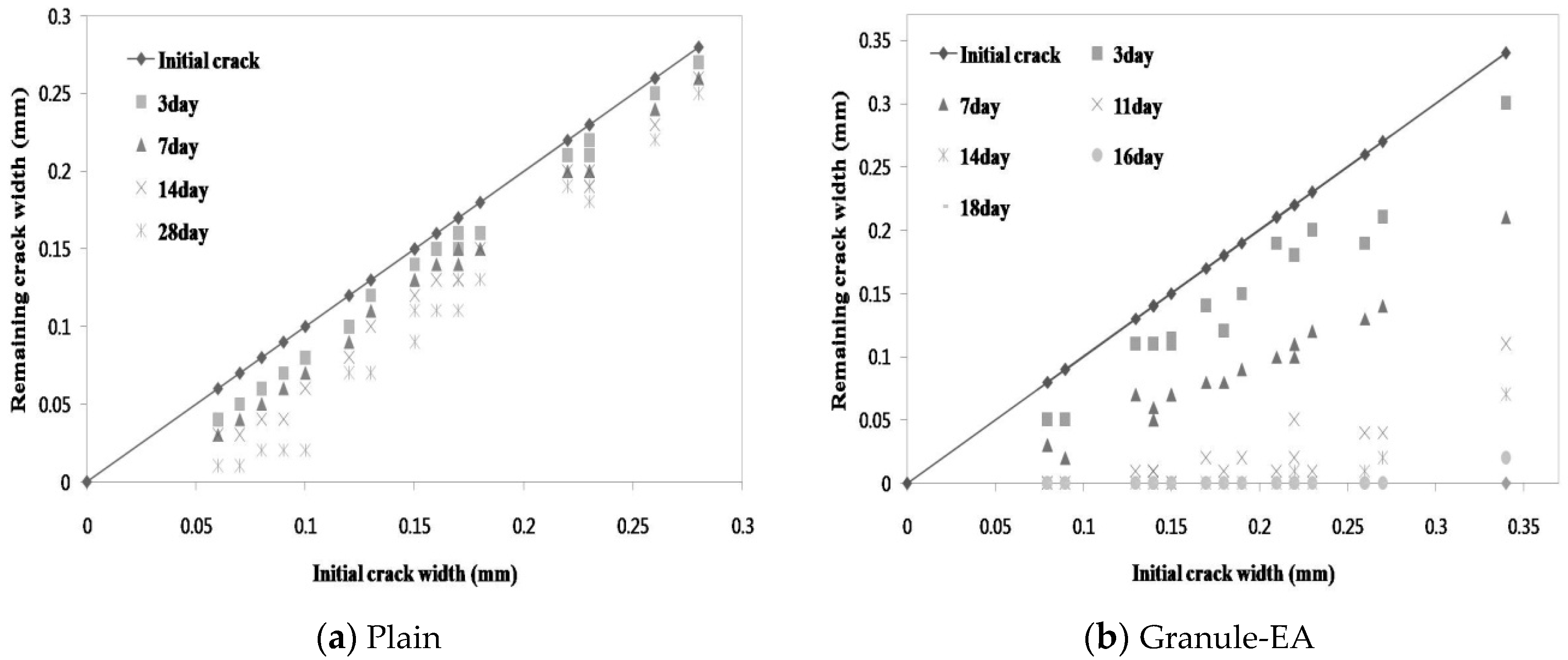
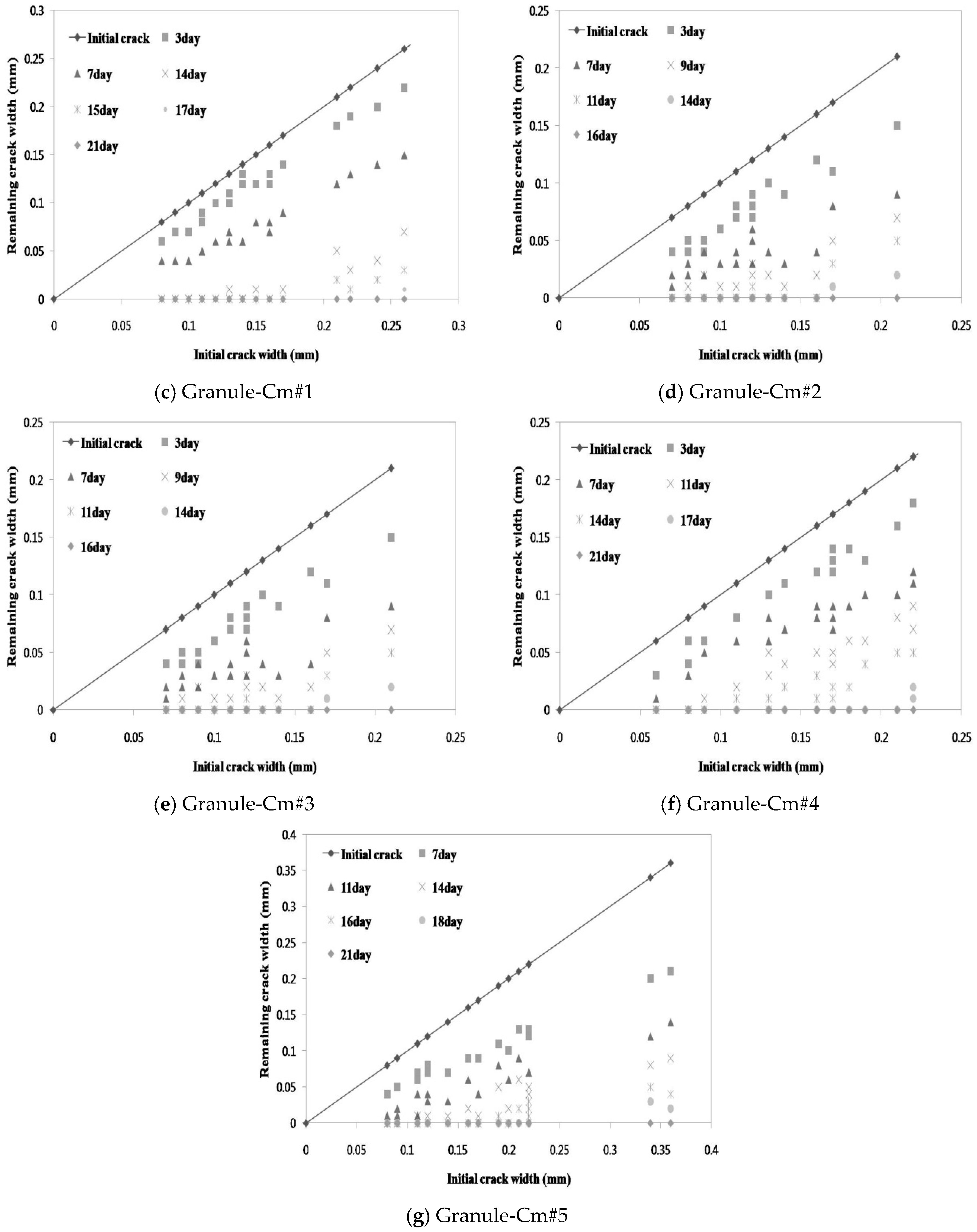
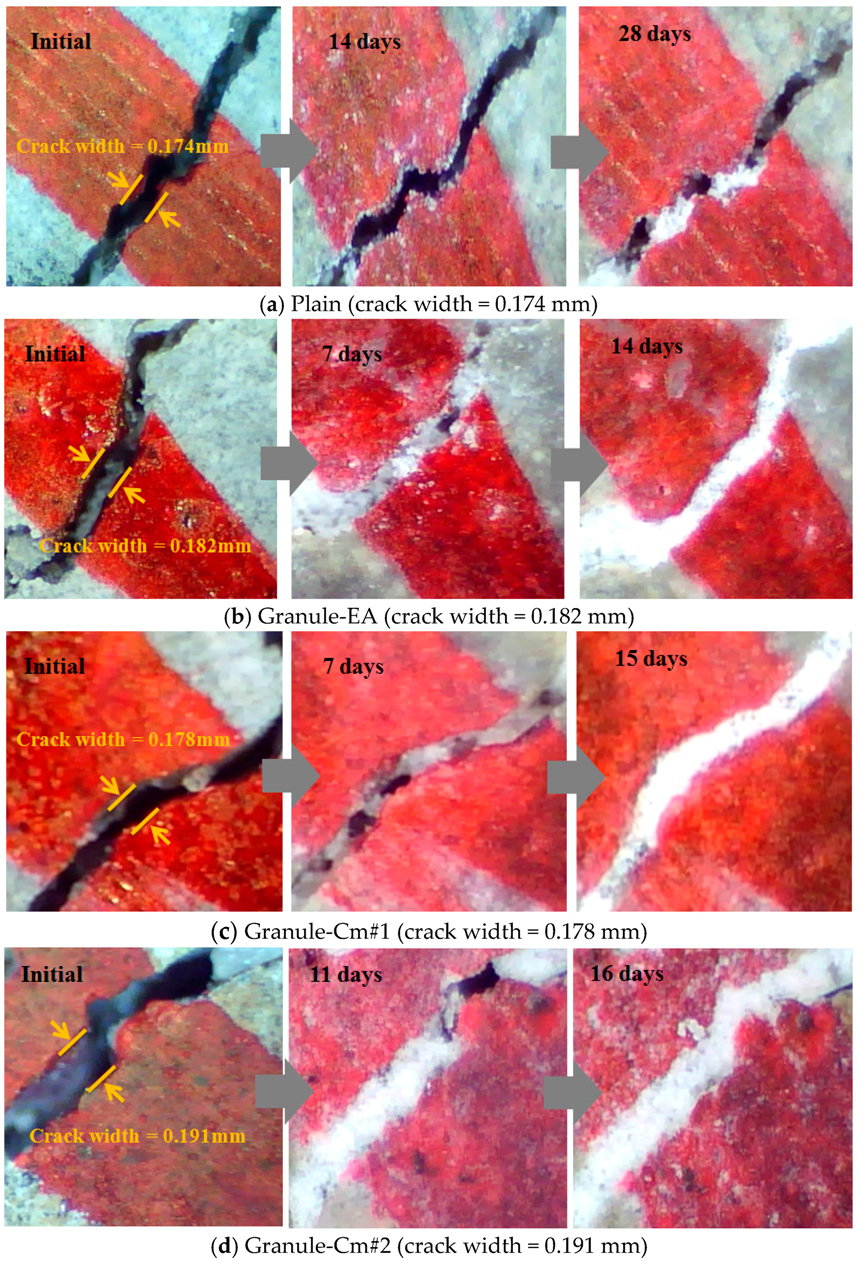
3.1. Microscopic Investigation of the Changes in the Crack Width
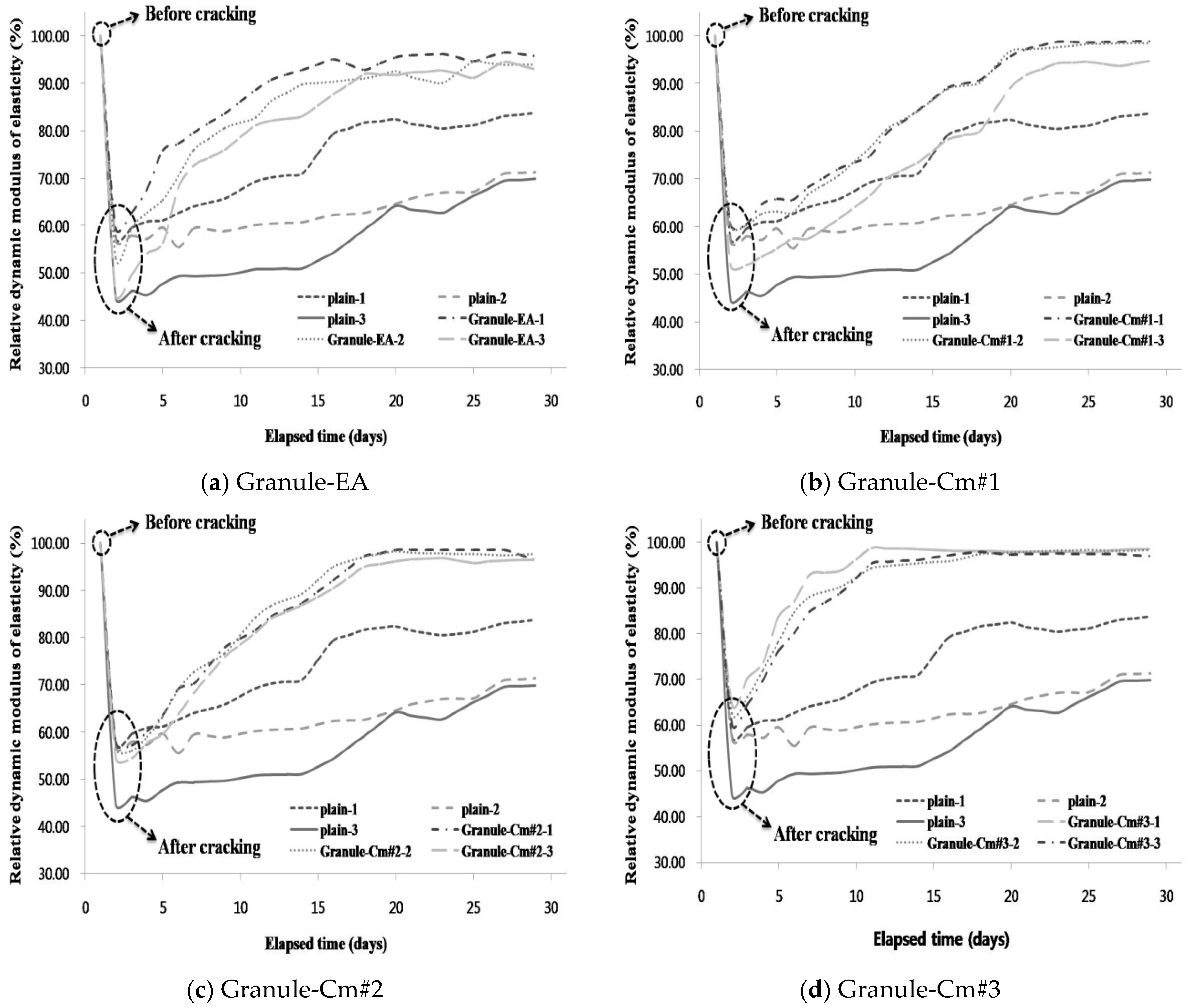
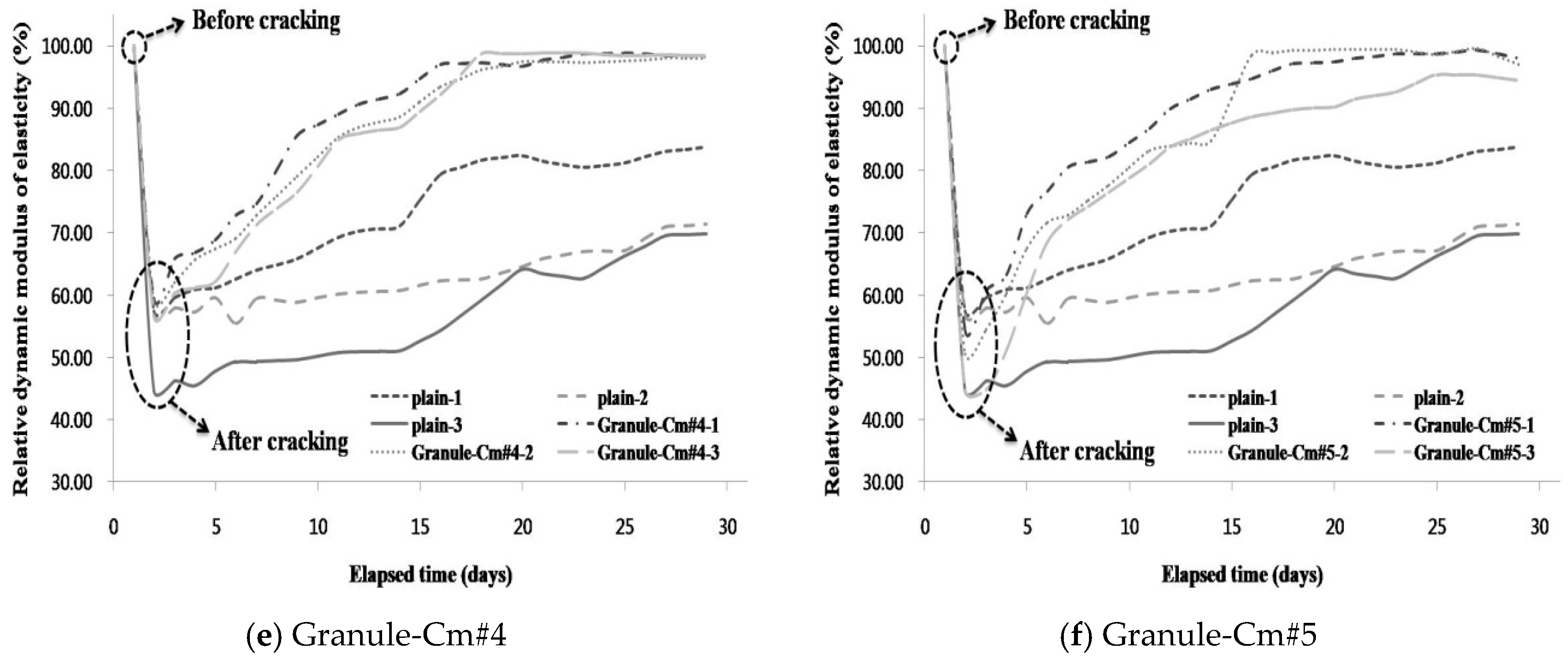
3.2. Evaluation of the Internal Crack Closing via the Dynamic Modulus of Elasticity
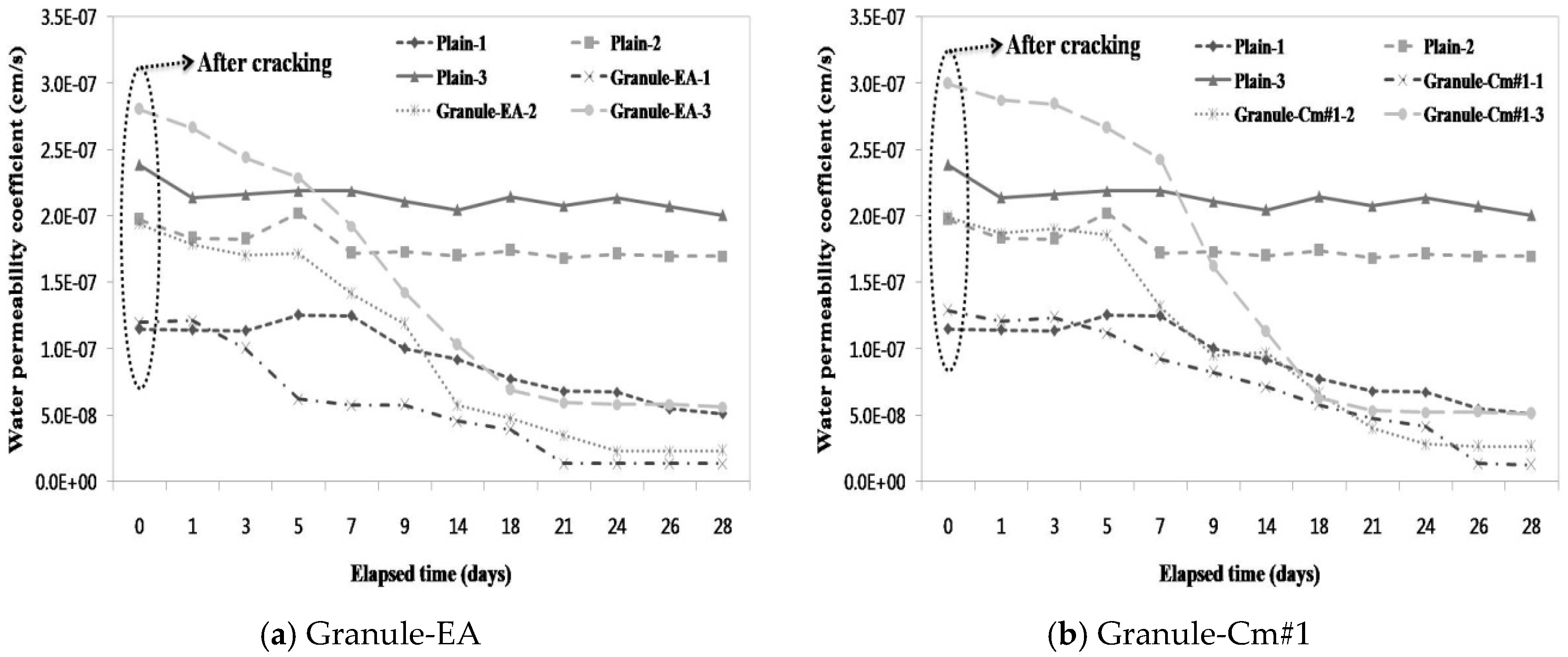
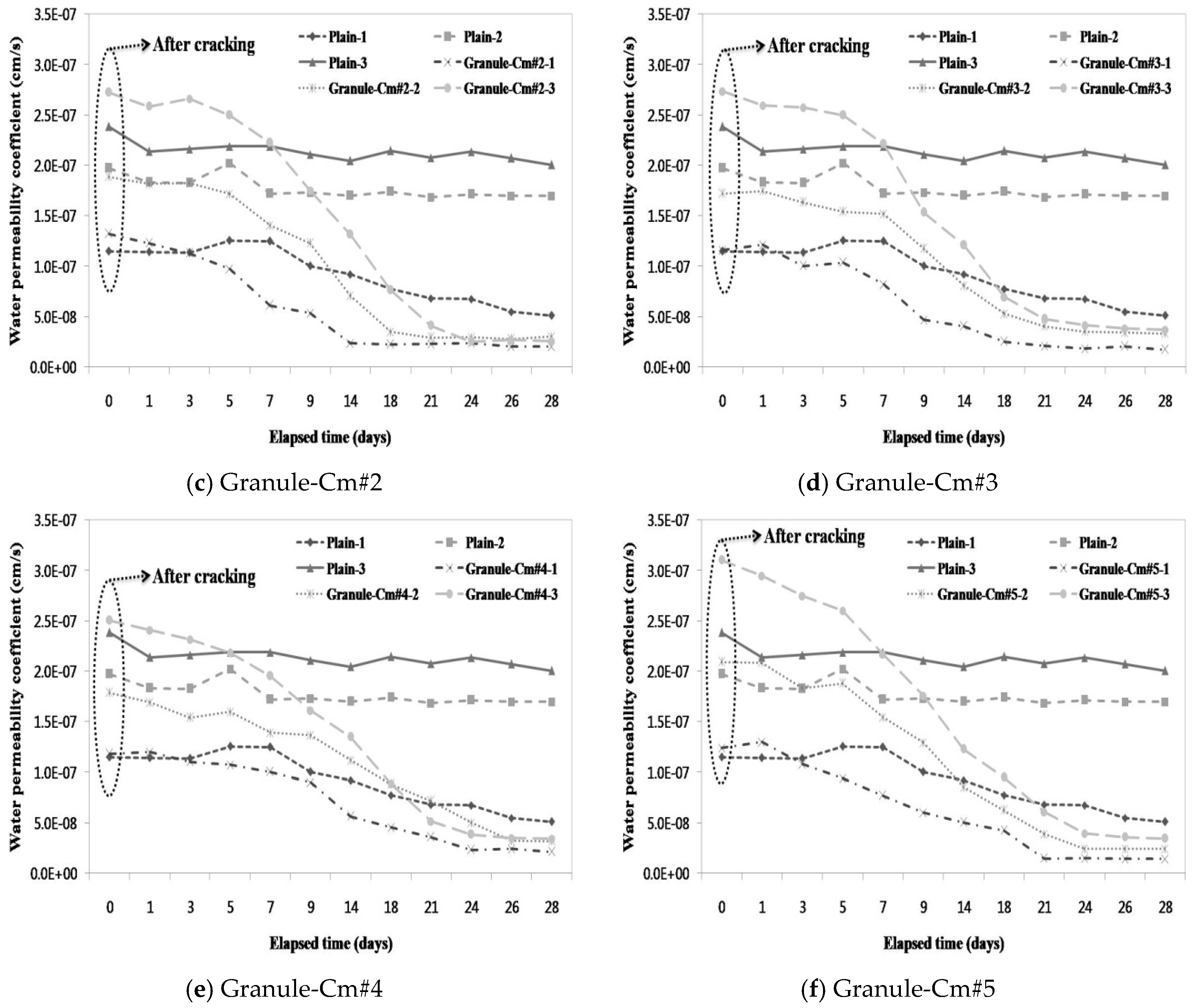
3.3. Evaluation of the Prevention of Water Migration by the Water Permeability Coefficient
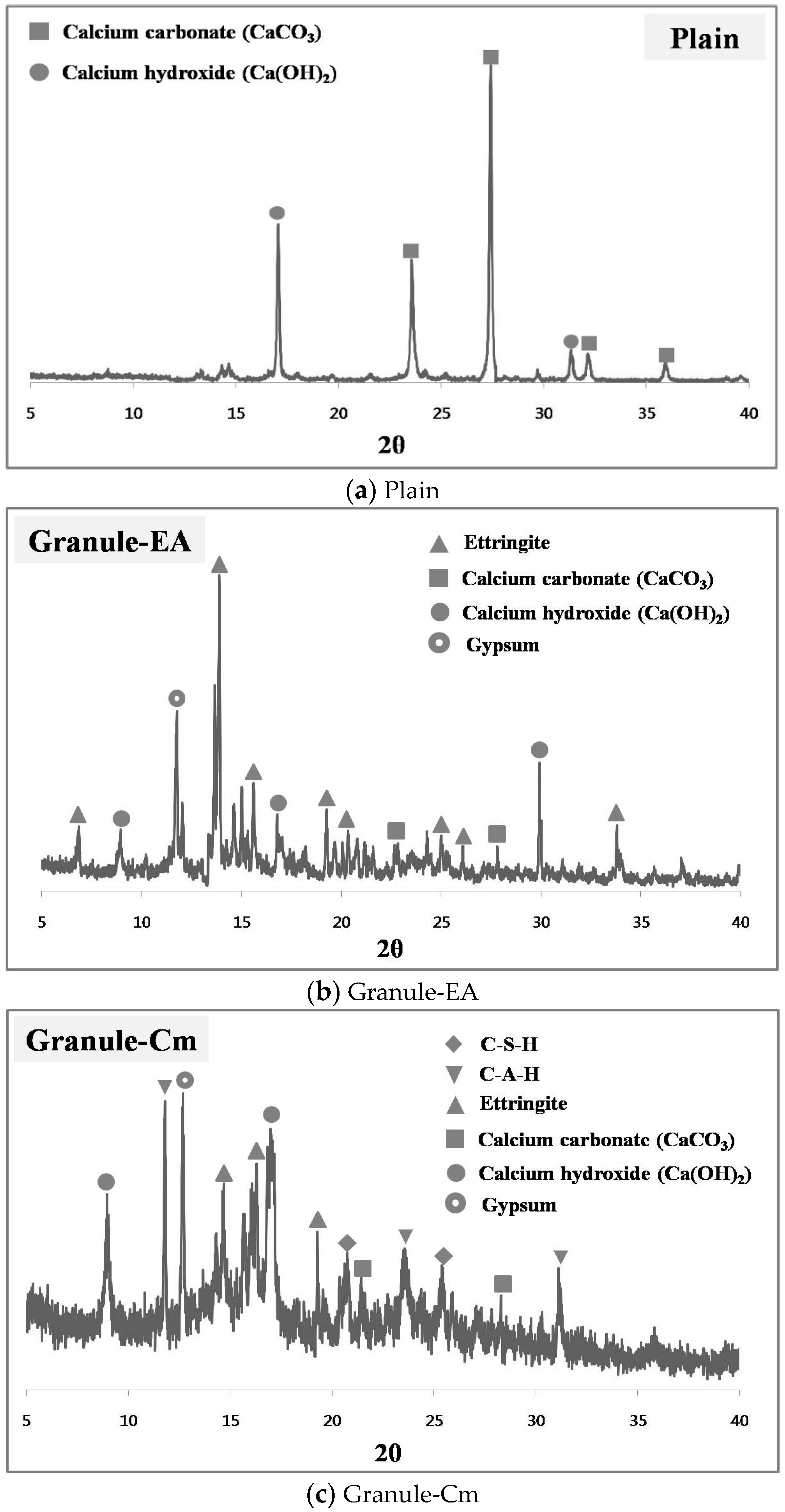
3.4. Analysis of the Healing Products Formed on the Crack Faces
4. Conclusions
- (1)
- For the granulated cementitious material with a PVA film coating (Granule-Cm), the <0.1 mm crack widths indicated complete crack closing within 14 days; the 0.1–0.2 mm crack widths, within 17 days; and the >0.2 mm crack widths, within 21 days.
- (2)
- In the process of crack closing via self-healing with regard to the typical specimens Plain and those containing granules coated with PVA (Granule-EA and Granule-Cm), which involved 0.1–0.2 mm crack widths within the crack width range, Plain showed crack closing in some parts of the crack even after 28-day immersion. The specimen incorporated with Granule-EA, however, was observed to have had crystals in the crack, except for some parts, after seven-day immersion, with complete crack closing after 14-day immersion. In the same way, in the specimens incorporating Granule-Cm (Granule-Cm#1, Granule-Cm#2, Granule-Cm#3, Granule-Cm#4, and Granule-Cm#5), the extent of crack closing was smaller than that in Granule-EA after seven-day immersion while complete crack closing was observed after 14- to 17-day immersion.
- (3)
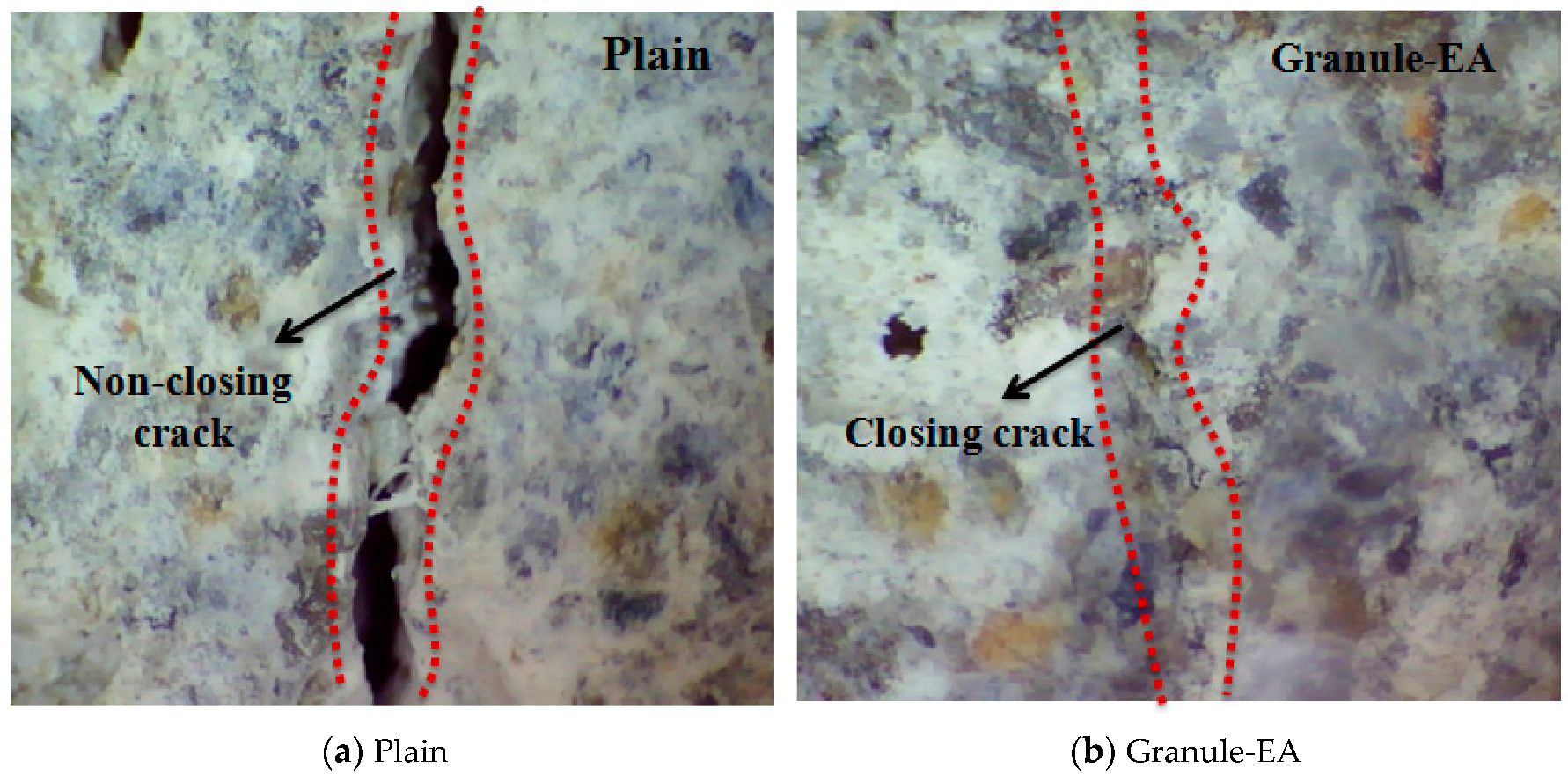
- The internal crack closing was evaluated based on the relative dynamic modulus of elasticity. The dynamic modulus of elasticity of Granule-EA rapidly increased early and then stabilized whereas that of Granule-Cm gradually increased and then stabilized. After the completion of the measurement of the dynamic modulus of elasticity, the occurrence of internal crack closing was examined with a microscope. It was seen that Granule-Cm achieved internal crack closing because marks of the cracks could no longer be observed.
- (4)
- A water passing test was conducted to evaluate the prevention of aggressive agents such as water migration due to crack propagation. After the cracking, a similar water permeability coefficient appeared for each crack width range (<0.1 mm, 0.1–0.2 mm, and >0.2 mm). This verified that, unlike the water permeability coefficient of Granule-EA, that of Granule-Cm gradually decreased up to seven days and then rapidly decreased.
- (5)
- The X-ray diffraction (XRD) patterns of the healing products of Granule-Cm showed the peaks of C–S–H, C–A–H, ettringite, Ca(OH)2, etc., which were formed by cement hydration. They also showed that acicular ettringite was visible in the form of a spider web in the scanning electron microscopy (SEM) micrographs. Ettringite had become entangled with the C–S–H gel, which had formed as a result of the hydration of the cement particles.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Allen, R.T.L.; Edwards, S.C.; Shaw, J.D.N. Repair of Concrete Structures; CRC Press: Boca Raton, FL, USA, 1992; pp. 13–23. [Google Scholar]
- Jacobsen, S.; Marchand, J.; Boisvert, L. Effect of cracking and healing on chloride transport in OPC concrete. Cem. Concr. Res. 1996, 26, 869–881. [Google Scholar] [CrossRef]
- Van Breugel, K. Is there a market for self-healing cement-based materials? In Proceedings of the First International Conference on Self-Healing Materials, Noordwijkaan Zee, The Netherlands, 18–20 April 2007.
- Wu, M.; Johannesson, B.R.; Geiker, M. A review: Self-healing in cementitious materials and engineered cementitious composite as a self-healing material. Constr. Build. Mater. 2012, 28, 571–583. [Google Scholar] [CrossRef]
- Thao, T.D.P.; Johnson, T.J.S.; Tong, Q.S.; Dai, P.S. Implementation of self-healing in concrete—Proof of concept. IES J. Part A 2009, 2, 116–125. [Google Scholar] [CrossRef]
- Neville, A. Autogenous healing—A concrete miracle. Concr. Int. 2002, 24, 76–82. [Google Scholar]
- Igarashi, S.; Kunieda, M.; Nishiwaki, T. Research activity of JCI technical committee TC-075B: Autogenous healing in cementitious materials. In Proceedings of the 4th International Conference on Construction Materials: Performance, Innovations and Structural Implications, Con Mat, Nagoya, Japan, 24–26 August 2009.
- Huang, H.; Ye, G.; Qian, C.; Schlangen, E. Self-healing in cementitious materials: Materials, methods and service conditions. Mater. Des. 2016, 92, 499–511. [Google Scholar] [CrossRef]
- Bekas, D.G.; Tsirka, K.; Baltzis, D.; Paipetis, A.S. Self-healing materials: A review of advances in materials, evaluation, characterization and monitoring techniques. Compos. Part B 2016, 87, 92–119. [Google Scholar] [CrossRef]
- Van Tittelboom, K.; de Belie, N. Self-healing in cementitious materials—A review. Materials 2013, 6, 2182–2217. [Google Scholar] [CrossRef]
- Li, V.C.; Herbert, E. Robust self-healing concrete for sustainable infrastructure. J. Adv. Concr. Technol. 2012, 10, 207–218. [Google Scholar] [CrossRef]
- Qian, S.; Zhou, J.; de Rooij, M.R.; Schlangen, E.; Ye, G.; van Breugel, K. Self-healing behavior of strain hardening cementitious composites incorporating local waste materials. Cem. Concr. Compos. 2009, 31, 613–621. [Google Scholar] [CrossRef]
- Sisomphon, K.; Copuroglu, O.; Koenders, E.A.B. Self-healing of surface cracks in mortars with expansive additive and crystalline additive. Cem. Concr. Compos. 2012, 34, 566–574. [Google Scholar] [CrossRef]
- Ahn, T.H.; Kishi, T. The effect of geomaterials on the autogenous healing behavior of cracked concrete. In Proceedings of the 2nd ICCRRR, Cape Town, South Africa, 24–26 November 2008; pp. 235–240.
- Nagataki, S.; Gomi, H. Expansive admixtures (mainly ettringite). Cem. Concr. Compos. 1998, 20, 163–170. [Google Scholar] [CrossRef]
- Ahn, T.; Kishi, T. Crack self-healing behavior of cementitious composites incorporating various mineral admixtures. J. Adv. Concr. Technol. 2010, 8, 171–186. [Google Scholar] [CrossRef]
- Sahamitmongkol, R.; Kishi, T. Tensile behavior of restrained expansive mortar and concrete. Cem. Concr. Compos. 2011, 33, 131–141. [Google Scholar] [CrossRef]
- Tousey, M.D. The Granulation Process 101 Basic Technologies for Tablet Making. Pharm. Technol. 2002, I, 8–13. [Google Scholar]
- Shangraw, R.F.; Demarest, D.A. A survey of current industrial practices in the formulation of tablets and capsules. Pharm. Tech. 1993, 17, 32–44. [Google Scholar]
- Allen, L.V.; Popovich, N.G. Ansel’s Pharmaceutical Dosage Forms and Drug Delivery Systems; Lippincott Williams & Wilkins: Baltimore, MD, USA; Philadelphia, PA, USA, 2005; pp. 83–125. [Google Scholar]
- Kamble, N.D.; Chaudhari, P.S.; Oswal, R.J.; Kshirsagar, S.S.; Antre, R.V.; Wagholi, P. Innovations in tablet coating technology: A review. Int. J. Appl. Biol. Pharm. Technol. 2011, 2, 214–218. [Google Scholar]
- Lee, Y.S.; Ryou, J.S. Self-healing behavior for crack closing of expansive agent via granulation/film coating method. Constr. Build. Mater. 2014, 71, 188–193. [Google Scholar] [CrossRef]
- Ryou, J.S.; Lee, Y.S. Use of tableting & coating accelerator for the prevention of early-frost of concrete in cold weather. Cold Reg. Sci. Technol. 2013, 87, 1–5. [Google Scholar]
- KS L ISO 679 Methods of Testing Cements-Determination of Strength; Korean Standards Association (KSA): Seoul, Korea, 2006.
- Arai, Y. Chemistry of Cement Materials; Dai-Nippon Tosho: Tokyo, Japan, 1993; pp. 156–252. [Google Scholar]
- Neville, A.M. Properties of Concrete; PEARSON: London, UK, 2011; pp. 320–353. [Google Scholar]
- Malhotra, V.M.; Carino, N.J. Handbook on Nondestructive Testing of Concrete, 2nd ed.; ASTM International: Boca Raton, FL, USA, 2003; Chapter 8. [Google Scholar]
- ASTM Designation C215-14, Standard Test Method for Fundamental Transverse, Longitudinal and Torsional Resonant Frequencies of Concrete Specimens; ASTM International: Boca Raton, FL, USA, 2014.
- Hearn, N. Self-sealing, autogenous healing and continued hydration: What is the difference? Mater. Struct. 1998, 31, 563–567. [Google Scholar] [CrossRef]
- Test Method No. II.4. Water Absorption Tube Test; RILEM: Paris, France, 2006.
- Cernica, J.N. Geotechnical Engineering: Soil Mechanics; Holt, Reinhart & Winston: New York, NY, USA, 1982; pp. 97–99. [Google Scholar]
- Walker, H.N.; Lane, D.S.; Stutzman, P.E. Petrographic Methods of Examining Hardened Concrete: A Petrographic Manual. Revised 2004; No. FHWA-HRT-04-150; National Technical Information Service: Alexandria, VA, USA, 2006. [Google Scholar]
- Van der Zwaag, S. An Introduction to Material Design Principles: Damage Prevention versus Damage Management; Self Healing Materials; Springer: Delft, The Netherlands, 2007; pp. 1–18. [Google Scholar]
- Edvardsen, C. Water permeability and autogenous healing of cracks in concrete. ACI Mater. J. 1999, 96, 448–454. [Google Scholar]
- Kumar, M.P. Concrete. Structure, Properties and Materials; Prentice-Hall, Incorporated: Englewood Cliffs, NJ, USA, 1986. [Google Scholar]
- Lee, Y.S.; Lim, D.S.; Chun, B.S.; Ryou, J.S. Characterization of a sodium aluminate (NaAlO2)-based accelerator made via a tablet processing method. J. Ceram. Process. Res. 2013, 14, 87–91. [Google Scholar]






| Type | Advantage | Disadvantage |
|---|---|---|
| Microcapsule |
|
|
| Expansive agent and mineral admixture |
|
|
| S. No. | Specimens | Material Proportions |
|---|---|---|
| 1 | Plain | CSA (0%), OPC (0%) |
| 2 | Granule-EA | CSA (10%), OPC (90%) |
| 3 | Granule-Cm#1 | CSA (10%), OPC (90%), and Na2CO3 (1%) |
| 4 | Granule-Cm#2 | CSA (20%), OPC (80%), and Na2CO3 (1%) |
| 5 | Granule-Cm#3 | CSA (30%), OPC (70%), and Na2CO3 (1%) |
| 6 | Granule-Cm#4 | CSA (40%), OPC (60%), and Na2CO3 (1%) |
| 7 | Granule-Cm#5 | CSA (50%), OPC (50%), and Na2CO3 (1%) |
| Type | Healing Products |
|---|---|
| Plain | Calcium carbonate (CaCO3), calcium hydroxide [Ca(OH)2] |
| Granule-EA | Ettringite, calcium carbonate (CaCO3), calcium hydroxide [Ca(OH)2] |
| Granule-Cm | C–S–H, C–A–H, ettingite, calcium carbonate (CaCO3), calcium hydroxide [Ca(OH)2] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-S.; Ryou, J.-S. Crack Healing Performance of PVA-Coated Granules Made of Cement, CSA, and Na2CO3 in the Cement Matrix. Materials 2016, 9, 555. https://doi.org/10.3390/ma9070555
Lee Y-S, Ryou J-S. Crack Healing Performance of PVA-Coated Granules Made of Cement, CSA, and Na2CO3 in the Cement Matrix. Materials. 2016; 9(7):555. https://doi.org/10.3390/ma9070555
Chicago/Turabian StyleLee, Yong-Soo, and Jae-Suk Ryou. 2016. "Crack Healing Performance of PVA-Coated Granules Made of Cement, CSA, and Na2CO3 in the Cement Matrix" Materials 9, no. 7: 555. https://doi.org/10.3390/ma9070555
APA StyleLee, Y.-S., & Ryou, J.-S. (2016). Crack Healing Performance of PVA-Coated Granules Made of Cement, CSA, and Na2CO3 in the Cement Matrix. Materials, 9(7), 555. https://doi.org/10.3390/ma9070555