Recent Progress in Fabrication and Applications of Superhydrophobic Coating on Cellulose-Based Substrates
Abstract
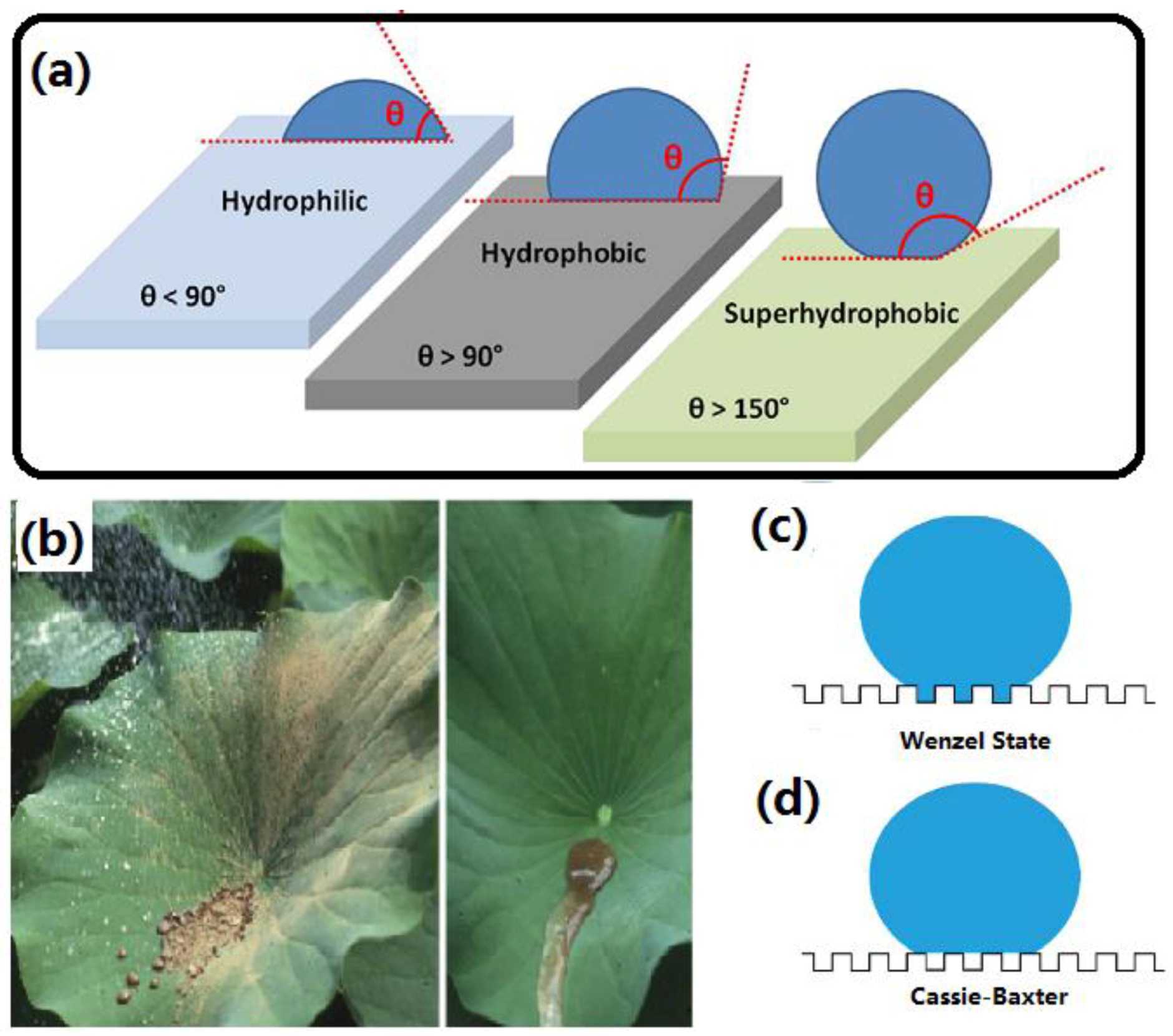
:1. Introduction
2. Construction of Superhydrophobic Coating on Cellulose-Based Substrates
2.1. Wet Chemical Methods
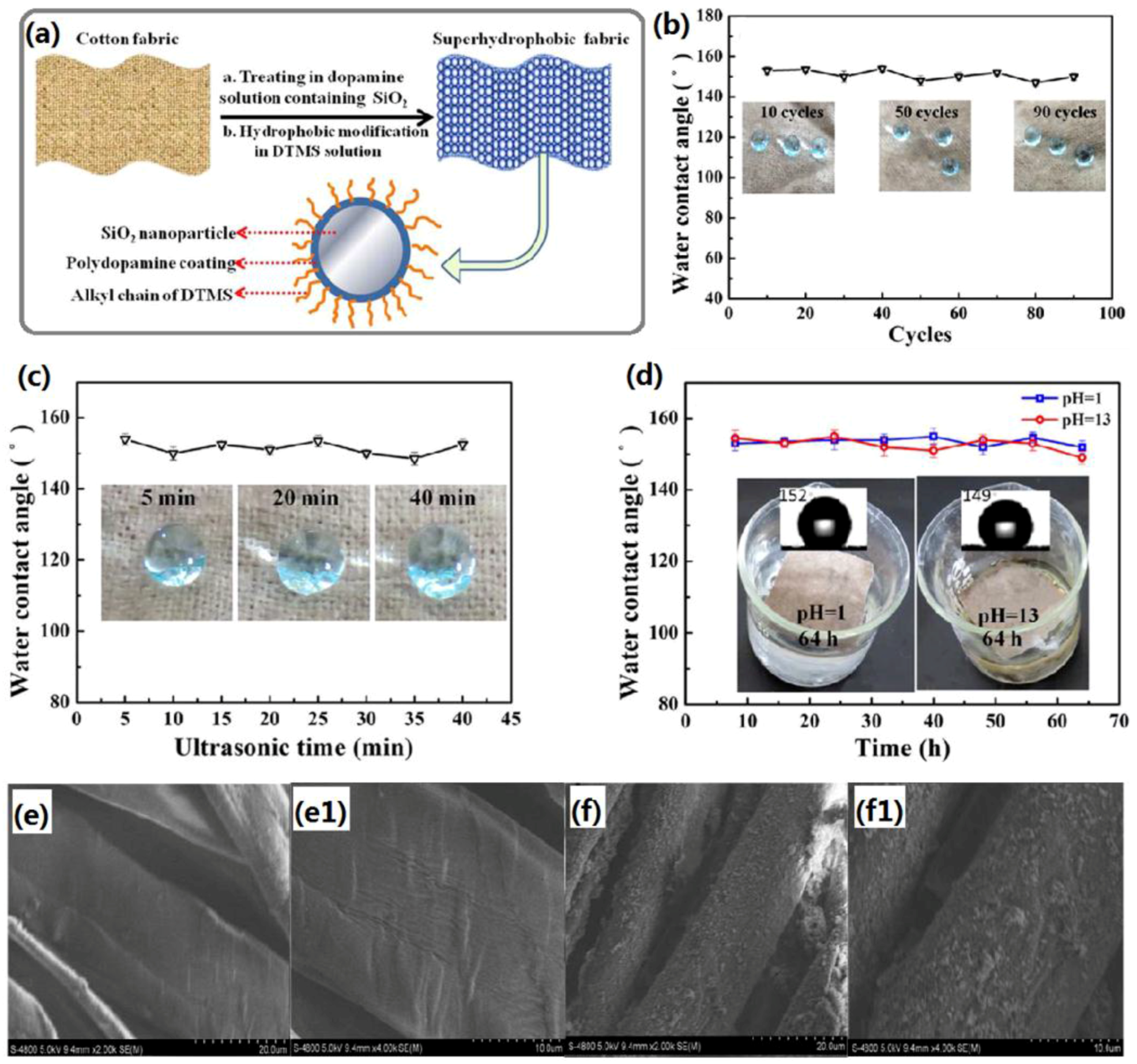
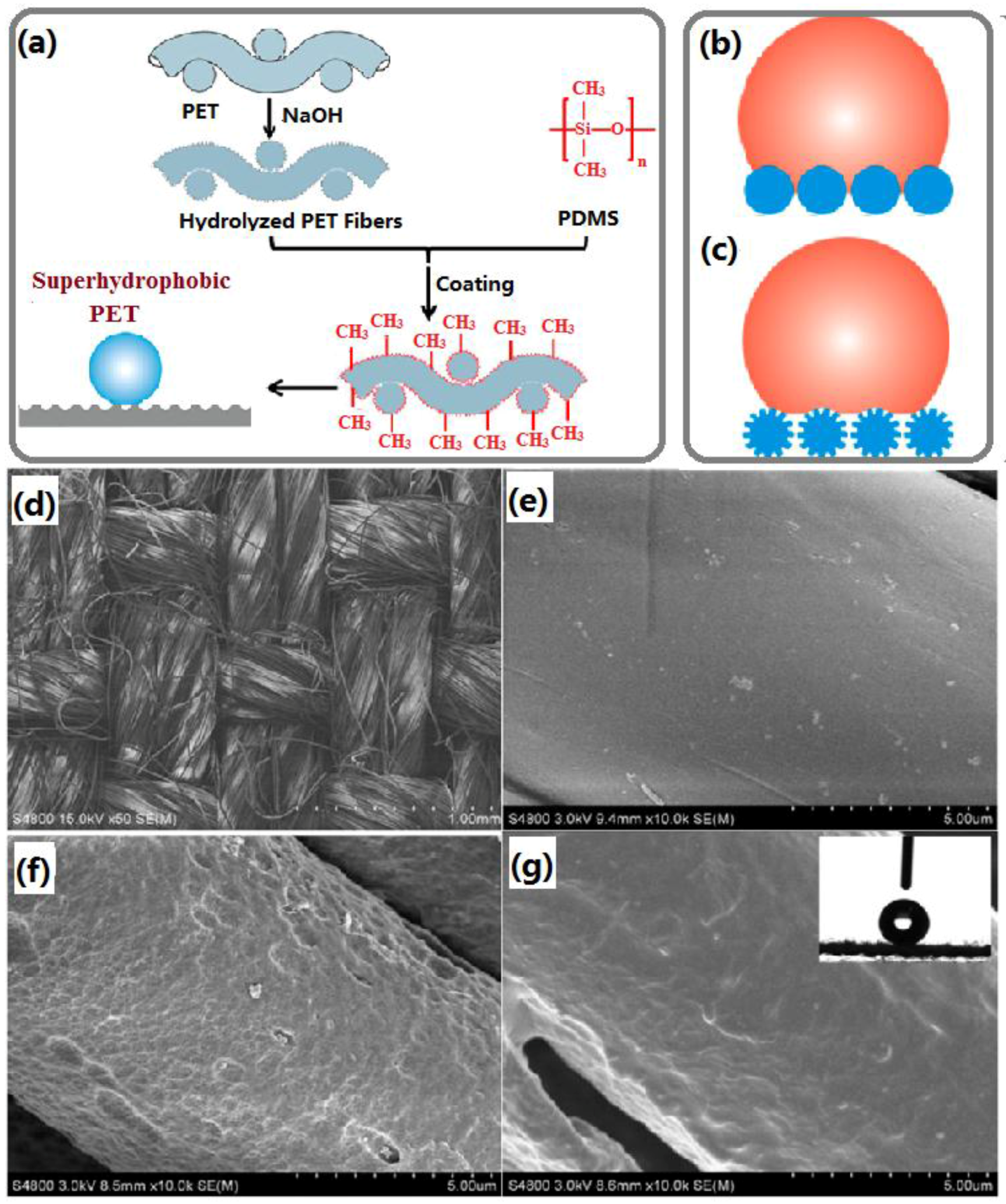
2.1.1. Dip-Coating Methods
2.1.2. Wet Chemical Etching
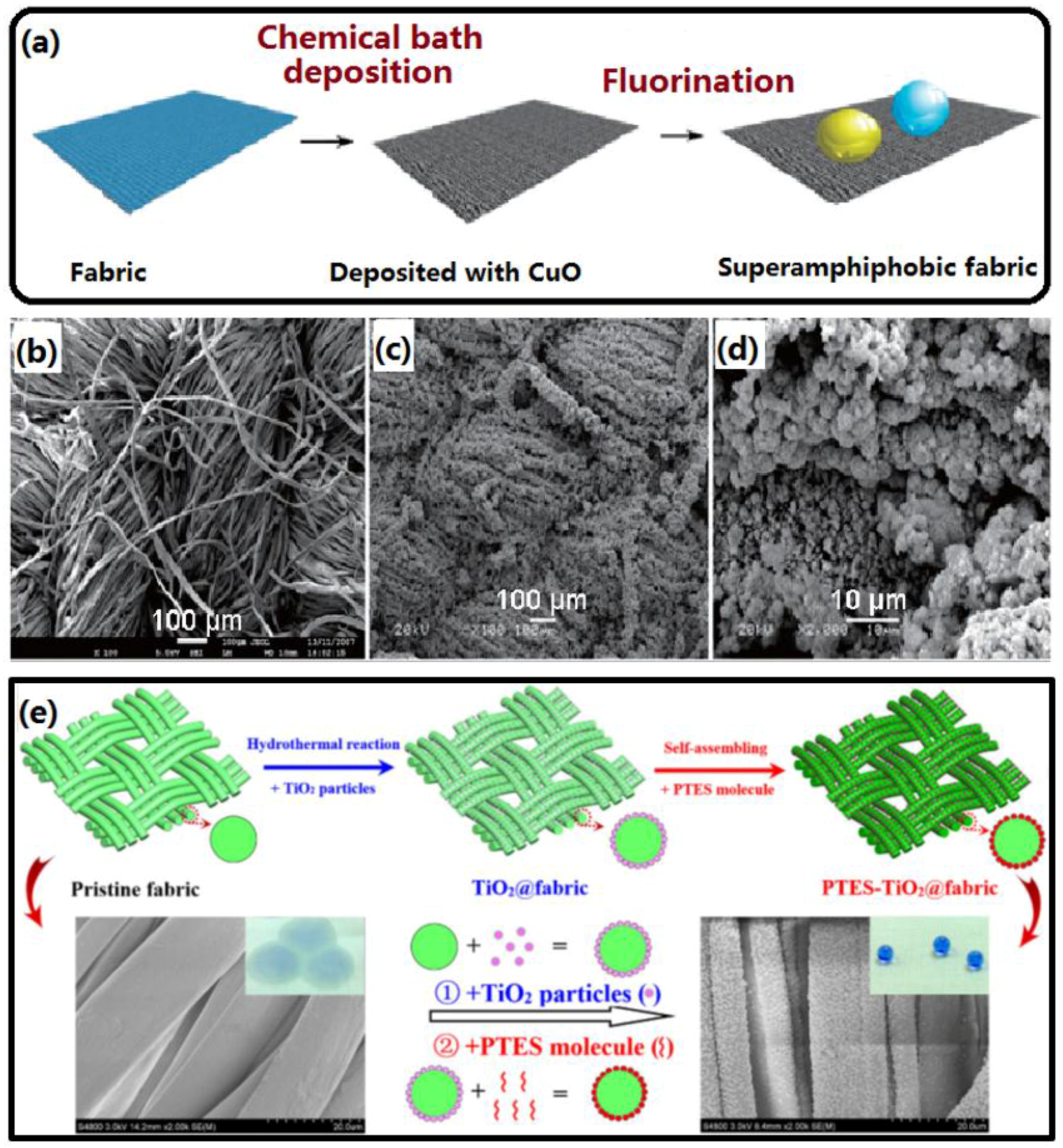
2.1.3. Chemical Bath Deposition
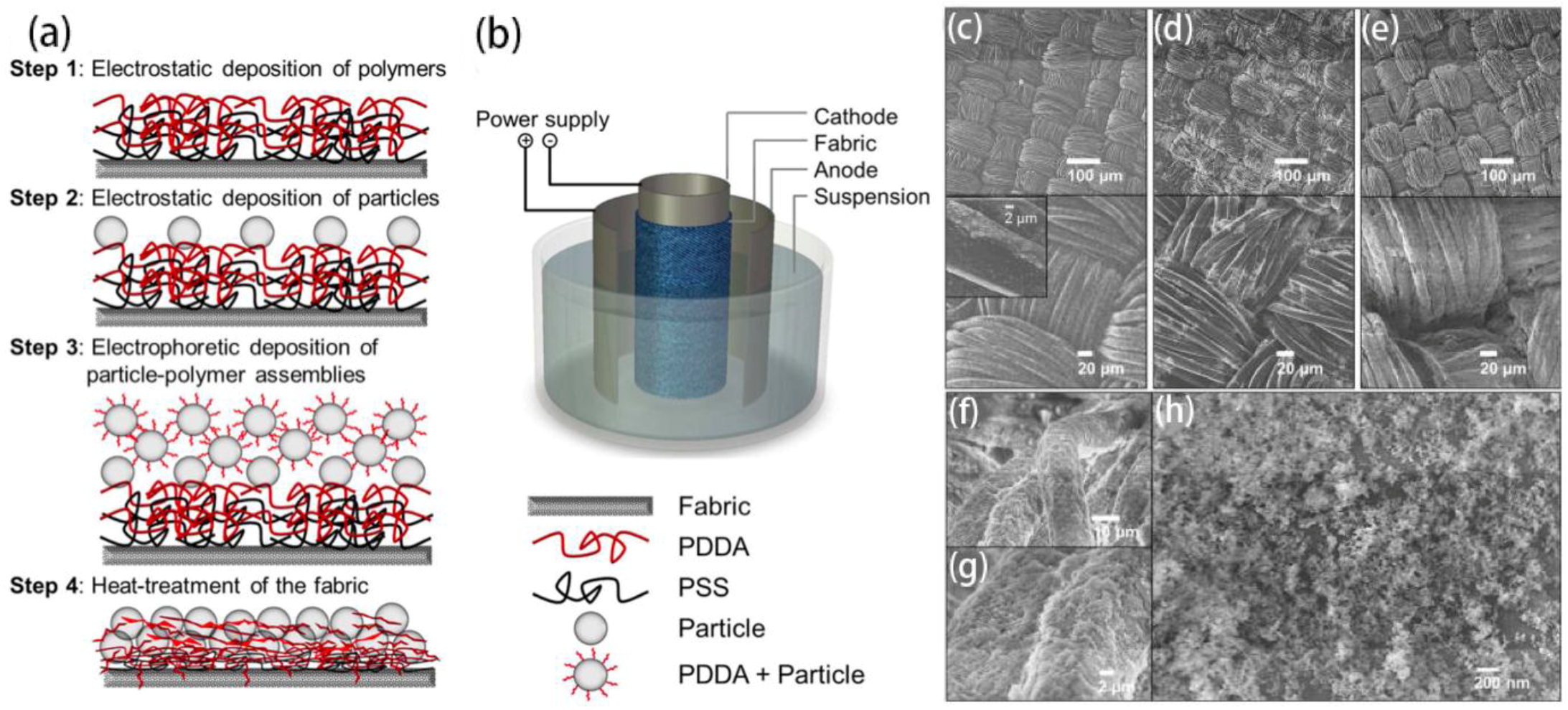
2.1.4. Electric-Field Assisted Etching/Deposition
2.1.5. Spray-Coating
2.1.6. Other Wet Methods
2.2. Dry Methods
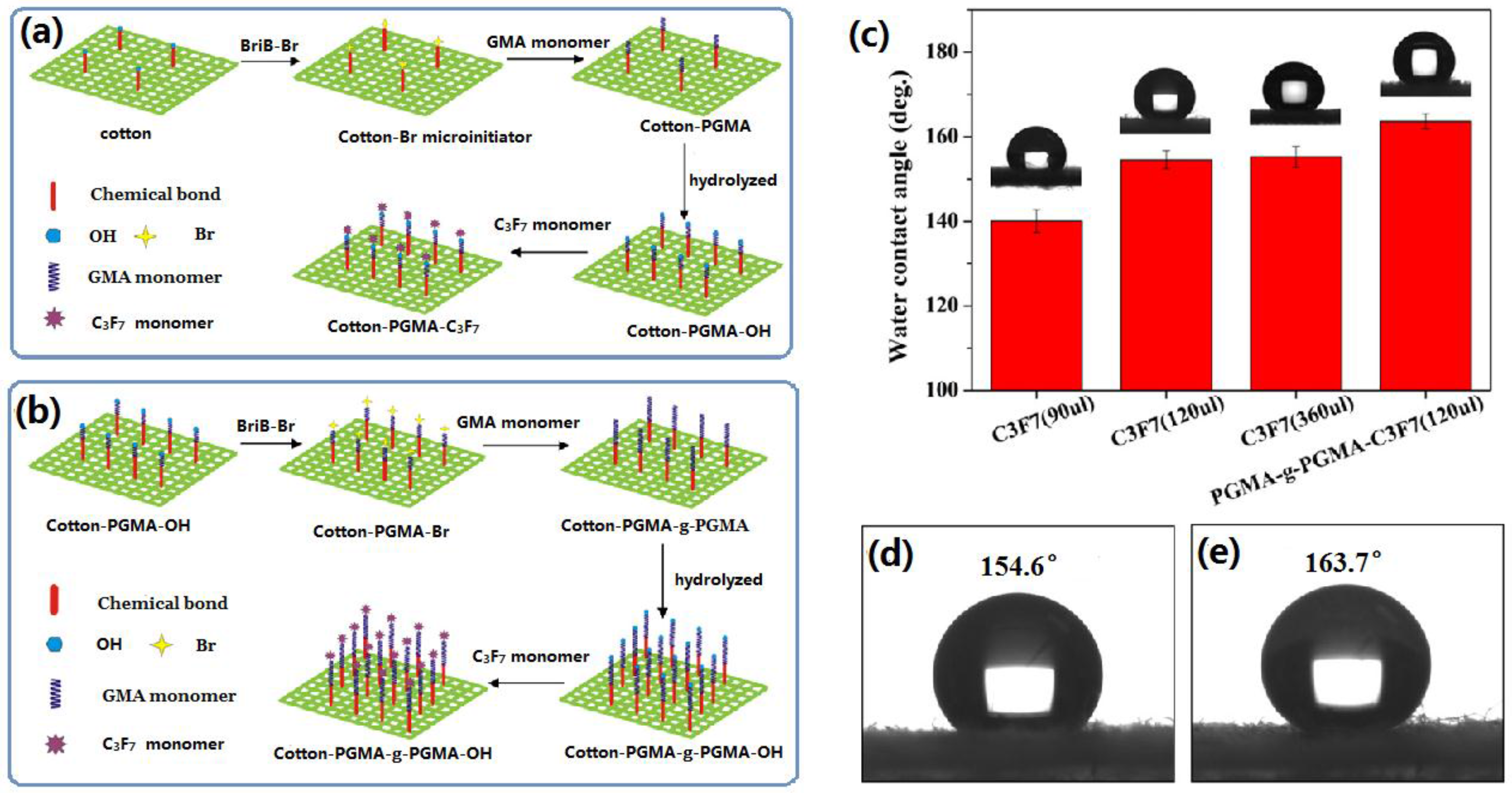
2.2.1. Chemical Vapour Deposition
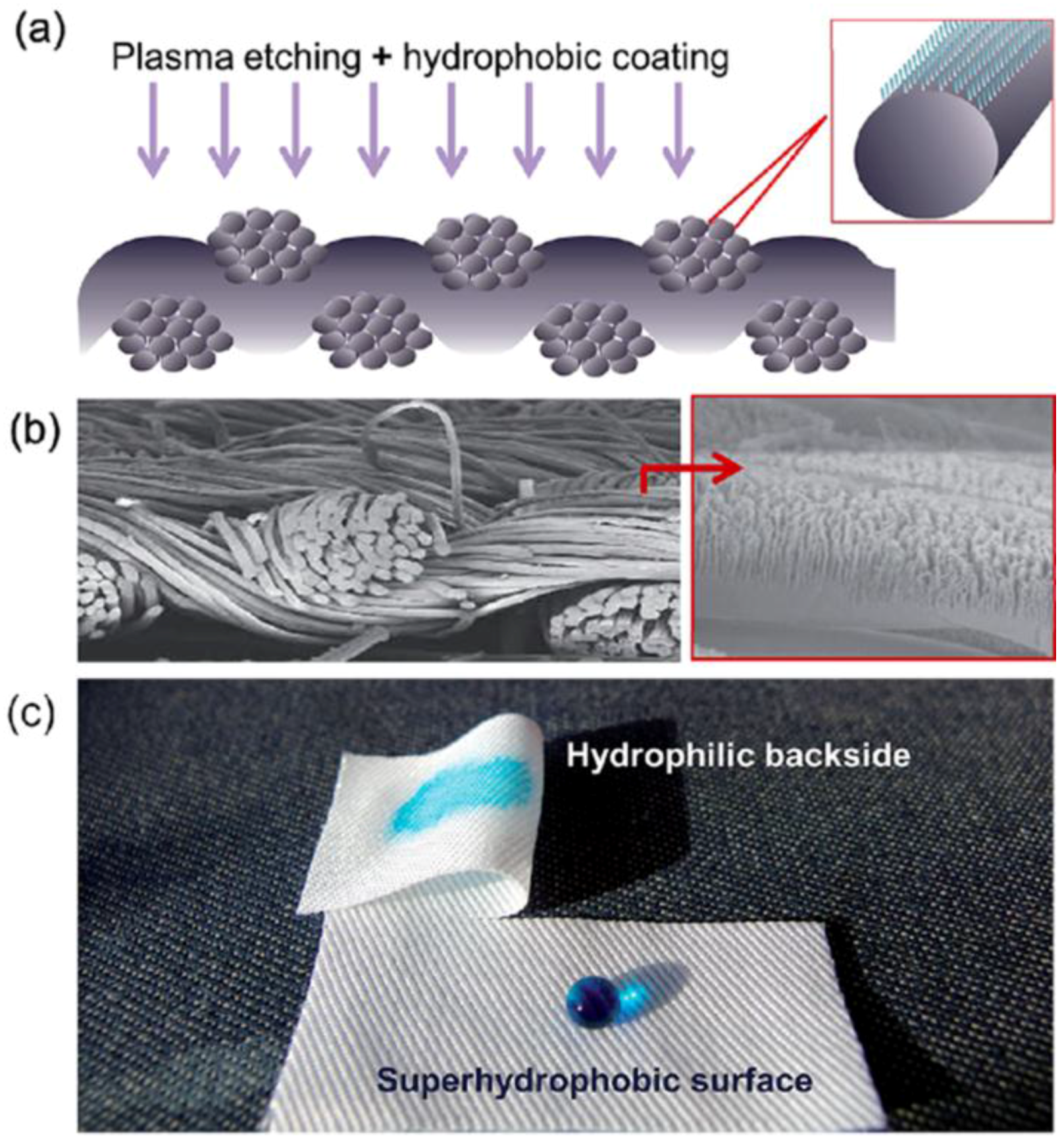
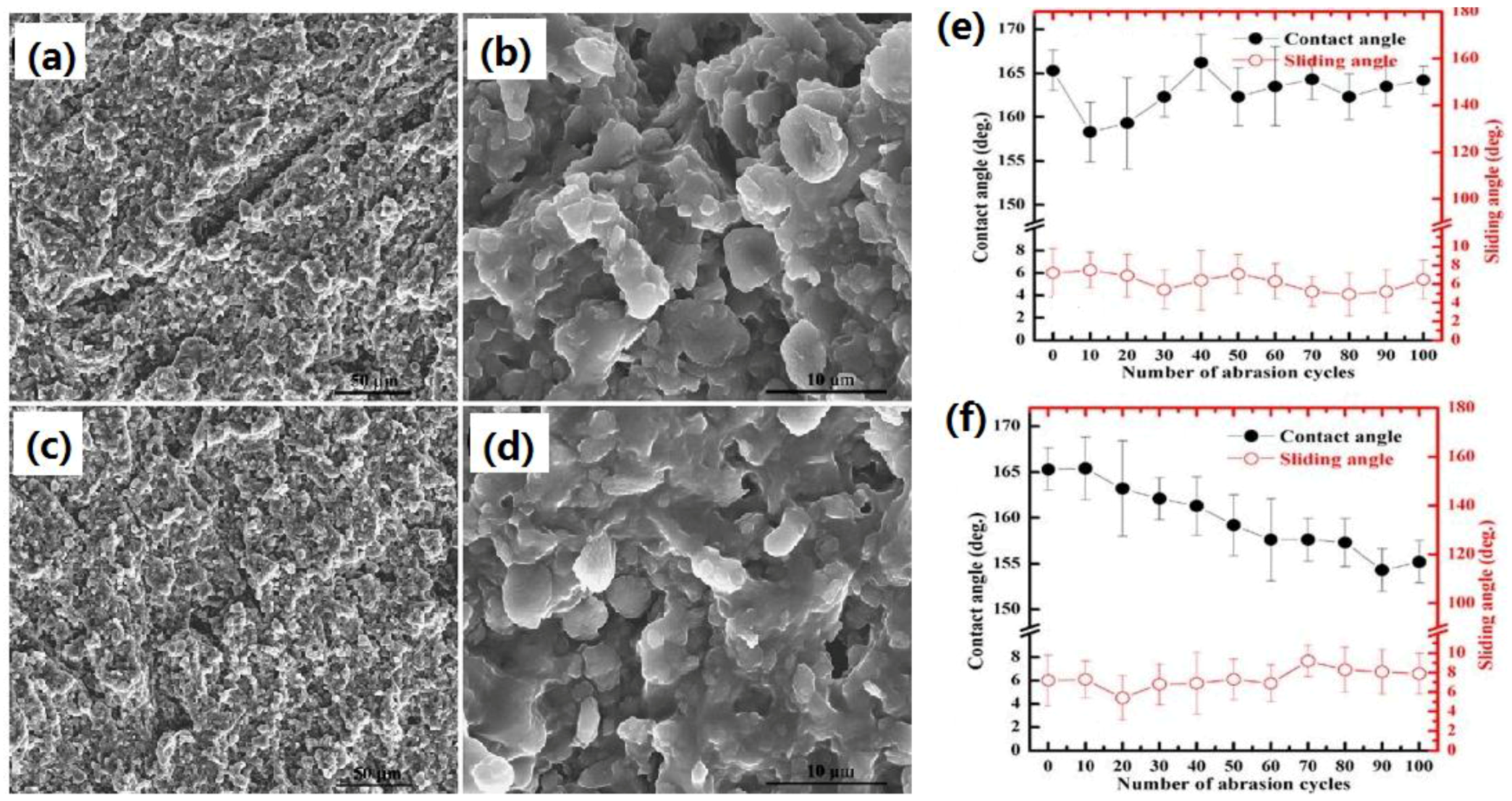
2.2.2. Plasma Etching Processing
2.2.3. Other Dry Methods
3. Promising Applications of Superhydrophobic Fabrics
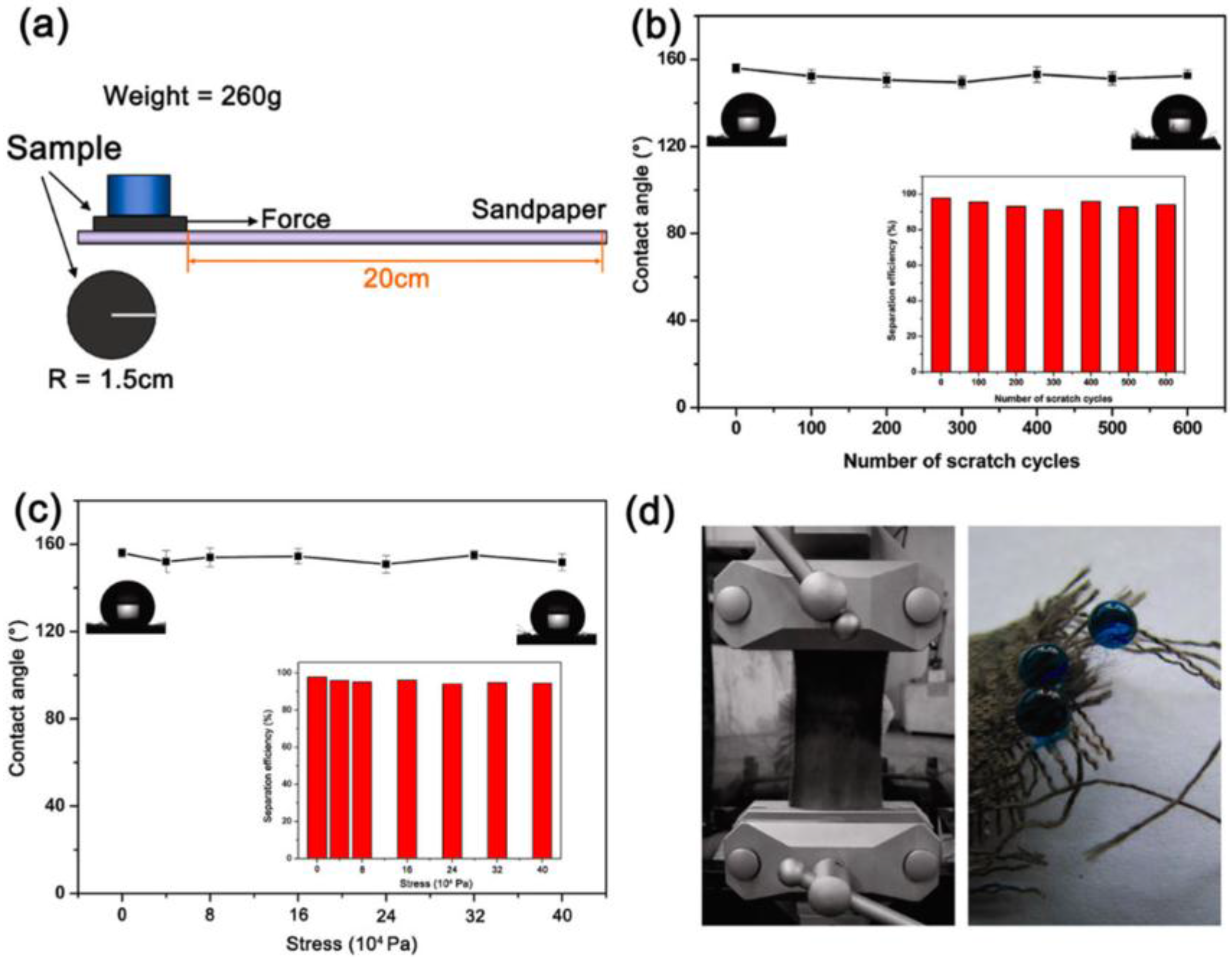
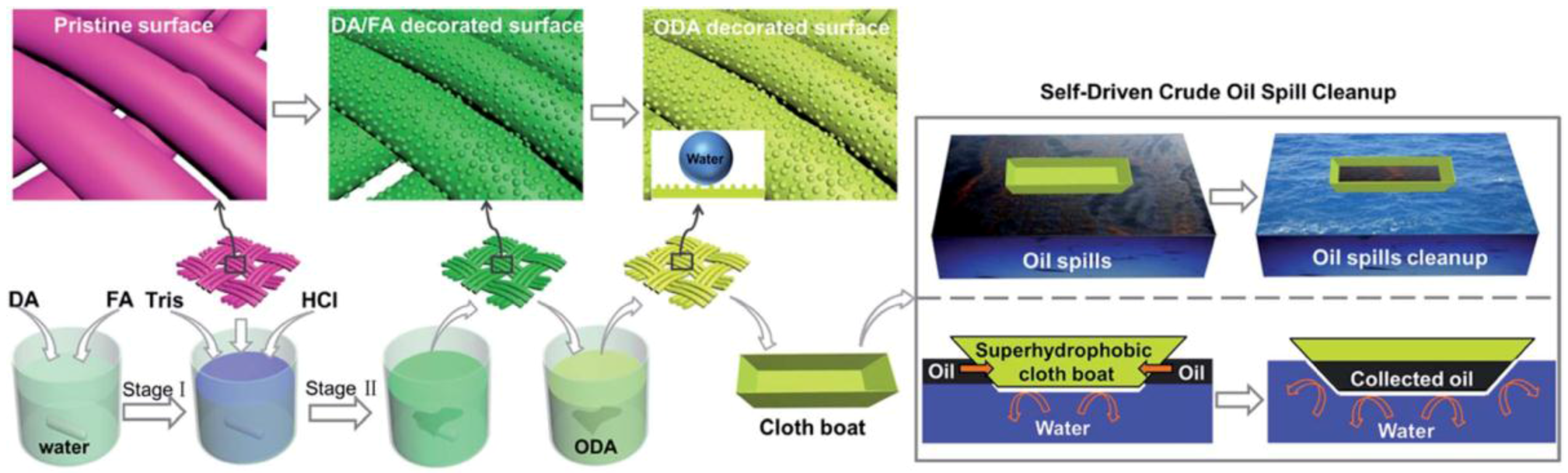
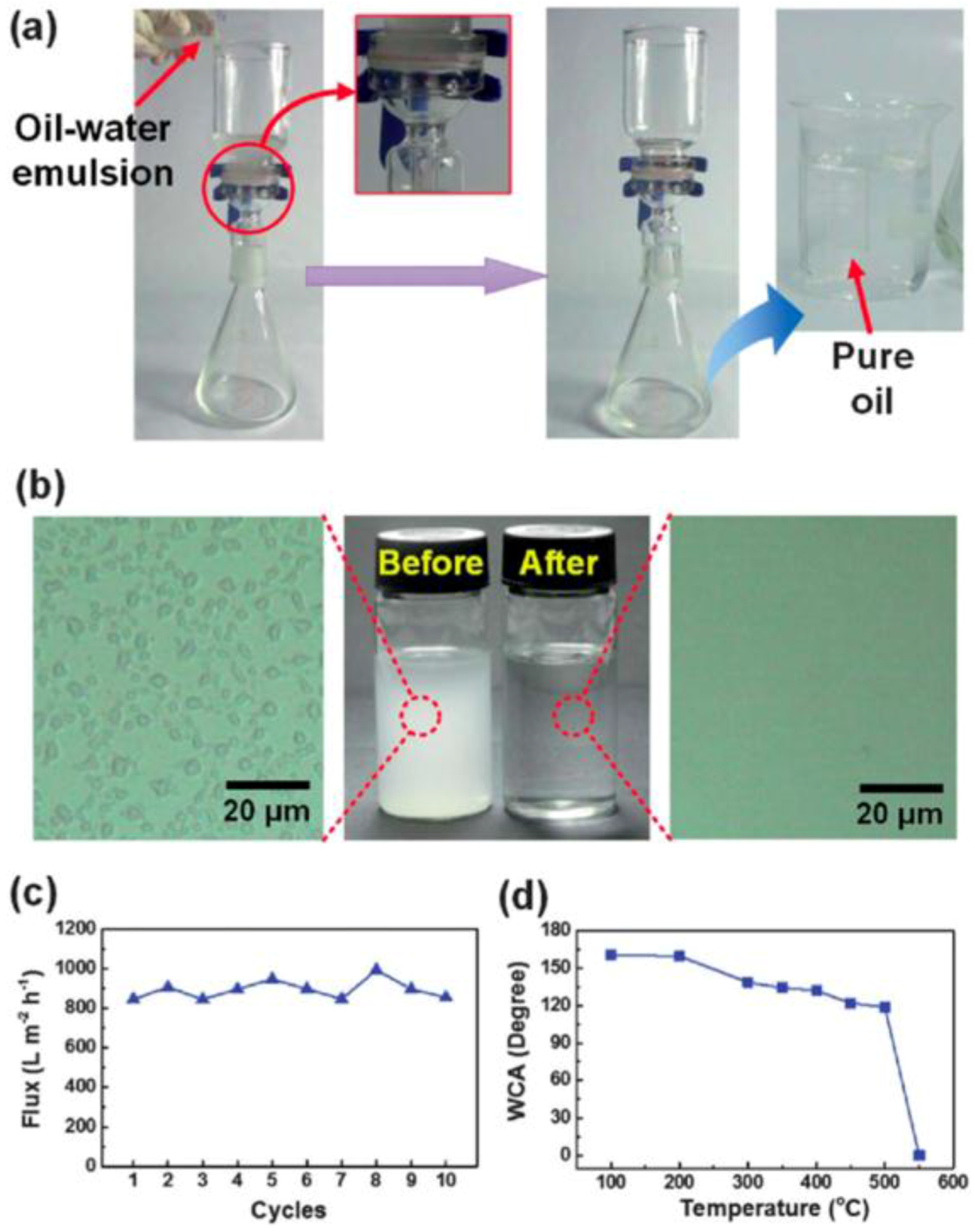
3.1. Oil-Water Separation
3.2. Self-Cleaning
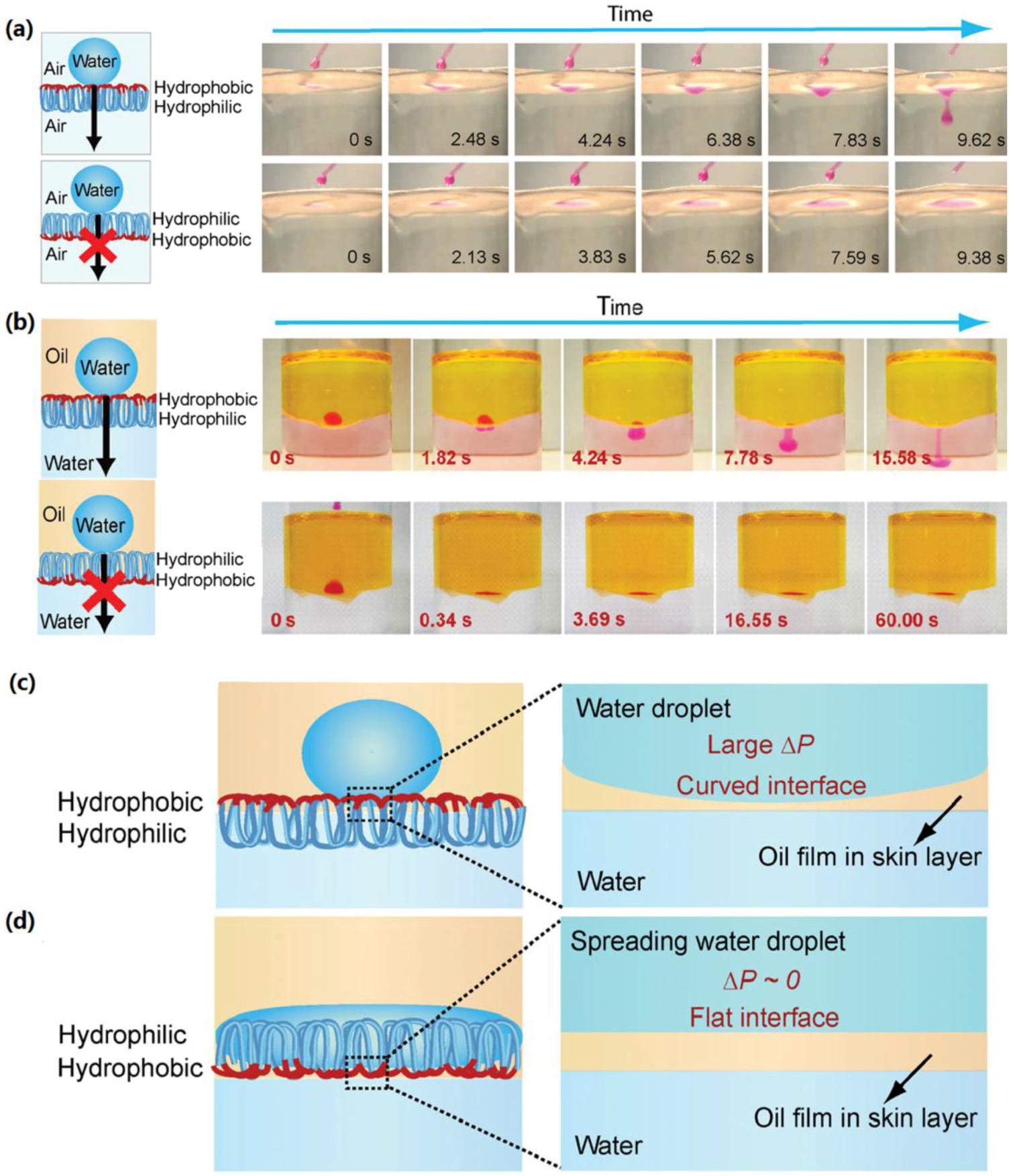
3.3. Asymmetric Superhydrophobic Fabrics
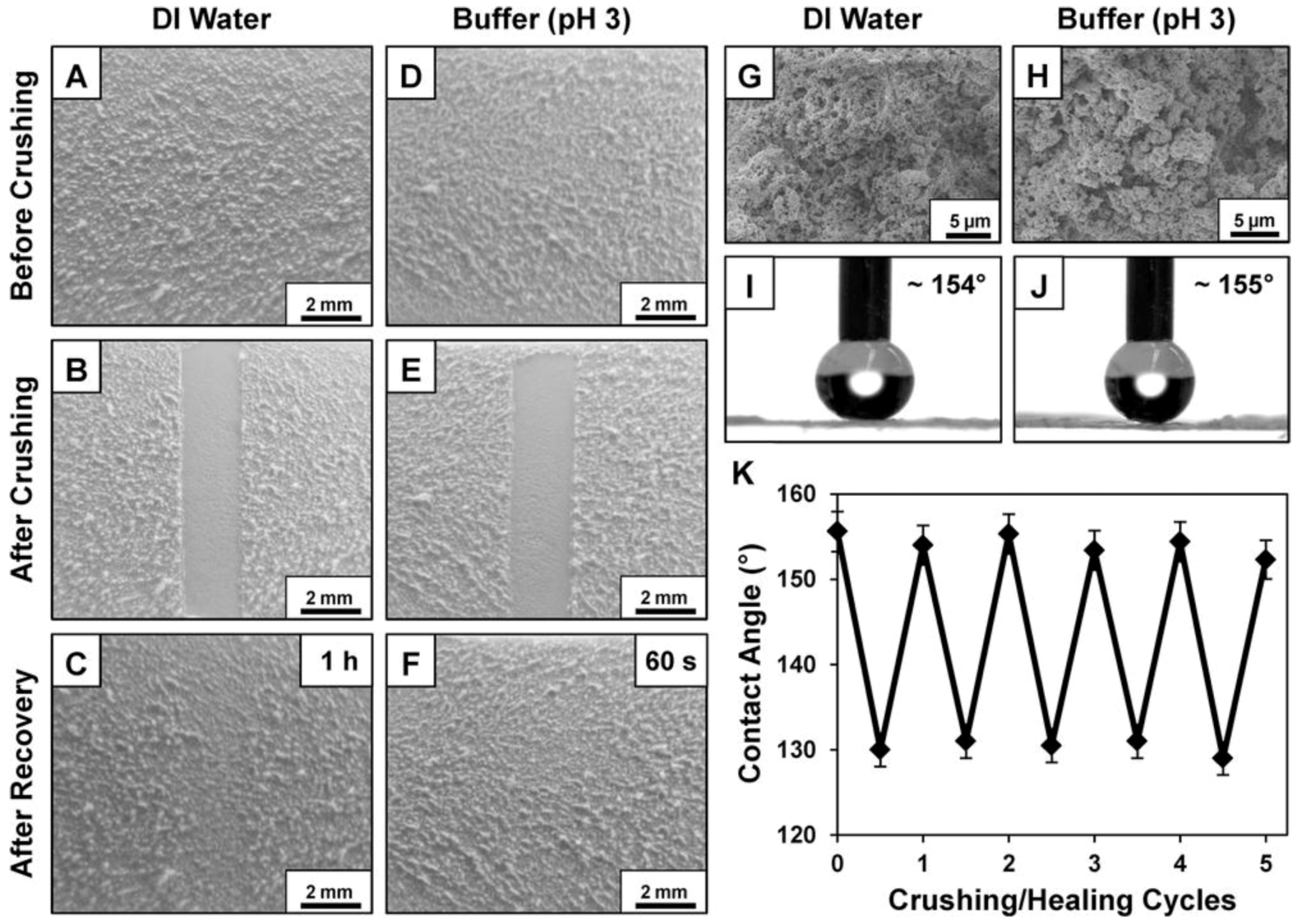
3.4. UV-Shielding/Durable and Self-Healing Superhydrophobic Fabrics
3.5. Other Potential Applications with Special Properties
4. Summary and Future Perspectives
Acknowledgments
Author Contributions
Conflict of Interests
References
- Wang, R.; Hashimoto, K.; Fujishima, A.; Chikuni, M.; Kojima, E.; Kitamura, A.; Shimohigoshi, M.; Watanabe, T. Light-induced amphiphilic surfaces. Nature 1997, 388, 431–432. [Google Scholar] [CrossRef]
- Feng, L.; Li, S.H.; Li, Y.S.; Li, H.J.; Zhang, L.J.; Zhai, J.; Song, Y.L.; Liu, B.Q.; Jiang, L.; Zhu, D.B. Super-hydrophobic surfaces: From natural to artificial. Adv. Mater. 2002, 14, 1857–1860. [Google Scholar] [CrossRef]
- Sun, T.L.; Feng, L.; Gao, X.F.; Jiang, L. Bioinspired surfaces with special wettability. Acc. Chem. Res. 2005, 38, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.T.; Liu, K.S.; Yao, X.; Jiang, L. Bioinspired surfaces with superwettability: New insight on theory, design, and applications. Chem. Rev. 2015, 115, 8230–8293. [Google Scholar] [CrossRef] [PubMed]
- Oberli, L.; Caruso, D.; Hall, C.; Fabretto, M.; Murphy, P.J.; Evans, D. Condensation and freezing of droplets on superhydrophobic surfaces. Adv. Colloid Interfaces Sci. 2014, 210, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Lafuma, A.; Quéré, D. Superhydrophobic states. Nat. Mater. 2003, 2, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Jiang, L. Bio-inspired, smart, multiscale interfacial materials. Adv. Mater. 2008, 20, 2842–2858. [Google Scholar] [CrossRef]
- Liu, M.J.; Zheng, Y.M.; Zhai, J.; Jiang, L. Bioinspired super-antiwetting interfaces with special liquid-solid adhesion. Acc. Chem. Res. 2010, 43, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Darmanin, T.; Guittard, F. Recent advances in the potential applications of bioinspired superhydrophobic materials. J. Mater. Chem. A 2014, 2, 16319–16359. [Google Scholar] [CrossRef]
- Li, X.M.; Reinhoudt, D.; Crego-Calama, M. What do we need for a superhydrophobic surface? A review on the recent progress in the preparation of superhydrophobic surfaces. Chem. Soc. Rev. 2007, 36, 1350–1368. [Google Scholar] [CrossRef] [PubMed]
- Bhushan, B.; Jung, Y.C. Natural and biomimetic artificial surfaces for superhydrophobicity, self-cleaning, low adhesion, and drag reduction. Prog. Mater. Sci. 2011, 56, 1–108. [Google Scholar] [CrossRef]
- Jiang, T.; Guo, Z.G.; Liu, W.M. Biomimetic superoleophobic surfaces: Focusing on their fabrication and applications. J. Mater. Chem. A 2015, 3, 1811–1827. [Google Scholar] [CrossRef]
- Liu, K.S.; Cao, M.Y.; Fujishima, A.; Jiang, L. Bio-inspired titanium dioxide materials with special wettability and their applications. Chem. Rev. 2014, 114, 10044–10094. [Google Scholar] [CrossRef] [PubMed]
- Nagappan, S.; Ha, C.S. Emerging trends in superhydrophobic surface based magnetic materials: Fabrications and their potential applications. J. Mater. Chem. A 2015, 3, 3224–3251. [Google Scholar] [CrossRef]
- Sahoo, B.N.; Kandasubramanian, B. Recent progress in fabrication and characterisation of hierarchical biomimetic superhydrophobic structures. RSC Adv. 2014, 4, 22053–22093. [Google Scholar] [CrossRef]
- Furstner, R.; Barthlott, W.; Neinhuis, C.; Wallzel, P. Wetting and self-cleaning properties of artificial superhydrophobic surfaces. Langmuir 2005, 21, 956–961. [Google Scholar] [CrossRef] [PubMed]
- Marmur, A. The lotus effect: Superhydrophobicity and metastability. Langmuir 2004, 20, 3517–3519. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.C.; McCarthy, T.J. The “lotus effect” explained: Two reasons why two length scales of topography are important. Langmuir 2006, 22, 2966–2967. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. Trans. Faraday Soc. 1944, 40, 546–550. [Google Scholar] [CrossRef]
- Wang, S.; Jiang, L. Definition of superhydrophobic states. Adv. Mater. 2007, 19, 3423–3424. [Google Scholar] [CrossRef]
- Barthlott, W.; Neinhuis, C. Purity of the sacred lotus, or escape from contamination in biological surfaces. Planta 1997, 202, 1–8. [Google Scholar] [CrossRef]
- Feng, L.; Zhang, Y.; Xi, J.; Zhu, Y.; Wang, N.; Xia, F.; Jiang, L. Petal effect: A superhydrophobic state with high adhesive force. Langmuir 2008, 24, 4114–4119. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.M.; Bai, H.; Huang, Z.B.; Tian, X.L.; Nie, F.Q.; Zhao, Y.; Zhai, J.; Jiang, L. Directional water collection on wetted spider silk. Nature 2010, 463, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Parker, A.R.; Lawrence, C.R. Water capture by a desert beetle. Nature 2001, 414, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.F.; Jiang, L. Biophysics: Water-repellent legs of water striders. Nature 2004, 432, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, B.P.; Messersmith, P.B. A reversible wet/dry adhesive inspired by mussels and geckos. Nature 2007, 448, 338–341. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.P.; Wu, L.; Zhang, Y.J.; Wang, A.Q. Mussel and fish scale-inspired underwater superoleophobic kapok membranes for continuous and simultaneous removal of insoluble oils and soluble dyes in water. J. Mater. Chem. A 2015, 3, 18475–18482. [Google Scholar] [CrossRef]
- Teisala, H.; Tuominen, M.; Kuusipalo, J. Superhydrophobic coatings on cellulose-based materials: Fabrication, properties, and applications. Adv. Mater. Interfaces 2014, 1. [Google Scholar] [CrossRef]
- Lai, Y.K.; Chen, Z.; Lin, C.J. Recent progress on the superhydrophobic surfaces with special adhesion: From natural to biomimetic to functional. J. Nanoeng. Nanomanuf. 2011, 1, 18–34. [Google Scholar] [CrossRef]
- Hejazi, I.; Sadeghi, G.M.M.; Jafari, S.H.; Khonakdar, H.A.; Seyfi, J.; Holzschuh, M.; Simon, F. Transforming an intrinsically hydrophilic polymer to a robust self-cleaning superhydrophobic coating via carbon nanotube surface embedding. Mater. Design 2015, 86, 338–346. [Google Scholar] [CrossRef]
- Nguyen, S.H.; Webb, H.K.; Mahon, P.J.; Crawford, R.J.; Ivanova, E.P. Natural insect and plant micro-/nanostructsured surfaces: An excellent selection of valuable templates with superhydrophobic and self-cleaning properties. Molecules 2014, 19, 13614–13630. [Google Scholar] [CrossRef] [PubMed]
- Erbil, H.Y.; Demirel, A.L.; Avci, Y.; Mert, O. Transformation of a simple plastic into a superhydrophobic surface. Science 2003, 299, 1377–1380. [Google Scholar] [CrossRef] [PubMed]
- Xiao, M.; Guo, X.P.; Cheng, M.J.; Ju, G.N.; Zhang, Y.J.; Shi, F. pH-responsive on-off motion of a superhydrophobic boat: Towards the design of a minirobot. Small 2014, 10, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.J.; Liu, Q.; Ju, G.N.; Zhang, Y.J.; Jiang, L.; Shi, F. Superhydrophilic-superhydrophobic-superhydrophilic double transformation on a pH-responsive smart surface. Adv. Mater. 2014, 26, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.K.; Lin, C.J.; Huang, J.Y.; Zhuang, H.F.; Sun, L.; Nguyen, T. Markedly controllable adhesion of superhydrophobic sponge-like nanostructure TiO2 films. Langmuir 2008, 24, 3867–3873. [Google Scholar] [CrossRef] [PubMed]
- Celia, E.; Darmanin, T.; de Givenchy, E.T.; Amigoni, S.; Guittard, F. Recent advances in designing superhydrophobic surfaces. J. Colloid. Interface Sci. 2013, 402, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Wu, X.H.; Fu, Q.T.; Ho, J.W.C.; Kanhere, P.D.; Li, L.; Chen, Z. Hydrophobic sol-gel coatings based on polydimethylsiloxane for self-cleaning applications. Mater. Design 2015, 86, 855–862. [Google Scholar] [CrossRef]
- Lai, Y.K.; Huang, J.Y.; Cui, Z.Q.; Ge, M.Z.; Zhang, K.Q.; Chen, Z.; Chi, L.F. Recent advances in TiO2-based nanostructure surfaces with controllable wettability and adhesion. Small 2016, 12. [Google Scholar] [CrossRef]
- Teng, Y.Q.; Zhang, Y.Q.; Heng, L.P.; Meng, X.F.; Yang, Q.W.; Jiang, L. Conductive polymer porous film with tunable wettability and adhesion. Materials 2015, 8, 1817–1830. [Google Scholar] [CrossRef]
- Lai, Y.K.; Chen, Y.C.; Tang, Y.X.; Gong, D.G.; Chen, Z.; Lin, C.J. Electrophoretic deposition of titanate nanotube films with extremely large wetting contrast. Electrochem. Commun. 2009, 11, 2268–2271. [Google Scholar] [CrossRef]
- Cheng, M.J.; Song, M.M.; Dong, H.Y.; Shi, F. Surface adhesive forces: A metric describing the drag-reducing effects of superhydrophobic coatings. Small 2015, 11, 1665–1671. [Google Scholar] [CrossRef] [PubMed]
- Huan, S.Q.; Liu, G.X.; Han, G.P.; Cheng, W.L.; Fu, Z.Y.; Wu, Q.L.; Wang, Q.W. Effect of experimental parameters on morphological, mechanical and hydrophobic properties of electrospun polystyrene fibers. Materials 2015, 8, 2718–2734. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, Y.B.; Liang, W.X.; Wang, G.Y.; Guo, Z.G.; Liu, W.M. A simple route to transform normal hydrophilic cloth into a superhydrophobic-superhydrophilic hybrid surface. J. Mater. Chem. A 2015, 2, 7845–7852. [Google Scholar] [CrossRef]
- Huang, J.Y.; Lai, Y.K.; Wang, L.N.; Li, S.H.; Ge, M.Z.; Zhang, K.Q.; Fuchs, H.; Chi, L.F. Controllable wettability and adhesion on bioinspired multifunctional TiO2 nanostructure surfaces for liquid manipulation. J. Mater. Chem. A 2014, 2, 18531–18538. [Google Scholar] [CrossRef]
- Lai, Y.K.; Tang, Y.X.; Huang, J.Y.; Wang, H.; Li, H.Q.; Gong, D.G.; Ji, X.B.; Gong, J.J.; Lin, C.J.; Sun, L.; et al. Multi-functional hybrid protonated titanate nanobelts with tunable wettability. Soft Matter 2011, 7, 6313–6319. [Google Scholar] [CrossRef]
- Ma, M.L.; Mao, Y.; Gupta, M.; Gleason, K.K.; Rutledge, G.C. Superhydrophobic fabrics produced by electrospinning and chemical vapor deposition. Macromolecules 2005, 38, 9742–9748. [Google Scholar] [CrossRef]
- Tian, D.L.; Zhang, X.F.; Wang, X.; Zhai, J.; Jiang, L. Micro/nanoscale hierarchical structured ZnO mesh film for separation of water and oil. Phys. Chem. Chem. Phys. 2011, 13, 14606–14610. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.J.; Kota, A.K.; Mabry, J.M.; Tuteja, A. Superomniphobic surfaces for effective chemical shielding. J. Am. Chem. Soc. 2013, 315, 578–581. [Google Scholar] [CrossRef] [PubMed]
- Darmanin, T.; Tarrade, J.; Celia, E.; Bellanger, H.; Guittard, F. Superoleophobic meshes with relatively low hysteresis and sliding angles by electropolymerization: Importance of polymer-growth control. ChemPlusChem 2014, 3, 382–386. [Google Scholar] [CrossRef]
- Zhang, X.; Geng, T.; Guo, Y.G.; Zhang, Z.J.; Zhang, P.Y. Facile fabrication of stable superhydrophobic SiO2/polystyrene coating and separation of liquids with different surface tention. Chem. Eng. J. 2013, 231, 414–419. [Google Scholar] [CrossRef]
- Hu, H.B.; Gao, L.; Chen, C.L.; Chen, Q.W. Low-cost, acid/alkaline-resistant, and fluorine-free superhydrophobic fabric coating from onionlike carbon microspheres converted from waste polyethylene terephthalate. Environ. Sci. Technol. 2014, 48, 2928–2933. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, U.; Erbil, H.Y. Superhydrophobic perfluoropolymer surfaces having heterogeneous roughness created by dip-coating from solutions containing a nonsolvent. Appl. Surf. Sci. 2014, 292, 591–597. [Google Scholar] [CrossRef]
- Lin, J.; Zheng, C.; Ye, W.J.; Wang, H.Q.; Feng, D.Y.; Li, Q.Y.; Huan, B.W. A facile dip-coating approach to prepare SiO2/fluoropolymer coating for superhydrophobic and superoleophobic fabrics with self-cleaning property. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Makowski, T.; Kowalczyk, D.; Fortuniak, W.; Jeziorska, D.; Brzezinski, S.; Tracz, A. Superhydrophobic properties of cotton woven fabrics with conducting 3D networks of multiwall carbon nanotubes, MWCNTs. Cellulose 2014, 21, 4659–4670. [Google Scholar] [CrossRef]
- Wang, J.T.; Chen, Y.H. Oil-water separation capability of superhydrophobic fabrics fabricated via combining polydopamine adhesion with lotus-leaf-like structure. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Nateghi, M.R.; Shateri-Khalilabad, M.R. Silver nanowire-functionalized cotton fabric. J. Carbohyd. Polym. 2015, 117, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Bayer, I.S.; Fragouli, D.; Attanasio, A.; Sorce, B.; Bertoni, G.; Brescia, R.; di Corato, R.; Pellegrino, T.; Kalyva, M.; Sabella, S.; et al. Water-repellent cellulose fiber networks with multifunctional properties. ACS Appl. Mater. Interfaces 2011, 3, 4024–4031. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.H.; Li, M.; Lu, Q.H. Filter paper with selective absorption and separation of liquids that differ in surface tension. ACS Appl. Mater. Interfaces 2010, 2, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Li, S.H.; Zhang, S.B.; Wang, X.H. Fabrication of superhydrophobic cellulose-based materials through a solution-immersion process. Langmuir 2008, 24, 5585–5590. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, H.X.; Niu, H.T.; Gestos, A.; Wang, X.G.; Lin, T. Fluoroalkyl silane modified silicone rubber/nanoparticle composite: A super durable, robust superhydrophobic fabric coating. Adv. Mater. 2012, 24, 2409–2412. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, H.X.; Niu, H.T.; Gestos, A.; Lin, T. Robust, self-healing superamphiphobic fabrics prepared by two-step coating of fluoro-containing polymer, fluoroalkyl silane, and modified silica nanoparticles. Adv. Funct. Mater. 2013, 23, 1664–1670. [Google Scholar] [CrossRef]
- Wang, H.X.; Fang, J.; Cheng, T.; Ding, J.; Qu, L.T.; Dai, L.M.; Wang, X.G.; Lin, T. One-step coating of fluoro-containing silica nanoparticles for universal generation of surface sueperhydrophobicity. Chem. Commun. 2008, 7, 877–879. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhang, J.; Li, B.; Fan, L.; Li, L.; Wang, A. Facile preparation of super durable superhydrophobic materials. J. Colloid. Interface Sci. 2014, 432, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Sasikala, S.; Barshilia, H.C. Corrosion study of superhydrophobic magnesium alloy AZ31 surfaces prepared by wet chemical etching process. Nanosci. Nanotech. Lett. 2012, 4, 757–765. [Google Scholar] [CrossRef]
- Lai, Y.K.; Gao, X.F.; Zhuang, H.F.; Huang, J.Y.; Lin, C.J.; Jiang, L. Designing superhydrophobic porous nanostructures with tunable water adhesion. Adv. Mater. 2009, 21, 3799–3803. [Google Scholar] [CrossRef]
- Lai, Y.K.; Sun, L.; Chen, Y.C.; Zhuang, H.F.; Lin, C.J.; Chin, J.W. Effects of the structure of TiO2 nanotube array on Ti substrate on its photocatalytic activity. J. Electrochem. Soc. 2006, 153, D123–D127. [Google Scholar] [CrossRef]
- Xue, C.H.; Li, Y.R.; Zhang, P.; Ma, J.Z.; Jia, S.T. Washable and wear-resistant superhydrophobic surfaces with self-cleaning property by chemical etching of fibers and hydrophobization. ACS Appl. Mater. Interfaces 2014, 6, 10153–10161. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.W.; Wang, B.; Zhang, Y.B.; Zhu, H.; Guo, Z.G. Strong amphiphobic porous films with oily-self-cleaning property beyond nature. Chem. Lett. 2014, 43, 1566–1568. [Google Scholar] [CrossRef]
- Huang, J.Y.; Li, S.H.; Ge, M.Z.; Wang, L.N.; Xing, T.L.; Chen, G.Q.; Liu, X.F.; Al-Deyab, S.S.; Zhang, K.Q.; Chen, T.; et al. Robust superhydrophobic TiO2@fabrics for UV shielding, self-cleaning and oil-water separation. J. Mater. Chem. A 2015, 3, 2825–2832. [Google Scholar] [CrossRef]
- Zhang, M.; Zang, D.L.; Shi, J.Y.; Gao, Z.X.; Wang, C.Y.; Li, J. Superhydrophobic cotton textile with robust composite film and flame retardancy. RSC Adv. 2015, 5, 67780–67786. [Google Scholar] [CrossRef]
- Kivotidi, S.; Tsioptsias, C.; Pavlidou, E.; Panayiotou, C. Flame-retarded hydrophobic cellulose through impregnation with aqueous solutions and supercritical CO2. J. Therm. Anal. Calorim. 2012, 111, 475–482. [Google Scholar] [CrossRef]
- Xu, B.; Cai, Z.S. Fabrication of a superhydrophobic ZnO nanorod array film on cotton fabrics via a wet chemical route and hydrophobic modification. Appl. Surf. Sci. 2008, 254, 5899–5904. [Google Scholar]
- Li, S.H.; Huang, J.Y.; Ge, M.Z.; Cao, C.Y.; Deng, S.; Zhang, S.N.; Chen, G.Q.; Zhang, K.Q.; Al-Deyab, S.S.; Lai, Y.K. Robust flower-like TiO2@Cotton fabrics with special wettability for effective self-cleaning and versatile oil/water separation. Adv. Mater. Interfaces 2015, 2. [Google Scholar] [CrossRef]
- Zhang, T.; Yan, H.Q.; Fang, Z.P.; E, Y.P.; Wu, T.; Chen, F. Superhydrophobic and conductive properties of carbon nanotubes/polybenzoxazine nanocomposites coated ramie fabric prepared by solution-immersion process. Appl. Surf. Sci. 2014, 309, 218–224. [Google Scholar] [CrossRef]
- Lai, Y.K.; Tang, Y.X.; Gong, J.J.; Gong, D.G.; Chi, L.F.; Lin, C.J.; Chen, Z. Transparent superhydrophobic/superhydrophilic TiO2-based coatings for self-cleaning and anti-fogging. J. Mater. Chem. 2012, 22, 7420–7426. [Google Scholar] [CrossRef]
- Joung, Y.S.; Buie, C.R. Anti-wetting fabric produced by a combination of layer-by-layer assembly and electrophoretic deposition of hydrophobic nanoparticles. ACS Appl. Mater. Interfaces 2015, 7, 20100–20110. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Sarkar, D.K.; Chen, X.G. Superhydrophobic nanostructured ZnO thin films on aluminum alloy substrates by electrophoretic deposition process. Appl. Surf. Sci. 2015, 327, 327–334. [Google Scholar] [CrossRef]
- Ogihara, H.; Katayama, T.; Saji, T. One-step electrophoretic deposition for the preparation of superhydrophobic silica particle/trimethylsiloxysilicate composite coatings. J. Colloid Interface Sci. 2011, 362, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Shi, F.; Wang, Z.Q.; Zhang, X. Combining layer-by-layer assembly with electrodeposition of silver aggregates for fabricating superhydrophobic surfaces. Langmuir 2005, 21, 4713–4716. [Google Scholar] [CrossRef] [PubMed]
- Oktay, B.; Toker, R.D.; Kayaman-Apohan, N. Superhydrophobic behavior of polyimide-siloxane mats produced by electrospinning. Polym. Bull. 2015, 72, 2831–2842. [Google Scholar] [CrossRef]
- Cakir, M.; Kartal, I.; Yildiz, Z. The preparation of UV-cured superhydrophobic cotton fabric surfaces by electrospinning method. Textile Res. J. 2014, 84, 1528–1538. [Google Scholar] [CrossRef]
- Han, D.W.; Steckl, A.J. Superhydrophobic and oleophobic fibers by coaxial electrospinning. Langmuir 2009, 25, 9454–9462. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.P.; Wu, X.F. Electrospinning superhydrophobic-superoleophilic fibrous PVDF membranes for high-efficiency water-oil separation. Mater. Lett. 2015, 160, 423–427. [Google Scholar] [CrossRef]
- Wang, X.F.; Ding, B.; Yu, J.Y.; Wang, M.R. Engineering biomimetic superhydrophobic surfaces of electrospun nanomaterials. Nano Today 2011, 6, 510–530. [Google Scholar] [CrossRef]
- Zhu, M.F.; Zuo, W.W.; Yu, H.; Yang, W.; Chen, Y.M. Superhydrophobic surface directly created by electrospinning based on hydrophilic material. J. Mater. Sci. 2006, 41, 3793–3797. [Google Scholar] [CrossRef]
- Nuraje, N.; Khan, W.S.; Lei, Y.; Ceylan, M.; Asmatulu, R. Superhydrophobic electrospun nanofibers. J. Mater. Chem. A 2013, 1, 1929–1946. [Google Scholar] [CrossRef]
- Spark, B.J.; Hoff, E.F.T.; Xiong, L.; Goetz, J.T.; Patton, D.L. Superhydrophobic hybrid inorganic-organic thiol-ene surfaces fabricated via spray-deposition and photopolymerization. ACS Appl. Mater. Interfaces 2013, 5, 1811–1817. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Tang, Y.C.; Xu, J.Q.; Chen, B.B.; Tang, H.; Li, C.S. Durable superhydrophobic/superoleophilic epoxy/attapulgite nanocomposite coatings for oil/water separation. Surf. Coat. Technol. 2015, 272, 285–290. [Google Scholar] [CrossRef]
- Ge, D.T.; Yang, L.L.; Wu, G.X.; Yang, S. Spray coating of superhydrophobic and angle-independent coloured films. Chem. Commun. 2014, 50, 2469–2472. [Google Scholar] [CrossRef] [PubMed]
- Li, B.C.; Zhang, J.P.; Wu, L.; Wang, A.Q. Durable superhydrophobic surfaces prepared by spray coating of polymerized organosilane/attapulgite nanocomposites. ChemPlusChem 2013, 78, 1503–1509. [Google Scholar] [CrossRef]
- Chu, Z.L.; Seeger, S. Robust superhydrophobic wood obtained by spraying silicone nanoparticles. RSC Adv. 2015, 5, 21999–22004. [Google Scholar] [CrossRef]
- Li, J.; Jing, Z.J.; Zha, F.; Yang, Y.X.; Wang, Q.T.; Lei, Z.Q. Facile spray-coating process for the fabrication of tunable adhesive superhydrophobic surfaces with heterogeneous chemical compositions used for selective transportation of microdroplets with different volumes. ACS Appl. Mater. Interfaces 2014, 6, 8868–8877. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Z.; Zhang, X.Y.; Bao, Y.; Liu, J.L. A facile spraying method for fabricating superhydrophobic leather coating. Colloids Surf. A 2015, 472, 21–25. [Google Scholar] [CrossRef]
- Kim, D.Y.; Lee, J.G.; Joshi, B.N.; Latthe, S.S.; Al-Deyab, S.S.; Yoon, S.S. Self-cleaning superhydrophobic films by supersonic-spraying polytetrafluoroethylene-titania nanoparticles. J. Mater. Chem. A 2015, 3, 3975–3983. [Google Scholar] [CrossRef]
- Artus, C.R.J.; Jung, S.; Zimmermann, J.; Gautschi, H.P.; Marquardt, K.; Seeger, S. Silicone nanofilaments and their application as superhydrophobic coatings. Adv. Mater. 2006, 18, 2758–2762. [Google Scholar] [CrossRef]
- Balu, B.; Breedveld, V.; Hess, D.W. Fabrication of “roll-off” and “sticky” superhydrophobic cellulose surfaces via plasma processing. Langmuir 2008, 24, 4785–4790. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.M.; Cui, J.F.; Sun, H.X.; Liang, W.D.; Zhu, Z.Q.; An, J.; Yang, B.P.; La, P.Q.; Li, A. Facile preparation of superhydrophobic surfaces based on metal oxide nanoparticles. Appl. Surf. Sci. 2014, 303, 473–480. [Google Scholar] [CrossRef]
- Zhou, X.Y.; Zhang, Z.Z. Robust and durable superhydrophobic cotton fabrics for oil/water separation. ACS Appl. Mater. Interfaces 2013, 5, 7208–7214. [Google Scholar] [CrossRef] [PubMed]
- Aminayi, P.; Abidi, N. Ultra-oleophobic cotton fabric prepared using molecular and nanoparticle vapor deposition methods. Surf. Coat. Technol. 2015, 276, 636–644. [Google Scholar] [CrossRef]
- Aminayi, P.; Abidi, N. Imparting super hydro/oleophobic properties to cotton fabric by means of molecular and nanoparticles vapor deposition methods. Appl. Surf. Sci. 2013, 287, 223–231. [Google Scholar] [CrossRef]
- Oh, J.; Ko, T.; Moon, M.; Park, C.H. Nanostructured superhydrophobic silk fabric fabricated using the ion beam method. RSC Adv. 2014, 4, 38966–38973. [Google Scholar] [CrossRef]
- Zheng, Z.R.; Gu, Z.Y.; Huo, R.T.; Ye, Y.H. Superhydrophobicity of polyvinylidene fluoride membrane fabricated by chemical vapor deposition from solution. Appl. Surf. Sci. 2009, 255, 7263–7267. [Google Scholar] [CrossRef]
- Chakraborty, A.; Xiang, M.M.; Luo, C. Fabrication of super-hydrophobic microchannels via strain-recovery deformations of polystyrene and oxygen reactive ion etch. Materials 2013, 6, 3610–3623. [Google Scholar] [CrossRef]
- Caschera, D.; Mezzi, A.; Cerri, L.; de Caro, T.; Riccucci, C.; Ingo, G.M.; Padeletti, G.; Biasiucci, M.; Gigli, G.; Cortese, B. Effects of plasma treatments for improving extreme wettability behavior of cotton fabrics. Cellulose 2014, 21, 741–756. [Google Scholar] [CrossRef]
- Caschera, D.; Toro, R.G.; Federici, F.; Riccucci, C.; Ingo, G.M.; Gigli, G.; Cortese, B. Flame retardant properties of plasma pre-treated/diamond-like carbon (DLC) coated cotton fabrics. Cellulose 2015, 22, 2797–2809. [Google Scholar] [CrossRef]
- Kwon, S.O.; Ko, T.J.; Yu, E.S.; Kim, J.; Moon, M.W.; Park, C.H. Nanostructured self-cleaning lyocell fabrics with asymmetric wettability and moisture absorbency (part I). RSC Adv. 2014, 4, 45442–45448. [Google Scholar] [CrossRef]
- Kinoshita, H.; Ogasahara, A.; Fukuda, Y.; Ohmae, N. Superhydrophobic/superhydrophilic micropatterning on a carbon nanotube film using a laser plasma-type hyperthermal atom beam facility. Carbon 2010, 48, 4403–4408. [Google Scholar] [CrossRef]
- Yang, C.; Li, X.M.; Gilron, J.; Kong, D.F.; Yin, Y.; Oren, Y.; Linder, C.; He, T. CF4 plasma-modified superhydrophobic PVDF membranes for direct contact membrane distillation. J. Membrane Sci. 2014, 456, 155–161. [Google Scholar] [CrossRef]
- Ellinas, K.; Pujari, S.P.; Dragatogiannis, D.A.; Charitidis, C.A.; Tserepi, A.; Zuilhof, H.; Gogolides, E. Plasma micro-nanotextured, scratch, water and hexadecane resistant, superhydrophobic, and superamphiphobic polymeric surfaces with perfluorinated monolayers. ACS Appl. Mater. Interfaces 2014, 6, 6510–6524. [Google Scholar] [CrossRef] [PubMed]
- Li, S.H.; Huang, J.Y.; Ge, M.Z.; Li, S.W.; Xing, T.L.; Chen, G.Q.; Liu, Y.Q.; Zhang, K.Q.; Al-Deyab, S.S.; Lai, Y.K. Controlled grafting superhydrophobic cellulose surface with environmentally-friendly short fluoroalkyl chains by ATRP. Mater. Design 2015, 85, 815–822. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.Q.; Shao, J.Z.; Zou, C. Fabrication of superhydrophobic fluorine-free films on cotton fabrics through plasma-induced grafting polymerization of 1,3,5,7-tetravinyl-1,3,5,7-tetramethyl cyclotetrasiloxane. Surf. Coat. Technol. 2015, 276, 16–22. [Google Scholar] [CrossRef]
- Li, Y.W.; Zheng, X.W.; Zhu, H.Y.; Wu, K.; Lu, M.G. Synthesis and self-assembly of well-defined binary graft copolymer and its use in superhydrophobic cotton fabrics preparation. RSC Adv. 2015, 5, 46132–46145. [Google Scholar] [CrossRef]
- Duan, W.; Xie, A.J.; Shen, Y.H.; Wang, Y.F.; Wang, F.; Zhang, Y.; Li, J.L. Fabrication of superhydrophobic cotton fabrics with UV protection based on CeO2 particles. Ind. Eng. Chem. Res. 2011, 50, 4441–4445. [Google Scholar] [CrossRef]
- Wang, H.X.; Ding, J.; Xue, Y.H.; Wang, X.G.; Lin, T. Superhydrophobic fabrics from hybrid silica sol-gel coatings: Structural effect of precursors on wettability and washing durability. J. Mater. Res. 2010, 25, 1336–1343. [Google Scholar] [CrossRef]
- Gao, Q.W.; Zhu, Q.; Guo, Y.L.; Yang, C.Q. Formation of highly hydrophobic surfaces on cotton and polyester fabrics using silica sol nanoparticles and nonfluorinated alkylsilane. Ind. Eng. Chem. Res. 2009, 48, 9797–9803. [Google Scholar] [CrossRef]
- Gurav, A.B.; Xu, Q.F.; Latthe, S.S.; Vhatkar, R.S.; Liu, S.H.; Yoon, H.; Yoon, S.S. Superhydrophobic coatings prepared from methyl-modified silica particles using simple dip-coating method. Ceram. Int. 2015, 41, 3017–3023. [Google Scholar] [CrossRef]
- Zhu, T.; Cai, C.; Duan, C.T.; Zhai, S.; Liang, S.M.; Jin, Y.; Zhao, N.; Xu, J. Robust polypropylene fabrics super-repelling various liquids: A simple, rapid and scalable fabrication method by solvent swelling. ACS Appl. Mater. Interfaces 2015, 7, 13996–14003. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.X.; Liu, X.; Yang, H.Y.; Dong, B.H.; Zhou, Y.S.; Chen, D.Z.; Hu, H.; Xiao, X.F.; Fan, D.F.; Zhang, C.H.; et al. A simple one-step approach to fabrication of highly hydrophobic silk fabrics. Appl. Surf. Sci. 2016, 360, 207–212. [Google Scholar] [CrossRef]
- Xiao, X.F.; Cao, G.Y.; Chen, F.X.; Tang, Y.R.; Liu, X.; Xu, W.L. Durable superhydrophobic wool fabrics coating with nanoscale Al2O3 layer by atomic layer deposition. Appl. Surf. Sci. 2015, 349, 876–879. [Google Scholar] [CrossRef]
- Cortese, B.; Caschera, D.; Federici, F.; Ingo, G.M.; Gigli, G. Superhydrophobic fabrics for oil-water separation through a diamond like carbon (DLC) coating. J. Mater. Chem. A 2014, 2, 6781–6789. [Google Scholar] [CrossRef]
- Wang, F.J.; Yu, S.; Ou, J.F.; Xue, M.S.; Li, W. Mechanically durable superhydrophobic surfaces prepared by abrading. J. Appl. Phys. 2013, 114. [Google Scholar] [CrossRef]
- Yu, M.; Wang, Z.Q.; Liu, H.Z.; Xie, S.Y.; Wu, J.X.; Jiang, H.Q.; Zhang, J.Y.; Li, L.F.; Li, J.Y. Laundering durability of photocatalyzed self-cleaning cotton fabric with TiO2 nanoparticles covalently immobilized. ACS Appl. Mater. Interfaces 2013, 5, 3697–3703. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.T.; Zhang, B.W.; Wu, J.X.; Wang, Z.Q.; Ma, H.J.; Yu, M.; Li, L.F.; Li, J.Y. Electrical switchability and dry-wash durability of conductive textiles. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Li, J.; Wang, G.Y.; Liang, W.X.; Zhang, Y.B.; Shi, L.; Guo, Z.G.; Liu, W.M. Methodology for robust superhydrophobic fabrics and sponges from in situ growth of transition metal/metal oxide nanocrystals with thiol modification and their applications in oil/water separation. ACS Appl. Mater. Interfaces 2013, 5, 1827–1839. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.G.; Zhou, F.; Hao, J.C.; Liu, W.M. Stable biomimetic super-hydrophobic engineering materials. J. Am. Chem. Soc. 2005, 127, 15670–15671. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liang, W.X.; Guo, Z.G.; Liu, W.M. Biomimetic super-lyophobic and super-lyophilic materials applied for oil/water separation: a new strategy beyond nature. Chem. Soc. Rev. 2015, 44, 336–361. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ma, M.L.; Zang, D.L.; Gao, Z.X.; Wang, C.Y. Fabrication of superhydrophobic/superoleophilic cotton for application in the field of water/oil separation. Carbohyd. Poly. 2014, 103, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Y. Mussel-inspired one-step copolymerization to engineer hierarchically structured surface with superhydrophobic properties for removing oil from water. ACS Appl. Mater. Inter 2014, 6, 17144–17150. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.X.; Xu, Y.C.; Liu, Y.Y.; Shao, L.J. A novel mussel-inspired strategy toward superhydrophobic surfaces for self-driven crude oil spill cleanup. J. Mater. Chem. A 2015, 3, 12171–12178. [Google Scholar] [CrossRef]
- Wang, Y.C.; Tao, S.Y.; An, Y.G. A reverse membrane emulsification process based on a hierarchically porous monolith for high efficiency water-oil separation. J. Mater. Chem. A 2013, 1, 1701–1708. [Google Scholar] [CrossRef]
- Zhu, X.T.; Zhang, Z.Z.; Ge, B.; Men, X.H.; Zhou, X.Y.; Xue, Q.J. A versatile approach to produce superhydrophobic materials used for oil-water separation. J. Colloid. Interface Sci. 2014, 432, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.F.; Lu, X.; Xin, Z.; Zhou, C.L. A self-cleaning polybenzoxazine/TiO2 surface with superhydrophobicity and superoleophilicity for oil/water separation. Nanoscale 2015, 7, 19476–19483. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zhou, Y.; Jiang, G.H.; Wang, R.J.; Wang, X.H.; Hu, R.B.; Xi, X.G. Transformation of hydrophilic cotton fabrics into superhydrophobic surfaces for oil/water separation. J. Text. Inst. 2013, 104, 305–311. [Google Scholar] [CrossRef]
- Song, J.; Huang, S.; Lu, Y.; Bu, X.; Mates, J.E.; Ghosh, A.; Ganguly, R.; Carmalt, C.J.; Parkin, I.P.; Xu, W.; et al. Self-driven one-step oil removal from oil spill on water via selective-wettability steel mesh. ACS Appl. Mater. Interfaces 2014, 6, 19858–19865. [Google Scholar] [CrossRef] [PubMed]
- Zang, D.L.; Liu, F.; Zhang, M.; Niu, X.G.; Gao, Z.X.; Wang, C.Y. Superhydrophobic coating on fiberglass cloth for selective removal of oil from water. Chem. Eng. J. 2015, 262, 210–216. [Google Scholar] [CrossRef]
- Li, J.; Yan, L.; Zhao, Y.Z.; Zha, F.; Wang, Q.T.; Lei, Z.Q. Correction: One-step fabrication of robust fabrics with both-faced superhydrophobicity for the separation and capture of oil from water. Phys. Chem. Chem. Phys. 2015, 17, 6451–6457. [Google Scholar] [CrossRef] [PubMed]
- Dudchenko, A.V.; Rolf, J.; Shi, L.; Olivas, L.; Duan, W.Y.; Jassby, D. Coupling underwater superoleophobic membranes with magnetic pickering emulsions for fouling-free separation of crude oil/water mixtures: An experimental and theoretical study. ACS Nano 2015, 9, 9930–9941. [Google Scholar] [CrossRef] [PubMed]
- Li, K.Q.; Zeng, X.R.; Li, H.Q.; Lai, X.J.; Xie, H. Facile fabrication of superhydrophobic filtration fabric with honeycomb structures for the separation of water and oil. Mater. Lett. 2014, 120, 255–258. [Google Scholar] [CrossRef]
- Arumugham, T.; Kaleekkal, N.J.; Rana, D.; Doraiswamy, M. Separation of oil/water emulsions using nano MgO anchored hybrid ultrafiltration membranes for environmental abatement. J. Appl. Poly. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Huang, M.L.; Si, Y.; Tang, X.M.; Zhu, Z.G.; Ding, B.; Liu, L.F.; Zheng, G.; Luo, W.J.; Yu, J.Y. Gravity driven separation of emulsified oil-water mixtures utilizing in situ polymerized superhydrophobic and superoleophilic nanofibrous membranes. J. Mater. Chem. A 2013, 1, 14071–14074. [Google Scholar] [CrossRef]
- Si, Y.; Fu, Q.; Wang, X.; Zhu, J.; Yu, J.; Sun, G.; Ding, B. Superelastic and superhydrophobic nanofiber-assembled cellular aerogels for effective separation of oil/water emulsions. ACS Nano 2015, 9, 3791–3799. [Google Scholar] [CrossRef] [PubMed]
- Si, Y.; Yan, CC.; Hong, F.F.; Yu, J.Y.; Ding, B. A general strategy for fabricating flexible magnetic silica nanofibrous membranes with multifunctionality. Chem. Commun. 2015, 51, 12521–12524. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.B.; Zhang, Z.H.; Wang, P. Smart surfaces with switchable superoleophilicity and superoleophobicity in aqueous media: toward controllable oil/water separation. NPG Asia Mater. 2012, 4. [Google Scholar] [CrossRef]
- Jin, Y.X.; Ke, Q.P.; Jiang, P.; Zhu, Y.S.N.; Cheng, F.H.; Zhang, Y.X. High efficiently oil/water separation and excellent self-cleaning surfaces based on 1-triacontanol-polymerized octadecylsiloxane coatings. Appl. Surf. Sci. 2015, 351, 358–366. [Google Scholar] [CrossRef]
- Poortavasoly, H.; Montazer, M.; Harifi, T. Aminolysis of polyethylene terephthalate surface along with in situ synthesis and stabilizing ZnO nanoparticles using triethanolamine optimized with response surface methodology. Mater. Sci. Eng. C 2016, 58, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Greppi, G.; Malfatti, L.; Lasio, B.; Sanna, V.; Mura, M.E.; Marceddu, S.; Lugliè, L. Multifunctionalization of wool fabrics through nanoparticles: A chemical route towards smart textiles. J. Colloid Interface Sci. 2015, 456, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Dionysiou, D.D.; Pillai, S.C. Self-cleaning applications of TiO2 by photo-induced hydrophilicity and photocatalysis. Appl. Catal. B 2015, 176, 396–428. [Google Scholar] [CrossRef]
- Shahidi, S.; Ahmadi, M.; Rashidi, A.; Ghoranneviss, M. Effect of plasma treatment on self-cleaning of textile fabric using titanium dioxide. Micro Nano Lett. 2015, 10, 408–413. [Google Scholar]
- Manna, J.; Goswami, S.; Shilpa, N.; Sahu, N.; Rana, R.K. Biomimetic method to assemble nanostructured Ag@ZnO on cotton fabrics: Application as self-cleaning flexible materials with visible-light photocatalysis and antibacterial activities. ACS Appl. Mater. Interfaces 2015, 7, 8076–8082. [Google Scholar] [CrossRef] [PubMed]
- Ge, M.Z.; Cao, C.Y.; Huang, J.Y.; Li, S.H.; Chen, Z.; Zhang, K.Q.; Al-Deyab, S.S.; Lai, Y.K. A review of one-dimensional TiO2 nanostructured materials for environmental and energy applications. J. Mater. Chem. A 2016, 4. [Google Scholar] [CrossRef]
- Xu, B.; Ding, J.; Feng, L.; Ding, Y.Y.; Ge, F.Y.; Cai, Z.S. Self-cleaning cotton fabrics via combination of photocatalytic TiO2 and superhydrophobic SiO2. Surf. Coat. Technol. 2015, 262, 70–76. [Google Scholar] [CrossRef]
- Zheng, X.; Guo, Z.Y.; Tian, D.L.; Zhang, X.F.; Li, W.X.; Jiang, L. Underwater self-cleaning scaly fabric membrane for oily water separation. ACS Appl. Mater. Interfaces 2015, 7, 4336–4343. [Google Scholar] [CrossRef] [PubMed]
- Li, J.H.; Park, E.J.; Kim, D.H.; Jeong, M.G.; Kim, Y.D. Superhydrophobic surfaces with photocatalytic activity under UV and visible light irradiation. Catal. Today 2016, 260, 32–38. [Google Scholar]
- Afzal, S.; Daoud, W.A.; Langford, S.J. Superhydrophobic and photocatalytic self-cleaning cotton. J. Mater. Chem. A 2014, 2, 18005–18011. [Google Scholar] [CrossRef]
- Khajavi, R.; Berendjchi, A. Effect of dicarboxylic acid chain length on the self-cleaning property of nano-TiO2-coated cotton fabrics. ACS Appl. Mater. Interfaces 2014, 6, 18795–18799. [Google Scholar] [CrossRef] [PubMed]
- Behzadnia, A.; Montazer, M.; Rashidi, A.; Rad, M.M. Rapid sonosynthesis of N-doped nano TiO2 on wool fabric at low temperature: introducing self-cleaning, hydrophilicity, antibacterial/antifungal properties with low alkali solubility, yellowness and cytotoxicity. J. Photochem. Photobiol. 2014, 90, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- Karimi, L.; Yazdanshenas, M.E.; Khajavi, R.; Rashidi, A.; Mirjalili, M. Using graphene/TiO2 nanocomposite as a new route for preparation of electroconductive, self-cleaning, antibacterial and antifungal cotton fabric without toxicity. Cellulose 2014, 21, 3813–3827. [Google Scholar] [CrossRef]
- Teng, C.; Lu, X.Y.; Ren, G.Y.; Zhu, Y.; Wan, M.X.; Jiang, L. Underwater self-cleaning PEDOT-PSS hydrogel mesh for effective separation of corrosive and hot oil/water mixtures. Adv. Mater. Interfaces 2014, 1. [Google Scholar] [CrossRef]
- Zheng, C.H.; Qi, Z.M.; Shen, W.C.; Chen, G.Q. Self-cleaning Bombyx mori silk: Room-temperature preparation of anatase nano-TiO2 by the sol-gel method and its application. Color. Technol. 2014, 130, 280–287. [Google Scholar] [CrossRef]
- Ragesh, P.; Nair, S.V.; Nair, A.S. An attempt to fabricate a photocatalytic and hydrophobic self-cleaning coating via electrospinning. RSC Adv. 2014, 4, 38498–38504. [Google Scholar] [CrossRef]
- Barletta, M.; Vesco, S.; Tagliaferri, V. In situ sonosynthesis of nano TiO2 on cotton fabric. J. Ultrason. Sonochem. 2014, 21, 681–691. [Google Scholar]
- Zohoori, S.; Karimi, L.; Nazari, A. Photocatalytic self-cleaning synergism optimization of cotton fabric using nano SrTiO3 and nano TiO2. J. Fibres. Text. East. Eur. 2014, 2, 91–95. [Google Scholar]
- Xue, C.H.; Jia, S.T.; Zhang, J.; Ma, J.Z. Large-area fabrication of superhydrophobic surfaces for practical applications: An overview. Sci. Technol. Adv. Mater. 2010, 11. [Google Scholar] [CrossRef]
- Barletta, M.; Vesco, S.; Tagliaferri, V. Self-cleaning and self-sanitizing coatings on plastic fabrics: Design, manufacture and performance. J. Colloid Surface B 2014, 120, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.Y.; Liu, Y.; Xu, Q.F.; Barahman, M.; Lyons, A.M. Catalytic, self-cleaning surface with stable superhydrophobic properties: Printed polydimethylsiloxane (PDMS) arrays embedded with TiO2 nanoparticles. ACS Appl. Mater. Interfaces 2015, 7, 2632–2640. [Google Scholar] [CrossRef] [PubMed]
- Ge, M.Z.; Cao, C.Y.; Huang, J.Y.; Li, S.H.; Zhang, S.N.; Deng, S.; Li, Q.S.; Zhang, K.Q.; Lai, Y.K. Synthesis, modification, and photo/photoelectro catalytic degradation applications of TiO2 nanotube arrays: A review. Nanotechnol. Rev. 2016, 5, 75–112. [Google Scholar]
- Wang, L.; Xi, G.H.; Wan, S.J.; Zhao, C.H.; Liu, X.D. Asymmetrically superhydrophobic cotton fabrics fabricated by mist polymerization of lauryl methacrylate. Cellulose 2014, 21, 2983–2994. [Google Scholar] [CrossRef]
- Xi, G.H.; Fan, W.C.; Wang, L.; Liu, X.D.; Endo, T. Fabrication of asymmetrically superhydrophobic cotton fabrics via mist copolymerization of 2,2,2-trifluoroethyl methacrylate. J. Polym. Sci. Part A 2015, 53, 1862–1871. [Google Scholar] [CrossRef]
- Liu, Y.; Xin, J.H.; Choi, C.H. Cotton fabrics with single-faced superhydrophobicity. Langmuir 2012, 28, 17426–17434. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.C.; Xiao, P.; Chen, J.; Zhang, J.W.; Huang, Y.J.; Chen, T. Janus polymer/carbon nanotube hybrid membranes for oil/water separation. ACS Appl. Mater. Interfaces 2014, 6, 16204–16209. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.X.; Ding, J.; Dai, L.M.; Wang, X.G.; Lin, T. Directional water-transfer through fabrics induced by asymmetric wettability. J. Mater. Chem. 2010, 20, 7938–7940. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.W.; Fei, B.; Hu, H.W.; Lai, C.L.; Xin, J.H. Bioinspired, stimuli-responsive, multifunctional superhydrophobic surface with directional wetting, adhesion, and transport of water. Adv. Funct. Mater. 2015, 25, 5047–5056. [Google Scholar] [CrossRef]
- Wang, H.X.; Zhou, H.; Yang, W.D.; Zhao, Y.; Fang, J.; Lin, T. Selective, spontaneous one-way oil-transport fabrics and their novel use for gauging liquid surface tension. ACS Appl. Mater. Interfaces 2015, 7, 22874–22880. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Liu, Y.; Xin, J.H. Fabrics with self-adaptive wettability controlled by “light-and-dark”. J. Mater. Chem. 2011, 21, 17978–17987. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, H.X.; Niu, H.T.; Lin, T. Superphobicity/philicity Janus fabrics with switchable, spontaneous, directional transport ability to water and oil fluids. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.S.; Park, S.H.; Koo, S.H.; Kwark, Y.J.; Thomas, E.L.; Jeong, Y.J.; Cho, J.H. Superamphiphilic Janus fabric. Langmuir 2010, 26, 19159–19162. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhou, H.; Niu, H.; Zhang, J.; Du, Y.; Lin, T. Dual-layer superamphiphobic/superhydrophobic-oleophilic nanofibrous membranes with unidirectional oil-transport ability and strengthened oil-water separation performance. Adv. Mater. Interfaces 2015, 2. [Google Scholar] [CrossRef]
- Tian, X.L.; Jin, H.; Sainio, J.; Ras, R.H.A.; Ikkala, O. Droplet and fluid gating by biomimetic Janus membranes. Adv. Funct. Mater. 2014, 24, 6023–6028. [Google Scholar] [CrossRef]
- Xue, C.H.; Yin, W. UV-durable superhydrophobic textiles with UV-shielding properties by coating fibers with ZnO/SiO2 core/shell particles. Nat Nanotechnol. 2011, 22. [Google Scholar] [CrossRef]
- Xue, C.H.; Yin, W. UV-durable superhydrophobic textiles with UV-shielding properties by introduction of ZnO/SiO2 core/shell nanorods on PET fibers and hydrophobization. Colloid Surf. A. 2013, 427, 7–12. [Google Scholar] [CrossRef]
- Wu, J.X.; Li, J.Y.; Wang, Z.Q.; Yu, M.; Jiang, H.Q.; Li, L.F.; Zhang, B.W. Designing breathable superhydrophobic cotton fabrics. RSC Adv. 2015, 5, 27752–27758. [Google Scholar] [CrossRef]
- Zeng, C.; Wang, H.X.; Zhou, H.; Lin, T. Self-cleaning, superhydrophobic cotton fabrics with excellent washing durability, solvent resistance and chemical stability prepared from an SU-8 derived surface coating. RSC Adv. 2015, 5, 61044–61050. [Google Scholar] [CrossRef]
- Brown, P.S.; Bhushan, B. Mechanically durable, superoleophobic coatings prepared by layer-by-layer technique for anti-smudge and oil-water separation. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Zhang, X.T. Superhydrophobic and ultraviolet-blocking cotton textiles. ACS Appl. Mater. Interfaces 2011, 3, 1277–1281. [Google Scholar] [CrossRef] [PubMed]
- Verho, T.; Bower, C. Mechanically durable superhydrophobic surfaces. Adv. Mater. 2011, 23, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Huang, W.Q. Preparation of durable superhydrophobic surface by sol-gel method with water glass and citric acid. Sol-Gel Sci. Technol. 2011, 58, 18–23. [Google Scholar] [CrossRef]
- Zou, L.H.; Lan, C.T. Superhydrophobization of cotton fabric with multiwalled carbon nanotubes for durable electromagnetic interference shielding. Fiber. Polym. 2015, 16, 2158–2164. [Google Scholar] [CrossRef]
- Abbas, R.; Khereby, M.A. Fabrication of durable and cost effective superhydrophobic cotton textiles via simple one step process. Cellulose 2015, 22, 887–896. [Google Scholar] [CrossRef]
- Zhou, H.; Wang, H.; Niu, H.; Fang, J.; Zhao, Y.; Lin, T. Superstrong, chemically stable, superamphiphobic fabrics from particle-free polymer coatings. Adv. Mater. Interfaces 2015, 2. [Google Scholar] [CrossRef]
- Tian, D.; Zhang, X.; Tian, Y.; Wu, Y.; Wang, X.; Zhai, J.; Jiang, L. Photo-induced water-oil separation based on switchable superhydrophobicity-superhydrophilicity and underwater superoleophobicity of the aligned ZnO nanorod array-coated mesh films. J. Mater. Chem. 2012, 22, 19652–19657. [Google Scholar] [CrossRef]
- Dunderdale, G.J.; Urata, C.; Miranda, D.F.; Hozumi, A. Large-scale and environmentally friendly synthesis of pH-responsive oil-repellent polymer brush surfaces under ambient conditions. ACS Appl. Mater. Interfaces 2014, 6, 11864–11868. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.T.; Choi, C.H.; Gao, N.; Yang, E.H. Tunable wetting mechanism of polypyrrole surfaces and low-voltage droplet manipulation via redox. Langmuir 2011, 27, 4249–4256. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.M.; Sarshar, M.A.; Du, K.; Chou, T.; Choi, C.H.; Sukhishvili, S.A. Large-amplitude, reversible, pH-triggered wetting transitions enabled by layer-by-layer films. ACS Appl. Mater. Interfaces 2013, 5, 12617–12623. [Google Scholar] [CrossRef] [PubMed]
- Kota, A.K.; Kwon, G.; Choi, W.; Mabry, J.M.; Tuteja, A. Hygro-responsive membranes for effective oil-water separation. Nat. Commun. 2012, 3, 1025. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Guo, Z.G.; Liu, W.M. pH-responsive smart fabrics with controllable wettability in different surroundings. RSC Adv. 2014, 4, 14684–14690. [Google Scholar] [CrossRef]
- Shateri-Khalilabad, M.; Yazdanshenas, M.E. Preparation of superhydrophobic electroconductive graphene-coated cotton cellulose. Cellulose 2013, 20, 963–972. [Google Scholar] [CrossRef]
- Li, M.M.; Qing, G.Y.; Xiong, Y.T.; Lai, Y.K.; Sun, T.L. CH-π interaction driven macroscopic property transition on smart polymer surface. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.H.; Ma, J.Z. Long-lived superhydrophobic surfaces. J. Mater. Chem. A. 2013, 1, 4146–4161. [Google Scholar] [CrossRef]
- Yin, X.Y.; Liu, Z.L.; Wang, D.A.; Pei, X.W.; Yu, B.; Zhou, F. Bioinspired self-healing organic materials: Chemical mechanisms and fabrications. J. Bionic. Eng. 2015, 12, 1–16. [Google Scholar] [CrossRef]
- Li, Y.; Li, L.; Sun, J.Q. Bioinspired self-healing superhydrophobic coatings. Angew. Chem. Int. Ed. 2010, 49, 6129–6133. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, S.S.; Wu, M.C.; Sun, J.Q. All spraying processes for the fabrication of robust, self-healing, superhydrophobic coatings. Adv. Mater. 2014, 26, 3344–3348. [Google Scholar] [CrossRef] [PubMed]
- Manna, U.; Lynn, D.M. Restoration of superhydrophobicity in crushed polymer films by treatment with water: Self-healing and recovery of damaged topographic features aided by an unlikely source. Adv. Mater. 2013, 25, 5104–5108. [Google Scholar] [CrossRef] [PubMed]
- Shillingford, C.; MacCallum, N. Fabrics coated with lubricated nanostructures display robustomniphobicity. Nanotechnology 2014, 25. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.S.; Li, X.; Li, Y.; Sun, J.Q. Intumescent flame-retardant and self-healing superhydrophobic coatings on cotton fabric. ACS Nano. 2015, 9, 4070–4076. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.H.; Zhang, Z.D. Lasting and self-healing superhydrophobic surfaces by coating of polystyrene/SiO2 nanoparticles and polydimethylsiloxane. J. Mater. Chem. A. 2014, 2. [Google Scholar] [CrossRef]
- Si, Y.F.; Zhu, H.; Chen, L.W.; Jinag, T.; Guo, Z.G. A multifunctional transparent superhydrophobicgel nanocoating with self-healing properties. Chem. Comm. 2015, 51, 16794–16797. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.L.; Zhou, S.X. Fabrication of all-water-based self-repairing superhydrophbic coatings based on UV-responsive microcapsules. Adv. Funct. Mater. 2015, 25, 1035–1041. [Google Scholar] [CrossRef]
- Esteves, A.C.C.; Luo, Y.; van de Put, M.W.P. Self-Replenishing dual structured superhydrophobic coatings prepared by drop-casting of an all-in-one dispersion. Adv. Funct. Mater. 2014, 24, 986–992. [Google Scholar] [CrossRef]
- Wu, J.X.; Li, J.Y. Self-healing of the sueperhydrophobicity by ironing for the abrasion durable superhydrophobic cotton fabrics. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef]
- Wang, H.X.; Xue, Y.H.; Deng, J.; Feng, L.F.; Wang, X.G.; Lin, T. Durable, self-healing superhydrophobic and superoleophobic surfaces from fluorinated-decyl polyhedral oligomeric silsesquioxane and hydrolyzed fluorinated alkyl silane. Angew. Chem. Int. Ed. 2011, 50, 11433–11436. [Google Scholar] [CrossRef] [PubMed]
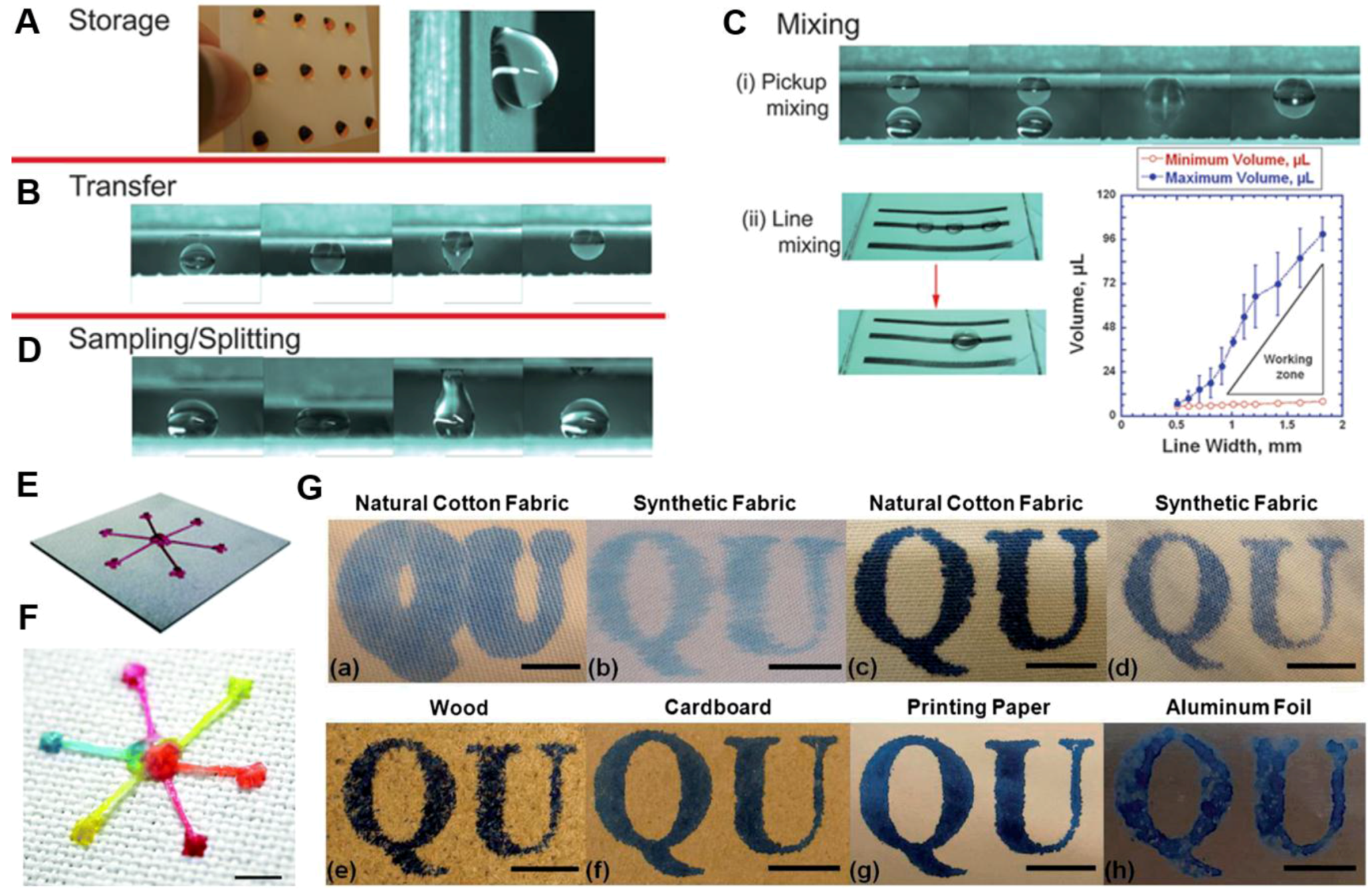
- Nilghaz, A.; Wicaksono, D.H.B.; Gustiono, D.; Majid, F.A.A.; Supriyanto, E.; Kadir, M.R.A. Flexible microfluidic cloth-based analytical devices using a low-cost wax patterning technique. Lab Chip 2012, 12, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.K.; Pan, F.; Xu, C.; Fuchs, H.; Chi, L.F. In situ surface-modification-induced superhydrophobic patterns with reversible wettability and adhesion. Adv. Mater. 2013, 25, 1682–1686. [Google Scholar] [CrossRef] [PubMed]
- Shin, B.S.; Lee, K.R.; Moon, M.W.; Kim, H.Y. Extreme water repellency of nanostructured low-surface-energy non-woven fabrics. Soft Matter 2012, 8, 1817–1823. [Google Scholar] [CrossRef]
- Rana, M.; Hao, B.; Mu, L.; Chen, L.; Ma, P.C. Development of multi-functional cotton fabrics with Ag/AgBr-TiO2 nanocomposite coating. Compos. Sci. Technol. 2016, 122, 104–112. [Google Scholar] [CrossRef]
- Xue, C.H.; Guo, X.J.; Zhang, M.M.; Ma, J.Z.; Jia, S.T. Fabrication of robust superhydrophobic surfaces by modification of chemically roughened fibers via thiol-ene click chemistry. J. Mater. Chem. A 2015, 3, 21797–21804. [Google Scholar] [CrossRef]
- Lai, Y.K.; Lin, L.X.; Pan, F.; Huang, J.Y.; Song, R.; Huang, Y.X.; Lin, C.J.; Fuchs, H.; Chi, L.F. Bioinspired patterning with extreme wettability contrast on TiO2 nanotube array surface: A versatile platform for biomedical applications. Small 2013, 9, 2945–2953. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Q.; Lai, Y.K.; Huang, J.Y.; Tang, Y.X.; Yang, L.; Chen, Z.; Zhang, K.Q.; Wang, X.C.; Tang, L.P. Multifunctional wettability patterns prepared by laser processing on superhydrophobic TiO2 nanostructured surfaces. J. Mater. Chem. B 2015, 3, 342–347. [Google Scholar] [CrossRef]
- Lai, Y.K.; Tang, Y.X.; Huang, J.Y.; Pan, F.; Chen, Z.; Zhang, K.Q.; Fuchs, H.; Chi, L.F. Bioinspired TiO2 nanostructure films with special wettability and adhesion for droplets manipulation and patterning. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Balu, B.; Berry, A.D.; Hess, D.W.; Breedveld, V. Patterning of superhydrophobic paper to control the mobility of micro-liter drops for two-dimensional lab-on-paper applications. Lab Chip. 2009, 9, 3066–3075. [Google Scholar] [CrossRef] [PubMed]
- Rabnawaz, M.; Wang, Z.J.; Wang, Y.; Wyman, L.; Hu, H.; Liu, G.J. Synthesis of poly(dimethylsiloxane)-block-poly[3-(triisopropyloxysilyl) propyl methacrylate] and its use in the facile coating of hydrophilically patterned superhydrophobic fabrics. RSC Adv. 2015, 5, 39505–39511. [Google Scholar] [CrossRef]
- Xing, S.Y.; Jiang, J.; Pan, T.R. Interfacial microfluidic transport on micropatterned superhydrophobic textile. Lab Chip 2013, 13, 1937–1947. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Y.; Lai, Y.K.; Pan, F.; Wang, H.; Zhang, K.Q.; Fuchs, H.; Chi, L.F. Multifunctional superamphiphobic TiO2 nanostructure surfaces with facile wettability and adhesion engineering. Small 2014, 10, 4865–4873. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.Y.; Hu, H.; Liu, G.J.; Rabnawaz, M. Hydrophilically patterned superhydrophobic cotton fabrics and their use in ink printing. J. Mater. Chem. A 2014, 2, 8094–8102. [Google Scholar] [CrossRef]
- Lai, Y.; Huang, J.; Gong, J.; Huang, Y.; Wang, C.; Chen, Z.; Lin, C. Superhydrophilic-superhydrophobic template: A simple approach to micro- and nanostructure patterning of TiO2 films. J. Electrochem. Soc. 2009, 156, D480–D484. [Google Scholar] [CrossRef]
- Lai, Y.K.; Lin, C.J.; Wang, H.; Huang, J.Y.; Zhuang, H.F.; Sun, L. Superhydrophilic-superhydrophobic micropattern on TiO2 nanotube films by photocatalytic lithography. Electrochem. Commun. 2008, 10, 387–391. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Jiang, Z.L.; Huang, J.Y.; Lim, L.Y.; Li, W.L.; Deng, J.Y.; Gong, D.G.; Tang, Y.X.; Lai, Y.K.; Chen, Z. Titanate and titania nanostructured materials for environmental and energy applications: A review. RSC Adv. 2015, 5, 79479–79510. [Google Scholar] [CrossRef]
- Su, B.; Tian, Y.; Jiang, L. Bioinspired interfaces with superwettability: From materials to chemistry. J. Am. Chem. Soc. 2016, 138. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Mammen, L.; Butt, H.J.; Vollmer, D. Candle soot as a template for a transparent robust superamphiphobic coating. Science 2012, 335, 67–70. [Google Scholar] [CrossRef] [PubMed]
- Tuteja, A.; Choi, W.; Ma, M.L.; Mabry, J.M.; Mazzella, S.A.; Rutledge, G.C.; McKinley, G.H.; Cohen, R.E. Designing superoleophobic surfaces. Science 2007, 318, 1618–1622. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Sathasivam, S.; Song, J.L.; Crick, C.R.; Carmalt, C.J.; Parkin, I.P. Robust self-cleaning surfaces that function when exposed to either air or oil. Science 2015, 347, 1132–1135. [Google Scholar] [CrossRef] [PubMed]


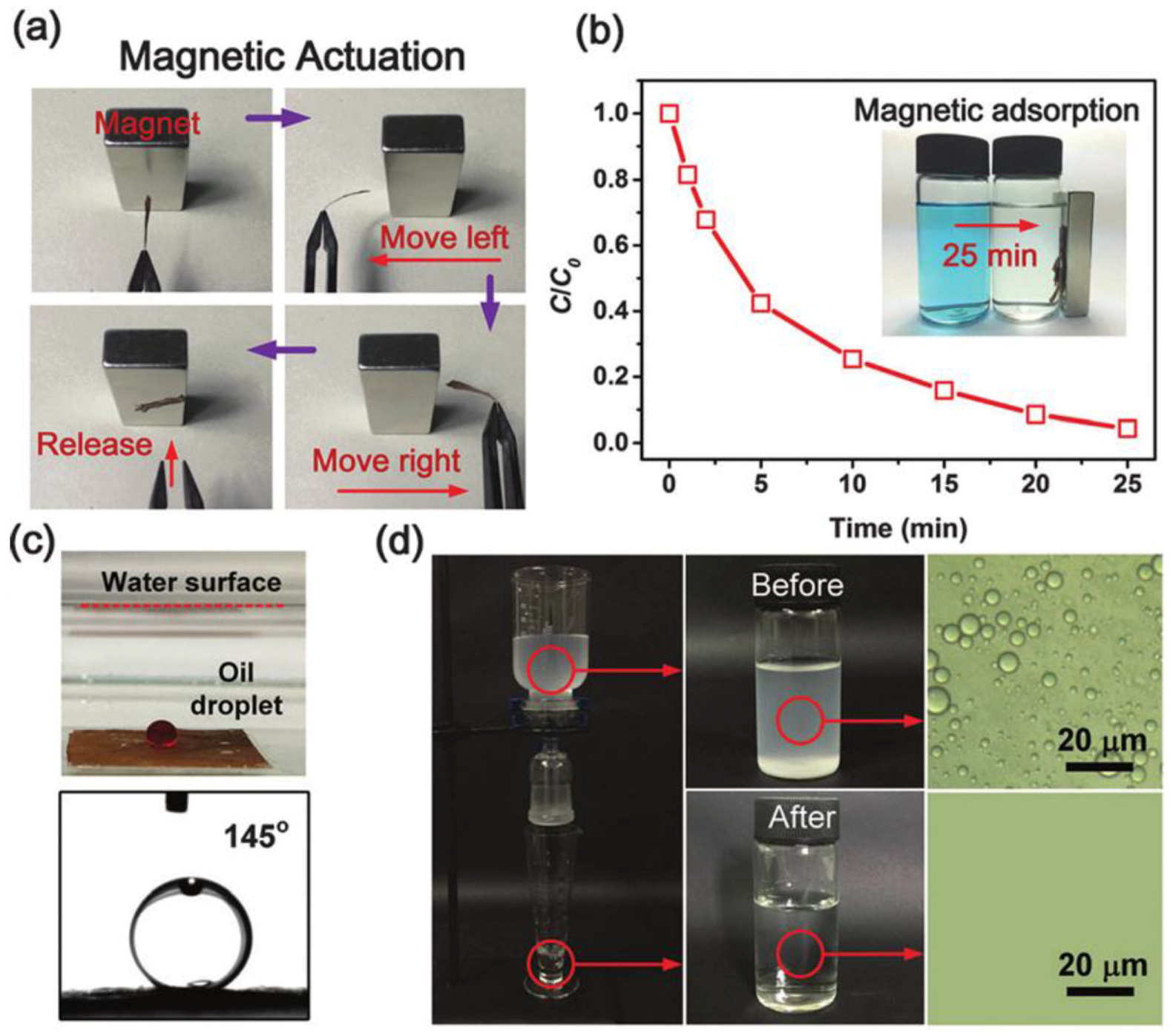

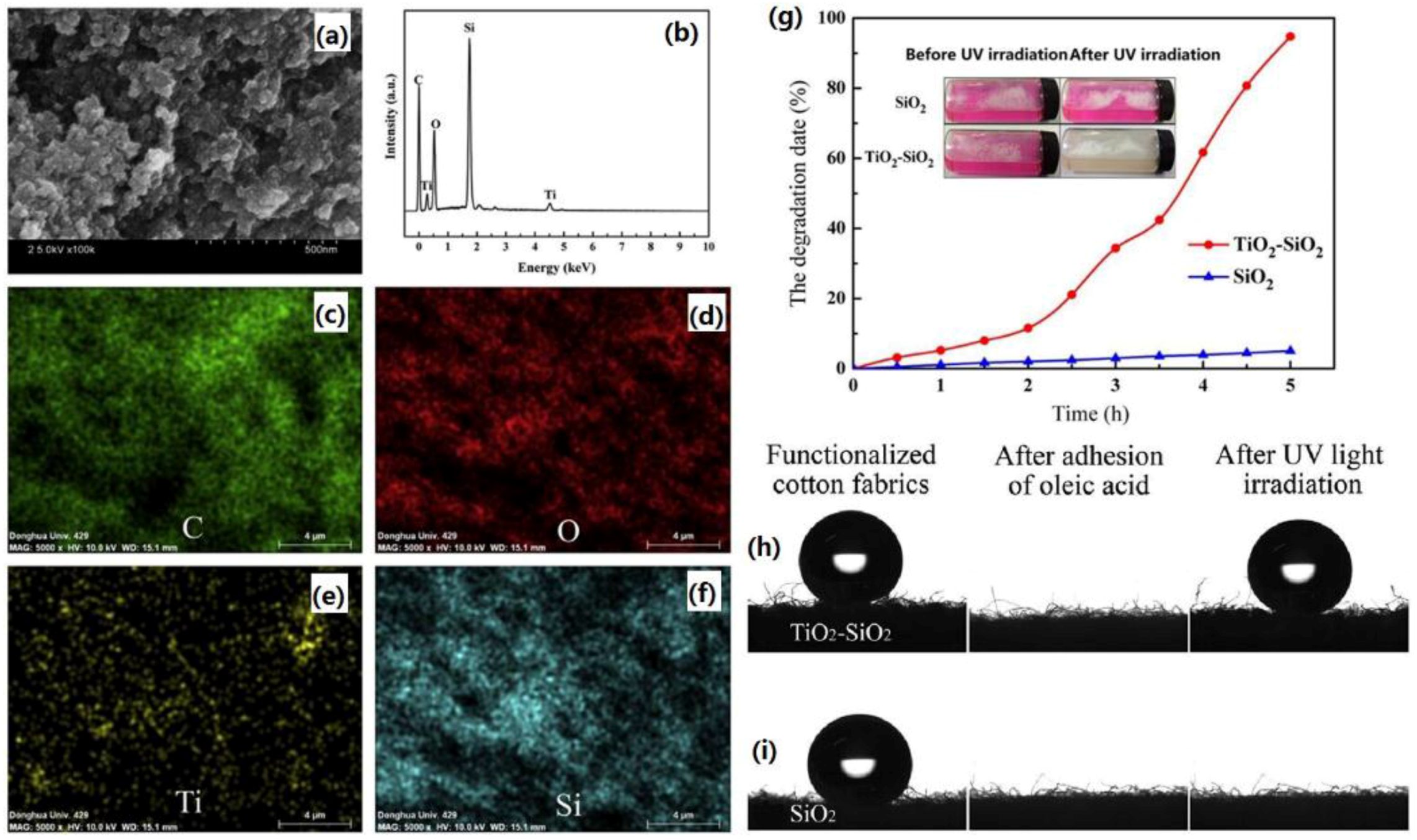
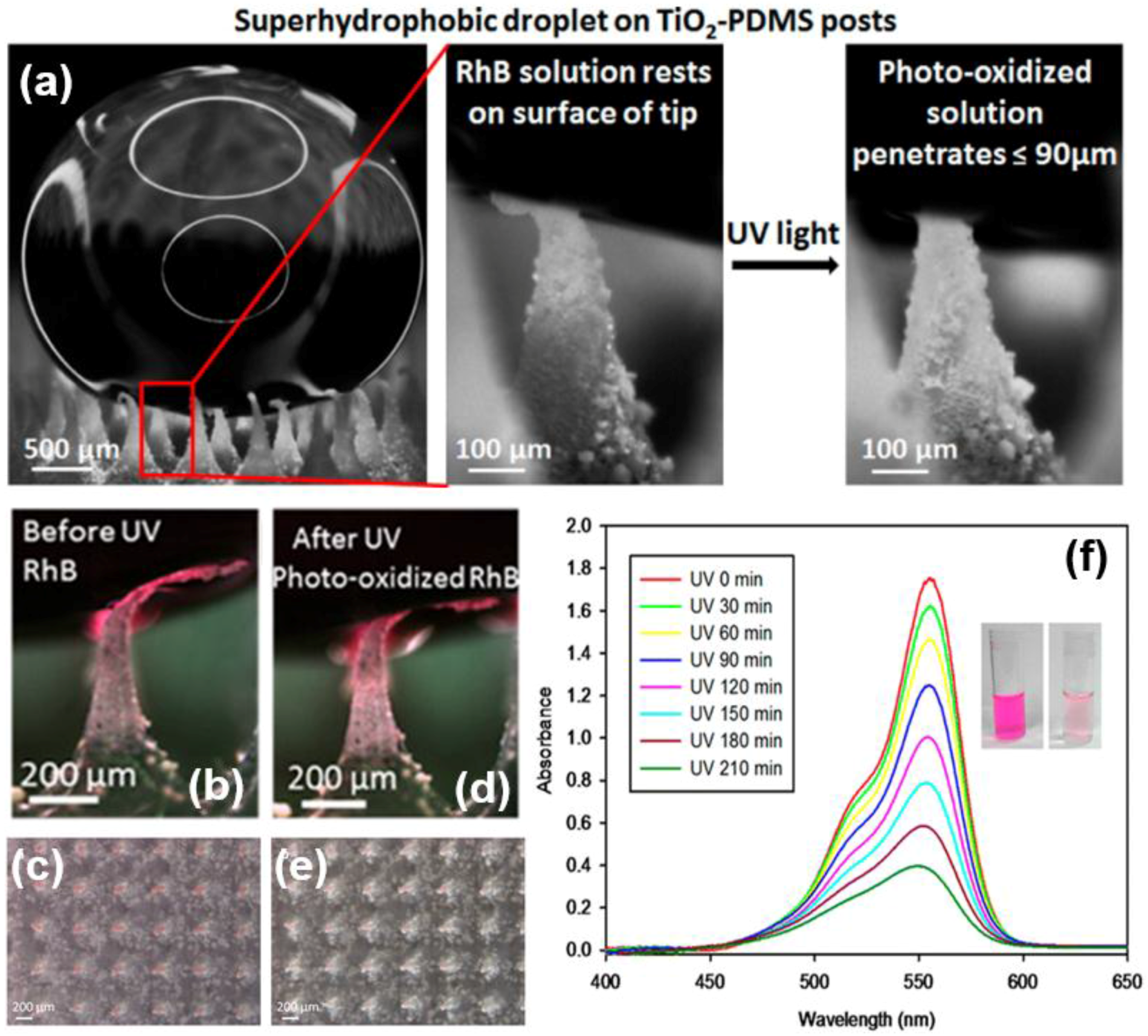
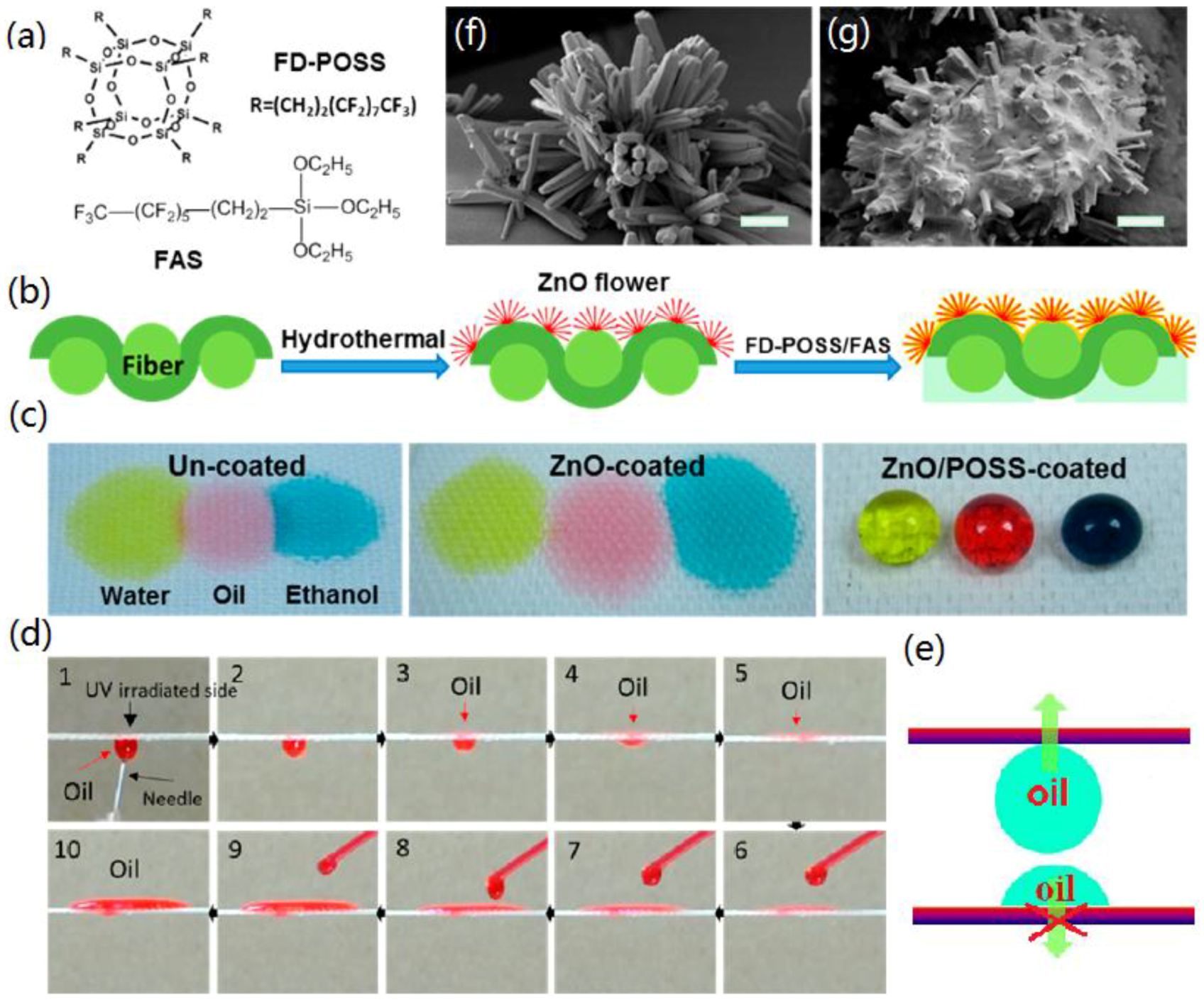

| Methods | Roughness Formation | Time-Scale and Requirement a | Properties | Ref. |
|---|---|---|---|---|
| Dip-coating | Nanoparticle coating | Slow | Mechanical and environmental stability | [51,52,53,54,55,56,57,58,59,60,61,62,63] |
| Wet chemical etching | Growth of nano-structures by etching | Rapid/slow | Excellent resistance to washing, abrasion | [64,65,66,67] |
| Chemical bath disposition | Nanoparticle film deposition | Slow and temperature requirement | Moderate durability | [68,69,70,71,72,73,74,75] |
| Electrophoretic deposition | Nanoparticle coating | Rapid and conductive substrate requirement | Chemical stability, highly transparent | [76,77,78,79,80] |
| Electrospinning | Nanofibers by electrospinning | Slow and solvent requirement | Porous membrane | [81,82,83,84,85,86,87] |
| Spray-coating methods | Micro/nanostructures by spraying | Rapid and scalable under ambient conditions | Moderate stability, easy reparability | [88,89,90,91,92,93,94,95] |
| Chemical vapor deposition | Growth of nano structures by polymerization | Slow and need heating | Separation of oils or organic contaminates from water | [96,97,98,99,100,101] |
| Plasma etching process | Growth of nanostructures by etching | Moderate and require specific equipment | Self-cleaning | [102,103,104,105,106,107,108,109,110] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Gao, S.-W.; Cai, J.-S.; He, C.-L.; Mao, J.-J.; Zhu, T.-X.; Chen, Z.; Huang, J.-Y.; Meng, K.; Zhang, K.-Q.; et al. Recent Progress in Fabrication and Applications of Superhydrophobic Coating on Cellulose-Based Substrates. Materials 2016, 9, 124. https://doi.org/10.3390/ma9030124
Liu H, Gao S-W, Cai J-S, He C-L, Mao J-J, Zhu T-X, Chen Z, Huang J-Y, Meng K, Zhang K-Q, et al. Recent Progress in Fabrication and Applications of Superhydrophobic Coating on Cellulose-Based Substrates. Materials. 2016; 9(3):124. https://doi.org/10.3390/ma9030124
Chicago/Turabian StyleLiu, Hui, Shou-Wei Gao, Jing-Sheng Cai, Cheng-Lin He, Jia-Jun Mao, Tian-Xue Zhu, Zhong Chen, Jian-Ying Huang, Kai Meng, Ke-Qin Zhang, and et al. 2016. "Recent Progress in Fabrication and Applications of Superhydrophobic Coating on Cellulose-Based Substrates" Materials 9, no. 3: 124. https://doi.org/10.3390/ma9030124
APA StyleLiu, H., Gao, S.-W., Cai, J.-S., He, C.-L., Mao, J.-J., Zhu, T.-X., Chen, Z., Huang, J.-Y., Meng, K., Zhang, K.-Q., Al-Deyab, S. S., & Lai, Y.-K. (2016). Recent Progress in Fabrication and Applications of Superhydrophobic Coating on Cellulose-Based Substrates. Materials, 9(3), 124. https://doi.org/10.3390/ma9030124