Lignin as a Binder Material for Eco-Friendly Li-Ion Batteries
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Pretreatment of Lignin
2.3. Preparation of LiFePO4 Positive and Graphite Negative Electrodes
2.4. Characterization
2.4.1. Analysis of the Lignin and Morphology of the Electrodes
2.4.2. Electrochemical Evaluation
2.5. Geometry
3. Results and Discussion
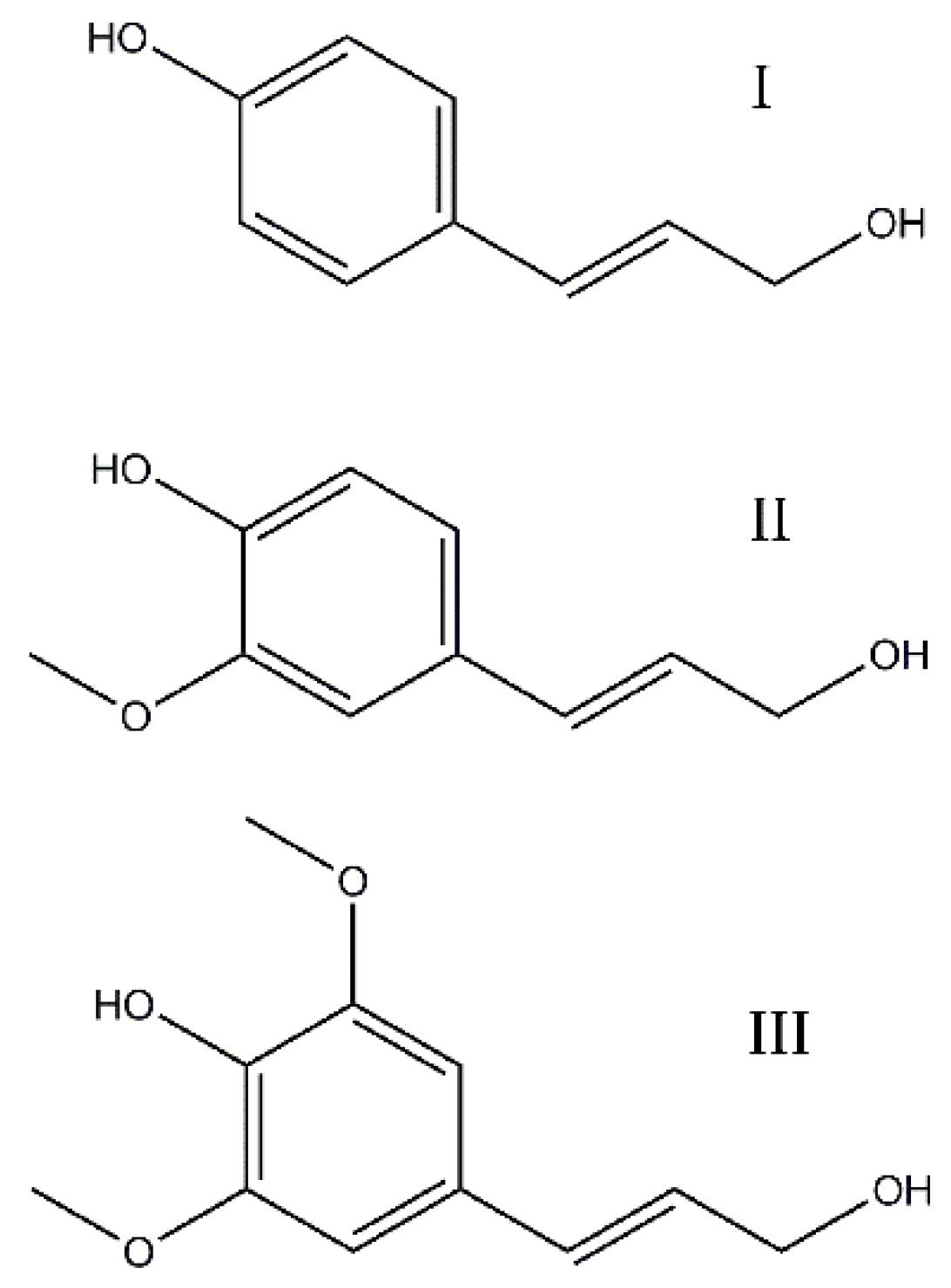
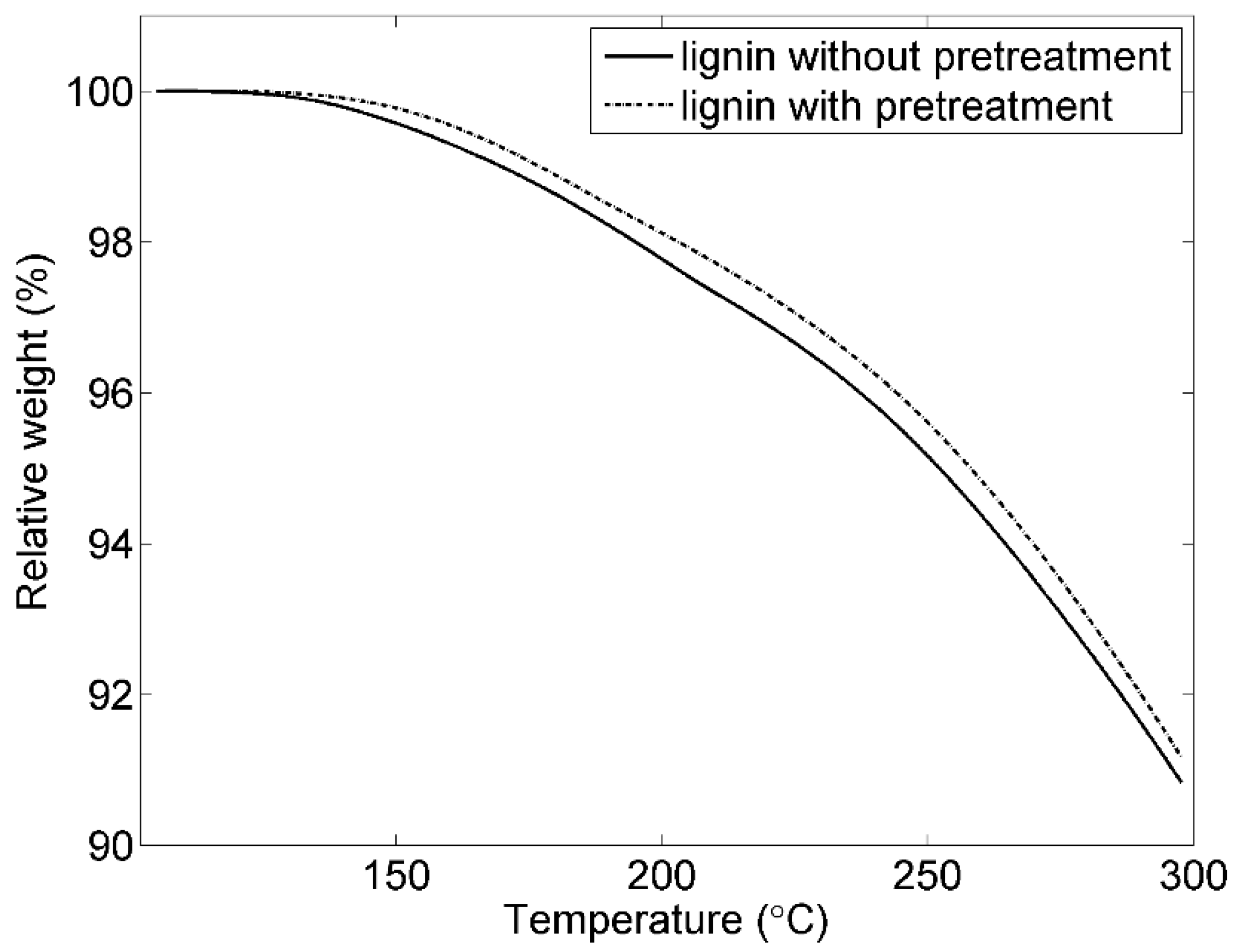
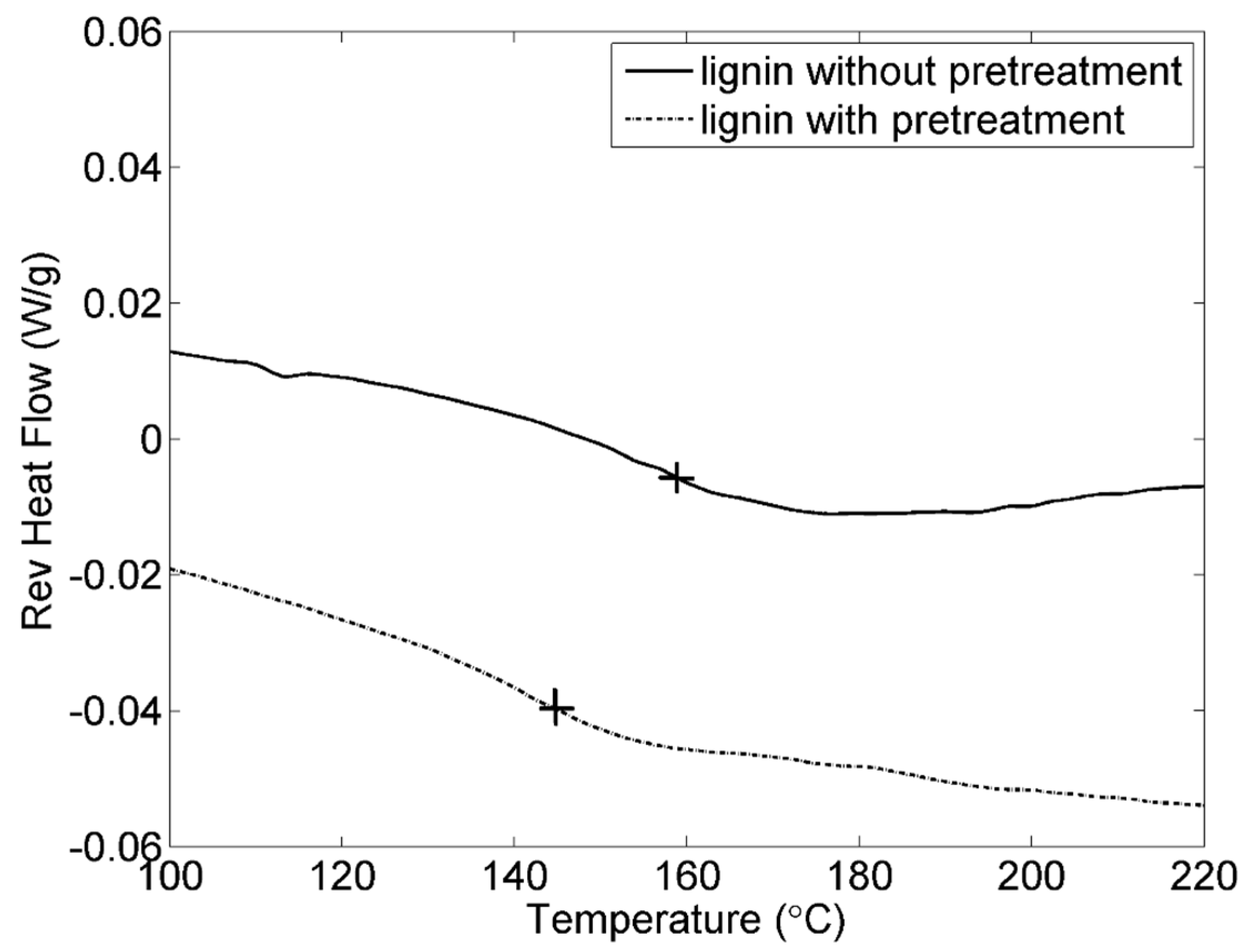
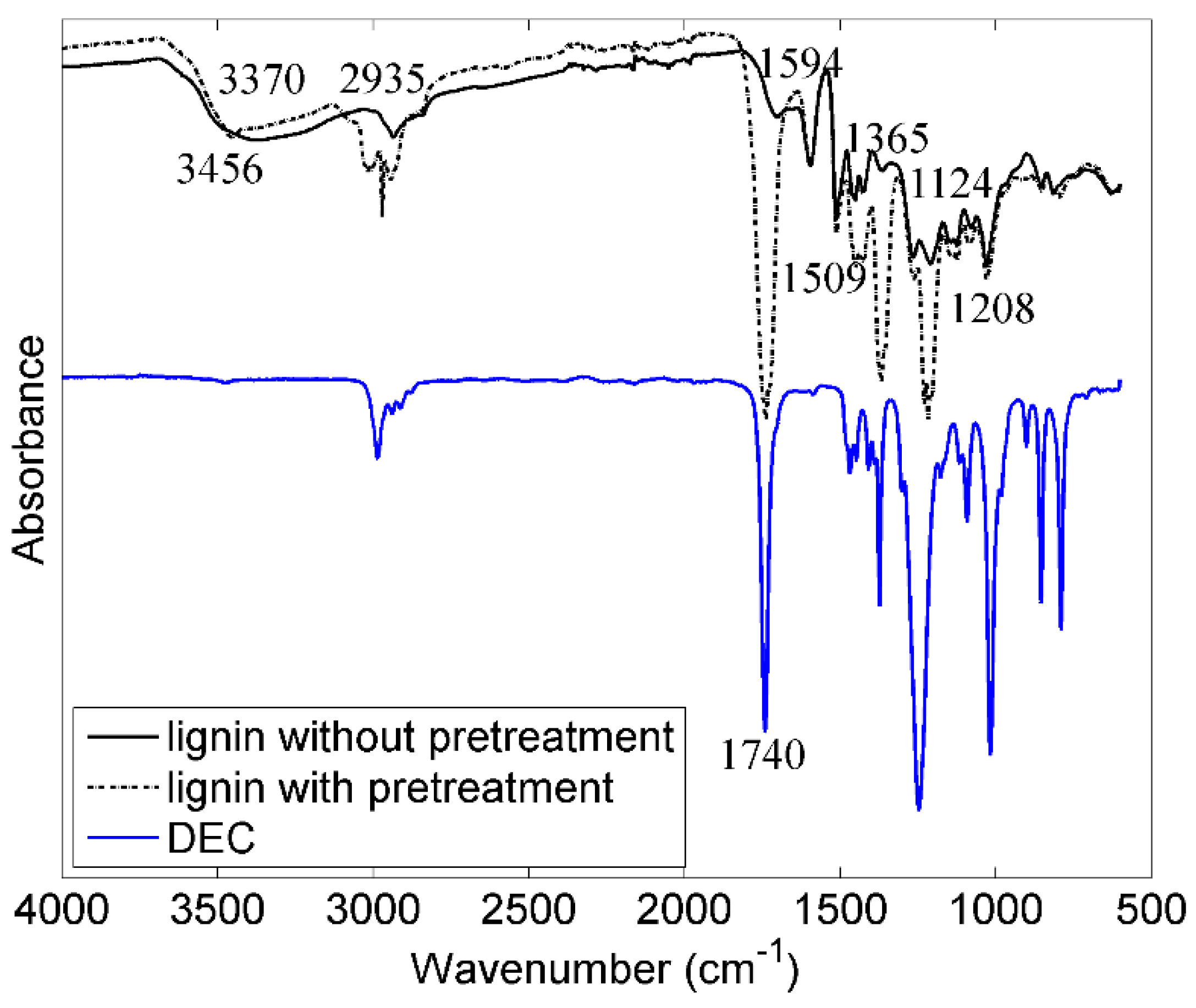
3.1. Pretreatment of Lignin
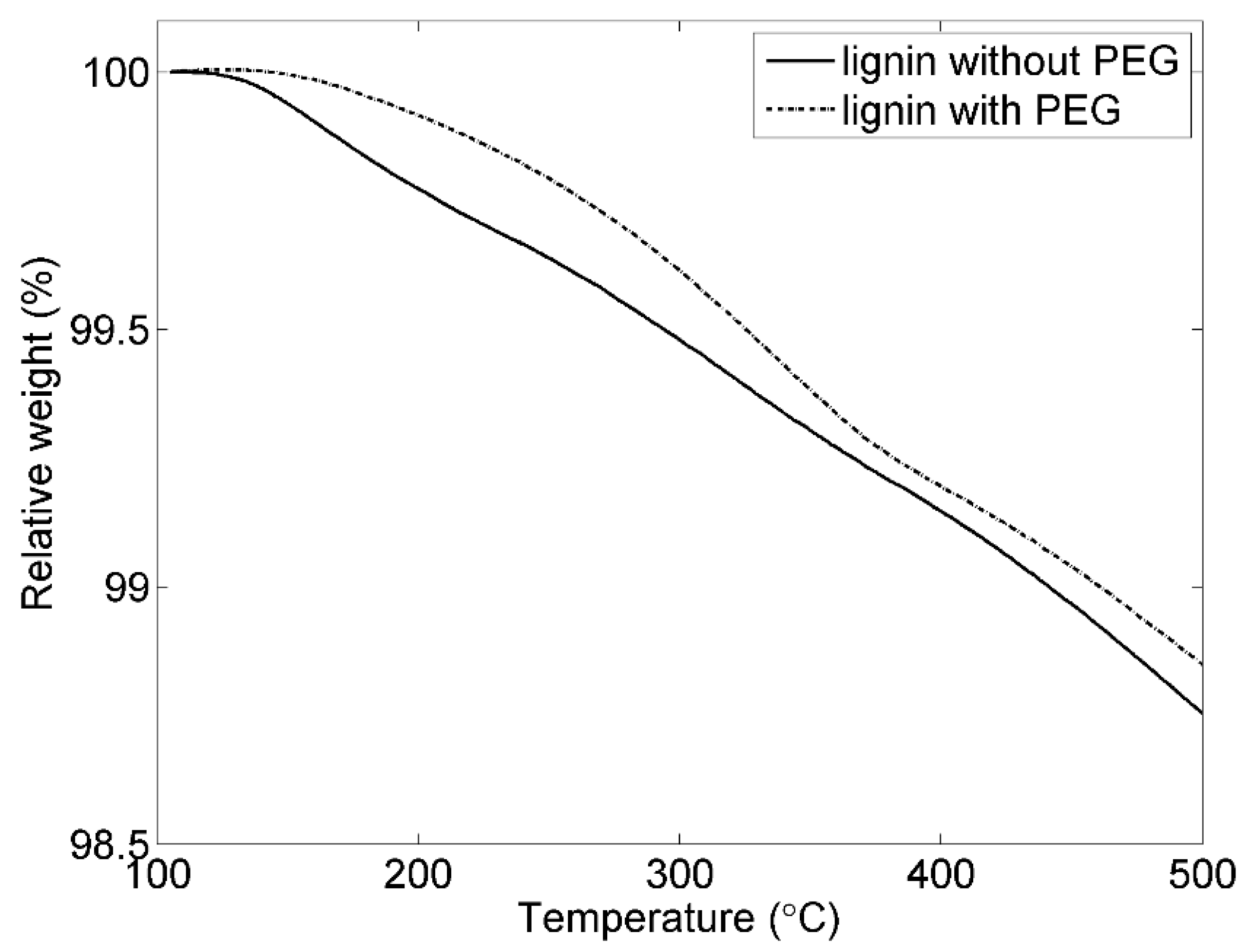
3.2. The Effect of PEG for the Thermal Behaviours of the Electrodes
3.3. Morphology of the Electrodes
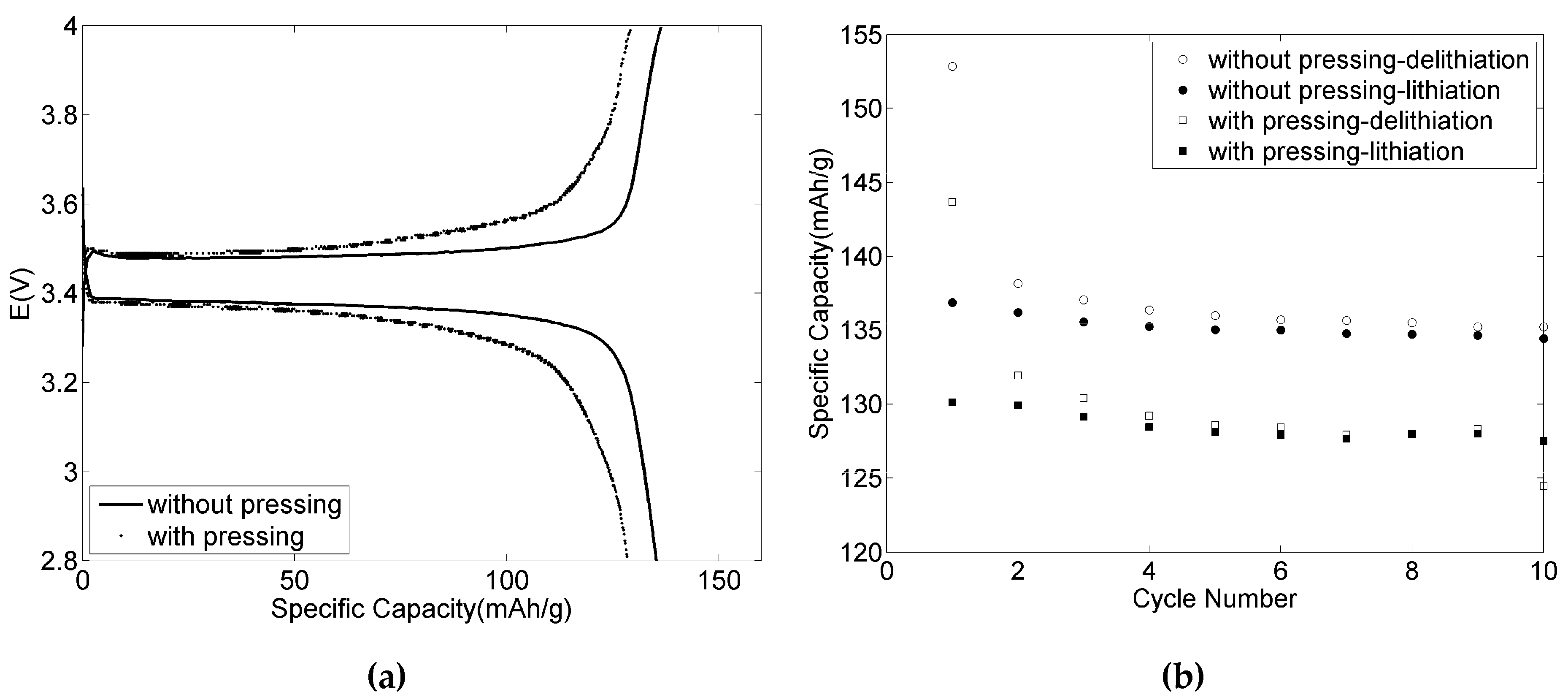
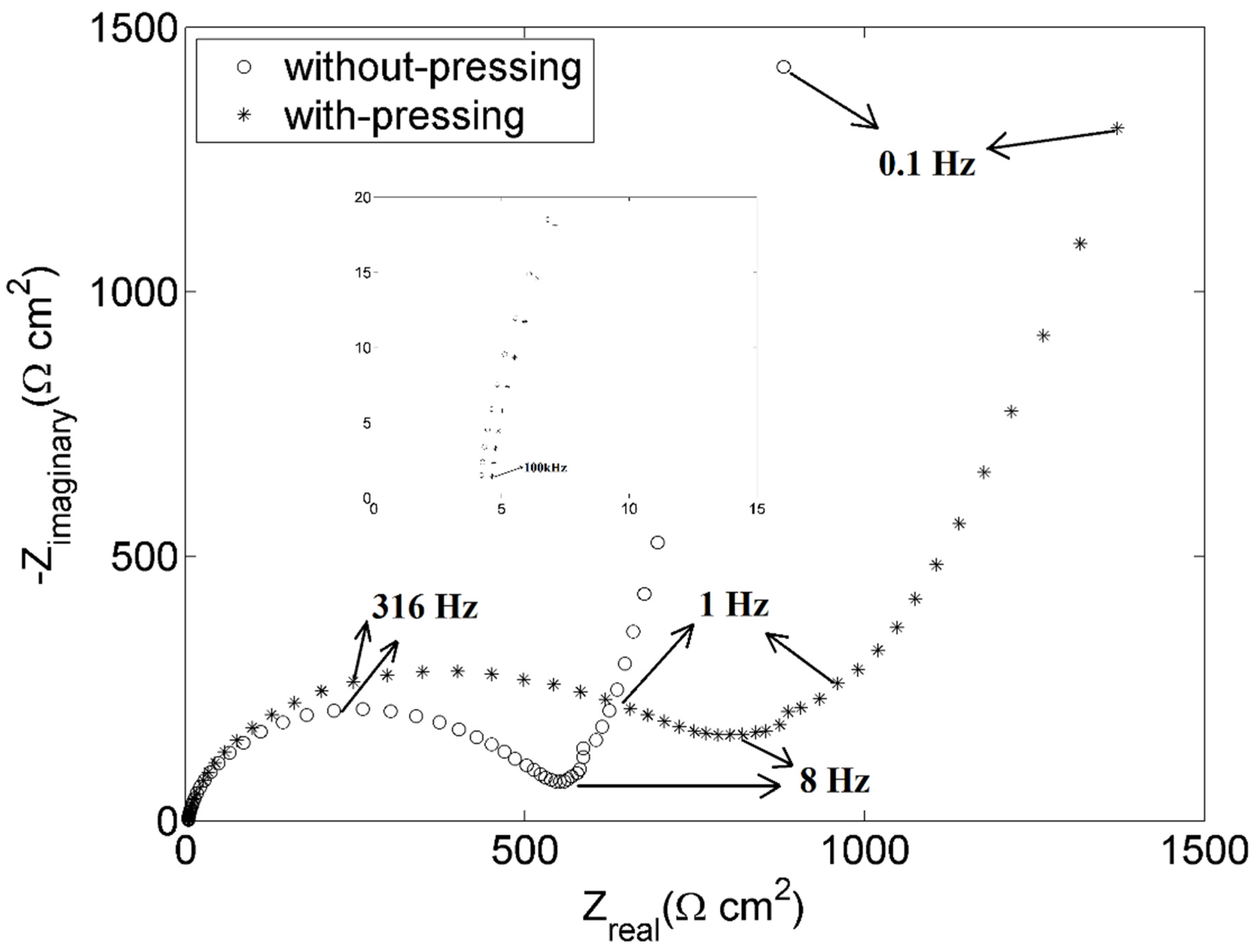
3.4. The Effect of Pressing for the Positive Electrodes
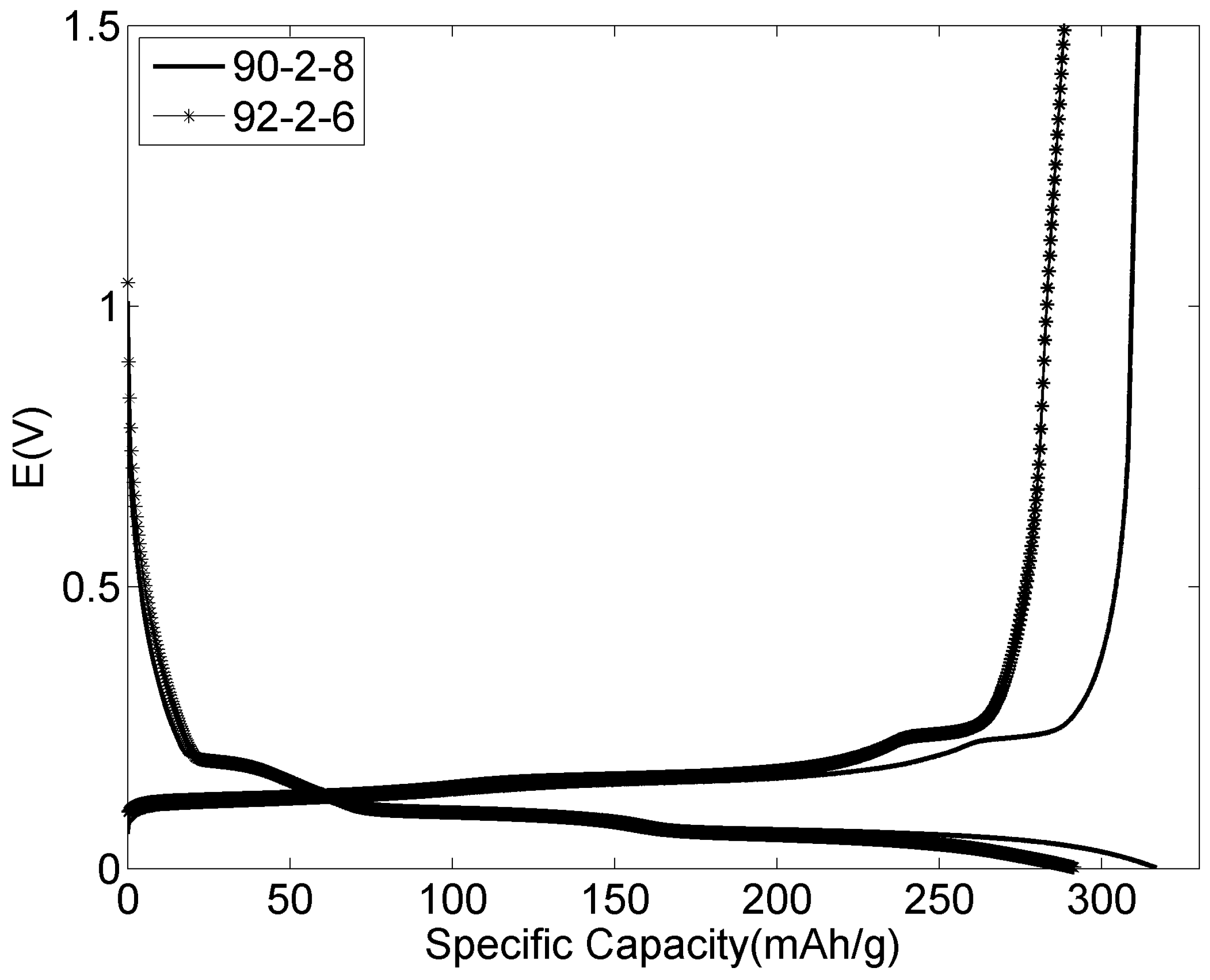
3.5. Variation of Component Ratios for the Positive Electrodes
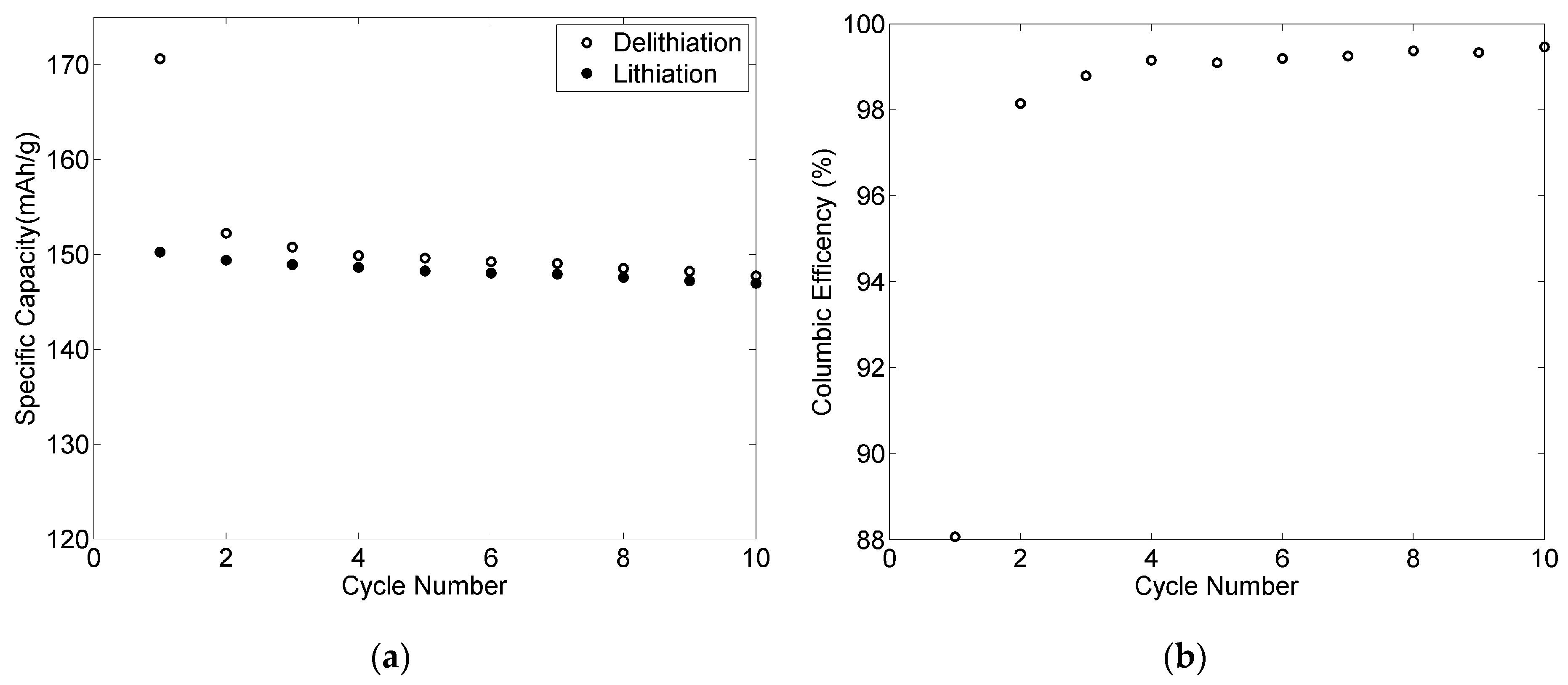
3.6. Electrochemical Performance for the 80-11-9 Positive Electrode
3.7. Voltage Profile of the Graphite Negative Electrodes
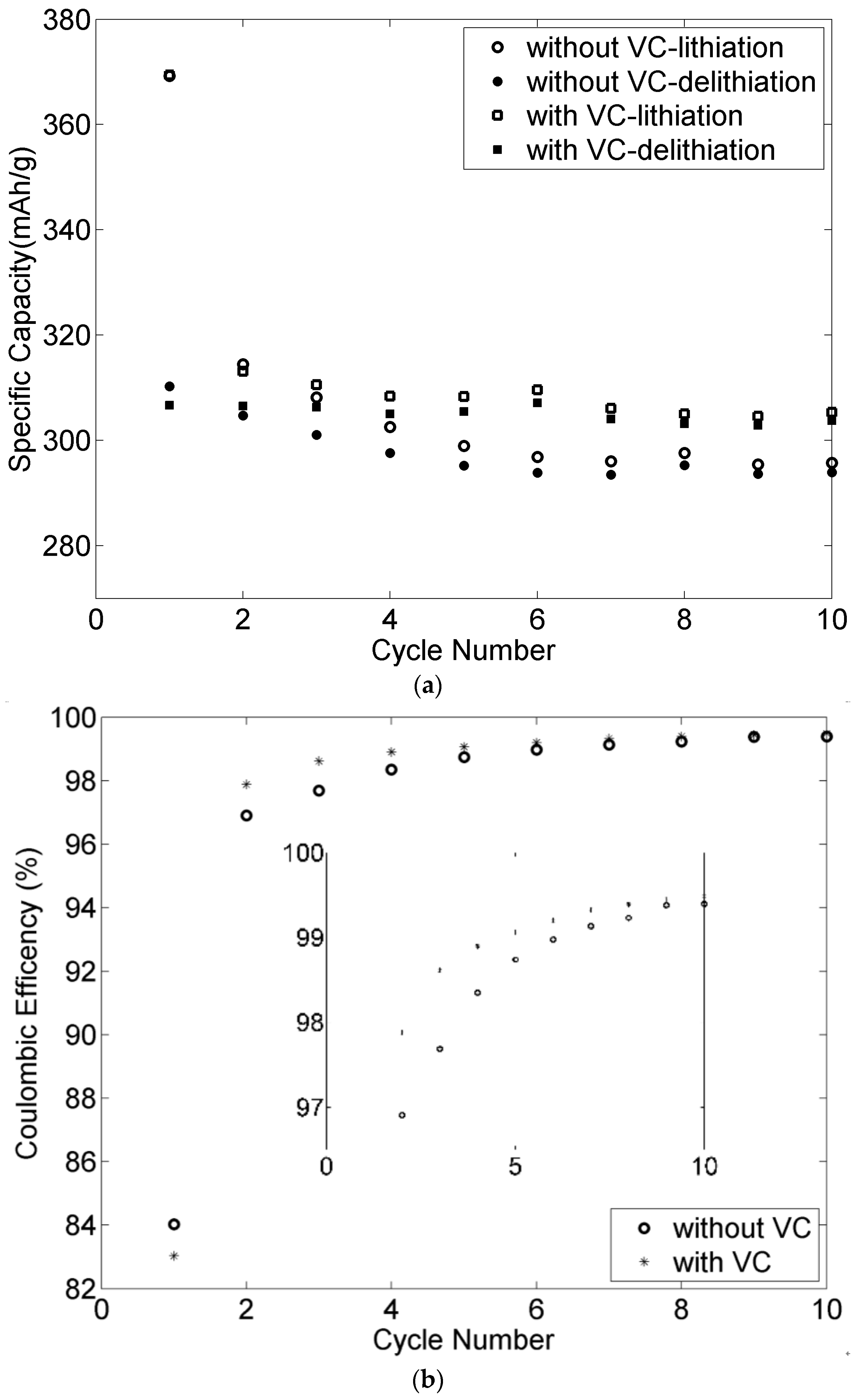
3.8. Effect of VC for the Graphite Negative Electrodes
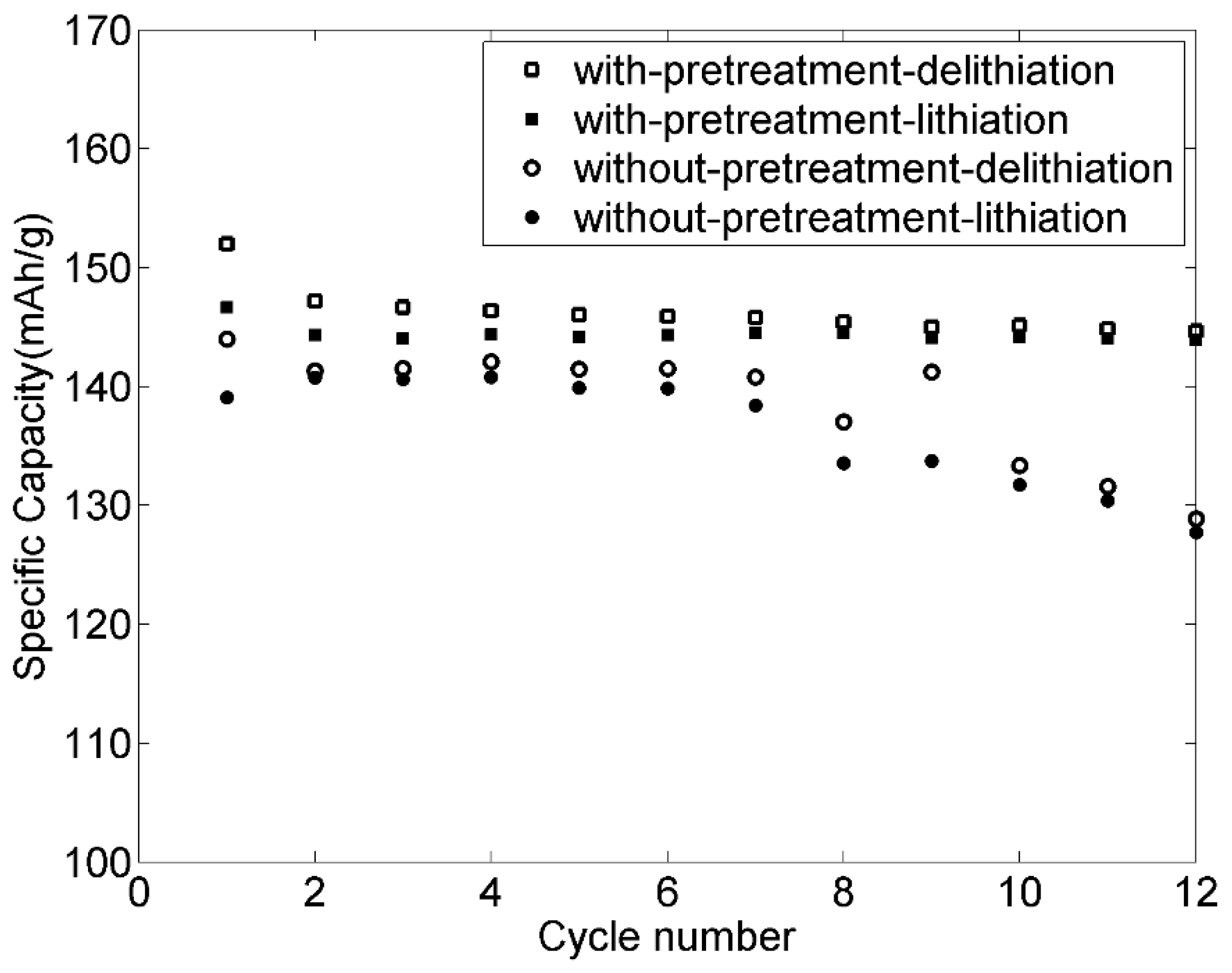
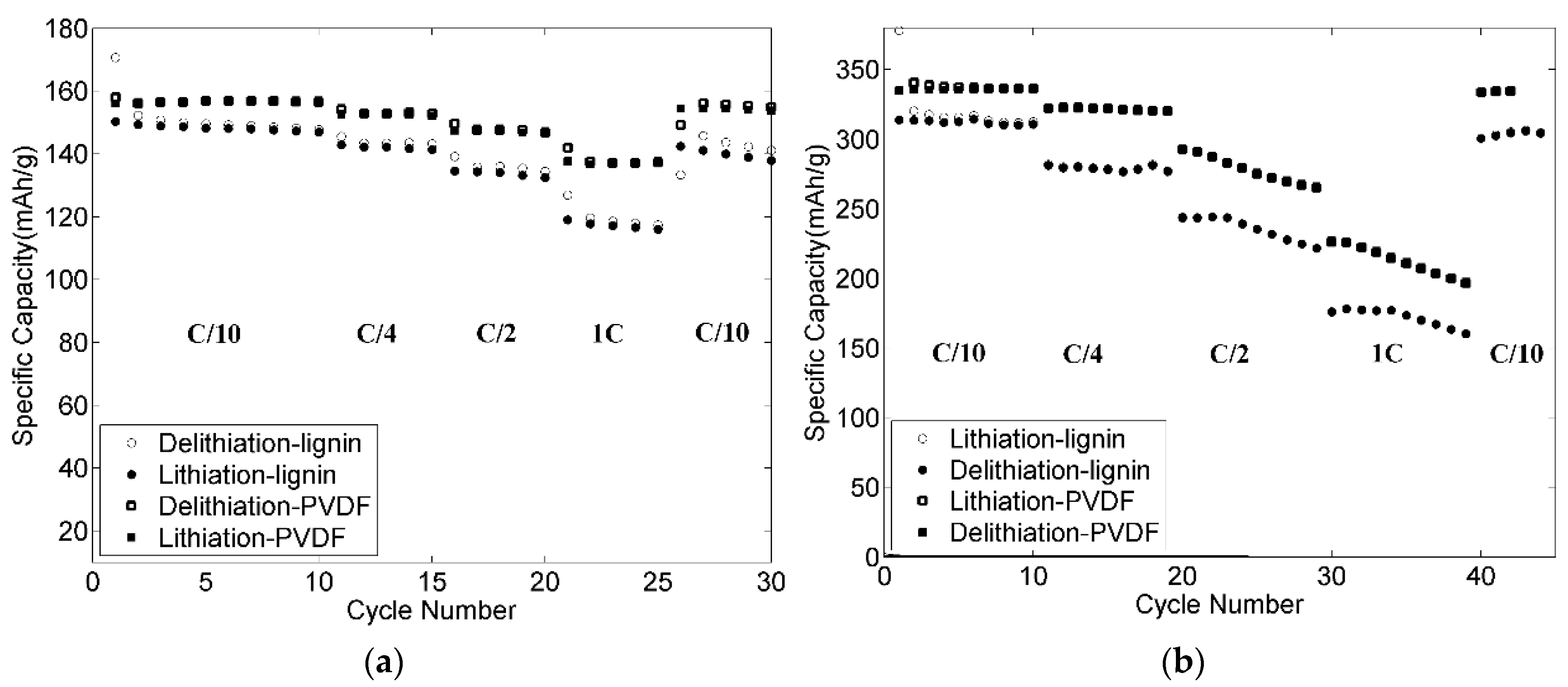
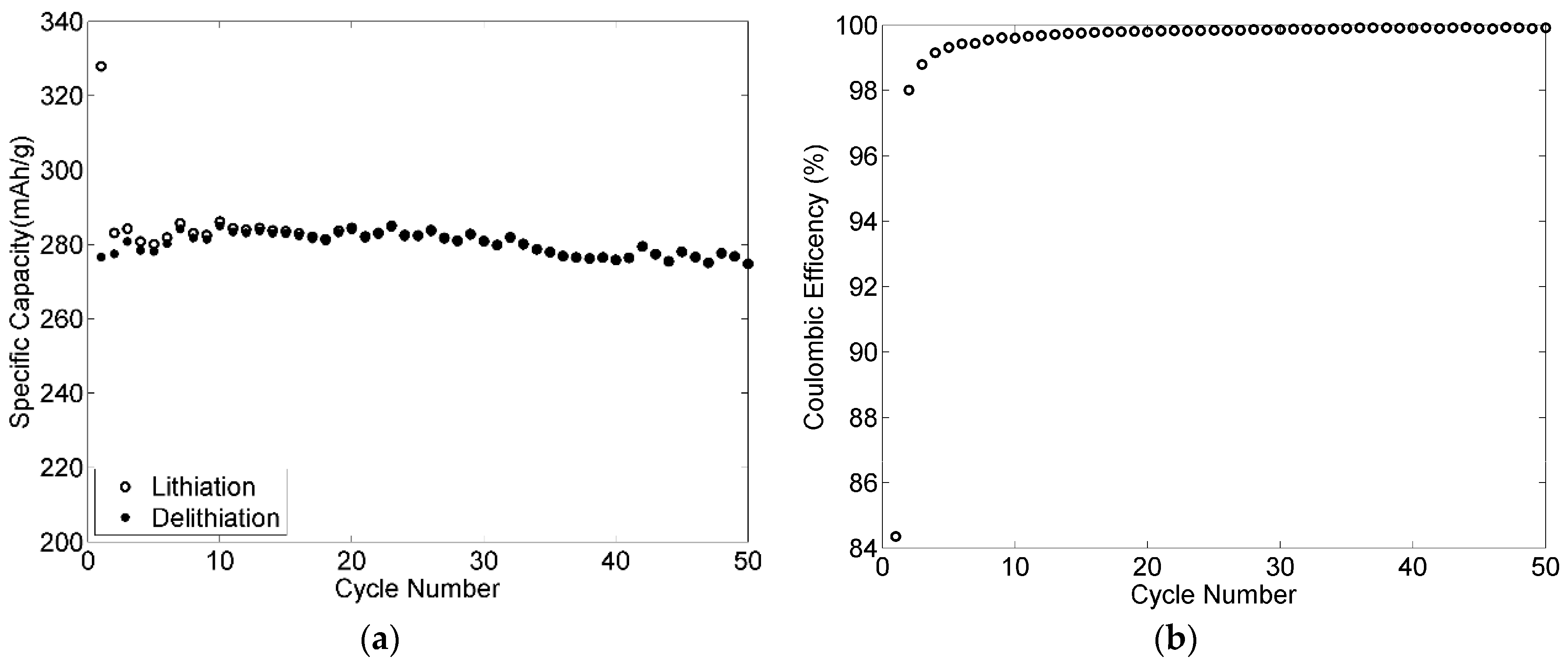
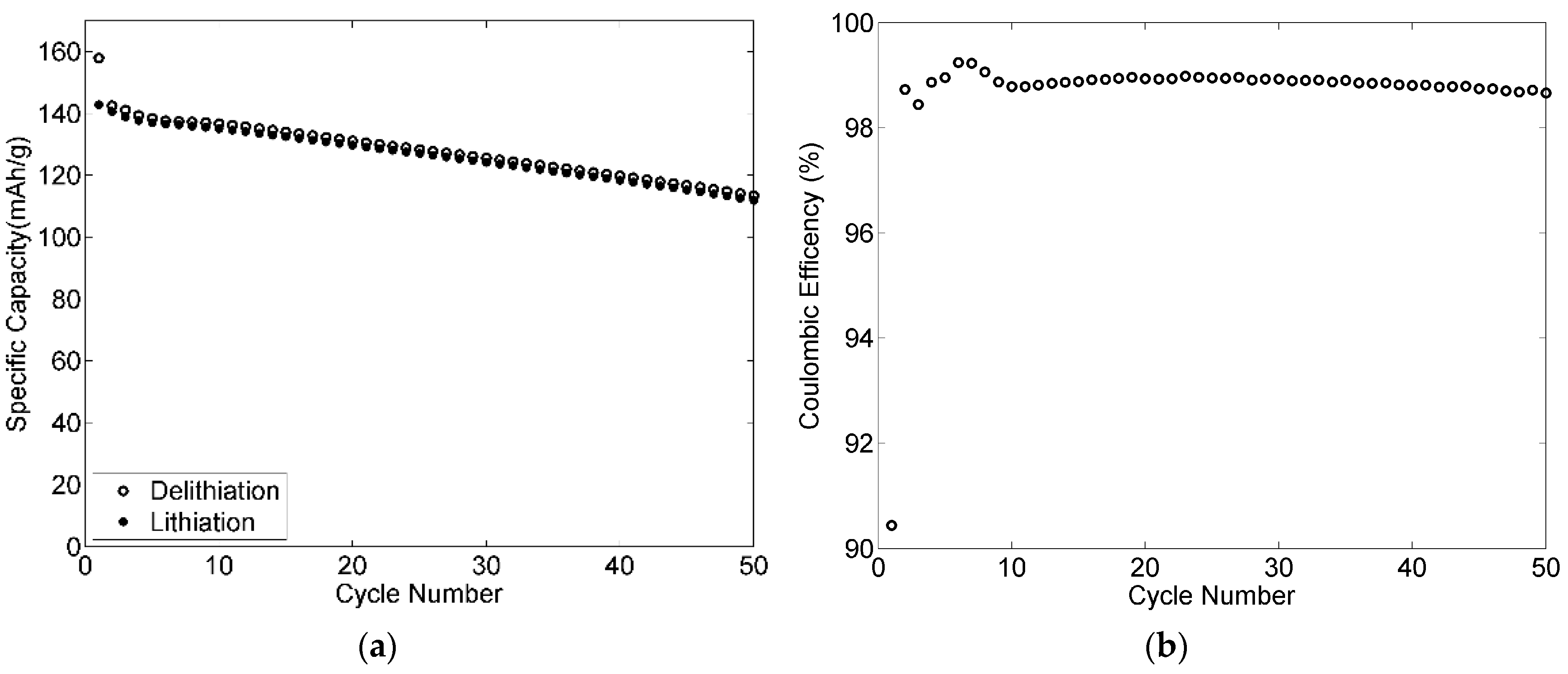
3.9. Rate Capabilities and Cycling Stability of the Electrodes
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Scrosati, B.; Garche, J. Lithium batteries: Status, prospects and future. J. Power Sour. 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Tarascon, J.-M.; Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 2001, 414, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Manickam, M.; Takata, M. Effect of cathode binder on capacity retention and cycle life in transition metal phosphate of a rechargeable lithium battery. Electrochim. Acta 2003, 48, 957–963. [Google Scholar] [CrossRef]
- Chen, Z.; Christensen, L.; Dahn, J.R. Comparison of PVDF and PVDF-TFE-P as binders for electrode materials showing large volume changes in lithium-ion batteries. J. Electrochem. Soc. 2003, 150, A1073–A1078. [Google Scholar] [CrossRef]
- Du Pasquier, A.; Disma, F.; Bowmer, T.; Gozdz, A.; Amatucci, G.; Tarascon, J.M. Differential scanning calorimetry study of the reactivity of carbon anodes in plastic Li-ion batteries. J. Electrochem. Soc. 1998, 145, 472–477. [Google Scholar] [CrossRef]
- Maleki, H.; Deng, G.; Kerzhner-Haller, I.; Anani, A.; Howard, J.N. Thermal stability studies of binder materials in anodes for lithium-ion batteries. J. Electrochem. Soc. 2000, 147, 4470–4475. [Google Scholar] [CrossRef]
- Zhang, S.S.; Xu, K.; Jow, T.R. Evaluation on a water-based binder for the graphite anode of Li-ion batteries. J. Power Sour. 2004, 138, 226–231. [Google Scholar] [CrossRef]
- Guerfi, A.; Kaneko, M.; Petitclerc, M.; Mori, M.; Zaghib, K. LiFePO4 water-soluble binder electrode for Li-ion batteries. J. Power Sour. 2007, 163, 1047–1052. [Google Scholar] [CrossRef]
- Drofenik, J.; Gaberscek, M.; Dominko, R.; Poulsen, F.W.; Mogensen, M.; Pejovnik, S.; Jamnik, J. Cellulose as a binding material in graphitic anodes for Li ion batteries: A performance and degradation study. Electrochim. Acta 2003, 48, 883–889. [Google Scholar] [CrossRef]
- Buqa, H.; Holzapfel, M.; Krumeich, F.; Veit, C.; Novak, P. Study of styrene butadiene rubber and sodium methyl cellulose as binder for negative electrodes in lithium-ion batteries. J. Power Sour. 2006, 161, 617–622. [Google Scholar] [CrossRef]
- Liu, W.R.; Yang, M.H.; Wu, H.C.; Chiao, S.; Wu, N.L. Enhanced cycle life of Si anode for Li-ion batteries by using modified elastomeric binder. Electrochem. Solid State Lett. 2005, 8, A100–A103. [Google Scholar] [CrossRef]
- Chou, S.L.; Wang, J.Z.; Liu, H.K.; Dou, S.X. Rapid synthesis of Li4Ti5O12 microspheres as anode materials and its binder effect for lithium-ion battery. J. Phys. Chem. C 2011, 115, 16220–16227. [Google Scholar] [CrossRef]
- Magasinski, A.; Zdyrko, B.; Kovalenko, I.; Hertzberg, B.; Burtovyy, R.; Huebner, C.F.; Fuller, T.F.; Luzinov, I.; Yushin, G. Toward efficient binders for Li-ion battery Si-based anodes: Polyacrylic acid. ACS Appl. Mater. Interfaces 2010, 2, 3004–3010. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Yu, G.H.; Pan, L.J.; Liu, N.; McDowell, M.T.; Bao, Z.N.; Cui, Y. Stable Li-ion battery anodes by in-situ polymerization of conducting hydrogel to conformally coat silicon nanoparticles. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Peng, L.; Ding, Y.; Zhao, Y.; Yu, G.H. Nanostructured conductive polymers for advanced energy storage. Chem. Soc. Rev. 2015, 44, 6684–6696. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Peng, L.; Yu, G.H. Nanostructured conducting polymer hydrogels for energy storage applications. Nanoscale 2015, 7, 12796–12806. [Google Scholar] [CrossRef] [PubMed]
- Tomani, P. The lignoboost process. Cell. Chem. Technol. 2010, 44, 53–58. [Google Scholar]
- Fengel, D.; Lignin, G.W. Wood: Chemistry, Ultrastructure, Reactions; Walter de Gruyter & Co.: Berlin, Germany, 1983; pp. 132–174. [Google Scholar]
- Grabber, J.H.; Ralph, J.; Hatfield, R.D.; Quideau, S. p-Hydroxyphenyl, guaiacyl, and syringyl lignins have similar inhibitory effects on wall degradability. J. Agric. Food Chem. 1997, 45, 2530–2532. [Google Scholar] [CrossRef]
- Lumadue, M.R.; Cannon, F.S.; Brown, N.R. Lignin as both fuel and fusing binder in briquetted anthracite fines for foundry coke substitute. Fuel 2012, 97, 869–875. [Google Scholar] [CrossRef]
- Pan, T. A first-principles based chemophysical environment for studying lignins as an asphalt antioxidant. Constr. Build. Mater. 2012, 36, 654–664. [Google Scholar] [CrossRef]
- Gellerstedt, G.; Li, J.; Eide, I.; Kleinert, M.; Barth, T. Chemical structures present in biofuel obtained from lignin. Energy Fuel 2008, 22, 4240–4244. [Google Scholar] [CrossRef]
- Milczarek, G. Preparation and characterization of a lignin modified electrode. Electroanalysis 2007, 19, 1411–1414. [Google Scholar] [CrossRef]
- Milczarek, G.; Inganäs, O. Renewable cathode materials from biopolymer/conjugated polymer interpenetrating networks. Science 2012, 335, 1468–1471. [Google Scholar] [CrossRef] [PubMed]
- He, Z.W.; Yang, J.; Lü, Q.F.; Lin, Q. Effect of structure on the electrochemical performance of nitrogen- and oxygen-containing carbon micro/nanospheres prepared from lignin-based composites. ACS Sustain. Chem. Eng. 2013, 1, 334–340. [Google Scholar] [CrossRef]
- Tenhaeff, W.E.; Rios, O.; More, K.; McGuire, M.A. Highly robust lithium ion battery anodes from lignin: An abundant, renewable, and low-cost material. Adv. Funct. Mater. 2014, 24, 86–94. [Google Scholar] [CrossRef]
- Hergert, H.L.; Kurth, E.F. The infrared spectra of lignin and related compounds. I. Characteristic carbonyl and hydroxyl frequencies of some flavanones, flavones, chalcones and acetophenones. J. Am. Chem. Soc. 1953, 75, 1622–1625. [Google Scholar] [CrossRef]
- Hergert, H.L. Infrared spectra of lignin and related compounds. II. Conifer lignin and model compounds. J. Org. Chem. 1960, 25, 405–413. [Google Scholar] [CrossRef]
- Fransson, L.; Eriksson, T.; Edström, K.; Gustafsson, T.; Thomas, J.O. Influence of carbon black and binder on Li-ion batteries. J. Power Sour. 2001, 101, 1–9. [Google Scholar] [CrossRef]
- Chen, L.; Wang, K.; Xie, X.; Xie, J. Effect of vinylene carbonate (VC) as electrolyte additive on electrochemical performance of Si film anode for lithium ion batteries. J. Power Sour. 2007, 174, 538–543. [Google Scholar] [CrossRef]
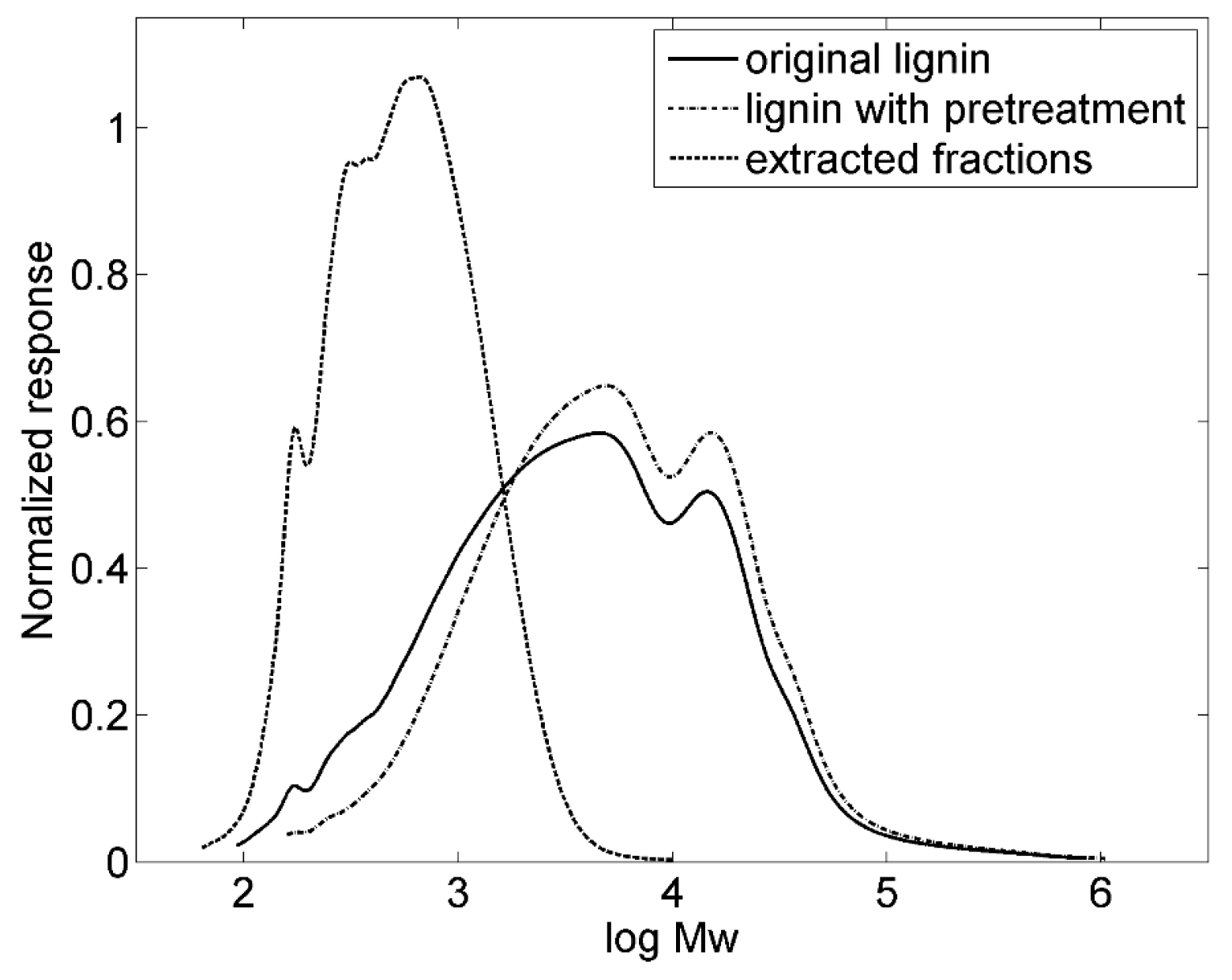


| Sample | Mw (g/mol) | Mn (g/mol) | PD |
|---|---|---|---|
| Original lignin | 14,000 | 1400 | 9.9 |
| Lignin with pretreatment | 16,000 | 2400 | 6.8 |
| Extracted fractions | 700 | 400 | 1.8 |
| Sample | Tg (°C) | Td (5%, °C) | V (250 °C, %) |
|---|---|---|---|
| Original lignin | 156 | 255 | 95.3 |
| Lignin with pretreatment | 146 | 257 | 95.5 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, H.; Cornell, A.; Alvarado, F.; Behm, M.; Leijonmarck, S.; Li, J.; Tomani, P.; Lindbergh, G. Lignin as a Binder Material for Eco-Friendly Li-Ion Batteries. Materials 2016, 9, 127. https://doi.org/10.3390/ma9030127
Lu H, Cornell A, Alvarado F, Behm M, Leijonmarck S, Li J, Tomani P, Lindbergh G. Lignin as a Binder Material for Eco-Friendly Li-Ion Batteries. Materials. 2016; 9(3):127. https://doi.org/10.3390/ma9030127
Chicago/Turabian StyleLu, Huiran, Ann Cornell, Fernando Alvarado, Mårten Behm, Simon Leijonmarck, Jiebing Li, Per Tomani, and Göran Lindbergh. 2016. "Lignin as a Binder Material for Eco-Friendly Li-Ion Batteries" Materials 9, no. 3: 127. https://doi.org/10.3390/ma9030127
APA StyleLu, H., Cornell, A., Alvarado, F., Behm, M., Leijonmarck, S., Li, J., Tomani, P., & Lindbergh, G. (2016). Lignin as a Binder Material for Eco-Friendly Li-Ion Batteries. Materials, 9(3), 127. https://doi.org/10.3390/ma9030127






