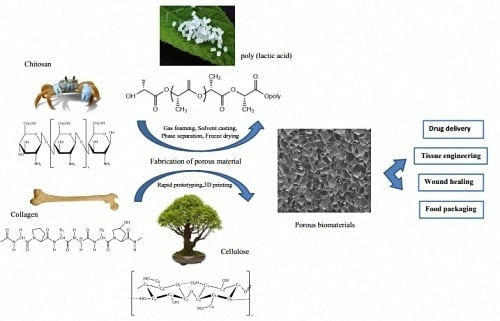
Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites
Abstract
:1. Introduction
2. Fabrication of Natural Biopolymers
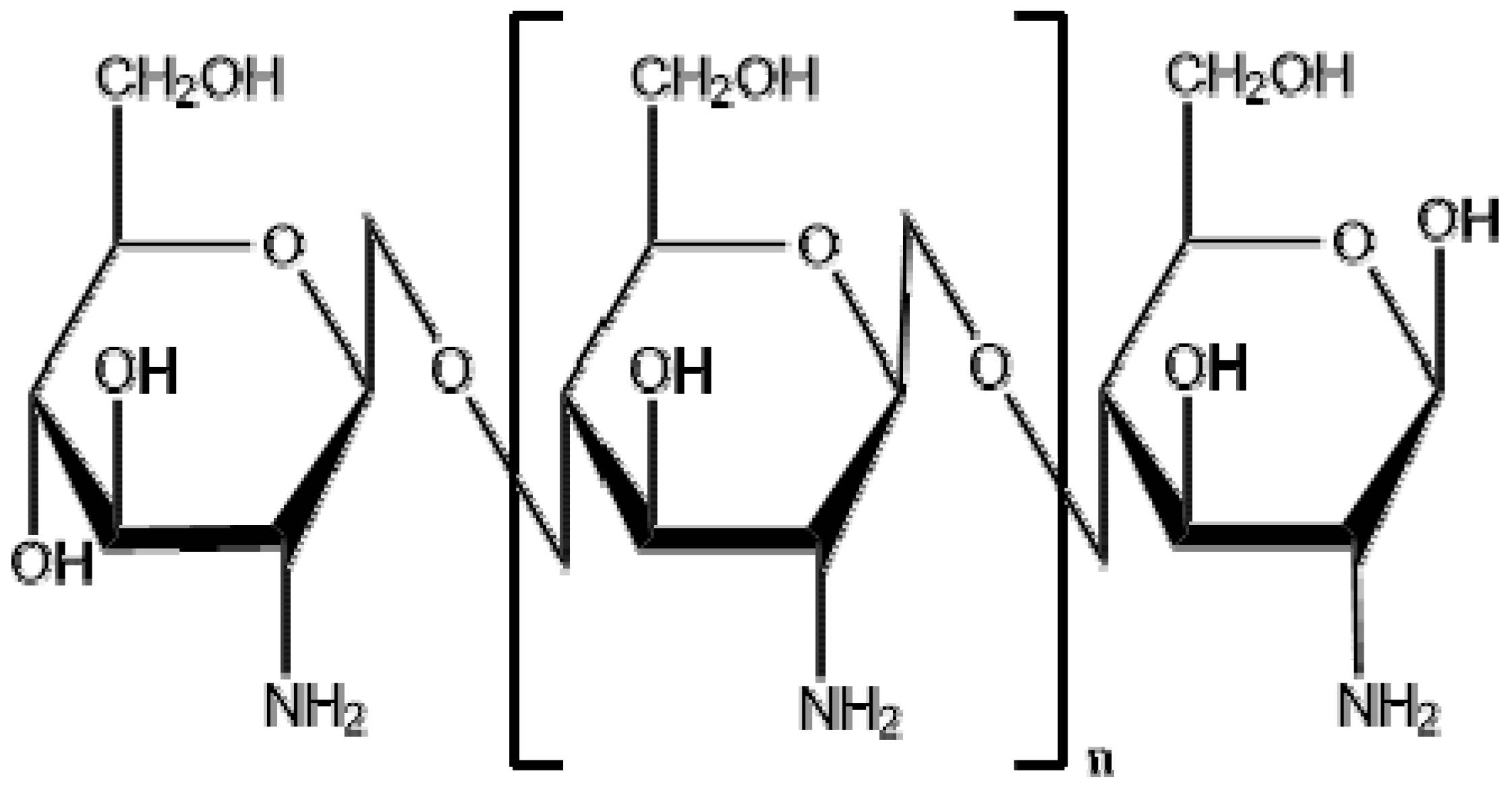
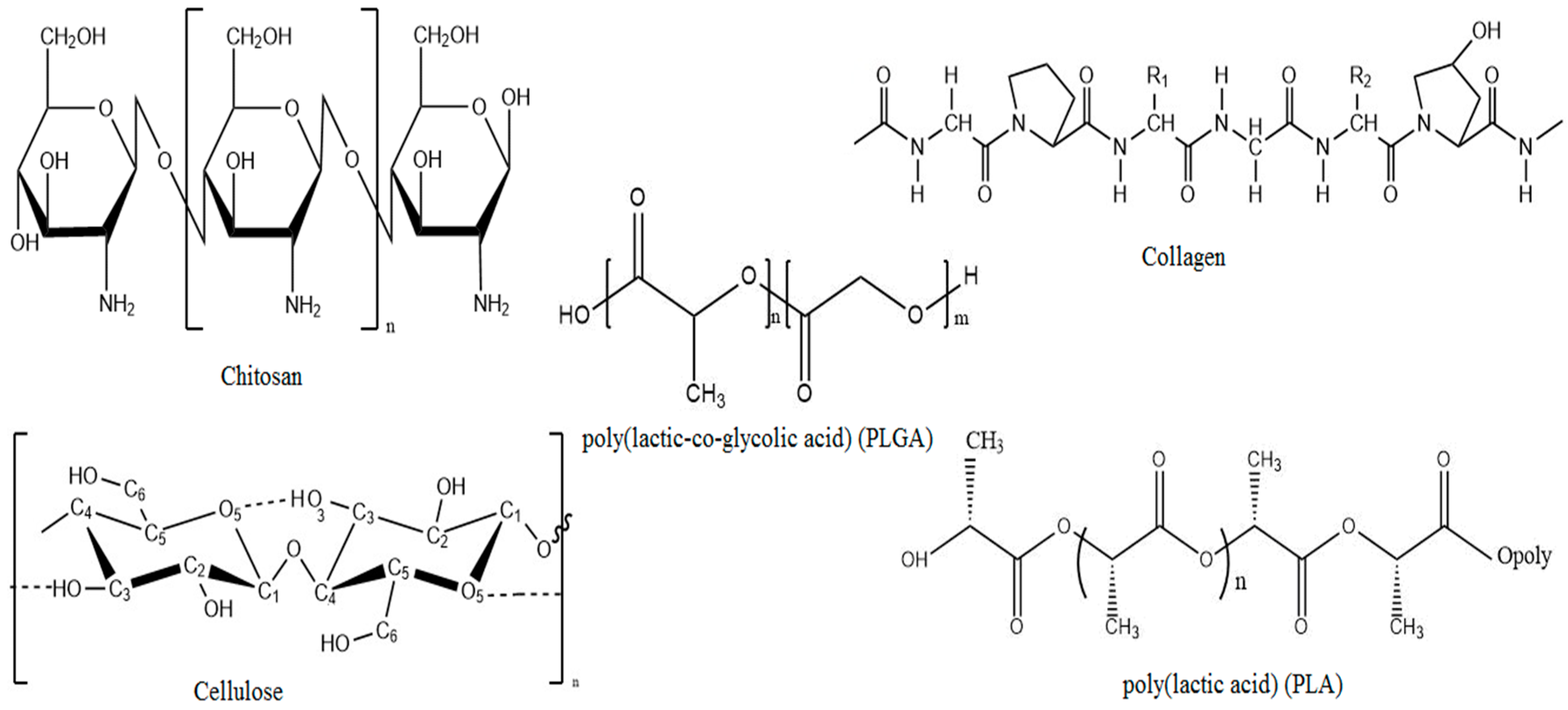
2.1. Chitosan
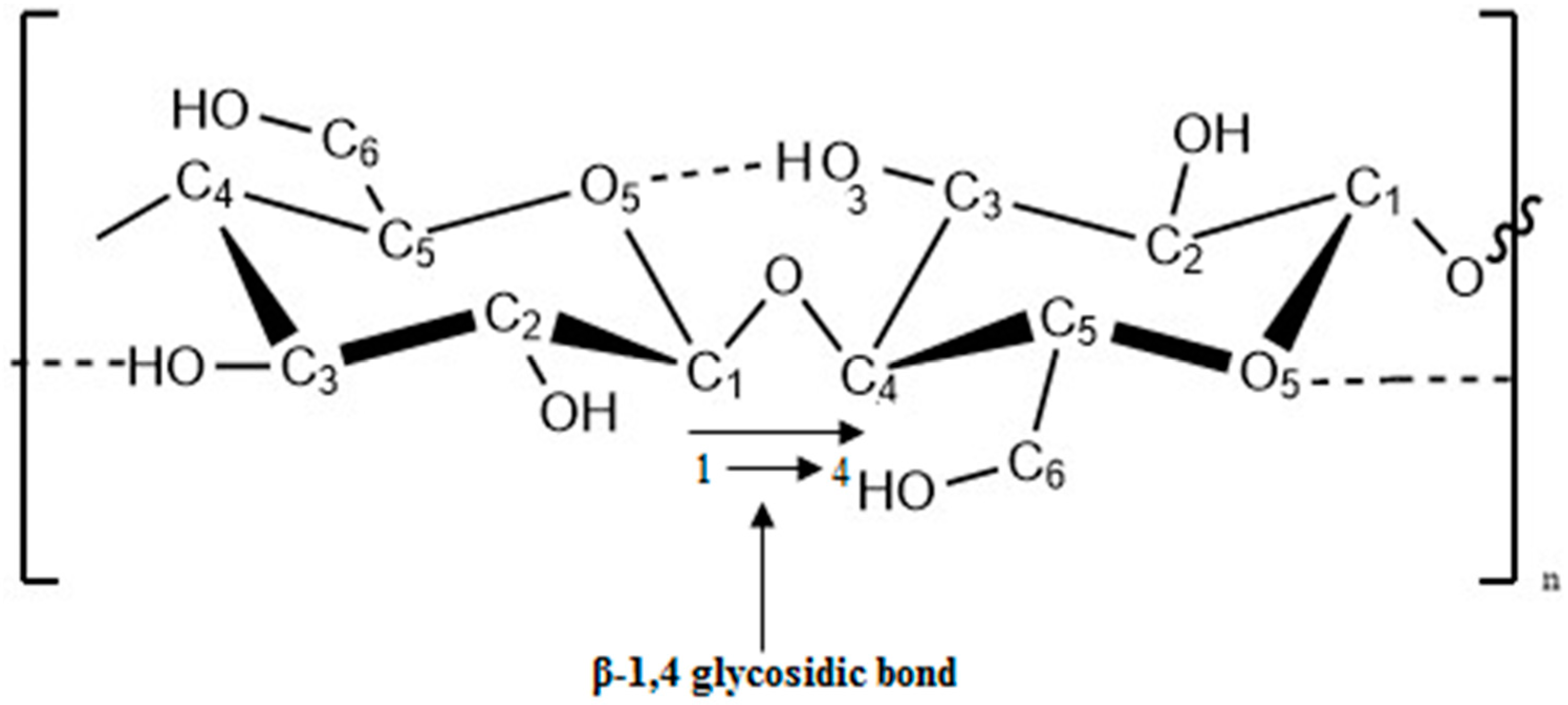
2.2. Cellulose
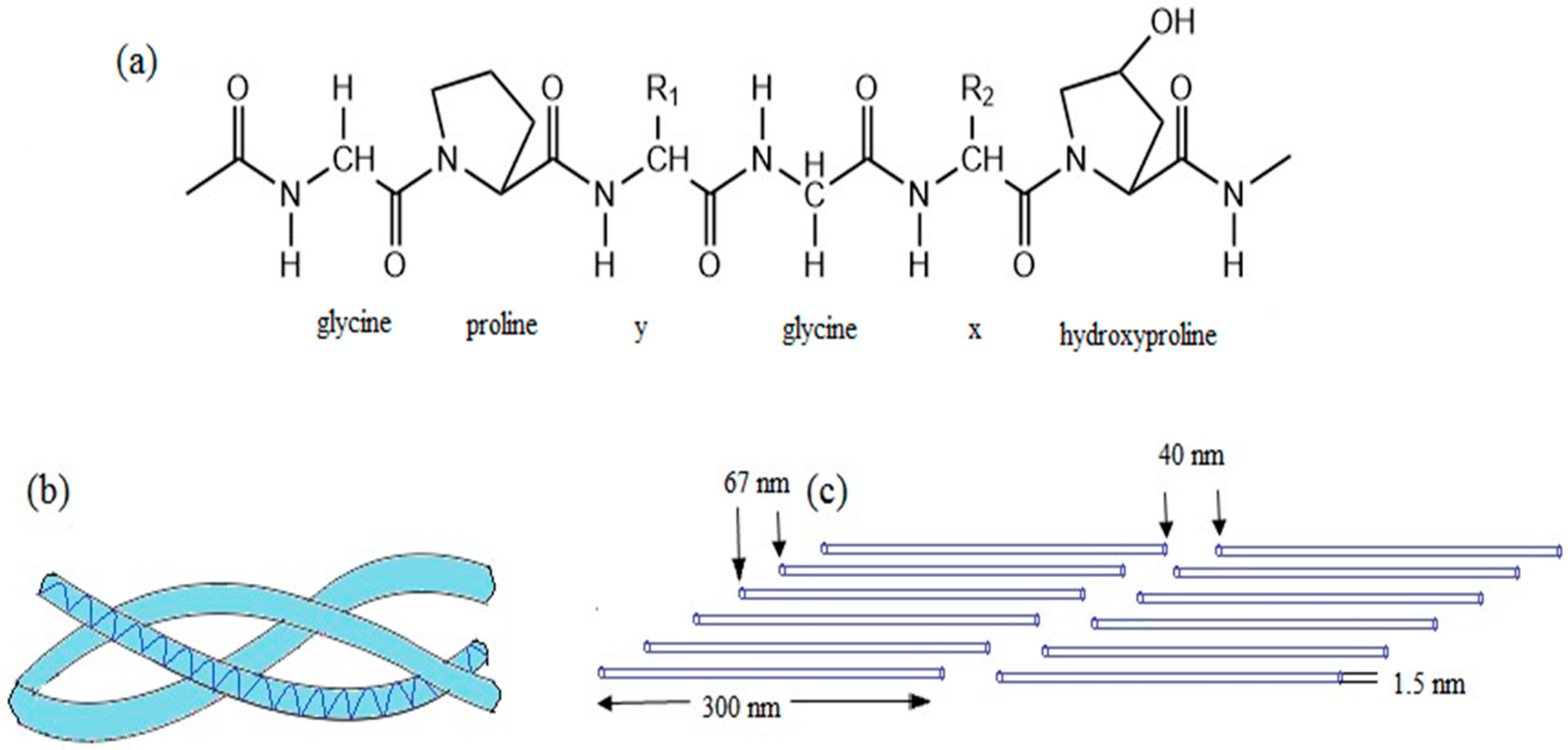
2.3. Collagen
3. Fabrication of Synthetic Biopolymers
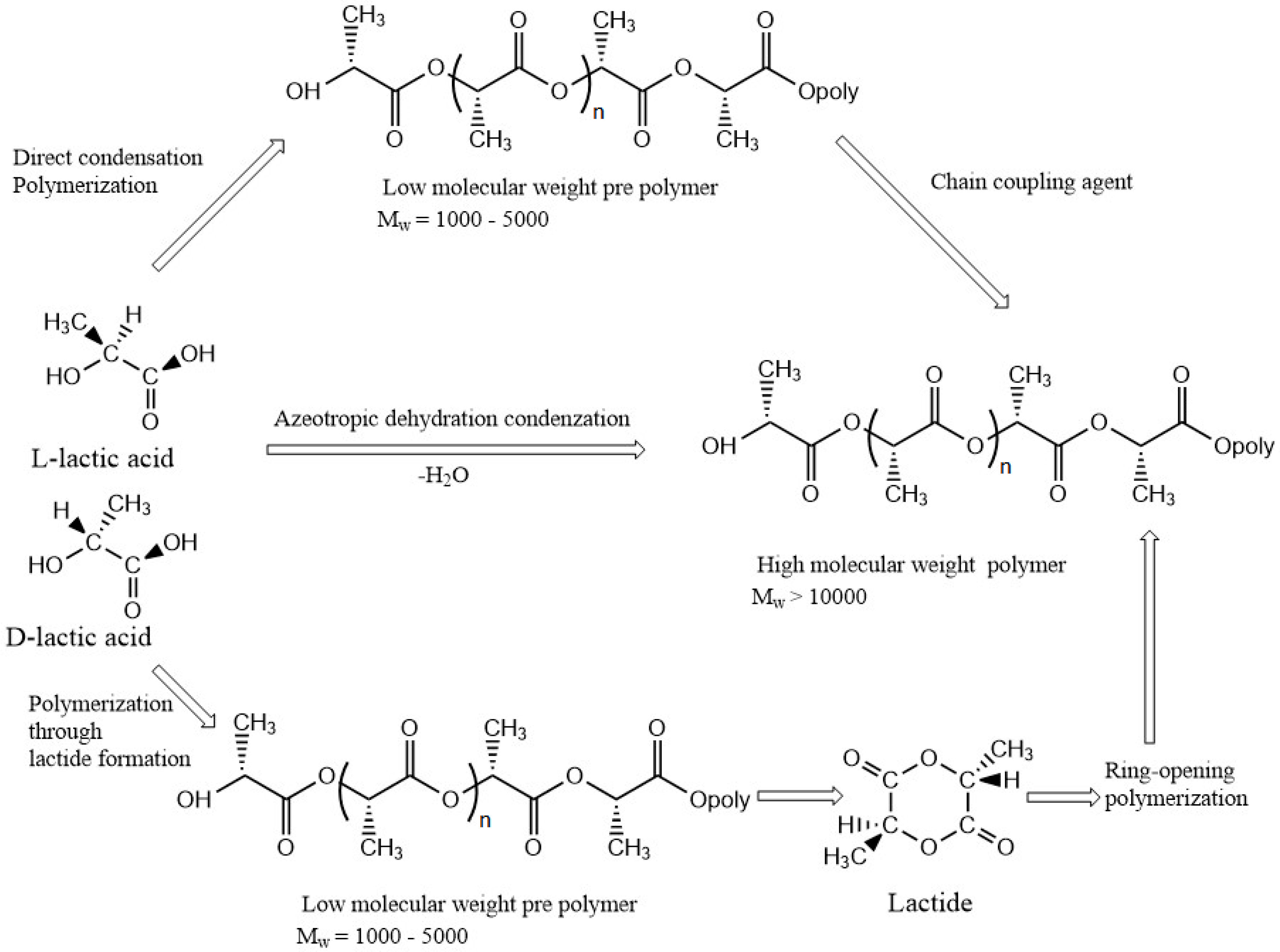
3.1. Poly(lactic acid)
3.2. Poly(lactic-co-glycolic acid)
4. Biocomposite Materials
4.1. Fabrication of Porous Biocomposite Materials
4.1.1. Chitosan/PLA Composite Materials
4.1.2. PLA/Cellulose Nanocomposites
4.1.3. Cellulose/Chitosan Composite Materials
4.1.4. Chitosan/PLGA Composite Materials
4.1.5. Chitosan/Collagen Composite Material
5. Fabrication Methods
5.1. Solvent Casting and Particulate Leaching
5.2. Thermally Induced Phase Separation
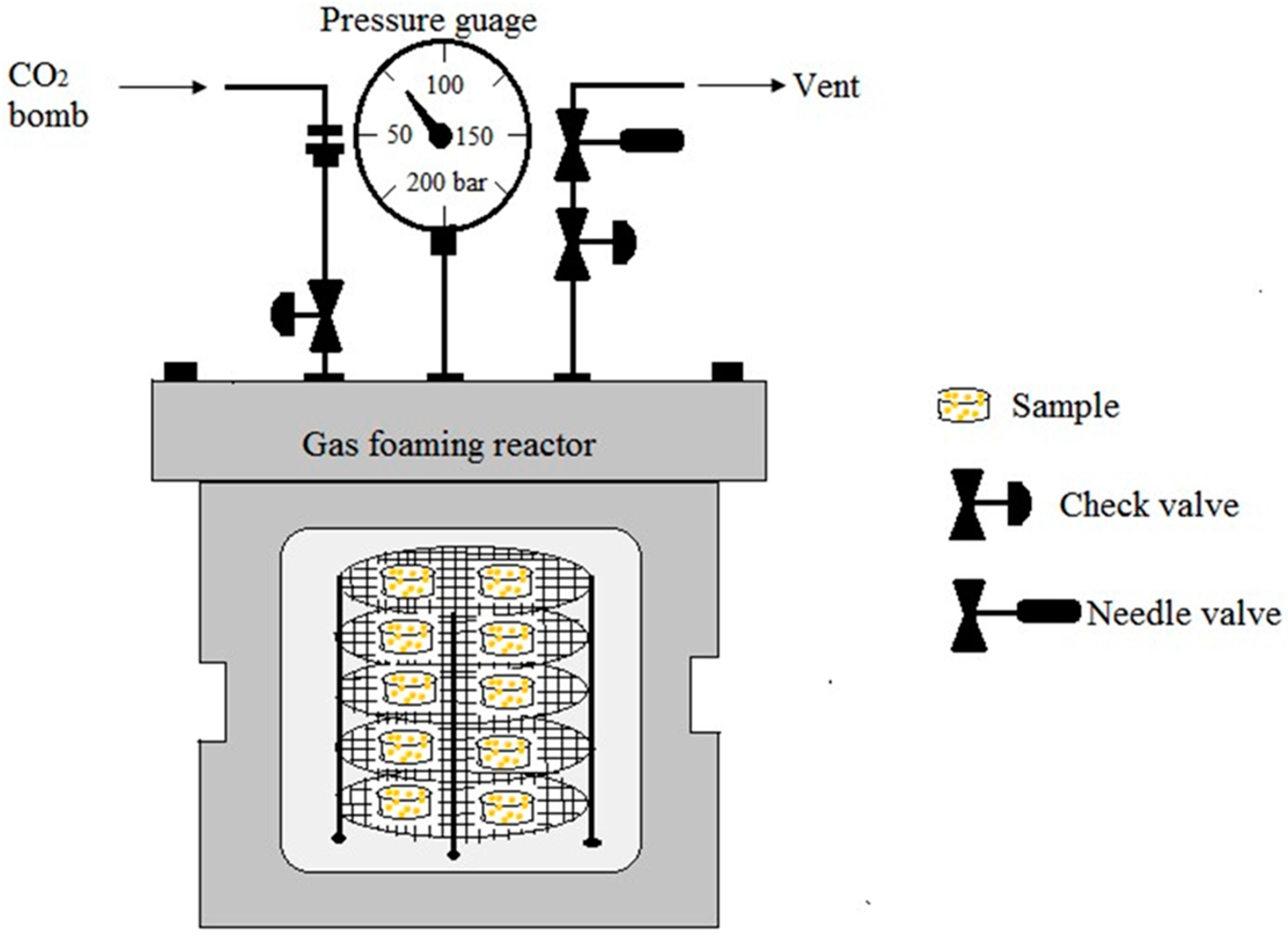
5.3. Gas Foaming
5.4. Emulsion Freeze-Drying
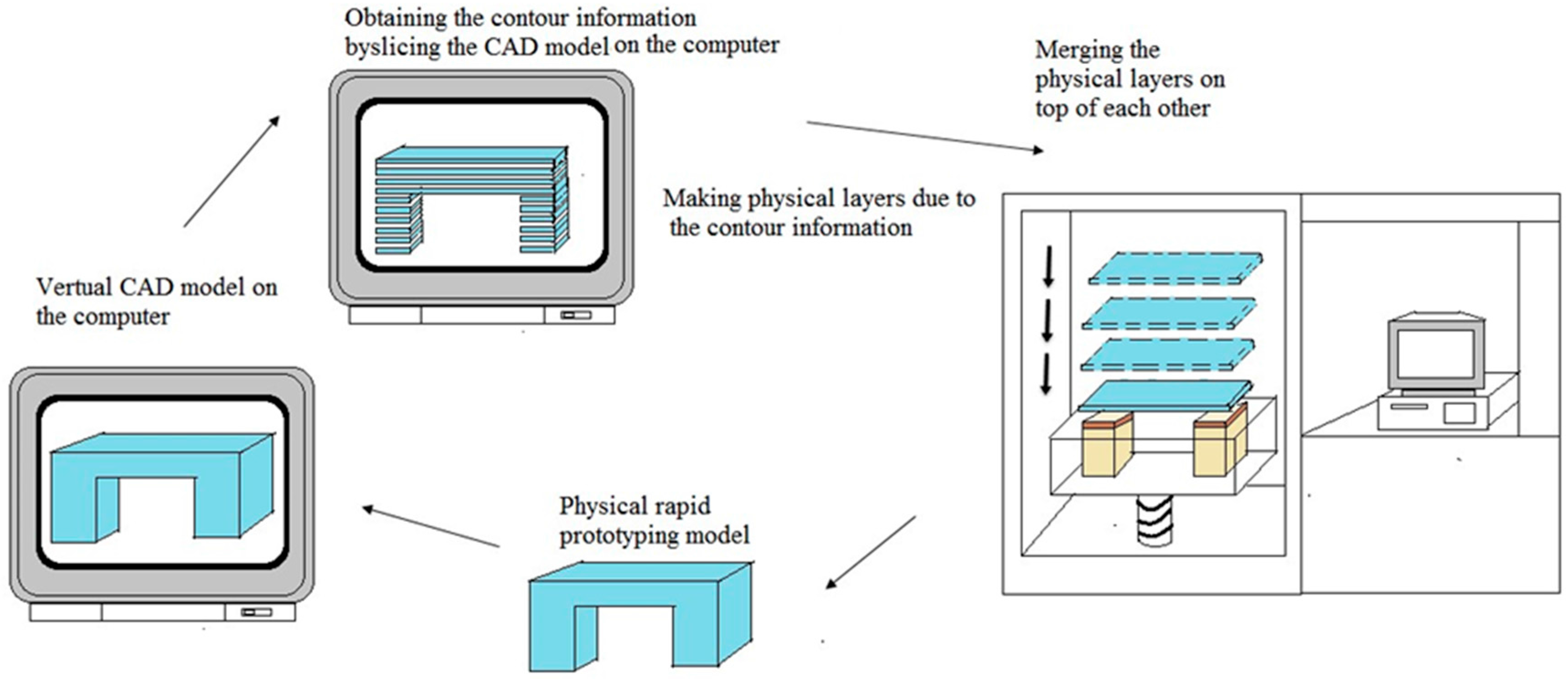
5.5. Rapid Prototyping Technique
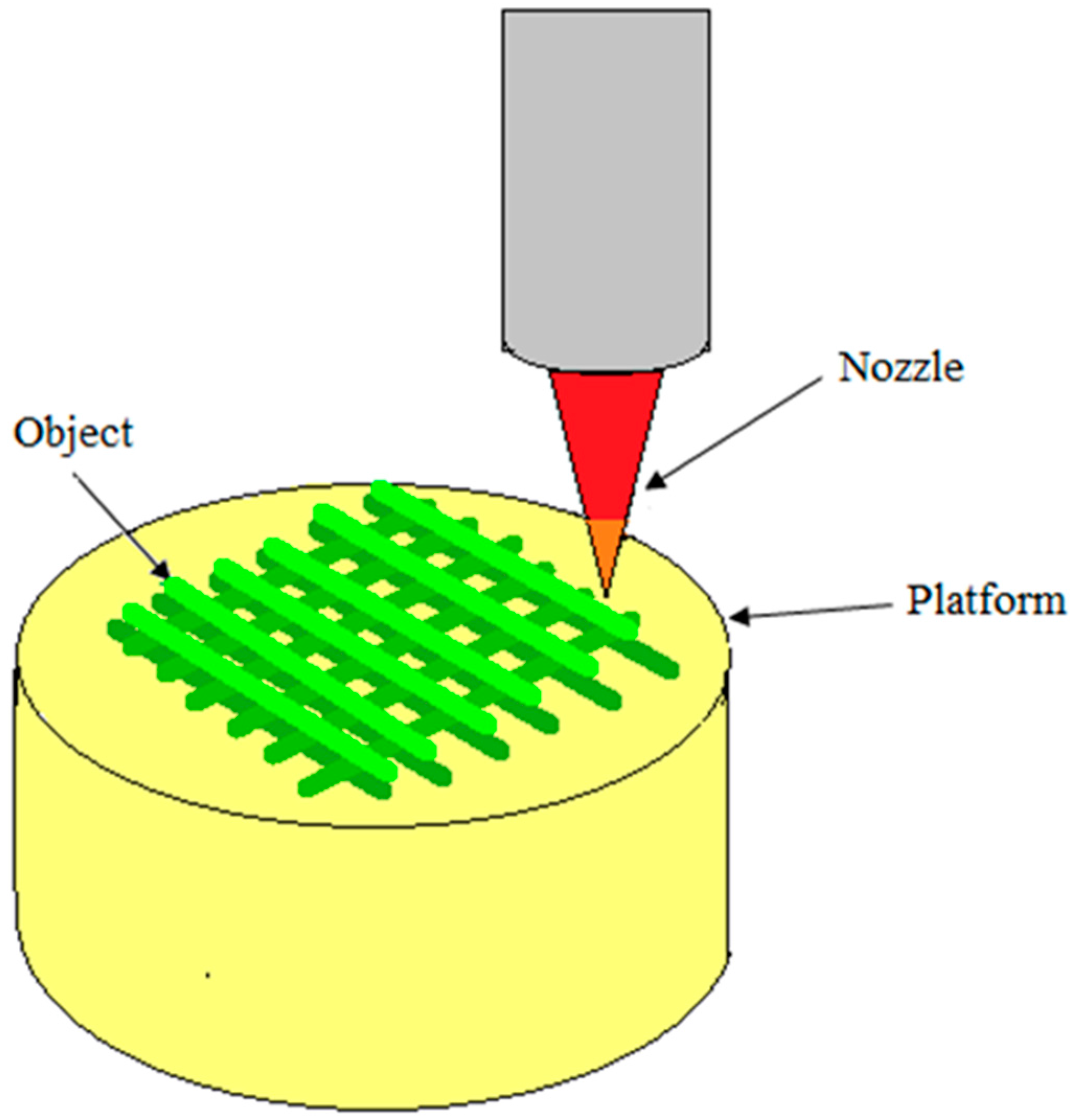
5.6. 3D Printing
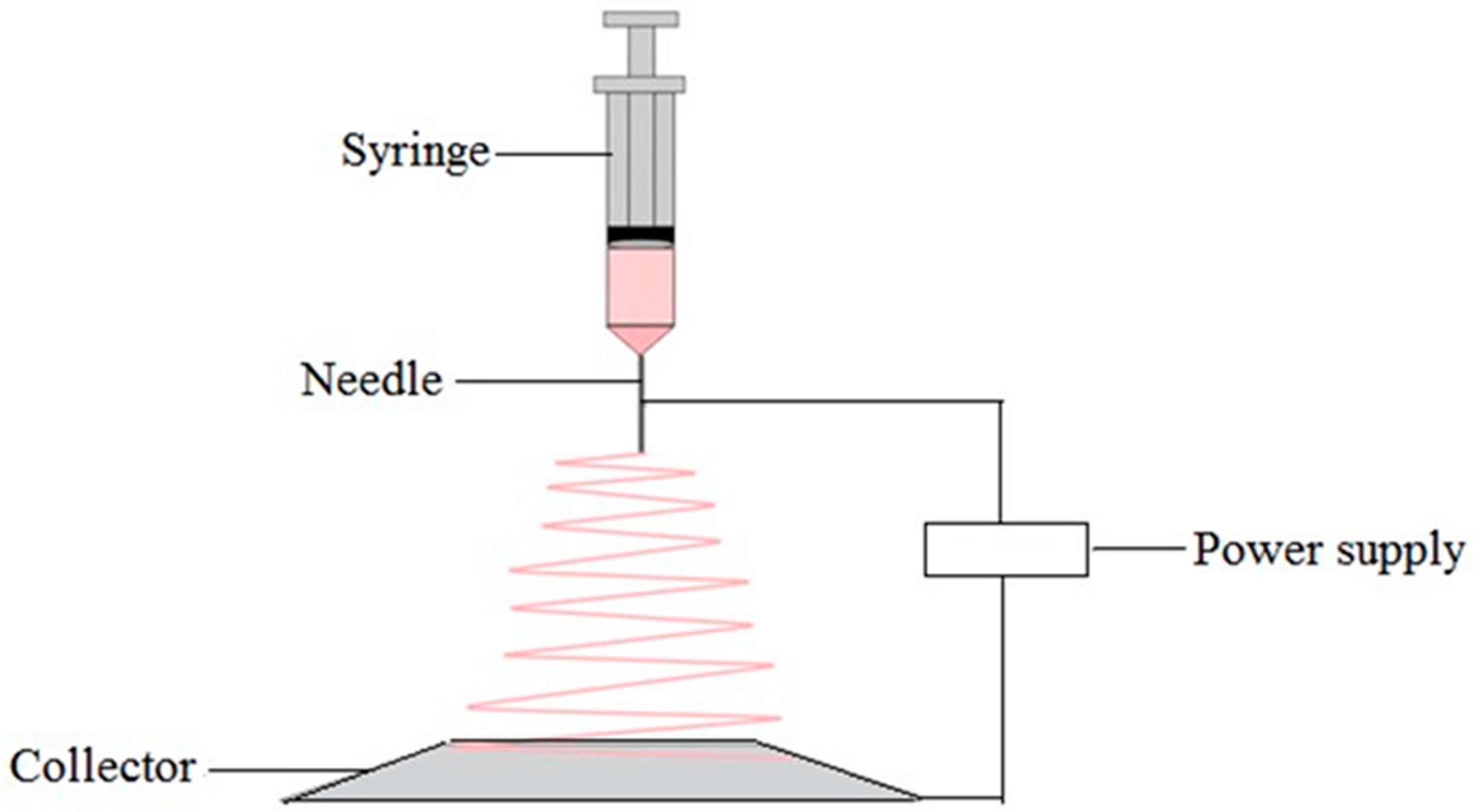
5.7. Electrospinning Technique
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vroman, I.; Tighzert, L. Biodegradable polymers. Materials 2009, 2, 307–344. [Google Scholar] [CrossRef]
- Lim, K.M.; Ching, Y.C.; Gan, S.N. Effect of palm oil bio-based plasticizer on the morphological, thermal and mechanical properties of poly(vinyl chloride). Polymers 2015, 7, 2031–2043. [Google Scholar] [CrossRef]
- Othman, S.H. Bio-nanocomposite materials for food packaging applications: Types of biopolymer and nano-sized filler. Agric. Agric. Sci. Procedia 2014, 2, 296–303. [Google Scholar] [CrossRef]
- Niaounakis, M. Biopolymers: Applications and Trends; William Andrew Publishing: Oxford, UK, 2015; pp. 91–138. [Google Scholar]
- Ershad, A.; Yong, K.C.; Ching, Y.C.; Chuah, C.H.; Liou, N.S. Effect of single and double stage chemically treated kenaf fibers on mechanical properties of polyvinyl alcohol film. Bioresource 2015, 10, 822–838. [Google Scholar]
- Tan, B.K.; Ching, Y.C.; Poh, S.C.; Luqman, C.A.; Gan, S.N. Review of natural fiber reinforced poly(vinyl alcohol) based composites: Application and Opportunity. Polymers 2015, 7, 2205–2222. [Google Scholar] [CrossRef]
- Gavillon, R.; Budtova, T. Aerocellulose: New highly porous cellulose prepared from cellulose-NaOH aqueous solutions. Biomacromolecules 2007, 9, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.; Sun, D. Graded/gradient porous biomaterials. Materials 2009, 3, 26–47. [Google Scholar] [CrossRef]
- Dhandayuthapani, B.; Yoshida, Y.; Maekawa, T.; Kumar, D.S. Polymeric scaffolds in tissue engineering application: A review. Int. J. Polym. Sci. 2011, 2011, 290602. [Google Scholar] [CrossRef]
- Nwe, N.; Furuike, T.; Tamura, H. The mechanical and biological properties of chitosan scaffolds for tissue regeneration templates are significantly enhanced by chitosan from gongronella butleri. Materials 2009, 2, 374–398. [Google Scholar] [CrossRef]
- Rodriguez-Vazquez, M.; Vega-Ruiz, B.; Ramos-Zuniga, R.; Saldana-Koppel, D.A.; Quinones-Olvera, L.F. Chitosan and its potential use as a scaffold for tissue engineering in regenerative medicine. BioMed Res. Int. 2015, 2015, 821279. [Google Scholar] [CrossRef] [PubMed]
- Croisier, F.; Jerome, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef]
- Hsieh, W.-C.; Chang, C.-P.; Lin, S.-M. Morphology and characterization of 3D micro-porous structured chitosan scaffolds for tissue engineering. Colloids Surf. B Biointerfaces 2007, 57, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Simonescu, C.M.; Marin, I.; Tardei, C.; Marinescu, V.; Oprea, O.; Capatina, C. Chitosan and chitosan modified with glutaraldehyde microparticles for Pb(II) biosorption I. Microparticles preparation and characterization. Rev. Chim. 2014, 65, 627–632. [Google Scholar]
- Ji, C.; Annabi, N.; Khademhosseini, A.; Dehghani, F. Fabrication of porous chitosan scaffolds for soft tissue engineering using dense gas CO2. Acta Biomater. 2011, 7, 1653–1664. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, E.; Sendemir-Urkmez, A.; Yesil-Celiktas, O. Supercritical CO2 processing of a chitosan-based scaffold: Can implantation of osteoblastic cells be enhanced? J. Supercrit. Fluids 2013, 75, 120–127. [Google Scholar] [CrossRef]
- Sivashankari, P.; Prabaharan, M. Prospects of chitosan-based scaffolds for growth factor release in tissue engineering. Int. J. Biol. Macromol. 2016, 93 Pt B, 1382–1389. [Google Scholar] [CrossRef] [PubMed]
- Ching, Y.C.; Ershad, A.; Luqman, C.A.; Choo, K.W.; Yong, C.K.; Sabariah, J.J.; Chuah, C.H.; Liou, N.S. Rheological properties of cellulose nanocrystal-embedded polymer composites: A review. Cellulose 2016, 23, 1011–1030. [Google Scholar] [CrossRef]
- Ching, Y.C.; Ng, T.S. Effect of preparation conditions on cellulose from oil palm empty fruit bunch fiber. Bioresource 2014, 9, 6373–6385. [Google Scholar] [CrossRef]
- Rubentheren, V.; Thomas, W.; Ching, Y.C.; Praveena, N. Effects of heat treatment on chitosan nanocomposite film reinforced with nanocrystalline cellulose and tannic acid. Carbohydr. Polym. 2016, 140, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.Y.; Ching, Y.C.; Chuah, C.H.; Luqman, C.A.; Liou, N.S. Individualization of microfibrillated celluloses from oil palm empty fruit bunch: Comparative studies between acid hydrolysis and ammonium persulfate oxidation. Cellulose 2016, 23, 379–390. [Google Scholar] [CrossRef]
- Mhd Haniffa, M.A.C.; Ching, Y.C.; Luqman, C.A.; Poh, S.C.; Chuah, C.H. Review of bionanocomposite coating films and their applications. Polymers 2016, 8, 246. [Google Scholar] [CrossRef]
- Medina-Gonzalez, Y.; Camy, S.; Condoret, J.-S. Cellulosic materials as biopolymers and supercritical CO2 as a green process: Chemistry and applications. Int. J. Sustain. Eng. 2012, 5, 47–65. [Google Scholar] [CrossRef]
- White, L.J.; Hutter, V.; Tai, H.; Howdle, S.M.; Shakesheff, K.M. The effect of processing variables on morphological and mechanical properties of supercritical CO2 foamed scaffolds for tissue engineering. Acta Biomater. 2012, 8, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Baldino, L.; Cardea, S.; Reverchon, E. Production of antimicrobial membranes loaded with potassium sorbate using a supercritical phase separation process. Innov. Food Sci. Emerg. Technol. 2016, 34, 77–85. [Google Scholar] [CrossRef]
- Xiong, G.; Luo, H.; Gu, F.; Zhang, J.; Hu, D.; Wan, Y. A novel in vitro three-dimensional macroporous scaffolds from bacterial cellulose for culture of breast cancer cells. J. Biomater. Nanobiotechnol. 2013, 4, 316–326. [Google Scholar] [CrossRef]
- Chang, S.-W.; Shefelbine, S.J.; Buehler, M.J. Structural and mechanical differences between collagen homo-and heterotrimers: Relevance for the molecular origin of brittle bone disease. Biophys. J. 2012, 102, 640–648. [Google Scholar] [CrossRef] [PubMed]
- Friess, W. Collagen-biomaterial for drug delivery. Eur. J. Pharm. Biopharm. 1998, 45, 113–136. [Google Scholar] [CrossRef]
- Ferreira, A.M.; Gentile, P.; Chiono, V.; Ciardelli, G. Collagen for bone tissue regeneration. Acta Biomater. 2012, 8, 3191–3200. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.H.; Ko, Y.G.; Lu, H.; Kawazoe, N.; Chen, G. Preparation of porous collagen scaffolds with micropatterned structures. Adv. Mater. 2012, 24, 4311–4316. [Google Scholar] [CrossRef] [PubMed]
- Nanda, H.S.; Chen, S.; Zhang, Q.; Kawazoe, N.; Chen, G. Collagen scaffolds with controlled insulin release and controlled pore structure for cartilage tissue engineering. BioMed Res. Int. 2014, 2014, 623805. [Google Scholar] [CrossRef] [PubMed]
- Pourjavadi, A.; Kurdtabar, M.; Ghasemzadeh, H. Salt-and ph-resisting collagen-based highly porous hydrogel. Polym. J. 2008, 40, 94–103. [Google Scholar] [CrossRef]
- Yee, Y.Y.; Ching, Y.C.; Shaifulazuar, R.; Awanis, H.; Ramesh, S. Preparation and characterization of poly(lactic acid) based composite reinforced with oil palm empty fruit bunch film and nanosilica. Bioresource 2016, 11, 2269–2286. [Google Scholar] [CrossRef]
- Ching, Y.C.; Rosiyah, Y.Y.; Li, G. Preparation and characterization of nano particle reinforced polyactides composite. J. Nano Res. 2013, 25, 128–136. [Google Scholar]
- Conde, C.M.; Demarco, F.F.; Casagrande, L.; Alcazar, J.C.; Nör, J.E.; Tarquinio, S.B.C. Influence of poly-l-lactic acid scaffold’s pore size on the proliferation and differentiation of dental pulp stem cells. Braz. Dent. J. 2015, 26, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Chavalitkul, J.; Likittanaprasong, N.; Seansala, P.; Suttiruengwong, S.; Seadan, M. Application of ultrasonic atomization for biopolymer particles and tube fabrication. Energy Procedia 2014, 56, 458–465. [Google Scholar] [CrossRef]
- Lee, H.-Y.; Jin, G.-Z.; Shin, U.S.; Kim, J.-H.; Kim, H.-W. Novel porous scaffolds of poly(lactic acid) produced by phase-separation using room temperature ionic liquid and the assessments of biocompatibility. J. Mater. Sci. Mater. Med. 2012, 23, 1271–1279. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Ma, Z.; Zhou, Q.; Li, J.; Gao, C.; Shen, J. Poly(lactic acid) scaffold fabricated by gelatin particle leaching has good biocompatibility for chondrogenesis. J. Biomater. Sci. Polym. Ed. 2008, 19, 207–221. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, W.; Kumar, V. A method for solvent-free fabrication of porous polymer using solid-state foaming and ultrasound for tissue engineering applications. Biomaterials 2006, 27, 1924–1929. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.-M.; Qian, J.; Zhang, J.; Lin, Z.-F.; Li, J.-S.; Xu, J.-Z.; Li, Z.-M. Engineering porous poly(lactic acid) scaffolds with high mechanical performance via a solid state extrusion/porogen leaching approach. Polymers 2016, 8, 213. [Google Scholar] [CrossRef]
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Chiono, V.; Carmagnola, I.; Hatton, P.V. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int. J. Mol. Sci. 2014, 15, 3640–3659. [Google Scholar] [CrossRef] [PubMed]
- Azimi, B.; Nourpanah, P.; Rabiee, M.; Arbab, S. Poly(lactide-co-glycolide) fiber: An overview. J. Eng. Fabr. Fibers (JEFF) 2014, 9, 47–66. [Google Scholar]
- Krebs, M.D.; Sutter, K.A.; Lin, A.S.; Guldberg, R.E.; Alsberg, E. Injectable poly(lactic-co-glycolic) acid scaffolds with in situ pore formation for tissue engineering. Acta Biomater. 2009, 5, 2847–2859. [Google Scholar] [CrossRef] [PubMed]
- Singh, L.; Kumar, V.; Ratner, B.D. Generation of porous microcellular 85/15 poly(dl-lactide-co-glycolide) foams for biomedical applications. Biomaterials 2004, 25, 2611–2617. [Google Scholar] [CrossRef] [PubMed]
- Day, R.M.; Boccaccini, A.R.; Maquet, V.; Shurey, S.; Forbes, A.; Gabe, S.M.; Jérôme, R. In vivo characterisation of a novel bioresorbable poly(lactide-co-glycolide) tubular foam scaffold for tissue engineering applications. J. Mater. Sci. Mater. Med. 2004, 15, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kwag, D.S.; Lee, D.J.; Lee, E.S. Acidic pH-stimulated tiotropium release from porous poly(lactic-co-glycolic acid) microparticles containing 3-diethylaminopropyl-conjugated hyaluronate. Macromol. Res. 2016, 24, 176–181. [Google Scholar] [CrossRef]
- Rubentheren, V.; Ward, T.A.; Chee, C.Y.; Tang, C.K. Processing and analysis of chitosan nanocomposites reinforced with chitin whiskers and tannic acid as a crosslinker. Carbohydr. Polym. 2015, 115, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Ching, Y.C.; Rahman, A.; Ching, K.Y.; Sukiman, N.L.; Cheng, H.C. Preparation and characterization of polyvinyl alcohol-based composite reinforced with nanocellulose and nanosilica. BioResources 2015, 10, 3364–3377. [Google Scholar] [CrossRef]
- Yong, K.C.; Ching, Y.C.; Afzan, M.; Lim, Z.K.; Chong, K.E. Mechanical and thermal properties of chemical treated oil palm empty fruit bunches fiber reinforced polyvinyl alcohol composite. J. Biobased Mater. Bioenergy 2015, 9, 231–235. [Google Scholar]
- Kumar, A.; Negi, Y.S.; Choudhary, V.; Bhardwaj, N.K. Microstructural and mechanical properties of porous biocomposite scaffolds based on polyvinyl alcohol, nano-hydroxyapatite and cellulose nanocrystals. Cellulose 2014, 21, 3409–3426. [Google Scholar] [CrossRef]
- Li, L.; Ding, S.; Zhou, C. Preparation and degradation of PLA/chitosan composite materials. J. Appl. Polym. Sci. 2004, 91, 274–277. [Google Scholar] [CrossRef]
- Zhang, Z.; Cui, H. Biodegradability and biocompatibility study of poly(chitosan-g-lactic acid) scaffolds. Molecules 2012, 17, 3243–3258. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Li, Q.F.; Gu, B.; Liu, K.; Shen, G.X. In vitro and in vivo evaluation of a biodegradable chitosan-PLA composite peripheral nerve guide conduit material. Microsurgery 2008, 28, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Choo, K.; Ching, Y.C.; Chuah, C.H.; Sabariah, J.; Liou, N.S. Preparation and characterization of polyvinyl alcohol-chitosan composite films reinforced with cellulose nanofiber. Materials 2016, 9, 644. [Google Scholar] [CrossRef]
- Ng, T.S.; Ching, Y.C.; Awanis, N.; Ishenny, N.; Rahman, M.R. Effect of bleaching condition on thermal properties and UV-transmittance of PVA/cellulose biocomposites. Mater. Res. Innov. 2014, 18, 400–404. [Google Scholar] [CrossRef]
- Cho, S.Y.; Park, H.H.; Yun, Y.S.; Jin, H.-J. Influence of cellulose nanofibers on the morphology and physical properties of poly(lactic acid) foaming by supercritical carbon dioxide. Macromol. Res. 2013, 21, 529–533. [Google Scholar] [CrossRef]
- Dlouha, J.; Suryanegara, L.; Yano, H. The role of cellulose nanofibres in supercritical foaming of polylactic acid and their effect on the foam morphology. Soft Matter 2012, 8, 8704–8713. [Google Scholar] [CrossRef]
- Xiang, C.; Taylor, A.G.; Hinestroza, J.P.; Frey, M.W. Controlled release of nonionic compounds from poly(lactic acid)/cellulose nanocrystal nanocomposite fibers. J. Appl. Polym. Sci. 2013, 127, 79–86. [Google Scholar] [CrossRef]
- Twu, Y.-K.; Huang, H.-I.; Chang, S.-Y.; Wang, S.-L. Preparation and sorption activity of chitosan/cellulose blend beads. Carbohydr. Polym. 2003, 54, 425–430. [Google Scholar] [CrossRef]
- Rubentheren, V.; Ward, T.A.; Chee, C.Y.; Nair, P. Physical and chemical reinforcement of chitosan film using nanocrystalline cellulose and tannic acid. Cellulose 2015, 22, 2529–2541. [Google Scholar] [CrossRef]
- Li, M.; Wang, Z.; Li, B. Adsorption behaviour of congo red by cellulose/chitosan hydrogel beads regenerated from ionic liquid. Desalination Water Treat. 2016, 57, 16970–16980. [Google Scholar] [CrossRef]
- Kim, J.; Cai, Z.; Lee, H.S.; Choi, G.S.; Lee, D.H.; Jo, C. Preparation and characterization of a bacterial cellulose/chitosan composite for potential biomedical application. J. Polym. Res. 2011, 18, 739–744. [Google Scholar] [CrossRef]
- Kim, S.J.; Yang, D.H.; Chun, H.J.; Chae, G.T.; Jang, J.W.; Shim, Y.B. Evaluations of chitosan/poly(d,l-lactic-co-glycolic acid) composite fibrous scaffold for tissue engineering applications. Macromol. Res. 2013, 21, 931–939. [Google Scholar] [CrossRef]
- Yuanyuan, Z.; Song, L. Preparation of chitosan/poly(lactic-co glycolic acid)(PLGA) nanocoposite for tissue engineering scaffold. Optoelectron. Adv. Mater.-Rapid Commun. 2012, 6, 516–519. [Google Scholar]
- Cui, Z.; Zhao, H.; Peng, Y.; Han, J.; Turng, L.-S.; Shen, C. Fabrication and characterization of highly porous chitosan/poly(dl lactic-co-glycolic acid) nanocomposite scaffolds using electrospinning and freeze drying. J. Biobased Mater. Bioenergy 2014, 8, 281–291. [Google Scholar] [CrossRef]
- Ajalloueian, F.; Tavanai, H.; Hilborn, J.; Donzel-Gargand, O.; Leifer, K.; Wickham, A.; Arpanaei, A. Emulsion electrospinning as an approach to fabricate PLGA/chitosan nanofibers for biomedical applications. BioMed Res. Int. 2014, 2014, 475280. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Cheng, X.R.; Wang, J.W.; Xu, D.X.; Wang, G. Preparation and evaluation of porous chitosan/collagen scaffolds for periodontal tissue engineering. J. Bioact. Compat. Polym. 2006, 21, 207–220. [Google Scholar] [CrossRef]
- Ma, L.; Gao, C.; Mao, Z.; Zhou, J.; Shen, J.; Hu, X.; Han, C. Collagen/chitosan porous scaffolds with improved biostability for skin tissue engineering. Biomaterials 2003, 24, 4833–4841. [Google Scholar] [CrossRef]
- Mighri, N.; Mao, J.; Mighri, F.; Ajji, A.; Rouabhia, M. Chitosan-coated collagen membranes promote chondrocyte adhesion, growth, and interleukin-6 secretion. Materials 2015, 8, 7673–7689. [Google Scholar] [CrossRef]
- Wang, L.; Stegemann, J.P. Glyoxal crosslinking of cell-seeded chitosan/collagen hydrogels for bone regeneration. Acta Biomater. 2011, 7, 2410–2417. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, J.; Best, S.; Cameron, R. Quantitative architectural description of tissue engineering scaffolds. Mater. Technol. 2014, 29, 281–295. [Google Scholar] [CrossRef]
- Mallick, S.; Tripathi, S.; Srivastava, P. Advancement in scaffolds for bone tissue engineering: A review. IOSR J. Pharm. Biol. Sci. 2015, 10, 37–54. [Google Scholar]
- Lim, J.I.; Lee, Y.-K.; Shin, J.-S.; Lim, K.-J. Preparation of interconnected porous chitosan scaffolds by sodium acetate particulate leaching. J. Biomater. Sci. Polym. Ed. 2011, 22, 1319–1329. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Lee, H.K.; Kim, J.Y.; Hyon, S.H.; Kim, S.C. Thermally induced phase separation in poly(lactic acid)/dialkyl phthalate systems. J. Appl. Polym. Sci. 2003, 88, 2224–2232. [Google Scholar] [CrossRef]
- Li, D.; Krantz, W.B.; Greenberg, A.R.; Sani, R.L. Membrane formation via thermally induced phase separation (TIPS): Model development and validation. J. Membr. Sci. 2006, 279, 50–60. [Google Scholar] [CrossRef]
- Yang, H.C.; Wu, Q.Y.; Liang, H.Q.; Wan, L.S.; Xu, Z.K. Thermally induced phase separation of poly(vinylidene fluoride)/diluent systems: Optical microscope and infrared spectroscopy studies. J. Polym. Sci. B Polym. Phys. 2013, 51, 1438–1447. [Google Scholar] [CrossRef]
- Kim, J.F.; Kim, J.H.; Lee, Y.M.; Drioli, E. Thermally induced phase separation and electrospinning methods for emerging membrane applications: A review. AIChE J. 2016, 62, 461–490. [Google Scholar] [CrossRef]
- Wei, G.; Ma, P.X. Structure and properties of nano-hydroxyapatite/polymer composite scaffolds for bone tissue engineering. Biomaterials 2004, 25, 4749–4757. [Google Scholar] [CrossRef] [PubMed]
- La Carrubba, V.; Pavia, F.C.; Brucato, V.; Piccarolo, S. PLLA/PLA scaffolds prepared via thermally induced phase separation (TIPS): Tuning of properties and biodegradability. Int. J. Mater. Form. 2008, 1, 619–622. [Google Scholar] [CrossRef]
- Liu, X.; Ma, P.X. Polymeric scaffolds for bone tissue engineering. Ann. Biomed. Eng. 2004, 32, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Sachlos, E.; Czernuszka, J. Making tissue engineering scaffolds work. Review: The application of solid freeform fabrication technology to the production of tissue engineering scaffolds. Eur. Cell Mater. 2003, 5, 39–40. [Google Scholar]
- Poursamar, S.A.; Hatami, J.; Lehner, A.N.; da Silva, C.L.; Ferreira, F.C.; Antunes, A.P.M. Gelatin porous scaffolds fabricated using a modified gas foaming technique: Characterisation and cytotoxicity assessment. Mater. Sci. Eng. C 2015, 48, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Bhamidipati, M.; Scurto, A.M.; Detamore, M.S. The future of carbon dioxide for polymer processing in tissue engineering. Tissue Eng. B Rev. 2013, 19, 221–232. [Google Scholar]
- Mooney, D.J.; Baldwin, D.F.; Suh, N.P.; Vacanti, J.P.; Langer, R. Novel approach to fabricate porous sponges of poly(d,l-lactic-co-glycolic acid) without the use of organic solvents. Biomaterials 1996, 17, 1417–1422. [Google Scholar] [CrossRef]
- Bak, T.-Y.; Kook, M.-S.; Jung, S.-C.; Kim, B.-H. Biological effect of gas plasma treatment on CO2 gas foaming/salt leaching fabricated porous polycaprolactone scaffolds in bone tissue engineering. J. Nanomater. 2014, 2014, 657542. [Google Scholar] [CrossRef]
- Annabi, N.; Mithieux, S.M.; Weiss, A.S.; Dehghani, F. Cross-linked open-pore elastic hydrogels based on tropoelastin, elastin and high pressure CO2. Biomaterials 2010, 31, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- Whang, K.; Thomas, C.; Healy, K.; Nuber, G. A novel method to fabricate bioabsorbable scaffolds. Polymer 1995, 36, 837–842. [Google Scholar] [CrossRef]
- Sultana, N.; Wang, M. Fabrication of tissue engineering scaffolds. In Integrated Biomaterials in Tissue Engineering; Scrivener Publishing: Beverly, MA, USA, 2012; pp. 63–89. [Google Scholar]
- Mikos, A.G.; Temenoff, J.S. Formation of highly porous biodegradable scaffolds for tissue engineering. Electron. J. Biotechnol. 2000, 3, 23–24. [Google Scholar] [CrossRef]
- Sultana, N.; Wang, M. Fabrication of HA/PHBV composite scaffolds through the emulsion freezing/freeze-drying process and characterisation of the scaffolds. J. Mater. Sci. Mater. Med. 2008, 19, 2555–2561. [Google Scholar] [CrossRef] [PubMed]
- Torabi, K.; Farjood, E.; Hamedani, S. Rapid prototyping technologies and their applications in prosthodontics, a review of literature. J. Dent. (Shiraz) 2014, 16, 1–9. [Google Scholar]
- Sobral, J.M.; Caridade, S.G.; Sousa, R.A.; Mano, J.F.; Reis, R.L. Three-dimensional plotted scaffolds with controlled pore size gradients: Effect of scaffold geometry on mechanical performance and cell seeding efficiency. Acta Biomater. 2011, 7, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.E.; Chuan, Y.L.; Pashby, I. Extrusion based rapid prototyping technique: An advanced platform for tissue engineering scaffold fabrication. Biopolymers 2012, 97, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, K.; Nakajima, H. Metallic scaffolds for bone regeneration. Materials 2009, 2, 790–832. [Google Scholar] [CrossRef]
- Stanek, M.; Manas, D.; Manas, M.; Navratil, J.; Kyas, K.; Senkerik, V.; Skrobak, A. Comparison of different rapid prototyping methods. Intern. J. Math. Comput. Simul. 2012, 6, 550–557. [Google Scholar]
- Peltola, S.M.; Melchels, F.P.; Grijpma, D.W.; Kellomäki, M. A review of rapid prototyping techniques for tissue engineering purposes. Ann. Med. 2008, 40, 268–280. [Google Scholar] [CrossRef] [PubMed]
- Abdelaal, O.A.; Darwish, S.M. Fabrication of tissue engineering scaffolds using rapid prototyping techniques. Int. J. Mech. Aerosp. Ind. Mechatron. Manuf. Eng. 2011, 5, 2317–2325. [Google Scholar]
- Zhu, N.; Li, M.; Cooper, D.; Chen, X. Development of novel hybrid poly(l-lactide)/chitosan scaffolds using the rapid freeze prototyping technique. Biofabrication 2011, 3, 034105. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cui, R.; Sun, L.; Aifantis, K.E.; Fan, Y.; Feng, Q.; Cui, F.; Watari, F. 3D-printed biopolymers for tissue engineering application. Int. J. Polym. Sci. 2014, 2014, 829145. [Google Scholar] [CrossRef]
- Cox, S.C.; Thornby, J.A.; Gibbons, G.J.; Williams, M.A.; Mallick, K.K. 3D printing of porous hydroxyapatite scaffolds intended for use in bone tissue engineering applications. Mater. Sci. Eng. C 2015, 47, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Lam, C.X.F.; Mo, X.; Teoh, S.-H.; Hutmacher, D. Scaffold development using 3d printing with a starch-based polymer. Mater. Sci. Eng. C 2002, 20, 49–56. [Google Scholar] [CrossRef]
- Lee, M.; Dunn, J.C.; Wu, B.M. Scaffold fabrication by indirect three-dimensional printing. Biomaterials 2005, 26, 4281–4289. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zhou, W.; Ma, D.; Ma, Q.; Bridges, D.; Ma, Y.; Hu, A. Electrospinning of nanofibers and their applications for energy devices. J. Nanomater. 2015, 2015, 140716. [Google Scholar] [CrossRef]
- Agarwal, S.; Wendorff, J.H.; Greiner, A. Use of electrospinning technique for biomedical applications. Polymer 2008, 49, 5603–5621. [Google Scholar] [CrossRef]
- Sundararaghavan, H.G.; Metter, R.B.; Burdick, J.A. Electrospun fibrous scaffolds with multiscale and photopatterned porosity. Macromol. Biosci. 2010, 10, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Dahlin, R.L.; Kasper, F.K.; Mikos, A.G. Polymeric nanofibers in tissue engineering. Tissue Eng. B Rev. 2011, 17, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Chew, S.; Wen, Y.; Dzenis, Y.; Leong, K.W. The role of electrospinning in the emerging field of nanomedicine. Curr. Pharm. Des. 2006, 12, 4751–4770. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Feng, Y.; Huang, Z.; Ramakrishna, S.; Lim, C.T. Fabrication of porous electrospun nanofibres. Nanotechnology 2006, 17, 901–908. [Google Scholar] [CrossRef]
- Roozbahani, F.; Sultana, N.; Almasi, D.; Naghizadeh, F. Effects of chitosan concentration on the protein release behaviour of electrospun poly(ε-caprolactone)/chitosan nanofibers. J. Nanomater. 2015, 2015, 11. [Google Scholar] [CrossRef]
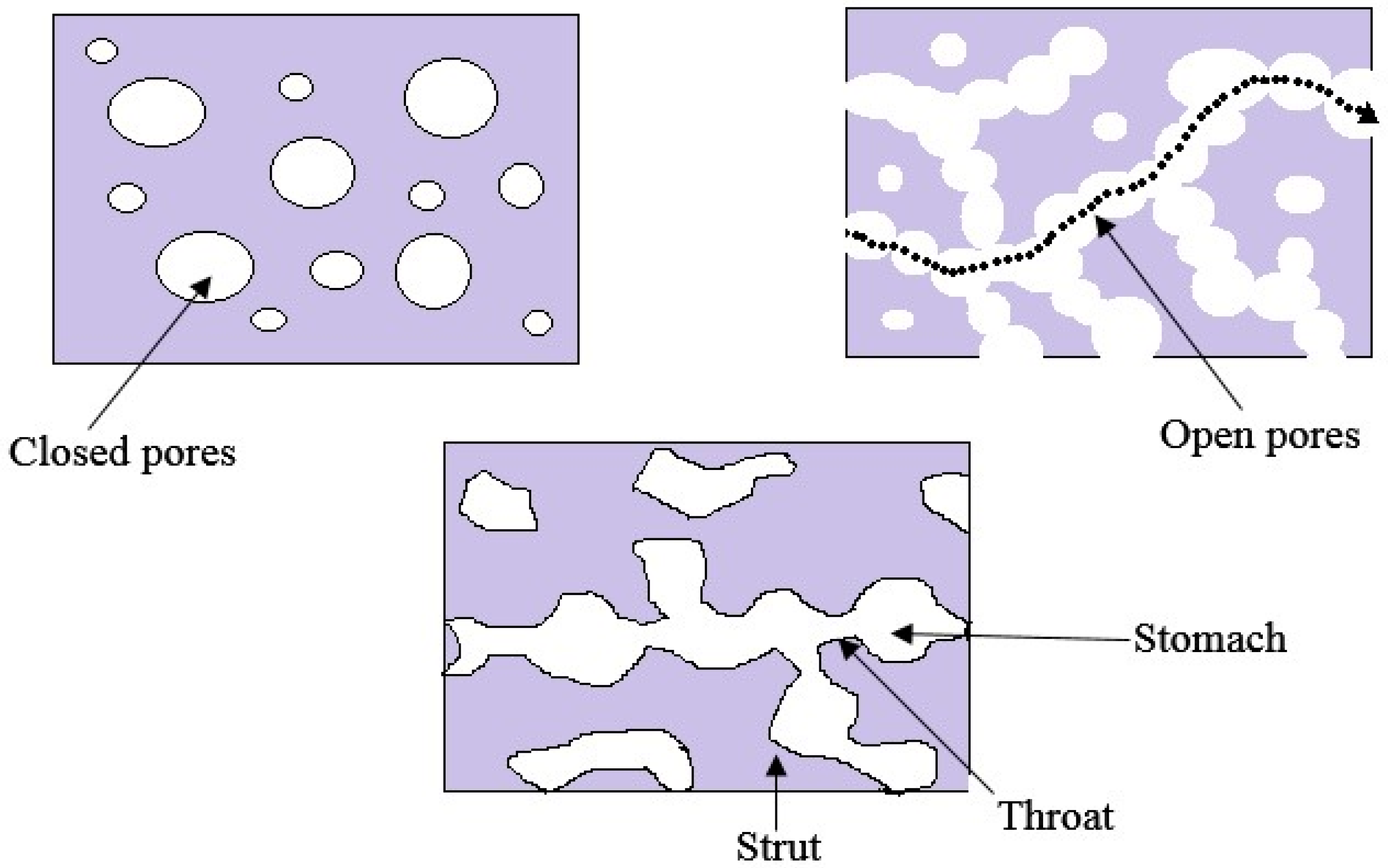



| Biopolymer | Fabrication Method | Application | Pore Characteristics | Reference |
|---|---|---|---|---|
| Chitosan | Freeze-drying | Scaffold | Polygonal pores formed with low molecular weight chitosan and elongated pores formed with high molecular weight chitosan. Average pore sizes of scaffold were approximately 60–90 μm. | [10] |
| Chitosan | Liquid hardening | Scaffold | Pore diameter decreased with increasing the stirring rate and decreasing the concentration of chitosan. Average pore sizes of 200–500 μm and 80% porosity could be obtained by varying the concentration of chitosan and the stirring rate. | [13] |
| Chitosan | Dense gas CO2 | Scaffold | The porous structure obtained without formation of a nonporous skin layer. The average pore size in the scaffold produced at 60 bar and 4 °C was 30 to 40 μm using glutaraldehyde and genipin as crosslinker, respectively. | [15] |
| Chitosan | Supercritical CO2 | Scaffold | Under optimum condition (CO2 pressure of 250 bar, 45 °C, 5 g/min CO2 flow rate for 2 h) that yielded 87.03% porosity. The pore sizes were in the range of 20–100 μm. | [16] |
| Chitosan | Liquid hardening | Scaffold | Pore sizes from 70 to 900 µm were obtained when transferring the stirred chitosan solution to sodium hydroxide solution. Macroporous chitosan scaffold with porosity 85% ± 2% was obtained. | [17] |
| Cellulose | Supercritical CO2 | Polymeric foam | Pore size increased as decreasing the pressure and pore morphology varied with the depressurization rate. | [22] |
| Cellulose | Supercritical CO2 | Antimicrobial food packaging | Mean pore size decreased with increasing the operative pressure and decreasing the operative temperature. Porosity increased with decreasing the pressure and with increasing the temperature. | [24] |
| Cellulose | Infrared laser | Scaffold | Pore size varied with adjusting the distance between specimen and laser focus. Patterned macropores with smooth surface and diameter larger than 100 μm was introduced to the scaffold with this method. | [25] |
| Collagen | Freeze-drying (using ice particulates as templates) | Scaffold | Two types of pores formed; one from the negative replica of ice templates and other from ice crystals developed by freeze-drying. Pore size decreased with decreasing the freezing temperature. The micropatterned pores of the scaffold can be controlled by designing a desirable micropattern for the ice template. | [30] |
| Collagen | Freeze-drying (mixing with ice particulates) | Drug delivery system | Interconnected pore structure obtained with pore size equivalent to ice particulates (150–250 µm). All the scaffolds had large controlled pore structure. | [31] |
| Poly(l-lactic acid) | Solvent-casting and particulate leaching | Scaffold | Two ranges of pore size formed using two particle sizes of NaCl as porogen: 150–250 µm and 251–425 µm. Pore structures were formed after Poly(l-lactic acid) dissolved in chloroform was dropped over the salt and leached with distilled water. | [35] |
| PLA | Phase-separation | Scaffold | Open porous PLA network formed with pore sizes greater than 100 µm and porosities of about 86%–94%. | [37] |
| Poly(l-lactic acid) | Solvent casting and particulate leaching | Scaffold | Interconnected pore structure formed with pre-designed pore sizes (280–450 µm) and porosity >94%. | [38] |
| PLA | CO2 blowing with the application of ultrasound | Scaffold | Interconnectivity of pores improved by ultrasound (by breaking the pore walls of closed pores). The diameters of the closed pores were from 30 to 70 µm. After the ultrasound treatment, pore sizes changed to 30–90 µm due to the formation of interconnected pores. | [39] |
| PLA | Solid state extrusion combined with porogen (NaCl) leaching method | Scaffold | Interconnected porous architecture formed with high connectivity exceeding 97% and with enhanced porosity over 60%. Smaller pore sizes (9 µm) were resulted due to the fragmentation of bulky NaCl during the processing. | [40] |
| PLGA | Phase inversion | Scaffold | Microporous interconnected architecture formed on the surface and within the bulk. The total porosities were 32.19% ± 11.4% and 72.24% ± 4.0% for the control (nonporous) and porous scaffolds, respectively. | [44] |
| PLGA | Supercritical CO2 | Scaffold | Highly interconnected pores formed with relative pore densities ranging from 0.107 to 0.232 and porosities as high as 89%. The pore sizes were within the range from 30 to 100 μm. | [45] |
| PLGA | Thermally induced phase separation | Scaffold | Macropores with average diameter ~100 µm and interconnected micropores of 10–50 µm diameter formed with porosity > 93%. Tubular pores consited of radially oriented. | [46] |
| PLGA | Multi-emulsion method | Drug delivery system | Incorporation of pH-sensitive drug release activator increased the average pore diameter and surface area of microparticles in acidic medium. The average pore diameters of the microparticles at pH 7.4 were within the range of 11, 12, and 27 nm, respectively. It decreased at pH 6.0 to 13, 23, and 120 nm. | [47] |
| Biocomposite Material | Fabrication Method | Application | Pore Characteristics | Reference |
|---|---|---|---|---|
| Chitosan/PLA | Melt molding and particulate (NaCl) leaching | Scaffold | The pore sizes were larger than 100 µm and all the pores including inner pores were interconnected. Porosity increased with the weight fraction of NaCl. | [52] |
| Chitosan/PLA | Freeze drying | Scaffold | Scaffold with interconnected porous structures and pore size around 100–500 µm was obtained. The pore size of the scaffolds decreased with increasing lactic acid/chitosan feed ratio. The chitosan scaffold had a porosity of 62.3% and pore size of 500 µm, and the lactic acid/chitosan scaffold (4:1, wt/wt) had a porosity of 34.37% and pore size of 100 µm. | [53] |
| Chitosan/PLA | Mold casting/infrared dehydration | Scaffold | Well-distributed 0.2 µm pores on the surface of the conduit was formed. | [54] |
| PLA/nanocellulose | Electrospinning | Release of nonionic compounds | There was no significant difference in the mean pore size between the nonwoven fabrics electrospun from PLA containing 0% and 1% cellulose nanocrystals. The mean pore size increased twice as big with PLA containing 10% cellulose nanocrystals. The mean pore sizes of the PLA nonwoven fabrics with 0%, 1% and 10% of cellulose nanocrystals were 0.48 ± 0.04 µm, 0.51 ± 0.08 µm and 0.94 ± 0.14 µm, respectively. | [59] |
| Cellulose/chitosan | Freeze drying | Sorption of trimethylamine and metal ions | The mean pore diameter was within the range of 100–300 μm. The pore diameters decreased with increasing chitosan concentration. | [60] |
| Cellulose/chitosan | Freeze drying | Dye adsorption | The beads were nanoporous with pore sizes from 10 nm to 20 nm. | [62] |
| Bacterial cellulose nanofiber/chitosan | Freeze drying | Scaffold | After the bacterial cellulose was treated by chitosan, porous structure remained but pore sizes became larger. Nanofibrous bacterial cellulose and bacterial cellulose/chitosan composite had well interconnected pore network structure. | [63] |
| Chitosan/PLGA | Electrospinning | Scaffold | In the electrospinning process, the spinning parameters, solution viscosity, polymer concentration, applied voltage, and flow rate highly influenced the porosity and pore size distribution of the composite material. | [64] |
| Chitosan/PLGA nanocomposite | Electrospinning and unidirectional freeze-drying | Scaffold | The porosity was found to be more than 96% and it decreased with increasing the chitosan concentration. | [65] |
| Chitosan/PLGA nanocomposite | Electrospinning and freeze drying | Scaffold | The porosity of chitosan/PLGA nanocomposite scaffolds decreased with increasing the chitosan solution concentration and electrospinning time. | [66] |
| Chitosan/collagen | Freeze drying | Scaffold | The chitosan scaffold showed the pore sizes between 500 and 700 µm while the chitosan/collagen composite scaffold showed a smaller pore sizes of 100–400 µm. The addition of collagen decreased the pore size of the composite scaffold. All samples composed of different proportions of chitosan and collagen showed porosities higher than 90%. The addition of collagen did not change the porosity. | [68] |
| Chitosan/collagen | Freeze drying | Scaffold | The mean pore size of the scaffold increased from 100 μm to >200 μm by crosslinking with glutaraldehyde. Elongated pores were formed with high concentration of glutaraldehyde. Refreeze-drying induced the fusion of some smaller pores to generate larger ones. | [69] |
| Chitosan/collagen | Freeze drying | Scaffold | At the highest chitosan/collagen ratio (75/25), the gels showed a sponge-like structure with larger pores than the gels containing lower chitosan content for both crosslinked and uncrosslinked scaffolds. | [71] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sampath, U.G.T.M.; Ching, Y.C.; Chuah, C.H.; Sabariah, J.J.; Lin, P.-C. Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites. Materials 2016, 9, 991. https://doi.org/10.3390/ma9120991
Sampath UGTM, Ching YC, Chuah CH, Sabariah JJ, Lin P-C. Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites. Materials. 2016; 9(12):991. https://doi.org/10.3390/ma9120991
Chicago/Turabian StyleSampath, Udeni Gunathilake T.M., Yern Chee Ching, Cheng Hock Chuah, Johari J. Sabariah, and Pai-Chen Lin. 2016. "Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites" Materials 9, no. 12: 991. https://doi.org/10.3390/ma9120991
APA StyleSampath, U. G. T. M., Ching, Y. C., Chuah, C. H., Sabariah, J. J., & Lin, P.-C. (2016). Fabrication of Porous Materials from Natural/Synthetic Biopolymers and Their Composites. Materials, 9(12), 991. https://doi.org/10.3390/ma9120991