Synthesis and Drug Delivery Application of Thermo- and pH-Sensitive Hydrogels: Poly(β-CD-co-N-Isopropylacrylamide-co-IAM)
Abstract
:1. Introduction
2. Experimental
2.1. Materials
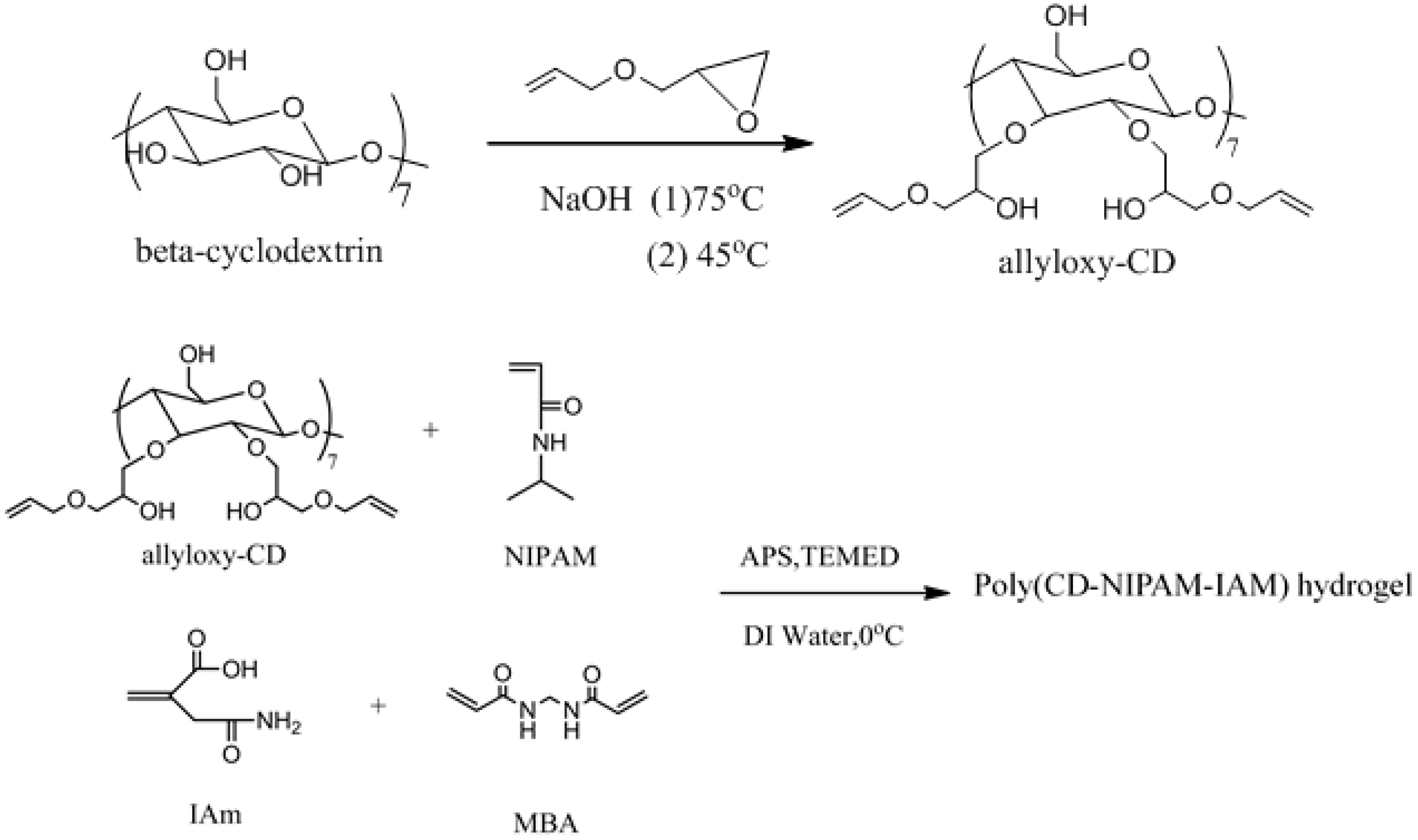
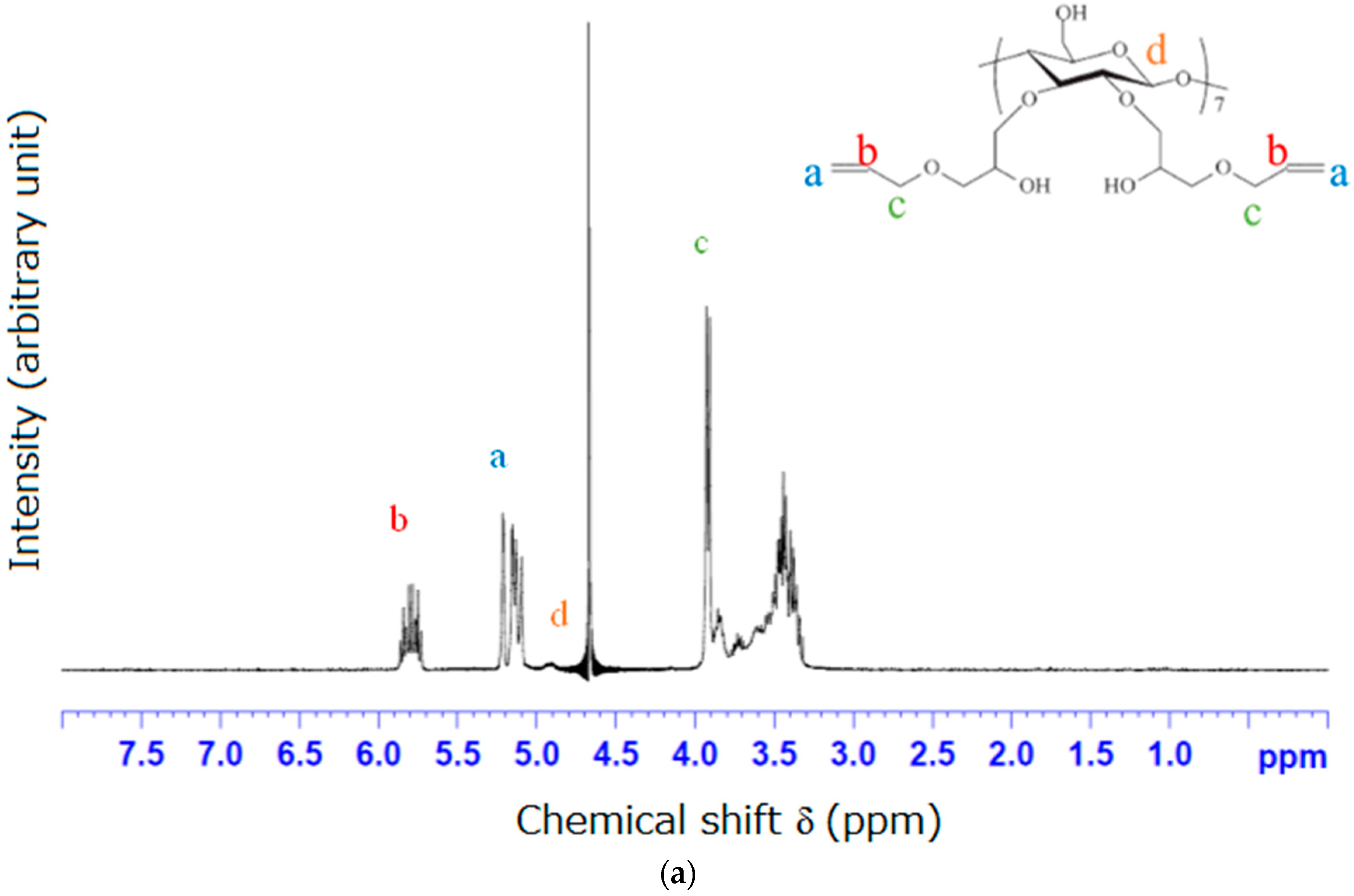
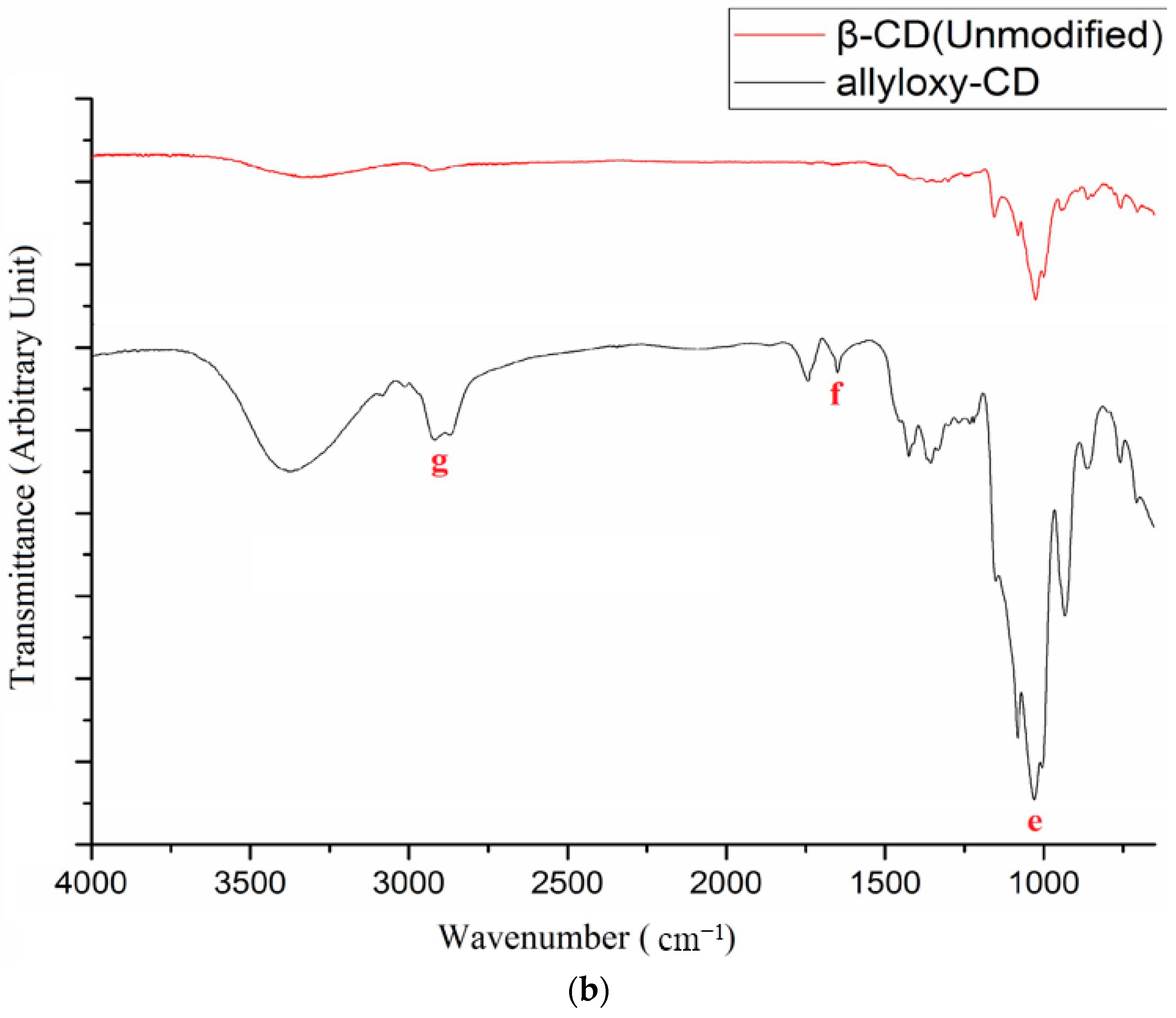
2.2. Synthesis of Allyloxy-CD
2.3. Synthesis of Poly(CD-NIPAM) and Poly(CD-NIPAM-IAM) Copolymers (CD-0, CD-8, CD-10 and CD-12)
2.4. Synthesis of Poly(CD-NIPAM) and Poly(CD-NIPAM-IAM) Hydrogels
2.5. Identification and Characterization
2.6. Drug Release
2.6.1. Encapsulation
2.6.2. Drug Release Experiment
3. Results and Discussion
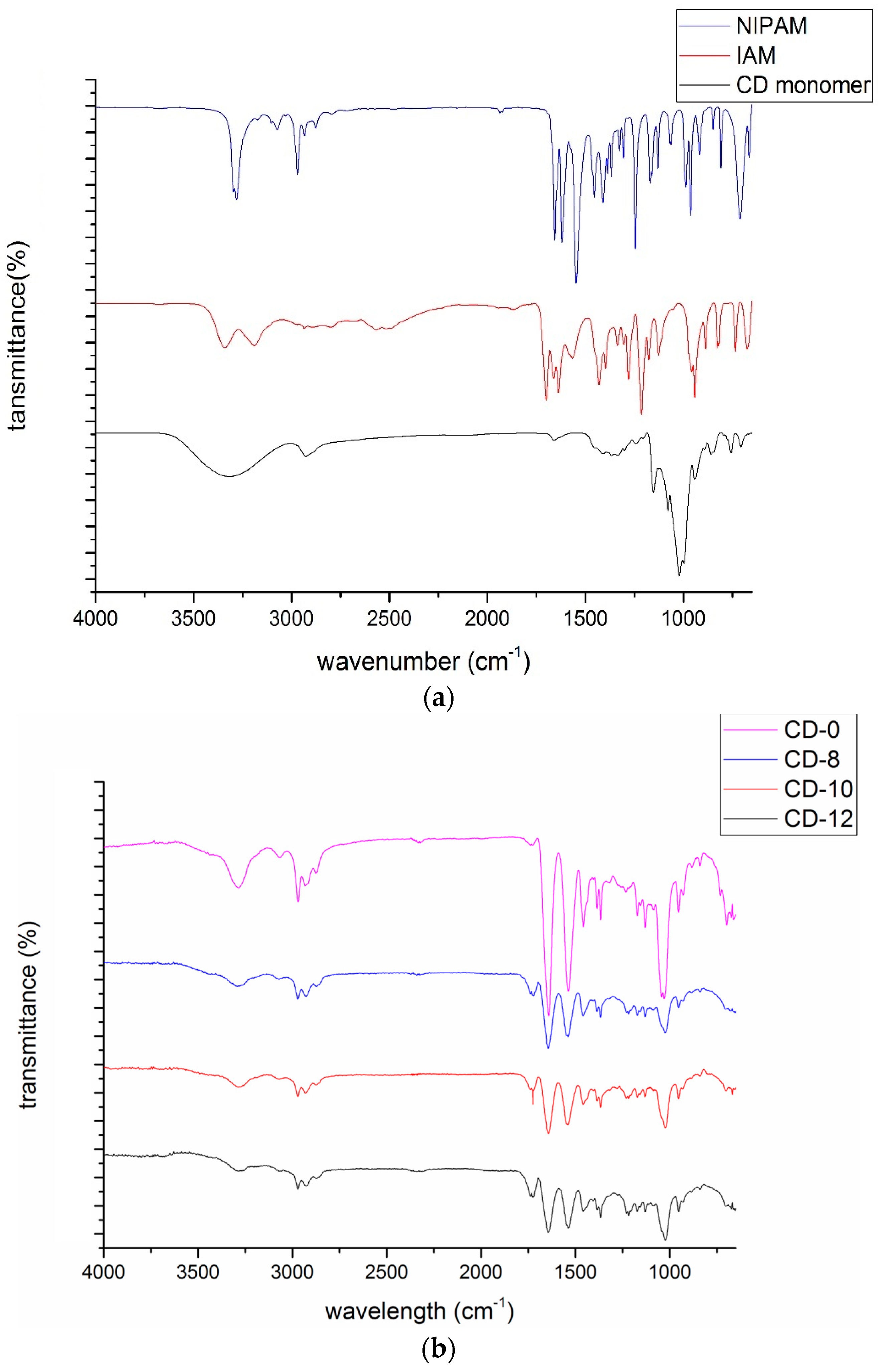
3.1. NMR and FTIR Results of Modified β-Cyclodextrin (β-CD)
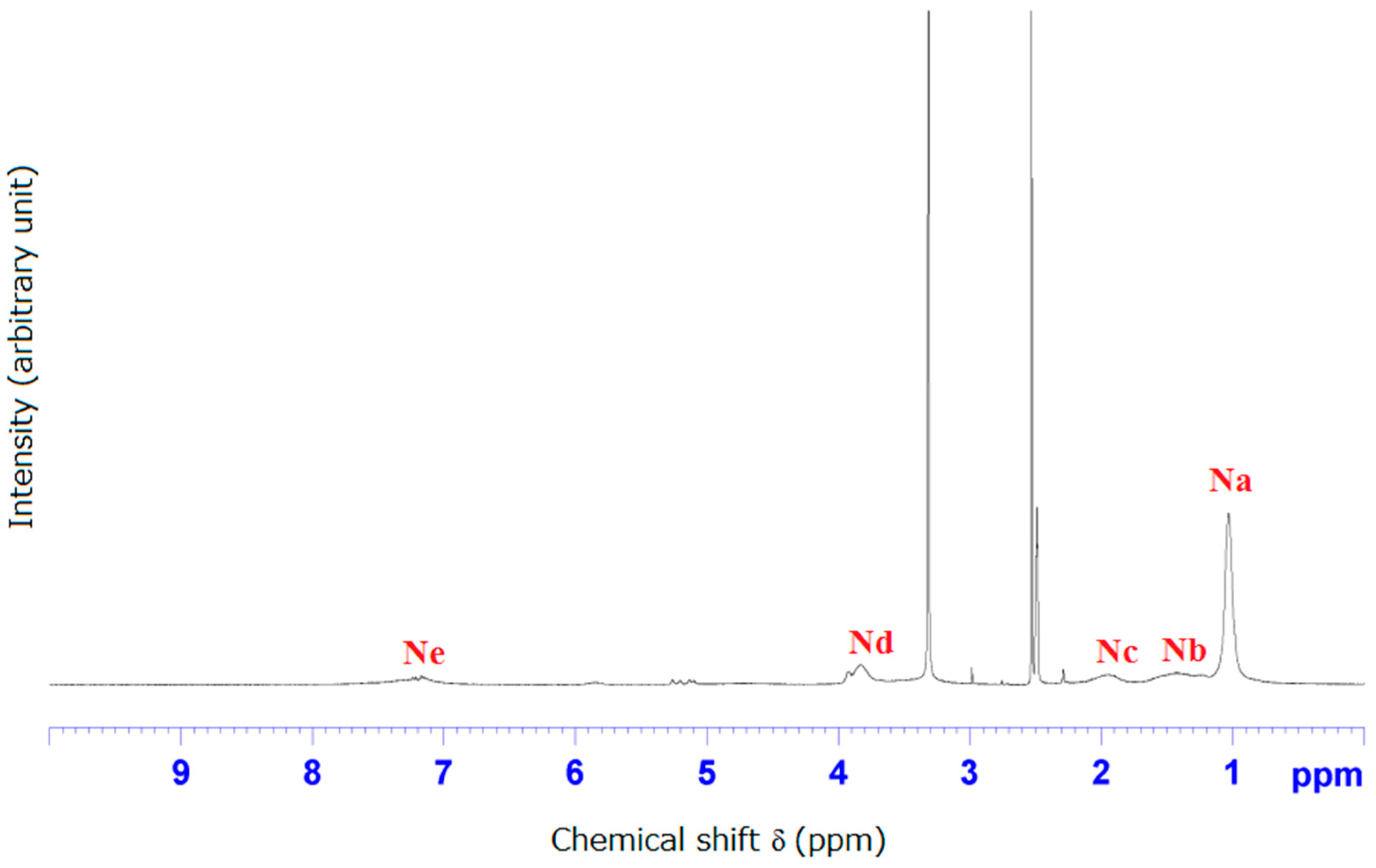
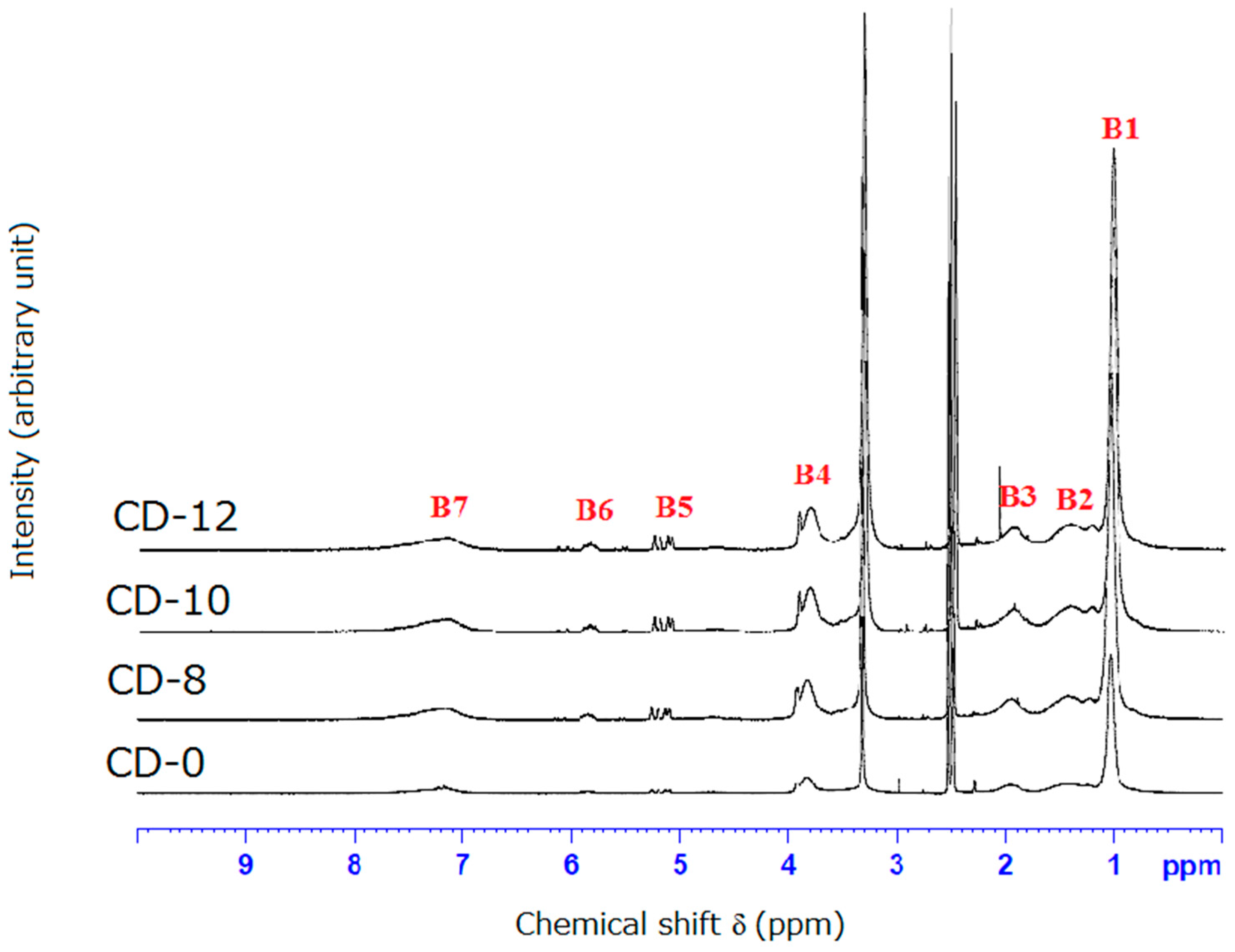
3.2. NMR and FTIR Results of Poly(CD-NIPAM) and Poly(CD-NIPAM-IAM) Copolymers (CD-0, CD-8, CD-10 and CD-12)
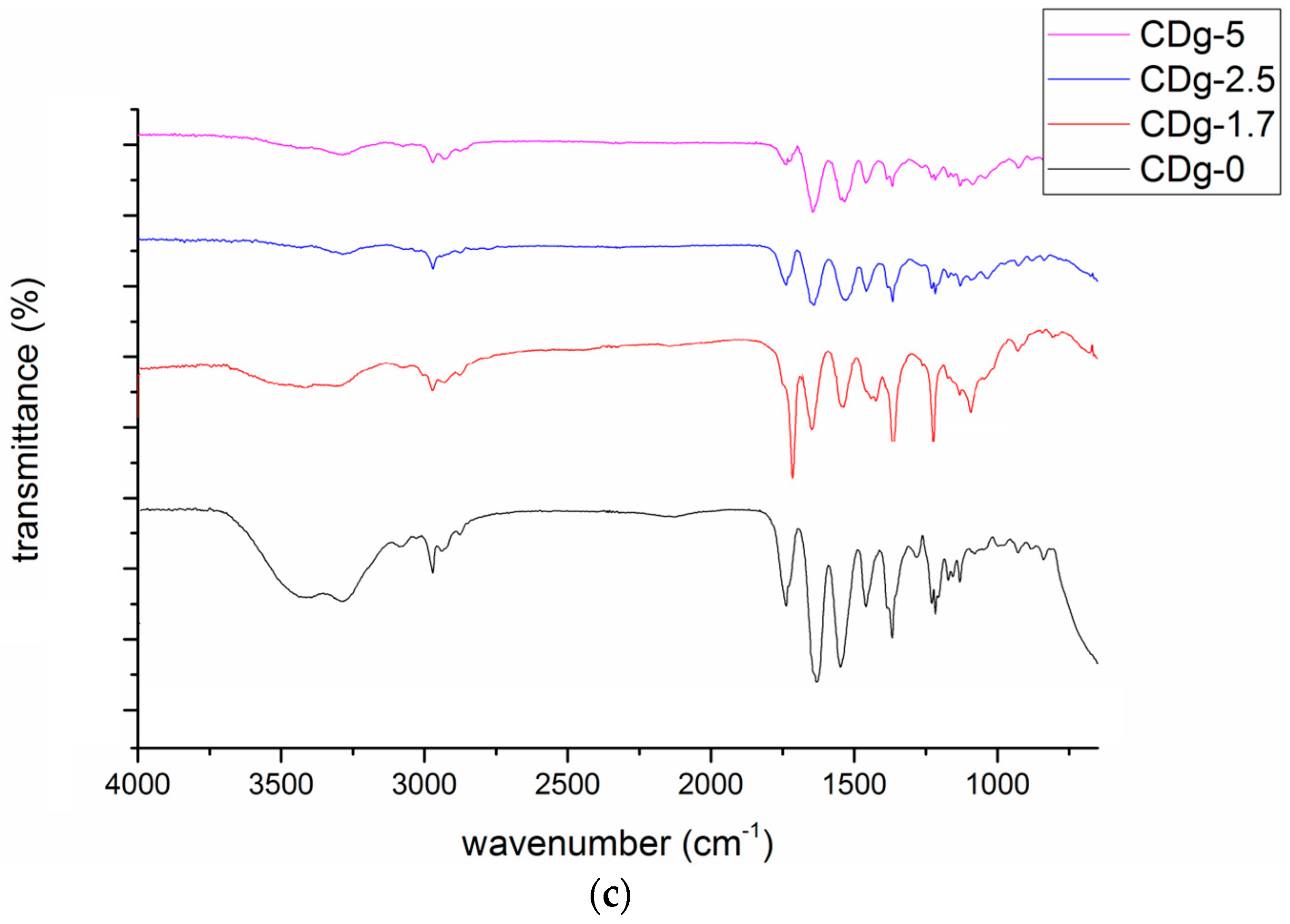
3.3. FTIR Results of Poly(CD-NIPAM) and Poly(CD-NIPAM-IAM) Copolymers
3.4. GPC Analysis
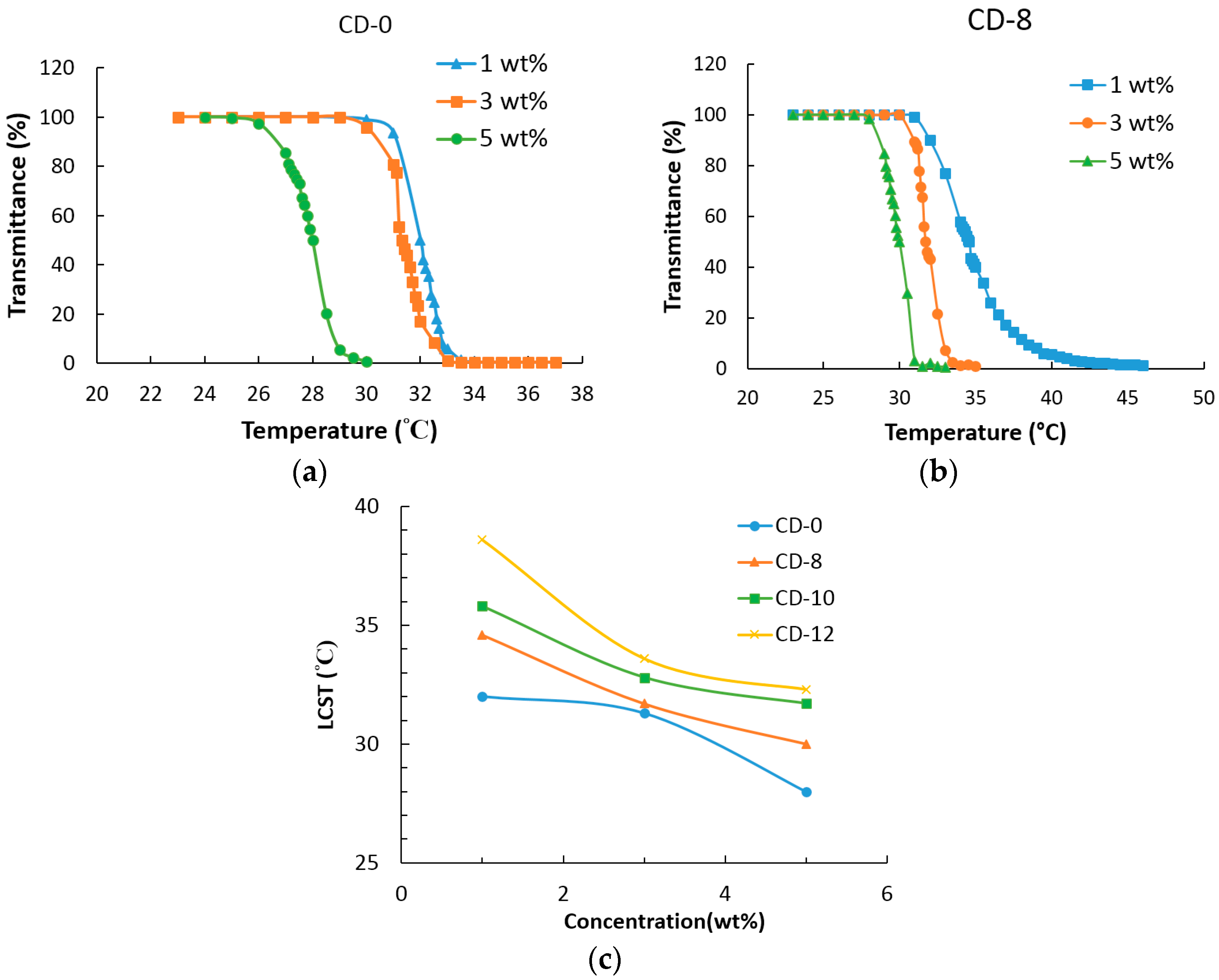
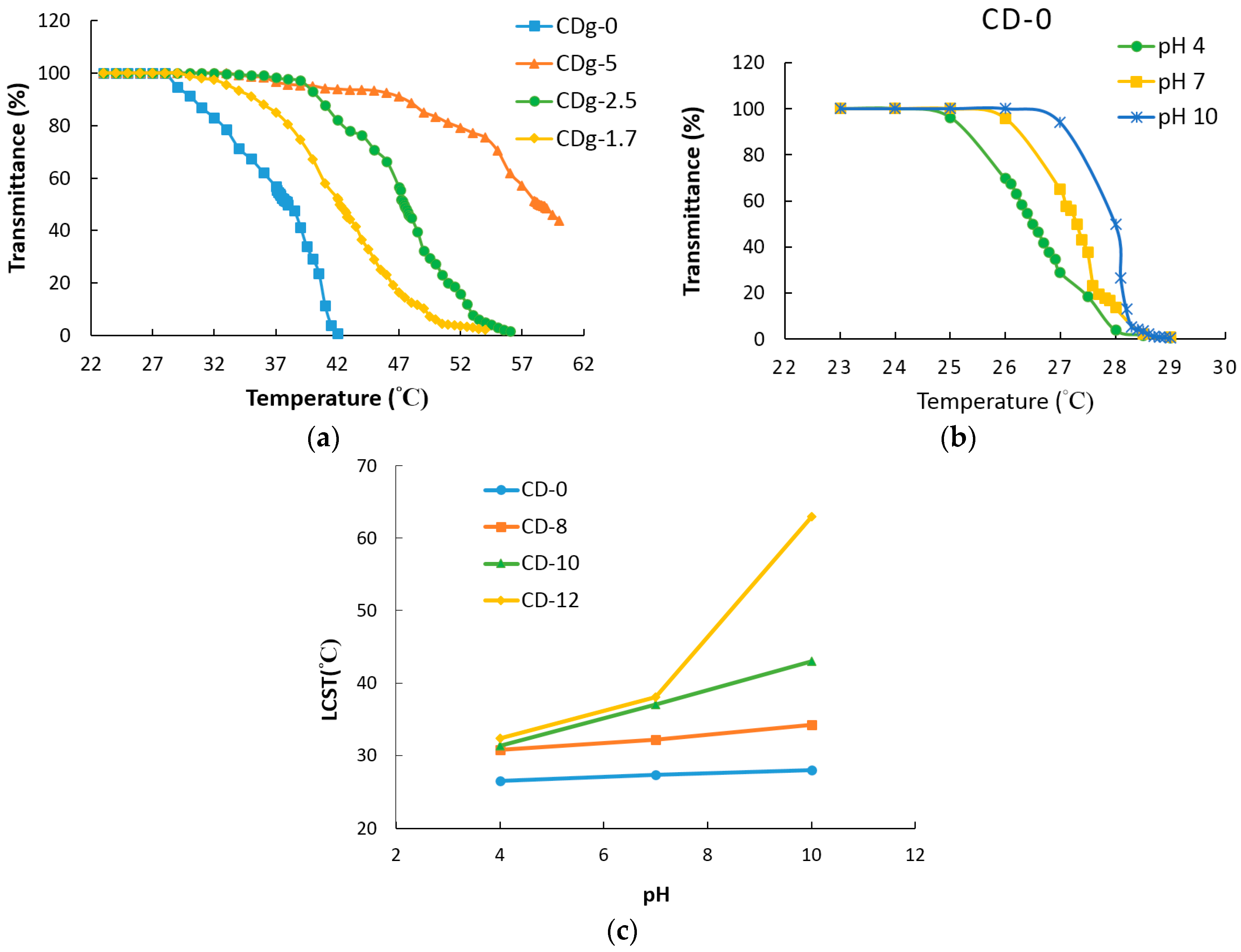
3.5. LCST Analysis
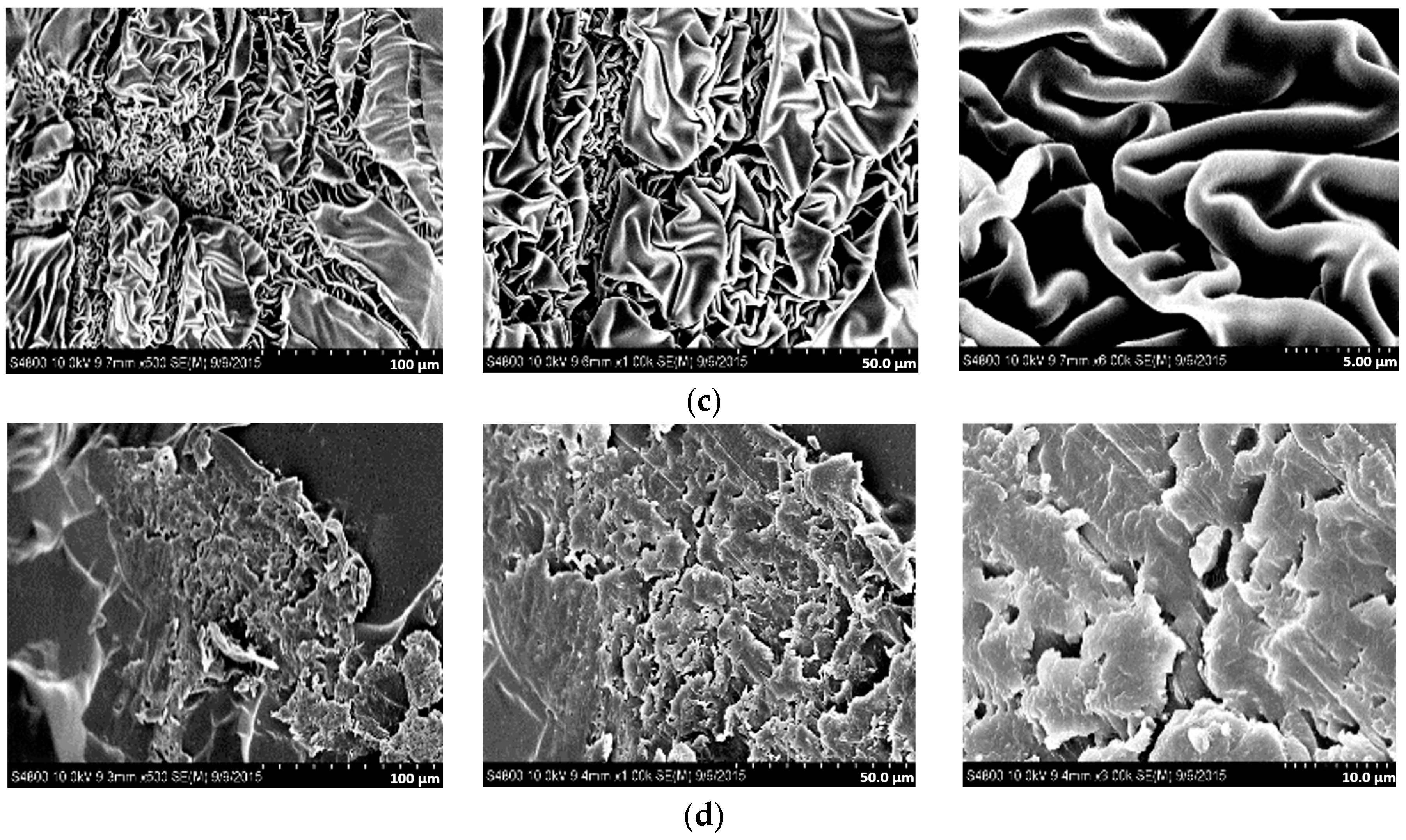
3.6. Morphological Analysis
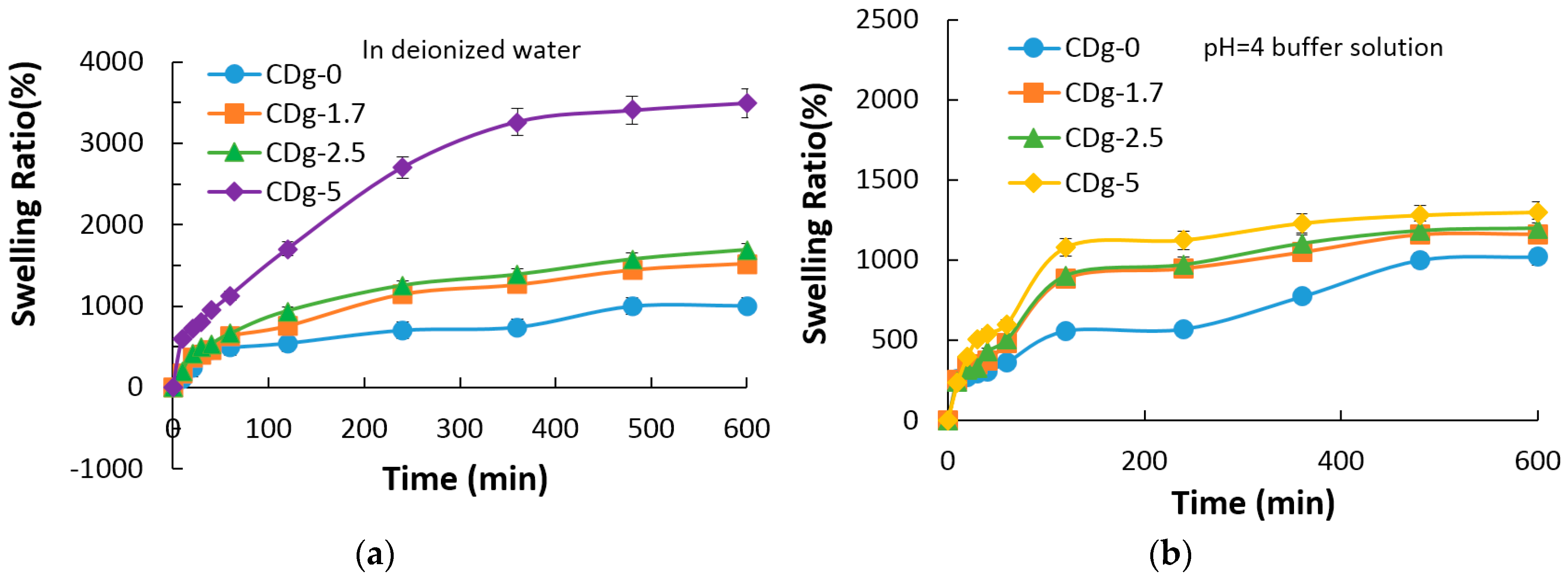
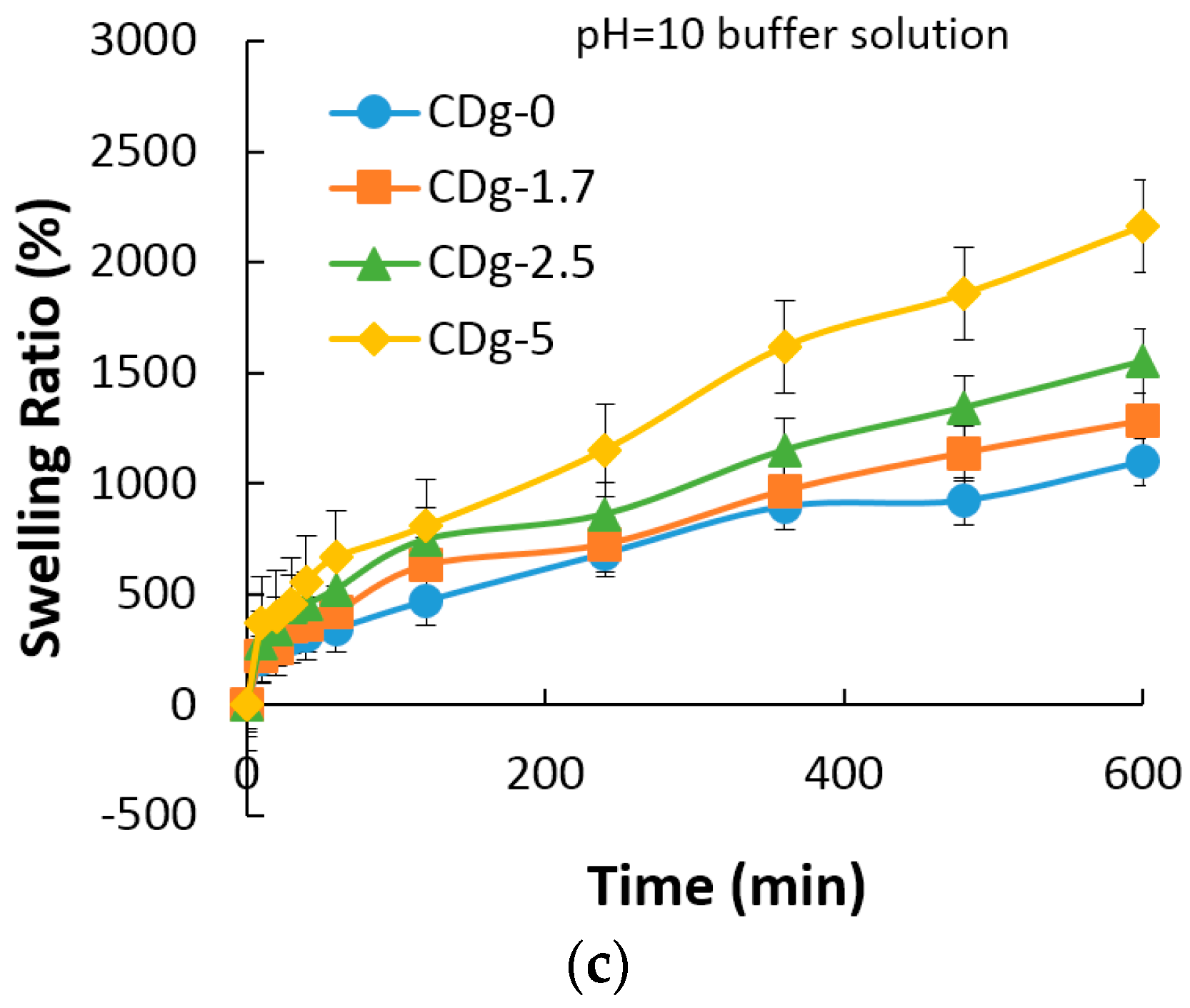
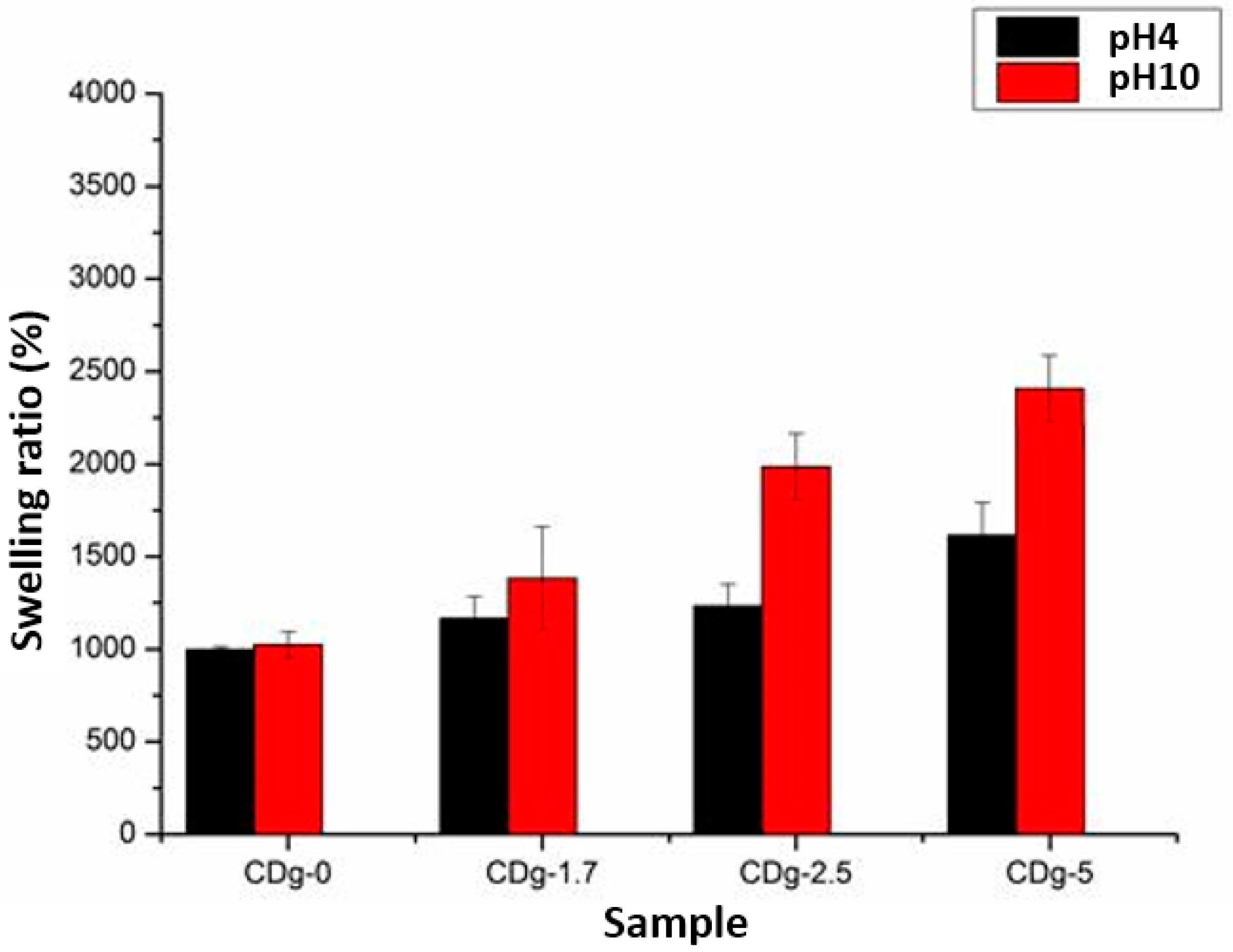
3.7. Swelling Property of Hydrogels
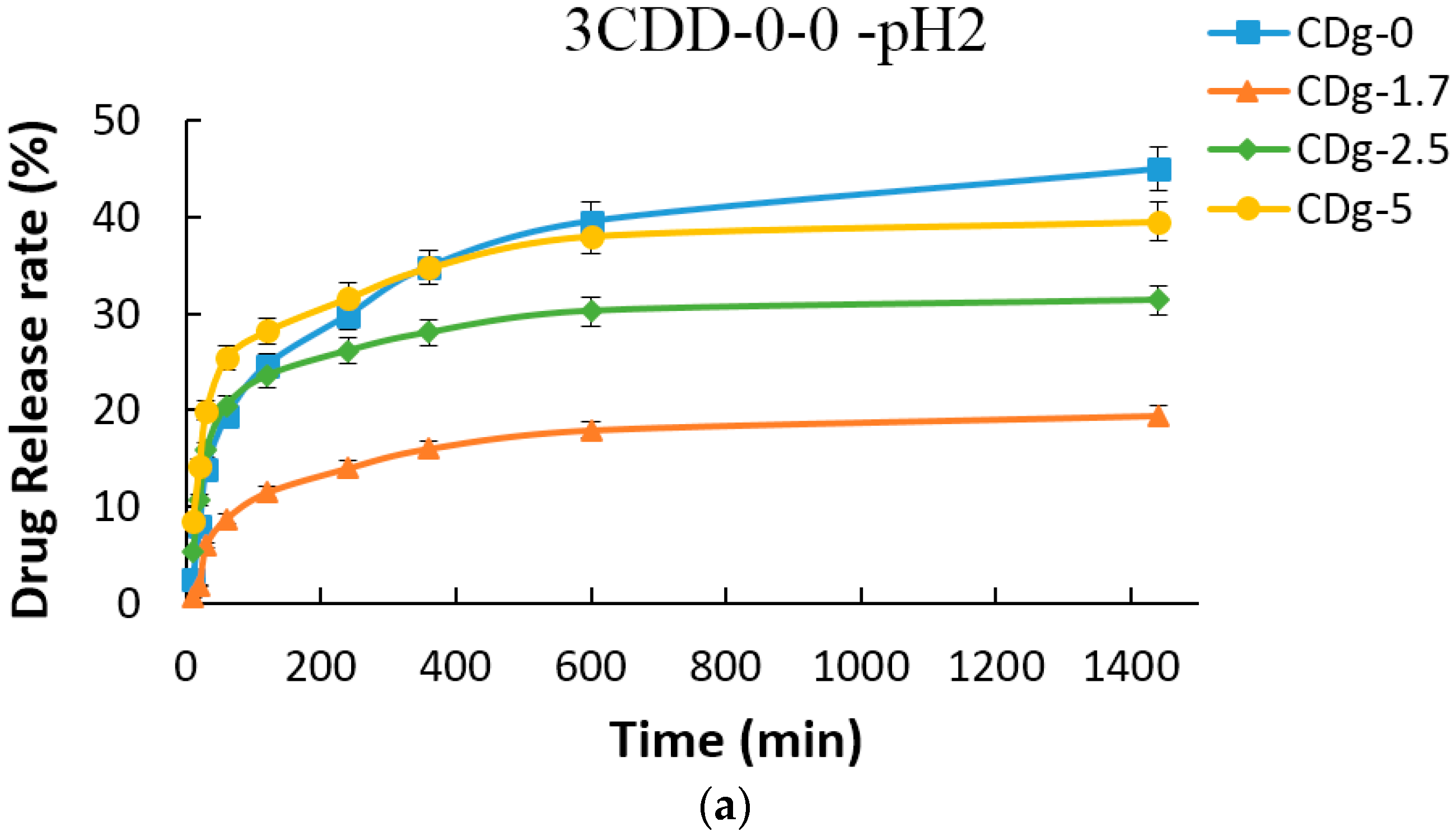
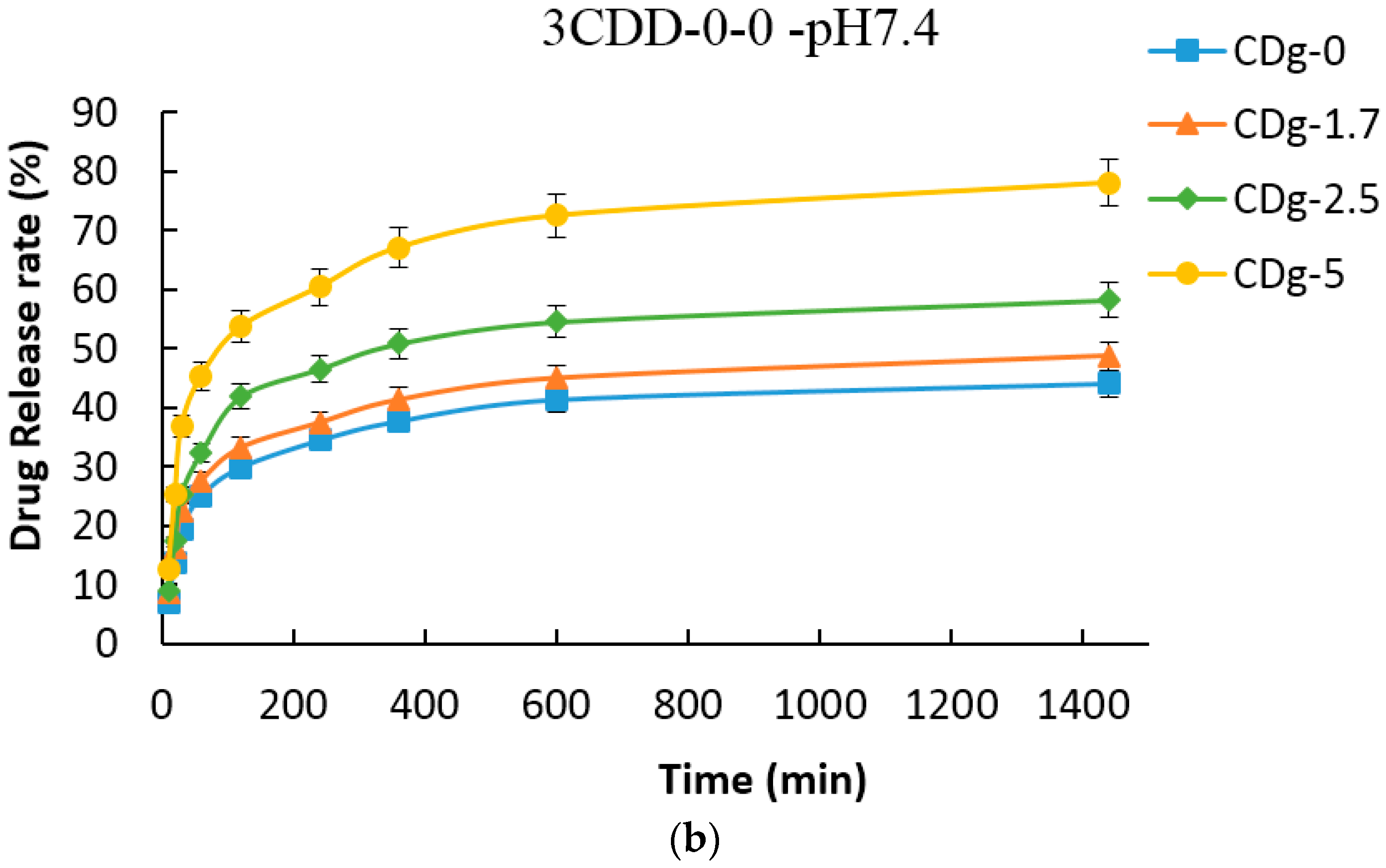
3.8. Drug Encapsulation and Release
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Aguilar, M.R.; Roman, J.S. Smart Polymers and Their Applications; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Kumar, A.; Srivastava, A.; Galaev, I.Y.; Mattiasson, B. Smart polymers: Physical forms and bioengineering applications. Prog. Polym. Sci. 2007, 32, 1203–1237. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, H.; Xue, H. Reversible pH stimulus-response material based on amphiphilic block polymer self-assembly and its electrochemical application. Materials 2016, 9, 478–489. [Google Scholar] [CrossRef]
- Fang, S.; Kawaguchi, H. A thermosensitive amphoteric microsphere and its potential application as a biological carrier. Colloid. Polym. Sci. 2002, 280, 984–989. [Google Scholar] [CrossRef]
- Cui, W.; Lu, X.; Cui, K.; Niu, L.; Wei, Y.; Lu, Q. Dual-responsive controlled drug delivery based on ionically assembled nanoparticles. Langmuir 2012, 28, 9413–9420. [Google Scholar] [CrossRef] [PubMed]
- Koch, B.; Rubino, I.; Quan, F.-S.; Yoo, B.; Choi, H.-J. Microfabrication for drug delivery. Materials 2016, 9, 646–682. [Google Scholar] [CrossRef]
- Constantin, M.; Bucatariu, S.; Ascenzi, P.; Simionescu, B.C.; Fundueanu, G. Poly(NIPAAm-co-β-cyclodextrin) microgels with drug hosting and temperature-dependent delivery properties. React. Funct. Polym. 2014, 84, 1–9. [Google Scholar] [CrossRef]
- Liu, T.-Y.; Hu, S.-H.; Liu, K.-H.; Liu, D.-M.; Chen, S.-Y. Preparation and characterization of smart magnetic hydrogels and its use for drug release. J. Magn. Magn. Mate. 2006, 304, e397–e399. [Google Scholar] [CrossRef]
- Hu, S.-H.; Liu, T.-Y.; Liu, D.-M.; Chen, S.-Y. Controlled pulsatile drug release from a ferrogel by a high-frequency magnetic field. Macromolecules 2007, 40, 6786–6788. [Google Scholar] [CrossRef]
- Kulkarni, R.V.; Biswanath, S. Electrically responsive smart hydrogels in drug delivery: A review. J. Appl. Biomater. Biomech. 2007, 5, 125–139. [Google Scholar] [PubMed]
- Qiu, Y.; Park, K. Environment-sensitive hydrogels for drug delivery. Adv. Drug Deliv. Rev. 2001, 53, 321–339. [Google Scholar] [CrossRef]
- Gupta, B.; Kumari, M.; Ikram, S. Drug release studies of N-isopropyl acrylamide/acrylic acid grafted polypropylene nonwoven fabric. J. Polym. Res. 2013, 20, 95–101. [Google Scholar] [CrossRef]
- Suzuki, A.; Tanaka, T. Phase transition in polymer gels induced by visible light. Nature 1990, 346, 345–347. [Google Scholar] [CrossRef]
- Mamada, A.; Tanaka, T.; Kungwatchakun, D.; Irie, M. Photoinduced phase transition of gels. Macromolecules 1990, 23, 1517–1519. [Google Scholar] [CrossRef]
- Cai, J.; Guo, J.; Ji, M.; Yang, W.; Wang, C.; Fu, S. Preparation and characterization of multiresponsive polymer composite microspheres with core–shell structure. Colloid. Polym. Sci. 2007, 285, 1607–1615. [Google Scholar] [CrossRef]
- Schild, H.G. Poly(N-isopropylacrylamide): Experiment, theory and application. Prog. Polym. Sci. 1992, 17, 163–249. [Google Scholar] [CrossRef]
- Yu, H.; Grainger, D.W. Thermo-sensitive swelling behavior in crosslinked N-isopropylacrylamide networks: Cationic, anionic, and ampholytic hydrogels. J. Appl. Polym. Sci. 1993, 49, 1553–1563. [Google Scholar] [CrossRef]
- Chun, S.-W.; Kim, J.-D. A novel hydrogel-dispersed composite membrane of poly(N-isopropylacrylamide) in a gelatin matrix and its thermally actuated permeation of 4-acetamidophen. J. Controll. Release 1996, 38, 39–47. [Google Scholar] [CrossRef]
- Ichikawa, H.; Fukumori, Y. A novel positively thermosensitive controlled-release microcapsule with membrane of nano-sized poly(N-isopropylacrylamide) gel dispersed in ethylcellulose matrix. J. Controll. Release 2000, 63, 107–119. [Google Scholar] [CrossRef]
- Lee, W.-F.; Shieh, C.-H. pH–thermoreversible hydrogels. II. Synthesis and swelling behaviors of N-isopropylacrylamide-co-acrylic acid-co-sodium acrylate hydrogels. J. Appl. Polym. Sci. 1999, 73, 1955–1967. [Google Scholar] [CrossRef]
- Liu, R.; Fraylich, M.; Saunders, B.R. Thermoresponsive copolymers: From fundamental studies to applications. Colloid Polym. Sci. 2009, 287, 627–643. [Google Scholar] [CrossRef]
- Rwei, S.-P.; Chuang, Y.-Y.; Way, T.-F.; Chiang, W.-Y. Thermosensitive copolymer synthesized by controlled-living radical polymerization: Phase behavior of diblockcopolymers of poly(N-isopropylacrylamide) families. J. Appl. Polym. Sci. 2016, 133, 43224. [Google Scholar] [CrossRef]
- Rwei, S.-P.; Way, T.-F.; Chang, S.-M.; Chiang, W.-Y.; Lien, Y.-Y. Thermo- and pH-responsive copolymers: Poly(N-isopropylacrylamide-co-IAM) copolymers. J. Appl. Polym. Sci. 2015, 132, 42367. [Google Scholar] [CrossRef]
- Rwei, S.-P.; Chuang, Y.-Y.; Way, T.-F.; Chiang, W.-Y.; Hsu, S.-P. Preparation of thermo- and pH-responsive star copolymers via ATRP and used in drug release application. Colloid Polym. Sci. 2015, 293, 493–503. [Google Scholar] [CrossRef]
- Rwei, S.-P.; Shu, K.-T.; Way, T.-F.; Chang, S.-M.; Chiang, W.-Y.; Pan, W.-C. Synthesis and characterization of hyperbranched copolymers hyper-g-(NIPAAm-co-IAM) via ATRP. Colloid Polym. Sci. 2016, 294, 291–301. [Google Scholar] [CrossRef]
- Zhao, C.; Gao, X.; He, P.; Xiao, C.; Zhuang, X.; Chen, X. Facile synthesis of thermo- and pH-responsive biodegradable microgels. Colloid Polym. Sci. 2011, 289, 447–451. [Google Scholar] [CrossRef]
- Manakker, F.; Vermonden, T.; Nostrum, C.; Hennink, W. Cyclodextrin-based polymeric materials: Synthesis, properties, and pharmaceutical/biomedical applications. Biomacromolecules 2009, 10, 3157–3175. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Ma, P. Cyclodextrin-based supramolecular systems for drug delivery: Recent progress and future perspective. Adv. Drug Deliv. Rev. 2013, 65, 1215–1233. [Google Scholar] [CrossRef] [PubMed]
- Way, T.-F.; Chen, Y.-T.; Chen, J.-J.; Teng, K. Copolymer and method for manufacturing the same. U.S. Patent Application No. 2013/0172490 A1, 4 July 2013. [Google Scholar]
- Schrage, S.; Sigel, R.; Schlaad, H. Formation of amphiphilic polyion complex vesicles from mixtures of oppositely charged block ionomers. Macromolecules 2003, 36, 1417–1420. [Google Scholar] [CrossRef]


| Sample | Mw | Mn | PDI (Mw/Mn) |
|---|---|---|---|
| CD-0 | 59,500 | 38,600 | 1.54 |
| CD-8 | 123,000 | 62,900 | 1.96 |
| CD-10 | 162,600 | 79,000 | 2.06 |
| CD-12 | 167,500 | 84,700 | 1.98 |
| Sample | 1 wt % | 3 wt % | 5 wt % | ΔLCST |
|---|---|---|---|---|
| CD-0 | 32.0 °C | 31.3 °C | 28.0 °C | 4 °C |
| CD-8 | 34.6 °C | 31.7 °C | 30.0 °C | 4.6 °C |
| CD-10 | 35.8 °C | 32.8 °C | 31.7 °C | 4.1 °C |
| CD-12 | 38.6 °C | 33.6 °C | 32.3 °C | 6.3 °C |
| Sample | NIPAM/IAM | LCST |
|---|---|---|
| CDg-0 | 100/0 | 38 °C |
| CDg-1.7 | 100/1.67 | 42.4 °C |
| CDg-2.5 | 100/2.5 | 47.4 °C |
| CDg-5 | 100/5 | 58.3 °C |
| Sample | pH 4 | pH 7 | pH 10 | ΔLCST |
|---|---|---|---|---|
| CD-0 | 26.5 °C | 27.3 °C | 28 °C | 1.5 °C |
| CD-8 | 30.8 °C | 32.3 °C | 34.2 °C | 3.4 °C |
| CD-10 | 31.3 °C | 37 °C | 43 °C | 11.7 °C |
| CD-12 | 32.4 °C | 38.1 °C | 63 °C | 30.6 °C |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rwei, S.-P.; Anh, T.H.N.; Chiang, W.-Y.; Way, T.-F.; Hsu, Y.-J. Synthesis and Drug Delivery Application of Thermo- and pH-Sensitive Hydrogels: Poly(β-CD-co-N-Isopropylacrylamide-co-IAM). Materials 2016, 9, 1003. https://doi.org/10.3390/ma9121003
Rwei S-P, Anh THN, Chiang W-Y, Way T-F, Hsu Y-J. Synthesis and Drug Delivery Application of Thermo- and pH-Sensitive Hydrogels: Poly(β-CD-co-N-Isopropylacrylamide-co-IAM). Materials. 2016; 9(12):1003. https://doi.org/10.3390/ma9121003
Chicago/Turabian StyleRwei, Syang-Peng, Tuan Huynh Nguyen Anh, Whe-Yi Chiang, Tun-Fun Way, and Yung-Jia Hsu. 2016. "Synthesis and Drug Delivery Application of Thermo- and pH-Sensitive Hydrogels: Poly(β-CD-co-N-Isopropylacrylamide-co-IAM)" Materials 9, no. 12: 1003. https://doi.org/10.3390/ma9121003
APA StyleRwei, S.-P., Anh, T. H. N., Chiang, W.-Y., Way, T.-F., & Hsu, Y.-J. (2016). Synthesis and Drug Delivery Application of Thermo- and pH-Sensitive Hydrogels: Poly(β-CD-co-N-Isopropylacrylamide-co-IAM). Materials, 9(12), 1003. https://doi.org/10.3390/ma9121003






