Abstract
Titanium carbide (TiC) is an important engineering material and has found widespread applications. Currently, TiC is typically synthesized through carbothermal reduction, requiring a high temperature (ca. 1700–2300 °C) and long reaction time (ca. 10–20 h), which is not eco-friendly. During a conventional reaction path, anatase TiO2 (A-TiO2) was first converted to rutile TiO2 (R-TiO2), which was subsequently reduced to TiC. Herein, we explored the synthesis of TiC powders with the assistance of microwave heating. In particular, we achieved the conversion of A-TiO2, which was more reactive than R-TiO2 for the carbothermal reduction, to TiC, which was directly due to quick microwave heating. As such, the carbothermal reduction started at a much lower temperature of ca. 1200 °C and finished within 30 min when reacting at 1400 °C, leading to significant energy saving. This study shows that microwave-assisted synthesis can be an effective and green process for preparing TiC powders, which is promising for future large-scale production. The influence of the reaction temperature, the reaction duration, and the carbon content on the synthesis of TiC powders was investigated.
1. Introduction
Titanium carbide (TiC) is an important engineering material because of its superior hardness, high melting temperature, good electrical conductivity, and outstanding abrasion resistance [1,2,3]. Therefore, TiC powders have been used extensively in cutting tools, grinding wheels, polishing pastes, etc. [3,4,5,6].
Currently, TiC powders are mainly synthesized by the carbothermal reduction of TiO2, and the heat during the process of reaction is usually provided by an external heating system. The synthesis is typically in the temperature range of 1700–2300 °C for 10–20 h [7]. The size of the synthesized TiC powders not only depends on the size of raw materials, but also on the synthesis conditions. Therefore, it is difficult to prepare uniform TiC powders by conventional heating processes [8,9].
As an alternative heating technology, microwave heating has advantages such as high thermal efficiency, selective heating, quick heating, and short processing time. It has been implemented in many industrial applications in recent years [10,11,12]. The major difference between microwave heating and conventional heating is the inverse temperature profile inside microwave-heated samples [13,14]. The center of the sample becomes hotter than the surface, which is exposed to the colder furnace atmosphere [15]. Many materials including ceramics, polymers, and metallic powders can be directly exposed to microwaves [16]. In microwave carbothermal reduction process, two factors might be favorable for lowering the onset reaction temperature. One is the existence of thermal and non-thermal effects from microwave heating [17]. The other is the uniform heating achieved by the energy directly delivered to the starting materials via molecular-level interactions under an electromagnetic field [18,19]. Microwave-assisted synthesis is attractive for the preparation of TiC because it is faster and more effective than conventional heating. Carbon, one of the reactants for the synthesis of TiC, is a very effective absorber of microwave radiation, offering extra facilitation of the reaction. Cross and coworkers [20,21] made some early explorations of the synthesis of TiC via microwave heating and proved that it was viable and indeed more effective to synthesize TiC via microwave heating.
In this report, microwave heating was adopted to synthesize TiC powders by carbothermal reduction of TiO2. The influence of reaction temperature and carbon content on the phase composition of the produced TiC powders was systematically investigated. In particular, we aimed to achieve the direct conversion of anatase TiO2 (A-TiO2), which was more reactive than rutile TiO2 (R-TiO2), for the carbothermal reduction to TiC by taking advantage of quick microwave heating.
2. Experimental
2.1. Materials
Both A-TiO2 powders (99%, ~40 nm in diameter) and carbon black (99%, ~10 nm in diameter) were purchased from Tuoyi Co. (Guangzhou, China). Ethanol was acquired from Fuyu Fine Chemical Co. (Fuzhou, China). Polyacrylic acid (PAA) was obtained from KeMiOU Chemical Co. (Tianjin, China). All the chemicals were used as received without further purification.
2.2. Synthesis
First, a 20.0 wt % A-TiO2 ethanol dispersion was ultrasonicated for 30 min to form a uniform system, which was mixed with polyacrylic acid (PAA/TiO2 = 0.5 wt %) to stabilize the dispersion of A-TiO2 in ethanol. Subsequently, a pre-determined amount of carbon black was mixed with the above dispersion. The reaction precursor was obtained after evaporating ethanol.
The synthesis was carried out in a KL-2D-16 microwave furnace (Kailin Microwave Equipment Co., Guangzhou, China). During reaction, the microwave frequency was 2450 ± 50 MHz, and microwave power was 6000 W. Thermocouples were used to measure the temperatures during reaction. The entire reaction was carried out under argon protection.
The carbothermal reduction of TiO2 is a complex process. Under a conventional heating process, before the reduction reaction, A-TiO2 is first converted to R-TiO2. With the rising of temperature, the reduction consists of a series of intermediate reactions, producing various intermediate products and phases (Equations (1)–(4)). The general procedures of the carbothermal reduction reaction between carbon black and R-TiO2 through conventional heating to generate TiC are as follows [22,23]:
Total reaction:
In this work, we aimed to achieve a direct reduction of A-TiO2 to TiC under the assistance of microwave heating, as illustrated in Figure 1. Such a direction reduction route is expected to occur at a lower temperature, thus saving energy and time. In order to investigate the reduction process of TiO2 by microwave heating, we monitored the phase transition of the reaction products at various temperatures and reaction durations by X-ray diffraction, and studied the influence of carbon content on the synthesis of the TiC powders.
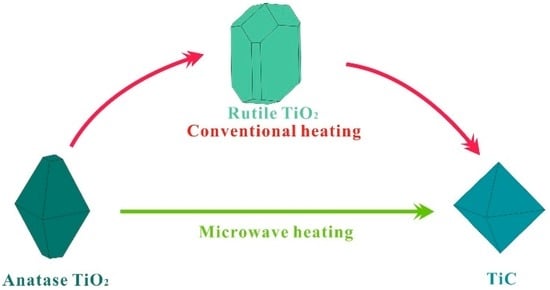
Figure 1.
Schematic of different reaction routes between conventional and microwave heating.
2.3. Characterization
Phase composition of the reaction products was determined by X-ray diffraction on a PANalytical X-ray diffractometer (XRD, monochromated Cu Kα radiation) at 25 °C. The morphology of the powders was imaged by scanning electron microscopy (SEM) on a Nova S-430 microscope operated at 20 kV, which was equipped with an energy-dispersive X-ray spectrometer (EDS).
3. Results and Discussion
3.1. Effect of Temperature and Reaction Time
The XRD patterns of the samples synthesized at various temperatures (1100, 1200, 1300, and 1400 °C) and durations of reaction (10 and 30 min) at a TiO2 and C molar ratio of 1:3.6 are presented in Figure 2.
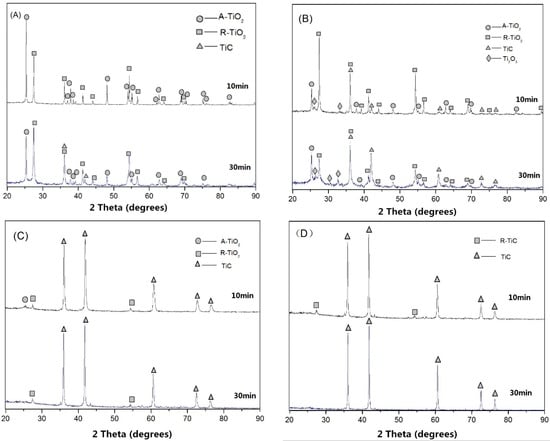
Figure 2.
X-ray diffractometer (XRD) patterns of the synthesized samples at various temperatures: (A) 1100 °C; (B) 1200 °C; (C) 1300 °C; (D) 1400 °C.
Figure 2A displays the XRD patterns of the samples reacted at 1100 °C for 10 and 30 min. It shows that TiC phase was not formed at 1100 °C for 10 min of reaction, but a small amount of TiC was generated after 30 min of reaction. The conversion from A-TiO2 to R-TiO2 was observed at this temperature. A number of studies reported that A-TiO2 began to transform to R-TiO2 at a temperature up to 610 °C [24,25]. With an increase in reaction time, more R-TiO2 was generated, as evidenced by stronger R-TiO2 phase diffraction peaks in the XRD pattern. The XRD patterns of the samples synthesized at 1200 °C (Figure 2B) showed that most A-TiO2 was converted to be R-TiO2; meanwhile, Ti2O3 started to form after 10 min of reaction at 1200 °C. After 30 min of reaction at 1200 °C, the TiC phase started to appear as evidenced by the corresponding XRD peaks. The TiC phase could be clearly observed on the XRD pattern when the reaction temperature was raised to 1300 °C. With an increase in reaction time, a higher concentration of TiC was generated, as supported by the more intensive diffraction peaks of TiC (Figure 2C). According to the XRD pattern, the virtually pure TiC phase was synthesized after 30 min of reaction at 1400 °C (Figure 2D).
Based on the above observations, the reaction mechanism is proposed as follows. At 1100 °C, the main reaction was the transformation from A-TiO2 to R-TiO2, but this is a relatively slow process:
It was reported that the activity of A-TiO2 with C was higher than that of R-TiO2 [26,27,28], so the rate of Reaction (6) is faster than that of Reaction (1):
During conventional heating, A-TiO2 tends to transform to R-TiO2 before the reduction reaction. This is one of the key reasons that a much higher temperature; thus, much more energy is required to convert the less active R-TiO2 to TiC. If one can quick heat to directly convert A-TiO2 to TiC, it is much more favorable in terms of energy consumption. Microwave heating can help achieve this process. Because the rate of microwave heating is very fast, it takes a very short time to raise the temperature from 1100 to 1200 °C. Therefore, when the temperature was quickly increased to 1200 °C, some A-TiO2 remained. Such unconverted A-TiO2 can quickly react with C to form TiC at relatively lower temperatures compared with the reduction reaction temperature of R-TiO2 to TiC. Figure 2C shows that, after 30 min of reaction at 1300 °C, there was still some R-TiO2 left but no A-TiO2. As such, a high heating rate is very desirable to directly reduce A-TiO2 to TiC, which allows for the synthesis of TiC at lower temperatures while saving energy. After 30 min of reaction at 1300 °C, most TiO2 was reacted to form TiC, which began to be the dominating phase.
With an increase in reaction temperature and time, it was observed that the (200) peak of the synthesized TiC shifted from 41.90° to 41.70° (Figure 3A,B). The lattice constant of the TiC (1300 °C, 10 min) and TiC (1400 °C, 30 min) were calculated to be 4.321 Å and 4.324 Å, lower than the standard value of 4.327 Å. The results show that a higher temperature and longer reaction time are beneficial for the growth of TiC, which was also reported by Preiss et al. [29,30].
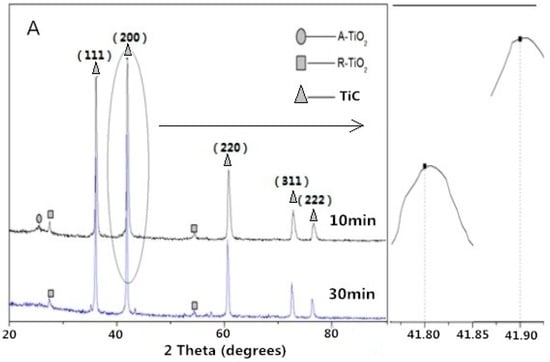
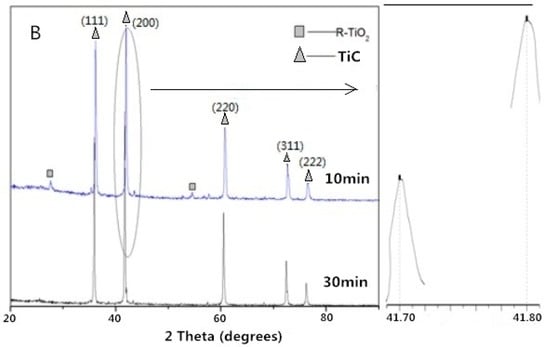
Figure 3.
XRD patterns of the samples synthesized at (A) 1300 °C and (B) 1400 °C.
SEM images of the samples synthesized at various temperatures (1100, 1200, 1300, and 1400 °C) for 30 min are shown in Figure 4. With an increase in reaction temperature, the size of the powders became larger, changing from ca. 0.2 μm at 1100 °C to ca. 0.4 μm at 1200 °C. When the reaction temperature was increased to 1300 °C, the TiC powders exhibited a pseudocubic morphology (Figure 4C) with a particle size of ca. 0.7 μm. It should be noted that, although no other diffraction peaks were observed in Figure 2D, amorphous carbon should still exist because excessive carbon was added to ensure a complete carbothermal reduction, because carbon black acts as both the carbon source and the media to transform microwave dielectric to heat.

Figure 4.
SEM images of the products synthesized at various temperatures for 30 min: (A) 1100 °C; (B) 1200 °C; (C) 1300 °C; (D) 1400 °C.
3.2. Effect of Carbon Content
TiO2 is a poor microwave absorber, while carbon black absorbs microwave very effectively. During the reaction, carbon black not only participates in the reaction but also serves as a medium to absorb microwave radiation to help heat the mixture of the reactants. Its content was reduced along the reaction; therefore, excessive carbon black was added during the reactions. In order to study the role of carbon content in the process of the reduction of TiO2, mixtures of TiO2 and C at various molar ratios (1:3.0; 1:3.2; 1:3.4, and 1:3.6) were reacted at 1400 °C for 30 min via microwave heating.
Table 1 presents the results calculated from the data of the XRD and EDS measurements of the products from the samples starting at various TiO2 and C mole ratios. The results showed that, with an increasing amount of carbon in the mixture, both the concentration and lattice constant of TiC increased, which suggests that a higher concentration of C is favorable for the growth of TiC.

Table 1.
Product composition after reaction at 1400 °C for 30 min under various TiO2 and C ratios.
4. Conclusions
In conclusion, our experimental results showed that pure and pseudocubic TiC phase could be synthesized by directly reducing A-TiO2 instead of R-TiO2 with the assistance of quick microwave heating. The lattice constant of TiC increases with an increasing reaction temperature and time. Carbon black acts as both the carbon source and the media to transform microwave dielectric to heat. A high ratio of C to TiO2 in the starting materials is favorable for improving the conversion rate and quality of TiC powders. Compared with the carbothermal reduction using conventional heating to synthesize TiC powders, which typically requires a temperature of 1700–2400 °C for 10 to 24 h, TiC powders could be synthesized at a temperature of as low as 1200 °C during microwave heating, and the carbothermal reduction can be finished within 30 min at 1400 °C. Therefore, the preparation of TiC powders via microwave heating is much more energy-effective and thus promising for large-scale production.
Acknowledgments
This work was financially supported by the National High-tech R&D Program (863 Program) of China and the Science and Technology Planning Project of Guangdong Province, China.
Author Contributions
Hui Wang and Lingke Zeng conceived and designed the experiments; Wencheng Zhu performed the experiments; Lingke Zeng and Luyi Sun analyzed the data; Yanchun Liu contributed in experiments and characterizations; Wang Hui and Luyi Sun wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Shin, Y.; Li, X.S.; Wang, C.; Coleman, J.R.; Exarhos, G.J. Synthesis of hierarchical titanium carbide from titania-coated cellulose paper. Adv. Mater. 2004, 16, 1212–1215. [Google Scholar] [CrossRef]
- Pierson, H.O. Handbook of Refractory Carbides & Nitrides: Properties, Characteristics, Processing and Apps; William Andrew: Norwich, NY, USA, 1996. [Google Scholar]
- Toth, L. Transition Metal Carbides and Nitrides; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Koc, R. Kinetics and phase evolution during carbothermal synthesis of titanium carbide from ultrafine titania/carbon mixture. J. Mater. Sci. 1998, 33, 1049–1055. [Google Scholar] [CrossRef]
- El-Eskandarany, M.S. Structure and properties of nanocrystalline TiC full-density bulk alloy consolidated from mechanically reacted powders. J. Alloys Compd. 2000, 305, 225–238. [Google Scholar] [CrossRef]
- Ushakov, A.; Karpov, I.; Lepeshev, A.; Krushenko, G. Physicochemical properties of nanomodifiers based on electric arc titanium nitride powder for polymer nanocomposite materials. Tekhnol. Met. 2011, 3, 16–21. [Google Scholar]
- Kappe, C.; Dallinger, D.; Murphree, S. Practical Microwave Synthesis for Organic Chemists: Strategies, Instruments, and Protocols; Wiley VCH Verlag GmbH & Co., KGaA: Weinheim, Germany, 2009. [Google Scholar]
- Choi, Y.; Rhee, S.-W. Effect of aluminium addition on the combustion reaction of titanium and carbon to form TiC. J. Mater. Sci. 1993, 28, 6669–6675. [Google Scholar] [CrossRef]
- Koc, R.; Folmer, J. Carbothermal synthesis of titanium carbide using ultrafine titania powders. J. Mater. Sci. 1997, 32, 3101–3111. [Google Scholar] [CrossRef]
- Thakur, S.K.; Kong, T.S.; Gupta, M. Microwave synthesis and characterization of metastable (Al/Ti) and hybrid (Al/Ti + SiC) composites. Mater. Sci. Eng. A 2007, 452, 61–69. [Google Scholar] [CrossRef]
- Rajkumar, K.; Aravindan, S. Microwave sintering of copper-graphite composites. J. Mater. Process. Technol. 2009, 209, 5601–5605. [Google Scholar] [CrossRef]
- Leparoux, S.; Vaucher, S.; Beffort, O. Assessment of microwave heating for sintering of Al/SiC and for in-situ synthesis of TiC. Adv. Eng. Mater. 2003, 5, 449–453. [Google Scholar] [CrossRef]
- Campanone, L.; Zaritzky, N. Mathematical analysis of microwave heating process. J. Food Eng. 2005, 69, 359–368. [Google Scholar] [CrossRef]
- Ciacci, T.; Galgano, A.; Di Blasi, C. Numerical simulation of the electromagnetic field and the heat and mass transfer processes during microwave-induced pyrolysis of a wood block. Chem. Eng. Sci. 2010, 65, 4117–4133. [Google Scholar] [CrossRef]
- Rosa, R.; Veronesi, P.; Leonelli, C. A review on combustion synthesis intensification by means of microwave energy. Chem. Eng. Process. Process Intensif. 2013, 71, 2–18. [Google Scholar] [CrossRef]
- Mishra, R.R.; Sharma, A.K. Microwave–material interaction phenomena: Heating mechanisms, challenges and opportunities in material processing. Compos. A Appl. Sci. Manuf. 2016, 81, 78–97. [Google Scholar] [CrossRef]
- Porcelli, M.; Cacciapuoti, G.; Fusco, S.; Massa, R.; d’Ambrosio, G.; Bertoldo, C.; De Rosa, M.; Zappia, V. Non-thermal effects of microwaves on proteins: Thermophilic enzymes as model system. FEBS Lett. 1997, 402, 102–106. [Google Scholar] [CrossRef]
- Moshtaghioun, B.; Poyato, R.; Cumbrera, F.; de Bernardi-Martin, S.; Monshi, A.; Abbasi, M.; Karimzadeh, F.; Dominguez-Rodriguez, A. Rapid carbothermic synthesis of silicon carbide nano powders by using microwave heating. J. Eur. Ceram. Soc. 2012, 32, 1787–1794. [Google Scholar] [CrossRef]
- Zhang, H.; Li, F.; Jia, Q.; Ye, G. Preparation of titanium carbide powders by sol-gel and microwave carbothermal reduction methods at low temperature. J. Sol-Gel Sci. Technol. 2008, 46, 217–222. [Google Scholar] [CrossRef]
- Binner, J.G.P.; Hassine, N.A.; Cross, T.E. The possible role of the pre-exponential factor in explaining the increased reaction rates observed during the microwave synthesis of titanium carbide. J. Mater. Sci. 1995, 30, 5389–5393. [Google Scholar] [CrossRef]
- Hassine, N.A.; Binner, J.G.P.; Cross, T.E. Synthesis of refractory metal carbide powders via microwave carbothermal reduction. Int. J. Refract. Met. Hard Mater. 1995, 13, 353–358. [Google Scholar] [CrossRef]
- Holt, J.; Munir, Z. Combustion synthesis of titanium carbide: Theory and experiment. J. Mater. Sci. 1986, 21, 251–259. [Google Scholar] [CrossRef]
- Zeng, L.K.; Liu, Y.C.; Zhu, W.C.; Liu, P.A.; Wang, H.; Cheng, X.S.; Liang, Q.Y. Investigation on the continuous microwave synthesis of nano titanium carbide powder. Adv. Mater. Res. 2014, 1064, 66–70. [Google Scholar] [CrossRef]
- Chen, Y.-F.; Lee, C.-Y.; Yeng, M.-Y.; Chiu, H.-T. The effect of calcination temperature on the crystallinity of TiO2 nanopowders. J. Cryst. Growth 2003, 247, 363–370. [Google Scholar] [CrossRef]
- Colmenares, J.; Aramendia, M.; Marinas, A.; Marinas, J.; Urbano, F. Synthesis, characterization and photocatalytic activity of different metal-doped titania systems. Appl. Catal A Gen. 2006, 306, 120–127. [Google Scholar] [CrossRef]
- Lucarelli, L.; Nadtochenko, V.; Kiwi, J. Environmental photochemistry: Quantitative adsorption and FTIR studies during the TiO2-photocatalyzed degradation of orange II. Langmuir 2000, 16, 1102–1108. [Google Scholar] [CrossRef]
- Diamandescu, L.; Vasiliu, F.; Tarabasanu-Mihaila, D.; Feder, M.; Vlaicu, A.; Teodorescu, C.; Macovei, D.; Enculescu, I.; Parvulescu, V.; Vasile, E. Structural and photocatalytic properties of iron-and europium-doped TiO2 nanoparticles obtained under hydrothermal conditions. Mater. Chem. Phys. 2008, 112, 146–153. [Google Scholar] [CrossRef]
- Jung, Y.-S.; Kim, D.-W.; Kim, Y.-S.; Park, E.-K.; Baeck, S.-H. Synthesis of alumina-titania solid solution by sol-gel method. J. Phys. Chem. Solids 2008, 69, 1464–1467. [Google Scholar] [CrossRef]
- Preiss, H.; Berger, L.-M.; Schultze, D. Studies on the carbothermal preparation of titanium carbide from different gel precursors. J. Eur. Ceram. Soc. 1999, 19, 195–206. [Google Scholar] [CrossRef]
- Cao, Y.; Zhang, H.; Li, F.; Lu, L.; Zhang, S. Preparation and characterization of ultrafine ZrB2–SiC composite powders by a combined sol-gel and microwave boro/carbothermal reduction method. Ceram. Int. 2015, 41, 7823–7829. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).