Determination of Insulator-to-Semiconductor Transition in Sol-Gel Oxide Semiconductors Using Derivative Spectroscopy
Abstract
:1. Introduction
2. Results and Discussion
2.1. Insulator-to-Semiconductor Transition of Sol-Gel Oxide Semiconductors
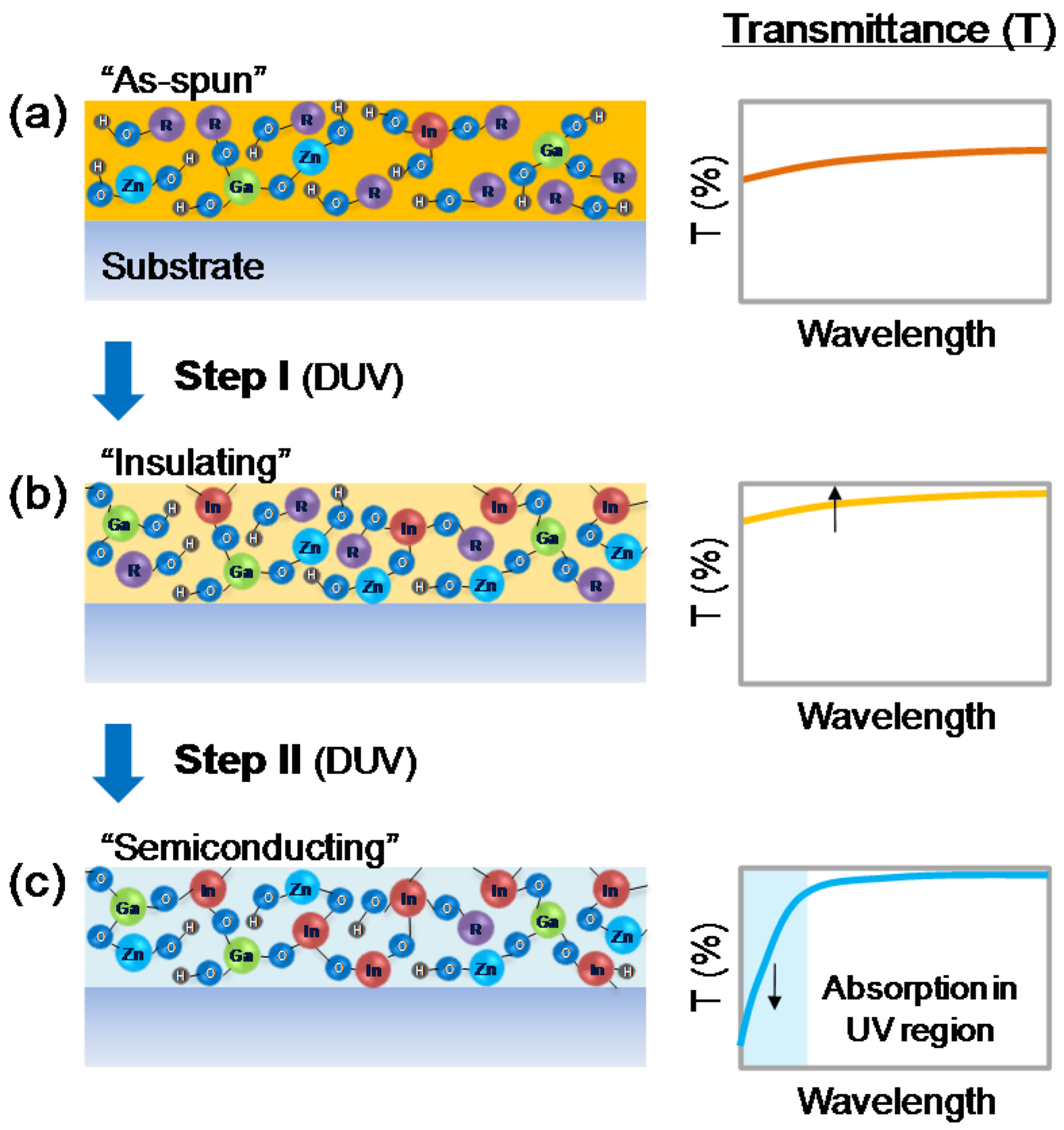
2.2. Determination of Insulator-to-Semiconductor Transition Using Derivative Spectroscopy
2.2.1. Optical Characteristics of Sol-Gel Oxide Films
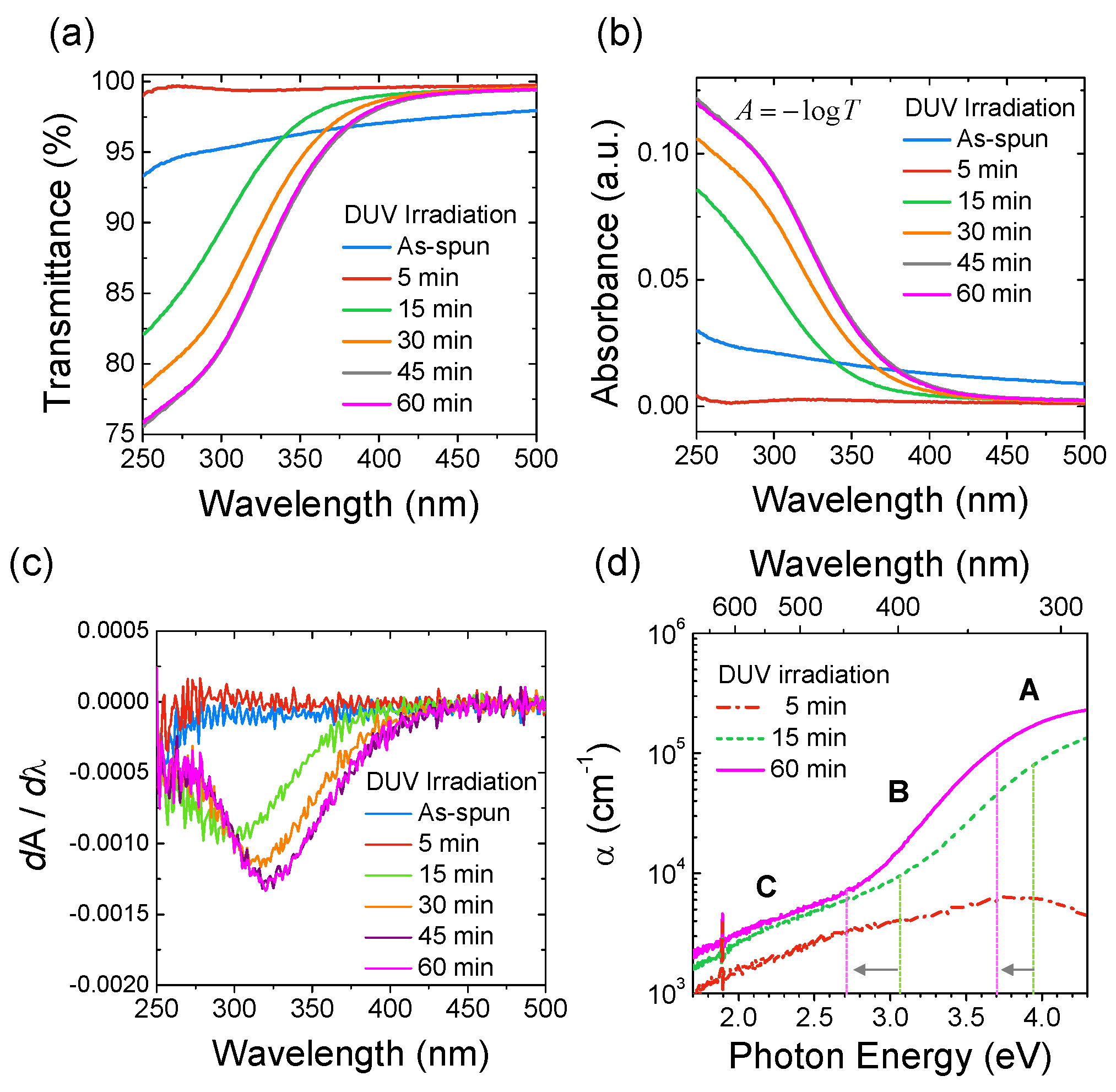
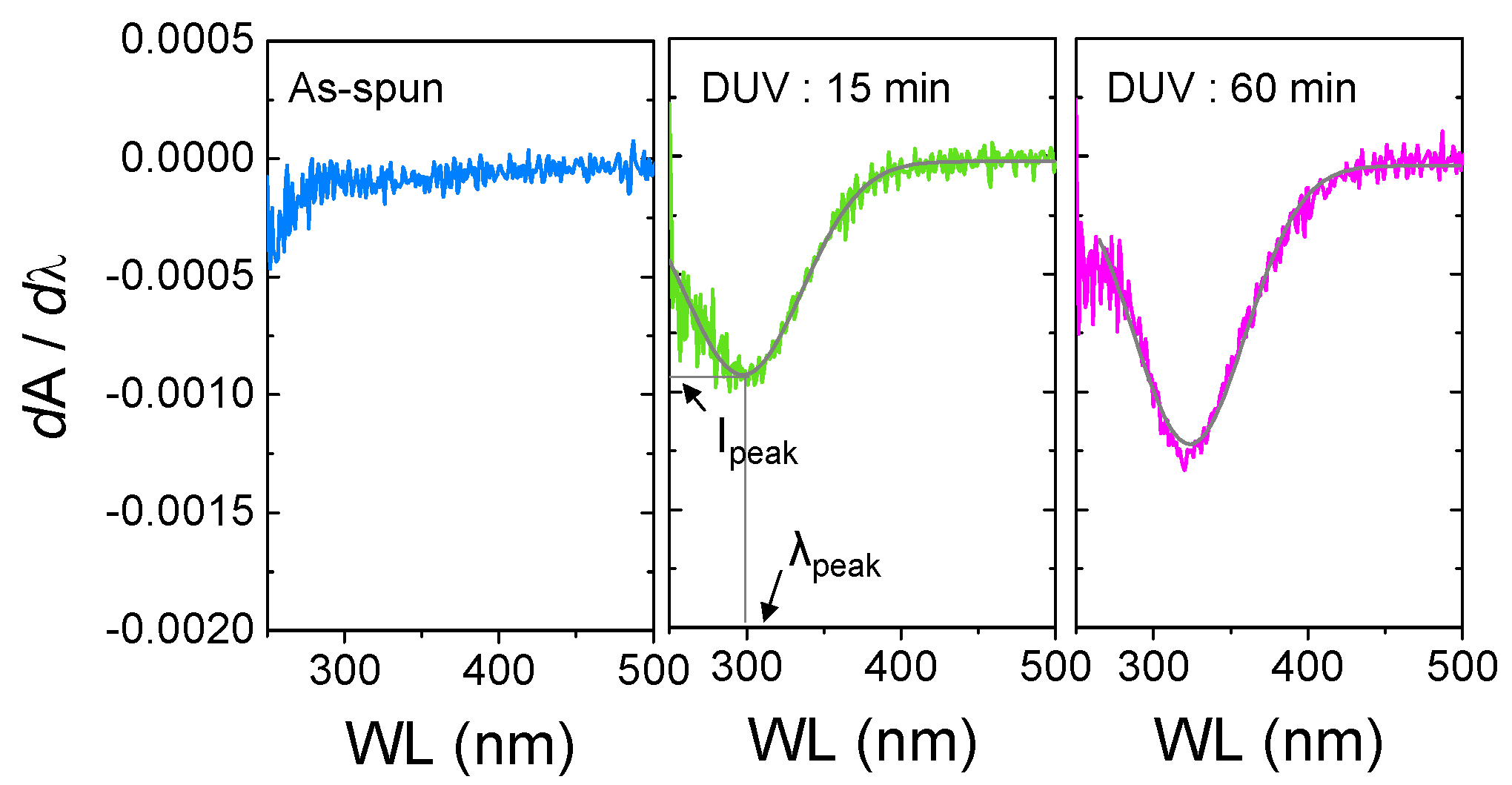
2.2.2. Extraction of Peak Center (λpeak) and Peak Intensity (Ipeak)
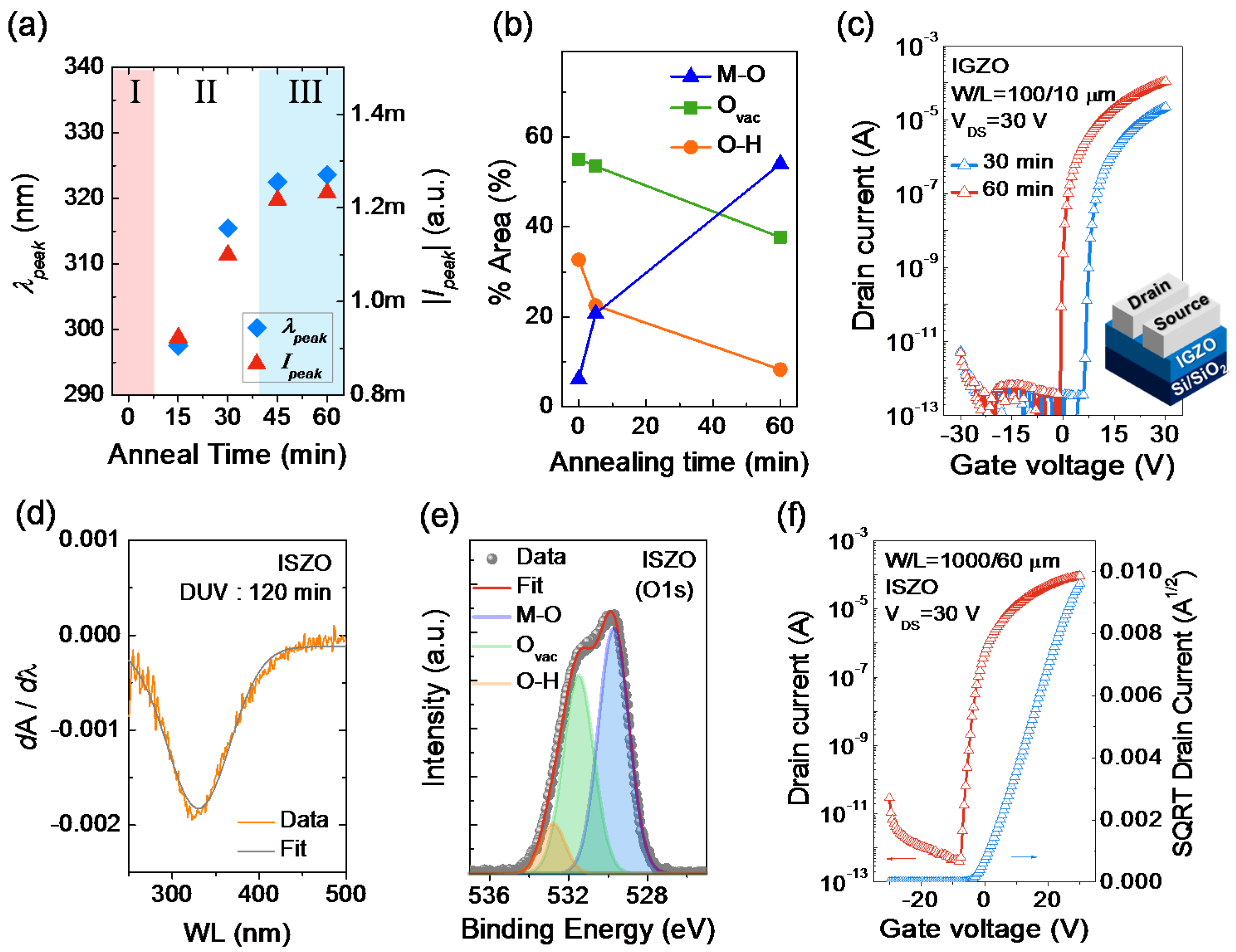
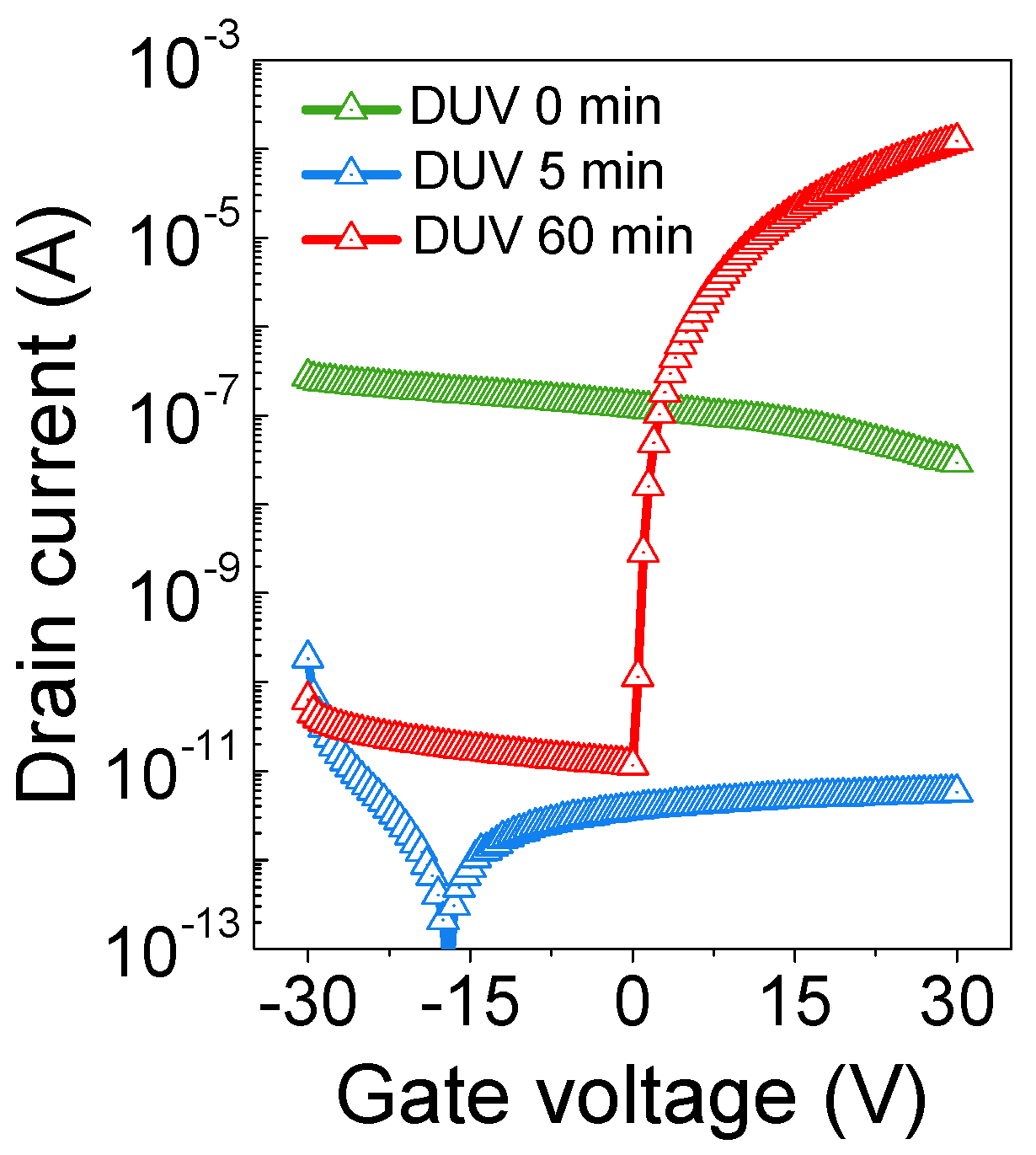
2.2.3. Correlation with TFT Performance
3. Materials and Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lin, Y.H.; Faber, H.; Zhao, K.; Wang, Q.; Amassian, A.; McLachlan, M.; Anthopoulos, T.D. High-performance ZnO transistors processed via an aqueous carbon-free metal oxide precursor route at temperatures between 80–180 °C. Adv. Mater. 2013, 25, 4340–4346. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Liu, A.; Zhu, H.; Shin, B.; Fortunato, E.; Martins, R.; Wang, Y.; Shan, F. Low-temperature, nontoxic water-induced metal-oxide thin films and their application in thin-film transistors. Adv. Funct. Mater. 2015, 25, 2564–2572. [Google Scholar] [CrossRef]
- Seo, J.S.; Jeon, J.H.; Hwang, Y.H.; Park, H.; Ryu, M.; Park, S.H.K.; Bae, B.S. Solution-processed flexible fluorine-doped indium zinc oxide thin-film transistors fabricated on plastic film at low temperature. Sci. Rep. 2013, 3, 2085:1–2085:9. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.T.; Park, J.; Ahn, B.D.; Kim, D.M.; Choi, S.J.; Kim, H.S.; Kim, D.H. Effect of direct current sputtering power on the behavior of amorphous indium-gallium-zinc-oxide thin-film transistors under negative bias illumination stress: A combination of experimental analyses and device simulation. Appl. Phys. Lett. 2015, 106, 123505:1–123505:5. [Google Scholar] [CrossRef]
- Chiang, H.Q.; McFarlane, B.R.; Hong, D.; Presley, R.E.; Wager, J.F. Processing effects on the stability of amorphous indium gallium zinc oxide thin-film transistors. J. Non-Cryst. Solids 2008, 354, 2826–2830. [Google Scholar] [CrossRef]
- Rim, Y.S.; Chen, H.; Liu, Y.; Bae, S.H.; Kim, H.J.; Yang, Y. Direct light pattern integration of low-temperature solution-processed all-oxide flexible electronics. ACS Nano 2014, 8, 9680–9686. [Google Scholar] [CrossRef] [PubMed]
- Street, R.A.; Ng, T.N.; Lujan, R.A.; Son, I.; Smith, M.; Kim, S.; Lee, T.; Moon, Y.; Cho, S. Sol-gel solution-deposited InGaZnO thin film transistors. ACS Appl. Mater. Interfaces 2014, 6, 4428–4437. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.J.; Yeh, Y.H.; Cheng, C.C.; Lin, C.Y.; Ho, G.T.; Lai, B.C.M.; Leu, C.M.; Hou, T.H.; Chan, Y.J. Amorphous InGaZnO thin-film transistors compatible with roll-to-roll fabrication at room temperature. IEEE Electron Dev. Lett. 2012, 33, 47–49. [Google Scholar] [CrossRef]
- Lin, Y.H.; Faber, H.; Labram, J.G.; Stratakis, E.; Sygellou, L.; Kymakis, E.; Hastas, N.A.; Li, R.; Zhao, K.; Amassian, A.; et al. High electron mobility thin-film transistors based on solution-processed semiconducting metal oxide heterojunctions and quasi-superlattices. Adv. Sci. 2015, 2, 1500058:1–1500058:12. [Google Scholar] [CrossRef]
- Ahn, C.H.; Karuppanan, S.; Cho, H.K.; Lee, S.Y. Artificial semiconductor/insulator superlattice channel structure for high-performance oxide thin-film transistors. Sci. Rep. 2013, 3, 2737:1–2737:6. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.H.; Ahn, B.D.; Shin, H.S.; Jeong, W.H.; Kim, H.J. Effect of indium composition ratio on solution-processed nanocrystalline InGaZnO thin film transistors. Appl. Phys. Lett. 2009, 94, 233501:1–233501:3. [Google Scholar] [CrossRef]
- Kim, Y.H.; Heo, J.S.; Kim, T.H.; Park, S.; Yoon, M.H.; Kim, J.; Oh, M.S.; Yi, G.R.; Noh, Y.Y.; Park, S.K. Flexible metal-oxide devices made by room-temperature photochemical activation of sol-gel films. Nature 2012, 489, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Moon, J. Low-temperature, solution-processed metal oxide thin film transistors. J. Mater. Chem. 2012, 22, 1243–1250. [Google Scholar] [CrossRef]
- Fung, T.C.; Chuang, C.S.; Chen, C.; Abe, K.; Cottle, R.; Townsend, M.; Kumomi, H.; Kanicki, J. Two-dimensional numerical simulation of radio frequency sputter amorphous In-Ga-Zn-O thin-film transistors. J. Appl. Phys. 2009, 106, 084511:1–084511:10. [Google Scholar] [CrossRef]
- Rim, Y.S.; Chen, H.; Kou, X.; Duan, H.S.; Zhou, H.; Cai, M.; Kim, H.J.; Yang, Y. Boost up mobility of solution-processed metal oxide thin-film transistors via confining structure on electron pathways. Adv. Mater. 2014, 26, 4273–4278. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Li, X.Y.; Zou, Z.X.; Lin, L.R.; Li, Y.Q. A novel baseline-correction method for standard addition based derivative spectra and its application to quantitative analysis of benzo(a)pyrene in vegetable oil samples. Analyst 2011, 136, 2802–2810. [Google Scholar] [CrossRef] [PubMed]
- Winer, K.; Ley, L. Surface states and the exponential valence-band tail in a-Si:H. Phys. Rev. 1987, 36, 6072–6073. [Google Scholar] [CrossRef]
- Tiedje, T.; Cebulla, H.J.M.; Morel, D.L. Evidence for exponential band tails in amorphous silicon hydride. Phys. Rev. Lett. 1981, 46, 1425–1426. [Google Scholar] [CrossRef]
- Ishida, T.; Kobayashi, H.; Nakako, Y. Structures and properties of electron-beam-evaporated indium tin oxide films as studied by X-ray photoelectron spectroscopy and work-function measurements. J. Appl. Phys. 1993, 73, 4344–4350. [Google Scholar] [CrossRef]
- Seo, S.J.; Hwang, Y.H.; Bae, B.S. Postannealing process for low temperature processed sol-gel zinc tin oxide thin film transistors. Electrochem. Solid-State Lett. 2010, 13, H357–H359. [Google Scholar] [CrossRef]
- Park, S.; Kim, K.H.; Jo, J.W.; Sung, S.; Kim, K.T.; Lee, W.J.; Kim, J.; Kim, H.J.; Yi, G.R.; Kim, Y.H.; et al. In-depth studies on rapid photochemical activation of various sol-gel metal oxide films for flexible transparent electronics. Adv. Funct. Mater. 2015, 25, 2807–2815. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, W.; Choi, S.; Kim, K.T.; Kang, J.; Park, S.K.; Kim, Y.-H. Determination of Insulator-to-Semiconductor Transition in Sol-Gel Oxide Semiconductors Using Derivative Spectroscopy. Materials 2016, 9, 6. https://doi.org/10.3390/ma9010006
Lee W, Choi S, Kim KT, Kang J, Park SK, Kim Y-H. Determination of Insulator-to-Semiconductor Transition in Sol-Gel Oxide Semiconductors Using Derivative Spectroscopy. Materials. 2016; 9(1):6. https://doi.org/10.3390/ma9010006
Chicago/Turabian StyleLee, Woobin, Seungbeom Choi, Kyung Tae Kim, Jingu Kang, Sung Kyu Park, and Yong-Hoon Kim. 2016. "Determination of Insulator-to-Semiconductor Transition in Sol-Gel Oxide Semiconductors Using Derivative Spectroscopy" Materials 9, no. 1: 6. https://doi.org/10.3390/ma9010006
APA StyleLee, W., Choi, S., Kim, K. T., Kang, J., Park, S. K., & Kim, Y.-H. (2016). Determination of Insulator-to-Semiconductor Transition in Sol-Gel Oxide Semiconductors Using Derivative Spectroscopy. Materials, 9(1), 6. https://doi.org/10.3390/ma9010006




