Influence of Different Post-Plasma Treatment Storage Conditions on the Shear Bond Strength of Veneering Porcelain to Zirconia
Abstract
:1. Introduction
2. Results
2.1. Scanning Electron Microscopy and Atomic Force Microscopy

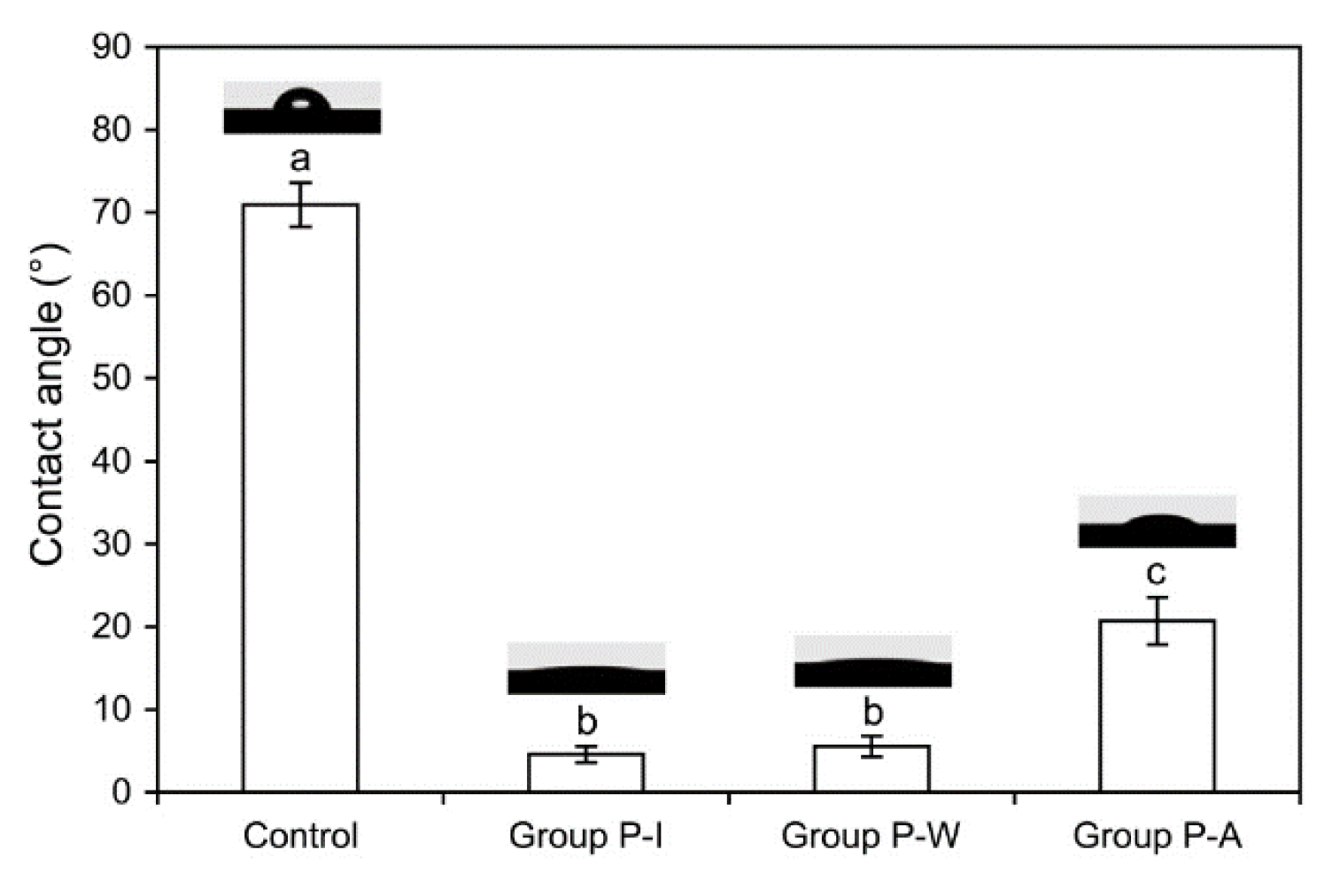
2.2. Water Contact Angles
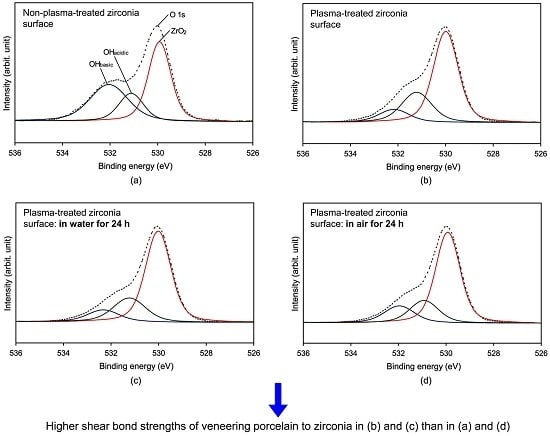
2.3. X-ray Photoelectron Spectroscopy
| Group | Zr 3d | O 1s | Y 3d | C 1s | O/C |
|---|---|---|---|---|---|
| Control | 15.05 | 48.32 | 1.32 | 35.31 | 1.37 |
| P-I | 20.73 | 64.93 | 1.71 | 12.63 | 5.14 |
| P-W | 22.31 | 62.04 | 1.66 | 13.99 | 4.43 |
| P-A | 22.11 | 61.23 | 1.65 | 15.01 | 4.08 |
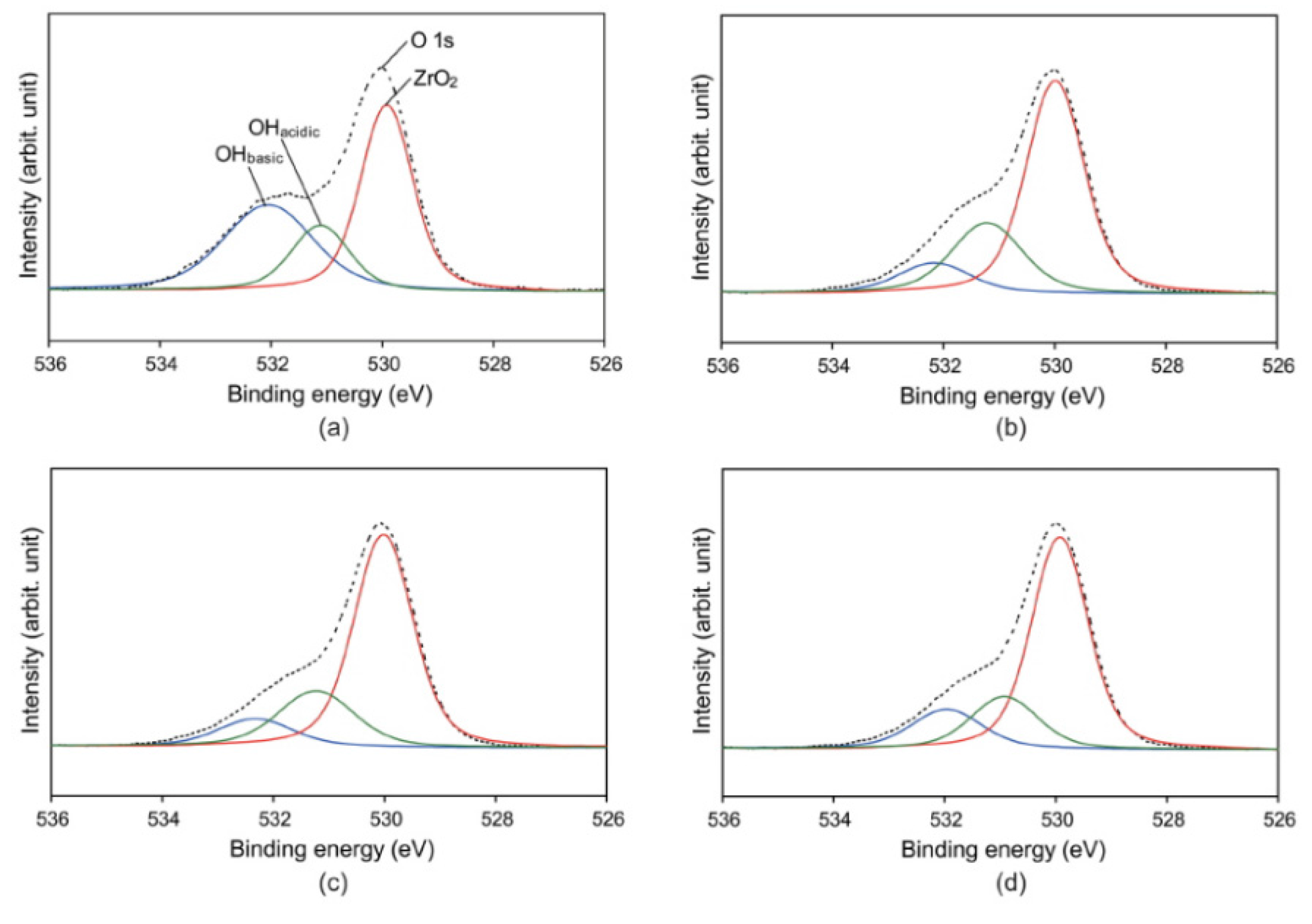
2.4. Shear Bond Strength and Failure Pattern
| Group | No-Liner Subgroups | Liner Subgroups |
|---|---|---|
| Control | 17.65 (2.98) Aa | 26.99 (2.93) b |
| P-I | 32.59 (3.58) Ba | 27.80 (3.00) b |
| P-W | 30.42 (2.27) Ba | 27.84 (2.51) b |
| P-A | 26.07 (1.93) Ca | 26.78 (2.25) a |
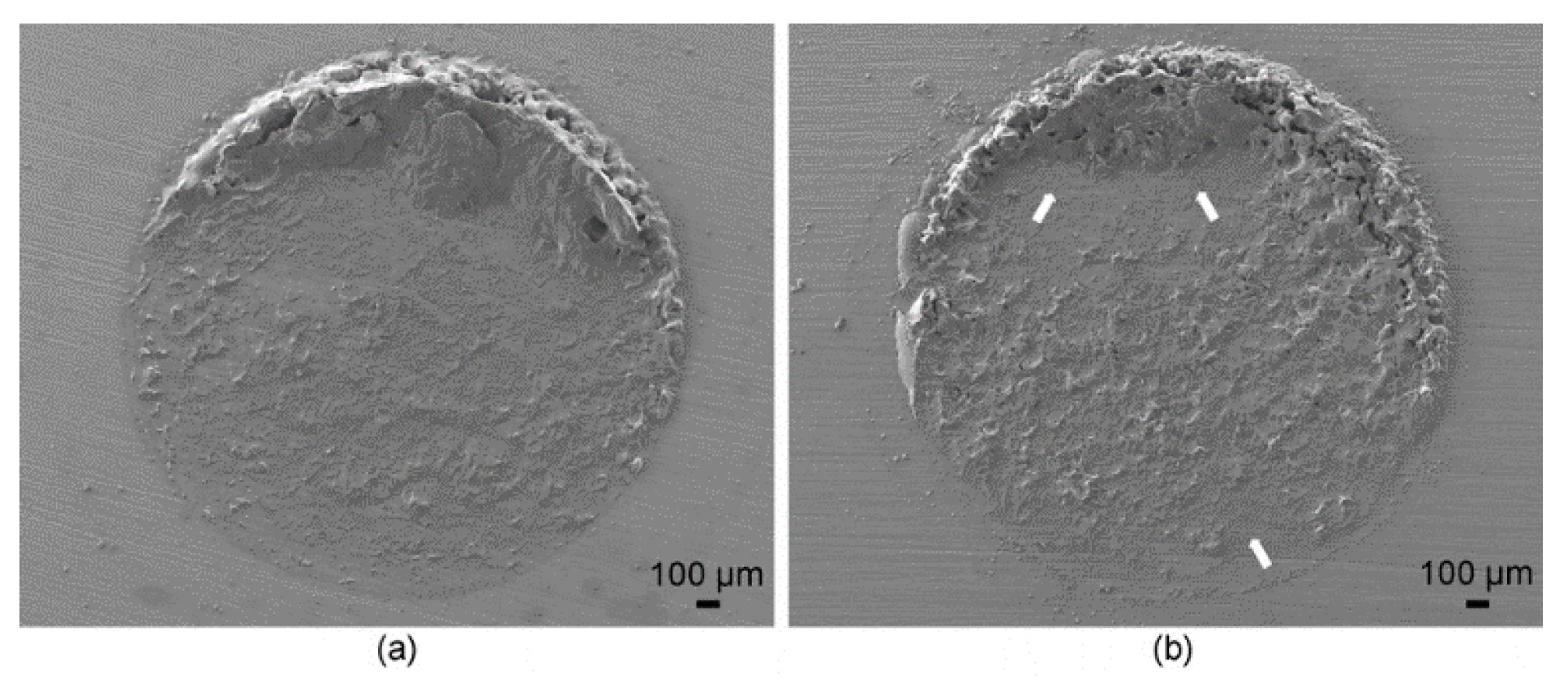
3. Discussion
4. Materials and Methods
4.1. Zirconia Specimen Preparation
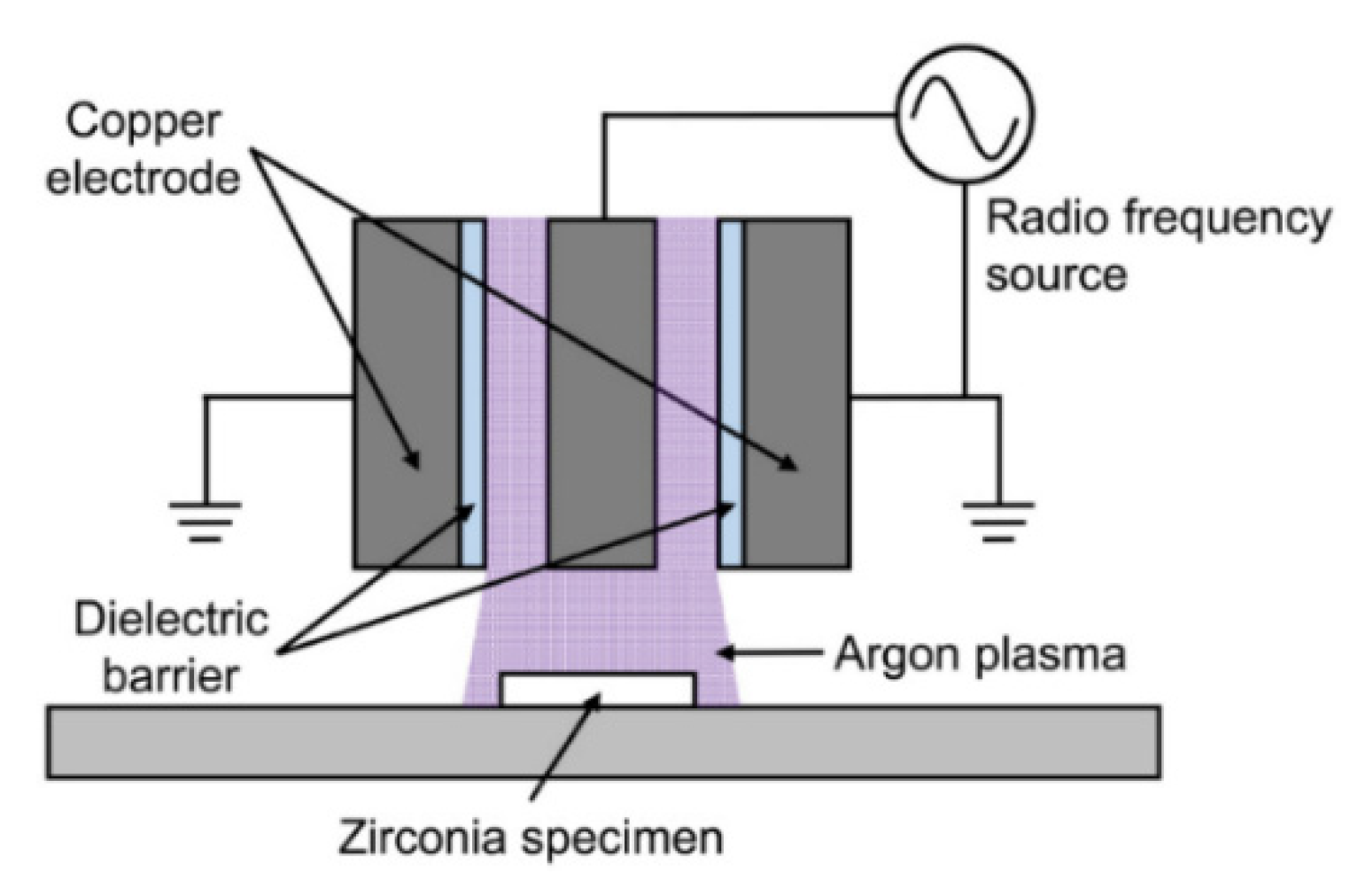
4.2. Plasma Treatment
4.3. Surface Examinations
4.4. Contact Angle Measurements
4.5. X-ray Photoelectron Spectroscopy
4.6. Veneering Procedures

4.7. Shear Bond Strength Testing
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kim, M.J.; Kim, Y.K.; Kim, K.H.; Kwon, T.Y. Shear bond strengths of various luting cements to zirconia ceramic: Surface chemical aspects. J. Dent. 2011, 39, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Janyavula, S.; Lawson, N.; Cakir, D.; Beck, P.; Ramp, L.C.; Burgess, J.O. The wear of polished and glazed zirconia against enamel. J. Prosthet. Dent. 2013, 109, 22–29. [Google Scholar] [CrossRef]
- Chaiyabutr, Y.; McGowan, S.; Phillips, K.M.; Kois, J.C.; Giordano, R.A. The effect of hydrofluoric acid surface treatment and bond strength of a zirconia veneering ceramic. J. Prosthet. Dent. 2008, 100, 194–202. [Google Scholar] [CrossRef]
- Canullo, L.; Micarelli, C.; Bettazzoni, L.; Magnelli, A.; Baldissara, P. Shear bond strength of veneering porcelain to zirconia after argon plasma treatment. Int. J. Prosthodont. 2014, 27, 137–139. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lim, H.P.; Park, Y.J.; Vang, M.S. Effect of zirconia surface treatments on the shear bond strength of veneering ceramic. J. Prosthet. Dent. 2011, 105, 315–322. [Google Scholar] [CrossRef]
- Sailer, I.; Feher, A.; Filser, F.; Gauckler, L.J.; Luthy, H.; Hammerle, C.H. Five-year clinical results of zirconia frameworks for posterior fixed partial dentures. Int. J. Prosthodont. 2007, 20, 383–388. [Google Scholar] [PubMed]
- Wang, G.; Zhang, S.; Bian, C.; Kong, H. Interface toughness of a zirconia-veneer system and the effect of a liner application. J. Prosthet. Dent. 2014, 112, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Kim, S.H.; Lee, J.B.; Han, J.S.; Yeo, I.S. In vitro evaluation of fracture strength of zirconia restoration veneered with various ceramic materials. J. Adv. Prosthodont. 2012, 4, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.; Goellner, M.; Lohbauer, U.; Wichmann, M.; Reich, S. Zirconia posterior fixed partial dentures: 5-year clinical results of a prospective clinical trial. Int. J. Prosthodont. 2012, 25, 585–589. [Google Scholar] [PubMed]
- Tarumi, N.; Uo, M.; Yamaga, E.; Watari, F. SEM observation and wettability of variously processed and fractured surface of dental zirconia. Appl. Surf. Sci. 2012, 262, 253–257. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, S.; Bian, C.; Kong, H. Effect of zirconia surface treatment on zirconia/veneer interfacial toughness evaluated by fracture mechanics method. J. Dent. 2014, 42, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Ritts, A.C.; Li, H.; Yu, Q.; Xu, C.; Yao, X.; Hong, L.; Wang, Y. Dentin surface treatment using a non-thermal argon plasma brush for interfacial bonding improvement in composite restoration. Eur. J. Oral Sci. 2010, 118, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhang, Y.; Sky Driver, M.; Caruso, A.N.; Yu, Q.; Wang, Y. Surface modification of several dental substrates by non-thermal, atmospheric plasma brush. Dent. Mater. 2013, 29, 871–880. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Villanueva, C.; Encinas, N.; Abenojar, J.; Martínez, M.A. Assessment of atmospheric plasma treatment cleaning effect on steel surfaces. Surf. Coat. Technol. 2013, 236, 450–456. [Google Scholar] [CrossRef]
- Liebermann, A.; Keul, C.; Bahr, N.; Edelhoff, D.; Eichberger, M.; Roos, M.; Stawarczyk, B. Impact of plasma treatment of PMMA-based CAD/CAM blanks on surface properties as well as on adhesion to self-adhesive resin composite cements. Dent. Mater. 2013, 29, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.H.; Kim, J.K.; Liu, D.; Liu, P.C.K.; Cheung, Y.M.; Ng, M.W. Effect of plasma treatment of Au-Ni-Cu bond pads on process windows of Au wire bonding. IEEE Trans. Adv. Packag. 2005, 28, 674–684. [Google Scholar] [CrossRef]
- Noro, A.; Kaneko, M.; Murata, I.; Yoshinari, M. Influence of surface topography and surface physicochemistry on wettability of zirconia (tetragonal zirconia polycrystal). J. Biomed. Mater. Res. B 2013, 101, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Lohbauer, U.; Zipperle, M.; Rischka, K.; Petschelt, A.; Müller, F.A. Hydroxylation of dental zirconia surfaces: Characterization and bonding potential. J. Biomed. Mater. Res. B. 2008, 87, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Piascik, J.R.; Swift, E.J.; Braswell, K.; Stoner, B.R. Surface fluorination of zirconia: Adhesive bond strength comparison to commercial primers. Dent. Mater. 2012, 28, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Nam, Y.; Sharratt, S.; Byon, C.; Kim, S.J.; Ju, Y.S. Fabrication and characterization of the capillary performance of superhydrophilic Cu micropost arrays. J. Microelectromech. Syst. 2010, 19, 581–588. [Google Scholar]
- Yaman, N.; Özdoğan, E.; Seventekin, N. Atmospheric plasma treatment of polypropylene fabric for improved dyeability with insoluble textile dyestuff. Fibers Polym. 2011, 12, 35–41. [Google Scholar] [CrossRef]
- Da Silva, M.A.M.; Guerra Neto, C.L.B.; Nunes Filho, A.; Freitas, D.O.; Braz, D.C.; Alves, C., Jr. Influence of topography on plasma treated titanium surface wettability. Surf. Coat. Technol. 2013, 235, 447–453. [Google Scholar] [CrossRef]
- Choi, Y.R.; Kwon, J.S.; Song, D.H.; Choi, E.H.; Lee, Y.K.; Kim, K.N.; Kim, K.M. Surface modification of biphasic calcium phosphate scaffolds by non-thermal atmospheric pressure nitrogen and air plasma treatment for improving osteoblast attachment and proliferation. Thin Solid Films 2013, 547, 235–240. [Google Scholar] [CrossRef]
- Fricke, K.; Steffen, H.; von Woedtke, T.; Schroder, K.; Weltmann, K.D. High rate etching of polymers by means of an atmospheric pressure plasma jet. Plasma Process. Polym. 2011, 8, 51–58. [Google Scholar] [CrossRef]
- Sun, S.; Sun, J.; Yao, L.; Qiu, Y. Wettability and sizing property improvement of raw cotton yarns treated with He/O2 atmospheric pressure plasma jet. Appl. Surf. Sci. 2011, 257, 2377–2382. [Google Scholar] [CrossRef]
- Yavirach, P.; Chaijareenont, P.; Boonyawan, D.; Pattamapun, K.; Tunma, S.; Takahashi, H.; Arksornnukit, M. Effects of plasma treatment on the shear bond strength between fiber-reinforced composite posts and resin composite for core build-up. Dent. Mater. J. 2009, 28, 686–692. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.; Wang, J.; Yang, Z.; Wang, Z.; Wang, H.; Yang, S. A simple one-step solution deposition process for constructing high-performance amorphous zirconium oxide thin film. RSC Adv. 2014, 4, 6060–6067. [Google Scholar] [CrossRef]
- Homola, T.; Matoušek, J.; Medvecká, V.; Zahoranová, A.; Kormunda, M.; Kováčik, D.; Černák, M. Atmospheric pressure diffuse plasma in ambient air for ITO surface cleaning. Appl. Surf. Sci. 2012, 258, 7135–7139. [Google Scholar] [CrossRef]
- Piascik, J.R.; Wolter, S.D.; Stoner, B.R. Development of a novel surface modification for improved bonding to zirconia. Dent. Mater. 2011, 27, e99–e105. [Google Scholar] [CrossRef] [PubMed]
- Maryanchik, I.; Brendlinger, E.J.; Fallis, D.W.; Vandewalle, K.S. Shear bond strength of orthodontic brackets bonded to various esthetic pontic materials. Am. J. Orthod. Dentofac. Orthop. 2010, 137, 684–689. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.-H.; Min, B.K.; Son, J.S.; Kwon, T.-Y. Influence of Different Post-Plasma Treatment Storage Conditions on the Shear Bond Strength of Veneering Porcelain to Zirconia. Materials 2016, 9, 43. https://doi.org/10.3390/ma9010043
Lee M-H, Min BK, Son JS, Kwon T-Y. Influence of Different Post-Plasma Treatment Storage Conditions on the Shear Bond Strength of Veneering Porcelain to Zirconia. Materials. 2016; 9(1):43. https://doi.org/10.3390/ma9010043
Chicago/Turabian StyleLee, Mun-Hwan, Bong Ki Min, Jun Sik Son, and Tae-Yub Kwon. 2016. "Influence of Different Post-Plasma Treatment Storage Conditions on the Shear Bond Strength of Veneering Porcelain to Zirconia" Materials 9, no. 1: 43. https://doi.org/10.3390/ma9010043
APA StyleLee, M.-H., Min, B. K., Son, J. S., & Kwon, T.-Y. (2016). Influence of Different Post-Plasma Treatment Storage Conditions on the Shear Bond Strength of Veneering Porcelain to Zirconia. Materials, 9(1), 43. https://doi.org/10.3390/ma9010043