1. Introduction
In recent years, the use of thermal energy storage (TES) with latent heat storage has become a very popular topic within researchers. The main advantage of latent heat storage is the high storage density in small temperature intervals, showing very big potential to be used in building applications [
1]. However, in most cases, the materials used in latent heat storage, known as phase change materials (PCM), need to be encapsulated to avoid leakage when it is in the liquid phase. There are three means of encapsulation: micro-encapsulation, macro-encapsulation and shape-stabilization [
2], although recently nano-encapsulation has also grown in interest [
3,
4].
Microencapsulation is the encapsulation in particles smaller than 1 mm in diameter, known as microcapsules, microparticles, microspheres [
5]. Microencapsulation serves several purposes, such as holding the liquid PCM and preventing changes of its composition through contact with the environment; improving material compatibility with the surrounding, through building a barrier; improving handling of the PCM in a production; reducing external volume changes, which is usually also a positive effect for an application; improving heat transfer to the surrounding through its large surface to volume ratio; and improving cycling stability since phase separation is restricted to microscopic distances.
Microencapsulation processes can be categorized into two groups: physical processes and chemical processes. Physical methods include spray cooling, spray drying, and fluidized bed processes; chemical processes include
in-situ polymerization (interfacial polycondensation, suspension polymerization, and emulsion polymerization), complex coacervation, sol-gel method, and solvent extraction/evaporation method. Physical methods are limited by their granulated sizes thus making them useful for producing microencapsulated PCM particles, and chemical methods can produce much smaller encapsulated PCM particles [
3,
5,
6,
7]. Hawlader
et al. [
8] reported a substantial drop in heat storage capacity with the physical methods as compared to that of chemical methods.
In 2011, Cabeza
et al. [
1] claimed that only hydrophobic PCM could be microencapsulated. In 2015, Su
et al. [
3] claimed that inorganic PCM micro-/nano-encapsulation is limited to the solvent extraction/evaporation method, probably based in the existence of the study from Salaun
et al. [
9]. In 2015, Khadiran
et al. [
5] and Giro-Paloma
et al. [
6] reviewed only the encapsulation techniques of organic PCM. Therefore, there is a research gap on finding ways to encapsulate inorganic PCM.
At the time of writing this paper, microencapsulation of inorganic PCM can be found in very few papers. For example, Salaun
et al. [
9] microencapsulated sodium phosphate dodecahydrate (DSP) by solvent evaporation-precipitation method using various organic solvents and cellulose acetated butyrate (CAB) crosslinked by methylene di-isocyanate (MDI) as coating polymer. Those authors identified that the nature of the solvent was one of the most influencing parameters in the final surface morphology of the microcapsule. Similarly, Huang
et al. [
10] microencapsulated disodium hydrogen phosphate heptahydrate (Na
2HPO
4·7H
2O) by means of the suspension copolymerization-solvent volatile method with modified PMMA as coating polymer. Hassabo
et al. [
11] microencapsulated six different salt hydrates (calcium nitrate tetrahydrate, calcium chloride hexahydrate, sodium sulphate decahydrate, disodium hydrogen phosphate dodecahydrate, ferric nitrate nonahydrate, and manganese (II) nitrate hexahydrate) by polycondensation of tetraethoxysilane.
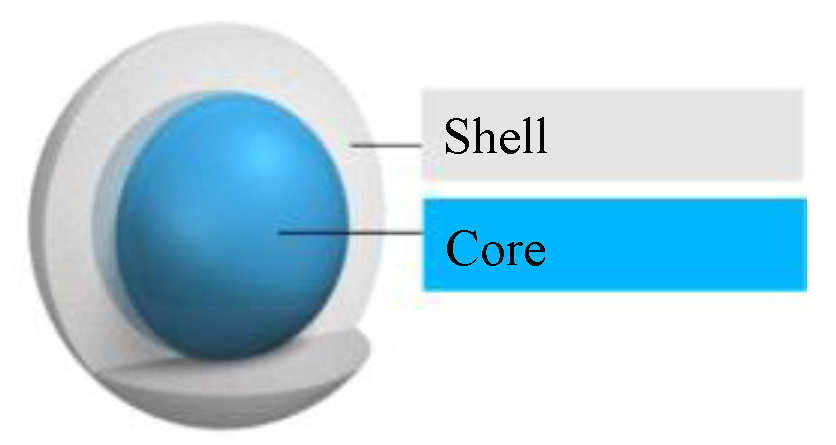
Moreover, microencapsulated PCM are composed of two main parts (
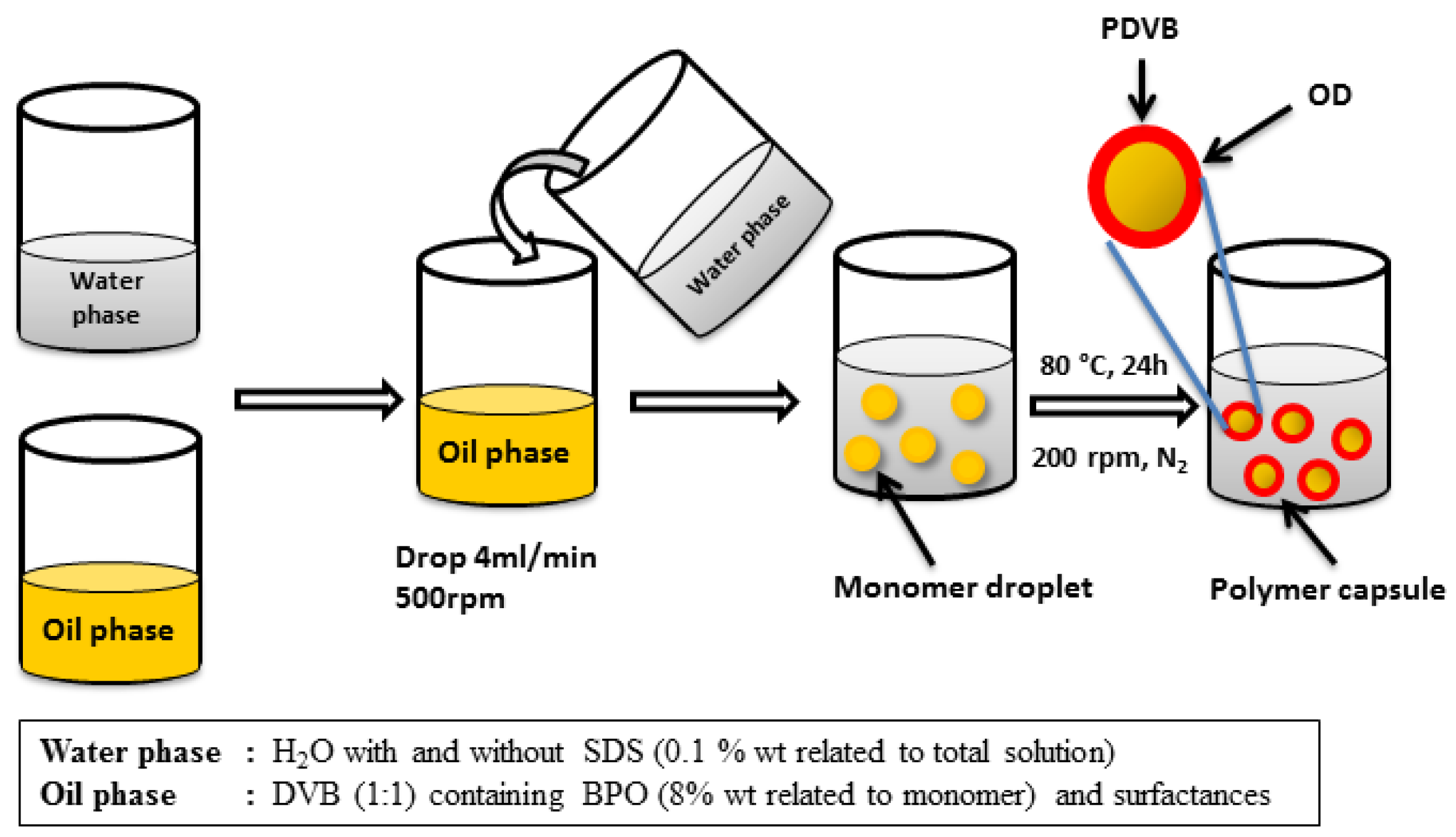
Figure 1), the core (the PCM) and the shell (usually a polymer). However, the process of microencapsulation always involves two solvents, that should not be miscible between them (
Figure 2 shows the process of microsuspension polymerization as example). So the problem of microencapsulating inorganic PCM is that water is always used as solvent in microencapsulation processes and salt hydrates are soluble in water.
Figure 1.
Structure of a microencapsulated phase change materials (PCM) (adapted from [
5]).
Figure 1.
Structure of a microencapsulated phase change materials (PCM) (adapted from [
5]).
Figure 2.
Microsuspension polymerization process [
12].
Figure 2.
Microsuspension polymerization process [
12].
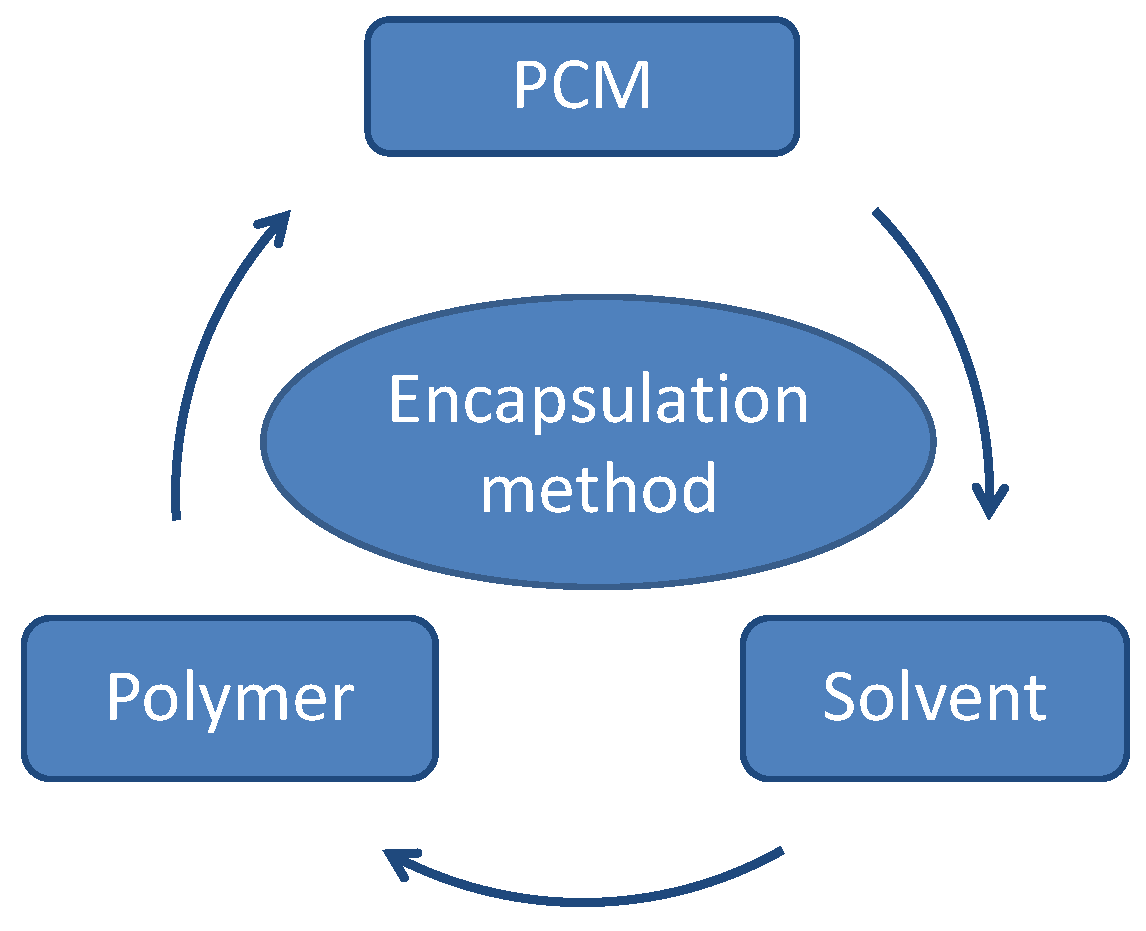
The aim of this paper was to microencapsulate inorganic PCM. To achieve this objective, an encapsulation method had to be selected taking into consideration that not only the PCM or the shell material (polymer) would influence the process, but also the solvent to be used during the encapsulation process (
Figure 3); therefore, between the available methods, a fluidized bed process was selected.
Figure 3.
Material selection influencing an encapsulation method.
Figure 3.
Material selection influencing an encapsulation method.
Encapsulation using fluidized bed has been used extensively in areas such as food [
13,
14,
15] and agriculture [
16] but to the author’s knowledge it has never been used for microencapsulated PCM. Notoriously, fluidization of PCM was used as early as 1988 by Sozen
et al. [
17] to increase the heat storage efficiency of Glauber salt, an inorganic PCM. In this study, fluidization provided enhanced heat transfer to or from the storage medium and resulted in a steady-state heat storage efficiency of about 60% after repeated heating and cooling cycles. However, this technology was not used again until 2013 when Izquierdo-Barrientos
et al. started a series of papers on the study of thermal energy storage in a fluidized bed of PCM [
18,
19,
20]. The results showed that fluidized PCM can increase the efficiency of the system.
3. Discussion
Although there is a need for microencapsulation of PCM and organic PCM have been widely microencapsulated by many different methods, microencapsulation of inorganic PCM has been not studied adequately. This paper shows that fluidization is a good method to do so, since this study shows that efficiencies of around 90% were achieved microencapsulting pure MgCl2·6H2O and the by-product bischofite, with 95% MgCl2·6H2O. To do so, not only the fluidization parameters such as fluidization time and atomization flow need to be selected, 2 min and 2 kg/h were used respectively, but also the compatibility between the three materials involved in the process: PCM, polymer and solvent. Results show that for the PCM studied, MgCl2·6H2O and bischofite, the solvent to be used should be chloroform and the polymer acrylic as shell material. The final microcapsules had excellent melting temperatures and enthalpy compared to the original PCM, 104.6 °C and 95 J/g for bischofite, and 95.3 °C and 118.3 J/g for MgCl2·6H2O.
4. Materials and Methods
4.1. Materials
As material to be encapsulated, two salt hydrates were used as PCM, magnesium chloride hexahydrate (99% Merck S.A, Santiago, Chile) and bischofite (Salmag, Antofagasta, Chile). Bischofite is a mineral that precipitates in the evaporation ponds during potassium chloride production process in the Salar de Atacama (Chile) [
27]. Bischofite is a by-product with a chemical composition of at least 95% MgCl
2·6H
2O that melts at 101 °C with a heat of fusion of 116.2 J/g.
For a material to be a good encapsulating material it needs to be compatible with the PCM that will encapsulated, needs to be thermally and physically stable during the melting and solidification cycles, should have low density and be non-corrosive, and should be easy to produce.
The materials chosen in the paper that fulfil the requirements listed above were high density polyethylene (HDPE), resin epoxy (both from Plastigen S.A., Antofagasta, Chile), polystyrene and acrylic (from Norglass, Santiago, Chile). The HDPE was used both as a bar and in granular form (prill or perl). The solvents used were acetone, chloroform, xylene, and tetrahydrofuran (THF), all >99.7% from Norglass.
4.2. Compatibility Studies
Since the polymers will be solubilized in the PCM in the fluidized bed, first of all, the compatibility between polymers and solvents were carried out. Between 1 and 50 mL of solvent were mixed with 1 to 5 g of polymer and the mixture was agitated constantly during 24 h. The polymer with the best solubility with the organic solvents was chosen.
When the mixture polymer-solvent covers the PCM, this could be partially solubilized by the organic solvent, which would mean PCM losses; therefore, the solubility between the PCM and the considered solvents was determined. For this study, two PCM-solvent ratios were used, 60:40 and 90:10. The variables measured were the initial mass and the final mass of PCM after the interaction with the solvent. The optimal mixture was that where there was not mass difference.
The compatibility between the PCM and the polymer was also studied to ensure the stability of the final microcapsules. The experiments were carried out immersing four sheets of each polymer, having previously been weight, in melted PCM at, approximately, 10 °C over the melting temperature of the PCM inside an oven during 60 days. On sheet was evaluated after 15, 30, 45 and 60 days of experimentation. The difference between the initial and the final mass of the polymer was used to determine the possible degradation of the polymer.
4.3. Microencapsulation via Fluidization
The used equipment was a glass fluidization chamber, an air source with a mess distributor to distribute the air flow evenly in the chamber, and a spray system. Based upon the literature [
28], it seemed necessary to use a pressure nozzle to achieve the desired droplets size.
The studied variables were:
The polymer concentration was established at 60% and 90%. Concentrations lower than 60% were tried but no good results could be achieved.
The polymer atomization flow was set at 2 kg/h and 4 kg/h.
The fluidization time was selected to be 60 s and 120 s.
The PCM mass was fixed at 100 g of salt hydrate.
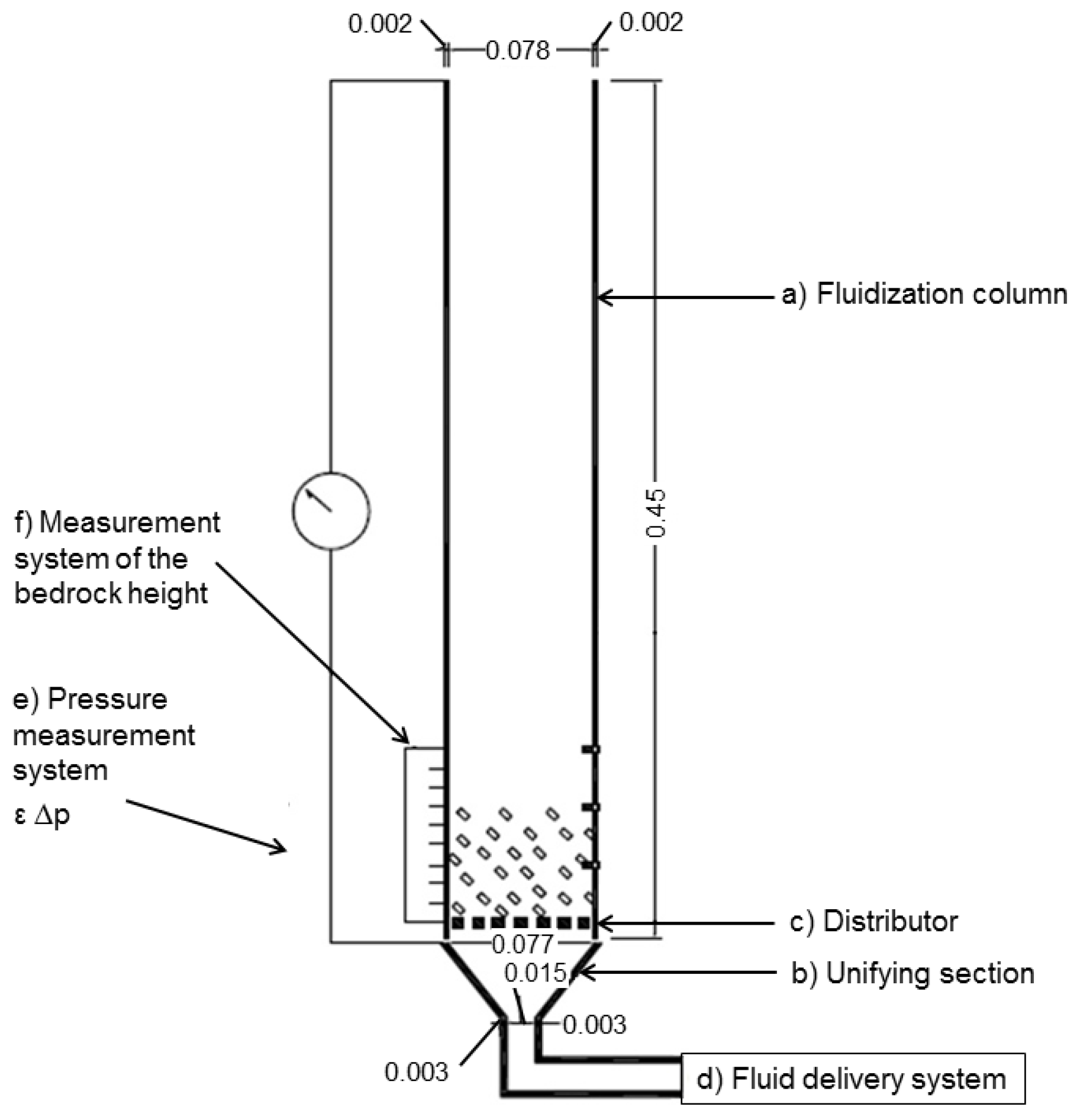
The fluidization method used was particles fluidization, where the crystals are suspended in an air flow as shown in
Figure 7. The dimensions of the fluidization chamber are summarized in
Table 8. As uniformization section, a cone of glass with the dimensions shown in
Table 9 was used. The distributor was PVC mesh with a diameter of 200 μm.
The particles properties studied were that required for fluidization method: sphericity, diameter and density. The characteristics of MgCl
2·6H
2O particles are presented in
Table 10 [
29]. The same values were considered for bischofite.
Figure 7.
Fluidization column used in this study (units: m).
Figure 7.
Fluidization column used in this study (units: m).
Table 8.
Dimensions of the fluidization chamber.
Table 8.
Dimensions of the fluidization chamber.
| Dimensions | Value (m) |
|---|
| Tube external diameter | 0.08 |
| Tube internal diameter | 0.077 |
| Thickness | 0.0015 |
| Height | 0.45 |
Table 9.
Dimensions of the uniformization section of the fluidization chamber.
Table 9.
Dimensions of the uniformization section of the fluidization chamber.
| Dimensions | Value (m) |
|---|
| Internal diameter of the tube in its upper part | 0.077 |
| Internal diameter of the tube in its lower part | 0.015 |
| Thickness | 0.0015 |
| Height | 0.03 |
Table 10.
MgCl
2·6H
2O particles properties [
29].
Table 10.
MgCl2·6H2O particles properties [29].
| Parameter | Value |
|---|
| Sphericity, | 0.86 |
| Average particle diameter, dp | 500 μm |
| Density | 1570 kg/m3 |
4.4. Chemical Analysis
Thermophysical properties of the encapsulated PCM were analyzed by differential scanning calorimetry (DSC) with a Foenix F 201 (NETZSCH Group, Santiago, Chile). Measurements were done with 40 µL micro-crucibles hermetically closed.
Morphological characteristics of the salts hydrates and the microcapsules were determined with an optical phase contrast microscopy Olympus with a mechanism coaxial coarse and fine focus adjustment objectives 4×, 10×, 40×, and 60× connected to a precision chamber. Bischofite polymer was dyed with a blue pigment and MgCl2·6H2O polymer was dyed with a red pigment, so microcapsules could be differentiated from the non-encapsulated PCM.