Effect of Growth Pressure on Epitaxial Graphene Grown on 4H-SiC Substrates by Using Ethene Chemical Vapor Deposition
Abstract
:1. Introduction
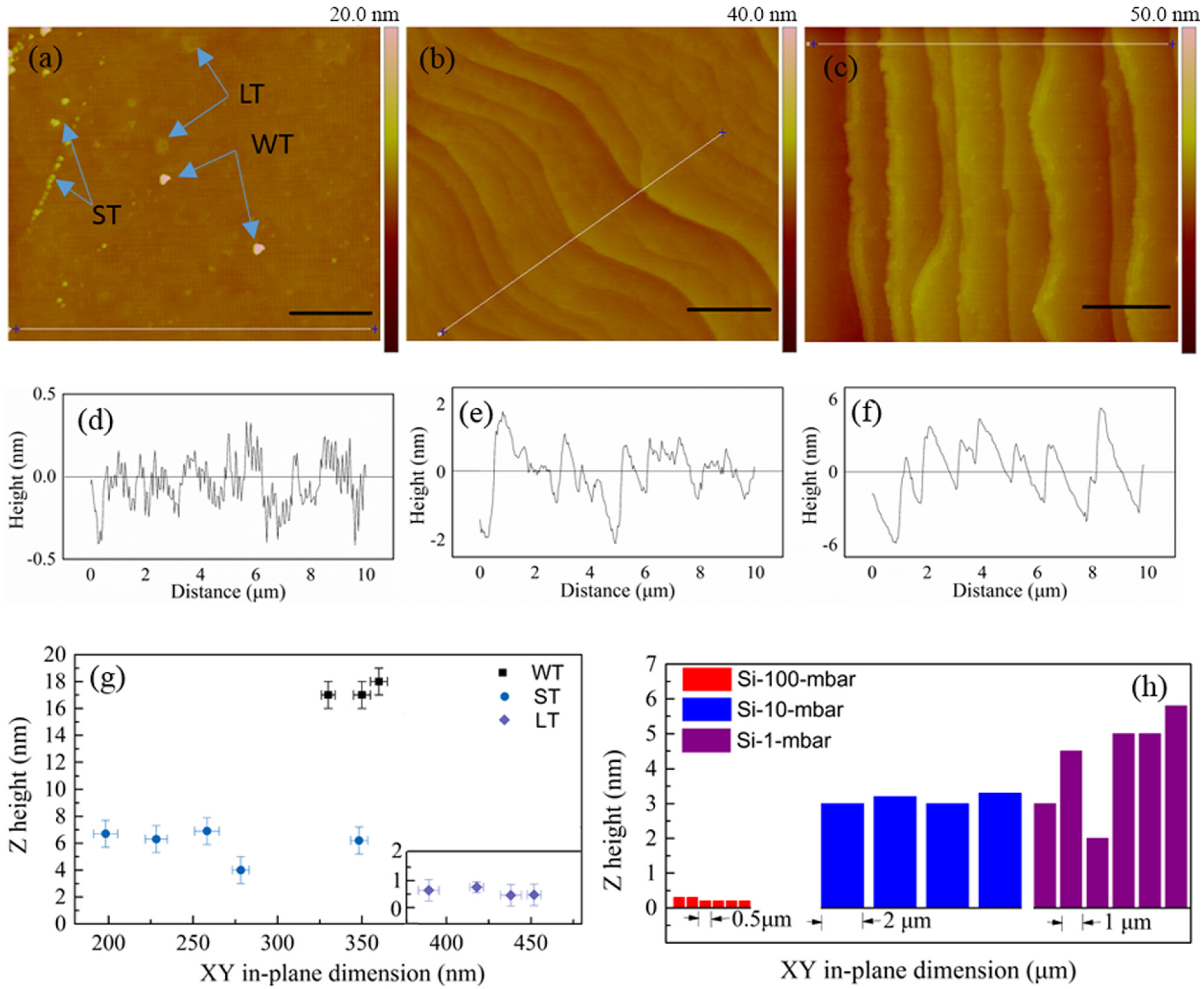
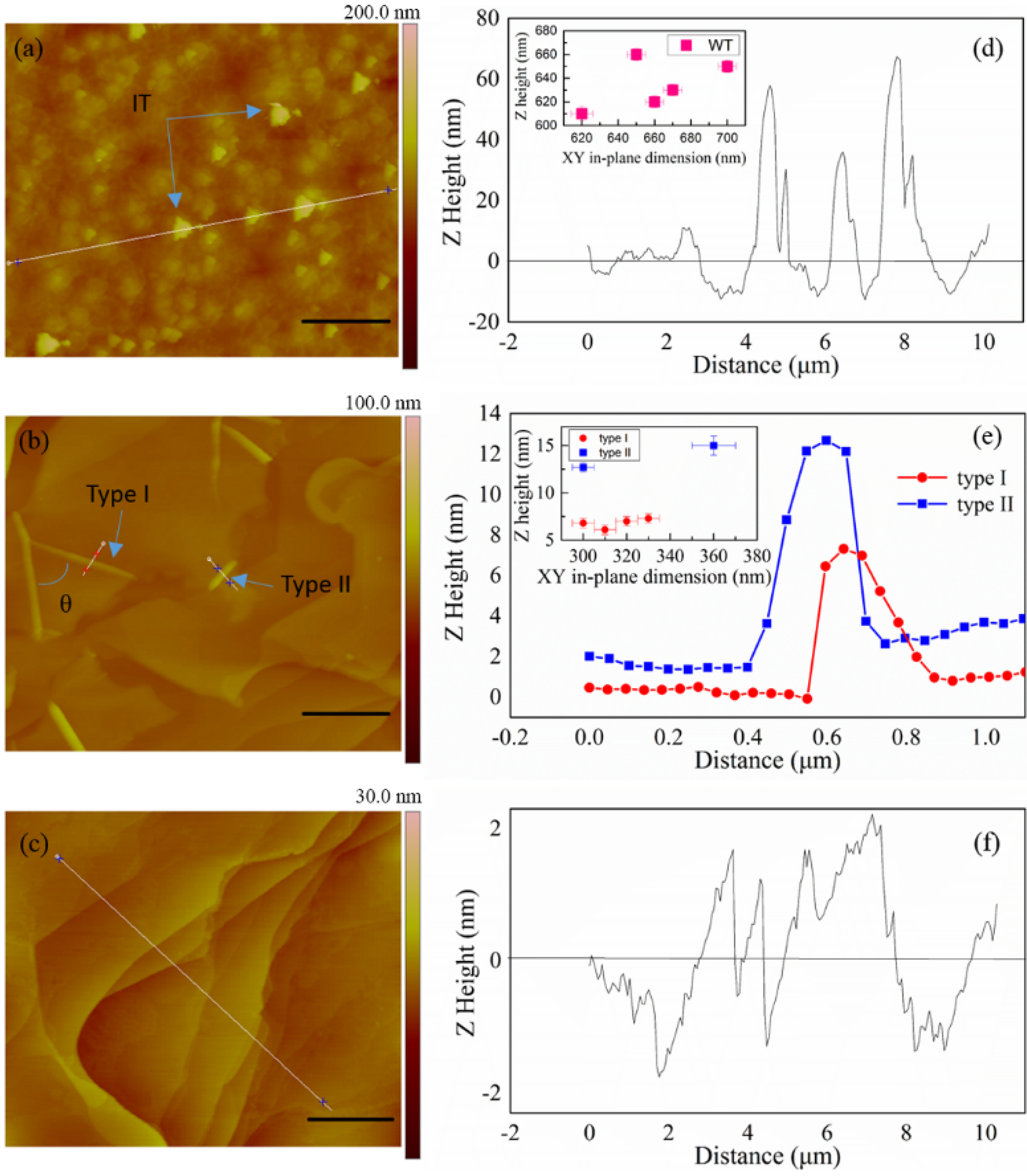
2. Results and Discussion
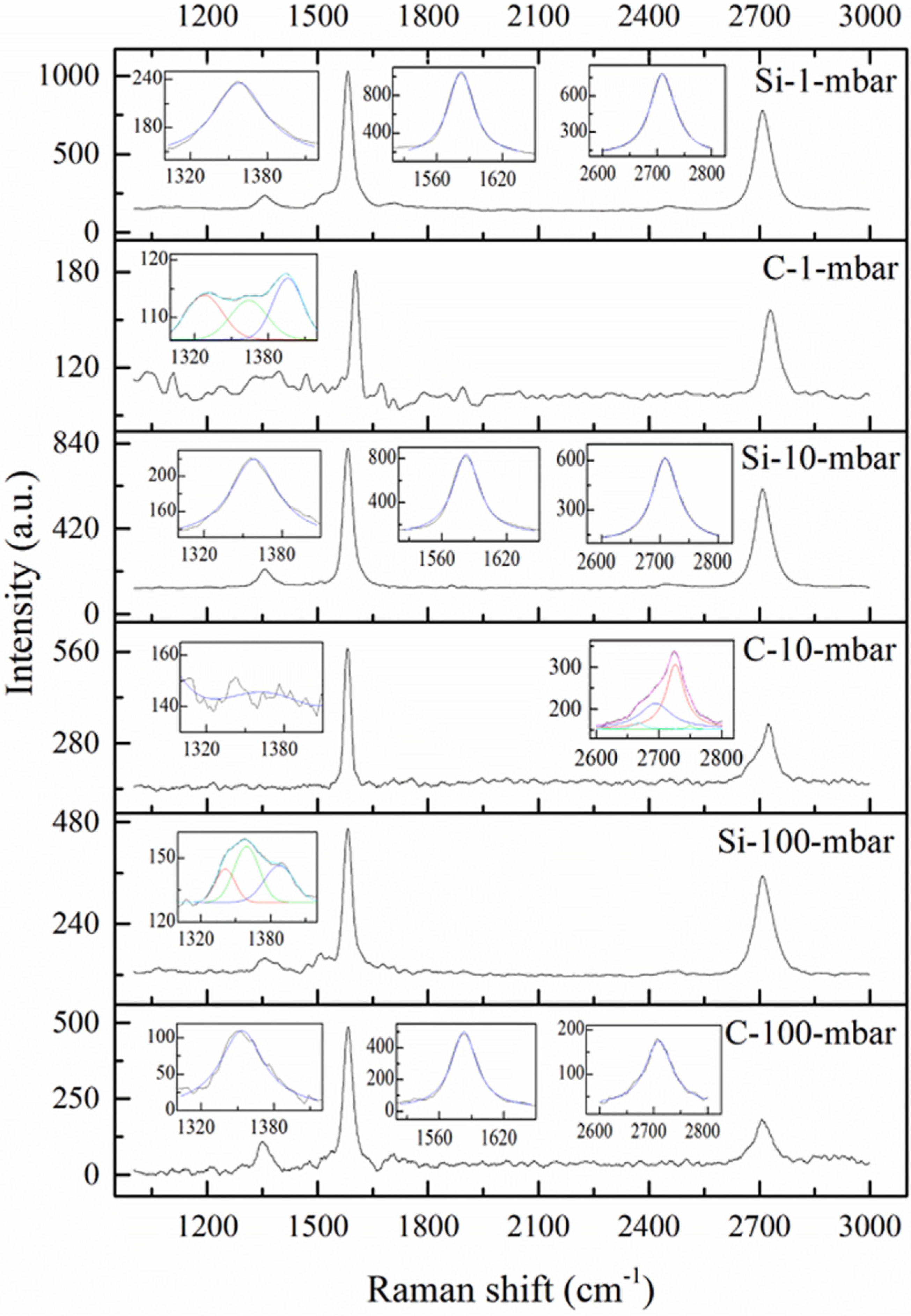
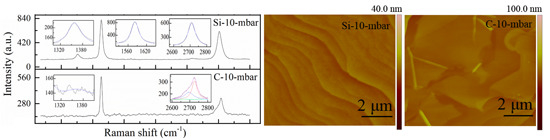
| Samples | D (cm−1) | G (cm−1) | Ratio | La (nm) | ||||
|---|---|---|---|---|---|---|---|---|
| X | FWHM | X | FWHM | ID/IG | AD/AG | (ID/IG) | (AD/AG) | |
| Si-1-mbar | 1358 | 50 | 1582 | 29 | 0.10 | 0.18 | 167.5 | 93.1 |
| C-1-mbar | / | / | 1602 | 30 | ---- | ---- | ---- | ---- |
| 1328 | 35 | / | / | ---- | ---- | ---- | ---- | |
| 1364 | 35 | / | / | ---- | ---- | ---- | ---- | |
| 1396 | 29 | / | / | ---- | ---- | ---- | ---- | |
| Si-10-mbar | 1358 | 45 | 1583 | 30 | 0.12 | 0.19 | 139.6 | 88.2 |
| C-10-mbar | / | / | 1581 | 21 | ---- | ---- | ---- | ---- |
| Si-100-mbar | / | / | 1582 | 28 | ---- | ---- | ---- | ---- |
| 1341 | 20 | / | / | ---- | ---- | ---- | ---- | |
| 1359 | 25 | / | / | ---- | ---- | ---- | ---- | |
| 1387 | 30 | / | / | ---- | ---- | ---- | ---- | |
| C-100-mbar | 1354 | 44 | 1583 | 29 | 0.22 | 0.33 | 76.1 | 50.8 |
3. Experimental Section
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kim, J.; Park, H.; Hannon, J.B.; Bedell, S.W.; Fogel, K.; Sadana, D.K.; Dimitrakopoulos, C. Layer-resolved graphene transfer via engineered strain layers. Science 2013, 342, 833–836. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Emtsev, K.V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Roehrl, J.; et al. Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Kim, H.; Lee, Y.; Xu, X.; Park, J.S.; Zheng, Y.; Balakrishnan, J.; Lei, T.; Kim, H.R.; Song, Y.I.; et al. Roll-to-roll production of 30-inch graphene films for transparent electrodes. Nat. Nanotechnol. 2010, 5, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Bando, M.; Kimura, N.; Shimizu, K.; Kadono, K.; Umezu, N.; Miyahara, K.; Hayazaki, S.; Nagai, S.; Mizuguchi, Y.; et al. Production of a 100-m-long high-quality graphene transparent conductive film by roll-to-roll chemical vapor deposition and transfer process. Appl. Phys. Lett. 2013, 102, 023112. [Google Scholar] [CrossRef]
- Berger, C.; Song, Z.; Li, X.; Wu, X.; Brown, N.; Naud, C.; Mayou, D.; Li, T.; Hass, J.; Marchenkov, A.N.; et al. Electronic confinement and coherence in patterned epitaxial graphene. Science 2006, 312, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.H.; Kim, P.; Choi, J.Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Song, Z.M.; Li, T.B.; Li, X.B.; Ogbazghi, A.Y.; Feng, R.; Dai, Z.T.; Marchenkov, A.N.; Conrad, E.H.; First, P.N.; et al. Ultrathin epitaxial graphite: 2D electron gas properties and a route toward graphene-based nanoelectronics. J. Phys. Chem. B 2004, 108, 19912–19916. [Google Scholar] [CrossRef]
- Lin, Y.M.; Dimitrakopoulos, C.; Jenkins, K.A.; Farmer, D.B.; Chiu, H.Y.; Grill, A.; Avouris, P. 100-GHz transistors from wafer-scale epitaxial graphene. Science 2010, 327, 662–662. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-M.; Valdes-Garcia, A.; Han, S.-J.; Farmer, D.B.; Meric, I.; Sun, Y.; Wu, Y.; Grill, A.; Dimitrakopoulos, C.; Avouris, P.; et al. Wafer-scale graphene integrated circuit. Science 2011, 332, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E.; et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 2009, 324, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, X.Y.; Liu, X.R.; Jin, Z.; Wang, D.; Wan, L.J. Facile growth of centimeter-sized single-crystal graphene on copper foil at atmospheric pressure. J. Mater. Chem. C 2015, 3, 3530–3535. [Google Scholar] [CrossRef]
- Gao, L.; Ni, G.X.; Liu, Y.; Liu, B.; Castro Neto, A.H.; Loh, K.P. Face-to-face transfer of wafer-scale graphene films. Nature 2014, 505, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Cummings, A.W.; Dinh Loc, D.; Van Luan, N.; Dinh Van, T.; Kotakoski, J.; Barrios Vargas, J.E.; Lee, Y.H.; Roche, S. Charge transport in polycrystalline graphene: Challenges and opportunities. Adv. Mater. 2014, 26, 5079–5094. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Sun, G.S.; Liu, X.F.; Zhang, F.; Dong, L.; Zheng, L.; Yan, G.G.; Liu, S.B.; Zhao, W.S.; Wang, L.; et al. Fast homoepitaxial growth of 4H-SiC films on 4 degrees off-axis substrates in a SiH4-C2H4-H2 system. Chin. Phys. Lett. 2013, 30, 128101. [Google Scholar] [CrossRef]
- Palacio, I.; Celis, A.; Nair, M.N.; Gloter, A.; Zobelli, A.; Sicot, M.; Malterre, D.; Nevius, M.S.; de Heer, W.A.; Berger, C.; et al. Atomic structure of epitaxial graphene sidewall nanoribbons: Flat graphene, miniribbons, and the confinement gap. Nano Lett. 2015, 15, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Liu, G.; Rothwell, S.; Nevius, M.; Tejeda, A.; Taleb-Ibrahimi, A.; Feldman, L.C.; Cohen, P.I.; Conrad, E.H. Wide-gap semiconducting graphene from nitrogen-seeded SiC. Nano Lett. 2013, 13, 4827–4832. [Google Scholar] [CrossRef] [PubMed]
- Strupinski, W.; Grodecki, K.; Wysmolek, A.; Stepniewski, R.; Szkopek, T.; Gaskell, P.E.; Grueneis, A.; Haberer, D.; Bozek, R.; Krupka, J.; et al. Graphene epitaxy by chemical vapor deposition on SiC. Nano Lett. 2011, 11, 1786–1791. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.H.; Kang, C.; Son, B.H.; Ahn, Y.H.; Lee, J.S. Correlating nucleation density with heating ramp rates in continuous graphene film formation. Carbon 2014, 80, 708–715. [Google Scholar] [CrossRef]
- Jung, D.H.; Kang, C.; Kim, M.; Cheong, H.; Lee, H.; Lee, J.S. Effects of hydrogen partial pressure in the annealing process on graphene growth. J. Phys. Chem. C 2014, 118, 3574–3580. [Google Scholar] [CrossRef]
- Kang, C.; Jung, D.H.; Lee, J.S. Growth mechanism of graphene on graphene films grown by chemical vapor deposition. Chem. Asian J. 2015, 10, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Jernigan, G.G.; VanMil, B.L.; Tedesco, J.L.; Tischler, J.G.; Glaser, E.R.; Davidson, A., III; Campbell, P.M.; Gaskill, D.K. Comparison of epitaxial graphene on Si-face and C-face 4H SiC formed by ultrahigh vacuum and RF furnace production. Nano Lett. 2009, 9, 2605–2609. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sun, G.; Liu, B.; Yan, G.; Guan, M.; Zhang, Y.; Zhang, F.; Chen, Y.; Dong, L.; Zheng, L.; et al. Growth of hexagonal columnar nanograin structured SiC thin films on silicon substrates with graphene-graphitic carbon nanoflakes templates from solid carbon sources. Materials 2013, 6, 1543–1553. [Google Scholar] [CrossRef]
- Prakash, G.; Capano, M.A.; Bolen, M.L.; Zemlyanou, D.; Reifenberger, R.G. AFM study of ridges in few-layer epitaxial graphene grown on the carbon-face of 4H-SiC(000(1)over-bar). Carbon 2010, 48, 2383–2393. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S.; et al. Raman spectrum of graphene and graphene layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef] [PubMed]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cancado, L.G.; Jorio, A.; Saito, R. Studying disorder in graphite-based systems by Raman spectroscopy. Phys. Chem. Chem. Phys. 2007, 9, 1276–1291. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, C.; Reich, S. Doable resonant Raman scattering in graphite. Phys. Rev. Lett. 2000, 85, 5214–5217. [Google Scholar] [CrossRef] [PubMed]
- Strudwick, A.J.; Creeth, G.L.; Johansson, N.A.B.; Marrows, C.H. Probing residual strain in epitaxial graphene layers on 4H-SiC (000(1)over-bar) with Raman spectroscopy. Appl. Phys. Lett. 2011, 98, 051910. [Google Scholar] [CrossRef]
- Robinson, J.A.; Wetherington, M.; Tedesco, J.L.; Campbell, P.M.; Weng, X.; Stitt, J.; Fanton, M.A.; Frantz, E.; Snyder, D.; VanMil, B.L.; et al. Correlating Raman spectral signatures with carrier mobility in epitaxial graphene: A guide to achieving high mobility on the wafer scale. Nano Lett. 2009, 9, 2873–2876. [Google Scholar] [CrossRef] [PubMed]
- Cancado, L.G.; Jorio, A.; Martins Ferreira, E.H.; Stavale, F.; Achete, C.A.; Capaz, R.B.; Moutinho, M.V.O.; Lombardo, A.; Kulmala, T.S.; Ferrari, A.C. Quantifying defects in graphene via Raman spectroscopy at different excitation energies. Nano Lett. 2011, 11, 3190–3196. [Google Scholar] [CrossRef] [PubMed]
- Martins Ferreira, E.H.; Moutinho, M.V.O.; Stavale, F.; Lucchese, M.M.; Capaz, R.B.; Achete, C.A.; Jorio, A. Evolution of the Raman spectra from single-, few-, and many-layer graphene with increasing disorder. Phys. Rev. B 2010, 82, 125429. [Google Scholar] [CrossRef]
- Casiraghi, C.; Ferrari, A.C.; Robertson, J. Raman spectroscopy of hydrogenated amorphous carbons. Phys. Rev. B 2005, 72, 085401. [Google Scholar] [CrossRef]
- Cancado, L.G.; Jorio, A.; Pimenta, M.A. Measuring the absolute Raman cross section of nanographites as a function of laser energy and crystallite size. Phys. Rev. B 2007, 76, 064304. [Google Scholar] [CrossRef]
- Urban, J.M.; Dabrowski, P.; Binder, J.; Kopciuszynski, M.; Wysmolek, A.; Klusek, Z.; Jalochowski, M.; Strupinski, W.; Baranowski, J.M. Nitrogen doping of chemical vapor deposition grown graphene on 4H-SiC (0001). J. Appl. Phys. 2014, 11, 5233504. [Google Scholar] [CrossRef]
- Luo, W.G.; Wang, H.F.; Cai, K.M.; Han, W.P.; Tan, P.H.; Hu, P.A.; Wang, K.Y. Synthesis of homogenous bilayer graphene on industrial Cu foil. Chin. Phys. Lett. 2014, 31, 067202. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, S.; Liu, Z.; Zhong, N.; Liu, S.; Liu, X. Effect of Growth Pressure on Epitaxial Graphene Grown on 4H-SiC Substrates by Using Ethene Chemical Vapor Deposition. Materials 2015, 8, 5586-5596. https://doi.org/10.3390/ma8095263
Cai S, Liu Z, Zhong N, Liu S, Liu X. Effect of Growth Pressure on Epitaxial Graphene Grown on 4H-SiC Substrates by Using Ethene Chemical Vapor Deposition. Materials. 2015; 8(9):5586-5596. https://doi.org/10.3390/ma8095263
Chicago/Turabian StyleCai, Shuxian, Zhonghua Liu, Ni Zhong, Shengbei Liu, and Xingfang Liu. 2015. "Effect of Growth Pressure on Epitaxial Graphene Grown on 4H-SiC Substrates by Using Ethene Chemical Vapor Deposition" Materials 8, no. 9: 5586-5596. https://doi.org/10.3390/ma8095263
APA StyleCai, S., Liu, Z., Zhong, N., Liu, S., & Liu, X. (2015). Effect of Growth Pressure on Epitaxial Graphene Grown on 4H-SiC Substrates by Using Ethene Chemical Vapor Deposition. Materials, 8(9), 5586-5596. https://doi.org/10.3390/ma8095263