Highly Stretchable, Biocompatible, Striated Substrate Made from Fugitive Glue
Abstract
:1. Introduction
2. Results
2.1. Stability
2.1.1. Mechanical Stability and Stretchability
2.1.2. pH Tolerance Test

2.1.3. Salt Solution Tolerance Test

2.2. Bio-Compatibility
2.2.1. Cell Growth on Fugitive Glue Substrate
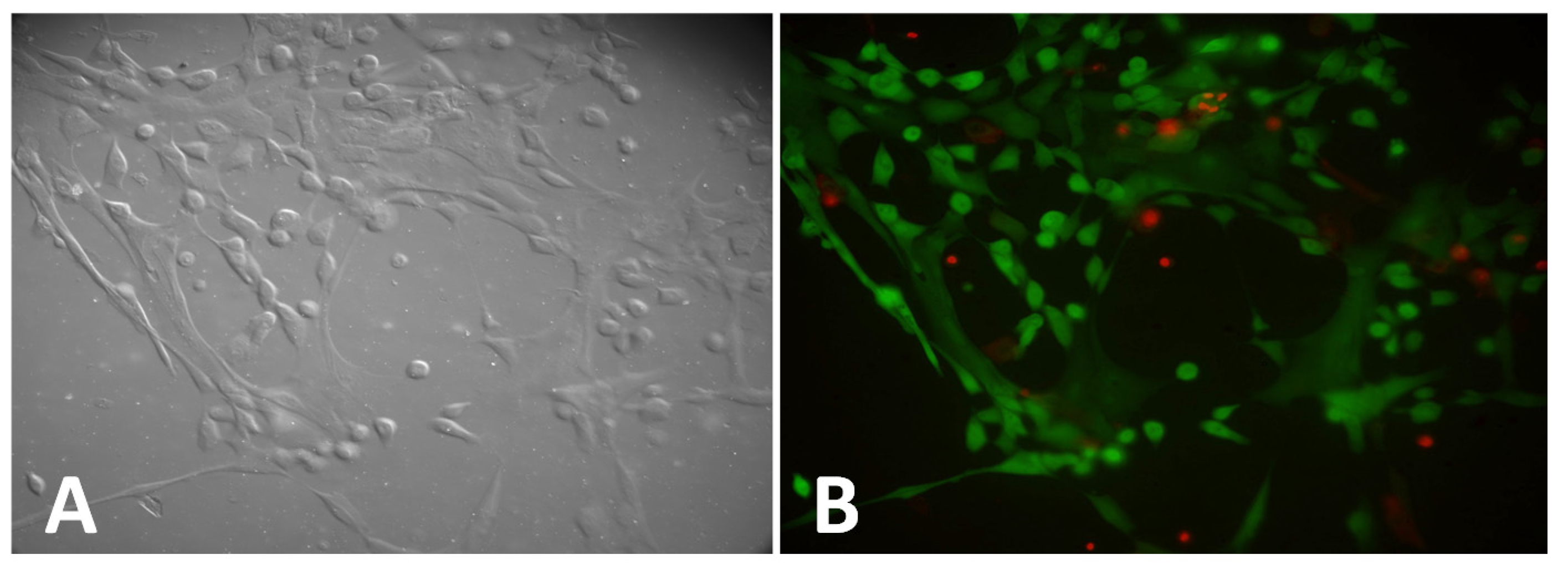
2.2.2. Fibrin Fiber Formed on Fugitive Glue Substrate

3. Discussion
4. Experimental Section
4.1. Stretchable Substrate Preparation

4.2. pH and Salt Solution Tolerance Test
4.3. Fibrin Fibers
4.4. Cell Growth
4.5. Cell Viability Assay
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Richard, M.N.; Deniset, J.F.; Kneesh, A.L.; Blackwood, D.; Pierce, G.N. Mechanical stretching stimulates smooth muscle cell growth, nuclear protein import, and nuclear pore expression through mitogen-activated protein kinase activation. J. Biol. Chem. 2007, 282, 23081–23088. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Bacabac, R.G.; Bakker, A.D. Mechanical Loading and How it Affects Bone Cells: The Role of the Osteocyte Cytoskeleton in Maintaining Our Skeleton. Eur. Cells Mater. 2012, 24, 278–291. [Google Scholar]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef]
- Kurpinski, K.; Chu, J.; Hashi, C.; Li, S. Anisotropic mechanosensing by mesenchymal stem cells. Proc. Natl. Acad. Sci. USA 2006, 103, 16095–16100. [Google Scholar] [CrossRef]
- Kim, T.J.; Sun, J.; Lu, S.Y.; Qi, Y.X.; Wang, Y.X. Prolonged Mechanical Stretch Initiates Intracellular Calcium Oscillations in Human Mesenchymal Stem Cells. PLoS ONE 2014, 9, e109378. [Google Scholar] [CrossRef]
- Park, J.S.; Chu, J.S.F.; Cheng, C.; Chen, F.Q.; Chen, D.; Li, S. Differential effects of equiaxial and uniaxial strain on mesenchymal stem cells. Biotechnol. Bioeng. 2004, 88, 359–368. [Google Scholar] [CrossRef]
- Gersh, K.C.; Edmondson, K.E.; Weisel, J.W. Flow rate and fibrin fiber alignment. J. Thromb. Haemost. 2010, 8, 2826–2828. [Google Scholar] [CrossRef]
- Campbell, R.A.; Aleman, M.M.; Gray, L.D.; Falvo, M.R.; Wolberg, A.S. Flow profoundly influences fibrin network structure: Implications for fibrin formation and clot stability in haemostasis. Thromb. Haemost. 2010, 104, 1281–1284. [Google Scholar] [CrossRef]
- Weiss, H.J.; Turitto, V.T.; Baumgartner, H.R. Role Of Shear Rate and Platelets in Promoting Fibrin Formation on Rabbit Subendothelium. Studies Utilizing Patients With Quantitative and Qualitative Platelet Defects. J. Clin. Invest. 1986, 78, 1072–1082. [Google Scholar] [CrossRef] [PubMed]
- Lemos, M.; Pozo, R.M.K.; Montes, G.S.; Saldiva, P.H.N. Organization of collagen and elastic fibers studied in stretch preparations of whole mounts of human visceral pleura. Ann. Anat. Anat. Anz. 1997, 179, 447–452. [Google Scholar] [CrossRef]
- Römgens, A.M.; van Donkelaar, C.C.; Ito, K. Contribution of collagen fibers to the compressive stiffness of cartilaginous tissues. Biomech. Model. Mechanobiol. 2013, 12, 1221–1231. [Google Scholar] [CrossRef]
- Munevar, S.; Wang, Y.L.; Dembo, M. Traction force microscopy of migrating normal and H-ras transformed 3T3 fibroblasts. Biophys. J. 2001, 80, 1744–1757. [Google Scholar] [CrossRef]
- Shao, Y.; Tan, X.Y.; Novitski, R.; Muqaddam, M.; List, P.; Williamson, L.; Fu, J.P.; Liu, A.P. Uniaxial cell stretching device for live-cell imaging of mechanosensitive cellular functions. Rev. Sci. Instrum. 2013, 84, 114304. [Google Scholar] [CrossRef] [PubMed]
- Richardson, W.J.; Metz, R.P.; Moreno, M.R.; Wilson, E.; Moore, J.E. A Device to Study the Effects of Stretch Gradients on Cell Behavior. J. Biomech. Eng. 2011, 133, 101008. [Google Scholar] [CrossRef]
- Liu, W.; Carlisle, C.R.; Sparks, E.A.; Guthold, M. The mechanical properties of single fibrin fibers. J. Thromb. Haemost. 2010, 8, 1030–1036. [Google Scholar] [CrossRef]
- Liu, W.; Jawerth, L.M.; Sparks, E.A.; Falvo, M.R.; Hantgan, R.R.; Superfine, R.; Lord, S.T.; Guthold, M. Fibrin fibers have extraordinary extensibility and elasticity. Science 2006, 313, 634. [Google Scholar] [CrossRef]
- Carlisle, C.R.; Coulais, C.; Guthold, M. The mechanical stress–strain properties of single electrospun collagen type I nanofibers. Acta Biomater. 2010, 6, 2997–3003. [Google Scholar] [CrossRef]
- Baker, S.; Sigley, J.; Helms, C.C.; Stitzel, J.; Berry, J.; Bonin, K.; Guthold, M. The mechanical properties of dry, electrospun fibrinogen fibers. Mater. Sci. Eng. C 2012, 32, 215–221. [Google Scholar] [CrossRef]
- Varjú, I.; Sótonyi, P.; Machovich, R.; Szabó, L.; Tenekedjiev, K.; Silva, M.M.C.G.; Longstaff, C.; Kolev, K. Hindered dissolution of fibrin formed under mechanical stress. J. Thromb. Haemost. 2011, 9, 979–986. [Google Scholar] [CrossRef]
- Hong, S.Y.; Yoon, J.; Jin, S.W.; Lim, Y.; Lee, S.J.; Zi, G.; Ha, J.S. High-Density, Stretchable, All-Solid-State Microsupercapacitor Arrays. ACS Nano 2014, 8, 8844–8855. [Google Scholar] [CrossRef]
- Lim, B.Y.; Yoon, J.; Yun, J.; Kim, D.; Hong, S.Y.; Lee, S.-J.; Zi, G.; Ha, J.S. Biaxially stretchable, integrated array of high performance microsupercapacitors. ACS Nano 2014, 8, 11639–11650. [Google Scholar] [CrossRef]
- Lazarus, N.; Meyer, C.D.; Bedair, S.S.; Nochetto, H.; Kierzewski, I.M. Multilayer liquid metal stretchable inductors. Smart Mater. Struct. 2014, 23, 085036. [Google Scholar] [CrossRef]
- Robinson, A.; Aziz, A.; Liu, Q.; Suo, Z.; Lacour, S.P. Hybrid stretchable circuits on silicone substrate. J. Appl. Phys. 2014, 115, 143511. [Google Scholar] [CrossRef]
- Xu, P.; Mark, J.E. Elasticity Measurements on Bimodal Networks in Elongation and Compression: Networks Crosslinked in Solution and Studied Unswollen, and Networks Crosslinked in the Undiluted State and Studied Swollen. Polymer 1992, 33, 1843–1848. [Google Scholar] [CrossRef]
- Serrell, D.B.; Oreskovic, T.L.; Slifka, A.J.; Mahajan, R.L.; Finch, D.S. A uniaxial bioMEMS device for quantitative force-displacement measurements. Biomed. Microdevices 2007, 9, 267–275. [Google Scholar] [CrossRef]
- Yang, S.Y.; Saif, T. Reversible and repeatable linear local cell force response under large stretches. Exp. Cell Res. 2005, 305, 42–50. [Google Scholar] [CrossRef]
- Sim, G.D.; Won, S.; Jin, C.Y.; Park, I.; Lee, S.B.; Vlassak, J.J. Improving the stretchability of as-deposited Ag coatings on poly-ethylene-terephthalate substrates through use of an acrylic primer. J. Appl. Phys. 2011, 109, 073511. [Google Scholar] [CrossRef]
- Jahanshahi, A.; Salvo, P.; Vanfleteren, J. Stretchable biocompatible electronics by embedding electrical circuitry in biocompatible elastomers. In Proceedings of the 2012 IEEE Annual Conference on Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August 2012; pp. 6007–6010.
- Habrard, F.; Patscheider, J.; Kovacs, G. Stretchable Metallic Electrodes for Electroactive Polymer Actuators. Adv. Eng. Mater. 2014, 16, 1133–1139. [Google Scholar] [CrossRef]
- Leong, K.W.; Brott, B.C.; Langer, R. Bioerodible Polyanhydeides as Drug-Carrier Matrices. I. Characterization, Degradation, and Release Characteristics. J. Biomed. Mater. Res. 1985, 19, 941–955. [Google Scholar] [CrossRef]
- Li, J.; Jiang, G.Q.; Ding, F.X. The effect of pH on the polymer degradation and drug release from PLGA-mPEG microparticles. J. Appl. Polym. Sci. 2008, 109, 475–482. [Google Scholar] [CrossRef]
- Grayson, A.C.R.; Shawgo, R.S.; Johnson, A.M.; Flynn, N.T.; Li, Y.W.; Cima, M.J.; Langer, R. A BioMEMS review: MEMS technology for physiologically integrated devices. Proc. IEEE 2004, 92, 6–21. [Google Scholar] [CrossRef]
- Heo, Y.J.; Kan, T.; Iwase, E.; Matsumoto, K.; Shimoyama, I. Stretchable cell culture platforms using micropneumatic actuators. Micro Nano Lett. 2013, 8, 865–868. [Google Scholar] [CrossRef]
- Kamotani, Y.; Bersano-Begey, T.; Kato, N.; Tung, Y.C.; Huh, D.; Song, J.W.; Takayama, S. Individually programmable cell stretching microwell arrays actuated by a Braille display. Biomaterials 2008, 29, 2646–2655. [Google Scholar] [CrossRef]
- Yang, S.; Saif, M.T.A. Microfabricated Force Sensors and Their Applications in the Study of Cell Mechanical Response. Exp. Mech. 2009, 49, 135–151. [Google Scholar] [CrossRef]
- Wipff, P.J.; Majd, H.; Acharya, C.; Buscemi, L.; Meister, J.J.; Hinz, B. The covalent attachment of adhesion molecules to silicone membranes for cell stretching applications. Biomaterials 2009, 30, 1781–1789. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.; Lucioni, T.; Guo, X.; Smelser, A.; Guthold, M. Highly Stretchable, Biocompatible, Striated Substrate Made from Fugitive Glue. Materials 2015, 8, 3508-3518. https://doi.org/10.3390/ma8063508
Li W, Lucioni T, Guo X, Smelser A, Guthold M. Highly Stretchable, Biocompatible, Striated Substrate Made from Fugitive Glue. Materials. 2015; 8(6):3508-3518. https://doi.org/10.3390/ma8063508
Chicago/Turabian StyleLi, Wei, Tomas Lucioni, Xinyi Guo, Amanda Smelser, and Martin Guthold. 2015. "Highly Stretchable, Biocompatible, Striated Substrate Made from Fugitive Glue" Materials 8, no. 6: 3508-3518. https://doi.org/10.3390/ma8063508
APA StyleLi, W., Lucioni, T., Guo, X., Smelser, A., & Guthold, M. (2015). Highly Stretchable, Biocompatible, Striated Substrate Made from Fugitive Glue. Materials, 8(6), 3508-3518. https://doi.org/10.3390/ma8063508





