Catalytically Enhanced Hydrogen Sorption in Mg-MgH2 by Coupling Vanadium-Based Catalyst and Carbon Nanotubes
Abstract
:1. Introduction
2. Experimental
2.1. Synthesis of VCat
2.2. Synthesis of MgH2 Storage Composites
2.3. Characterization
3. Result and Discussion
3.1. Characterization of Vanadium-Based Complex Catalyst
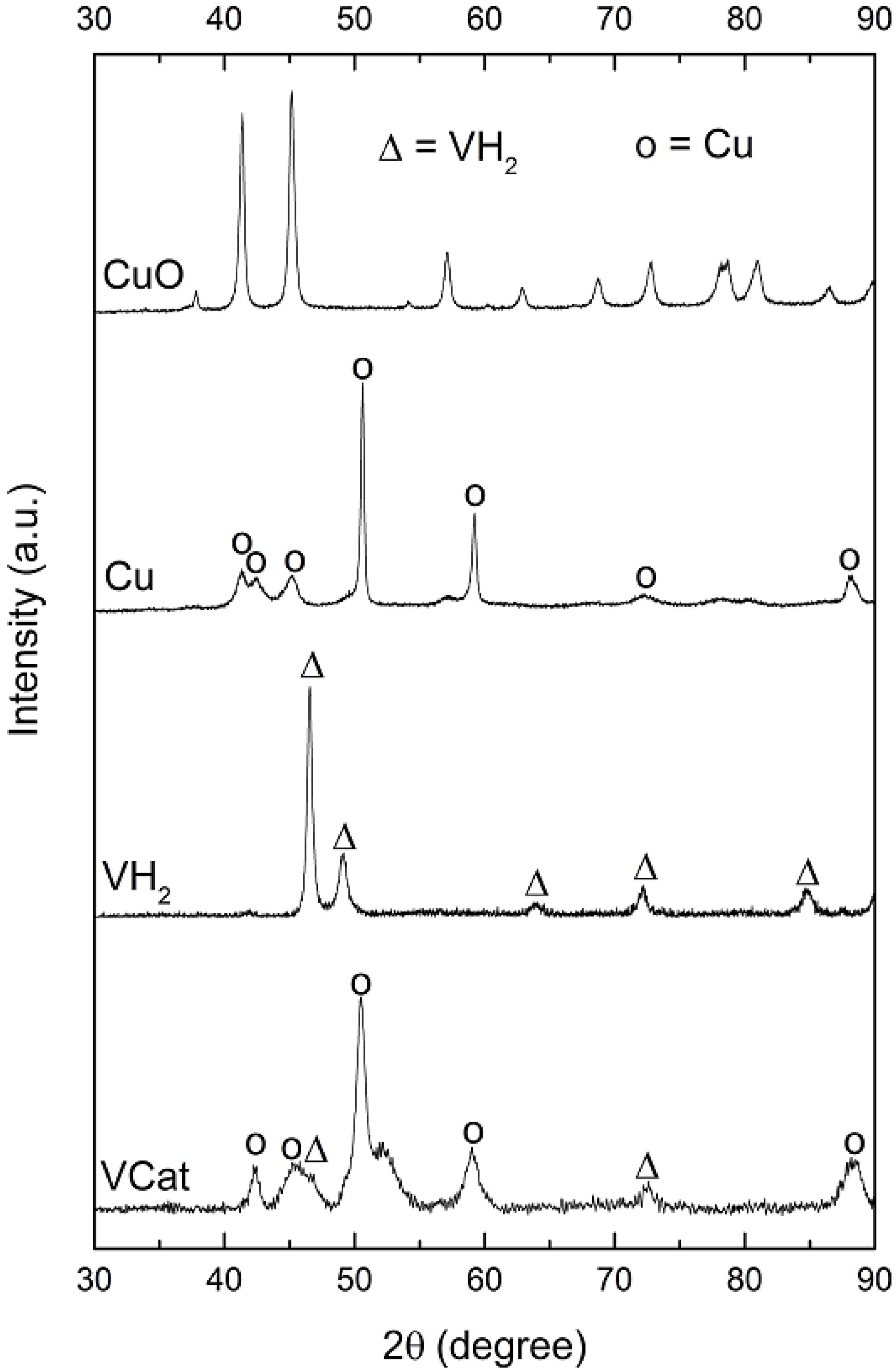
3.2. Characterization of MgH2-Based Composites
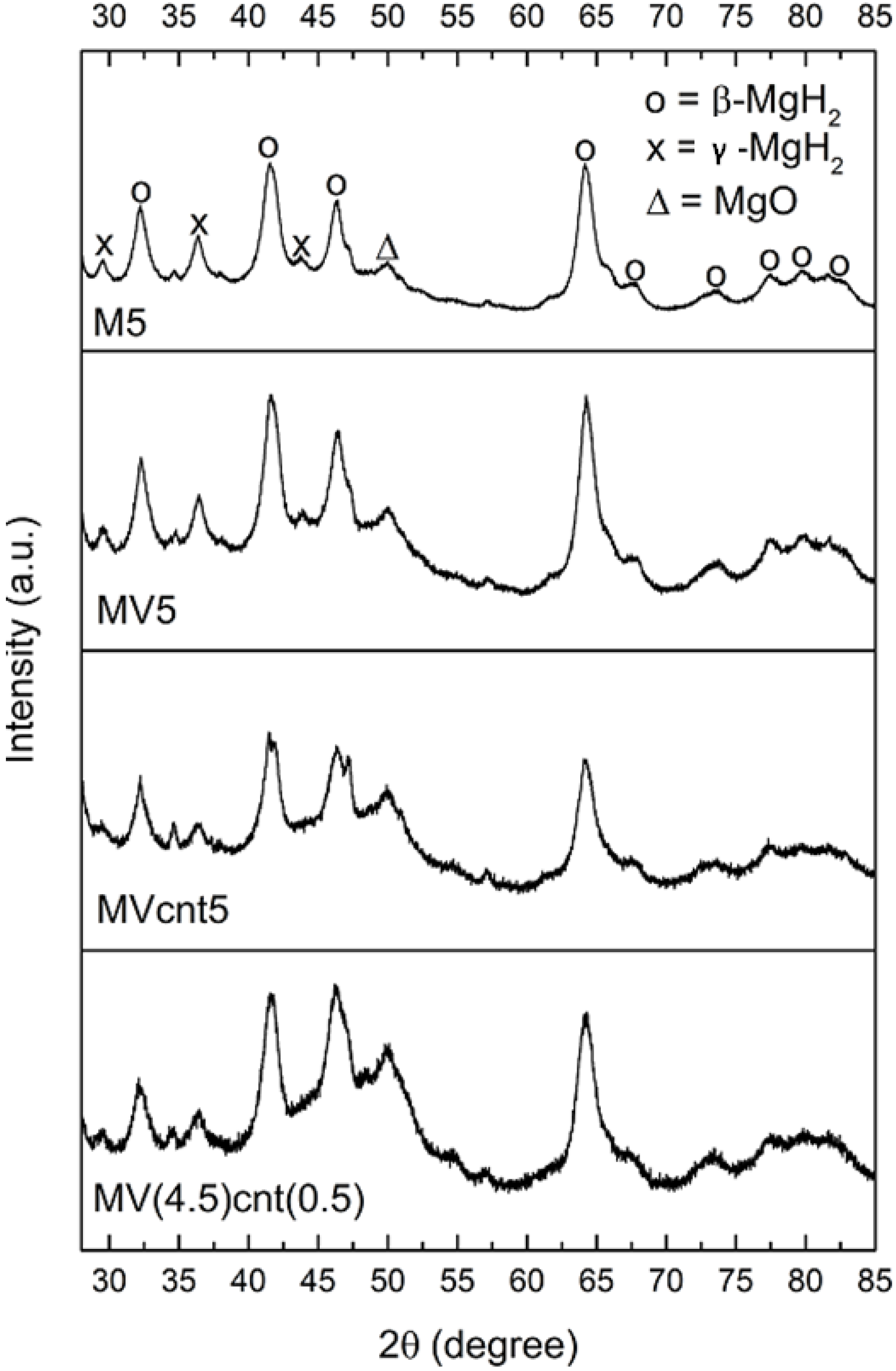
| Sample | β-MgH2 Grain Size (nm) |
|---|---|
| M5 | 11.6 |
| MV5 | 10.0 |
| MV(4.5)cnt(0.5) | 9.3 |
| MVcnt5 | 9.5 |
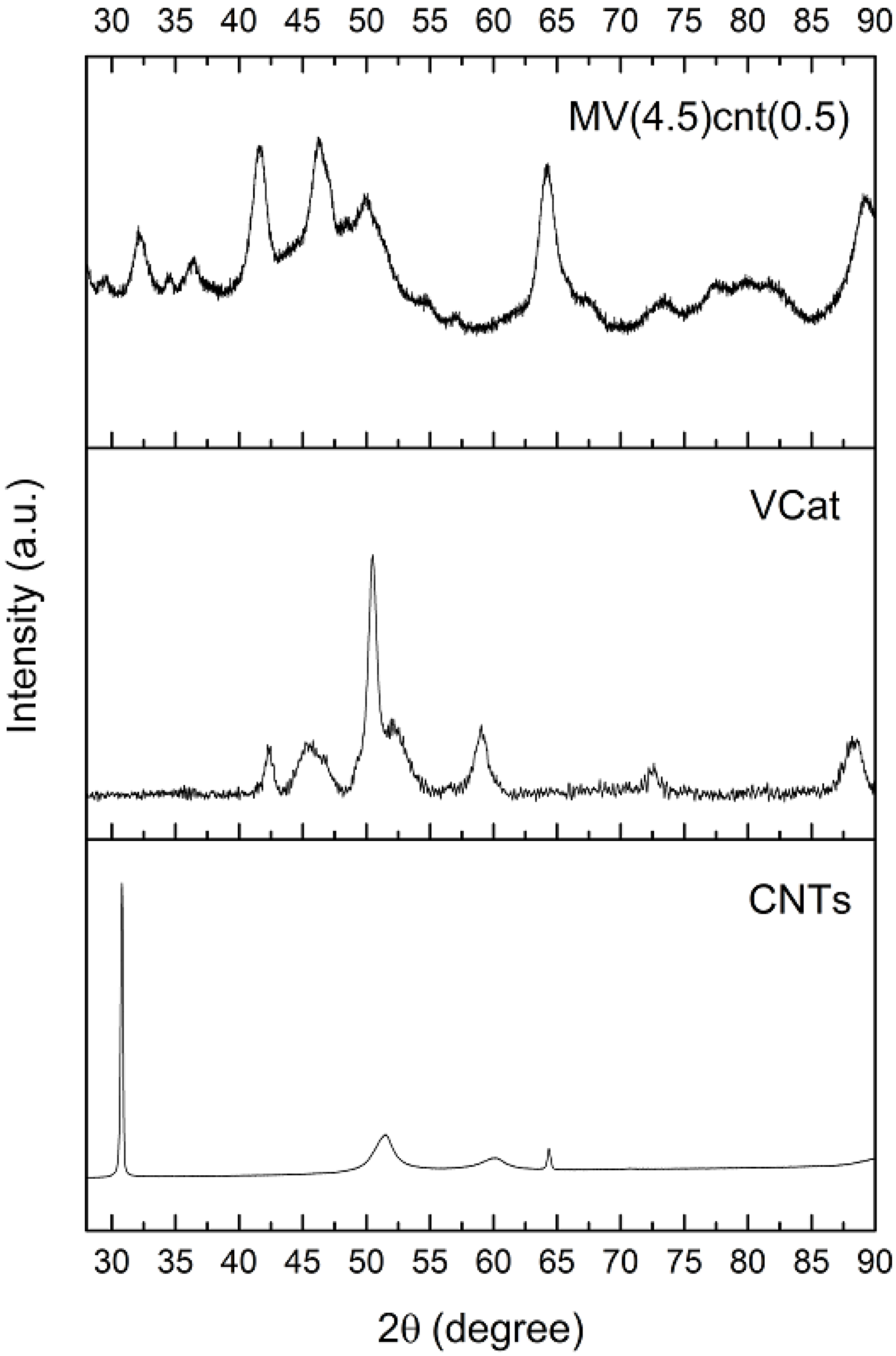
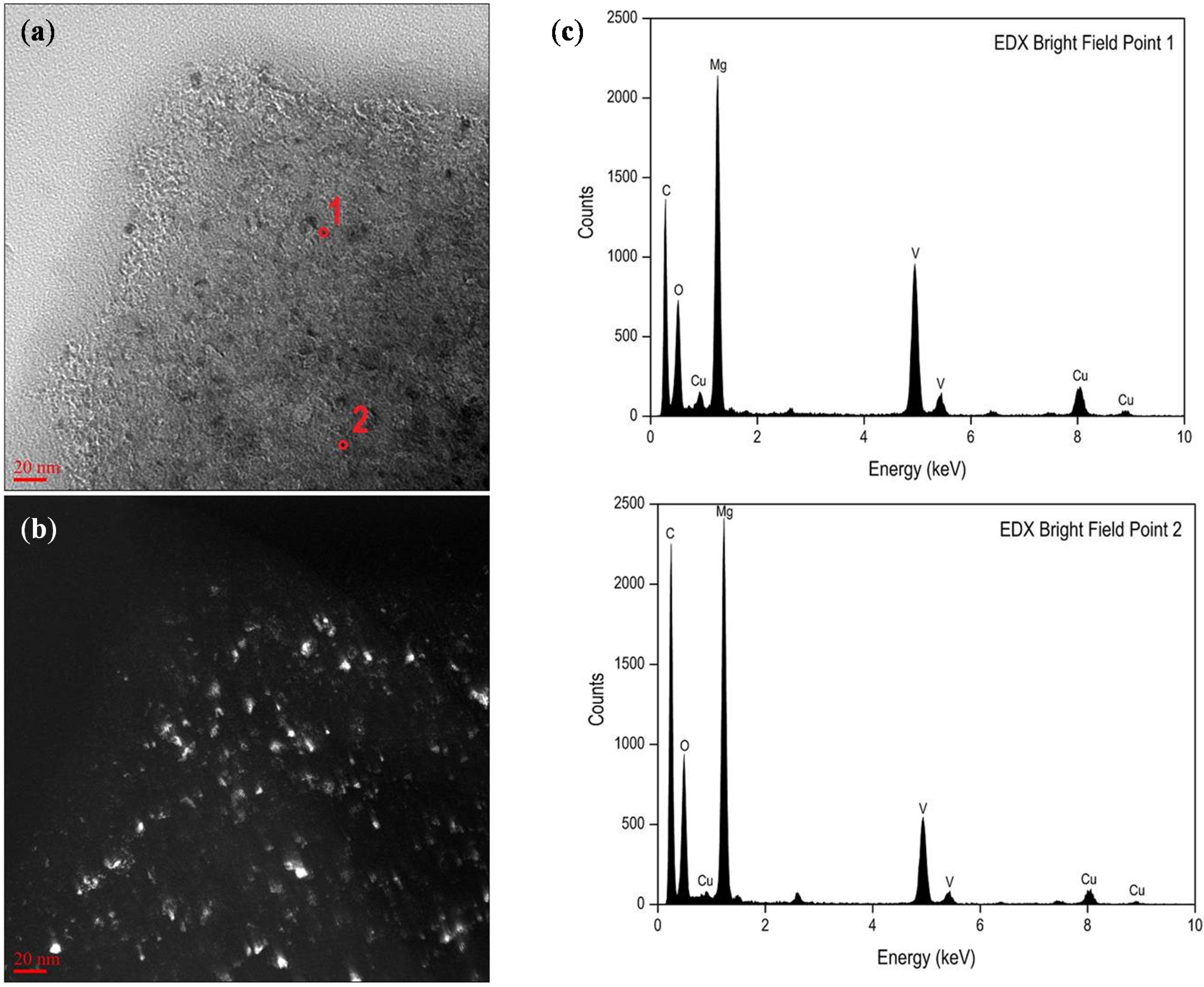
3.3. Tracing the Degree of Damage of CNTs in MgH2-Based Composites
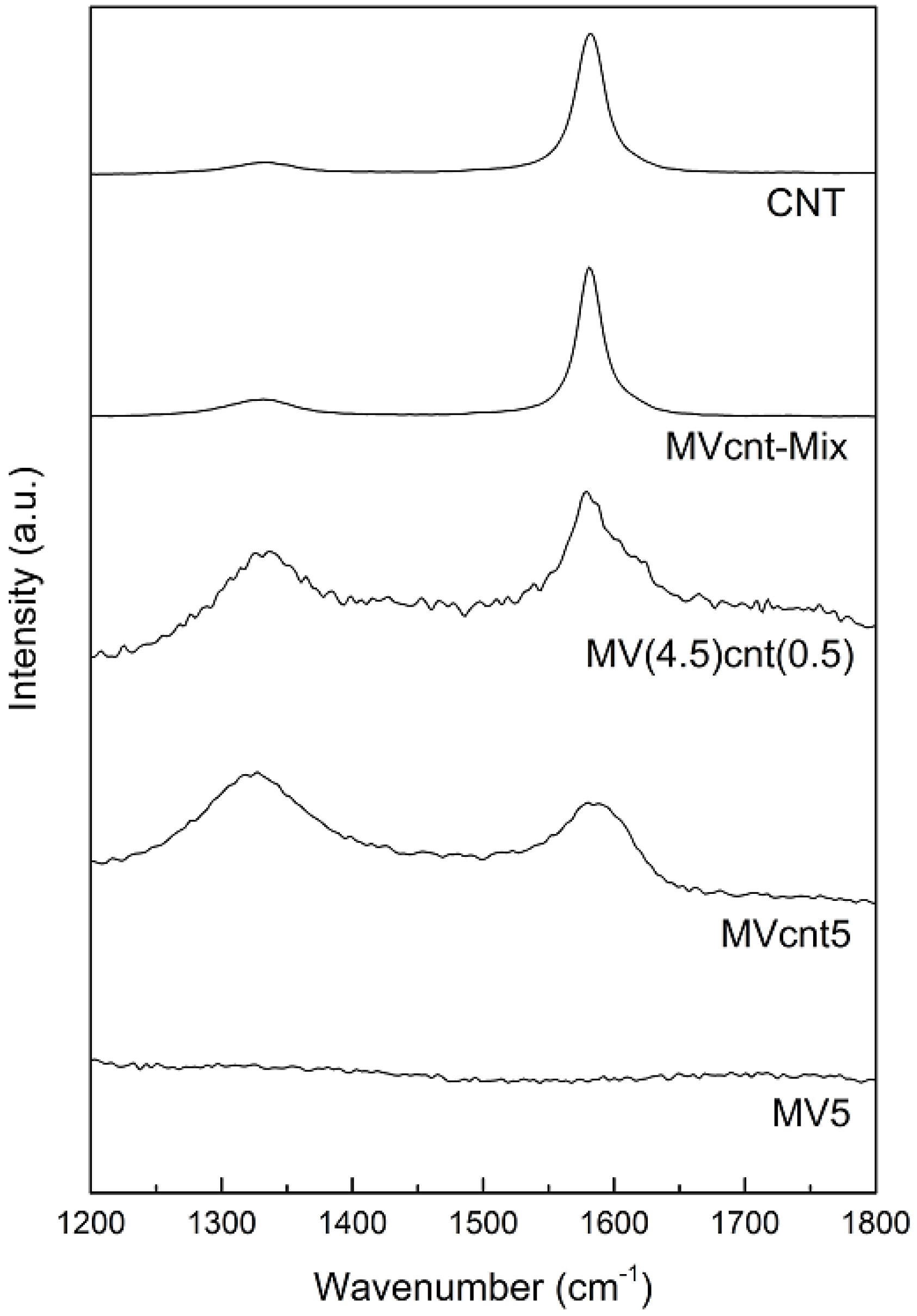
3.4. Hydrogen Storage Properties
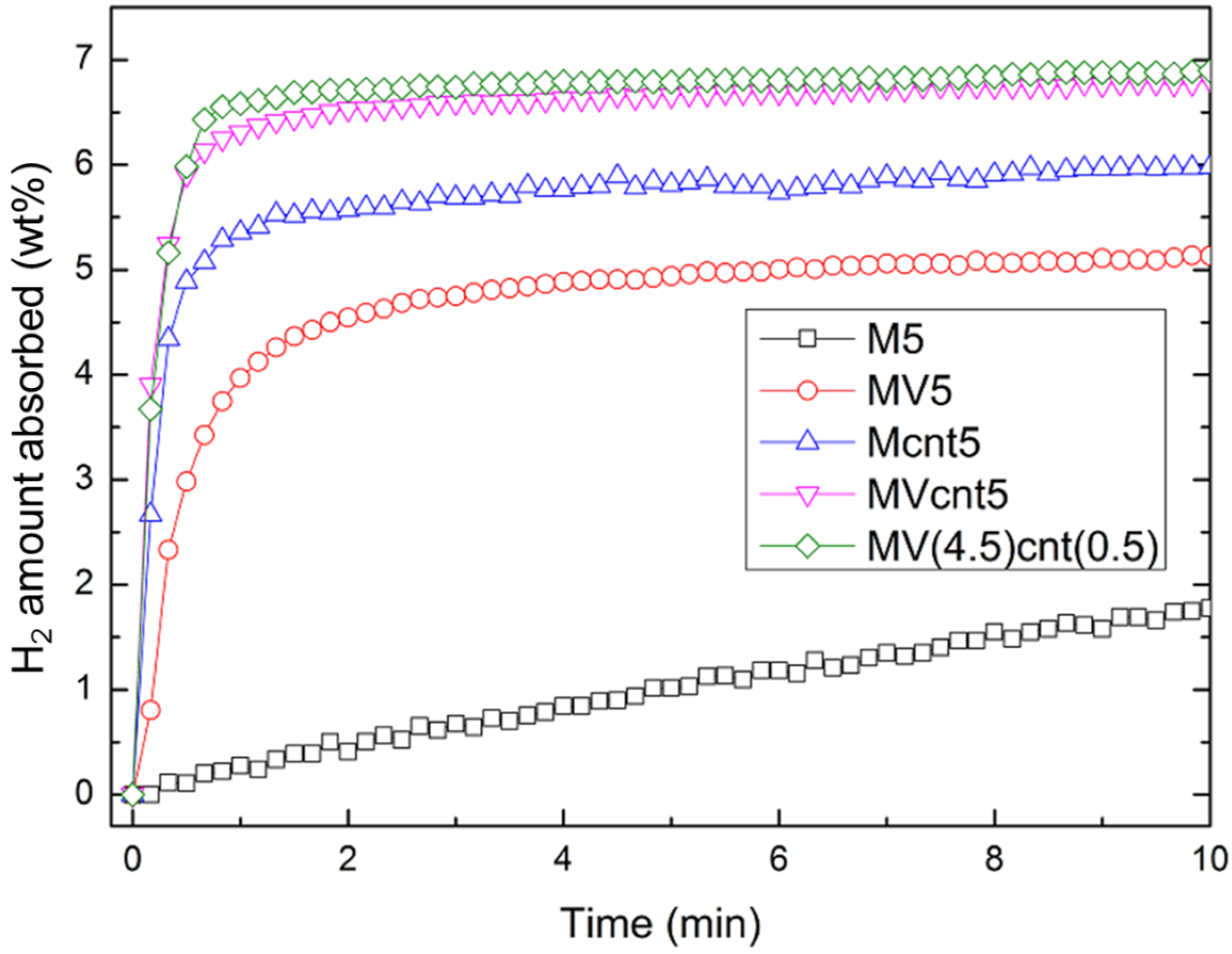
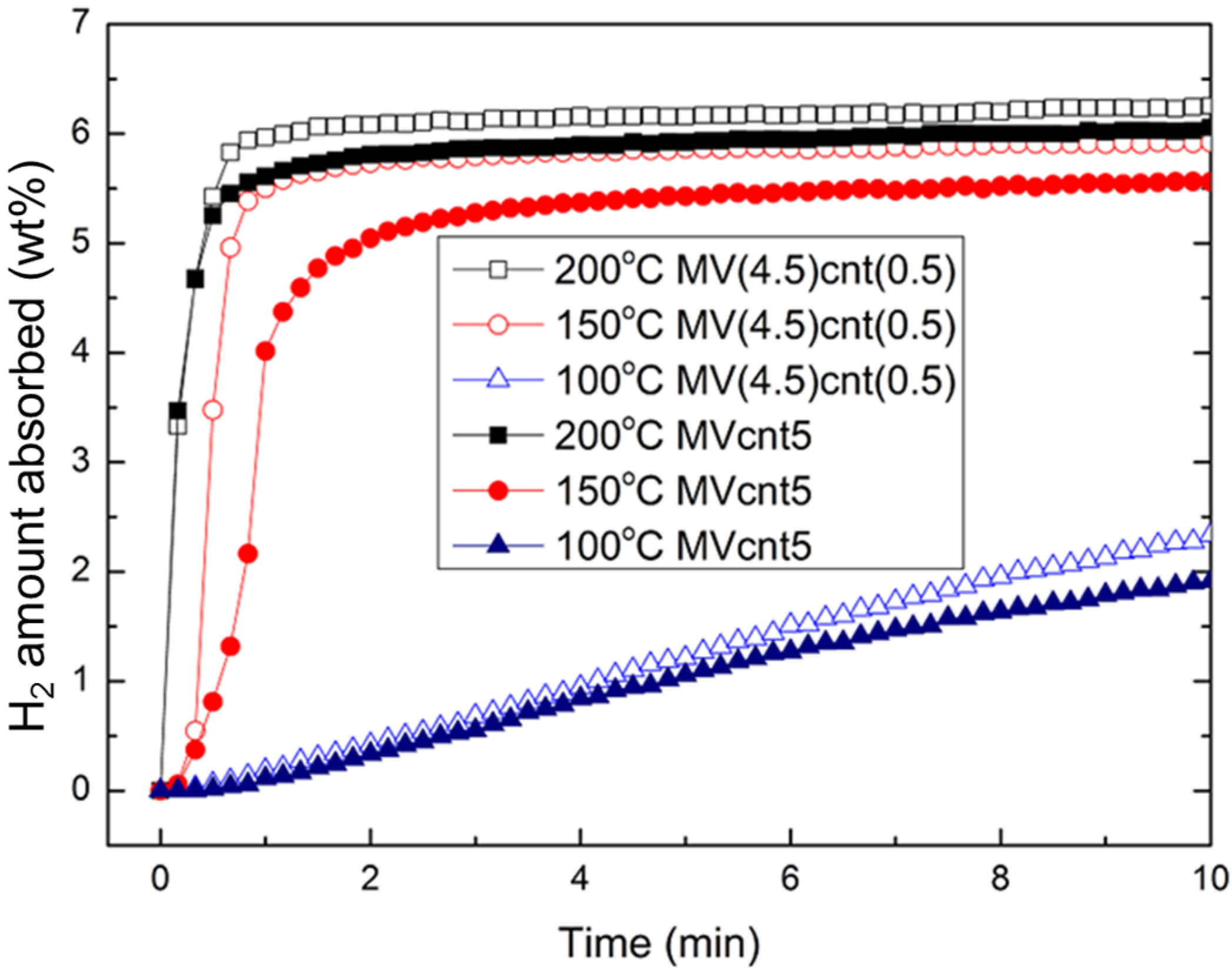
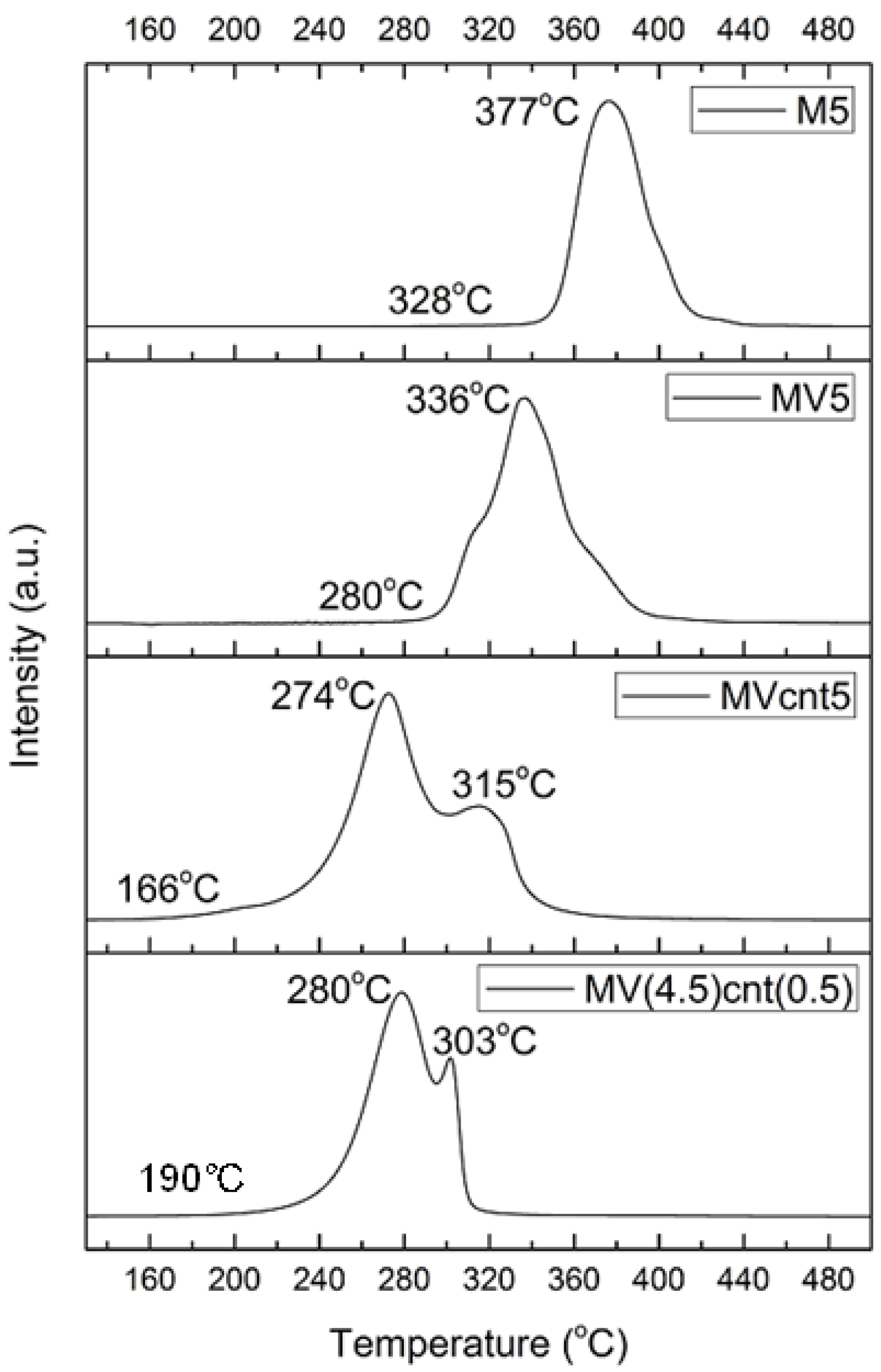
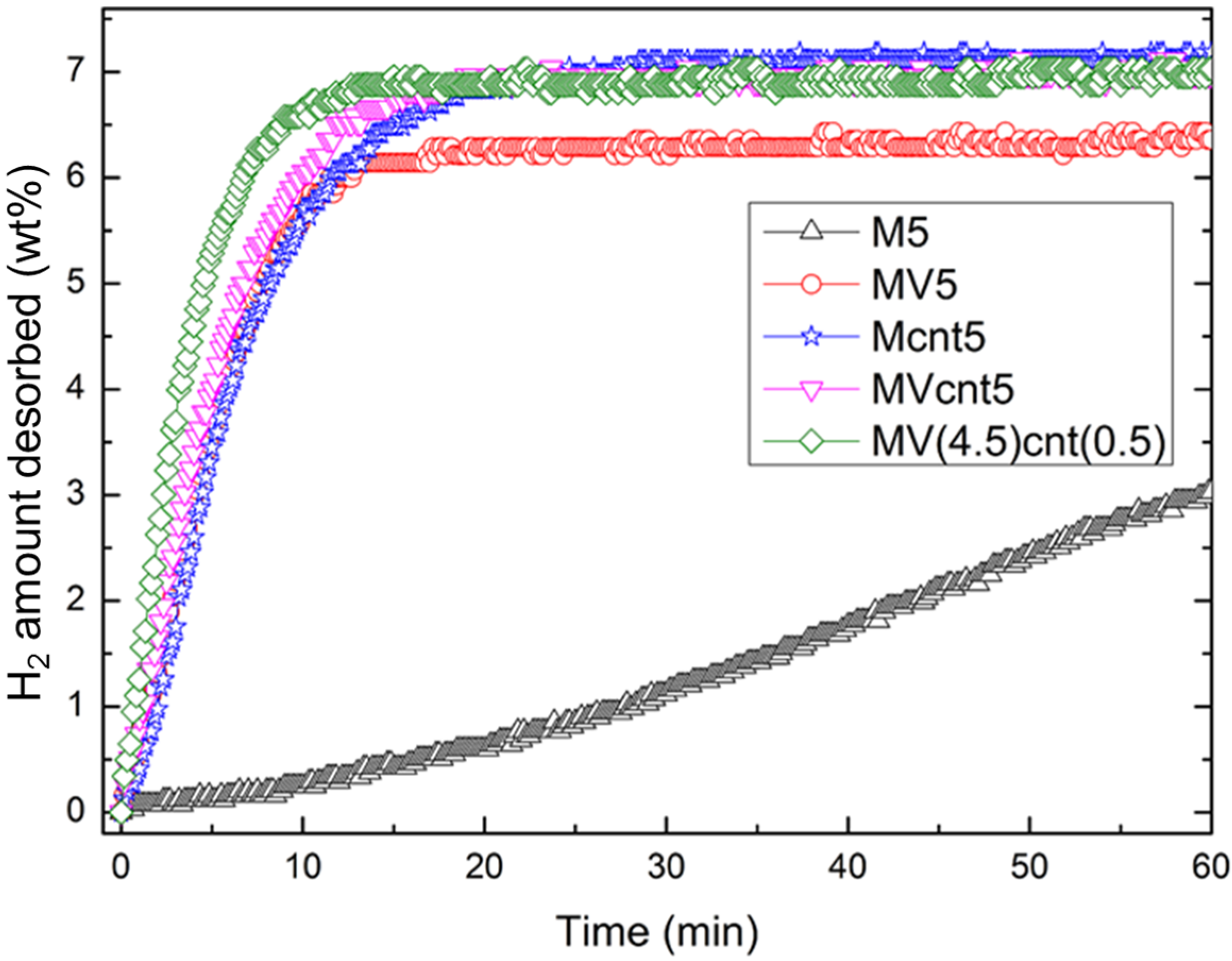
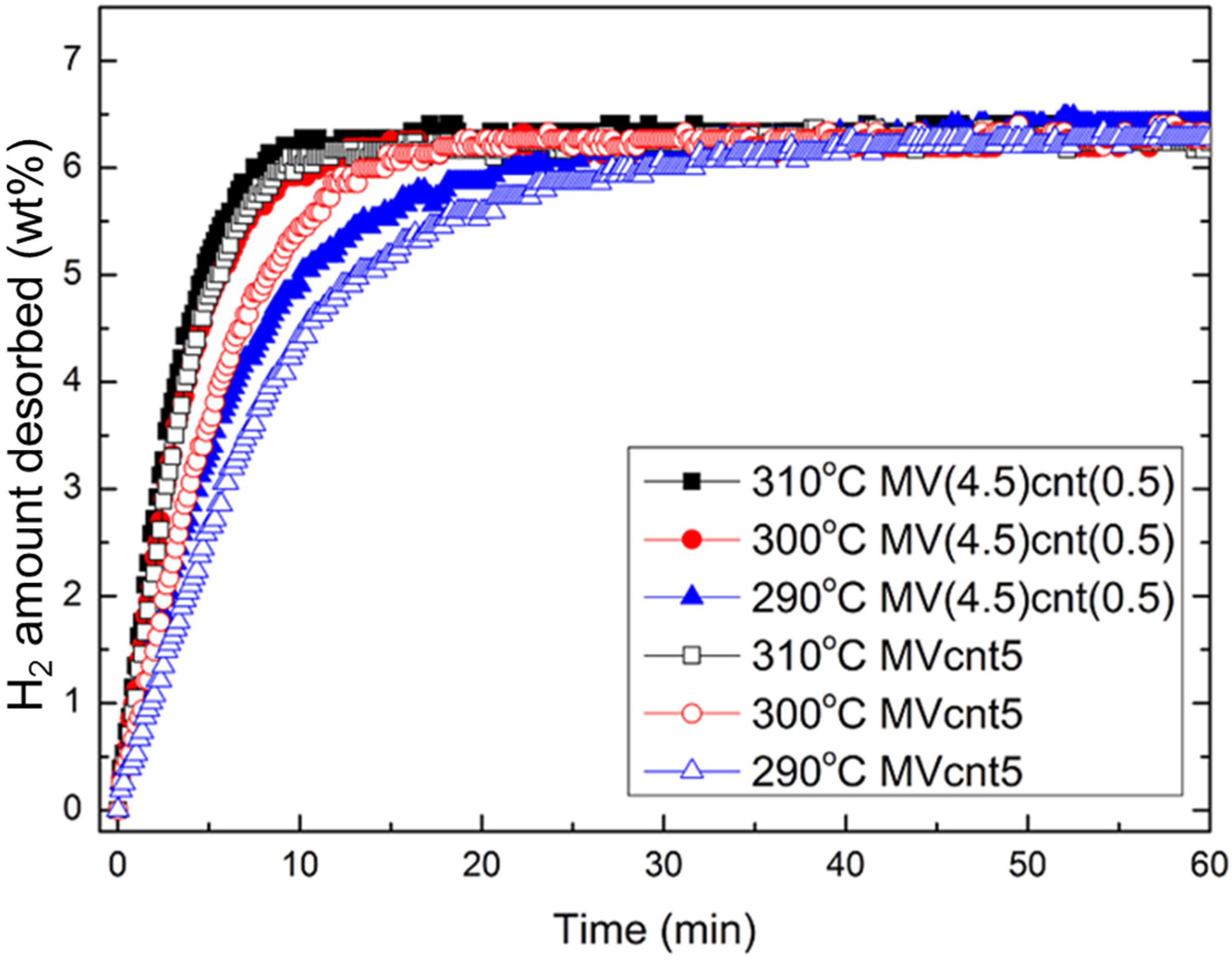
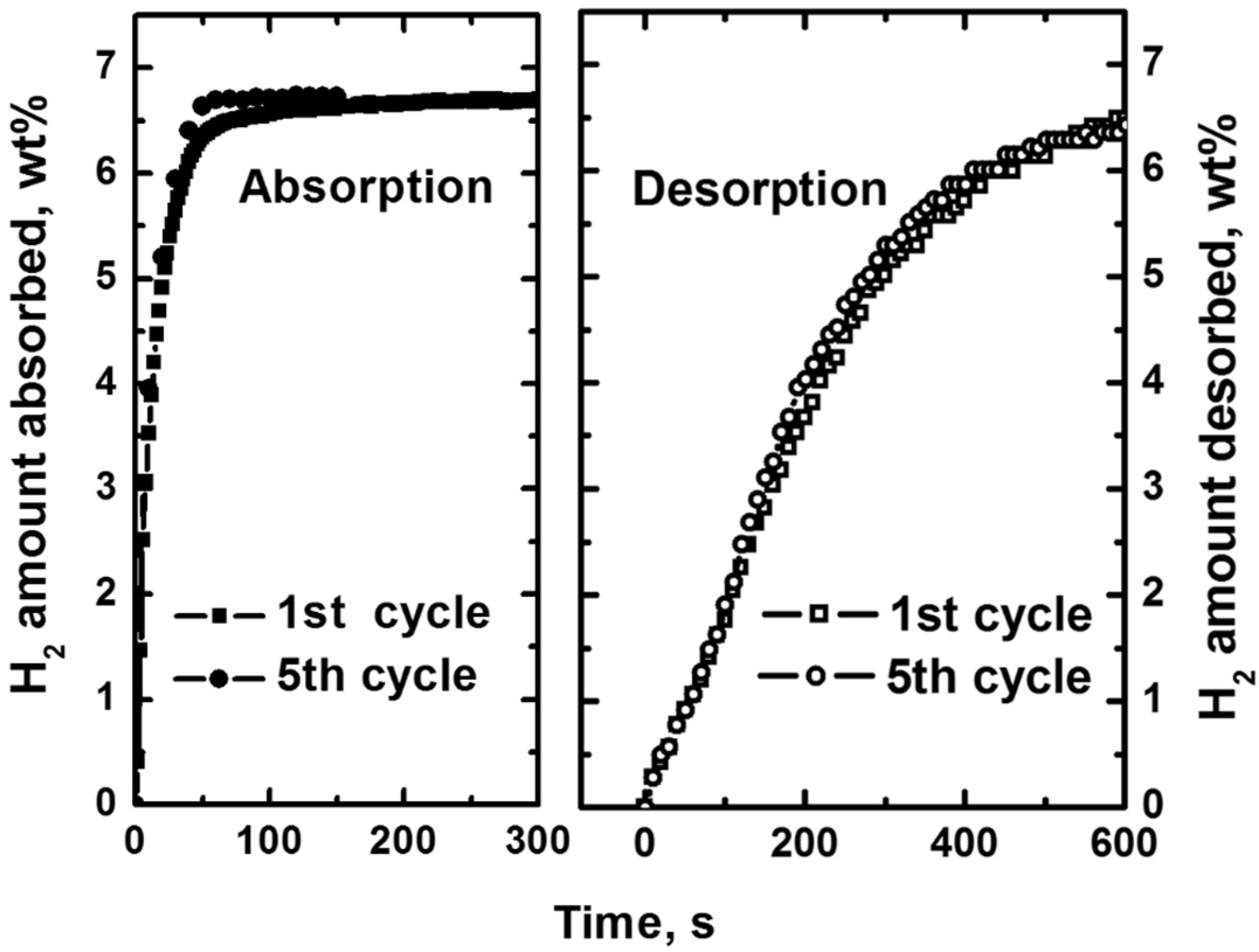
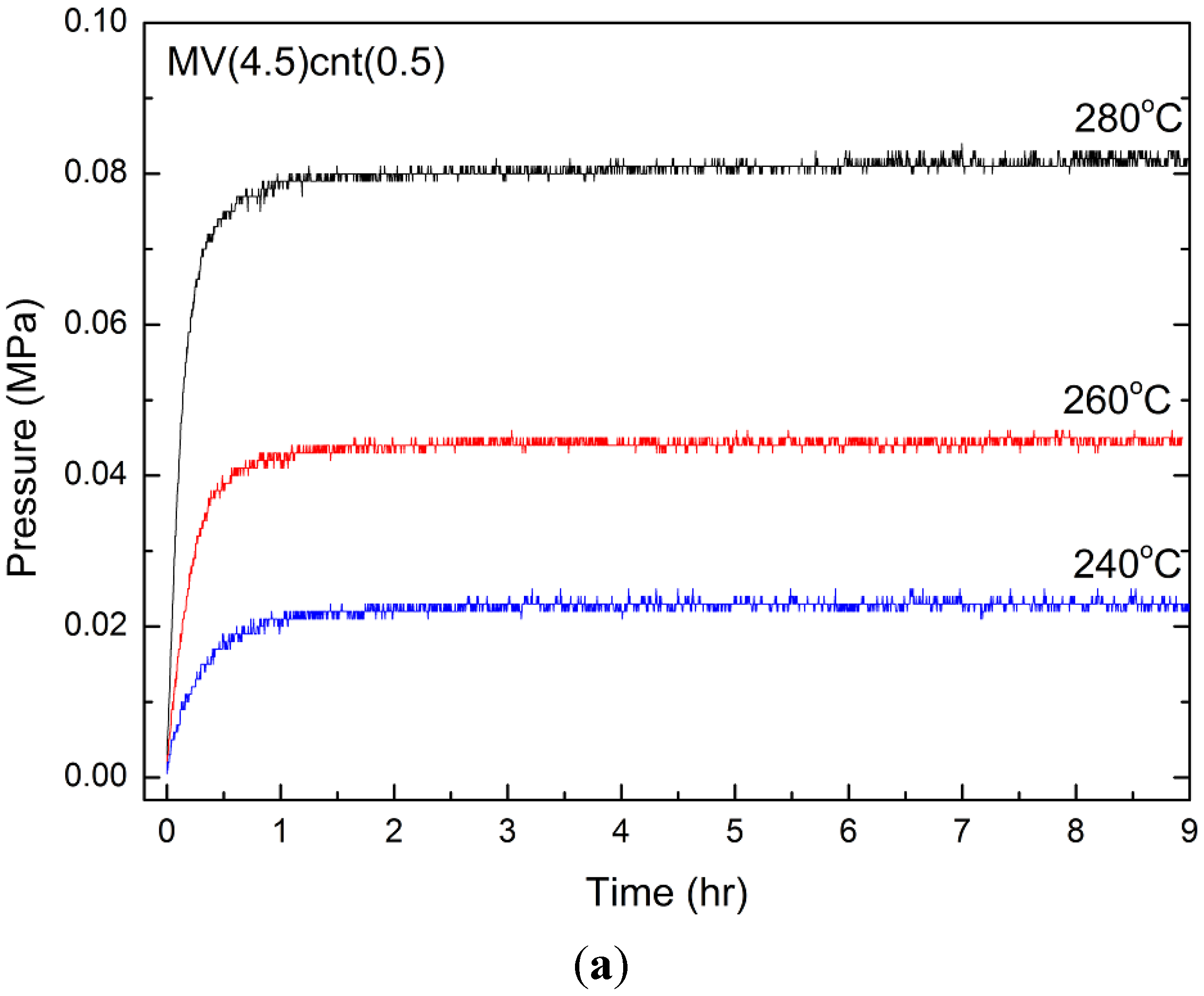
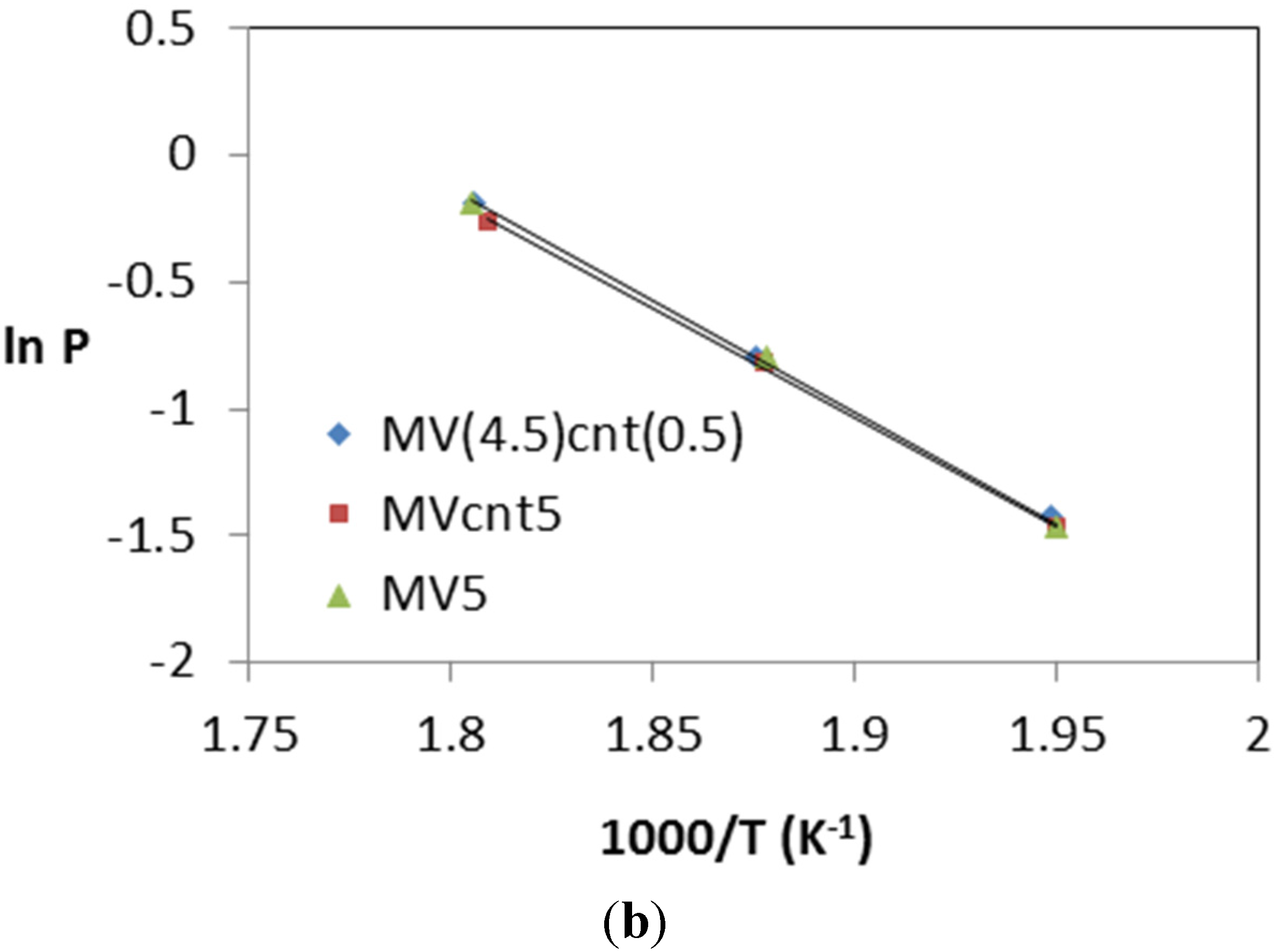
| Sample | Desorption Enthalpy, ΔH (kJ/mol H2) | Desorption Entropy, ΔS (J/mol H2·K) |
|---|---|---|
| MV5 | 73.9 | 132.0 |
| MVcnt5 | 71.6 | 127.4 |
| MV(4.5)cnt(0.5) | 72.2 | 128.8 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stampfer, J.F.; Holley, C.E.; Suttle, J.F. The Magnesium Hydrogen System. J. Am. Chem. Soc. 1960, 82, 3504–3508. [Google Scholar] [CrossRef]
- Lu, H.B.; Poh, C.K.; Zhang, L.C.; Guo, Z.P.; Yu, X.B.; Liu, H.K. Dehydrogenation characteristics of Ti- and Ni/Ti-catalyzed Mg hydrides. J. Alloys Compd. 2009, 481, 152–155. [Google Scholar] [CrossRef]
- Friedrichs, O.; Aguey-Zinsou, F.; Fernandez, J.R.A.; Sanchez-Lopez, J.C.; Justo, A.; Klassen, T.; Bormann, R.; Fernandez, A. MgH2 with Nb2O5 as additive, for hydrogen storage: Chemical, structural and kinetic behavior with heating. Acta Mater. 2006, 54, 105–110. [Google Scholar] [CrossRef]
- Barkhordarian, G.; Klassen, T.; Bormann, R. Effect of Nb2O5 content on hydrogen reaction kinetics of Mg. J. Alloys Compd. 2004, 364, 242–246. [Google Scholar] [CrossRef]
- Barkhordarian, G.; Klassen, T.; Bormann, R. Fast hydrogen sorption kinetics of nanocrystalline Mg using Nb2O5 as catalyst. Scr. Mater. 2003, 49, 213–217. [Google Scholar] [CrossRef]
- Khandelwal, A.; Agresti, F.; Capurso, G.; Russo, S.L.; Maddalena, A.; Gialanella, S.; Principi, G. Pellets of MgH(2)-based composites as practical material for solid state hydrogen storage. Int. J. Hydrog. Energy 2010, 35, 3565–3571. [Google Scholar] [CrossRef]
- Ma, L.P.; Kang, X.D.; Dai, H.B.; Liang, Y.; Fang, Z.Z.; Wang, P.J.; Wang, P.; Cheng, H.M. Superior catalytic effect of TiF3 over TiCl3 in improving the hydrogen sorption kinetics of MgH2: Catalytic role of fluorine anion. Acta Mater. 2009, 57, 2250–2258. [Google Scholar] [CrossRef]
- Ma, L.P.; Wang, P.; Cheng, H.M. Improving hydrogen sorption kinetics of MgH2 by mechanical milling with TiF3. J. Alloys Compd. 2007, 432, L1–L4. [Google Scholar] [CrossRef]
- Yavari, A.R.; LeMoulec, A.; de Castro, F.R.; Deledda, S.; Friedrichs, O.; Botta, W.J.; Vaughan, G.; Klassen, T.; Fernandez, A.; Kvick, A. Improvement in H-sorption kinetics of MgH2 powders by using Fe nanoparticles generated by reactive FeF3 addition. Scr. Mater. 2005, 52, 719–724. [Google Scholar] [CrossRef]
- Luo, Y.; Wang, P.; Ma, L.P.; Cheng, H.M. Hydrogen sorption kinetics of MgH2 catalyzed with NbF5. J. Alloys Compd. 2008, 453, 138–142. [Google Scholar] [CrossRef]
- Zaluska, A.; Zaluski, L. New catalytic complexes for metal hydride systems. J. Alloys Compd. 2005, 404–406, 706–711. [Google Scholar] [CrossRef]
- Adelhelm, P.; de Jongh, P.E. The impact of carbon materials on the hydrogen storage properties of light metal hydrides. J. Mater. Chem. 2011, 21, 2417–2427. [Google Scholar] [CrossRef]
- Wu, C.Z.; Cheng, H.M. Effects of carbon on hydrogen storage performances of hydrides. J. Mater. Chem. 2010, 20, 5390–5400. [Google Scholar] [CrossRef]
- Amirkhiz, B.S.; Danaie, M.; Mitlin, D. The influence of SWCNT-metallic nanoparticle mixtures on the desorption properties of milled MgH2 powders. Nanotechnology 2009, 20. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Z.; Wang, P.; Yao, X.D.; Liu, C.; Chen, D.M.; Lu, G.Q.; Cheng, H.M. Hydrogen storage properties of MgH2/SWNT composite prepared by ball milling. J. Alloys Compd. 2006, 420, 278–282. [Google Scholar] [CrossRef]
- Veron, M.G.; Troiani, H.; Gennari, F.C. Synergetic effect of Co and carbon nanotubes on MgH2 sorption properties. Carbon 2011, 49, 2413–2423. [Google Scholar] [CrossRef]
- Yao, X.D.; Wu, C.Z.; Du, A.J.; Lu, G.Q.; Cheng, H.M.; Smith, S.C.; Zou, J.; He, Y.H. Mg-based nanocomposites with high capacity and fast kinetics for hydrogen storage. J. Phys. Chem. B 2006, 110, 11697–11703. [Google Scholar] [CrossRef] [PubMed]
- Berube, V.; Radtke, G.; Dresselhaus, M.; Chen, G. Size effects on the hydrogen storage properties of nanostructured metal hydrides: A review. Int. J. Energy Res. 2007, 31, 637–663. [Google Scholar] [CrossRef]
- Cheung, S.; Deng, W.Q.; van Duin, A.C.T.; Goddard, W.A. ReaxFF(MgH) reactive force field for magnesium hydride systems. J. Phys. Chem. A 2005, 109, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Wagemans, R.W.P.; van Lenthe, J.H.; de Jongh, P.E.; van Dillen, A.J.; de Jong, K.P. Hydrogen Storage in Magnesium Clusters: Quantum Chemical Study. J. Am. Chem. Soc. 2005, 127, 16675–16680. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.G.; Guo, Z.P.; Calka, A.; Wexler, D.; Liu, H.K. Effects of carbon black, graphite and carbon nanotube additives on hydrogen storage properties of magnesium. J. Alloys Compd. 2007, 427, 94–100. [Google Scholar] [CrossRef]
- Yao, X.D.; Wu, C.Z.; Du, A.J.; Zou, J.; Zhu, Z.H.; Wang, P.; Cheng, H.M.; Smith, S.; Lu, G.Q. Metallic and carbon nanotube-catalyzed coupling of hydrogenation in magnesium. J. Am. Chem. Soc. 2007, 129, 15650–15654. [Google Scholar] [CrossRef] [PubMed]
- Amirkhiz, B.S.; Danaie, M.; Barnes, M.; Simard, B.; Mitlin, D. Hydrogen Sorption Cycling Kinetic Stability and Microstructure of Single-Walled Carbon Nanotube (SWCNT) Magnesium Hydride (MgH2) Nanocomposites. J. Phys. Chem. C 2010, 114, 3265–3275. [Google Scholar] [CrossRef]
- Kwon, S.; Baek, S.; Mumm, D.R.; Hong, S.-H.; Song, M. Enhancement of the hydrogen storage characteristics of Mg by reactive mechanical grinding with Ni, Fe and Ti. Int. J. Hydrog. Energy 2008, 33, 4586–4592. [Google Scholar] [CrossRef]
- Liang, G.; Huot, J.; Boily, S.; Schulz, R. Hydrogen desorption kinetics of a mechanically milled MgH2 + 5 at.% V nanocomposite. J. Alloys Compd. 2000, 305, 239–245. [Google Scholar] [CrossRef]
- Liang, G.; Huot, J.; Boily, S.; van Neste, A.; Schulz, R. Hydrogen storage properties of the mechanically milled MgH2-V nanocomposite. J. Alloys Compd. 1999, 291, 295–299. [Google Scholar] [CrossRef]
- He, Y.P.; Zhao, Y.P. Improved hydrogen storage properties of a V decorated Mg nanoblade array. Phys. Chem. Chem. Phys. 2009, 11, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Reilly, J.J.; Wiswall, R.H. Higher hydrides of vanadium and niobium. Inorg. Chem. 1970, 9, 1678–1682. [Google Scholar] [CrossRef]
- Zaluska, A.; Zaluski, L. New Type of Catalytic Materials Based on Active Metal-Hydrogen-Electronegative Element Complexes for Reactions Involving Hydrogen Transfer. WO 2004000453A2, 31 December 2003. [Google Scholar]
- Varin, R.A.; Czujko, T.; Wronski, Z.S.; Zaranski, Z. Nanomaterials for hydrogen storage produced by ball milling. Can. Metall. Q. 2009, 48, 11–25. [Google Scholar] [CrossRef]
- Revesz, A.; Gajdics, M.; Spassov, T. Microstructural evolution of ball-milled Mg-Ni powder during hydrogen sorption. Int. J. Hydrog. Energy 2013, 38, 8342–8349. [Google Scholar] [CrossRef]
- Schimmel, H.G.; Huot, J.; Chapon, L.C.; Tichelaar, F.D.; Mulder, F.M. Hydrogen cycling of niobium and vanadium catalyzed nanostructured magnesium. J. Am. Chem. Soc. 2005, 127, 14348–14354. [Google Scholar] [CrossRef] [PubMed]
- Hurst, K.E.; Dillon, A.C.; Keenan, D.A.; Lehman, J.H. Cleaning of carbon nanotubes near the π-plasmon resonance. Chem. Phys. Lett. 2007, 433, 301–304. [Google Scholar] [CrossRef]
- Wang, F.X.; Gao, X.P.; Lu, Z.W.; Ye, S.H.; Qu, J.Q.; Wu, F.; Yuan, H.T.; Song, D.Y. Electrochemical properties of Mg-based alloys containing carbon nanotubes. J. Alloys Compd. 2004, 370, 326–330. [Google Scholar] [CrossRef]
- Dillon, A.C.; Parilla, P.A.; Alleman, J.L.; Gennett, T.; Jones, K.M.; Heben, M.J. Systematic inclusion of defects in pure carbon single-wall nanotubes and their effect on the Raman D-band. Chem. Phys. Lett. 2005, 401, 522–528. [Google Scholar] [CrossRef]
- Pierard, N.; Fonseca, A.; Colomer, J.F.; Bossuot, C.; Benoit, J.M.; van Tendeloo, G.; Pirard, J.P.; Nagy, J.B. Ball milling effect on the structure of single-wall carbon nanotubes. Carbon 2004, 42, 1691–1697. [Google Scholar] [CrossRef]
- Darsono, N.; Yoon, D.H.; Kim, J.Y. Milling and dispersion of multi-walled carbon nanotubes in texanol. Appl. Surf. Sci. 2008, 254, 3412–3419. [Google Scholar] [CrossRef]
- Lee, J.H.; Rhee, K.Y.; Park, S.J. Effects of cryomilling on the structures and hydrogen storage characteristics of multi-walled carbon nanotubes. Int. J. Hydrog. Energy 2010, 35, 7850–7857. [Google Scholar] [CrossRef]
- He, H.Y.; Pan, B.C. Studies on structural defects in carbon nanotubes. Front. Phys. China 2009, 4, 297–306. [Google Scholar] [CrossRef]
- Robinson, J.A.; Snow, E.S.; Badescu, S.C.; Reinecke, T.L.; Perkins, F.K. Role of defects in single-walled carbon nanotube chemical sensors. Nano Lett. 2006, 6, 1747–1751. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.D.; Wang, D.L.; Hou, D.Y. Theoretical study of chemisorption of hydrogen atoms on the sidewalls of armchair single-walled carbon nanotubes with Stone-Wales defect. Comput. Theor. Chem. 2012, 999, 121–125. [Google Scholar] [CrossRef]
- Paskevicius, M.; Sheppard, D.A.; Buckley, C.E. Thermodynamic Changes in Mechanochemically Synthesized Magnesium Hydride Nanoparticles. J. Am. Chem. Soc. 2010, 132, 5077–5083. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.; Felderhoff, M.; Schuth, F. Hydrogen storage properties of nanostructured MgH2/TiH2 composite prepared by ball milling under high hydrogen pressure. Int. J. Hydrog. Energy 2011, 36, 10828–10833. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kadri, A.; Jia, Y.; Chen, Z.; Yao, X. Catalytically Enhanced Hydrogen Sorption in Mg-MgH2 by Coupling Vanadium-Based Catalyst and Carbon Nanotubes. Materials 2015, 8, 3491-3507. https://doi.org/10.3390/ma8063491
Kadri A, Jia Y, Chen Z, Yao X. Catalytically Enhanced Hydrogen Sorption in Mg-MgH2 by Coupling Vanadium-Based Catalyst and Carbon Nanotubes. Materials. 2015; 8(6):3491-3507. https://doi.org/10.3390/ma8063491
Chicago/Turabian StyleKadri, Atikah, Yi Jia, Zhigang Chen, and Xiangdong Yao. 2015. "Catalytically Enhanced Hydrogen Sorption in Mg-MgH2 by Coupling Vanadium-Based Catalyst and Carbon Nanotubes" Materials 8, no. 6: 3491-3507. https://doi.org/10.3390/ma8063491
APA StyleKadri, A., Jia, Y., Chen, Z., & Yao, X. (2015). Catalytically Enhanced Hydrogen Sorption in Mg-MgH2 by Coupling Vanadium-Based Catalyst and Carbon Nanotubes. Materials, 8(6), 3491-3507. https://doi.org/10.3390/ma8063491




_Jia.png)


