Photoluminescent ZnO Nanoparticles and Their Biological Applications
Abstract
:1. Introduction
2. Preparation and Functionalization of Luminescent ZnO NPs
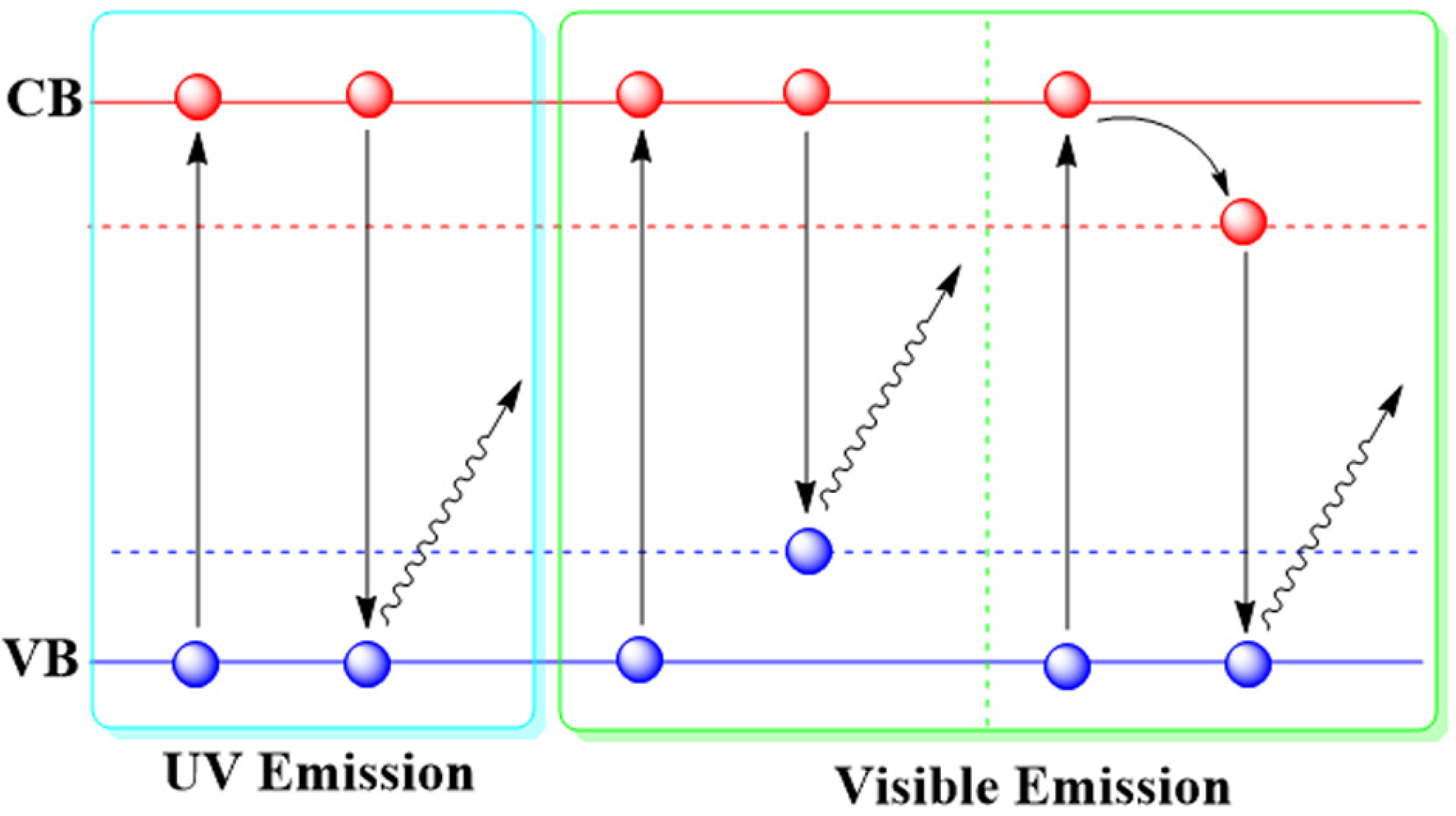
2.1. Luminescent Mechanisms of ZnO NPs
| Materials | Size (nm) | Excitation (nm) | Emission (nm) | Luminescent mechanism | Ref. |
|---|---|---|---|---|---|
| ZnO phosphors | 50–400; 500–1500 | 370 | 510 | oxygen vacancies | [23] |
| ZnO NPs | 3.4 | 248 | 510 | surface states | [24] |
| ZnO NPs | 1–3 | ~308 | 500–530 | oxygen vacancies | [25] |
| ZnO NPs | 3.5 ± 1.5 | – | Visible Emission | from conduction band to a deep trap | [21] |
| ZnO NPs | 4.3–7 | 325 | UV emission | exciton transition | [26] |
| Polyether-Grafted ZnO NPs | 1–4 | 320~365 | 450–570 | oxygen vacancy | [27] |
| ZnO@PMAA–PMMA NPs | 2.1 | UV light | ~420 | ZnO and organic species | [14] |
| ZnO NPs and NRs | NPs: 2–6 NRs: 2–4 × 6–50 | 360–420 280–360 | 440 580 | related to the ligands on surface | [28] |
| ionic liquid -ZnO NPs | 2.5–4 | UV light | 425~555 | transitions to trapped levels | [29] |
| ZnO@polymer | ZnO 2.1–2.3 ZnO@PMMA~150, ZnO@PS~450 | 328–366 | 420–464 | surface vacancies | [30] |
| ZnO NPs | – | – | Visible emission | defect-related | [31] |
| ZnO NPs | 2.2–7.8 | ~350 | 450–570 | defect-dependent & size-dependent | [22] |
| ZnO NPs | 10–20 | 340~390 | 400–600 | defect origins | [32] |
| ZnO@DDA-PVP NPs | 3–6.5 | UV light | 395–540 | quantum size effect | [33] |
| silane@ZnO NPs | 1.6–5 | UV light | 402–522 | surface defects | [34] |
| ZnO NPs | 3.5 ± 0.6 | 325 | UV emission | free exciton | [35] |
| ZnO NPs | 20 | 532 | 660 | zinc vacancy | [36] |
| ZnO NPs | < 50 | 532 | 793 | zinc vacancy | [36] |
| oleate-ZnO NPs | 3.6–5.2 | 365 | 500–560 | intrinsic defects | [37] |
| ZnO NPs | 3–7 | 320 | 348–362 | exciton emission | [38] |
2.2. Synthesis of Luminescent ZnO NPs
2.3. Functionalization of Luminescent ZnO NPs
3. Luminescence Sensors Based on ZnO NPs
3.1. Detection of Metal Ions
| Materials | Target | LOD | Strategy | Ref. |
|---|---|---|---|---|
| ZnO NPs | Cu2+ | 7.68 × 10−7 M | PL turn-off | [55] |
| Imine-linked-ZnO NPs | Co2+ | 4 × 10−10 M | PL turn-off | [56] |
| APTES-ZnO NPs | dopamine | 1.2 × 10−8 M | PL turn-off | [57] |
| APTES-ZnO NPs | picric acid | 2.86 × 10−6 M | PL turn-off | [58] |
| Metal-organic ZnO NPs | phosphate | 5.3 × 10−8 M | PL turn-on | [59] |
| silane-ZnO NPs | aldehyde | – | PL switch | [60] |
| Cd-doped ZnO NPs | bisphenol A | 1.31 × 10−8 g·mL−1 | immunosensor | [61] |
| ZnO NPs | carbohydrate antigen | 2.5 × 10−1 U·mL−1 | immunosensor | [62] |
3.2. Detection of Organic Molecules
3.3. Detection of Proteins
4. Biological Imaging by ZnO NPs
4.1. Fluorescence Biological Imaging
| Materials | Size (nm) | Models | Imaging modality | Ref. |
|---|---|---|---|---|
| ZnO@polymer NPs | 3–4 | human hepatoma cells | disk scanning confocal microscopy | [45] |
| ZnO NPs | 2–6 | NIH/3T3 cells | disk scanning confocal microscopy | [69] |
| ZnO@PMAA-co-PDMAEMA NPs | 4 | COS-7 cells | laser scanning confocal microscopy | [73] |
| ZnO@silica NPs | ZnO 4–7 ZnO@SiO2 ~50 | NIH/3T3 cells | disk scanning confocal microscopy | [49] |
| ZnO@polymer NPs | 3–4 | BALB/c mice | fluorescence microscopy | [70] |
| Gd-doped ZnO QDs | 3–6 | HeLa cells | confocal laser scanning microscopy | [74] |
| ZnO-Au@PEG NPs | 45–98 | B16F10 cells | confocal laser scanning microscopy | [68] |
| CdSe(S)/ZnO QDs | 2–4 | S. oneidensis | confocal microscopy | [75] |
| ZnO@silica NPs | 2–5 | Hela cells | Laser scanning confocal microscopy | [76] |
| ZnO nanocrystals | < 100 | KB cells | nonresonant nonlinear optical microscopy, SFG SHG FWM | [77] |
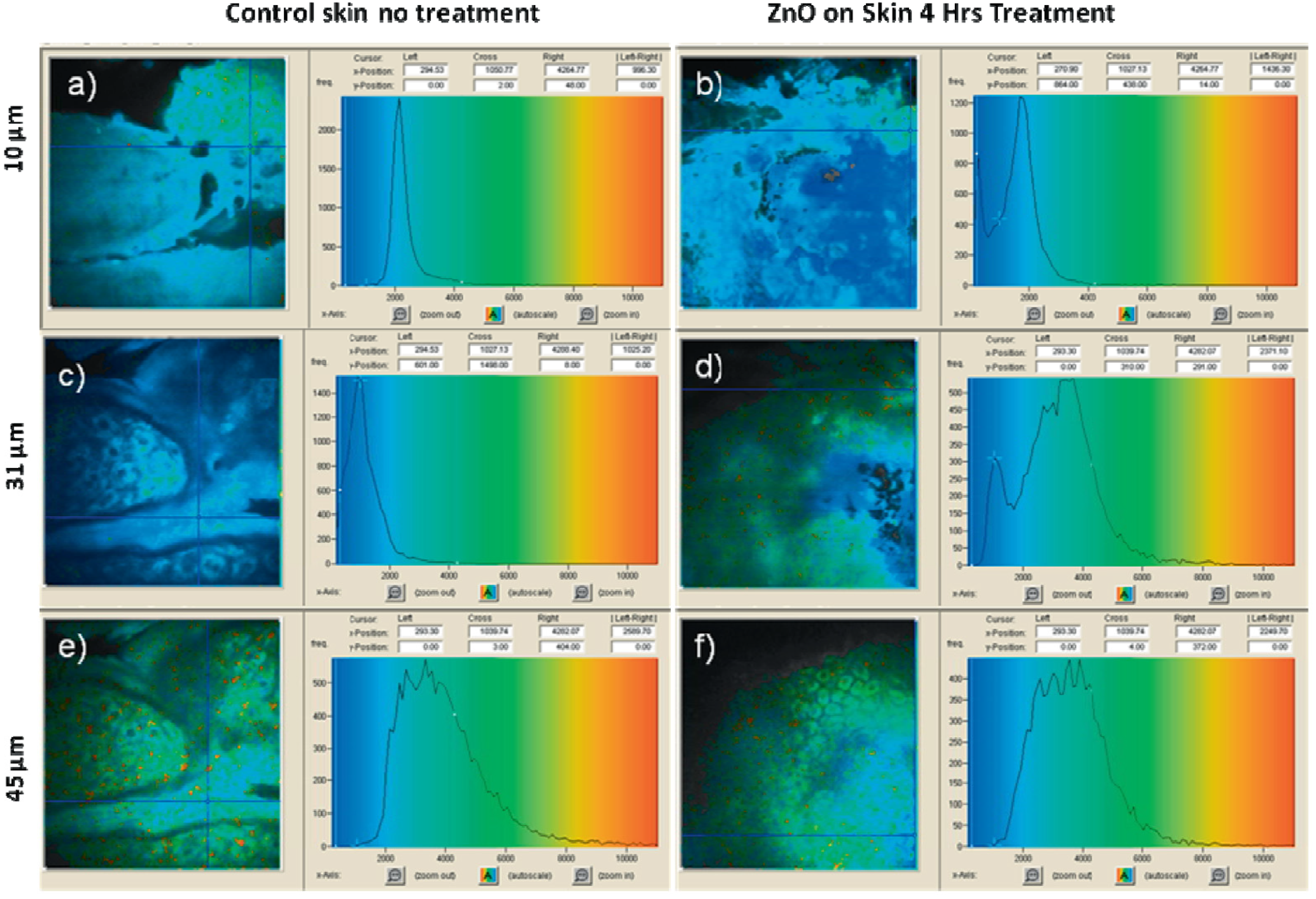
| ZnO NPs | 15–30 | skin tissue/cellular architecture | multiphoton microscopy, SHG | [78] |
| ZnO NPs | 21 | skin | nonlinear optical microscopy, SHG | [79] |
| ZnO NPs | 2–200 | plants tissues cell implosion | nonlinear optical microscopy, SHG | [80] |
| ZnO NPs | 10–300 | blood cells of zebrafish; roots and shoots of Arabidopsis plants | nonlinear imaging, SHG | [81] |
| ZnO NPs | 26–30 | human skin and rat liver | fluorescence lifetime imaging | [82] |
4.2. Multiphoton Biological Imaging
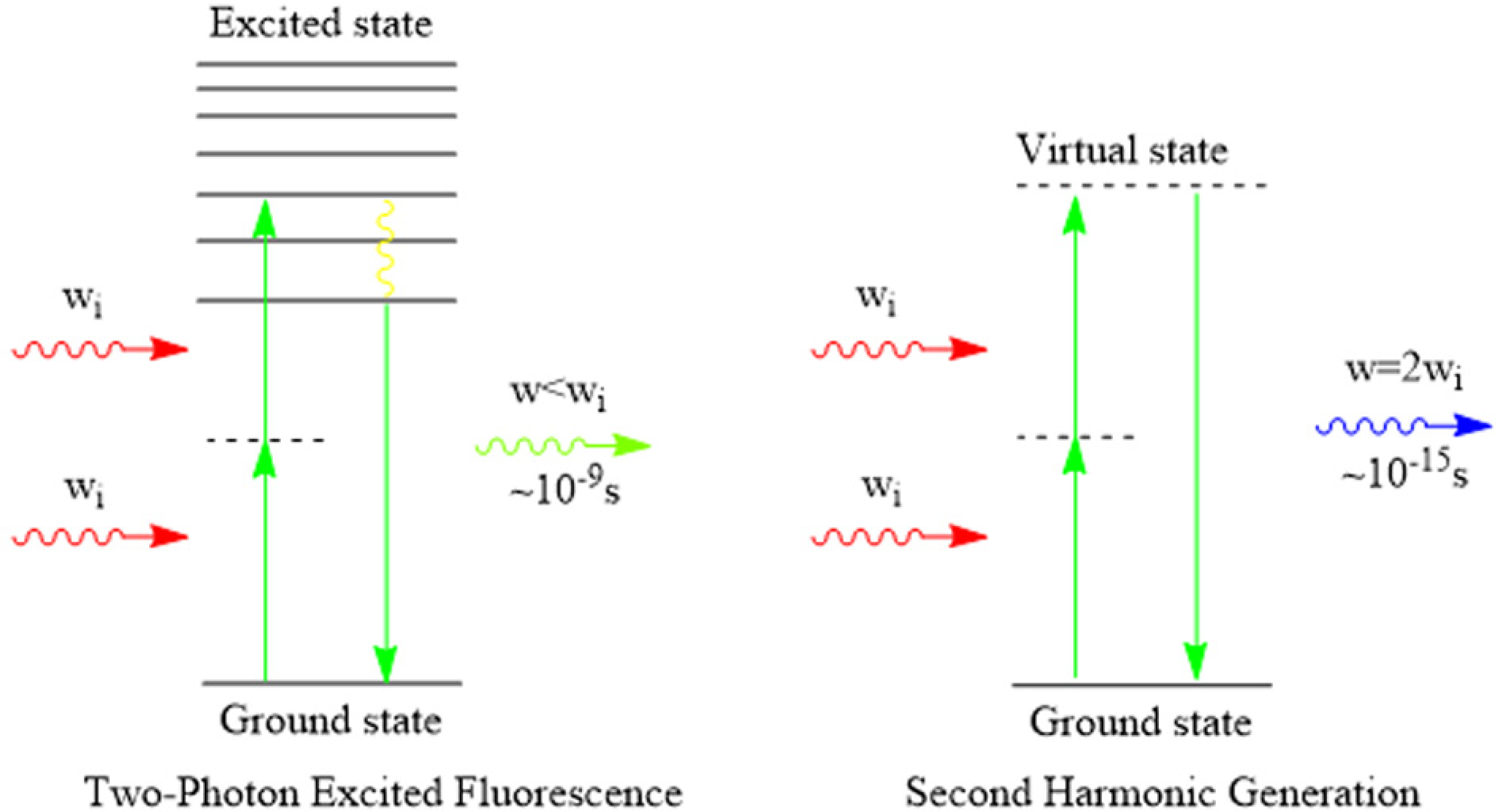
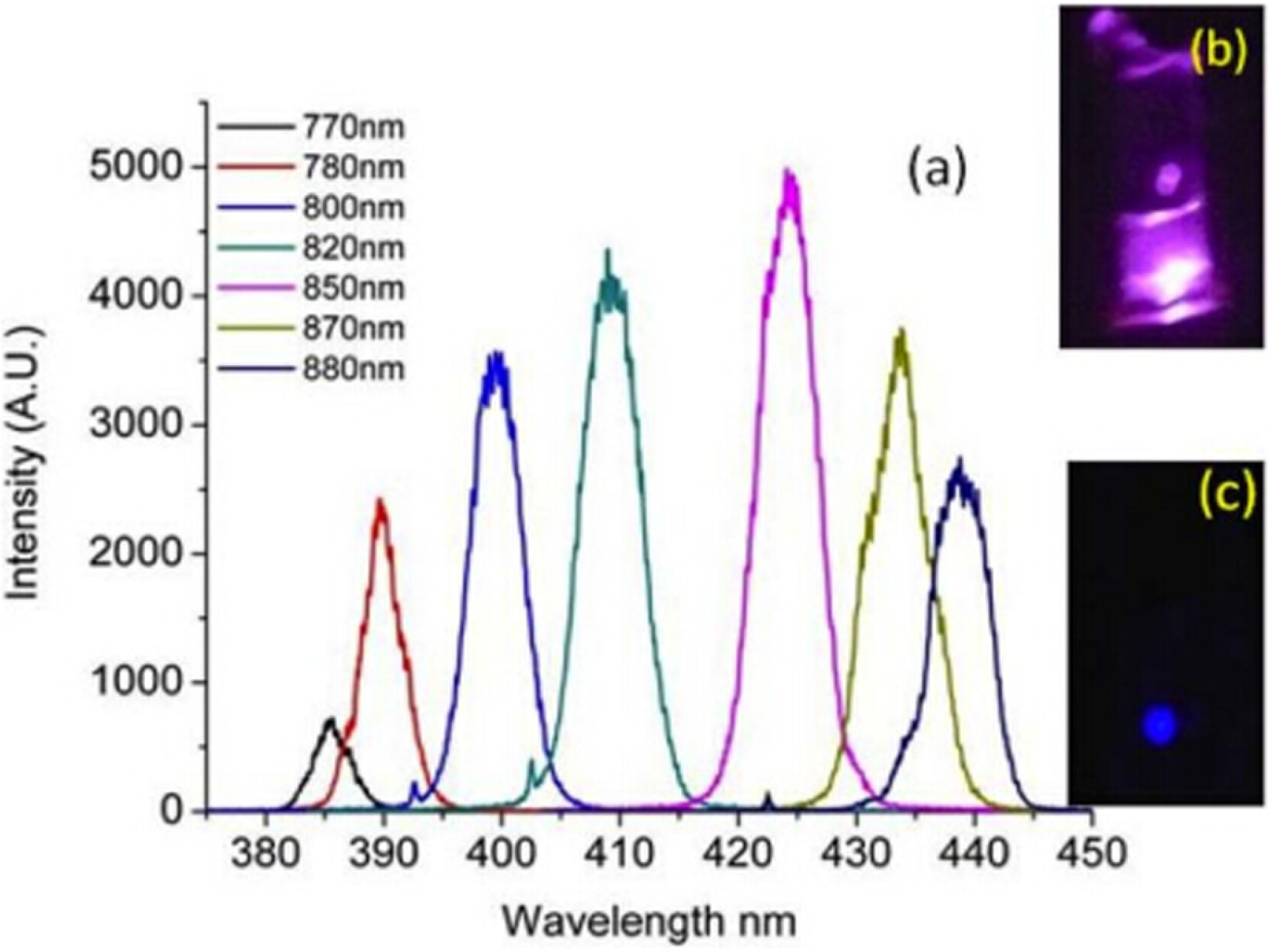
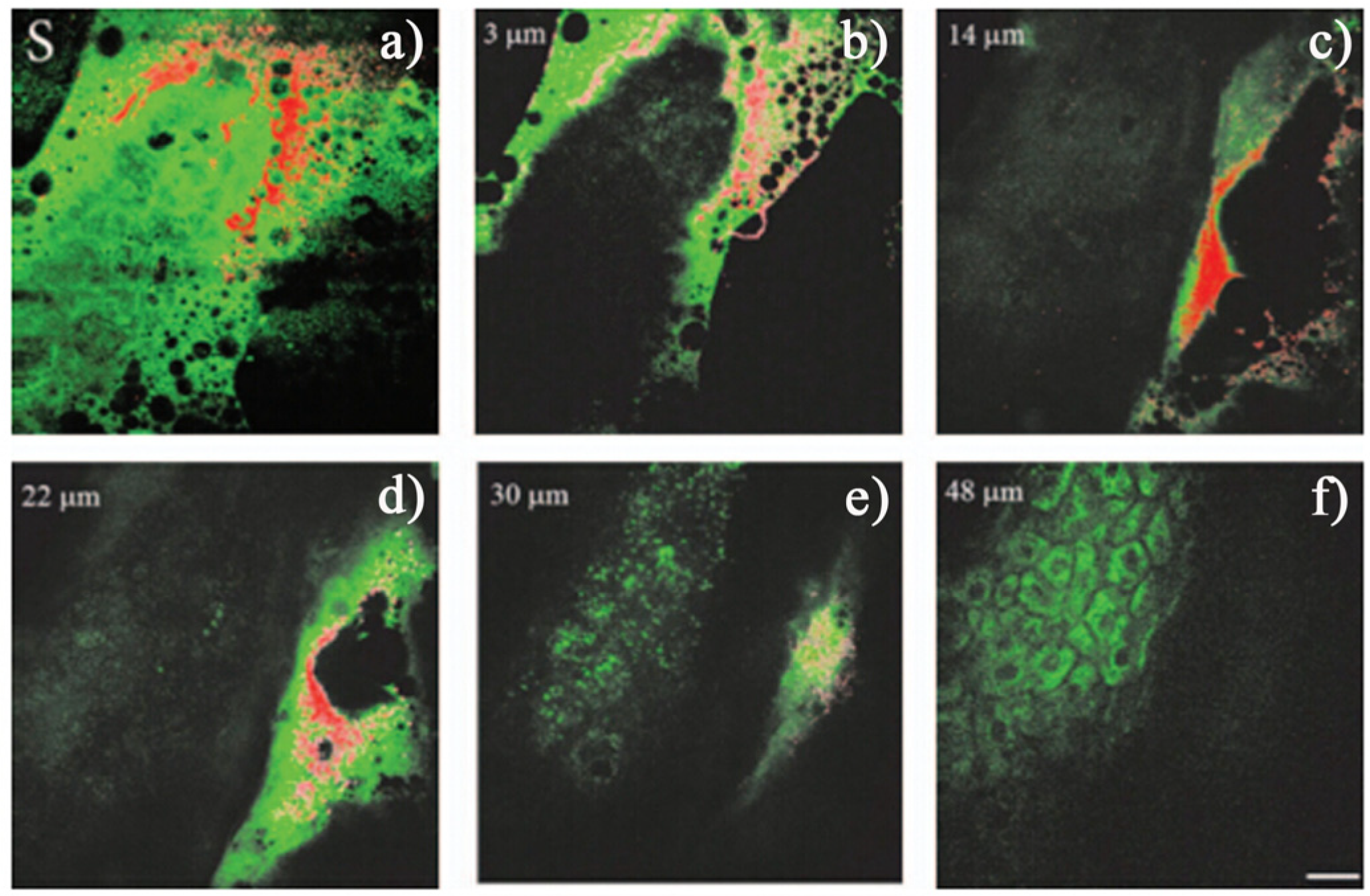
4.3. Fluorescence Lifetime Imaging
| Fluorophore | Excitation (nm) | Emission (nm) | Lifetime (ns) | Ref. |
|---|---|---|---|---|
| phenylalanine | 240–270 | 280 | 7.5 | [85,92] |
| tyrosine | 250–290 | 300 | 2.5 | [85,93] |
| tryptophan | 250–310 | 350 | 3.03 | [85,94] |
| flavin mononucleotide | 420–500 | 520–570 | 4.27–4.67 | [85,95] |
| riboflavin | 420–500 | 520–570 | 4.12 | [85,96] |
| protoporphyrin IX | 400–450 | 635, 710 | up to 15 | [85,97] |
| lipofuscin | 340–395 | 540, 430–460 | 1.34 | [85,98] |
| NAD (P) H free | 300–380 | 450–500 | 0.3 | [85,97] |
| NAD (P) H free | 730–800 (2P) | 450–460 | 0.3–0.7 | [86,99] |
| NAD (P) H bound | 300–380 | 450–500 | 2.0–2.3 | [85,97] |
| NAD (P) H bound | 730–800 (2P) | 450–460 | 2.5–3.0 | [86,99] |
| FAD | 420–500 | 520–570 | 2.91 | [85,97] |
| FAD | ~800 (2P) | 525–550 | 2.3–2.8 | [86,100] |
| FAD bound | 420–500 | weak in 520–570 | <0.01 | [85,98] |
| FAD bound | ~800 (2P) | 525–550 | 0.04–0.4 | [86,100] |
| Melanin | 300–800 | 440, 520, 575 | 0.1/1.9/8 | [85,97] |
| Melanin | 730–830 (2P) | ~550 | 0.1–0.2, 0.7–1.4 | [86,101] |
| Elastin | 290–340 | 420–460 | 0.2–2.5 | [82,97] |
| Elastin | 760–830 (2P) | 475–575 | 0.26, 1.96 | [86,102] |
| Keratin | 750–800 (2P) | 382, 450–500 | 1.4 | [86,103] |
| Collagen | 280–350 | 370–440 | ≤5.3 | [85,97] |
| Collagen, SHG | 730–880 (2P) | 1/2 of excitation wavelength | No lifetime | [86,87] |
| Materials | Size (nm) | Excitation (nm) | Emission (nm) | Lifetime (ns) | Ref. |
|---|---|---|---|---|---|
| ZnO NPs | ~4 | 248 | 510 | ~1000 | [24] |
| ZnO NPs | 1 | 308 | visible (500–530) | 920 | [25] |
| ZnO NPs | 3 | 308 | visible (500–530) | 1340 | [25] |
| ZnO NPs | ~4 | 380 | 460 | 2, 9 | [28] |
| ZnO NPs | –4 | 320 | 600 | 9, 60, 1850 | [28] |
| ZnO NPs | 26–30 | 405 | – | 21, 478, 2500 | [82] |
| ZnO NPs | 3–4 | 320–340 | 530 | 3066, 1000 | [104] |
| ZnO NPs | 3~4 | 320–340 | 510 | 2750, 885 | [104] |
| ZnO NPs | 3–4 | 320–340 | 490 | 729, 109 | [104] |
| ZnO QDs | 3–7 | 320 | 348–362 | (9.8~29.6) × 10−3 | [38] |
| ZnO NPs | – | 350 | ~370 | ~0.14, 3.12 | [105] |
| ZnO NPs | – | 730 (2P) | ~430 | ~0.13, 1.56 | [105] |
5. Antibacterial Activity of ZnO NPs
| Materials | Size (nm) | Models | Treatment | Antibacterial result | Antibacterial mechanism | Ref. |
|---|---|---|---|---|---|---|
| ZnO | ~480 | B. subtilis CB310 | 10 ppm | 90% growth reduction of B. subtilis | ROS | [114] |
| ZnO | ~480 | E. coli DH5α | 1000 ppm | 48% growth reduction of E. coli | ROS | [114] |
| Acetate-ZnO QDs | 3~5 | E. coli | 2.5 mM in light | MIC | ROS | [115] |
| Acetate-ZnO QDs | 3~5 | E. coli | 3 mM in dark | MIC | ROS | [115] |
| Nitrate-ZnO QDs | 4–7 | E. coli | 6 mM in light, 30 mM in dark | MIC | ROS | [115] |
| Nitrate-ZnO QDs | 4–7 | E. coli | 30 mM in dark | no significant bacterial growth inhibition | ROS | [115] |
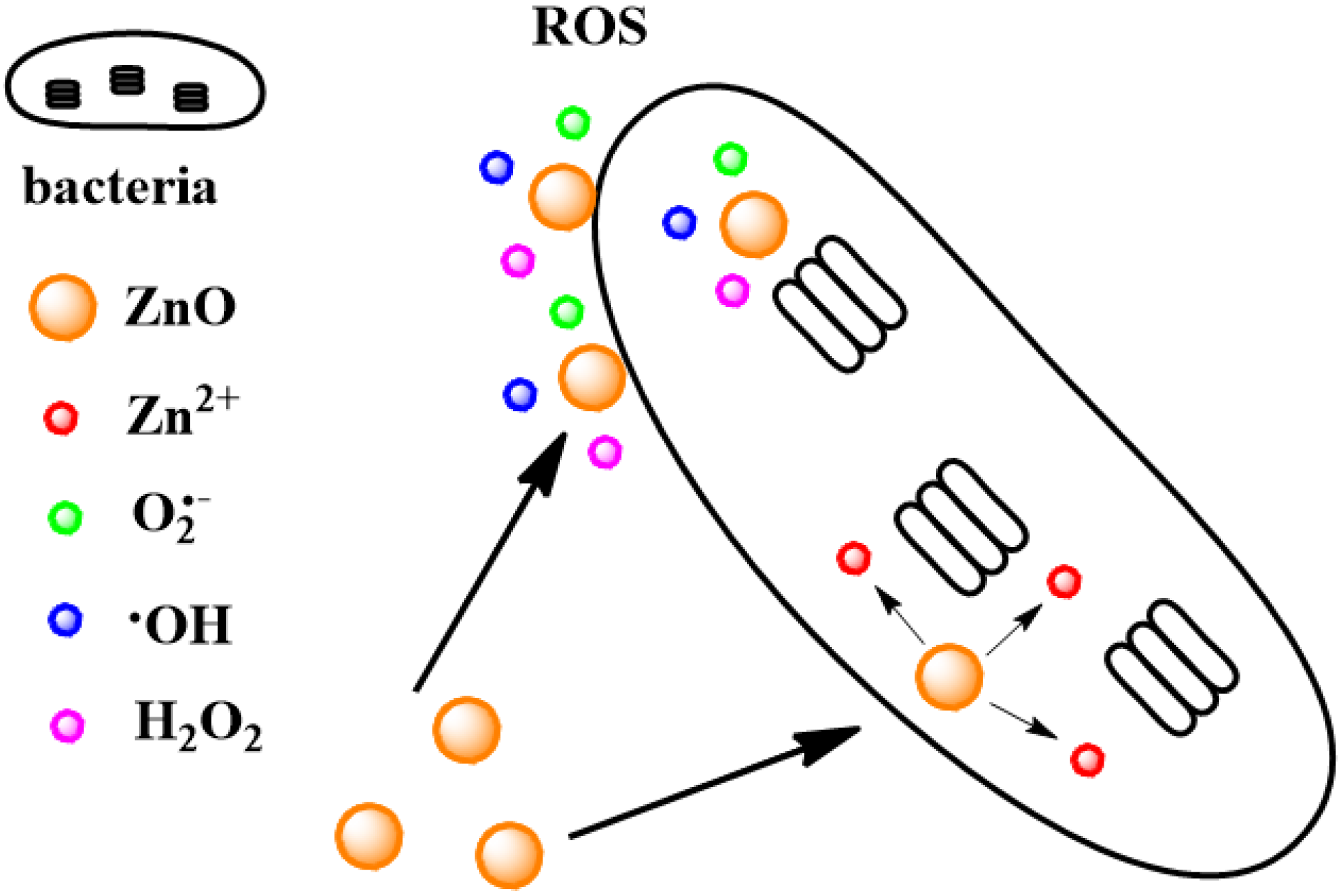
| ZnO NPs | 6.8 ± 2 | E. coli and S. aureus | 0.1 mg·mL−1 for 3 h | inactivation of E. coli by 99.8%, that of S. aureus by 98% | ROS | [113] |
| ZnO NPs | 260 ± 40 | E. coli and S. aureus | 0.1 mg·mL−1 for 3 h | inactivation of E. coli by 99.5%, that of S. aureus by 89% | ROS | [113] |
| ZnO NPs | 800 ± 300 | E. coli and S. aureus | 0.1 mg·mL−1 for 3 h | inactivation of E. coli by 87%, that of S. aureus by 68% | ROS | [113] |
| ZnO NPs | 800 ± 300 | E. coli and S. aureus | 5 mg·mL−1 for E. coli and 10 mg·mL−1 for S. aureus. | MIC | ROS | [113] |
| ZnO NPs | ~20 | E. coli 11634 | 543 nm, 1000 lux light for 24 h | <20 bacteria count (CFU) | hydrogen peroxide (H2O2) ROS | [116] |
| Ag-ZnO composite | ~64 | S. aureus and GFP E. coli | 550 μg·mL−1 for E. coli and 60 μg·mL−1 for S. aureus | MIC | ROS and the release of Ag+, Zn2+ | [117] |
| ZnO QDs | ~4 | E. coli MG1655 and C. metallidurans CH34 | 1.6 × 10−5 M for E. coli MG1655 and 2.8 × 10−4 M for C. metallidurans CH34 | IC50 | mainly result from the liberation of Zn2+ cations from the ZnO QDs | [110] |
| crystal violet-ZnO silicone polymers | ZnO NPs 3.3 ± 1.1 | S. aureus | 1 h incubation in dark or 1 h in standard hospital light | Significantly reduced under dark or 3.36 log kill in light | PDT | [118] |
| ROS | ||||||
| crystal violet-ZnO silicone polymers | ZnO NPs 3.3 ± 1.1 | E. coli | 6 h in the dark or in standard hospital light | 1.41 log kill in dark or below the detection limit | PDT | [118] |
| ROS | ||||||
| CdO-ZnO composite | ~27 | E. coli | 10 μg·mL−1 | MIC | ROS and Zn2+, Cd2+ release | [119] |
| CdO-ZnO composite | ~27 | S. aureus | 10 μg·mL−1 | MIC | ROS and Zn2+, Cd2+ release | [119] |
| CdO-ZnO composite | ~27 | P. aeruginosa | 15 μg·mL−1 | MIC | ROS and Zn2+, Cd2+ release | [119] |
| CdO-ZnO composite | ~27 | P. vulgaris | 20 μg·mL−1 | MIC | ROS and Zn2+, Cd2+ release | [119] |
| CdO-ZnO composite | ~27 | K. pneumonia | 20 μg·mL−1 | MIC | ROS and Zn2+, Cd2+ release | [119] |
| CdO-ZnO composite | ~27 | Bacillus spp. | 15 μg·mL−1 | MIC | ROS and Zn2+, Cd2+ release | [119] |
6. Drug Delivery by ZnO NPs
| Materials | The role of ZnO | Drugs | Models | Control method | Ref. |
|---|---|---|---|---|---|
| ZnO | drugs | itself | cancerous T cells, activated human T cells | – | [130] |
| chitosan-ZnO NPs | carriers | DOX | – | pH | [133] |
| ZnO@PMAA-co-PDMAEMA NPs | carriers | DNA | COS-7 cells | – | [73] |
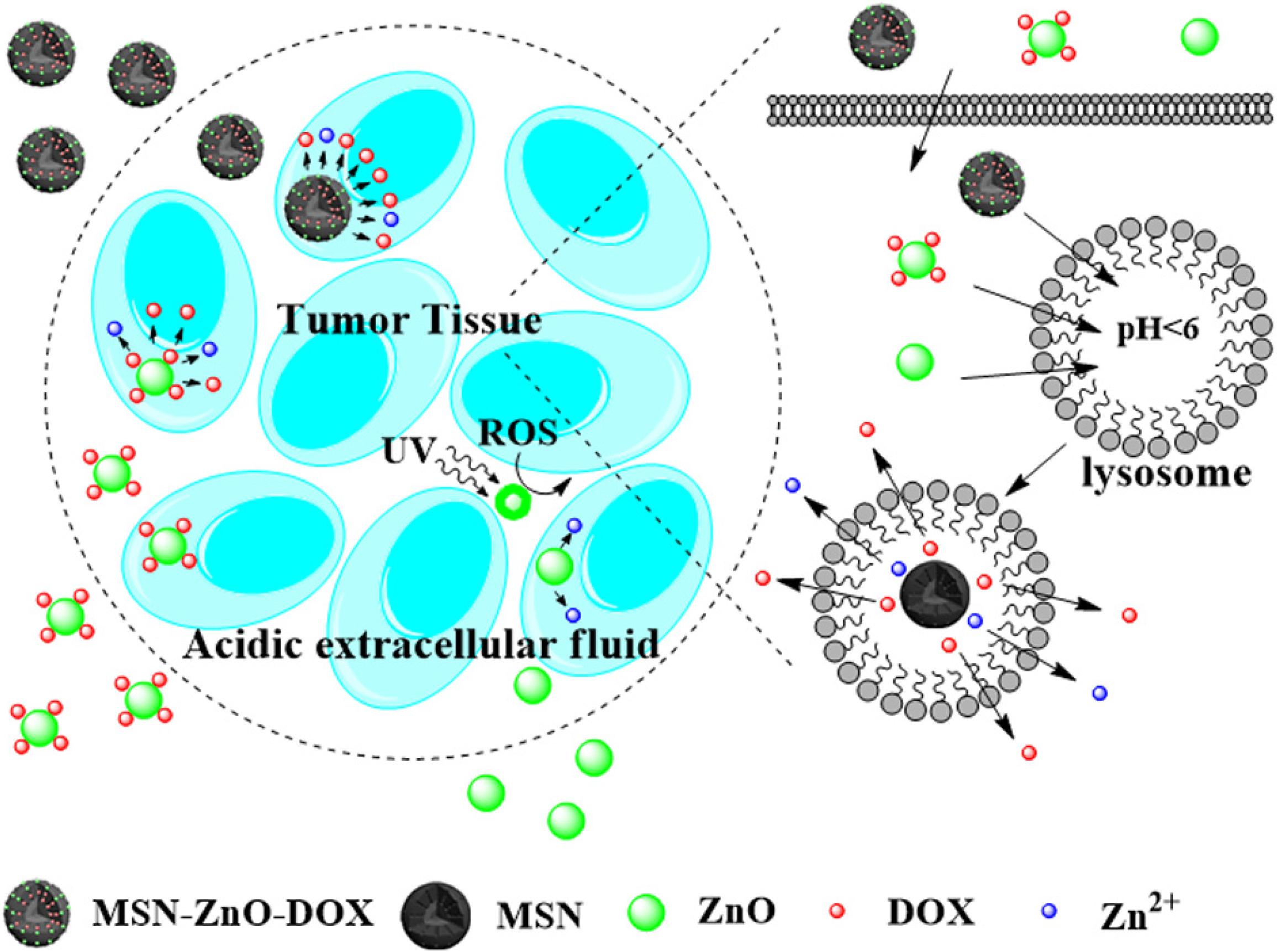
| mesoporous ZnO | carriers | DOX | – | pH | [134] |
| ZnO NRs | carriers | PPDME | T47D cells | PDT | [131] |
| ZnO-Mesoporous Silica NPs | lids | DOX | HeLa cells | pH | [135] |
| ZnO NPs | carriers | DOX | HeLa cells | pH | [136] |
| ZnO tetrapod | drugs | itself | CHO-K1 cells, HeLa cells, Vero cells and VK2/E6 cells | – | [137] |
| ZnO/PEG NPs | carriers | DOX | Gram-positive microorganisms | Photo-dynamic therapy | [138] |
| ZnO@polymer NPs | carriers | DOX | U251 cells | pH | [128] |
| ZnO-Au NPs | carriers | camptothecin | Hela cells | pH | [139] |
| ZnO@PNIPAM NPs | carriers | DOX | – | Thermal and pH | [140] |
| ZnO/carboxymethyl cellulose | carriers | curcumin | L929 and MA104 cell | pH | [141] |
| mesoporous ZnO NPs | carriers | DNA, DOX | normal lymphocyte and K562 cells | – | [142] |
| ZnO NRs | carriers | ALA | FM55P cells and AG01518 cells | PDT | [132] |
| ZnO QDs | drugs | itself | HepG2 cells | – | [143] |
| Curcumin/O-CMCS/n-ZnO nanocomposites | carriers | curcumin | MA 104 cells | pH | [144] |
| UCNP@mSiO2-ZnO | gatekeepers | DOX | HeLa cells and Balb/c mouse | pH | [125] |
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fountaine, T.J.; Wincovitch, S.M.; Geho, D.H.; Garfield, S.H.; Pittaluga, S. Multispectral imaging of clinically relevant cellular targets in tonsil and lymphoid tissue using semiconductor quantum dots. Mod. Pathol. 2006, 19, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.Q.; Zhao, J.B.; Zhang, R.; Liu, Y.J.; Liu, D.M.; Goldys, E.M.; Yang, X.S.; Xi, P.; Sunna, A.; Lu, J.; et al. Tunable lifetime multiplexing using luminescent nanocrystals. Nat. Photon. 2014, 8, 32–36. [Google Scholar] [CrossRef]
- Drbohlavova, J.; Adam, V.; Kizek, R.; Hubalek, J. Quantum dots-characterization, preparation and usage in biological systems. Int. J. Mol. Sci. 2009, 10, 656–673. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.A.; Xie, X.Y.; Patel, M.; Yang, Y.H.; Li, Z.J.; Cao, F.; Gheysens, O.; Zhang, Y.; Gambhir, S.; Rao, J.; Wu, J. Quantum dot imaging for embryonic stem cells. BMC Biotechnol. 2007, 7, 67. [Google Scholar] [CrossRef] [PubMed]
- Medintz, I.L.; Uyeda, H.T.; Goldman, E.R.; Mattoussi, H. Quantum dot bioconjugates for imaging, labelling and sensing. Nat. Mater. 2005, 4, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Yong, K.T.; Wang, Y.C.; Roy, I.; Rui, H.; Swihart, M.T.; Law, W.C.; Kwak, S.K.; Ye, L.; Liu, J.W.; Mahajan, S.D.; Reynolds, J.L. Preparation of quantum dot/drug nanoparticle formulations for traceable targeted delivery and therapy. Theranostics 2012, 2, 681–694. [Google Scholar] [CrossRef] [PubMed]
- Montalti, M.; Prodi, L.; Rampazzo, E.; Zaccheroni, N. Dye-doped silica nanoparticles as luminescent organized systems for nanomedicine. Chem. Soc. Rev. 2014, 43, 4243–4268. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.Y.; Shan, C.X.; Qu, S.N.; Shen, D.Z. Highly sensitive ultraviolet photodetectors fabricated from ZnO quantum dots/carbon nanodots hybrid films. Sci. Rep. 2014, 4, 7469. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Xu, L.J.; Li, H.X.; Kong, J.L. Wavelength-tunable luminescent gold nanoparticles generated by cooperation ligand exchange and their potential application in cellular imaging. RSC Adv. 2013, 3, 59–63. [Google Scholar] [CrossRef]
- Chen, L.Y.; Wang, C.W.; Yuan, Z.Q.; Chang, H.T. Fluorescent gold nanoclusters: Recent advances in sensing and imaging. Anal. Chem. 2015, 87, 216–229. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, Q.; Feng, W.; Sun, Y.; Li, F.Y. Upconversion luminescent materials: Advances and applications. Chem. Rev. 2015, 115, 395–465. [Google Scholar] [CrossRef] [PubMed]
- Spanhel, L.; Anderson, M.A. Semiconductor clusters in the sol-gel process: Quantized aggregation, gelation, and crystal growth in concentrated zinc oxide colloids. J. Am. Chem. Soc. 1991, 113, 2826–2833. [Google Scholar] [CrossRef]
- Fu, Y.S.; Du, X.W.; Kulinich, S.A.; Qiu, J.S.; Qin, W.J.; Li, R.; Sun, J.; Liu, J. Stable aqueous dispersion of ZnO quantum dots with strong blue emission via simple solution route. J. Am. Chem. Soc. 2007, 129, 16029–16033. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.M.; Wang, Z.D.; Xia, Y.Y. Polymerization initiated by inherent free radicals on nanoparticle surfaces: A simple method of obtaining ultrastable (ZnO) polymer core–shell nanoparticles with strong blue fluorescence. Adv. Mater. 2006, 18, 748–751. [Google Scholar] [CrossRef]
- Zhang, C.B.; Li, K.Y.; Song, S.Y.; Xue, D.F. Reversible phase transfer of luminescent ZnO quantum dots between polar and nonpolar media. Chem. Eur. J. 2013, 19, 6329–6333. [Google Scholar] [CrossRef] [PubMed]
- Salgueiro, M.J.; Zubillaga, M.; Lysionek, A.; Caro, R.; Weill, R.; Boccio, J. Fortification strategies to combat zinc and iron deficiency. Nutr. Rev. 2002, 60, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Cushen, M.; Kerry, J.; Morris, M.; Cruz-Romero, M.; Cummins, E. Nanotechnologies in the food industry-Recent developments, risks and regulation. Trends Food Sci. Technol. 2012, 24, 30–46. [Google Scholar] [CrossRef]
- Cross, S.E.; Innes, B.; Roberts, M.S.; Tsuzuki, T.; Robertson, T.A.; McCormick, P. Human skin penetration of sunscreen nanoparticles: In-vitro assessment of a novel micronized zinc oxide formulation. Skin Pharmacol. Physiol. 2007, 20, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Özgür, Ü.; Alivov, Y.I.; Liu, C.; Teke, A.; Reshchikov, M.A.; Doğan, S.; Avrutin, V.; Cho, S.J.; Morkoc, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 041301. [Google Scholar]
- Xiong, H.M. Photoluminescent ZnO nanoparticles modified by polymers. J. Mater. Chem. 2010, 20, 4251–4262. [Google Scholar] [CrossRef]
- van Dijken, A.; Meulenkamp, E.A.; Vanmaekelbergh, D.; Meijerink, A. Identification of the transition responsible for the visible emission in ZnO using quantum size effects. J. Lumin. 2000, 90, 123–128. [Google Scholar] [CrossRef]
- Zhang, L.Y.; Yin, L.W.; Wang, C.X.; Lun, N.; Qi, Y.X.; Xiang, D. Origin of visible photoluminescence of ZnO quantum dots: Defect-dependent and size-dependent. J. Phys. Chem. C 2010, 114, 9651–9658. [Google Scholar] [CrossRef]
- Vanheusden, K.; Seager, C.H.; Warren, W.L.; Tallant, D.R.; Voigt, J.A. Correlation between photoluminescence and oxygen vacancies in ZnO phosphors. Appl. Phys. Lett. 1996, 68, 403–405. [Google Scholar] [CrossRef]
- Monticone, S.; Tufeu, R.; Kanaev, A.V. Complex nature of the UV and visible fluorescence of colloidal ZnO nanoparticles. J. Phys. Chem. B 1998, 102, 2854–2862. [Google Scholar] [CrossRef]
- van Dijken, A.; Meulenkamp, E.A.; Vanmaekelbergh, D.; Meijerink, A. The kinetics of the radiative and nonradiative processes in nanocrystalline ZnO particles upon photoexcitation. J. Phys. Chem. B 2000, 104, 1715–1723. [Google Scholar] [CrossRef]
- Zhou, H.; Alves, H.; Hofmann, D.M.; Kriegseis, W.; Meyer, B.K.; Kaczmarczyk, G.; Hoffmann, A. Behind the weak excitonic emission of ZnO quantum dots: ZnO/Zn(OH)2 core-shell structure. Appl. Phys. Lett. 2002, 80, 210–212. [Google Scholar] [CrossRef]
- Xiong, H.M.; Liu, D.P.; Xia, Y.Y.; Chen, J.S. Polyether-grafted ZnO nanoparticles with tunable and stable photoluminescence at room temperature. Chem. Mater. 2005, 17, 3062–3064. [Google Scholar] [CrossRef]
- Kahn, M.L.; Cardinal, T.; Bousquet, B.; Monge, M.; Jubera, V.; Chaudret, B. Optical properties of zinc oxide nanoparticles and nanorods synthesized using an organometallic method. ChemPhysChem 2006, 7, 2392–2397. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.P.; Li, G.D.; Su, Y.; Chen, J.S. Highly luminescent ZnO nanocrystals stabilized by ionic-liquid components. Angew. Chem. Int. Ed. 2006, 45, 7370–7373. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.M.; Xie, D.P.; Guan, X.Y.; Tan, Y.J.; Xia, Y.Y. Water-stable blue-emitting ZnO@polymer core-shell microspheres. J. Mater. Chem. 2007, 17, 2490–2496. [Google Scholar] [CrossRef]
- Reshchikov, M.A.; Morkoc, H.; Nemeth, B.; Nause, J.; Xie, J.; Hertog, B.; Osinsky, A. Luminescence properties of defects in ZnO. Physica B 2007, 401–402, 358–361. [Google Scholar] [CrossRef]
- Zeng, H.B.; Duan, G.T.; Li, Y.; Yang, S.K.; Xu, X.X.; Cai, W.P. Blue luminescence of ZnO nanoparticles based on non-equilibrium processes: Defect origins and emission controls. Adv. Funct. Mater. 2010, 20, 561–572. [Google Scholar] [CrossRef]
- Wang, H.P.; Jiang, H.; Wang, X.M. Construction of strong alkaline microcavities for facile synthesis of fluorescence-tunable ZnO quantum dots. Chem. Commun. 2010, 46, 6900–6902. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.Q.; Li, W.N.; Sun, L.W.; Liu, Y.; Xiao, H.M.; Fu, S.Y. Synthesis of silane surface modified ZnO quantum dots with ultrastable, strong and tunable luminescence. Chem. Commun. 2011, 47, 11921–11923. [Google Scholar] [CrossRef] [PubMed]
- Kiliani, G.; Schneider, R.; Litvinov, D.; Gerthsen, D.; Fonin, M.; Rüdiger, U.; Leitenstorfer, A.; Bratschitsch, R. Ultraviolet photoluminescence of ZnO quantum dots sputtered at room-temperature. Opt. Express 2011, 19, 1641–1647. [Google Scholar] [CrossRef] [PubMed]
- Morfa, A.J.; Gibson, B.C.; Karg, M.; Karle, T.J.; Greentree, A.D.; Mulvaney, P.; Tomljenovic-Hanic, S. Single-photon emission and quantum characterization of zinc oxide defects. Nano Lett. 2012, 12, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Aleksandra, S.; Mathieu, F. Size-controlled synthesis of ZnO quantum dots in microreactors. Nanotechnology 2014, 25, 145606. [Google Scholar]
- Jacobsson, T.J.; Viarbitskaya, S.; Mukhtar, E.; Edvinsson, T. A size dependent discontinuous decay rate for the exciton emission in ZnO quantum dots. Phys. Chem. Chem. Phys. 2014, 16, 13849–13857. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.J.; Qiu, J.H.; Liang, C.H. Controllable fabrication and photocatalytic activity of ZnO nanobelt arrays. J. Phys. Chem. C 2008, 112, 715–721. [Google Scholar] [CrossRef]
- Djurišić, A.B.; Leung, Y.H. Optical properties of ZnO nanostructures. Small 2006, 2, 944–961. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.W.; Bi, Y.; Luo, X.; Zhang, L. Photoluminescence of monolithic zinc oxide aerogel synthesised by dispersed inorganic sol–gel method. Mater. Technol. 2015, 30, 65–69. [Google Scholar] [CrossRef]
- Li, F.; Bi, W.T.; Liu, L.Y.; Li, Z.; Huang, X.T. Preparation and characterization of ZnO nanospindles and ZnO@ZnS core–shell microspindles. Colloids Surf. A Physicochem. Eng. Aspects 2009, 334, 160–164. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Senthilkumar, O.; Yamauchi, K.; Sato, M.; Morito, S.; Ohba, T.; Nakamura, M.; Fujita, Y. Preparation of ZnO nanoparticles for bio-imaging applications. Phys. Status Solidi B 2009, 246, 885–888. [Google Scholar] [CrossRef]
- Yue, Q.L.; Cheng, J.M.; Li, G.; Zhang, K.; Zhai, Y.L.; Wang, L.; Liu, J.F. Fluorescence property of ZnO nanoparticles and the interaction with bromothymol blue. J. Fluoresc. 2011, 21, 1131–1135. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.M.; Xu, Y.; Ren, Q.G.; Xia, Y.Y. Stable aqueous ZnO@polymer core−shell nanoparticles with tunable photoluminescence and their application in cell imaging. J. Am. Chem. Soc. 2008, 130, 7522–7523. [Google Scholar] [CrossRef] [PubMed]
- Moussodia, R.O.; Balan, L.; Merlin, C.; Mustin, C.; Schneider, R. Biocompatible and stable ZnO quantum dots generated by functionalization with siloxane-core PAMAM dendrons. J. Mater. Chem. 2010, 20, 1147–1155. [Google Scholar] [CrossRef]
- Moussodia, R.O.; Balan, L.; Schneider, R. Synthesis and characterization of water-soluble ZnO quantum dots prepared through PEG-siloxane coating. New J. Chem. 2008, 32, 1388–1393. [Google Scholar] [CrossRef]
- Saliba, S.; Valverde Serrano, C.; Keilitz, J.; Kahn, M.L.; Mingotaud, C.; Haag, R.; Marty, J.D. Hyperbranched polymers for the formation and stabilization of ZnO nanoparticles. Chem. Mater. 2010, 22, 6301–6309. [Google Scholar] [CrossRef]
- Tang, X.S.; Choo, E.S.G.; Li, L.; Ding, J.; Xue, J.M. Synthesis of ZnO nanoparticles with tunable emission colors and their cell labeling applications. Chem. Mater. 2010, 22, 3383–3388. [Google Scholar] [CrossRef]
- Xiong, H.M.; Shchukin, D.G.; Möhwald, H.; Xu, Y.; Xia, Y.Y. Sonochemical synthesis of highly luminescent zinc oxide nanoparticles doped with magnesium (II). Angew. Chem. Int. Ed. 2009, 48, 2727–2731. [Google Scholar]
- Hancock, J.M.; Rankin, W.M.; Hammad, T.M.; Salem, J.S.; Chesnel, K.; Harrison, R.G. Optical and magnetic properties of ZnO nanoparticles doped with Co, Ni and Mn and synthesized at low temperature. J. Nanosci. Nanotechnol. 2015, 15, 3809–3815. [Google Scholar] [CrossRef]
- Radovanovic, P.V.; Norberg, N.S.; McNally, K.E.; Gamelin, D.R. Colloidal transition-metal-doped ZnO quantum dots. J. Am. Chem. Soc. 2002, 124, 15192–15193. [Google Scholar] [CrossRef] [PubMed]
- Spanhel, L. Colloidal ZnO nanostructures and functional coatings: A survey. J. Sol-Gel Sci. Technol. 2006, 39, 7–24. [Google Scholar] [CrossRef]
- Wang, X.; Kong, X.G.; Shan, G.Y.; Yu, Y.; Sun, Y.J.; Feng, L.Y.; Chao, K.F.; Lu, S.Z.; Li, Y.J. Luminescence spectroscopy and visible upconversion properties of Er3+ in ZnO nanocrystals. J. Phys. Chem. B 2004, 108, 18408–18413. [Google Scholar] [CrossRef]
- Ng, S.M.; Wong, D.S.N.; Phung, J.H.C.; Chua, H.S. Integrated miniature fluorescent probe to leverage the sensing potential of ZnO quantum dots for the detection of copper (II) ions. Talanta 2013, 116, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.; Singh, A.; Kaur, N.; Singh, N. ZnO-based imine-linked coupled biocompatible chemosensor for nanomolar detection of Co2+. ACS Sustain. Chem. Eng. 2013, 1, 1600–1608. [Google Scholar] [CrossRef]
- Zhao, D.; Song, H.J.; Hao, L.Y.; Liu, X.; Zhang, L.C.; Lv, Y. Luminescent ZnO quantum dots for sensitive and selective detection of dopamine. Talanta 2013, 107, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Chaudhary, G.R.; Singh, S.; Mehta, S.K. Synthesis of highly luminescent water stable ZnO quantum dots as photoluminescent sensor for picric acid. J. Lumin. 2014, 154, 148–154. [Google Scholar] [CrossRef]
- Zhao, D.; Wan, X.Y.; Song, H.J.; Hao, L.Y.; Su, Y.Y.; Lv, Y. Metal–organic frameworks (MOFs) combined with ZnO quantum dots as a fluorescent sensing platform for phosphate. Sens. Actuators B Chem. 2014, 197, 50–57. [Google Scholar] [CrossRef]
- Jana, N.R.; Yu, H.H.; Ali, E.M.; Zheng, Y.G.; Ying, J.Y. Controlled photostability of luminescent nanocrystalline ZnO solution for selective detection of aldehydes. Chem. Commun. 2007, 14, 1406–1408. [Google Scholar]
- Zhang, J.; Zhao, S.Q.; Zhang, K.; Zhou, J.Q. Cd-doped ZnO quantum dots-based immunoassay for the quantitative determination of bisphenol A. Chemosphere 2014, 95, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Gu, B.X.; Xu, C.X.; Yang, C.; Liu, S.Q.; Wang, M.L. ZnO quantum dot labeled immunosensor for carbohydrate antigen 19-9. Biosens. Bioelectron. 2011, 26, 2720–2723. [Google Scholar] [CrossRef] [PubMed]
- Sadollahkhani, A.; Hatamie, A.; Nur, O.; Willander, M.; Zargar, B.; Kazeminezhad, I. Colorimetric disposable paper coated with ZnO@ZnS core-shell nanoparticles for detection of copper ions in aqueous solutions. ACS Appl. Mater. Interfaces 2014, 6, 17694–17701. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.L.; Hermans, A.; Seipel, A.T.; Wightman, R.M. Monitoring rapid chemical communication in the brain. Chem. Rev. 2008, 108, 2554–2584. [Google Scholar] [CrossRef] [PubMed]
- Bruchez, M.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.C.W.; Nie, S.M. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 1998, 281, 2016–2018. [Google Scholar] [CrossRef] [PubMed]
- Jamieson, T.; Bakhshi, R.; Petrova, D.; Pocock, R.; Imani, M.; Seifalian, A.M. Biological applications of quantum dots. Biomaterials 2007, 28, 4717–4732. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.T.; Shen, J.; Banerjee, P.; Zhou, S.Q. A multifuntional nanoplatform based on responsive fluorescent plasmonic ZnO-Au@PEG hybrid nanogels. Adv. Funct. Mater. 2011, 21, 2830–2839. [Google Scholar] [CrossRef]
- Tang, X.S.; Choo, E.S.G.; Li, L.; Ding, J.; Xue, J.M. One-pot synthesis of water-stable ZnO nanoparticles via a polyol hydrolysis route and their cell labeling applications. Langmuir 2009, 25, 5271–5275. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.Y.; Liang, J.; Zheng, Z.Z.; Wang, H.H.; Xiong, H.M. The application of ZnO luminescent nanoparticles in labeling mice. Contrast Media Mol. Imaging 2011, 6, 328–330. [Google Scholar] [PubMed]
- Xiong, H.M. ZnO nanoparticles applied to bioimaging and drug delivery. Adv. Mater. 2013, 25, 5329–5335. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.J.; Yang, J.; Gong, J.W.; Zheng, N.F. Photo-and pH-triggered release of anticancer drugs from mesoporous silica-coated Pd@Ag nanoparticles. Adv. Funct. Mater. 2012, 22, 842–848. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, W.G. ZnO QD@PMAA-co-PDMAEMA nonviral vector for plasmid DNA delivery and bioimaging. Biomaterials 2010, 31, 3087–3094. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.L.; Ai, K.L.; Yuan, Q.H.; Lu, L.H. Fluorescence-enhanced gadolinium-doped zinc oxide quantum dots for magnetic resonance and fluorescence imaging. Biomaterials 2011, 32, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Aldeek, F.; Mustin, C.; Balan, L.; Medjahdi, G.; Roques-Carmes, T.; Arnoux, P.; Schneider, R. Enhanced photostability from CdSe (S)/ZnO core/shell quantum dots and their use in biolabeling. Eur. J. Inorg. Chem. 2011, 2011, 794–801. [Google Scholar] [CrossRef]
- Zhang, H.J.; Xiong, H.M.; Ren, Q.G.; Xia, Y.Y.; Kong, J.L. ZnO@silica core-shell nanoparticles with remarkable luminescence and stability in cell imaging. J. Mater. Chem. 2012, 22, 13159–13165. [Google Scholar] [CrossRef]
- Kachynski, A.V.; Kuzmin, A.N.; Nyk, M.; Roy, I.; Prasad, P.N. Zinc oxide nanocrystals for nonresonant nonlinear optical microscopy in biology and medicine. J. Phys. Chem. C 2008, 112, 10721–10724. [Google Scholar] [CrossRef] [PubMed]
- Zvyagin, A.V.; Zhao, X.; Gierden, A.; Sanchez, W.; Ross, J.A.; Roberts, M.S. Imaging of zinc oxide nanoparticle penetration in human skin in vitro and in vivo. J. Biomed. Opt. 2008, 13, 064031–064039. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Kelf, T.A.; Sanchez, W.H.; Roberts, M.S.; Ri Ka, J.; Frenz, M.; Zvyagin, A.V. Characterization of optical properties of ZnO nanoparticles for quantitative imaging of transdermal transport. Biomed. Opt. Express 2011, 2, 3321–3333. [Google Scholar] [CrossRef] [PubMed]
- Urban, B.E.; Neogi, P.B.; Butler, S.J.; Fujita, Y.; Neogi, A. Second harmonic imaging of plants tissues and cell implosion using two-photon process in ZnO nanoparticles. J. Biophoton. 2012, 5, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Urban, B.E.; Neogi, P.; Senthilkumar, K.; Rajpurohit, S.K.; Jagadeeshwaran, P.; Seongcheol, K.; Fujita, Y.; Neogi, A. Bioimaging using the optimized nonlinear optical properties of ZnO nanoparticles. IEEE J. Sel. Top. Quant. Electron. 2012, 18, 1451–1456. [Google Scholar] [CrossRef]
- Roberts, M.S.; Roberts, M.J.; Robertson, T.A.; Sanchez, W.; Thörling, C.; Zou, Y.H.; Zhao, X.; Becker, W.; Zvyagin, A.V. In vitro and in vivo imaging of xenobiotic transport in human skin and in the rat liver. J. Biophoton. 2008, 1, 478–493. [Google Scholar]
- Zhang, H.J.; Xiong, H.M. Biological applications of ZnO nanoparticles. Current Molecular Imaging 2013, 2, 177–192. [Google Scholar] [CrossRef]
- Pantazis, P.; Maloney, J.; Wu, D.; Fraser, S.E. Second harmonic generating (SHG) nanoprobes for in vivo imaging. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 14535–14540. [Google Scholar] [CrossRef] [PubMed]
- Berezin, M.Y.; Achilefu, S. Fluorescence lifetime measurements and biological imaging. Chem. Rev. 2010, 110, 2641–2684. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.S.; Dancik, Y.; Prow, T.W.; Thorling, C.A.; Lin, L.L.; Grice, J.E.; Robertson, T.A.; König, K.; Becker, W. Non-invasive imaging of skin physiology and percutaneous penetration using fluorescence spectral and lifetime imaging with multiphoton and confocal microscopy. Eur. J. Pharm. Biopharm. 2011, 77, 469–488. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.; McKee, T.; DiTomaso, E.; Pluen, A.; Seed, B.; Boucher, Y.; Jain, R.K. Dynamic imaging of collagen and its modulation in tumors in vivo using second-harmonic generation. Nat. Med. 2003, 9, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Hellström, S.; Ågren, H. Nonlinear optical properties of quantum dots: Excitons in nanostructures. J. Nonlinear Opt. Phys. Mater. 2009, 18, 195–226. [Google Scholar] [CrossRef]
- Urban, B.E.; Lin, J.; Kumar, O.; Senthilkumar, K.; Fujita, Y.; Neogi, A. Optimization of nonlinear optical properties of ZnO micro and nanocrystals for biophotonics. Opt. Mater. Express 2011, 1, 658–669. [Google Scholar] [CrossRef]
- Foreman, J.V.; Everitt, H.O.; Yang, J.; McNicholas, T.; Liu, J. Effects of reabsorption and spatial trap distributions on the radiative quantum efficiencies of ZnO. Phys. Rev. B 2010, 81, 115318. [Google Scholar] [CrossRef]
- Na, R.; Stender, I.M.; Ma, L.X.; Wulf, H.C. Autofluorescence spectrum of skin: Component bands and body site variations. Skin Res. Technol. 2000, 6, 112–117. [Google Scholar] [CrossRef] [PubMed]
- McGuinness, C.D.; Macmillan, A.M.; Sagoo, K.; McLoskey, D.; Birch, D.J.S. Excitation of fluorescence decay using a 265 nm pulsed light-emitting diode: Evidence for aqueous phenylalanine rotamers. Appl. Phys. Lett. 2006, 89, 063901. [Google Scholar] [CrossRef]
- Ashikawa, I.; Nishimura, Y.; Tsuboi, M.; Watanabe, K.; Iso, K. Lifetime of tyrosine fluorescence in nucleosome core particles. J. Biochem. 1982, 91, 2047–2055. [Google Scholar] [PubMed]
- Alcala, J.R.; Gratton, E.; Prendergast, F.G. Interpretation of fluorescence decays in proteins using continuous lifetime distributions. Biophys. J. 1987, 51, 925–936. [Google Scholar] [CrossRef]
- Grajek, H.; Gryczynski, I.; Bojarski, P.; Gryczynski, Z.; Bharill, S.; Kułak, L. Flavin mononucleotide fluorescence intensity decay in concentrated aqueous solutions. Chem. Phys. Lett. 2007, 439, 151–156. [Google Scholar] [CrossRef]
- Koziol, B.; Markowicz, M.; Kruk, J.; Plytycz, B. Riboflavin as a source of autofluorescence in eisenia fetida coelomocytes. Photochem. Photobiol. 2006, 82, 570–573. [Google Scholar] [CrossRef] [PubMed]
- König, K. Clinical multiphoton tomography. J. Biophoton. 2008, 1, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, D.; Schenke, S.; Hammer, M.; Schweitzer, F.; Jentsch, S.; Birckner, E.; Becker, W.; Bergmann, A. Towards metabolic mapping of the human retina. Microsc. Res. Tech. 2007, 70, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Niesner, R.; Peker, B.; Schlüsche, P.; Gericke, K.H. Noniterative biexponential fluorescence lifetime imaging in the investigation of cellular metabolism by means of NAD (P) H autofluorescence. ChemPhysChem 2004, 5, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- De Beule, P.A.A.; Dunsby, C.; Galletly, N.P.; Stamp, G.W.; Chu, A.C.; Anand, U.; Anand, P.; Benham, C.D.; Naylor, A.; French, P.M.W. A hyperspectral fluorescence lifetime probe for skin cancer diagnosis. Rev. Sci. Instrum. 2007, 78, 123101. [Google Scholar] [CrossRef] [PubMed]
- Dimitrow, E.; Riemann, I.; Ehlers, A.; Koehler, M.J.; Norgauer, J.; Elsner, P.; König, K.; Kaatz, M. Spectral fluorescence lifetime detection and selective melanin imaging by multiphoton laser tomography for melanoma diagnosis. Exp. Dermatol. 2009, 18, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Laiho, L.H.; Pelet, S.; Hancewicz, T.M.; Kaplan, P.D.; So, P.T.C. Two-photon 3-D mapping of ex vivo human skin endogenous fluorescence species based on fluorescence emission spectra. J. Biomed. Opt. 2005, 10, 024016:1–024016:10. [Google Scholar] [CrossRef] [PubMed]
- Pena, A.; Strupler, M.; Boulesteix, T.; Schanne-Klein, M. Spectroscopic analysis of keratin endogenous signal for skin multiphoton microscopy. Opt. Express 2005, 13, 6268–6274. [Google Scholar] [CrossRef] [PubMed]
- Xiong, H.M.; Ma, R.Z.; Wang, S.F.; Xia, Y.Y. Photoluminescent ZnO nanoparticles synthesized at the interface between air and triethylene glycol. J. Mater. Chem. 2011, 21, 3178–3182. [Google Scholar] [CrossRef]
- Lin, J.; Fujita, Y.; Neogi, A. Saturation of two photon emission in ZnO nanoparticles with second order nonlinearity. RSC Adv. 2015, 5, 10921–10926. [Google Scholar] [CrossRef]
- Espitia, P.; Soares, N.D.; Coimbra, J.D.; de Andrade, N.; Cruz, R.; Medeiros, E. Zinc oxide nanoparticles: Synthesis, antimicrobial activity and food packaging applications. Food Bioprocess Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Yang, Y.J.; Lan, J.F.; Xu, Z.G.; Chen, T.; Zhao, T.; Cheng, T.; Shen, J.M.; Lv, S.Y.; Zhang, H.X. Toxicity and biodistribution of aqueous synthesized ZnS and ZnO quantum dots in mice. Nanotoxicology 2014, 8, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.L.; Ding, Y.L.; Povey, M.; York, D. ZnO nanofluids-A potential antibacterial agent. Prog. Nat. Sci. 2008, 18, 939–944. [Google Scholar] [CrossRef]
- Raghupathi, K.R.; Koodali, R.T.; Manna, A.C. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir 2011, 27, 4020–4028. [Google Scholar] [CrossRef] [PubMed]
- Bellanger, X.; Billard, P.; Schneider, R.; Balan, L.; Merlin, C. Stability and toxicity of ZnO quantum dots: Interplay between nanoparticles and bacteria. J. Hazard. Mater. 2015, 283, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.E.; Madler, L.; Velegol, D.; Xia, T.; Hoek, E.M.V.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding biophysicochemical interactions at the nano-bio interface. Nat. Mater. 2009, 8, 543–557. [Google Scholar] [CrossRef] [PubMed]
- Jalal, R.; Goharshadi, E.K.; Abareshi, M.; Moosavi, M.; Yousefi, A.; Nancarrow, P. ZnO nanofluids: Green synthesis, characterization, and antibacterial activity. Mater. Chem. Phys. 2010, 121, 198–201. [Google Scholar] [CrossRef]
- Applerot, G.; Lipovsky, A.; Dror, R.; Perkas, N.; Nitzan, Y.; Lubart, R.; Gedanken, A. Enhanced antibacterial activity of nanocrystalline ZnO due to increased ROS-mediated cell injury. Adv. Funct. Mater. 2009, 19, 842–852. [Google Scholar] [CrossRef]
- Adams, L.K.; Lyon, D.Y.; Alvarez, P. J.J. Comparative eco-toxicity of nanoscale TiO2, SiO2, and ZnO water suspensions. Water Res. 2006, 40, 3527–3532. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Chakraborti, S.; Chakrabarti, P.; Haranath, D.; Shanker, V.; Ansari, Z.A.; Singh, S.P.; Gupta, V. Role of surface adsorbed anionic species in antibacterial activity of ZnO quantum dots against Escherichia coli. J. Nanosci. Nanotechnol. 2009, 9, 6427–6433. [Google Scholar] [CrossRef] [PubMed]
- Ghule, K.; Ghule, A.V.; Chen, B.J.; Ling, Y.C. Preparation and characterization of ZnO nanoparticles coated paper and its antibacterial activity study. Green Chem. 2006, 8, 1034–1041. [Google Scholar] [CrossRef]
- Matai, I.; Sachdev, A.; Dubey, P.; Uday Kumar, S.; Bhushan, B.; Gopinath, P. Antibacterial activity and mechanism of Ag-ZnO nanocomposite on S. aureus and GFP-expressing antibiotic resistant E. coli. Colloids Surf. B 2014, 115, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Noimark, S.; Weiner, J.; Noor, N.; Allan, E.; Williams, C.K.; Shaffer, M.S.P.; Parkin, I.P. Dual-mechanism antimicrobial polymer-ZnO nanoparticle and crystal violet-encapsulated silicone. Adv. Funct. Mater. 2015, 25, 1367–1373. [Google Scholar] [CrossRef]
- Karthik, K.; Dhanuskodi, S.; Gobinath, C.; Sivaramakrishnan, S. Microwave-assisted synthesis of CdO-ZnO nanocomposite and its antibacterial activity against human pathogens. Spectrochim. Acta A 2015, 139, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.D. Similarities and differences in the responses of microorganisms to biocides. J. Antimicrob. Chemother. 2003, 52, 750–763. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, M. Cancer nanotechnology: Opportunities and challenges. Nat. Rev. Cancer 2005, 5, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.J.; Su, T.Y.; Chen, H.L.; Liu, H.L.; Chiang, W.L.; Chu, P.C.; Xia, Y.N.; Sung, H.W. Smart multifunctional hollow microspheres for the quick release of drugs in intracellular lysosomal compartments. Angew. Chem. Int. Ed. 2011, 50, 8086–8089. [Google Scholar] [CrossRef] [PubMed]
- Jin, E.; Zhang, B.; Sun, X.R.; Zhou, Z.X.; Ma, X.P.; Sun, Q.H.; Tang, J.B.; Shen, Y.Q.; Van Kirk, E.; Murdoch, W.J.; Radosz, M. Acid-active cell-penetrating peptides for in vivo tumor-targeted drug delivery. J. Am. Chem. Soc. 2013, 135, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Pearce, T.R.; Shroff, K.; Kokkoli, E. Peptide targeted lipid nanoparticles for anticancer drug delivery. Adv. Mater. 2012, 24, 3803–3822. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.H.; Song, S.Y.; Liu, J.H.; Liu, D.P.; Zhang, H.J. ZnO-functionalized upconverting nanotheranostic agent: Multi-modality imaging-guided chemotherapy with on-demand drug release triggered by pH. Angew. Chem. Int. Ed. 2015, 54, 536–540. [Google Scholar] [CrossRef]
- Lin, Y.S.; Hurley, K.R.; Haynes, C.L. Critical considerations in the biomedical use of mesoporous silica nanoparticles. J. Phys. Chem. Lett. 2012, 3, 364–374. [Google Scholar] [CrossRef]
- Zhu, J.; Liao, L.; Bian, X.J.; Kong, J.L.; Yang, P.Y.; Liu, B.H. pH-Controlled delivery of doxorubicin to cancer cells, based on small mesoporous carbon nanospheres. Small 2012, 8, 2715–2720. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Xu, Y.D.; Ma, Y.Y.; Qiu, L.L.; Wang, Y.; Kong, J.L.; Xiong, H.M. Biodegradable ZnO@polymer core–shell nanocarriers: pH-triggered release of doxorubicin in vitro. Angew. Chem. Int. Ed. 2013, 52, 4127–4131. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, J.W.; Martinez, E.; Louka, P.; Wingett, D.G. Zinc oxide nanoparticles for selective destruction of tumor cells and potential for drug delivery applications. Expert Opin. Drug Deliv. 2010, 7, 1063–1077. [Google Scholar] [CrossRef] [PubMed]
- Cory, H.; Janet, L. Preferential killing of cancer cells and activated human T cells using ZnO nanoparticles. Nanotechnology 2008, 19, 295103. [Google Scholar]
- Kishwar, S.; Asif, M.H.; Nur, O.; Willander, M.; Larsson, P.O. Intracellular ZnO nanorods conjugated with protoporphyrin for local mediated photochemistry and efficient treatment of single cancer cell. Nanoscale Res. Lett. 2010, 5, 1669–1674. [Google Scholar] [CrossRef] [PubMed]
- Kishwar, S.; Siddique, M.; Israr-Qadir, M.; Nur, O.; Willander, M.; Öllinger, K. Investigation of the phototoxic effect of ZnO nanorods on fibroblasts and melanoma human cells. Laser Phys. Lett. 2014, 11, 115606. [Google Scholar] [CrossRef]
- Yuan, Q.; Hein, S.; Misra, R.D.K. New generation of chitosan-encapsulated ZnO quantum dots loaded with drug: Synthesis, characterization and in vitro drug delivery response. Acta Biomater. 2010, 6, 2732–2739. [Google Scholar] [CrossRef] [PubMed]
- Barick, K.C.; Nigam, S.; Bahadur, D. Nanoscale assembly of mesoporous ZnO: A potential drug carrier. J. Mater. Chem. 2010, 20, 6446–6452. [Google Scholar] [CrossRef]
- Muhammad, F.; Guo, M.Y.; Qi, W.X.; Sun, F.X.; Wang, A.F.; Guo, Y.J.; Zhu, G.S. pH-triggered controlled drug release from mesoporous silica nanoparticles via intracelluar dissolution of ZnO nanolids. J. Am. Chem. Soc. 2011, 133, 8778–8781. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, F.; Guo, M.Y.; Guo, Y.J.; Qi, W.X.; Qu, F.Y.; Sun, F.X.; Zhao, H.J.; Zhu, G.S. Acid degradable ZnO quantum dots as a platform for targeted delivery of an anticancer drug. J. Mater. Chem. 2011, 21, 13406–13412. [Google Scholar] [CrossRef]
- Antoine, T.E.; Mishra, Y.K.; Trigilio, J.; Tiwari, V.; Adelung, R.; Shukla, D. Prophylactic, therapeutic and neutralizing effects of zinc oxide tetrapod structures against herpes simplex virus type-2 infection. Antiviral Res. 2012, 96, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Hariharan, R.; Senthilkumar, S.; Suganthi, A.; Rajarajan, M. Synthesis and characterization of doxorubicin modified ZnO/PEG nanomaterials and its photodynamic action. J. Photoch. Photobio. B 2012, 116, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Zhao, T.; Wei, D.F.; Wei, Y.X.; Li, Y.Y.; Zhang, H.X. Core-shell nanocarriers with ZnO quantum dots-conjugated Au nanoparticle for tumor-targeted drug delivery. Carbohyd. Polym. 2013, 92, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.C.; Liu, J.; Zhou, W.H.; Wei, J.C.; Peng, Z.P. A novel thermal and pH responsive drug delivery system based on ZnO@PNIPAM hybrid nanoparticles. Mater. Sci. Eng. C 2014, 45, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, L.; Singh, J.; Agarwal, V.; Pandey, A.C.; Verma, S.; Das, P.; Tewari, R.P. In situ grafted nanostructured ZnO/carboxymethyl cellulose nanocomposites for efficient delivery of curcumin to cancer. J. Polym. Res. 2014, 21, 1–9. [Google Scholar] [CrossRef]
- Kumar, V.B.; Kumar, K.; Gedanken, A.; Paik, P. Facile synthesis of self-assembled spherical and mesoporous dandelion capsules of ZnO: Efficient carrier for DNA and anti-cancer drugs. J. Mater. Chem. B 2014, 2, 3956–3964. [Google Scholar] [CrossRef]
- Ahmad, J.; Wahab, R.; Siddiqui, M.A.; Musarrat, J.; Al-Khedhairy, A.A. Zinc oxide quantum dots: A potential candidate to detain liver cancer cells. Bioprocess Biosyst. Eng. 2015, 38, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, L.; Singh, J.; Agarwal, V.; Pandey, A.C.; Verma, S.P.; Das, P.; Tewari, R.P. Efficient water soluble nanostructured ZnO grafted O-carboxymethyl chitosan/curcumin-nanocomposite for cancer therapy. Process Biochem. 2015, 50, 678–688. [Google Scholar] [CrossRef]
- Wang, M.Y.; Yu, S.N.; Wang, C.; Kong, J.L. Tracking the endocytic pathway of recombinant protein toxin delivered by multiwalled carbon nanotubes. ACS Nano 2010, 4, 6483–6490. [Google Scholar] [CrossRef] [PubMed]
- Edson, J.A.; Kwon, Y.J. RNAi for silencing drug resistance in microbes toward development of nanoantibiotics. J. Control. Release 2014, 189, 150–157. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.-Y.; Xiong, H.-M. Photoluminescent ZnO Nanoparticles and Their Biological Applications. Materials 2015, 8, 3101-3127. https://doi.org/10.3390/ma8063101
Zhang Z-Y, Xiong H-M. Photoluminescent ZnO Nanoparticles and Their Biological Applications. Materials. 2015; 8(6):3101-3127. https://doi.org/10.3390/ma8063101
Chicago/Turabian StyleZhang, Zheng-Yong, and Huan-Ming Xiong. 2015. "Photoluminescent ZnO Nanoparticles and Their Biological Applications" Materials 8, no. 6: 3101-3127. https://doi.org/10.3390/ma8063101
APA StyleZhang, Z.-Y., & Xiong, H.-M. (2015). Photoluminescent ZnO Nanoparticles and Their Biological Applications. Materials, 8(6), 3101-3127. https://doi.org/10.3390/ma8063101





