Polypropylene Biocomposites with Boron Nitride and Nanohydroxyapatite Reinforcements
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials


2.2. Preparation of Nanocomposites
| Specimen | PP (wt%) | hBN (wt%) | nHA (wt%) |
|---|---|---|---|
| PP/5% hBN | 95 | 5 | 0 |
| PP/10% hBN | 90 | 10 | 0 |
| PP/15% hBN | 85 | 15 | 0 |
| PP /20% hBN | 80 | 20 | 0 |
| PP/20% nHA | 80 | 0 | 20 |
| PP/5% hBN-20% nHA | 75 | 5 | 20 |
| PP/10% hBN-20% nHA | 70 | 10 | 20 |
| PP/15% hBN-20% nHA | 65 | 15 | 20 |
2.3. Material Characterization
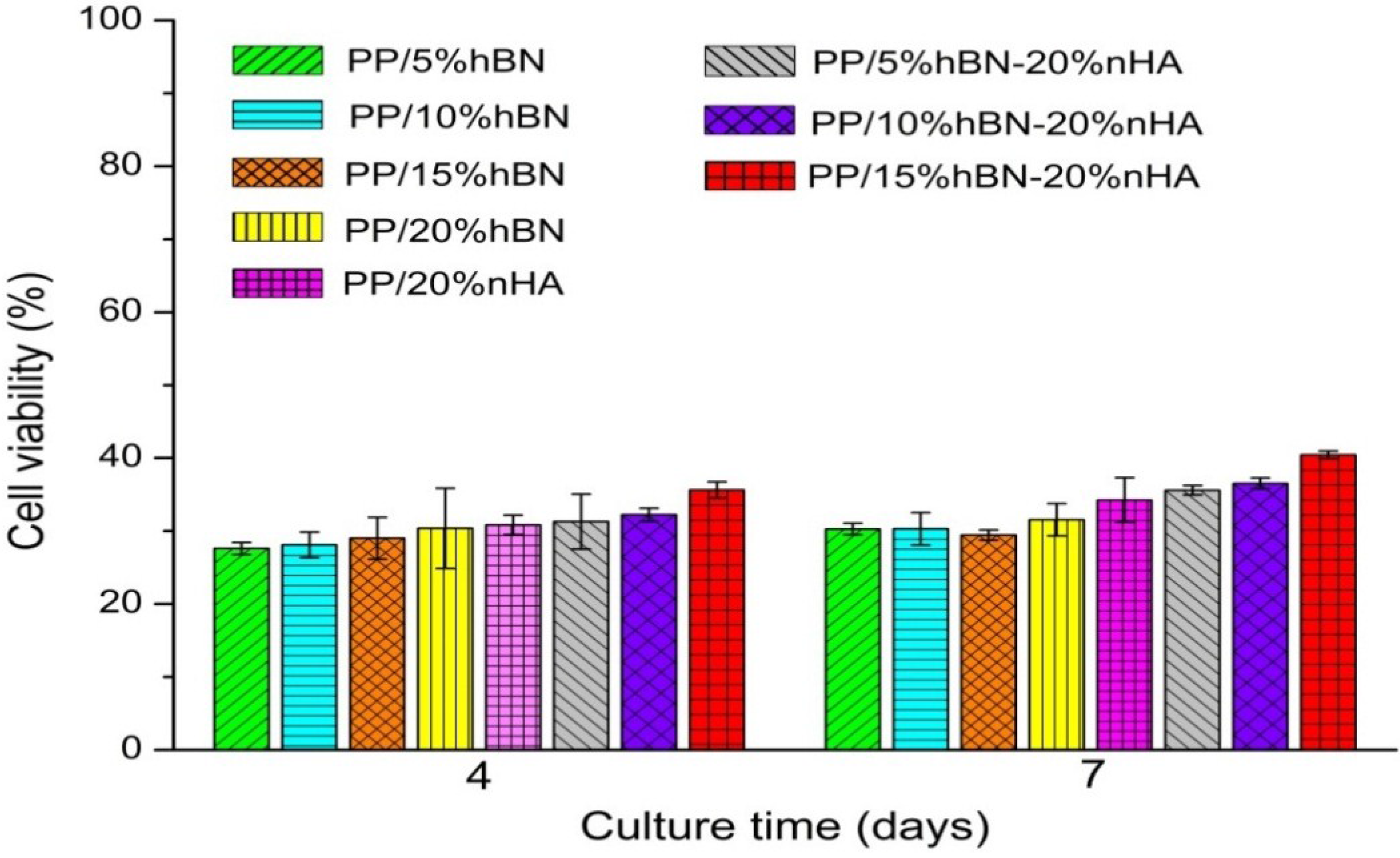
2.4. Cell Seeding and Proliferation
3. Results and Discussion
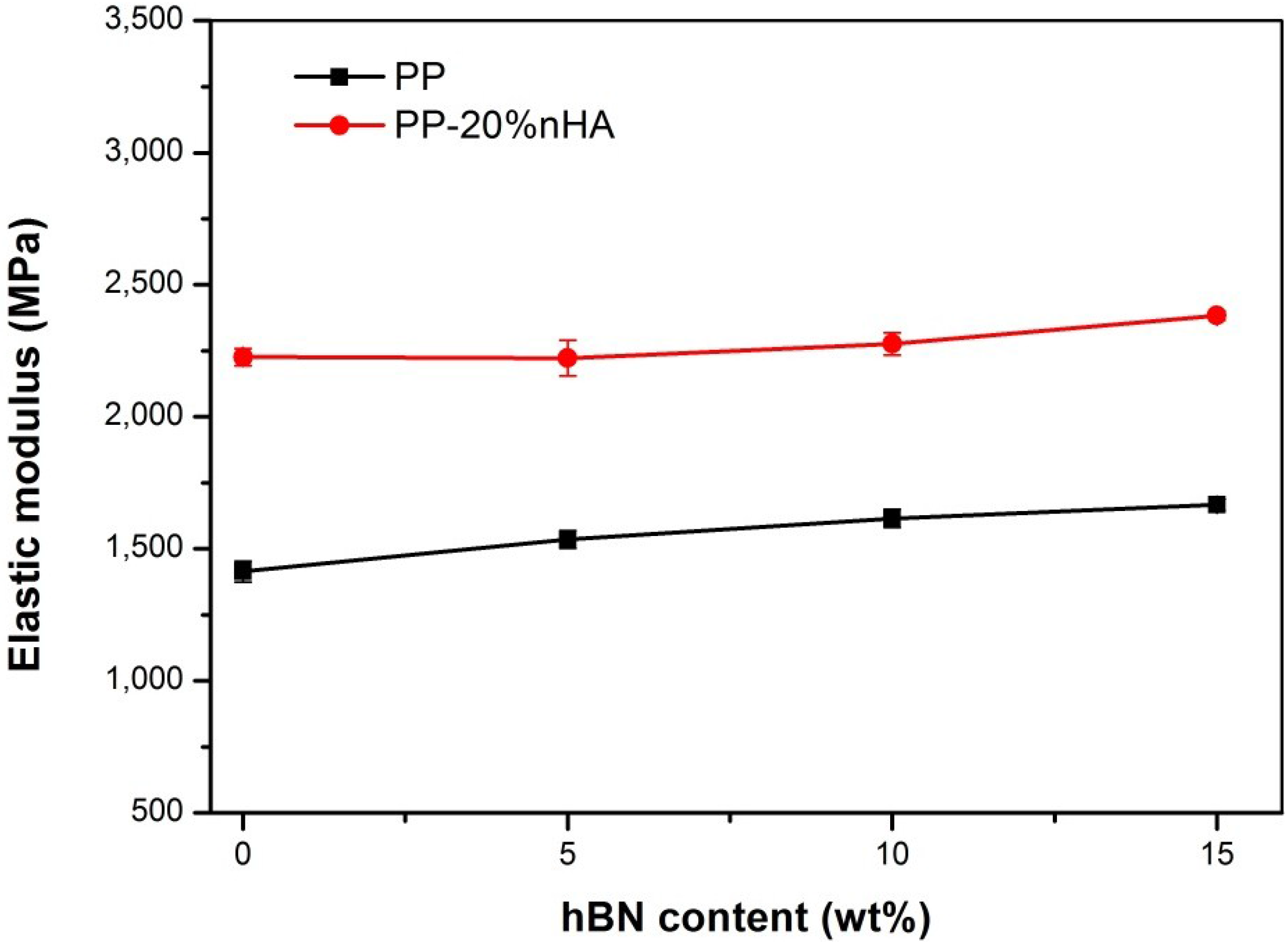
3.1. Morphology and Mechanical Behavior


| Specimen | Elastic modulus, MPa | Tensile stress, MPa | Elongation at break, % |
|---|---|---|---|
| PP | 1,414 ± 40 | 26.2 ± 0.5 | >600 |
| PP/5 wt% (2.2 vol%) hBN | 1,536 ± 34 | 26.8 ± 0.3 | >600 |
| PP/10 wt% (4.5 vol%) hBN | 1,615 ± 35 | 26.6 ± 0.4 | >600 |
| PP/15 wt% (7.0 vol%) hBN | 1,666 ± 21 | 26.4 ± 0.3 | 300 |
| PP/20 wt% (9.7 vol%) hBN | 1,758 ± 33 | 26.5 ± 0.4 | 43.0 ± 6.3 |
| PP/20 wt% (6.67 vol%) nHA | 2,226 ± 33 | 30.9 ± 0.4 | 9.9 ± 0.6 |
| PP/5 wt% hBN-20 wt% nHA | 2,222 ± 68 | 27.0 ± 0.1 | 9.0 ± 2.0 |
| PP/10 wt% hBN-20 wt% nHA | 2,276 ± 42 | 28.6 ± 0.3 | 7.1 ± 0.6 |
| PP/15 wt% hBN-20 wt% nHA | 2,383 ± 18 | 26.9 ± 0.2 | 5.8 ± 0.3 |
| HDPE/10 vol% mHA [44] | 980 ± 20 | 17.3 ± 0.3 | >200 |
| HDPE/20 vol% mHA [44] | 1,600 ± 20 | 17.8 ± 0.1 | 34.0 ± 9.5 |
| HDPE/30 vol% mHA [44] | 2,730 ± 10 | 19.5 ± 0.2 | 6.4 ± 0.5 |
| HDPE/40 vol% mHA [44] | 4,290 ± 17 | 20.7 ± 1.6 | 2.6 ± 0.4 |
| Cortical bone [48] | 7,000‒30,000 | ---------- | 1‒3 |
3.2. Cell Culture and Growth


4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jacobs, J.J.; Gilbert, J.L.; Urban, R.M. Corrosion of metal orthopedic implants. J. Bone Joint. Surg. Am. 1998, 80A, 268–282. [Google Scholar] [PubMed]
- Hallab, N.; Meritt, K.; Jacobs, J.J. Metal sensitivity in patients with orthopedic implants. J. Bone Joint. Surg. Am. 2001, 83A, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Scharf, B.; Clement, C.C.; Zolla, V.; Perino, G.; Yan, B.; Elci, S.G.; Purdue, E.; Goldring, S.; Macaluso, F.; Cobelli, N.; et al. Molecular analysis of chromium and cobalt-related toxicity. Sci. Rep. 2014, 4, 5729:1–5729:12. [Google Scholar] [CrossRef]
- Tjong, S.C.; Hoffman, R.W.; Yeager, E.B. Electron and ion spectroscopic studies of the passive film on iron-chromium alloys. J. Electrochem. Soc. 1982, 129, 1662–1668. [Google Scholar] [CrossRef]
- Tjong, S.C.; Yeager, E.B. ESCA and SIMS studies of the passive film on iron. J. Electrochem. Soc. 1981, 128, 2251–2254. [Google Scholar] [CrossRef]
- Tjong, S.C.; Lau, K.C. Abrasion resistance of stainless steel composites reinforced with hard TiB2 particles. Compos. Sci. Technol. 2000, 60, 1141–1146. [Google Scholar] [CrossRef]
- Schmutz, P.; Quach-Vu, N.C.; Gerber, I. Metallic medical implants: Electrochemical characterization of corrosion processes. Interface 2008, 17, 35–40. [Google Scholar]
- Tjong, S.C.; Meng, Y.Z. Effect of reactive compatibilizers on the mechanical properties of polycarbonate/poly(acrylonitrile-butadiene-styrene) blends. Eur. Polym. J. 2000, 36, 123–129. [Google Scholar] [CrossRef]
- Meng, Y.Z.; Hay, A.S.; Jian, X.G.; Tjong, S.C. Synthesis and properties of poly(aryl ether sulfone)s containing the phthalazinone moiety. J. Appl. Polym. Sci. 1998, 68, 137–143. [Google Scholar] [CrossRef]
- Meng, Y.Z.; Tjong, S.C.; Hay, A.S.; Wang, S.J. Synthesis and proton conductivities of phosphonic acid containing poly-(arylene ether)s. J. Polym. Sci. Part A Polym. Chem. 2001, 39, 3218–3226. [Google Scholar] [CrossRef]
- Du, L.C.; Meng, Y.Z.; Wang, S.J.; Tjong, S.C. Synthesis and degradation behavior of poly(propylene carbonate) derived from carbon dioxide and propylene oxide. J. Appl. Polym. Sci. 2004, 92, 1840–1846. [Google Scholar] [CrossRef]
- Orefice, R.; Clark, A.; West, J.; Brennan, A.; Hench, L. Processing, properties, and in vitro bioactivity of polysulfone-bioactive glass composites. J. Biomed. Mater. Res. A. 2007, 80, 565–580. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Gao, J.; You, X.; Huo, S.; Li, X.; Zhang, Y.; Zhang, W. Fabrication of calcium sulfate/PLLA composite for bone repair. J. Biomed. Mater. Res. A 2005, 73, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Fung, K.L.; Li, R.K.Y.; Tjong, S.C. Interface modification on the properties of sisal fiber-reinforced polypropylene composites. J. Appl. Polym. Sci. 2002, 85, 169–176. [Google Scholar] [CrossRef]
- Tjong, S.C.; Liu, S.L.; Li, R.K.Y. Mechanical properties of injection moulded blends of polypropylene with thermotropic liquid crystalline polymer. J. Mater. Sci. 1996, 31, 479–484. [Google Scholar] [CrossRef]
- Tjong, S.C.; Meng, Y.Z. Performance of potassium titanate whisker reinforced polyamide-6 composites. Polymer 1998, 39, 5461–5466. [Google Scholar] [CrossRef]
- Liang, J.Z.; Li, R.K.Y.; Tjong, S.C. Tensile properties and morphology of PP/EPDM/glass bead ternary composites. Polym. Compos. 1999, 20, 413–422. [Google Scholar] [CrossRef]
- Li, X.H.; Tjong, S.C.; Meng, Y.Z.; Zhu, Q. Fabrication and properties of poly(propylene carbonate)/calcium carbonate composites. J. Polym. Sci. Part B Polym. Phys. 2003, 41, 1806–1813. [Google Scholar] [CrossRef]
- Huang, J.; Disilvio, L.; Wang, M.; Tanner, K.E.; Bonfield, W. In vitro mechanical and biological assessment of hydroxyapatite-reinforced polyethylene composite. J. Mater. Sci. Mater. Med. 1997, 8, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Guild, F.J.; Bonfield, W. Predictive modeling of the mechanical properties and failure processes in hydroxyapatite- polyethylene (HapexTM) composite. J. Mater. Sci. Mater. Med. 1998, 9, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Abu Bakar, M.S.; Cheang, P.; Khor, K.A. Mechanical properties of injection molded hydroxyapatite-polyetheretherketone biocomposites. Compos. Sci. Technol. 2003, 63, 421–425. [Google Scholar] [CrossRef]
- Duncan, T.V. Applications of nanotechnology in food packaging and food safety: Barrier materials, antimicrobials and sensors. J. Colloid. Interf. Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef]
- Hule, R.A.; Pochan, D.J. Polymer nanocomposites for biomedical applications. MRS Bull. 2007, 32, 354–358. [Google Scholar] [CrossRef]
- Zhang, J.; Webster, T.J. Nanotechnology and nanomaterials: Promises for improved tissue regeneration. Nano Today 2009, 4, 66–80. [Google Scholar] [CrossRef]
- Liu, H.; Webster, T.J. Mechanical properties of dispersed ceramic nanoparticles in polymer composites for orthopedic applications. Int. J. Nanomed. 2010, 5, 299–313. [Google Scholar]
- Thein-Han, W.W.; Shah, J.; Misra, R.D.K. Superior in vitro biological response and mechanical properties of an implantable nanostructured biomaterial: Nanohydroxyapatite-silicone rubber composite. Acta Biomater. 2009, 5, 2668–2679. [Google Scholar] [CrossRef] [PubMed]
- Boissard, C.I.R.; Bourban, P.E.; Tami, A.E.; Alini, M.; Egli, D. Nanohydroxyapatite poly(ester urethane) scaffold for bone tissue engineering. Acta Biomater. 2009, 5, 3316–3327. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, C.; Zhai, H.; Zhu, G.; Pan, H.; Xu, X.; Tang, R. Biomimetic graphene oxide–hydroxyapatite composites via in situ mineralization and hierarchical assembly. RSC Adv. 2014, 4, 25398–25403. [Google Scholar] [CrossRef]
- Rajeswari, D.; Gopi, D.; Ramya, S.; Kavitha, L. Investigation of anticorrosive, antibacterial and in vitro biological properties of a sulphonated poly(etheretherketone)/strontium, cerium co-substituted hydroxyapatite composite coating developed on surface treated surgical grade stainless steel for orthopedic applications. RSC Adv. 2014, 4, 61525–61536. [Google Scholar] [CrossRef]
- Jiang, L.X.; Jiang, L.Y.; Xu, L.J.; Han, C.T.; Xiong, C.D. Effect of a new surface-grafting method for nano-hydroxyapatite on the dispersion and the mechanical enhancement for poly(lactide-co-glycolide). Express Polym. Lett. 2014, 8, 133–141. [Google Scholar] [CrossRef]
- Reves, B.T.; Jenning, J.A.; Burngardner, J.D.; Harrgard, W.O. Osteoinductivity assessment of BMP-2 loaded composite chitosan-nano-hydroxyapatite scaffolds in a rat muscle pouch. Materials 2011, 4, 1360–1374. [Google Scholar] [CrossRef]
- Liao, C.Z.; Li, K.; Wong, H.M.; Tong, W.Y.; Yeung, K.W.K.; Tjong, S.C. Novel polypropylene biocomposites reinforced with carbon nanotubes and hydroxyapatite nanorods for bone replacements. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.Z.; Wong, H.M.; Yeung, K.W.K.; Tjong, S.C. The development, fabrication and mechanical characterization of polypropylene composites reinforced with carbon nanofiber and hydroxyapatite nanorod hybrid fillers. Int. J. Nanomed. 2014, 9, 1299–1310. [Google Scholar]
- Muller, J.; Huaux, F.; Lison, D. Respiratory toxicity of carbon nanotubes: How worried should we be? Carbon 2006, 44, 1048–1056. [Google Scholar] [CrossRef]
- Porter, A.; Gass, M.; Muller, K.; Skepper, J.N.; Midgley, P.A.; Welland, M. Direct imaging of single-walled carbon nanotubes in cells. Nat. Nanotechnol. 2007, 2, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Muller, K.H.; Koziol, K.K.; Skepper, J.N.; Midgley, P.A.; Welland, M.E.; Porter, A. Toxicity and imaging of multi-walled carbon nanotubes in human macrophage cells. Biomaterials 2009, 30, 4152–4160. [Google Scholar] [CrossRef] [PubMed]
- Ciofani, G.; Danti, S.; D’Alessandro, D.; Moscato, S.; Menciassi, A. Assessing cytotoxicity of boron nitride nanotubes: Interference with the MTT assay. Biochem. Biophy. Res. Commun. 2010, 394, 405–411. [Google Scholar] [CrossRef]
- Horvath, L.; Magrez, A.; Golberg, D.; Zhi, C.; Bando, Y.; Smajda, R.; Horvath, E.; Forro, L.; Schwaller, B. In vitro investigation of the cellular toxicity of boron nitride nanotubes. ACS Nano 2011, 5, 3800–3810. [Google Scholar] [CrossRef] [PubMed]
- Ciofani, G.; Danti, S.; Genchi, G.G.; D’Alessandro, D.; Pellequer, J.; Odorico, M.; Mattoli, V.; Giorgi, M. Pilot in vivo toxicological investigation of boron nitride nanotubes. Int. J. Nanomed. 2012, 7, 19–24. [Google Scholar]
- Zhang, H.; Yamazaki, T.; Zhi, C.; Hanagata, N. Identification of boron nitride nanosphere-binding peptide for intracellular delivery of CpG olideoxynucleotides. Nanoscale 2012, 4, 6343–6350. [Google Scholar] [CrossRef] [PubMed]
- Lipp, A.; Schwetz, K.A.; Hunold, K. Hexagonal boron nitride: Fabrication, properties and applications. J. Eur. Ceram. Soc. 1989, 5, 3–9. [Google Scholar] [CrossRef]
- Raddaha, N.S.; Cordero-Arias, L.; Cabanas-Polo, S.; Virtanen, S.; Roether, J.A.; Boccaacini, A.R. Electrophoretic deposition of chitosan/h-BN and chitosan/h-BN/TiO2 composite coatings on stainless steel (316L) substrates. Materials 2014, 7, 1814–1829. [Google Scholar] [CrossRef]
- Lu, F.; Wang, F.; Cao, L.; Kong, C.Y.; Huang, X. Hexagonal boron nitride nanomaterials: Advanced towards bioapplications. Nanosci. Nanotechnol. Lett. 2012, 4, 949–961. [Google Scholar] [CrossRef]
- Wang, M.; Joseph, R.; Bonfield, W. Hydroxyapatite-polyethylene composites for bone substitution: Effects of ceramic particle size and morphology. Biomaterials 1998, 19, 2357–2366. [Google Scholar] [CrossRef] [PubMed]
- Tjong, S.C.; Meng, Y.Z. Impact-modified polypropylene/vermiculite nanocomposites. J. Polym. Sci. Part B Polym. Phys. 2003, 41, 2332–2341. [Google Scholar] [CrossRef]
- Coskunses, F.I.; Yilmazer, U. Preparation and characterization of low density polyethylene/ethylene methyl acrylate glycidyl methacrylate/organoclay nanocomposites. J. Appl. Polym. Sci. 2011, 120, 3087–3097. [Google Scholar] [CrossRef]
- Standard Test Method for Tensile Properties of Plastics; ASTM D638-08; ASTM: West Conshohocken, PA, USA, 2008.
- Hench, L.L.; Wilson, J. An Introduction to Bioceramics; World Scientific: Singapore, 1993. [Google Scholar]
- Tjong, S.C.; Bao, S.P. Fracture toughness of high density polyethylene/SEBS-g-MA/montmorillonite nanocomposites. Compos. Sci. Technol. 2007, 67, 314–323. [Google Scholar] [CrossRef]
- Tjong, S.C.; Meng, Y.Z. Morphology and mechanical characteristics of compatibilized polyamide 6-liquid crystalline polymer composites. Polymer 1997, 38, 4609–4615. [Google Scholar] [CrossRef]
- Xie, X.L.; Li, B.G.; Pan, Z.R.; Li, R.K.Y.; Tjong, S.C. Effect of talc/MMA in situ polymerization on mechanical properties of PVC-matrix composites. J. Appl. Polym. Sci. 2001, 80, 2105–2112. [Google Scholar] [CrossRef]
- Downes, R.N.; Vardy, S.; Tanner, K.E.; Bonfield, W. Hydroxyapatite polyethylene composite in orbital surgery. Bioceramics 1991, 4, 239–246. [Google Scholar]
- Dalmas, F.; Cavaille, J.Y.; Gauthier, C.; Chazeau, L.; Dendievel, R. Viscoelastic behavior and electrical properties of flexible nanofiber filled polymer nanocomposites. Influence of processing conditions. Compos. Sci. Technol. 2007, 67, 829–839. [Google Scholar] [CrossRef]
- Capadona, J.R.; van Den Berg, O.; Capadona, L.; Schroeter, M.; Rowan, S.J.; Tyler, D.J.; Weder, C. A versatile approach for the processing of polymer nanocomposites with self-assembled nanofiber templates. Nat. Nanotechnol. 2007, 2, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Quali, N.; Cavaille, J.Y.; Perez, J. Elastic, viscoelastic and plastic behavior of multiphase polymer blends. Plast. Rubber Compos. Proces Appl. 1991, 16, 55–60. [Google Scholar]
- Kolarik, J. Simultaneous prediction of the modulus, tensile strength and gas permeability of binary polymer blends. Eur. Polym. J. 1997, 34, 585–590. [Google Scholar] [CrossRef]
- Webster, T.J.; Ergun, C.; Doremus, R.H.; Siegel, R.W.; Bizios, R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials 2000, 21, 1803–1810. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, K.W.; Wong, H.M.; Yeung, K.W.K.; Tjong, S.C. Polypropylene Biocomposites with Boron Nitride and Nanohydroxyapatite Reinforcements. Materials 2015, 8, 992-1008. https://doi.org/10.3390/ma8030992
Chan KW, Wong HM, Yeung KWK, Tjong SC. Polypropylene Biocomposites with Boron Nitride and Nanohydroxyapatite Reinforcements. Materials. 2015; 8(3):992-1008. https://doi.org/10.3390/ma8030992
Chicago/Turabian StyleChan, Kai Wang, Hoi Man Wong, Kelvin Wai Kwok Yeung, and Sie Chin Tjong. 2015. "Polypropylene Biocomposites with Boron Nitride and Nanohydroxyapatite Reinforcements" Materials 8, no. 3: 992-1008. https://doi.org/10.3390/ma8030992
APA StyleChan, K. W., Wong, H. M., Yeung, K. W. K., & Tjong, S. C. (2015). Polypropylene Biocomposites with Boron Nitride and Nanohydroxyapatite Reinforcements. Materials, 8(3), 992-1008. https://doi.org/10.3390/ma8030992






