Chitosan-Coated Collagen Membranes Promote Chondrocyte Adhesion, Growth, and Interleukin-6 Secretion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Engineering of Chitosan-Coated Collagen Membranes
2.3. Material Characterization
2.3.1. Fourier Transform Infrared (FTIR) Characterization
2.3.2. Scanning Electron Microscopy (SEM) Characterization
2.3.3. X-ray Photoelectron Spectroscopy (XPS) Characterization
2.3.4. Mechanical Properties and Tensile Strength Characterization
2.3.5. Evaluate Potential Degradation of the Membranes
2.4. Chondrocyte Culture on Chitosan-Coated Collagen Membranes
2.4.1. Cell Adhesion as Determined by Hoechst Staining
2.4.2. Investigation of Cell Proliferation by Mean of MTT Assay
2.4.3. Evaluation of the Number of Live Cells after Culture on Chitosan-Coated Collagen Membranes
2.4.4. Interleuk-6 Quantification Following Chondrocyte Culture on Chitosan-Coated Collagen Membranes
3. Statistical Analyses
4. Results and Discussion
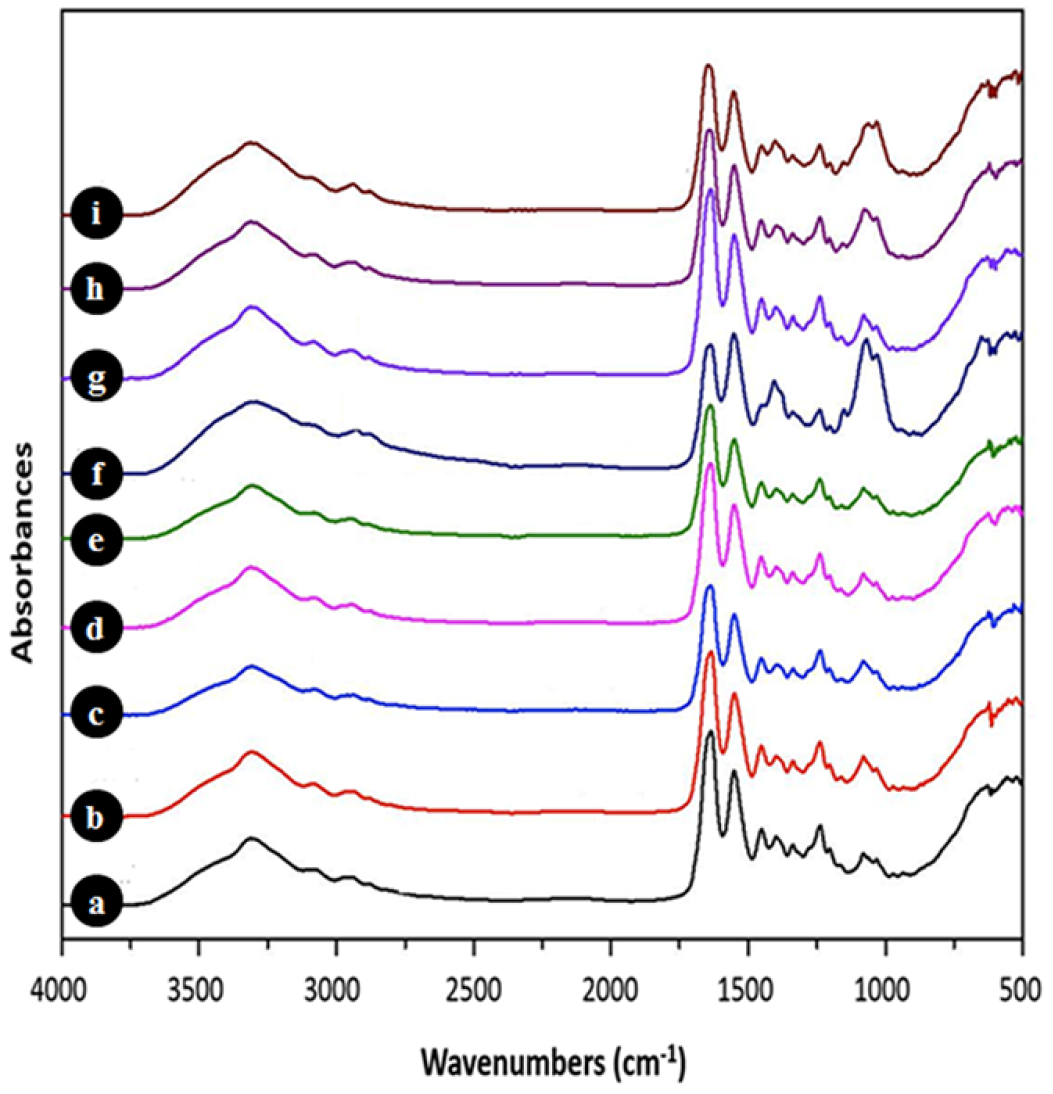


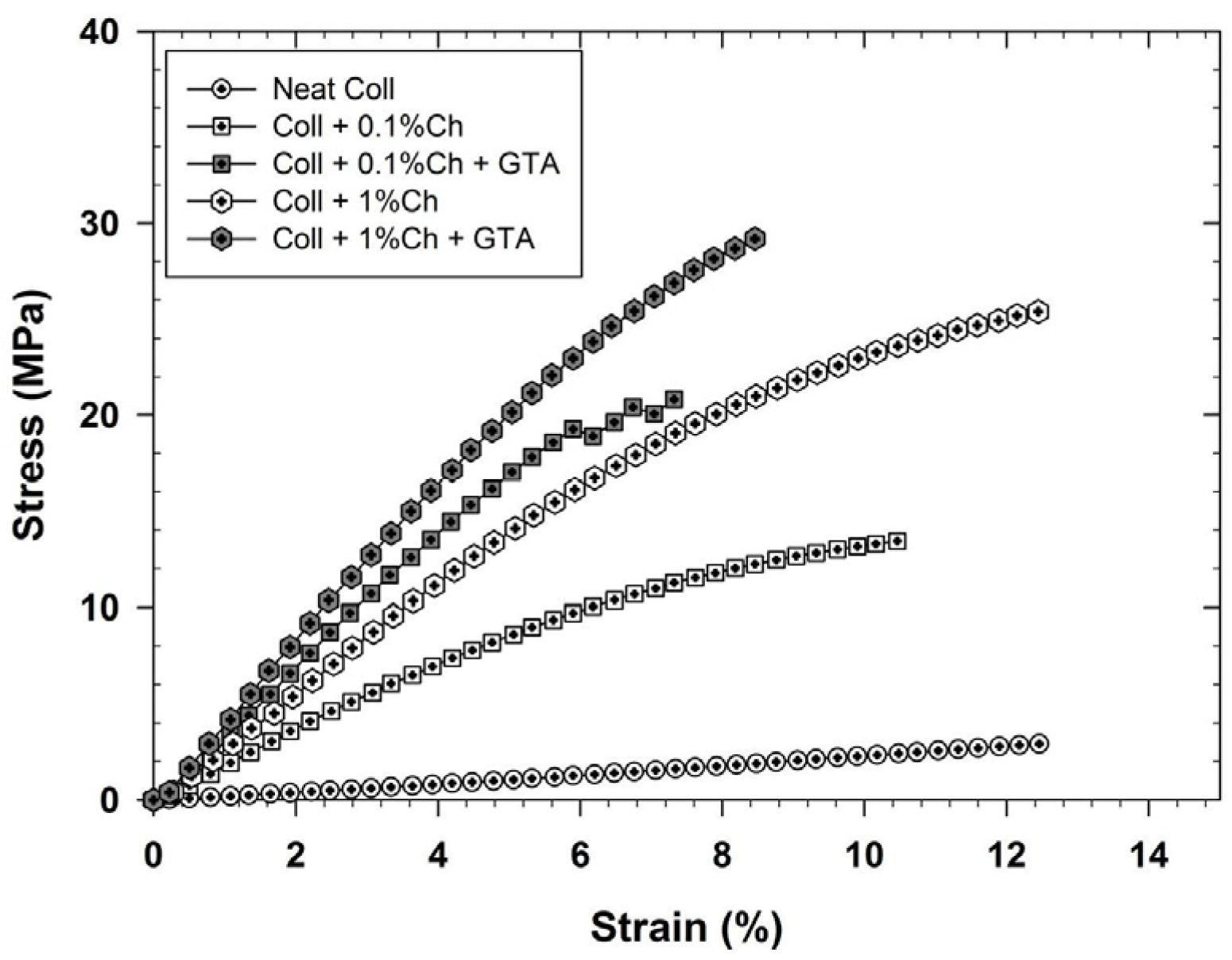
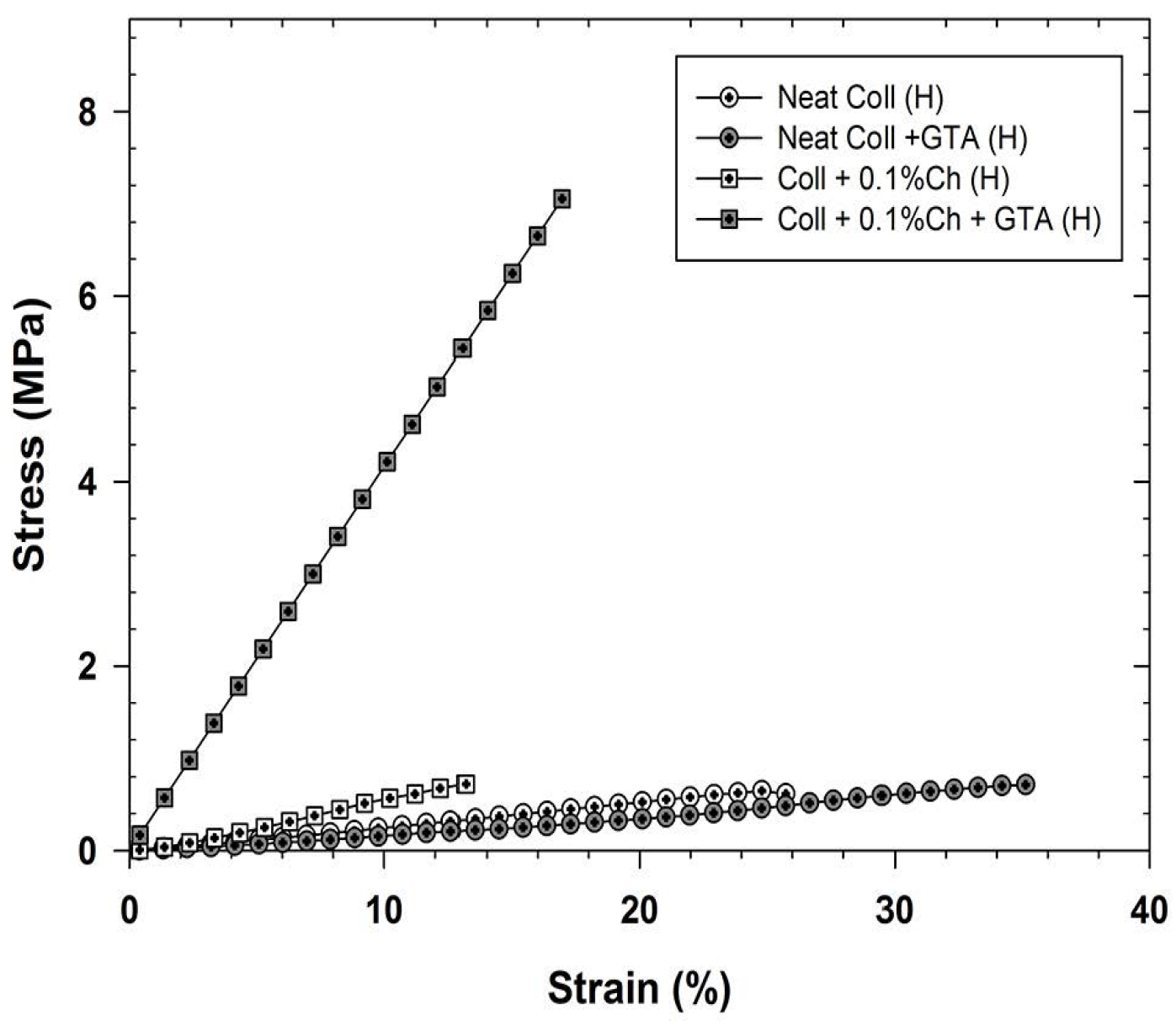
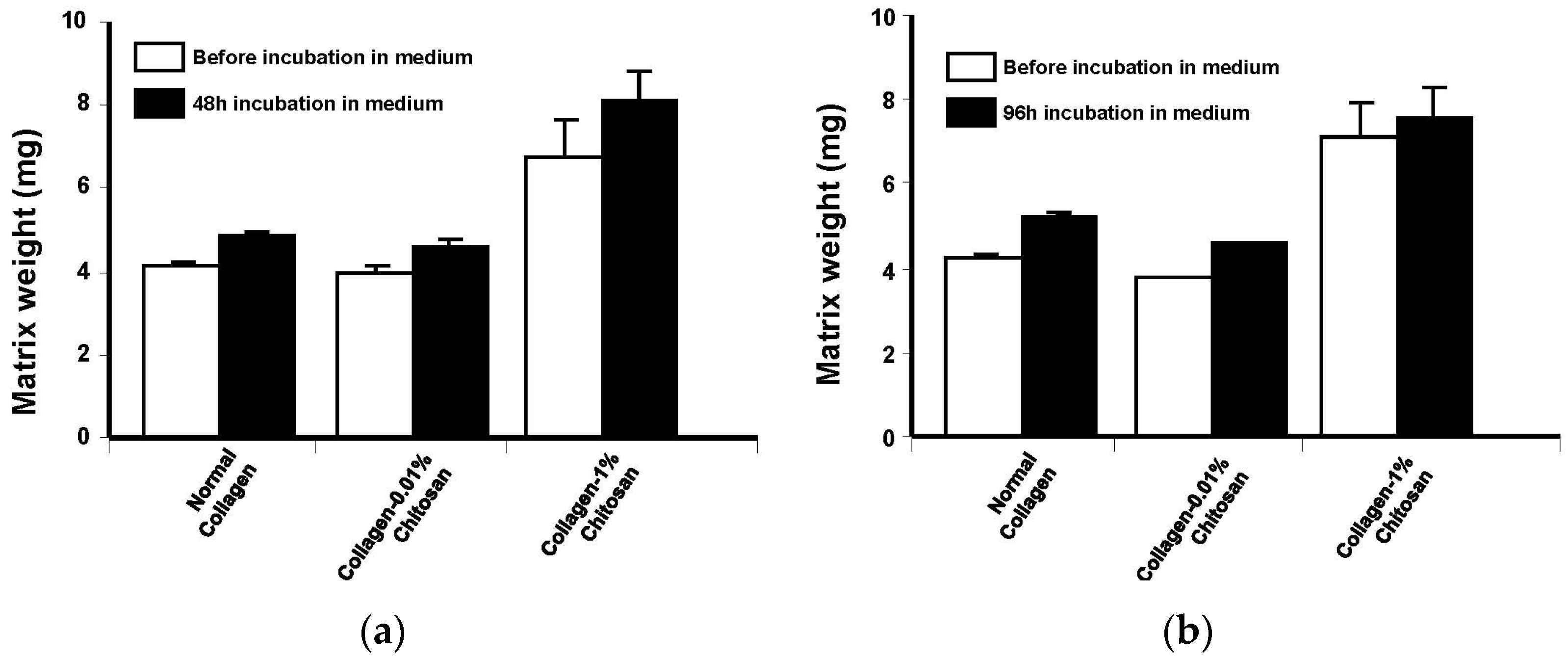
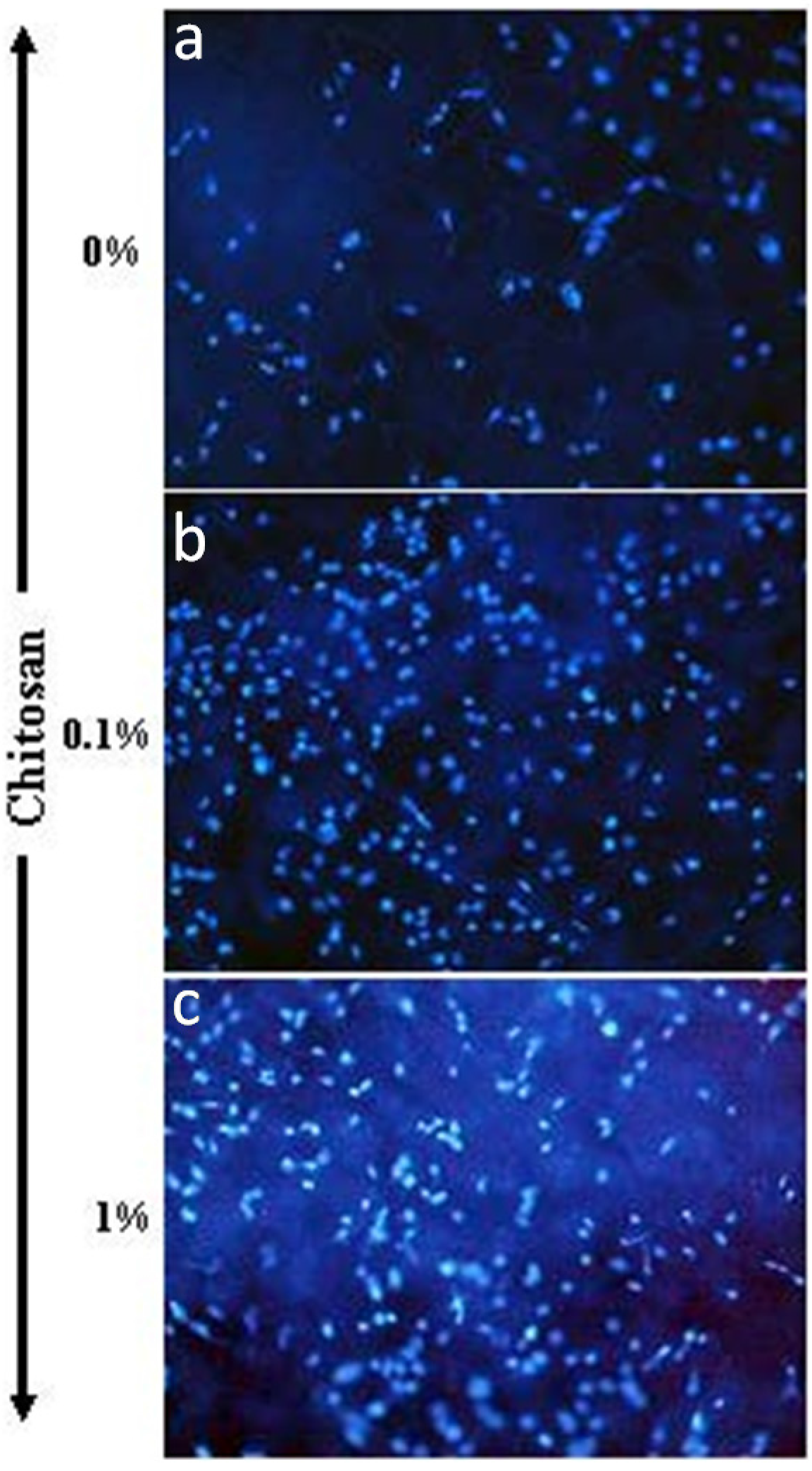
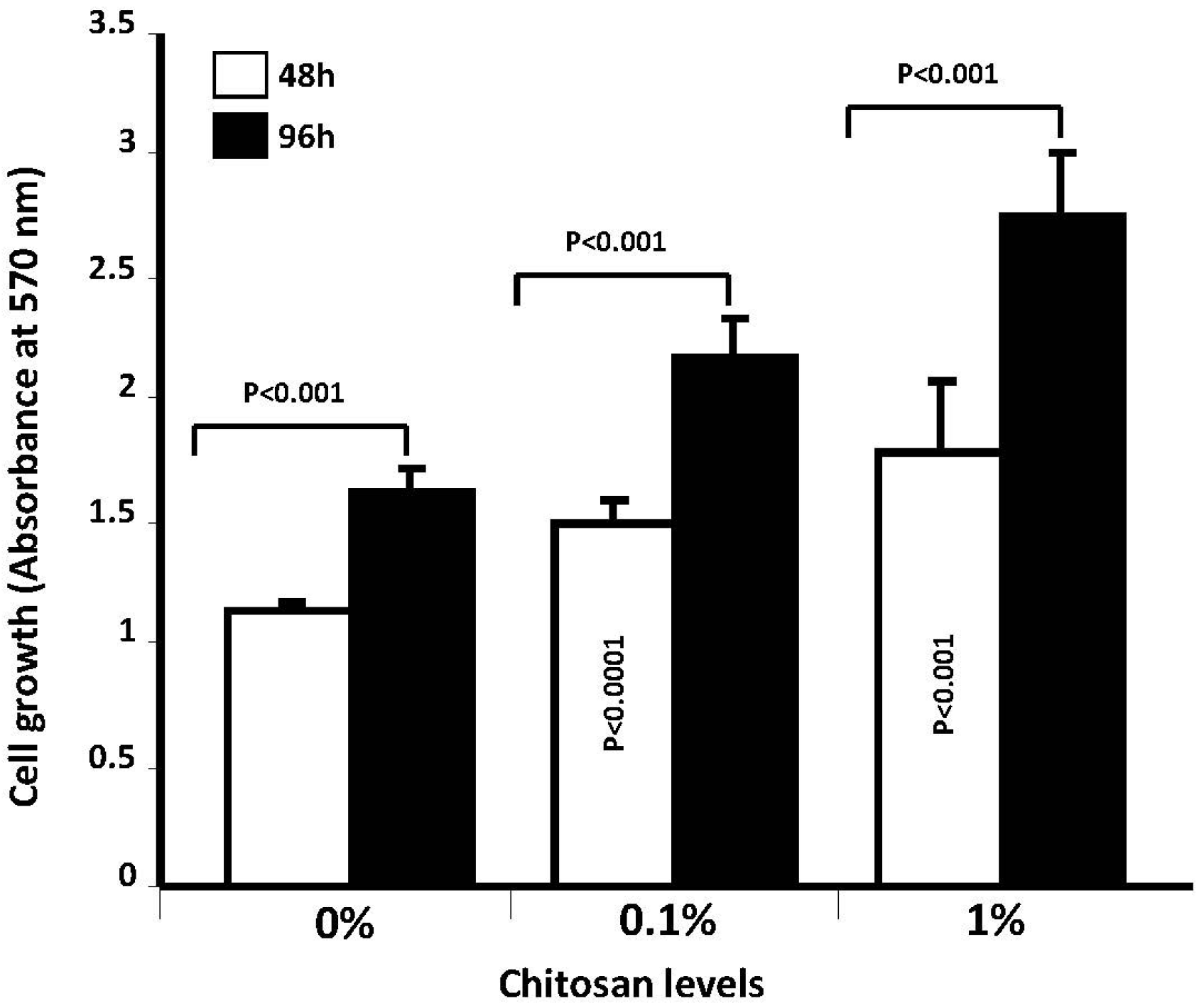
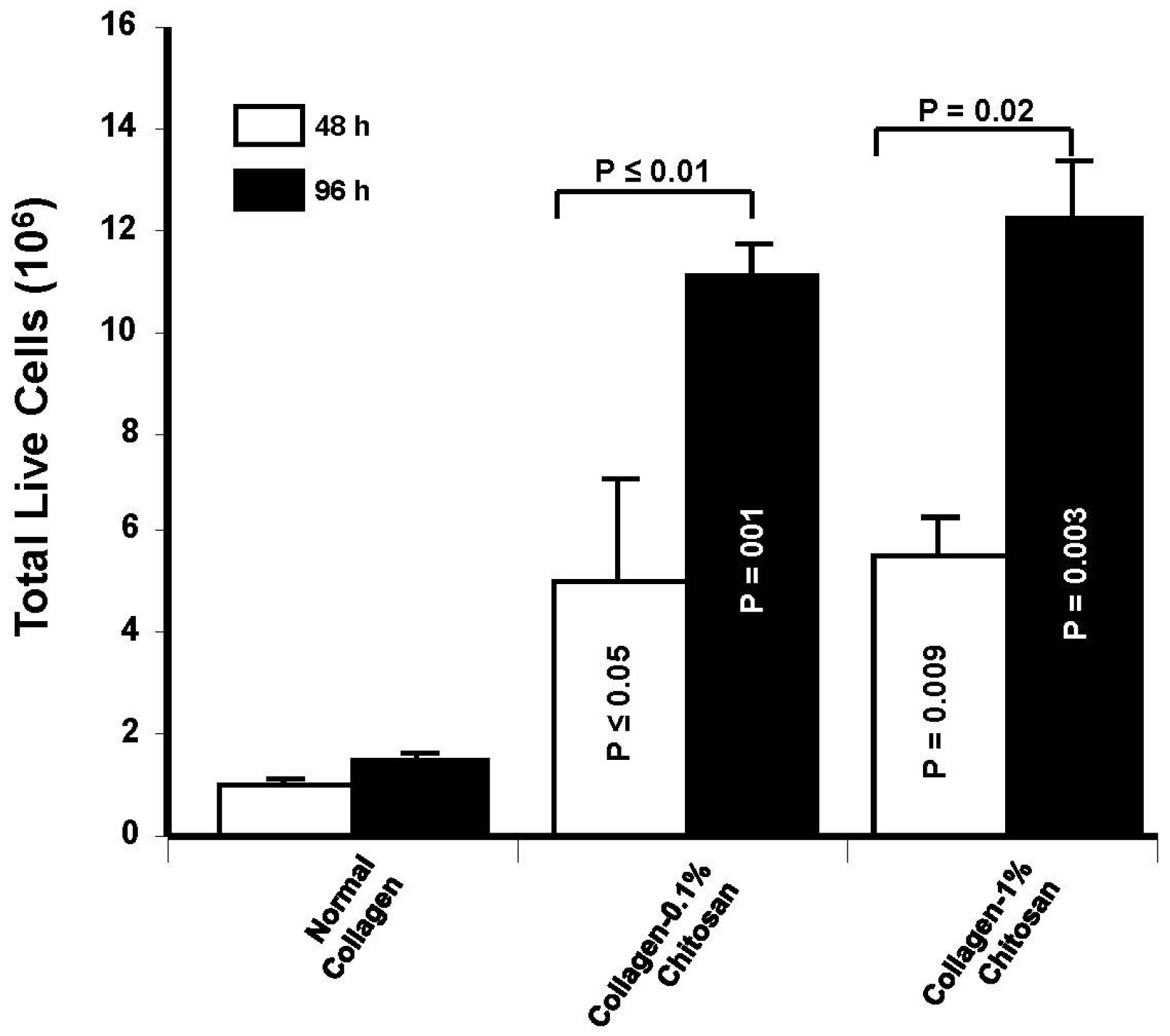

5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Curl, W.W.; Krome, J.; Gordon, E.S.; Rushing, J.; Smith, B.P.; Poehling, G.G. Cartilage injuries: A review of 31,516 knee arthroscopies. Arthrosc. J. Arthrosc. Relat. Surg. 1997, 13, 456–460. [Google Scholar] [CrossRef]
- Hjelle, K.; Solheim, E.; Strand, T.; Muri, R.; Brittberg, M. Articular cartilage defects in 1000 knee arthroscopies. Arthrosc. J. Arthrosc. Relat. Surg. 2002, 18, 730–734. [Google Scholar] [CrossRef]
- Chikanza, I.; Fernandes, L. Novel strategies for the treatment of osteoarthritis. Expert Opin. Investig. Drugs 2000, 9, 1499–1510. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Sanchez, M.; Orive, G.; Padilla, S.A. Biological therapy to osteoarthritis treatment using platelet-rich plasma. Expert Opin. Biol. Ther. 2013, 13, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Wise, B.L.; Niu, J.; Felson, D.T.; Hietpas, J.; Sadosky, A.; Torner, J.; Lewis, C.E.; Nevitt, M. Functional impairment is a risk factor for knee replacement in the multicenter osteoarthritis study. Clin. Orthop. Relat. Res. 2015, 473, 2505–2513. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.A.; Erickson, B.J.; Saltzman, B.M.; Mascarenhas, R.; Bach, B.R.; Cole, B.J.; Verma, N.N. Is Local Viscosupplementation Injection clinically superior to other therapies in the treatment of osteoarthritis of the knee: A systematic review of overlapping meta-analyses. Arthrosc. J. Arthrosc. Relat. Surg. 2015, 31, 2036–2045. [Google Scholar] [CrossRef] [PubMed]
- Sansone, V.; de Girolamo, L.; Pascale, W.; Melato, M.; Pascale, V. Long-term results of abrasion arthroplasty for full-thickness cartilage lesions of the medial femoral condyle. Arthrosc. J. Arthrosc. Relat. Surg. 2015, 31, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Solheim, E.; Hegna, J.; Oyen, J.; Harlem, T.; Strand, T. Results at 10 to 14 years after osteochondral autografting (mosaicplasty) in articular cartilage defects in the knee. Knee 2013, 20, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Valderrabano, V.; Leumann, A.; Rasch, H.; Egelhof, T.; Hintermann, B.; Pagenstert, G. Knee-to-ankle mosaicplasty for the treatment of osteochondral lesions of the ankle joint. Am. J. Sports Med. 2009, 37, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Levy, Y.D.; Gortz, S.; Pulido, P.A.; McCauley, J.C.; Bugbee, W.D. Do fresh osteochondral allografts successfully treat femoral condyle lesions? Clin. Orthop. Relat. Res. 2013, 471, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Shaha, J.S.; Cook, J.B.; Rowles, D.J.; Bottoni, C.R.; Shaha, S.H.; Tokish, J.M. Return to an athletic lifestyle after osteochondral allograft transplantation of the knee. Am. J. Sports Med. 2013, 41, 2083–2089. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.K.; Amiel, D.; Ball, S.T.; Allen, R.T.; Wong, V.W.; Chen, A.C.; Sah, R.L.; Bugbee, W.D. Prolonged storage effects on the articular cartilage of fresh human osteochondral allografts. J. Bone Joint Surg. Am. 2003, 85, 2111–2120. [Google Scholar] [PubMed]
- Luyten, F.P.; Vanlauwe, J. Tissue engineering approaches for osteoarthritis. Bone 2012, 51, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, A.M.; Hoque, M.E.; Prasad, R.G.; Uth, N. Current strategies in multiphasic scaffold design for osteochondral tissue engineering: A review. J. Biomed. Mater. Res. A 2015, 103, 2460–2481. [Google Scholar] [CrossRef] [PubMed]
- Makris, E.A.; Gomoll, A.H.; Malizos, K.N.; Hu, J.C.; Athanasiou, K.A. Repair and tissue engineering techniques for articular cartilage. Nat. Rev. Rheumatol. 2015, 11, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Ruszymah, B.H.; Lokman, B.S.; Asma, A.; Munirah, S.; Chua, K.; Mazlyzam, A.L.; Isa, M.R.; Fuzina, N.H.; Aminuddin, B.S. Pediatric auricular chondrocytes gene expression analysis in monolayer culture and engineered elastic cartilage. Int. J. Pediatr. Otorhinolaryngol. 2007, 71, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, S.; Raines, R.T. Review collagen-based biomaterials for wound healing. Biopolymers 2014, 101, 821–833. [Google Scholar] [CrossRef] [PubMed]
- Barsotti, M.C.; Felice, F.; Balbarini, A.; di Stefano, R. Fibrin as a scaffold for cardiac tissue engineering. Biotechnol. Appl. Biochem. 2011, 58, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Christensen, B.B.; Foldager, C.B.; Jensen, J.; Jensen, N.C.; Lind, M. Poor osteochondral repair by a biomimetic collagen scaffold: 1- to 3-year clinical and radiological follow-up. Knee Surg. Sports Traumatol. Arthrosc. 2015, in press. [Google Scholar] [CrossRef] [PubMed]
- Mushahary, D.; Wen, C.; Kumar, J.M.; Lin, J.; Harishankar, N.; Hodgson, P.; Pande, G.; Li, Y. Collagen type-I leads to in vivo matrix mineralization and secondary stabilization of Mg-Zr-Ca alloy implants. Colloids Surf. B Biointerfaces 2014, 122, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Hindle, P.; Hall, A.C.; Biant, L.C. Viability of chondrocytes seeded onto a collagen I/III membrane for matrix-induced autologous chondrocyte implantation. J. Orthop. Res. 2014, 32, 1495–1502. [Google Scholar] [CrossRef] [PubMed]
- Levengood, S.L.; Zhang, M. Chitosan-based scaffolds for bone tissue engineering. J. Mater. Chem. B Mater. Biol. Med. 2014, 2, 3161–3184. [Google Scholar] [CrossRef] [PubMed]
- Whu, S.W.; Hung, K.C.; Hsieh, K.H.; Chen, C.H.; Tsai, C.L.; Hsu, S.H. In vitro and in vivo evaluation of chitosan-gelatin scaffolds for cartilage tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 2855–2863. [Google Scholar] [CrossRef] [PubMed]
- Jancár, J.; Slovíková, A.; Amler, E.; Krupa, P.; Kecová, H.; Plánka, L.; Gál, P.; Necas, A. Mechanical response of porous scaffolds for cartilage engineering. Physiol. Res. 2007, 56, 17–25. [Google Scholar] [PubMed]
- Bhuiyan, D.; Jablonsky, M.J.; Kolesov, I.; Middleton, J.; Wick, T.M.; Tannenbaum, R. Novel synthesis and characterization of a collagen-based biopolymer initiated by hydroxyapatite nanoparticles. Acta Biomater. 2015, 15, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Petrigliano, F.A.; Arom, G.A.; Nazemi, A.N.; Yeranosian, M.G.; Wu, B.M.; McAllister, D.R. In vivo evaluation of electrospun polycaprolactone graft for anterior cruciate ligament engineering. Tissue Eng. Part A 2015, 21, 1228–1236. [Google Scholar] [CrossRef] [PubMed]
- Luitaud, C.; Laflamme, C.; Semlali, A.; Saidi, S.; Grenier, G.; Zakrzewski, A.; Rouabhia, M. Development of an engineering autologous palatal mucosa-like tissue for potential clinical applications. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 83, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Rouabhia, M.; Allaire, P. Gingival mucosa regeneration in athymic mice using in vitro engineered human oral mucosa. Biomaterials 2010, 31, 5798–5804. [Google Scholar] [CrossRef] [PubMed]
- Mostefaoui, Y.; Claveau, I.; Ross, G.; Rouabhia, M. Tissue structure, and IL-1beta, IL-8, and TNF-alpha secretions after contact by engineered human oral mucosa with dentifrices. J. Clin. Periodontol. 2002, 29, 1035–1041. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Jain, A.; Bansal, R. Applications of regenerative medicine in organ transplantation. J. Pharm. Bioallied. Sci. 2015, 7, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.D.; Grande, D.A. The current state of scaffolds for musculoskeletal regenerative applications. Nat. Rev. Rheumatol. 2015, 11, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Nehrer, S.; Chiari, C.; Domayer, S.; Barkay, H.; Yayon, A. Results of chondrocyte implantation with a fibrin-hyaluronan matrix: A preliminary study. Clin. Orthop. Relat. Res. 2008, 466, 1849–1855. [Google Scholar] [CrossRef] [PubMed]
- Romanova, O.A.; Grigor’ev, T.E.; Goncharov, M.E.; Rudyak, S.G.; Solov’yova, E.V.; Krasheninnikov, S.T.; Saprykin, V.P.; Sytina, E.V.; Chvalun, S.N.; Pal’tsev, M.A.; et al. Chitosan as a modifying component of artificial scaffold for human skin tissue engineering. Bull. Exp. Biol. Med. 2015, 159, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.; Blanco, M.D.; Davidenko, N.; Cameron, R.E. Tailoring chitosan/collagen scaffolds for tissue engineering: Effect of composition and different crosslinking agents on scaffold properties. Carbohydr. Polym. 2015, 5, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Staroszczyk, H.; Sztuka, K.; Wolska, J.; Wojtasz-Pająk, A.; Kołodziejska, I. Interactions of fish gelatin and chitosan in uncrosslinked and crosslinked with EDC films: FT-IR study. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 117, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Pielesz, A. Temperature-dependent FTIR spectra of collagen and protective effect of partially hydrolysed fucoidan. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 118, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.D.; Farrugia, B.L.; Dargaville, T.R.; Dhara, S. Chitosan-collagen scaffolds with nano/microfibrous architecture for skin tissue engineering. J. Biomed. Mater. Res. A 2013, 101, 3482–3492. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhu, C.; Fan, D.; Liu, B.; Ma, X.; Duan, Z.; Zhou, Y. A human-like collagen/chitosan electrospun nanofibrous scaffold from aqueous solution: Electrospun mechanism and biocompatibility. J. Biomed. Mater. Res. A 2011, 99, 395–409. [Google Scholar] [CrossRef] [PubMed]
- Geiger, M.; Li, R.H.; Friess, W. Collagen sponges for bone regeneration with rhBMP-2. Adv. Drug Deliv. Rev. 2003, 55, 1613–1629. [Google Scholar] [CrossRef] [PubMed]
- Tierney, C.M.; Haugh, M.G.; Liedl, J.; Mulcahy, F.; Hayes, B.; O’Brien, F.J. The effects of collagen concentration and crosslink density on the biological; structural and mechanical properties of collagen-GAG scaffolds for bone tissue engineering. J. Mech. Behav. Biomed. Mater. 2009, 2, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Yeung, T.; Georges, P.C.; Flanagan, L.A.; Marg, B.; Ortiz, M.; Funaki, M. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil. Cytoskelet. 2005, 60, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Awang, M.A.; Firdaus, M.A.; Busra, M.B.; Chowdhury, S.R.; Fadilah, N.R.; Wan Hamirul, W.K.; Reusmaazran, M.Y.; Aminuddin, M.Y.; Ruszymah, B.H. Cytotoxic evaluation of biomechanically improved crosslinked ovine collagen on human dermal fibroblasts. Biomed. Mater. Eng. 2014, 24, 1715–1724. [Google Scholar] [PubMed]
- Debnath, T.; Ghosh, S.; Potlapuvu, U.S.; Kona, L.; Kamaraju, S.R.; Sarkar, S.; Gaddam, S.; Chelluri, L.K. Proliferation and differentiation potential of human adipose-derived stem cells grown on chitosan hydrogel. PLoS ONE 2015, 10, e0120803. [Google Scholar] [CrossRef] [PubMed]
- Conti, B.; Modena, T.; Genta, I.; Perugini, P.; Pavanetto, F. A proposed new method for the crosslinking of chitosan microspheres. Drug Deliv. 1998, 5, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.W.; Caves, J.M.; Ravi, S.; Li, W.; Chaikof, E.L. Effects of crosslinking on the mechanical properties, drug release and cytocompatibility of protein polymers. Acta Biomater. 2014, 10, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Bianchera, A.; Salomi, E.; Pezzanera, M.; Ruwet, E.; Bettini, R.; Elviri, L. Chitosan hydrogels for chondroitin sulphate controlled release: An analytical characterization. J. Anal. Methods Chem. 2014, 808703. [Google Scholar] [CrossRef] [PubMed]
- Rajam, A.M.; Jithendral, P.; Mandal, A.B.; Rose, C. Evaluation of in vitro macrophage response and in vivo host response to growth factors incorporated chitosan nanoparticle impregnated collagen-chitosan scaffold. J. Biomed. Nanotechnol. 2014, 10, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, C.; Moritz, S.; Hessler, N.; Kralisch, D.; Wesarg, F.; Müller, F.A.; Fischer, D.; Hipler, U.C. Antimicrobial functionalization of bacterial nanocellulose by loading with polihexanide and povidone-iodine. J. Mater. Sci. Mater. Med. 2015, 26, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, S.C.; Sadocco, P.; Alonso-Varona, A.; Palomares, T.; Eceiza, A.; Silvestre, A.J.; Mondragon, I.; Freire, C.S. Bioinspired antimicrobial and biocompatible bacterial cellulosemembranes obtained by surface functionalization with aminoalkylgroups. ACS Appl. Mater. Interfaces 2013, 5, 3290–3297. [Google Scholar] [CrossRef] [PubMed]
- Rouabhia, M.; Asselin, J.; Tazi, N.; Messaddeq, Y.; Levinson, D.; Zhang, Z. Production of biocompatible and antimicrobial bacterial cellulosepolymers functionalized by RGDC grafting groups and gentamicin. ACS Appl. Mater. Interfaces 2014, 6, 1439–1446. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Fan, D.; Duan, Z.; Xue, W.; Shang, L.; Chen, F.; Luo, Y. Initial investigation of novel human-like collagen/chitosan scaffold for vascular tissue engineering. J. Biomed. Mater. Res. A 2009, 89, 829–840. [Google Scholar] [CrossRef] [PubMed]
- Aryaei, A.; Liu, J.; Jayatissa, A.H.; Champa Jayasuriya, A. Cross-linked chitosan improves the mechanical properties of calcium phosphate-chitosan cement. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 54, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Ma, L.; Zhou, J.; Mao, Z.; Gao, C. Collagen/chitosan-silicone membrane bilayer scaffold as a dermal equivalent. Polym. Adv. Technol. 2005, 16, 789–794. [Google Scholar] [CrossRef]
- Ti, D.; Hao, H.; Xia, L.; Tong, C.; Liu, J.; Dong, L.; Xu, S.; Zhao, Y.; Liu, H.; Fu, X.; Han, W. Controlled release of thymosin beta 4 using a collagen-chitosan sponge scaffold augments cutaneous wound healing and increases angiogenesis in diabetic rats with hindlimb ischemia. Tissue Eng. Part A 2015, 21, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xie, X.D.; Huang, X.; Liang, Z.H.; Zhou, C.R. A quantitative study of MC3T3-E1 cell adhesion; morphology and biomechanics on chitosan-collagen blend films at single cell level. Colloids Surf. B Biointerfaces 2015, 132, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ghorbani, F.M.; Kaffashi, B.; Shokrollahi, P.; Seyedjafari, E.; Ardeshirylajimi, A. PCL/chitosan/Zn-doped nHA electrospun nanocomposite scaffold promotes adipose derived stem cells adhesion and proliferation. Carbohydr. Polym. 2015, 118, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Irie, Y.; Nishimura, S.I. Endothelial cell responses to chitin and its derivatives. J. Biomed. Mater. Res. 1998, 43, 469–472. [Google Scholar] [CrossRef]
- Chicatun, F.; Pedraza, C.E.; Ghezzi, C.E.; Marelli, B.; Kaartinen, M.T.; McKee, M.D.; Nazhat, S.N. Osteoid-mimicking dense collagen/chitosan hybrid gels. Biomacromolecules 2011, 12, 2946–2956. [Google Scholar] [CrossRef] [PubMed]
- Ragetly, G.; Griffon, D.J.; Chung, Y.S. Effect of Type II collagen coating of chitosan fibrous scaffolds on mesenchymal stem cell adhesion and chondrogenesis. Acta Biomater. 2010, 6, 3988–3997. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhang, L.; Wang, C. Effect of silanization on chitosan porous scaffolds for peripheral nerve regeneration. Carbohydr. Polym. 2014, 101, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Lahiji, A.; Sohrabi, A.; Hungerford, D.S.; Frondoza, C.G. Chitosan supports expression of extracellular matrix proteins in human osteoblasts and chondrocytes. J. Biomed. Mater. Res. 2000, 51, 586–595. [Google Scholar] [CrossRef]
- Aigner, T.; Soeder, S.; Haag, J. IL-1beta and BMPs-interactive players of cartilage matrix degradation and regeneration. Eur. Cell Mater. 2006, 12, 49–56. [Google Scholar] [PubMed]
- Fernandes, J.C.; Martel-Pelletier, J.; Pelletier, J.P. The role of cytokines in osteoarthritis pathophysiology. Biorheology 2002, 39, 237–246. [Google Scholar] [PubMed]
- Kondo, M.; Yamaoka, K.; Sakata, K.; Sonomoto, K.; Lin, L.; Nakano, K.; Tanaka, Y. Contribution of the interleukin-6/STAT-3 signaling pathway to chondrogenic differentiation of human mesenchymal stem cells. Arthritis Rheumatol. 2015, 67, 1250–1260. [Google Scholar] [CrossRef] [PubMed]
- Mathews, S.; Gupta, P.K.; Bhonde, R.; Totey, S. Chitosan enhances mineralization during osteoblast differentiation of human bone marrow-derived mesenchymal stem cells; by upregulating the associated genes. Cell Prolif. 2011, 44, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Barnes, T.C.; Anderson, M.E.; Moots, R.J. The many faces of interleukin-6: The role of IL-6 in inflammation; vasculopathy; and fibrosis in systemic sclerosis. Int. J. Rheumatol. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Yagi, Y.; Shioya, M.; Nishida, A.; Tsujikawa, T.; Fujiyama, Y. Mucosal cytokine network in inflammatory bowel disease. World J. Gastroenterol. 2008, 14, 5154–5161. [Google Scholar] [CrossRef] [PubMed]
- Kon, E.; Roffi, A.; Filardo, G.; Tesei, G.; Marcacci, M. Scaffold-based cartilage treatments: With or without cells? A systematic review of preclinical and clinical evidence. Arthrosc. J. Arthrosc. Relat. Surg. 2015, 31, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Dhollander, A.A.; Liekens, K.; Almqvist, K.F.; Verdonk, R.; Lambrecht, S.; Elewaut, D.; Verbruggen, G.; Verdonk, P.C. A pilot study of the use of an osteochondral scaffold plug for cartilage repair in the knee and how to deal with early clinical failures. Arthrosc. J. Arthrosc. Relat. Surg. 2012, 28, 225–233. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mighri, N.; Mao, J.; Mighri, F.; Ajji, A.; Rouabhia, M. Chitosan-Coated Collagen Membranes Promote Chondrocyte Adhesion, Growth, and Interleukin-6 Secretion. Materials 2015, 8, 7673-7689. https://doi.org/10.3390/ma8115413
Mighri N, Mao J, Mighri F, Ajji A, Rouabhia M. Chitosan-Coated Collagen Membranes Promote Chondrocyte Adhesion, Growth, and Interleukin-6 Secretion. Materials. 2015; 8(11):7673-7689. https://doi.org/10.3390/ma8115413
Chicago/Turabian StyleMighri, Nabila, Jifu Mao, Frej Mighri, Abdallah Ajji, and Mahmoud Rouabhia. 2015. "Chitosan-Coated Collagen Membranes Promote Chondrocyte Adhesion, Growth, and Interleukin-6 Secretion" Materials 8, no. 11: 7673-7689. https://doi.org/10.3390/ma8115413
APA StyleMighri, N., Mao, J., Mighri, F., Ajji, A., & Rouabhia, M. (2015). Chitosan-Coated Collagen Membranes Promote Chondrocyte Adhesion, Growth, and Interleukin-6 Secretion. Materials, 8(11), 7673-7689. https://doi.org/10.3390/ma8115413




