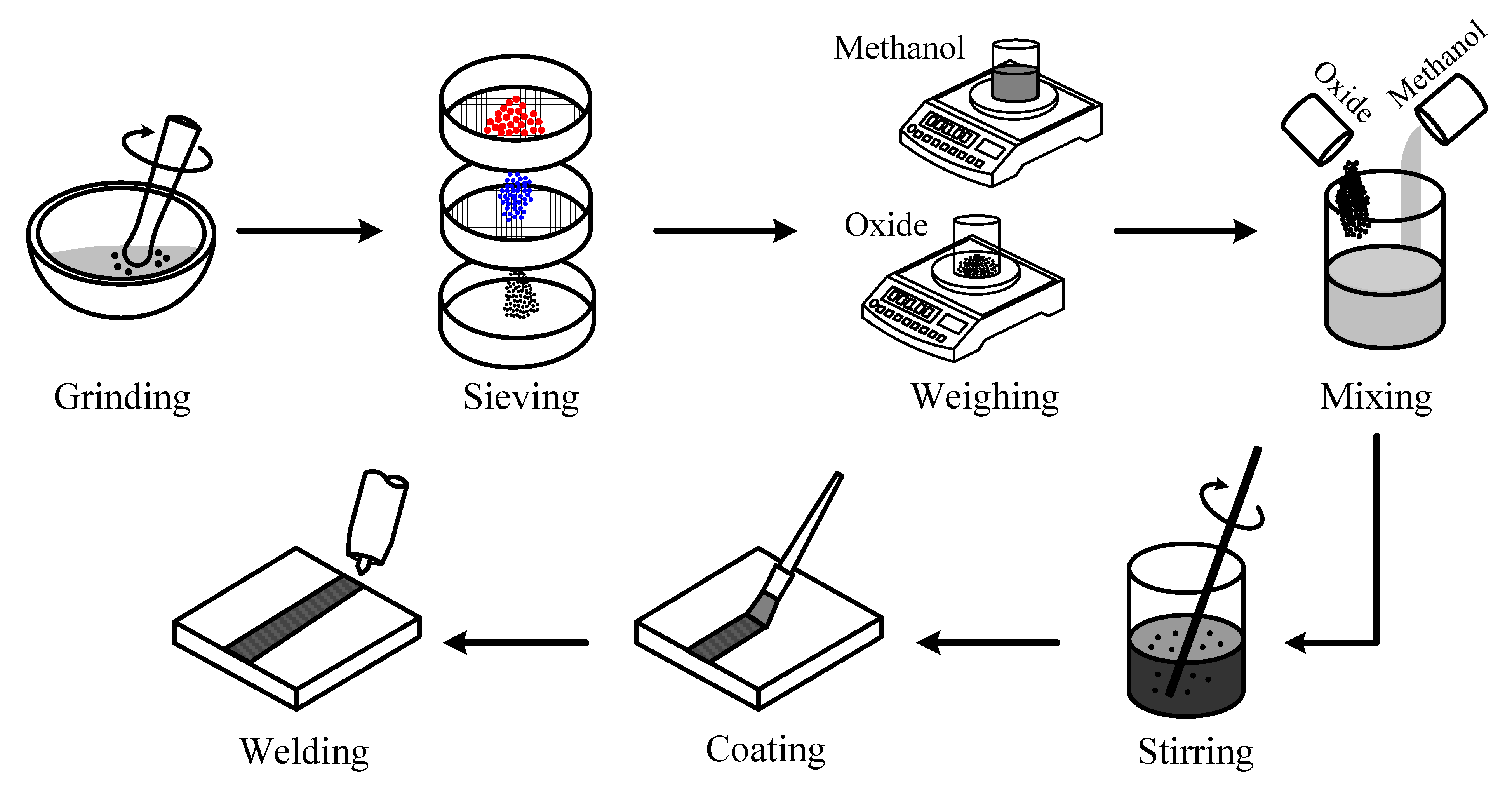
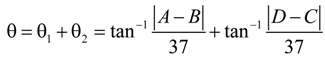The purpose of this study was to demonstrate that the surface appearance, geometric shape, angular distortion and metallurgical properties of the UNS S31603 stainless steel A-TIG welds greatly depend on the thermal stability and particle size of the activated oxides. In addition, the influence of polar solvents on the coverability, spreadability and volatility of the flux-coated layer were investigated.
3.1. Evaluation of Activated Oxides Mixed with Polar Solvents
It is necessary to choose a suitable solvent resulting in a homogeneous distribution of the flux-coated layer for A-TIG welding. This is because the A-TIG weld appearance becomes irregular when there is an inconsistent quantity of the flux-coated layer. In this study, water, methanol and acetone were used as carrier solvents. The differences in weight concentration for microparticle and nanoparticle fluxes were used in the trial after specific surface areas were considered. Although the weight concentrations of the fluxes are different, the quantity of the flux-coated layer is near constant. In this study, 500 mg of microparticle oxide was mixed with 0.75 mL of solvent, while 250 mg of nanoparticle oxide was mixed with 4.5 mL of solvent to form a paint-like consistency. The results in
Figure 3 clearly show that SiO
2 and Al
2O
3 mixed with water or methanol provides a homogeneous distribution of the coated layer, while acetone is the worst solvent in terms of coated layer consistency. Selecting a carrier solvent involves the evaluation of many factors. The two most important factors are the coatability and the volatility of the flux-coated layer. The coatability can be further characterized by the spreadability and coverability.
Table 3 lists the physical properties of water, methanol and acetone [
16]. The dielectric constant and dynamic viscosity of water and methanol are substantially higher than those of acetone. SiO
2 and Al
2O
3 mixed with water or methanol and then applied to a steel plate surface provide good spreadability and coverability of the coated layer. In A-TIG welding, the evaporation rate of the carrier solvent also plays an important role, because the solvent needs to leave the flux at an ideal rate to obtain a dry coated layer.
Table 4 shows the volatile time of the activated fluxes. The results show that water takes the longest time to evaporate from the flux-coated layer, whereas acetone has an evaporation time shorter than that of methanol. Compared with methanol and acetone, water has the highest boiling point and the lowest vapor pressure, leading to poor volatility after flux coating. For powdered SiO
2 and Al
2O
3, methanol is the preferred solvent in an activated flux, resulting in a good coatability, volatility of the coated layer. Compared with microparticle SiO
2 and Al
2O
3, nanoparticle SiO
2 and Al
2O
3 mixed with water or methanol provides good flux coverability of the coated layer. However, this is accompanied by poor flux spreadability of the layer, because the nanoparticles have a strong tendency to cluster, forming an agglomerate state [
17].
Figure 3.
Evaluation of activated oxides mixed with polar solvents.
Figure 3.
Evaluation of activated oxides mixed with polar solvents.
Table 3.
Physical properties of polar solvents used in this study.
Table 3.
Physical properties of polar solvents used in this study.
| Properties | Polar Solvent |
|---|
| Water | Methanol | Acetone |
|---|
| Dielectric constant | 80.1 | 33.0 | 21.0 |
| Dynamic viscosity (mPa·s) | 0.89 | 0.54 | 0.31 |
| Boiling point (°C) | 100.0 | 64.6 | 56.1 |
| Vapor pressure (kPa) | 3.2 | 16.9 | 30.8 |
Table 4.
Evaporation time of activated fluxes (at an operating temperature of 25 °C).
Table 4.
Evaporation time of activated fluxes (at an operating temperature of 25 °C).
| Activated Oxide | Polar Solvent |
|---|
| Water | Methanol | Acetone |
|---|
| Microparticle SiO2 | 31.78 min | 1.72 min | 0.45 min |
| Nanoparticle SiO2 | 61.13 min | 3.13 min | 1.28 min |
| Microparticle Al2O3 | 24.22 min | 1.30 min | 0.33 min |
| Nanoparticle Al2O3 | 41.35 min | 2.12 min | 0.87 min |
3.2. Effect of Microparticle or Nanoparticle Oxide on Surface Appearance of A-TIG Weld
A-TIG welding has the potential to substantially increase productivity [
18]. However, industry has been slow to exploit the benefits from the increased joint penetration, partially because A-TIG welding tends to produce excessive flux slag and a rough surface compared with C-TIG welding. The flux forms slag that is less dense than the molten metal, causing the slag to float up to the weld surface. The surface appearance of the weld will be a special consideration for some manufactured products. Producing a clean, smooth weld surface is thus a key competitive challenge for the adoption of A-TIG welding in industrial applications. In this study, microparticle oxide was mixed with methanol at a weight ratio of 1:1.2; nanoparticle oxide was mixed with methanol at a weight ratio of 1:14.3.
Figure 4 shows the surface appearance of the TIG welds made with and without oxides.
Figure 4a shows the results of C-TIG welding, which produced the clean, smooth surface of the weld.
Figure 4b shows that the microparticle SiO
2 produced little slag (20 ± 2% surface coverage of a weld). Similarly,
Figure 4c shows that the nanoparticle SiO
2 produced little slag (10 ± 1% surface coverage of a weld). As presented in
Figure 4d, a large amount of slag (40 ± 3% surface coverage) was produced when using microparticle Al
2O
3. In contrast, a small amount of slag (25 ± 2% surface coverage) was produced when using nanoparticle Al
2O
3 (
Figure 4e). Compared with the surface appearance of the TIG weld made with microparticle or nanoparticle SiO
2, the surface of the TIG weld made with microparticle or nanoparticle Al
2O
3 showed more particles of unmelted powder remaining on both of its sides. The melting point of SiO
2 (1723 °C) is lower than that of Al
2O
3 (2100 °C) [
19]. The SiO
2 could thus have been easily melted by the TIG arc heat. A bulk material has a fixed melting point regardless of its size. However, small particles exhibit a melting point dependent on the particle size [
20]. The melting point of nanoparticles is always lower than that of the corresponding microparticles, because ultrafine particles have a high proportion of surface atoms [
21]. Thus, the nanoparticle oxides could have been melted by the welding arc heat more easily. The surface appearance of the TIG weld made with nanoparticle oxide has less flux slag compared with the one made with microparticle oxide of the same type.
3.3. Effect of Microparticle or Nanoparticle Oxide on Geometric Shape of A-TIG Weld
Various thermal stabilities and particle sizes of the activated flux have different effects on the fluid flow and heat transfer in the molten pool. These effects will greatly affect the shape and size of the weld.
Figure 5 shows the weld shape and arc plasma of the TIG welding with and without oxides. The photograph is a combination of metallographic images (weld part) and CCD camera images (arc part).
Figure 5a shows the result of C-TIG welding, which typically exhibits a wide, shallow weld.
Figure 5b shows that use of the microparticle SiO
2 results in a narrow, partial penetration weld.
Figure 5c shows that the use of the nanoparticle SiO
2 results in a narrow, complete penetration weld. As presented in
Figure 5d,e, a wide, shallow weld was produced when using microparticle or nanoparticle Al
2O
3. The geometric shape of the TIG weld is strongly characterized by the weld depth and bead width.
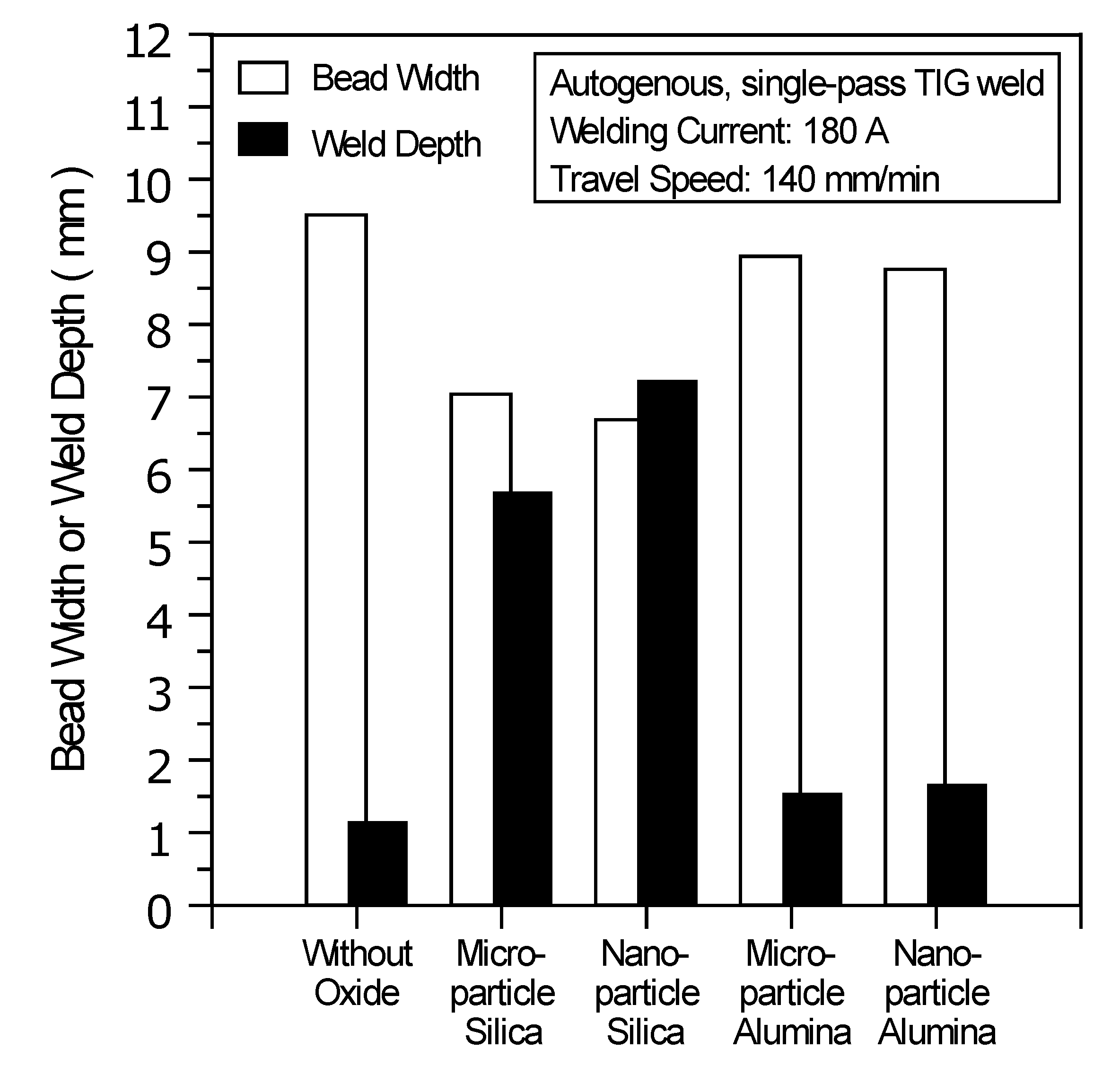
Figure 6 shows the geometric size of the TIG welds made with and without oxides. Compared with the C-TIG weld, there is an increase in weld depth and a decrease in bead width resulting from the use of microparticle or nanoparticle SiO
2; however, the use of microparticle or nanoparticle Al
2O
3 results in no significant difference in weld depth and bead width.
Figure 4.
Surface appearance of TIG welds made with and without oxides.
Figure 4.
Surface appearance of TIG welds made with and without oxides.
Figure 5.
Weld shape and arc plasma of TIG welding with and without oxides.
Figure 5.
Weld shape and arc plasma of TIG welding with and without oxides.
Figure 6.
Geometric size of TIG welds made with and without oxides.
Figure 6.
Geometric size of TIG welds made with and without oxides.
Tseng
et al. [
4,
5,
6,
7,
8,
10] reported experimental observations of an interactive mechanism between the arc column and the molten pool in A-TIG welding. Determining the joint penetration capability of the A-TIG weld can be conditionally subdivided into the mechanisms that take place in the arc column and the molten pool. In A-TIG welding, the arc column is characterized by the constricted arc plasma and the molten pool is characterized by the centripetal Marangoni convection. The mechanisms occurring in the arc column and the molten pool are interrelated and should be regarded as a unified system.
According to work by Lucas and Howse [
22], the vaporized flux constricts the arc plasma by capturing electrons in the outer regions of the arc column. The attachment of electrons to vaporized molecules and dissociated atoms affect electron absorption, thereby forming negatively charged particles. As a result, restricting the current flow to the central region of the arc column increases the current density of the arc plasma and raises the arc temperature at the molten pool surface (anode spot). A-TIG welding provides a greater number of vaporized molecules and dissociated atoms in the outer regions of the arc column, and constriction of the arc plasma occurs as the number of electrons in the outer regions of the arc column significantly decreases. The reduction in the number of electrons is dependent on the electron affinity of an atom or molecule (defined as the change in energy of a neutral atom or molecule in the gaseous phase when an electron is added to the atom or molecule to form a negatively charged ion) in the outer regions of the arc column.
According to work by Heiple
et al. [
23,
24,
25], fluid flow in the molten pool determines the fusion zone geometry, and the dominant force that drives fluid flow is the temperature coefficient of surface tension (dγ/d
T). If dγ/d
T is non-zero in the molten pool, then fluid flows from regions of lower surface tension to regions of higher surface tension. Furthermore, dγ/d
T is dependent on the oxygen content in the molten pool. In the TIG welding without activated oxide, dγ/d
T in the molten pool generally has a negative value, resulting in centrifugal Marangoni convection. In the TIG welding with activated oxide, a certain amount of oxygen from the decomposition of activated oxide induces the centripetal Marangoni convection by inverting the dγ/d
T in the molten pool. Lu
et al. [
26] showed that when the oxygen content in the stainless steel TIG weld metal was in the range of 70–300 ppm, the value of dγ/d
T in the molten pool was positive; otherwise, the value of dγ/d
T was negative.
In the TIG welding with SiO
2, the number of electrons in the outer regions of the arc column may be reduced, because the Si molecule has a higher electron affinity (1.385 ± 0.005 eV [
27]), and thus, the arc plasma is constricted (
Figure 5b,c). The constricted arc plasma increases the current density of the arc plasma and generates a high arc heat at the anode hotspot. The resulting large Lorentz force contributes a forceful downward flow of arc heat within the molten pool. Moreover, according to the Ellingham diagram, SiO
2 has a lower thermal stability than Al
2O
3.
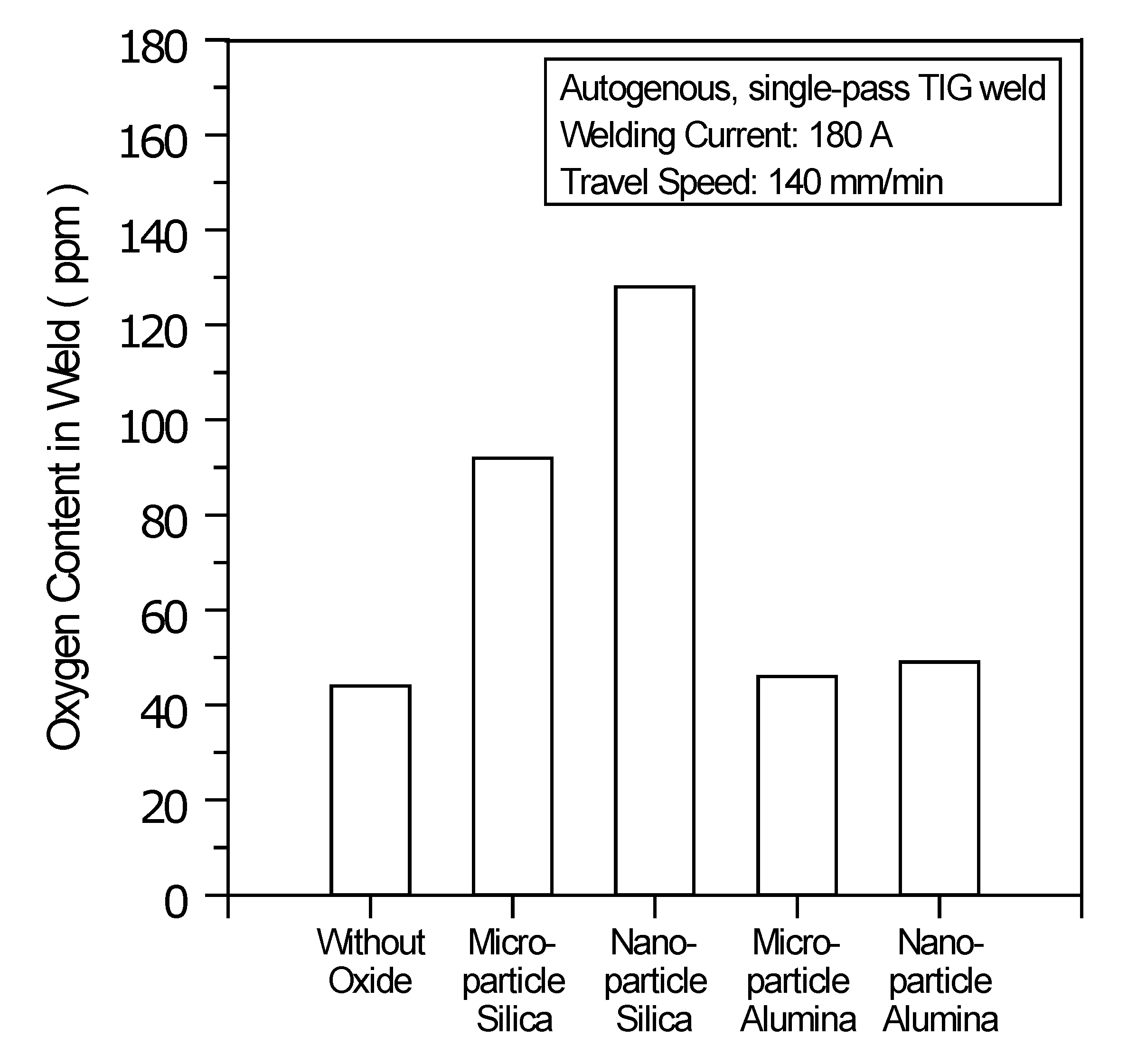
Figure 7 shows the oxygen content in the UNS S31603 stainless steel TIG welds made with and without oxides. The results show that the oxygen content in the stainless steel TIG weld metal made with microparticle SiO
2 is 92 ppm, resulting in a sufficient oxygen content (> 70 ppm) for the molten metal, thus causing the Marangoni convection to reverse and move inward along the molten pool surface. The centripetal Marangoni convection further promotes the energy transfer of the arc heat from the surface to the bottom of the molten pool, resulting in a narrow, deep weld. Compared with microparticle SiO
2, nanoparticle SiO
2 has greater efficiency in improving the joint penetration capability of the A-TIG weld. In TIG arc heating and vaporization, the thermal dissociation and decomposition of nanoparticle SiO
2 may occur much more readily than do the thermal dissociation and decomposition of microparticle SiO
2. This results in the maximum capability of the joint penetration in the A-TIG weld. The TIG welding with nanoparticle SiO
2 can result in a 524% increase in weld depth relative to the depth of the C-TIG weld.
Figure 7.
Oxygen content in TIG welds made with and without oxides.
Figure 7.
Oxygen content in TIG welds made with and without oxides.
Under the same welding conditions, the TIG welding with Al
2O
3 appears to have little effect on the arc column and the molten pool. In the TIG welding with Al
2O
3, the number of electrons in the outer regions of the arc column cannot be significantly reduced, because the Al molecule has a lower electron affinity (0.442 ± 0.010 eV [
27]), and thus, the arc plasma cannot be constricted (
Figure 5d,e). In addition, Al
2O
3 exhibits high thermal stability and possesses greater resistance to decomposition at high temperature. The oxygen content in the stainless steel TIG weld metal made with microparticle Al
2O
3 is 46 ppm, resulting in an insufficient oxygen content (< 70 ppm) in the molten metal and leaving the direction of Marangoni convection in the molten pool unchanged. Because Al
2O
3 could not make a positive contribution in constricting the arc plasma and inverting the Marangoni convection, The TIG welding with Al
2O
3 does not promote an increase in weld depth and a decrease in bead width. Moreover, the use of nanoparticle Al
2O
3 does not lead to a significant improvement in joint penetration capability compared with the capability achieved with microparticle Al
2O
3.
In arc welding, the power density of the welding heat source can be characterized by the D/W ratio, cross-sectional area and HAZ of the weld.
Table 5 shows the D/W ratio, cross-sectional area and HAZ of the TIG welds made with and without oxides. Compared with C-TIG weld, the TIG weld made with microparticle or nanoparticle SiO
2 leads to a high D/W ratio, large cross-sectional area and narrow HAZ; however, there is no significant improvement in the D/W ratio, cross-sectional area or HAZ resulting from the use of microparticle or nanoparticle Al
2O
3. A high D/W ratio, large cross-sectional area and narrow HAZ of the weld are characteristic of the increased power density of the heat source used in welding, which results in a high-energy density arc jet. Compared with microparticle SiO
2, nanoparticle SiO
2 produces a higher D/W ratio, larger cross-sectional area and a narrower HAZ of the A-TIG weld, resulting in a stronger, more forceful arc jet that has higher energy density. However, the use of nanoparticle Al
2O
3 does not lead to an enhancement of the energy density of TIG welding arc, unlike the use of nanoparticle SiO
2.
Table 5.
Depth-to-width (D/W) ratio, cross-sectional area and heat-affected zone (HAZ) of TIG welds made with and without oxides.
Table 5.
Depth-to-width (D/W) ratio, cross-sectional area and heat-affected zone (HAZ) of TIG welds made with and without oxides.
| Characteristics | Activated Oxide |
|---|
Without
oxide | Microparticle
SiO2 | Nanoparticle
SiO2 | Microparticle
Al2O3 | Nanoparticle
Al2O3 |
|---|
| D/W ratio | 0.12 | 0.81 | 1.08 | 0.17 | 0.19 |
| Cross-sectional area | 9.72 mm2 | 26.11 mm2 | 29.07 mm2 | 12.48 mm2 | 13.30 mm2 |
| HAZ | 1.72 mm | 0.85 mm | 0.60 mm | 1.51 mm | 1.42 mm |
3.4. Effect of Microparticle or Nanoparticle Oxide on Angular Distortion of A-TIG Weldment
Angular distortion is one of the most common types of out-of-plane deformations. Angular distortion often occurs in a butt joint when transverse shrinkage is not uniform in the thickness direction of the welded plate [
28,
29,
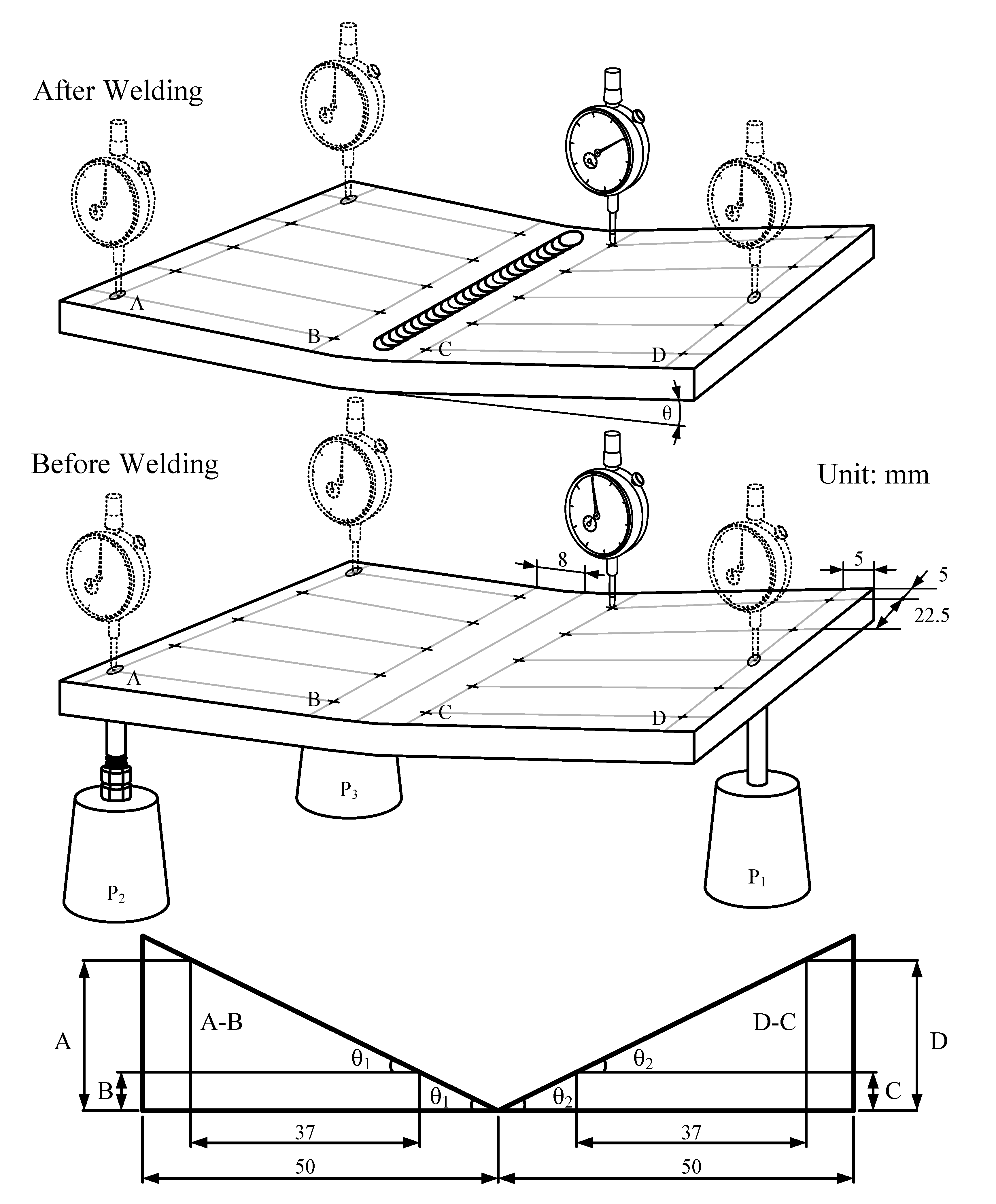
30]. Generally, the weld-induced distortion not only degrades the performance of the weldments, but also increases the rectification cost of the welded constructions. An investigation into the angular distortion of the A-TIG weldment is necessary in order to minimize weld-induced distortion. After the weldments were cooled to room temperature, all experiments were carried out to measure the angular distortion caused by welding.
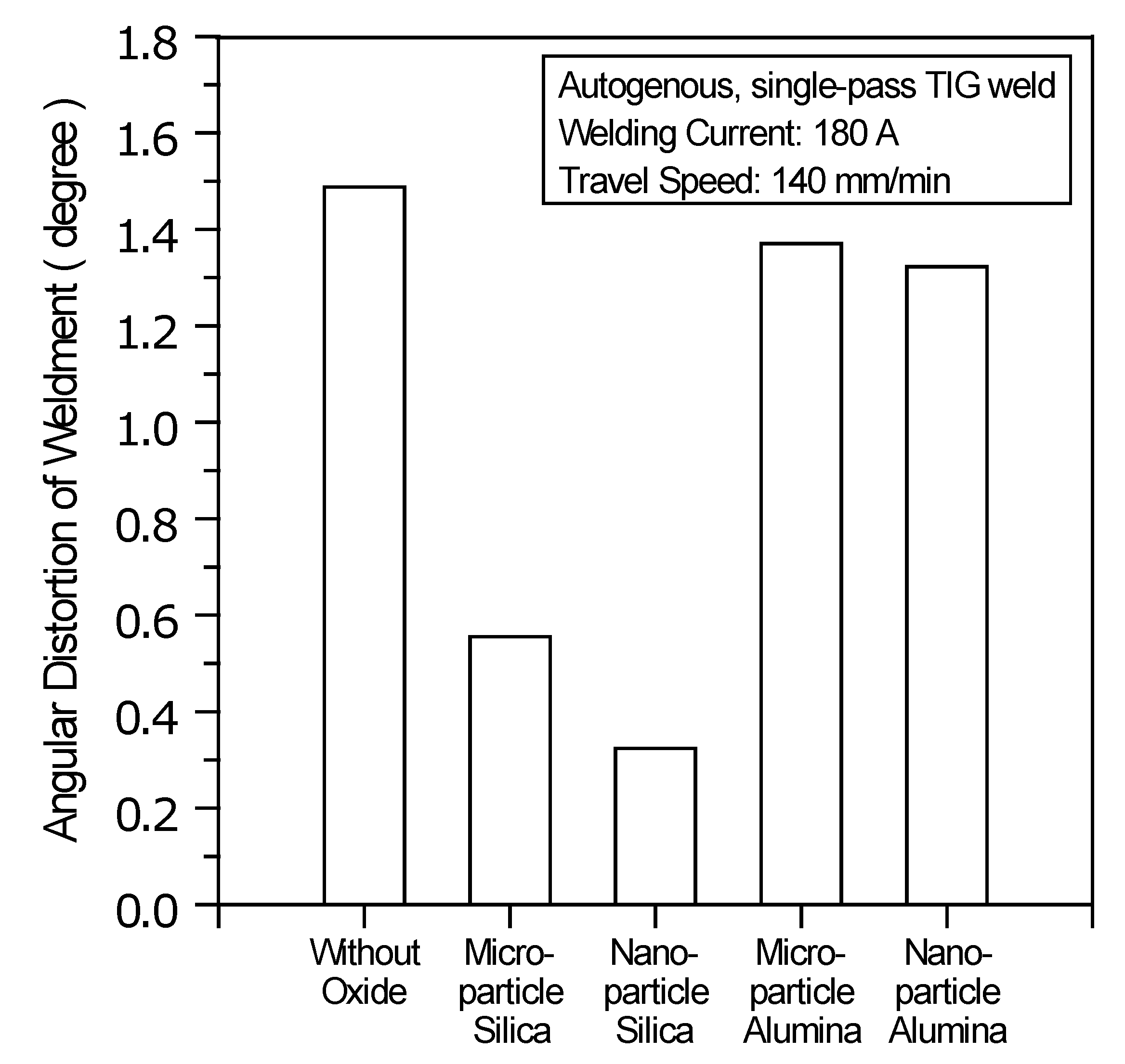
Figure 8 shows the angular distortion of the TIG weldments made with and without oxides. The results show a significant reduction in the angular distortion of the TIG weldment made with SiO
2. In contrast, the TIG weldment made with Al
2O
3 shows no significant reduction in angular distortion compared with the C-TIG weldment.
In the case of TIG welding, the energy generated by the heat source is distributed in two ways; a portion is lost to the environment, and the remainder is transferred to the base metal [
31]. The net energy of the arc heat delivered to the base metal is also distributed in two ways; a portion is used for fusion of the base metal, while the remainder is dissipated into the HAZ by heat conduction [
32]. Therefore, a relatively small portion (maximum value of 0.35) of the net energy of the arc heat is used for the fusion of the base metal [
31]. The arc heat dissipated into the HAZ causes a non-uniform thermal stress. Consequently, distortions are permanently produced in the weldment. A high power density of the welding heat source can significantly reduce arc heat loss to the HAZ, resulting in low thermal stresses acting on the weld metal and the adjacent HAZ.
3.5. Effect of Microparticle or Nanoparticle Oxide on Metallurgical Properties of A-TIG Weld Metal
Lippold and Savage indicated that the austenitic stainless steel weld metals, which exhibit delta ferrite content of 5–12 FN provided the best resistance to hot cracking [
33]. It is necessary to attain the desired levels of delta ferrite content in the UNS S31603 stainless steel A-TIG weld metal.
Figure 8.
Angular distortion of TIG weldments made with and without oxides.
Figure 8.
Angular distortion of TIG weldments made with and without oxides.
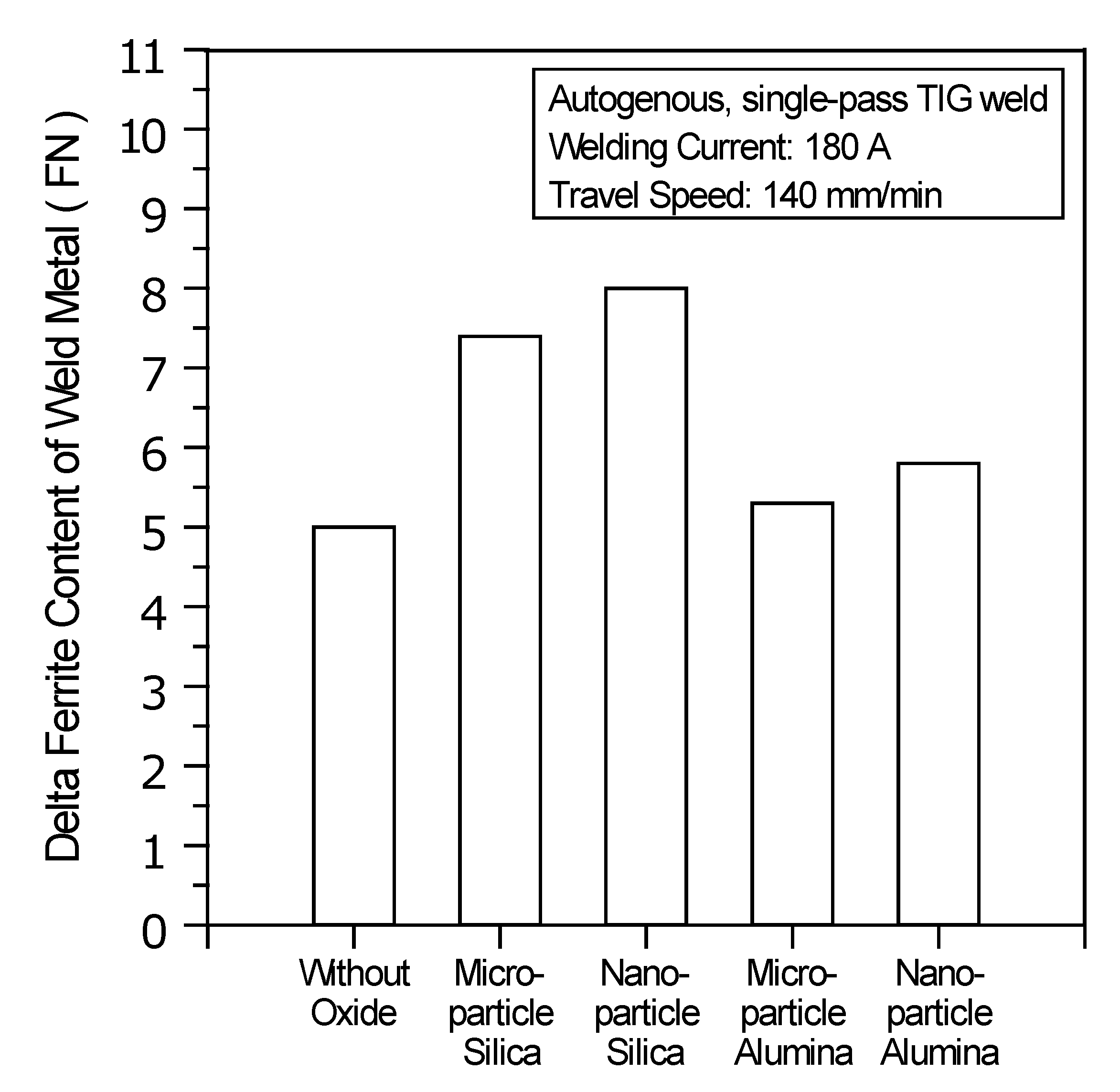
Figure 9 shows the delta ferrite content of the UNS S31603 stainless steel TIG weld metals made with and without oxides. The base metal used in this study was hot-rolled stainless steel having an ultra-low ferrite content of 0.4 FN. In the stainless steel C-TIG weld metal, the ferrite content was increased to 5.0 FN from its initial value of 0.4 FN. This is because the UNS S31603 stainless steel weld metal solidified in the ferritic-austenitic (FA) mode, which has delta ferrite as the primary phase. During welding, the cooling rate of the weld metal is so rapid, that phase transformation of the delta ferrite to the austenite is not complete, resulting in more delta ferrite being retained in the stainless steel weld metal after solidification.
The delta ferrite content of the UNS S31603 stainless steel TIG weld made with SiO2 is in the range of 7.4–8.0 FN; with the use of Al2O3, the ferrite content is in the range of 5.3–5.8 FN. The power density of the welding heat source directly affects the amount of arc heat supplied for the fusion of the base metal and affects the formation of the delta ferrite phase in the weld metal. An increase in the power density decreases the arc heat required for the fusion of the base metal, because it decreases the time required for using the heat source for fusing the base metal. Compared with C-TIG welding, the TIG welding with SiO2 uses a heat source with a higher power density, resulting in a higher ferrite content in the weld metal, as the transformation of delta ferrite to the austenite phase has less time to occur at a high cooling rate. However, the use of microparticle or nanoparticle Al2O3 does not lead to a significant increase in ferrite content, in contrast to welding without any oxide.
Figure 9.
Delta ferrite content of TIG weld metals made with and without oxides.
Figure 9.
Delta ferrite content of TIG weld metals made with and without oxides.
Figure 10 shows the hardness distribution of the TIG weldments made with and without oxides. The results show that the weld metal has the highest Vickers hardness, whereas the HAZ has the lowest Vickers hardness. In the UNS S31603 stainless steel TIG welding with or without oxide, higher hardness is achieved by forming microstructures that consist of the delta ferrite in the weld metal; lower hardness is associated with a relatively coarse austenite grain size in the HAZ. As previously mentioned, the TIG weld metal made with SiO
2 has a higher delta ferrite content, resulting in a higher Vickers hardness compared to that of the C-TIG weld metal or the TIG weld metal made with Al
2O
3. The results also show that the use of nanoparticle oxide does not lead to a significant increase in Vickers hardness compared with that achieved with microparticle oxide. Furthermore, the lower power density of the heat source for C-TIG welding or the TIG welding with Al
2O
3 requires a larger amount of arc heat for fusion of the base metal, resulting in a greater amount of arc heat being dissipated to the HAZ. The HAZ thus has a coarser austenite grain than that of the base metal, which tends to decrease the hardness of the HAZ. Compared with C-TIG welding or the TIG welding with Al
2O
3, the TIG welding assisted by SiO
2 uses a heat source with a higher power density. Therefore, a smaller amount of the arc heat dissipates to the HAZ, resulting in only a slight decrease in the hardness of the HAZ compared to that of the base metal. In conclusion, the use of nanoparticle oxide results in no significant difference in the delta ferrite content and Vickers hardness compared with the weld metal made with microparticle oxide or without any oxide.
Figure 10.
Hardness distribution of TIG weldments made with and without oxides.
Figure 10.
Hardness distribution of TIG weldments made with and without oxides.