Copper Tube Pitting in Santa Fe Municipal Water Caused by Microbial Induced Corrosion
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optical Microscopy


2.2. X-ray Diffraction

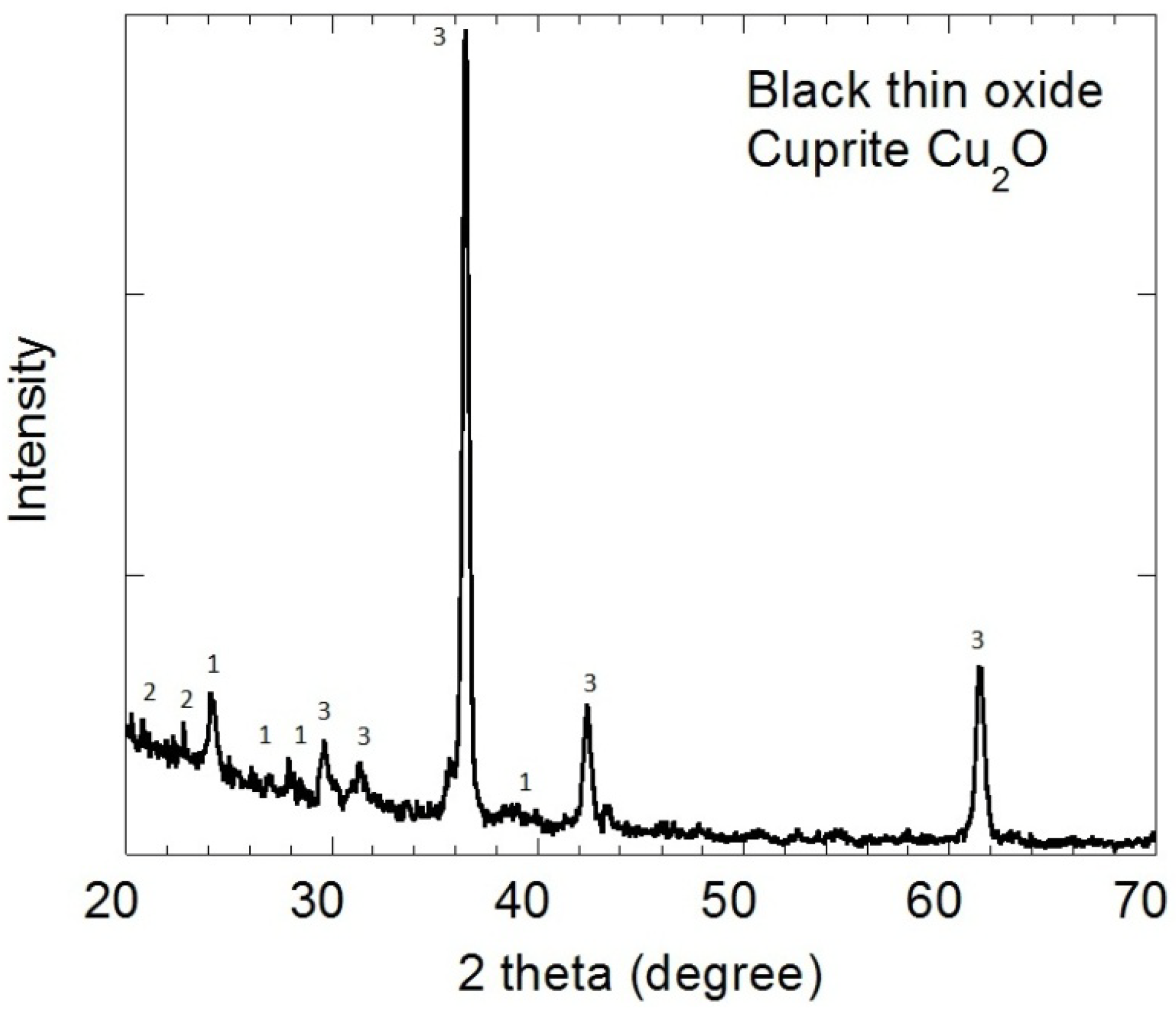
2.3. Scanning Electron Microscopy (SEM)








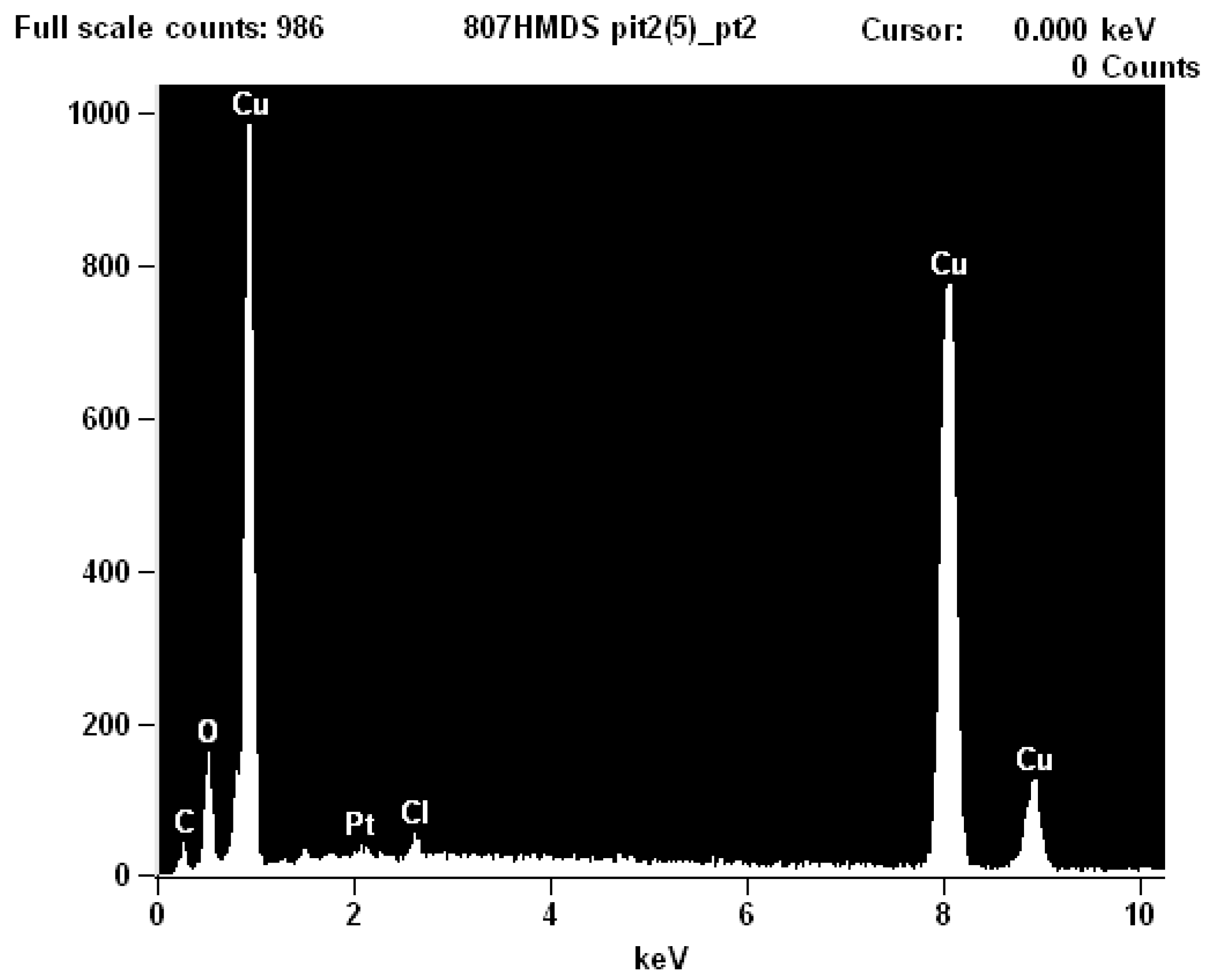



2.4. Discussion
3. Experimental Section
3.1. Study Sites
| Parameter | Low | High |
|---|---|---|
| Chloride (mg/L) | ≤10 | 45 |
| Sulfate (mg/L) | Not detected | 75 |
| Hardness (Ca & Mg) (mg/L) | 90 | 263 |
| pH | 7.0 | 8.2 |
3.2. Copper Tube Sampling
3.3. Characterization Techniques
3.4. Fixation in Hexamethyldisilazane (HMDS)
- (1)
- Immersion in Santa Fe municipal water during storage and shipping;
- (2)
- Immersion in 2.5% gluteraldehyde-water solution overnight;
- (3)
- Immersion in 25% ethanol-water solution overnight;
- (4)
- Immersion in 50% ethanol-water solution overnight;
- (5)
- Immersion in 75% ethanol-water solution overnight;
- (6)
- Immersion in 95% ethanol-water solution overnight;
- (7)
- Immersion in 100% ethanol-water solution overnight;
- (8)
- Immersion in 33% HMDS–ethanol solution for 10 min;
- (9)
- Immersion in 50% HMDS–ethanol solution for 10 min;
- (10)
- Immersion in 100% HMDS–ethanol solution for 10 min;
- (11)
- Dry overnight in a desiccant chamber;
- (12)
- Platinum sputter coat for viewing in SEM.
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lytle, D.A.; Nadagouda, M.N. A comprehensive investigation of copper pitting corrosion in a drinking water distribution system. Corros. Sci. 2010, 52, 1927–1938. [Google Scholar] [CrossRef]
- Farooqi, O.E.; Loganathan, G.; Edwards, M.; Bosch, D.; Lee, J.; Scardina, P. Copper Pinhole Failures: Plumbing Susceptitibility and Management. J. Water Resour. Plan. Manag. 2009, 135, 227–236. [Google Scholar] [CrossRef]
- Francis, R. The Corrosion of Copper and Its Alloys: A Practical Guide for Engineers; NACE International: Houston, TX, USA, 2010. [Google Scholar]
- Oliphant, R.J.; Jönsson, J. Review of Current Knowledge: Causes of Copper Corrosion in Plumbing Systems; Foundation for Water Research: Buckinghamshire, UK, 2003. [Google Scholar]
- Geesey, G.G.; Bremer, P.J.; Fischer, W.R.; Wagner, D.; Keevil, C.W.; Walker, J.; Chamberlain, A.H.L.; Angell, P. Unusual Types of Pitting Corrosion of Copper Tubes in Potable Water Systems. In Biofouling and Biocorrosion in Industrial Water Systems; Geesey, G.G., Lewandowski, Z., Flemming, H.C., Eds.; Lewis Publishers, CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Lucey, V.F. Mechanism of Pitting Corrosion of Copper in Supply Waters. Br. Corros. J. 1967, 2, 175–185. [Google Scholar] [CrossRef]
- Emily, S.; Kara, D.; Paolo, S.R.; Rebecca, L.-S.; Marc, E.; Caroline, N. Copper pitting in chlorinated, high-pH potable water. J. Am. Water Work. Assoc. 2011, 103, 86–98. [Google Scholar]
- Ha, H.; Taxen, C.; Williams, K.; Scully, J. Effects of selected water chemistry variables on copper pitting propagation in potable water. Electrochim. Acta 2011, 56, 6165–6183. [Google Scholar] [CrossRef]
- Ha, H.M.; Scully, J.R. Artificial Pit Study on Effects of Bulk Solution Composition Charnges on Copper Pitting Propagation in Synthetic Potable Waters. J. Electrochem. Soc. 2012, 159, C571–C582. [Google Scholar] [CrossRef]
- Campbell, H.S. Pitting Corrosion in Copper Water pipes caused by films of carbonaceous material produced during manufacture. J. Inst. Met. 1950, 77, 345–356. [Google Scholar]
- Wagner, D.; Chamberlain, A.H.L. Microbiologically influenced copper corrosion in potable water with emphasis on practical relevance. Biodegradation 1997, 8, 177–187. [Google Scholar] [CrossRef]
- Reyes, A.; Letelier, M.V.; de la Iglesia, R.; González, B.; Lagos, G. Microbiologically induced corrosion of copper pipes in low-pH water. Int. Biodeterior. Biodegrad. 2008, 61, 135–141. [Google Scholar] [CrossRef]
- Cantor, A.F.; Bushman, J.B.; Glodoski, M.S.; Kiefer, E.; Bersch, R.; Wallenkamp, H. Copper Pipe Failure by Microbiologically Influenced Corrosion. Mater. Perform. 2006, 45, 38–41. [Google Scholar]
- Labuda, E.M.; Bartholomew, R.D.; Hull, E.H. Morphologies of MIC in Copper and Carbon Steel Tubes—Case Histories. In Proceedings of the NACE EXPO Corrosion 2012, Salt Lake City, UT, USA, 11–15 March 2012.
- Polan, N.W. Corrosion of Copper and Copper Alloys. In ASM Handbook; ASM International: Metals Park, OH, 1987; Volume 13. [Google Scholar]
- Michels, H.T.; Anderson, D.G. Antimicrobial regulatory efficacy testing of solid copper alloy surfaces in the USA. In Metal Ions in Biology and Medicine; Collery, P., Maymard, I., Theophanides, T., Khassanova, L., Collery, T., Eds.; John Libbey Eurotext: Paris, France, 2008; Volume 10, pp. 185–190. [Google Scholar]
- Goodfellow, M.; Boone, D.R.; Castenholz, R.W.; Garrity, G.M.; Brenner, D.J.; Krieg, N.R.; Staley, J.T. The Actinobacteria. In Bergey’s Manual of Systematic Bacteriology, 2nd ed.; Springer: New York, NY, USA, 2012. [Google Scholar]
- Ventura, M.; Canchaya, C.; Tauch, A.; Chandra, G.; Fitzgerald, G.F.; Chater, K.F.; van Sinderen, D. Genomics of Actinobacteria: Tracing the evolutionary history of an ancient phylum. Microbiol. Mol. Biol. Rev. 2007, 71, 495–548. [Google Scholar] [CrossRef]
- Boston, P.J.; Summers-Engel, A.; Barton, H.A.; Northrup, D.E.; Curry, M.C.; Spilde, M.N. Geomicrobiology of Caves. In Caves and Karst of the USA; In, A.N., Palmer, M.V., Eds.; National Speleological Society Press: Huntsville, AL, USA, 2009; pp. 403–410. [Google Scholar]
- Spilde, M.N.; Boston, P.J.; Francis, C.A. Diseased minerals: Microbial degradation of copper sulfide museum specimens. Geochim. Cosmochim. Acta 2006, 70, A607–A607. [Google Scholar] [CrossRef]
- White, C.; Tancos, M.; Lytle, D.A. Microbial Community Profile of a Lead Service LIne Removed from a Drinking Water Distribution System. Appl. Environ. Microbiol. 2011, 77, 5557–5561. [Google Scholar] [CrossRef]
- Kjellerup, B.V.; Thomsen, T.R.; Nielsen, J.L.; Olesen, B.H.; Frølund, B.; Nielsen, P.H. Microbial diversity in biofilms from corroding heating systems. Biofouling 2005, 21, 19–29. [Google Scholar] [CrossRef]
- Teixeira, C.M.; Ramires, I.; Guastaldi, O.G.J.A.C. Electrochemical techniques applied to study the oxidative dissolution of the covellite—CuS by Thiobacillus ferrooxidans. Quím. Nova 2002, 25, 20–26. [Google Scholar]
- Dold, B.; Blowes, D.W.; Dickhout, R.; Spangenberg, J.E.; Pfeifer, H.R. Low molecular weight carboxylic acids in oxidizing porphyry copper tailings. Environ. Sci. Technol. 2005, 39, 2515–2521. [Google Scholar] [CrossRef]
- Little, B.; Ray, R.; Pope, R. Relationship between corrosion and the biological sulfur cycle: A review. Corrosion 2000, 56, 433–443. [Google Scholar] [CrossRef]
- Lorowitz, W.; Nagle, D.; Tanner, R. Anaerobic oxidation of elemental metals coupled to methanogenesis by methanobacterium-thermoautotrophicum. Environ. Sci. Technol. 1992, 26, 1606–1610. [Google Scholar] [CrossRef]
- Snyder, B. Santa Fe Water Quality. Available online: http://www.santafenm.gov/index.aspx?NID=1029 (accessed on 30 May 2014).
- Dekker, N.P.; Lammel, C.J.; Brooks, G.F. Scanning electron microscopy of piliated Neisseria gonorrhoeae processed with hexamethyldisilazane. J. Electron Microsc. Tech. 1991, 19, 461–467. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Burleigh, T.D.; Gierke, C.G.; Fredj, N.; Boston, P.J. Copper Tube Pitting in Santa Fe Municipal Water Caused by Microbial Induced Corrosion. Materials 2014, 7, 4321-4334. https://doi.org/10.3390/ma7064321
Burleigh TD, Gierke CG, Fredj N, Boston PJ. Copper Tube Pitting in Santa Fe Municipal Water Caused by Microbial Induced Corrosion. Materials. 2014; 7(6):4321-4334. https://doi.org/10.3390/ma7064321
Chicago/Turabian StyleBurleigh, Thomas D., Casey G. Gierke, Narjes Fredj, and Penelope J. Boston. 2014. "Copper Tube Pitting in Santa Fe Municipal Water Caused by Microbial Induced Corrosion" Materials 7, no. 6: 4321-4334. https://doi.org/10.3390/ma7064321
APA StyleBurleigh, T. D., Gierke, C. G., Fredj, N., & Boston, P. J. (2014). Copper Tube Pitting in Santa Fe Municipal Water Caused by Microbial Induced Corrosion. Materials, 7(6), 4321-4334. https://doi.org/10.3390/ma7064321





