Fluorescent Magnetic Bioprobes by Surface Modification of Magnetite Nanoparticles
Abstract
:1. Introduction
| Surface coating | Fluorophore | Potential bioapplications | Reference |
|---|---|---|---|
| None | FITC | MRI | [14] |
| None | FITC | MRI, cell tracking and drug delivery | [15] |
| None | QDs | Cell tracking and drug delivery | [16] |
| PNIPAM | None | Drug delivery | [17] |
| PEG | FITC | Drug delivery | [18] |
| PS | Pyrene | Bio-separation | [19] |
| PDADMAC/PSS | QDs | Cell tracking | [20] |
| APTS | Protoporphyrin IX | In vitro biological image | [21] |

2. Experimental
2.1. Chemicals
2.2. Synthesis of Magnetite (Fe3O4) and Silica Coated Magnetite (Fe3O4@SiO2)
2.3. Surface Functionalization of Fe3O4@ SiO2 with Fluorophore FITC
2.4. Characterization of the Nanoparticles
2.5. Interaction of Magnetic Nanostructures with Living Cells
3. Results and Discussion
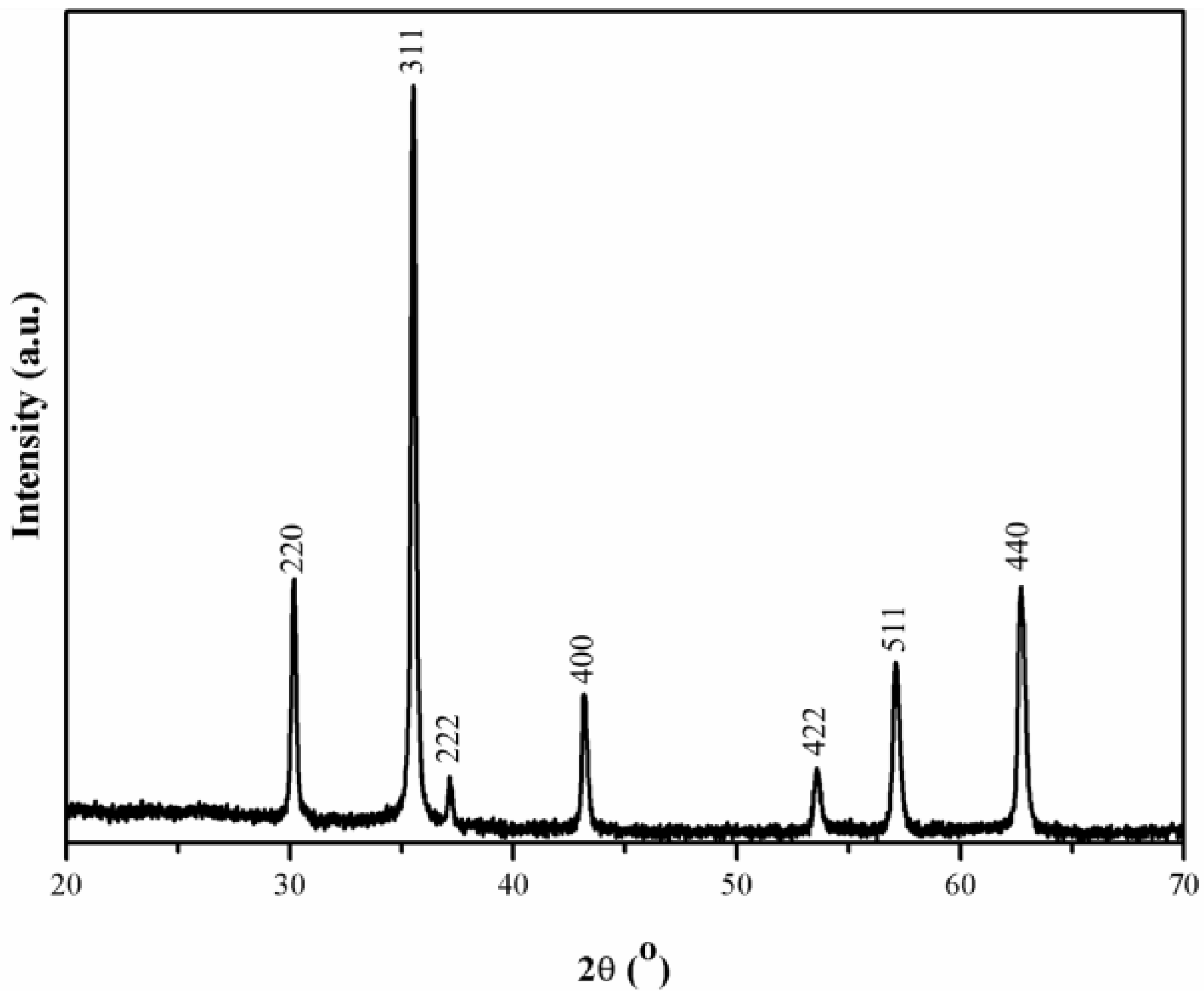


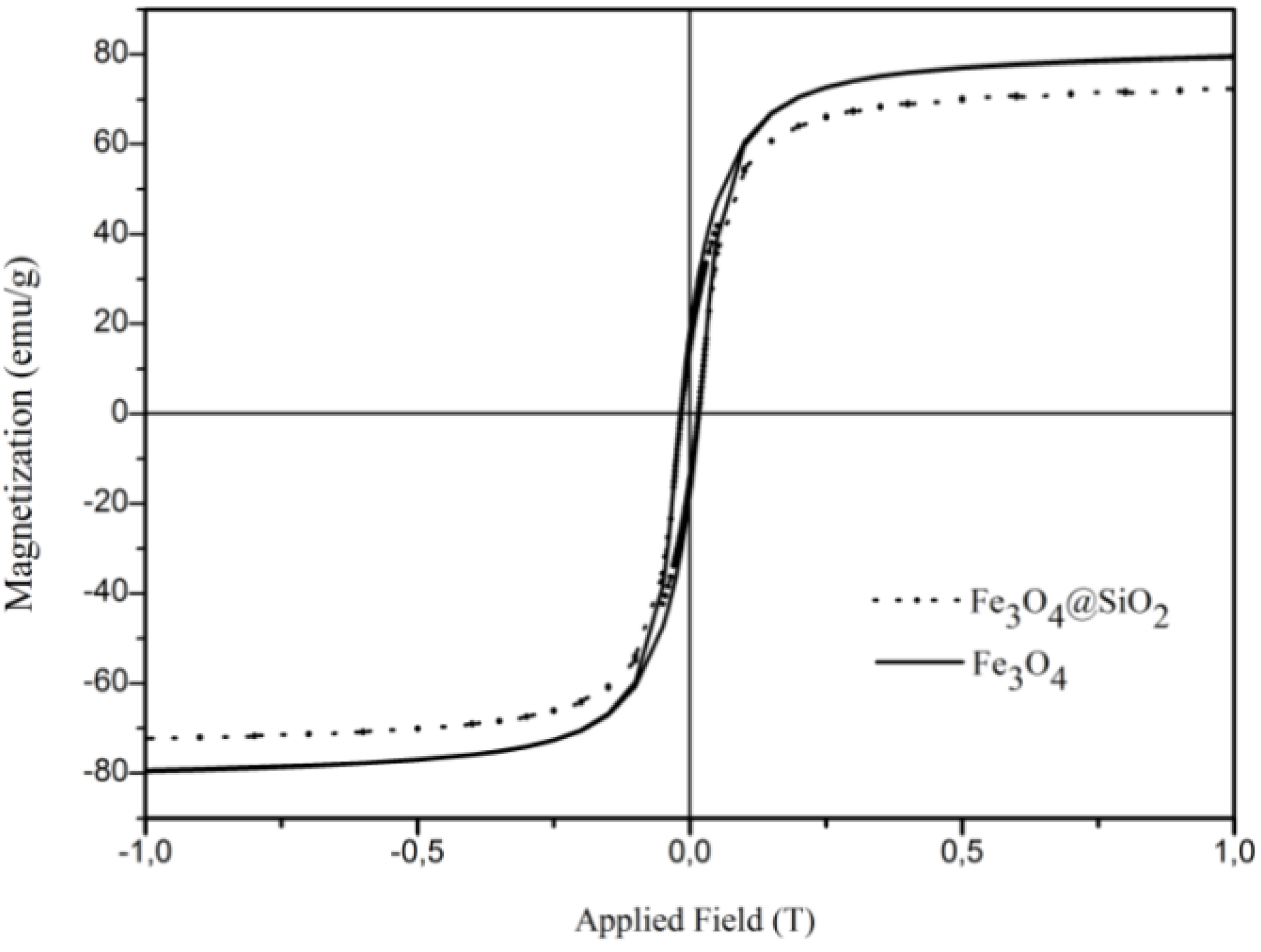
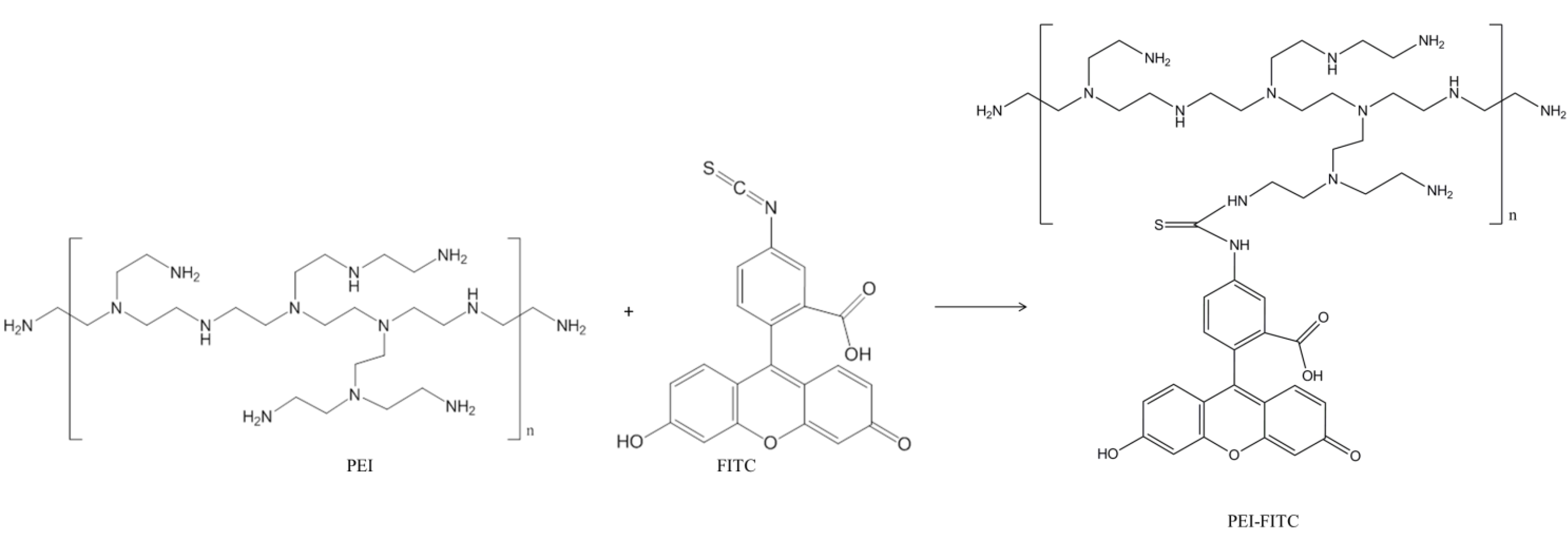
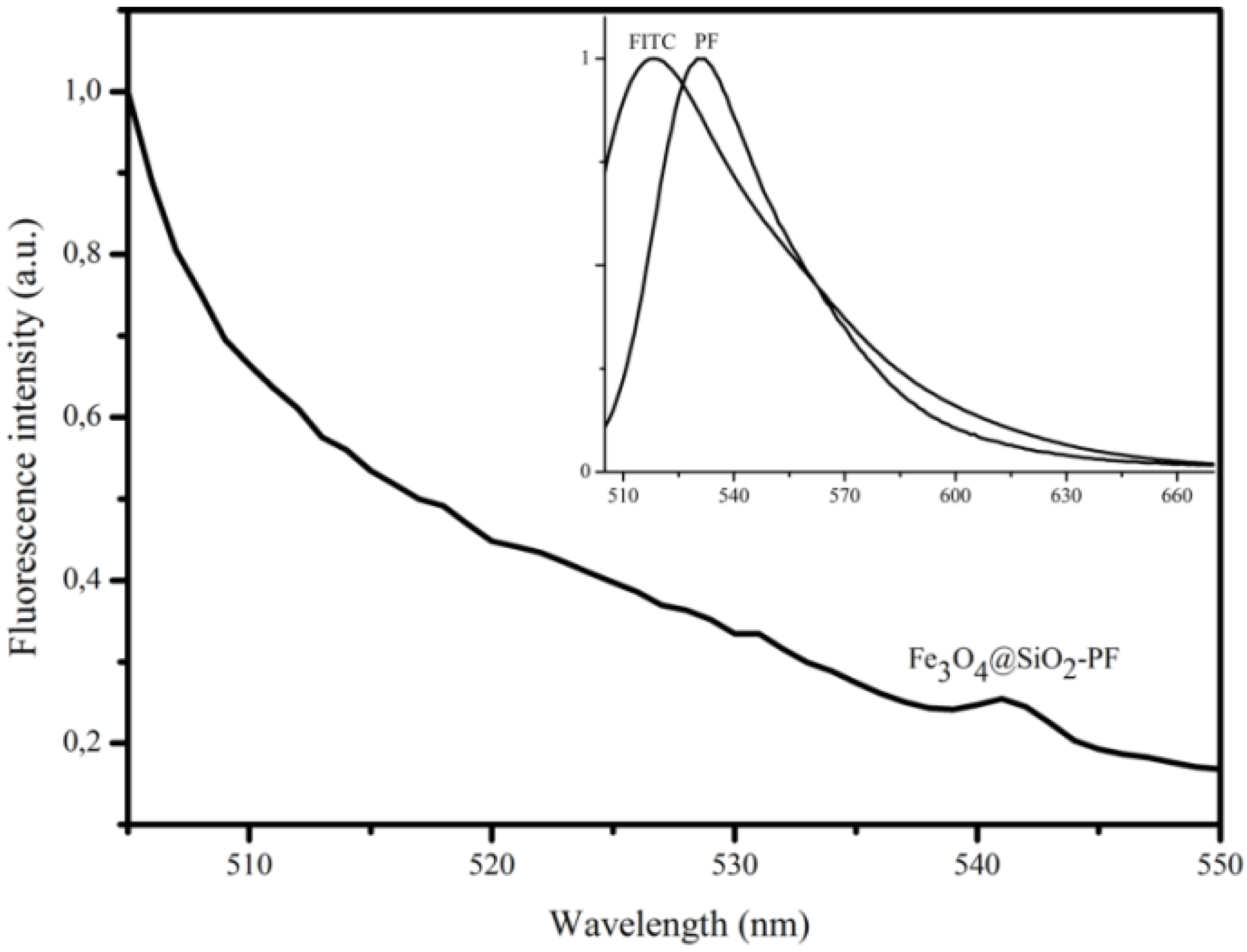
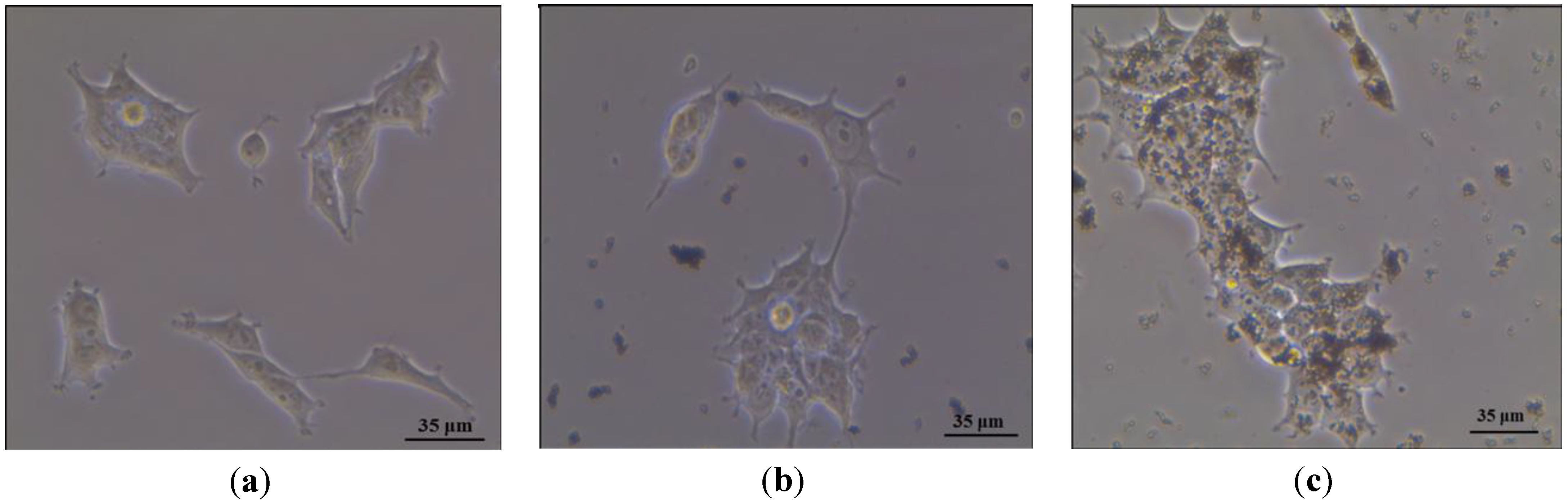


4. Conclusions
Acknowledgements
References
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R167–R181. [Google Scholar] [CrossRef]
- McBain, S.C.; Yiu, H.H.P.; Dobson, J. Magnetic nanoparticles for gene and drug delivery. Int. J. Nanomed. 2008, 2, 169–180. [Google Scholar]
- Sun, C.; Lee, J.S.H.; Zhang, M. Magnetic nanoparticles in MR imaging and drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1252–1265. [Google Scholar] [CrossRef] [PubMed]
- Corr, S.A.; Rakovich, Y.P.; Gun’ko, Y.K. Multifunctional magnetic-fluorescent nanocomposites for biomedical applications. Nanoscale Res. Lett. 2008, 3, 87–104. [Google Scholar] [CrossRef]
- Arruebo, M.; Férnandez-Pacheco, R.; Ibarra, M.R.; Santamaria, J. Magnetic nanoparticles for drug delivery. Nanotoday 2007, 2, 22–32. [Google Scholar] [CrossRef]
- Zhou, L.; Yuan, J.; Wei, Y. Core-shell structural iron oxide hybrid nanoparticles: From controlled synthesis to biomedical applications. J. Mater. Chem. 2011, 21, 2823–2840. [Google Scholar] [CrossRef]
- Zhang, C.; Wängler, B.; Morgenstern, B.; Zentgraf, H.; Eisenhut, M.; Untenecker, H.; Krüger, R.; Huss, R.; Seliger, C.; Semmler, W.; Kiessling, F. Silica- and alkoxysilane-coated ultrasmall superparamagnetic iron oxide particles: A promising tool to label cells for magnetic resonance imaging. Langmuir 2007, 23, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Kim, T.H.; Patel, K.D.; Knowles, J.C.; Kim, H.W. Biocompatible magnetite nanoparticles with varying silica-coating layer for use in biomedicine: Physicochemical and magnetic properties, and cellular compatibility. J. Biomed. Mater. Res. A 2012, 100A, 1734–1742. [Google Scholar] [CrossRef] [PubMed]
- Schneider, G.; Decher, G. Functional core/shell nanoparticles via layer-by-layer assembly. Investigation of the experimental parameters for controlling particle aggregation and for enhancing dispersion stability. Langmuir 2008, 24, 1778–1789. [Google Scholar] [CrossRef] [PubMed]
- Azzazy, H.M.E.; Mansour, M.M.H. In vitro diagnostic prospects of nanoparticles. Clin. Chim. Acta 2009, 403, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mornet, S.; Vasseur, S.; Grasset, F.; Duguet, E. Magnetic nanoparticle design for medical diagnosis and therapy. J. Mater. Chem. 2004, 14, 2161–2175. [Google Scholar] [CrossRef]
- Gallagher, J.J.; Tekoriute, R.; O’Reilly, J.A.; Kerskens, C.; Gun’ko, Y.K.; Lynch, M. Bimodal magnetic-fluorescent nanostructures for biomedical applications. J. Mater. Chem. 2009, 19, 4081–4084. [Google Scholar] [CrossRef]
- Wang, L.; Neoh, K.G.; Kang, E.T.; Shuter, B.; Wang, S.C. Biodegradable magnetic-fluorescent magnetite/poly(DL-lactic acid-co-alpha,beta-malic acid) composite nanoparticles for stem cell labeling. Biomaterials 2010, 31, 3502–3511. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Meng, X.; Liu, E.; Chen, K. Incorporation of magnetite nanoparticle clusters in fluorescent silica nanoparticles for high-performance brain tumor delineation. Nanotechnology 2010, 21, 235104:1–235104:8. [Google Scholar] [CrossRef]
- Lin, Y.S.; Wu, S.H.; Hung, Y.; Chou, Y.H.; Chang, C.; Lin, M.L.; Tsai, C.P.; Mou, C.Y. Multifunctional Composite Nanoparticles: Magnetic, Luminescent, and Mesoporous. Chem. Mater. 2006, 18, 5170–5172. [Google Scholar] [CrossRef]
- Guo, J.; Yang, W.; Wang, C.; He, J.; Chen, J. Poly(N-isopropylacrylamide)-coated luminescent/magnetic silica microspheres: preparation, characterization, and biomedical applications. Chem. Mater. 2006, 18, 5554–5562. [Google Scholar] [CrossRef]
- Lien, Y.H.; Wu, T.M. Preparation and characterization of thermosensitive polymers grafted onto silica-coated iron oxide nanoparticles. J. Colloid Interface Sci. 2008, 326, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kohler, N.; Zhang, M. Surface modification of superparamagnetic magnetite nanoparticles and their intracellular uptake. Biomaterials 2002, 23, 1553–1561. [Google Scholar] [CrossRef] [PubMed]
- Nagao, D.; Yokoyama, M.; Saeki, S.; Kobayashi, Y.; Konno, M. Preparation of composite particles with magnetic silica core and fluorescent polymer shell. Colloid Polym. Sci. 2008, 286, 959–964. [Google Scholar] [CrossRef]
- Salgueiriño-Maceira, V.; Correa-Duarte, M.A.; Spasova, M.; Liz-Marzán, L.M.; Farle, M. Composite silica spheres with magnetic and luminescent functionalities. Adv. Funct. Mater. 2006, 16, 509–514. [Google Scholar] [CrossRef]
- Nowostawska, M.; Corr, S.A.; Byrne, S.J.; Conroy, J.; Volkov, Y.; Gun’ko, Y.K. Porphyrin-magnetite nanoconjugates for biological imaging. J. Nanobiotechnol. 2011, 9, 1–12. [Google Scholar] [CrossRef]
- Girginova, P.I.; Daniel-da-Silva, A.L.; Lopes, C.B.; Figueira, P.; Otero, M.; Amaral, V.S.; Pereira, E.; Trindade, T. Silica coated magnetite particles for magnetic removal of Hg2+ from water. J. Colloid Interface Sci. 2010, 345, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Vergés, M.A.; Costo, R.; Roca, A.G.; Marco, J.F.; Goya, G.F.; Serna, C.J.; Morales, M.P. Uniform and water stable magnetite nanoparticles with diameters around the monodomain–multidomain limit. J. Phys. D Appl. Phys. 2008, 41, 134003:1–134003:10. [Google Scholar]
- Hermanson, G.T. Bioconjugate Techniques; Academic Press: London, UK, 1996; pp. 303–305. [Google Scholar]
- Zhang, N.; Ding, E.; Feng, X.; Xu, Y.; Cai, H. Synthesis, characterizations of dye-doped silica nanoparticles and their application in labeling cells. Colloids Surf. B 2012, 89, 133–138. [Google Scholar] [CrossRef]
- Ge, Y.; Zhang, Y.; He, S.; Nie, F.; Teng, G.; Gu, N. Fluorescence modified chitosan-coated magnetic nanoparticles for high-efficient cellular imaging. Nanoscale Res. Lett. 2009, 4, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Lex, A.; Pacher, P.; Werzer, O.; Track, A.; Shen, Q.; Schennach, R.; Koller, G.; Hlawacek, G.; Zojer, E.; Resel, R.; Ramsey, M.; Teichert, C.; Kern, W.; Trimmel, G. Synthesis of a photosensitive thiocyanate-functionalized trialkoxysilane and its application in patterned surface modifications. Chem. Mater. 2008, 20, 2009–2015. [Google Scholar] [CrossRef]
- Pavia, D.L.; Lampmam, G.M.; Kriz, G.S.; Vyvyan, J.R. Introduction to Spectroscopy; Cengage Learning: Stamford, CT, USA, 2009. [Google Scholar]
- Wang, L.; Roitberg, A.; Meuse, C.; Gaigalas, A.K. Raman and FTIR spectroscopies of fluorescein in solutions. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2001, 57, 1781–1791. [Google Scholar] [CrossRef] [PubMed]
- Bertorelle, F.; Wilhelm, C.; Roger, J.; Gazeau, F.; Ménager, C.; Cabuil, V. Fluorescence-modified superparamagnetic nanoparticles: Intracellular uptake and use in cellular imaging. Langmuir 2006, 22, 5385–5391. [Google Scholar] [CrossRef] [PubMed]
- Voss, E.W., Jr.; Croney, J.C.; Jameson, D.M. Discrete bathochromic shifts exhibited by fluorescein ligand bound to rabbit polyclonal anti-fluorescein fab fragments. J. Protein Chem. 2002, 21, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Imhof, A.; Megens, M.; Engelberts, J.J.; Lang, D.T.N.; Sprik, R.; Vos, W.L. Spectroscopy of Fluorescein (FITC) Dyed Colloidal Silica Spheres. J. Phys. Chem. B 1999, 103, 1408–1415. [Google Scholar] [CrossRef]
- Xiao, Q.; Xiao, C. Preparation and Characterization of Silica-Coated Magnetic–Fluorescent Bifunctional Microspheres. Nanoscale Res. Lett. 2009, 4, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.T.; Wang, L.Y.; Ho, Y.P. Use of polyethylenimine-modified magnetic nanoparticles for highly specific enrichment of phosphopeptides for mass spectrometric analysis. Anal. Bioanal. Chem. 2011, 399, 2795–2806. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Yang, H.; Chang, L.; Bala, H.; Li, M.; Zou, G. Anatase TiO2 nanolayer coating on strontium ferrite nanoparticles for magnetic photocatalyst. Colloid Surf. A Physicochem. Eng. Aspect 2006, 289, 47–52. [Google Scholar] [CrossRef]
- Pimpha, N.; Chaleawlert-umpon, S.; Sunintaboon, P. Core/shell polymethyl methacrylate/ polyethyleneimine particles incorporating large amounts of iron oxide nanoparticles prepared by emulsifier-free emulsion polymerization. Polymer 2012, 53, 2015–2022. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pinheiro, P.C.; Daniel-da-Silva, A.L.; Tavares, D.S.; Calatayud, M.P.; Goya, G.F.; Trindade, T. Fluorescent Magnetic Bioprobes by Surface Modification of Magnetite Nanoparticles. Materials 2013, 6, 3213-3225. https://doi.org/10.3390/ma6083213
Pinheiro PC, Daniel-da-Silva AL, Tavares DS, Calatayud MP, Goya GF, Trindade T. Fluorescent Magnetic Bioprobes by Surface Modification of Magnetite Nanoparticles. Materials. 2013; 6(8):3213-3225. https://doi.org/10.3390/ma6083213
Chicago/Turabian StylePinheiro, Paula C., Ana L. Daniel-da-Silva, Daniela S. Tavares, M. Pilar Calatayud, Gerardo F. Goya, and Tito Trindade. 2013. "Fluorescent Magnetic Bioprobes by Surface Modification of Magnetite Nanoparticles" Materials 6, no. 8: 3213-3225. https://doi.org/10.3390/ma6083213
APA StylePinheiro, P. C., Daniel-da-Silva, A. L., Tavares, D. S., Calatayud, M. P., Goya, G. F., & Trindade, T. (2013). Fluorescent Magnetic Bioprobes by Surface Modification of Magnetite Nanoparticles. Materials, 6(8), 3213-3225. https://doi.org/10.3390/ma6083213








