Kinetics of Corrosion Inhibition of Aluminum in Acidic Media by Water-Soluble Natural Polymeric Pectates as Anionic Polyelectrolyte Inhibitors
Abstract
:1. Introduction
2. Results
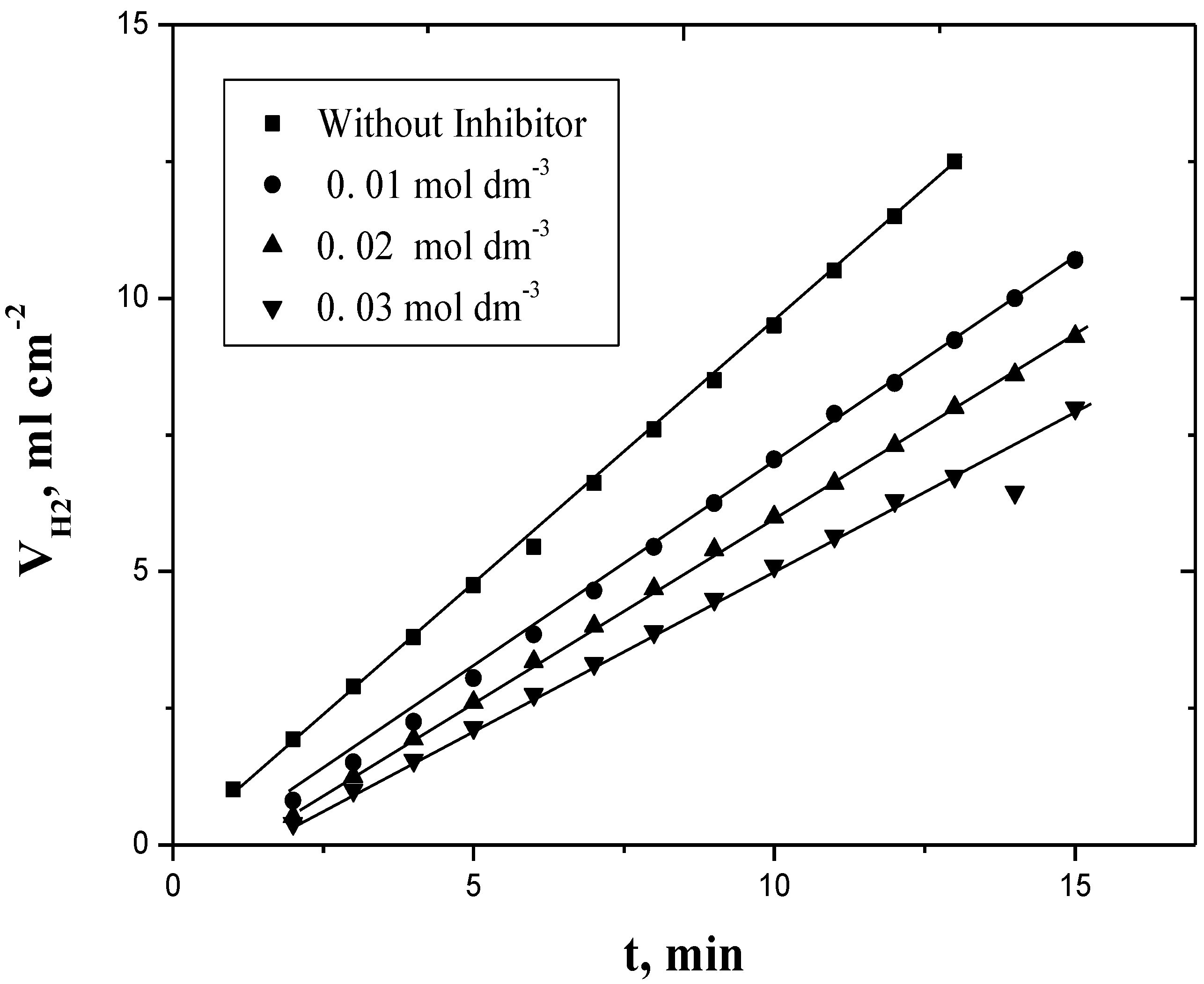
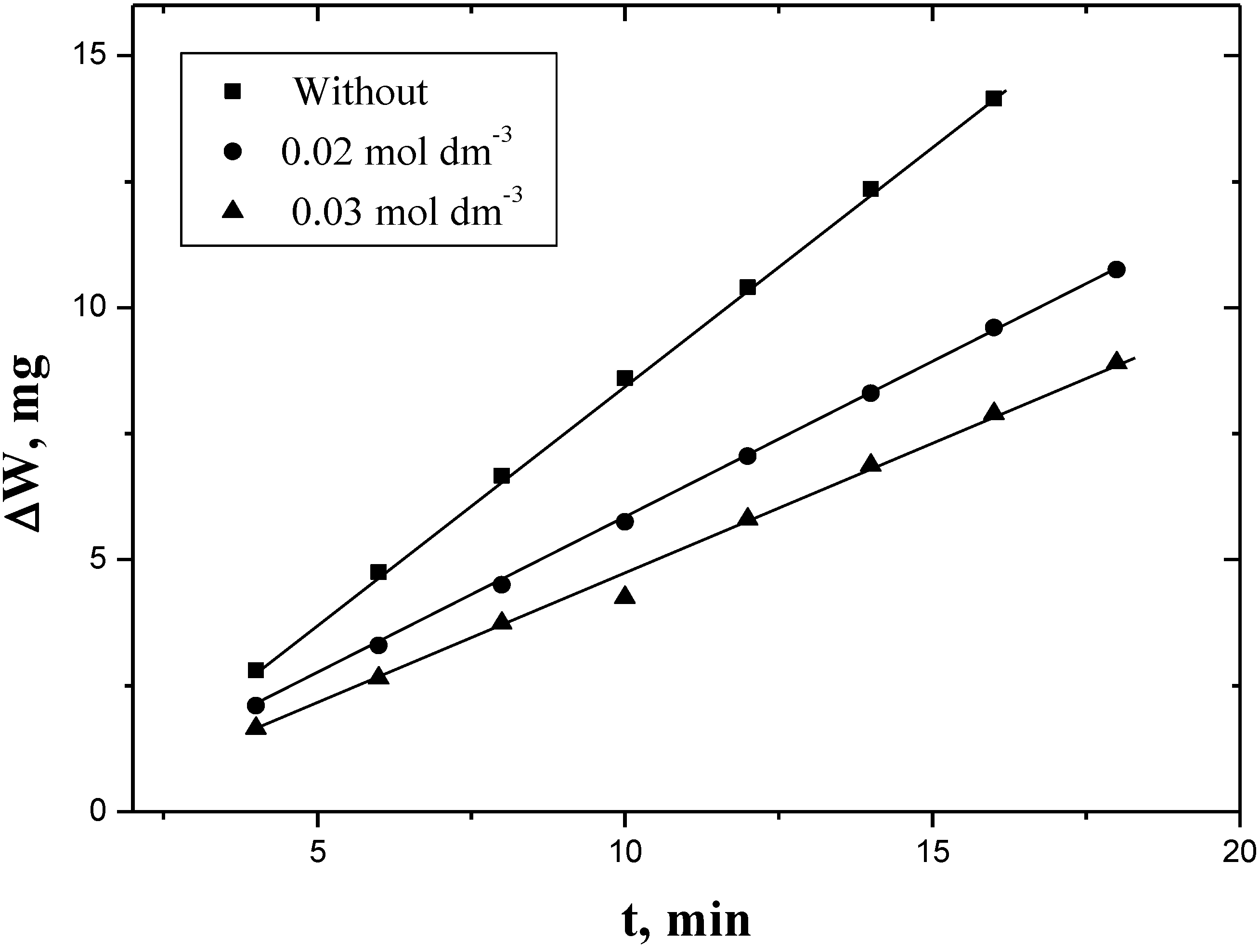
2.1. Evolved-Hydrogen (and Weight Loss)—Time Curves
2.2. Dependence of Corrosion Rate on [PEC]
| [PEC] | Temperature | |||
|---|---|---|---|---|
| % (w/v) | 102 mol dm−3 | 30 °C | 35 °C | 40 °C |
| 0.0 | 0.0 | 0.95 (0.88) * | 1.22 | 1.46 |
| 0.4 | 1.0 | 0.63 (0.59) | 0.74 | 0.84 |
| 0.6 | 2.0 | 0.55 (0.50) | 0.66 | 0.77 |
| 0.8 | 4.0 | 0.50 | 0.59 | 0.68 |
| 1.2 | 6.0 | 0.46 | 0.54 | 0.62 |
2.3. Dependence of Corrosion Rate on [H+]
| [H+], mol dm−3 | 2.0 | 3.0 | 4.0 | 5.0 |
|---|---|---|---|---|
| Roc (free), mL cm−3 min−1 | 0.39 | 0.77 | 1.46 | 1.98 |
| R′c * (inh.), mL cm−1 min−1 | 0.13 | 0.46 | 0.84 | 1.20 |
| [PEC] | 30 °C [H+], mol dm−3 | 40 °C [H+], mol dm−3 | |||
|---|---|---|---|---|---|
| %(w/v) | mol dm−3 | 3.0 | 4.0 | 3.0 | 4.0 |
| 0.4 | 0.02 | 37.04 | 33.68 | 40.26 | 42.47 |
| 0.6 | 0.03 | 48.15 | 42.11 | 50.65 | 47.26 |
| 0.8 | 0.04 | 59.26 | 47.38 | 61.03 | 53.42 |
| 1.2 | 0.06 | 68.52 | 51.57 | 70.12 | 57.53 |
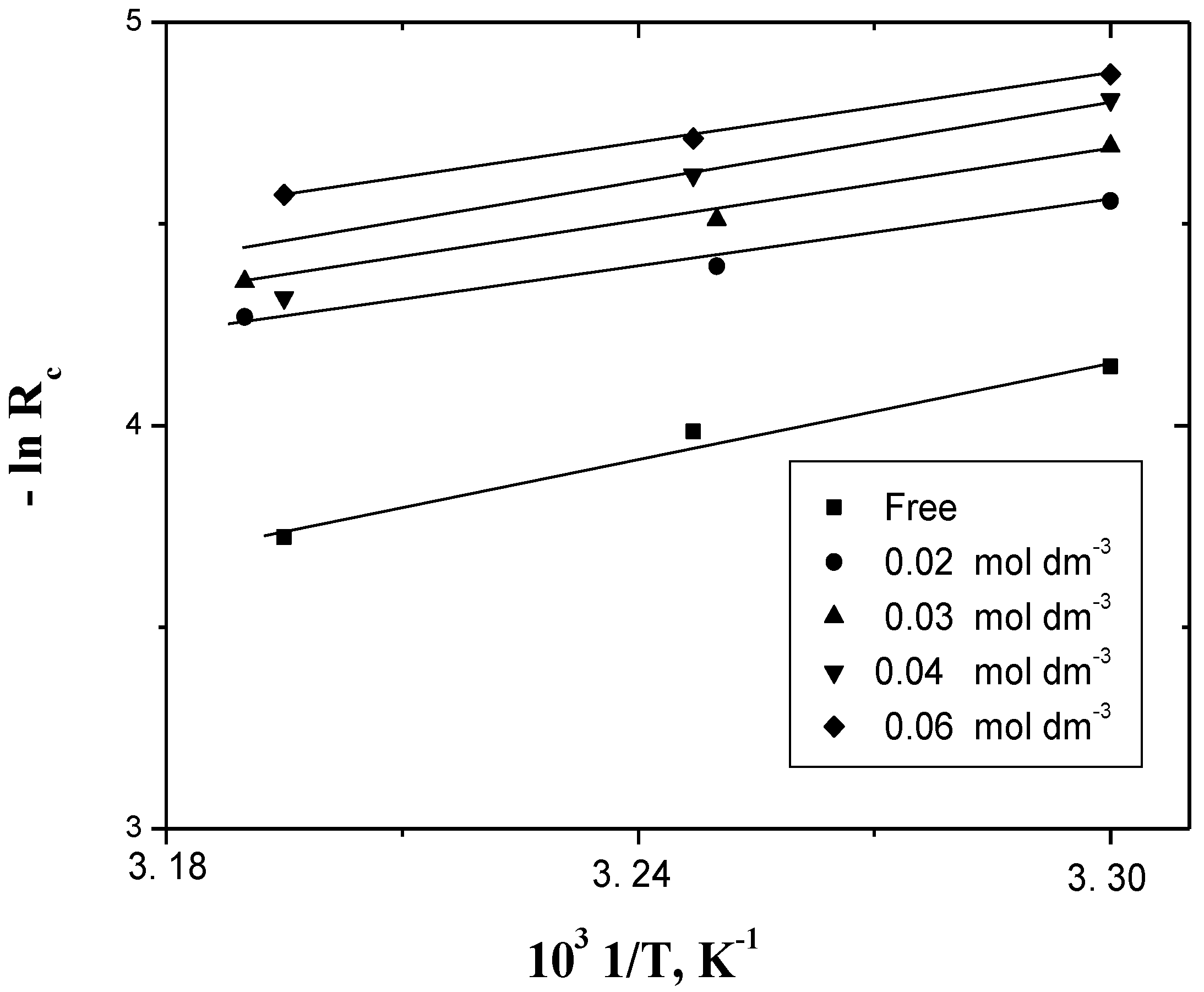
2.4. Dependence of Corrosion Rate on Temperature
3. Discussion
3.1. Corrosion Mechanism

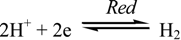

3.2. Adsorption Isotherm



| Parameter | Slope (n) 0.57 | Slope (n) 0.39 |
|---|---|---|
| 102 K, dm3 mol−1 | 28 | 62 |
| −ΔG0, kJ mol−1 | 6.91 | 2.81 |
| −ΔH0, kJ mol−1 | 15.28 | 16.05 |
| −ΔS0, J mol−1 K−1 | 73.30 | 82.40 |
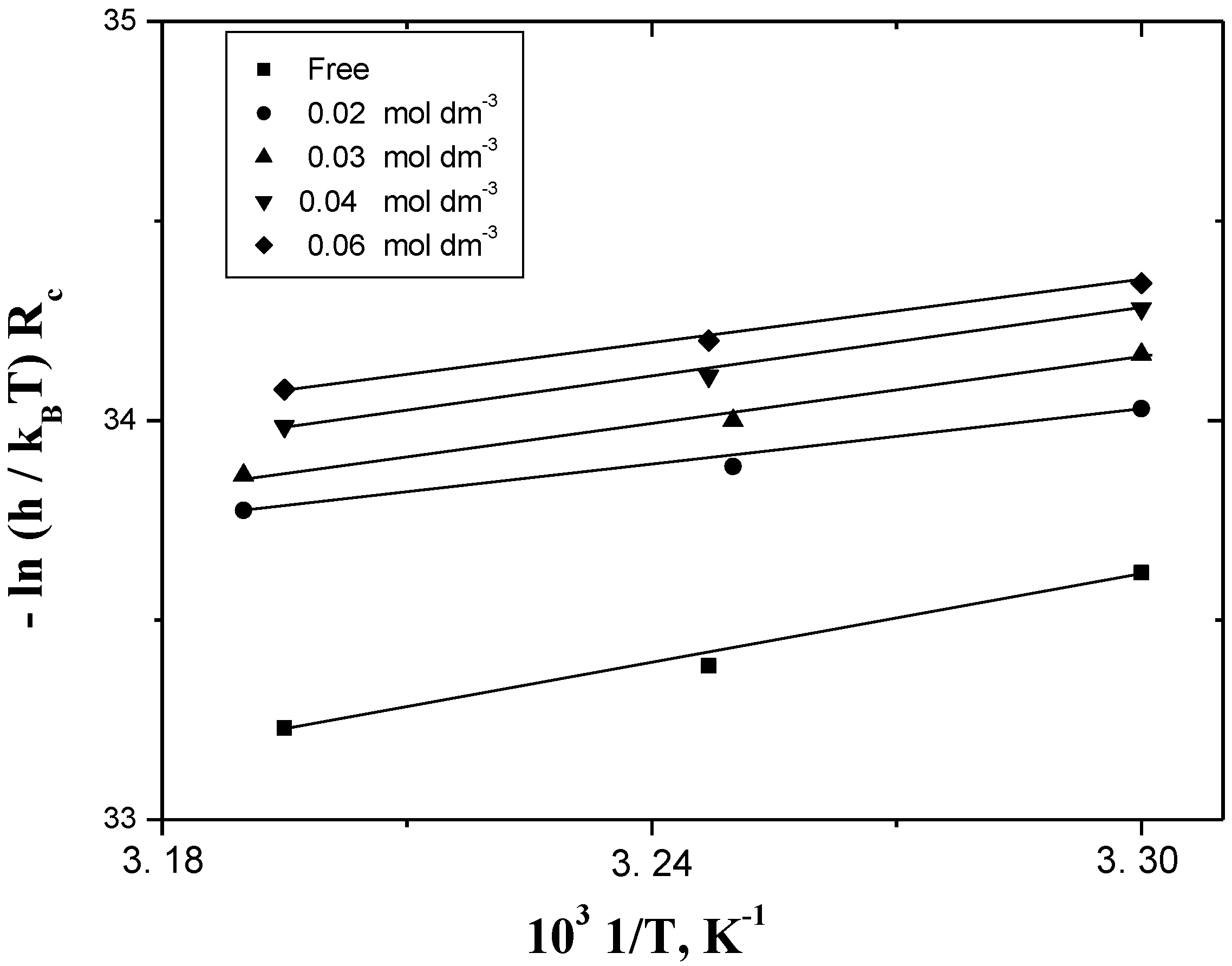
| Parameter [PEC] | ΔH≠ kJ mol−1 | ΔS≠ J mol−1K−1 | ΔG≠ kJ mol−1 | Ea≠ kJ mol−1 | A mol−1 s−1 | Rc * mL cm−2 min−1 | %IE * | |
|---|---|---|---|---|---|---|---|---|
| %(w/v) | mol dm−3 | |||||||
| 0 | 0 | 30.90 | −177.44 | 83.78 | 33.51 | 9.51 × 103 | 0.95 | – |
| 0.4 | 0.02 | 20.20 | −216.22 | 84.63 | 22.73 | 0.87 × 102 | 0.70 | 22.22 |
| 0.6 | 0.03 | 24.8 | −204.57 | 85.04 | 26.61 | 3.54 × 102 | 0.63 | 30.00 |
| 0.8 | 0.04 | 23.37 | −207. 84 | 85.31 | 24.08 | 1.19 × 102 | 0.50 | 44.44 |
| 1.2 | 0.06 | 21.07 | −215.99 | 85.43 | 23.66 | 0.91 × 102 | 0.46 | 48.89 |
| Activation parameters in alkaline solutions [16] | ||||||||
| 0 | 0 | 58.85 | −61.97 | 77.31 | 61.15 | 1.11x1011 | 0.63 | – |
| 0.4 | 0.02 | 88.35 | + 3.69 | 87.31 | 91.06 | 6.83x1013 | 0.32 | 46.67 |
| 0.8 | 0.04 | 92.63 | + 11.13 | 89.31 | 95.15 | 6.46x1013 | 0.16 | 72.67 |
| 1.2 | 0.06 | 88.82 | −4.77 | 90.42 | 91.78 | 1.16x1013 | 0.11 | 81.67 |
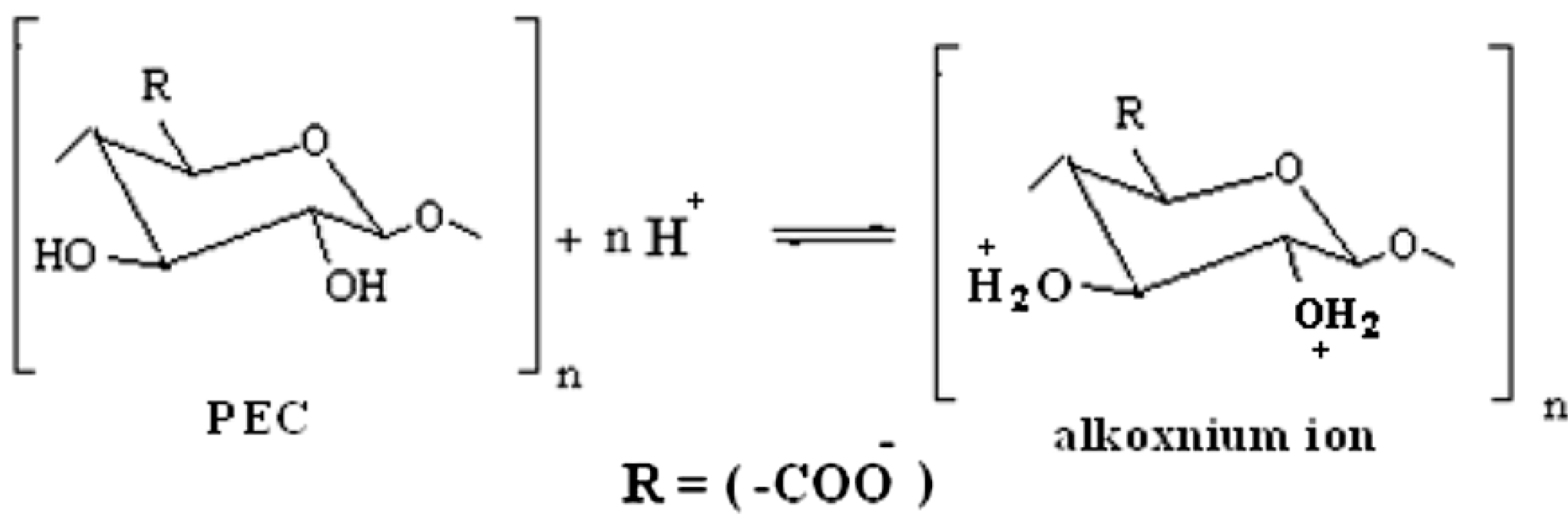
4. Experimental Section
4.1. Materials
4.2. Preparation of Pectate Sols
4.3. Hydrogen Evolution Measurements
5. Conclusions
References
- Binger, W.W. Corrosion Resistance of Metal and Alloy; Laque, P.L., Copson, M.R., Eds.; Reinhold Publishing Corp.: New York, NY, USA, 1963. [Google Scholar]
- Rengamani, S.; Muralidiharan, S.; Kulandainathan, M.A.; Venkata-kriskna, I.S. Inhibiting and accelerating effects of aminophenols on the corrosion and permeation of hydrogen through mild steel in acidic solutions. J. Appl. Electrochem. 1994, 24, 355–360. [Google Scholar] [CrossRef]
- Muller, B.; Schmelich, T. High-molecular weight styrene-maleic acid copolymers as corrosion inhibitors for aluminium pigments. Corros. Sci. 1995, 37, 877–883. [Google Scholar] [CrossRef]
- Muller, B.; Oughourlian, C.; Schubert, M. Amphiphilic Copolymers as Corrosion Inhibitors for Zinc Pigment. Corros. Sci. 2000, 42, 577–584. [Google Scholar] [CrossRef]
- Muller, B. Polymeric Corrosion Inhibitors for Aluminium Pigment. React. Funct. Polym. 1999, 39, 165–177. [Google Scholar] [CrossRef]
- Amin, M.A.; Hazzazi, O.A.; Abd El-Rhim, S.S.; El-Sherbini, E.F.; Abbas, M.N. Polyacrylic Acid as A Corrosion Inhibitor for Aluminium in Weakly Alkaline Solutions. Part I: Weight Loss, Polarization, Impedance EFM and EDX Studies. Corros. Sci. 2009, 51, 658–667. [Google Scholar] [CrossRef]
- Ramakrishnaih, K.; Subramanyan, N. Effect of Some Nitrogen Containing Organic Compounds on the Corrosion and Polarization Behaviour of Aluminium in 1 M Solutions of Sodium Hydroxide and Hydrochloric Acid with and Without Calcium. Corros. Sci. 1976, 16, 307–316. [Google Scholar] [CrossRef]
- Taiati, J.D.; Mold, R.M. p-Substituted phenols as corrosion inhibitors for aluminium-copper Alloy in Sodium Hydroxide. Corros. Sci. 1979, 19, 35–48. [Google Scholar] [CrossRef]
- Daufin, G.; Labre, J.P.; Pagetti, J. Corrosion Inhibition of an Aluminium–Silicon–Magnesium Alloy in Alkaline Media. Corros. Sci. 1977, 17, 901–912. [Google Scholar] [CrossRef]
- Solyneos, K.G.; Varhegyi, B.; Kalman, E.; Karman, F. H.; Gal, M.; Hencsei, P.; Bihatsi, L. Inhibition of Aluminium Corrosion in Alkaline Solutions by Silicon and Nitrogen Containing Compounds. Corros. Sci. 1993, 35, 1455–1457. [Google Scholar] [CrossRef]
- Muller, B.; Fisher, S. Epoxy ester resins as corrosion inhibitors for aluminium and zinc pigments. Corros. Sci. 2006, 48, 2406–2416. [Google Scholar] [CrossRef]
- Lunarska, E.; Chernagayeva, O. Effect of Corrosion Inhibitors on Hydrogen Uptake by Al from NaOH. Solution. Int. J. Hydrog. Eng 2006, 31, 285–293. [Google Scholar] [CrossRef]
- Awad, S.A.; Kamal, Kh.M.; Kassab, A. Effect of Anions on the Corrosion of Aluminium in Sodium: Part I. The Chromate Ion. J. Electroanal. Chem. 1981, 127, 203–209. [Google Scholar] [CrossRef]
- Arenos, M.A.; Bethencourt, M.; Botana, F.G.; Domborenena, J.; Marcos, M. Inhibition of 5083 Aluminium alloy and Galvanised Steel by Lanthanide Salts. Corros. Sci. 2001, 43, 157–170. [Google Scholar] [CrossRef]
- Abdel Gaber, A.M.; Khamis, E.; Abo El-Dahab, H.; Adel, Sh. Inhibition of Aluminium Corrosion in Alkaline Solutions using Natural Compound. Mater. Chem. Phys. 2008, 109, 297–305. [Google Scholar] [CrossRef]
- Zaafarany, I.A.; Khairou, K.S.; Hassan, R.M. Influence of Some Natural Polymeric Compounds Especially Alginate and Pectate Polysaccharides in Aqueous Alkaline Solutions. In Proceedings of Taibah International Fourth Saudi Science Conference, Al-Madina Al-Munawwara, KSA, Madina, Saudi Arabia, March 2010.
- Zaafarany, I.A. Inhibition of Acidic Corrosion of Iron by Some Carrageenan Compounds. Curr. World Environ. 2006, 12, 101–108. [Google Scholar]
- Khairou, K.S.; Zaafarany, I.A. Some Sulfated Water Soluble Natural Polymer (Carrageenans) Compounds as Corrosion Inhibitors for Dissolution of Iron in Hydrochloric Acid Solution. Mater. Sci. Res. India 2006, 3, 135–140. [Google Scholar]
- Solomon, M.M.; Umoren, S.A.; Udsoro, I.I.; Udoh, A.P. Synergistic and Antagonistic Effects Between Halide Ions and Carboxymethyl Cellulose for the Corrosion Inhibition of Mild Steel in Sulphuric Acid Solution. Cellulose 2010, 17, 635–648. [Google Scholar] [CrossRef]
- Solomon, M.M; Umoren, S.A.; Udosoro, I.I.; Udoh, A.P. Inhibitive and Adsorption Behaviour of Carboxymethyl Cellulose on Mild Steel Corrosion in Sulphuric Acid Solution. Corros. Sci. 2010, 52, 1317–1325. [Google Scholar] [CrossRef]
- Sorkhabi, H.A.; Jeddi, N.G.; Hashemzadeh, F.; Jahani, H. Corrosion Inhibition of Carbon Steel in Hydrochloric Acid by Some Polyethylene Glycols. Electrochem. Acta 2006, 51, 3848–3854. [Google Scholar] [CrossRef]
- Umeron, S.A.; Li, Y.; Wang, F.H. Effect of Polyacrylic Acid on the Corrosion Behaviour of Aluminium in Sulphuric Acid Solution. J. Solid State Electrochem. 2010, 14, 2293–2305. [Google Scholar] [CrossRef]
- Hassan, R.M.; Ahmed, S.A.; Fawzy, A.; Abdel-Kader, D.A.; Ikeda, Y.; Takagi, H.D. Acid-Catalyzed Oxidation of Carboxymethyl Cellulose Polysaccharide by Chromic Acid in Aqueous Perchlorate Solutions. Catal. Commun. 2010, 11, 611–615. [Google Scholar] [CrossRef]
- Zaafarany, I.A.; Fawzy, A.; Ahmed, G.A.; Ibrahim, S.A.; Hassan, R.M.; Takagi, H.D. Further Evidence For Detection of Short-Lived Transient Hypomanganate(V) And Manganate(VI) Intermediates During Oxidation of Some Sulfated Polysaccharides by Alkaline Permanganate using Conventional Spectrophotometric Techniques. Carbonhydr. Res. 2010, 345, 1588–1593. [Google Scholar] [CrossRef]
- Hassan, R.M.; Fawzy, A.; Ahmed, G.A.; Zaafarany, I.A.; Asghar, B.S.; Khairou, K.S. Acid-Catalyzed Oxidation of Some Sulfated Macromolecules. Kinetics and Mechanism of Permanganate Oxidation of Kappa-Carrageenan Polysaccharides in Acid Perchlorate Solutions. J. Mol. Catal. 2009, 309, 95–102. [Google Scholar] [CrossRef]
- Zaafarany, I.A.; Khairou, K.S.; Hassan, R.M. Acid-Catalysis of Chromic Acid Oxidation of Kappa-Carrageenan Polysaccharide in Aqueous Perchlorate Solutions. J. Mol. Catal. 2009, 302, 112–118. [Google Scholar] [CrossRef]
- Hassan, R.M. Alginate Polyelectrolyte Ionotropic Gels. XVIII. Oxidation of Alginate Polysaccharide by Potassium Permanganate in Alkaline Solutions. Kinetics of Decomposition of the Intermediate Complex. J. Polym. Sci. 1993, 31, 51–59. [Google Scholar] [CrossRef]
- Hassan, R.M. New Coordination Polymers. IV. Oxidation of Poly(Vinyl Alcohol) By Permanganate Ion in Alkaline Solutions. Kinetics and Mechanism of Decomposition of Intermediate Complex. Polym. Int. 1993, 32, 39–42. [Google Scholar] [CrossRef]
- Hassan, R.M.; Wahdan, M.H.; Hassan, A. Kinetics and Mechanism of Sol-Gel Transformation on Poly-electrolytes of Nickel Alginate Ionotropic Membranes. Eur. Polym. J. 1988, 24, 281–283. [Google Scholar] [CrossRef]
- Khairou, K.S.; El-Gethami, W.M. Ineticsand Mechanism of Sol gel Transformation Bbetween Sodium A Polyelectrolyte and Some Heavy Divalent Metal Ions with Formation of Capillary Polymembranes Ionotropic Gels. J. Membr. Sci. 2002, 209, 445–465. [Google Scholar] [CrossRef]
- Hassan, R.M. Alginate Polyelectrolyte Ionotropic Gels. II. Kinetics and Mechanism of Exchange of Chelated Nickel(II) by Hydrogen Ions In Capillary Ionotropic Nickel Alginate Polymembrane Gel Complex. J. Mater. Sci. 1993, 28, 384–388. [Google Scholar] [CrossRef]
- Hassan, R.M.; Awad, A.; Hassan, A. Separation of Metal Alginate Ionotropic Gels to Polymembranes with Special Evidence on the Position of Chelation in Copper Alginate Complex. J. Polym. Sci. A 1991, 29, 1645–1648. [Google Scholar] [CrossRef]
- Hassan, R.M.; Makhlouf, M.Th.; El-Shatoury, S.A. Alginate Polyelectrolyte Ionotropic Gels. IX. Diffusion Control Effects on the Relaxation Time of Sol-Gel Transformation of Divalent Metal Alginate Ionotropic Gel Complexes. Colloid Polym. Sci. 1992, 270, 1237–1242. [Google Scholar] [CrossRef]
- Hassan, R.M. Influence of Frequency on Electrical Properties of Acid and Trivalent Metal Alginate Ionotropic Gels. A Correlation between Strength of Chelation and Stability of Polye1ectrolyre Gels. High Perform. Polym. 1989, 1, 275–284. [Google Scholar]
- Hassan, R.M.; Makhlouf, M.Th.; Summan, A.M.; Awad, A. Influence of Frequency on Specific Conductance of Polyelectrolyte Gels with Special Correlation between Strength of Chelation and Stability of Divalent Metal Alginate Ionotropic Gels. Eur. Polym. J. 1989, 25, 993–996. [Google Scholar] [CrossRef]
- Hassan, R.M. Alginate Polyelectrolyte Ionotropic Gels. VII. Physicochemical Studies on Silver(I) Alginate Complex with Special Correlation to the Electrical Properties and Geometrical Structure. Colloids Surf. 1991, 60, 203–212. [Google Scholar] [CrossRef]
- Hassan, R.M.; Ikeda, Y.; Homiyasu, H. Alginate Polyelectrolyte Ionotropic Gels. XV. Physicochemical Properties of Uranyl Alginate Complex Especially the Chemical Equilibrium and Electrical Conductivity Related to the Coordination Geometry. J. Mater. Sci. 1993, 28, 5143–5147. [Google Scholar] [CrossRef]
- Khairou, K.S.; Hassan, R.M. Temperature-Dependence of Electrical Conductivity for Cross-Linked Mono- and Divalent Metal Alginate Complexes. High Perform. Polym. 2002, 14, 93–99. [Google Scholar] [CrossRef]
- Zaafarany, I.A.; Khairou, K.S.; Hassan, R.M. Physicochemical Studies on Some Cross-Linked Trivalent Metal-alginate Complexes especially the Electrical Conductivity and Chemical Equilibrium Related to the Coordination Geometry. High Perform. Polym. 2010, 22, 69–81. [Google Scholar] [CrossRef]
- Said, A.A.; Hassan, R.M. Thermal Decomposition of Some Divalent Metal Alginate Gel Compounds. Polym. Degrad. Stab. 1993, 39, 393–397. [Google Scholar] [CrossRef]
- Said, A.A.; Abd El-Wahab, M.M.; Hassan, R.M. Thermal and Electrical Studies on Some Metal Alginate Compounds. Thermochim. Acta 1994, 233, 13–24. [Google Scholar] [CrossRef]
- El-Gahami, M.A.; Khairou, K.S.; Hassan, R.M. Thermal Decomposition of Sn(II), Pb(II), Cd(II) and Hg(II) Cross-Linked Metal-Alginate Complexes. Bull. Pol. Acad. Sci. 2003, 51, 105–113. [Google Scholar]
- Hassan, R.M.; Abd-Alla, M.A. New Coordination Polymers. Part I. Novel Synthesis of Poly (Vinyl Alcohol) and Characterization as Chelating Agent. J. Mater. Chem. 1992, 2, 609–611. [Google Scholar] [CrossRef]
- Hassan, R.M.; El-Gahami, M.A.; Abd-Alla, M.A. New Coordination Polymers. Part II. Coordination of Poly (Vinyl Ketone) with Divalent Metal Ions. J. Mater. Chem. 1992, 2, 613–615. [Google Scholar] [CrossRef]
- Abdel-Hamid, M.I.; Khairou, K.S.; Hassan, R. M. Kinetics and Mechanism of Permanganate Oxidation of Pectin in Acid Perchlorate Media. Eur. Polym. J. 2003, 39, 381–387. [Google Scholar] [CrossRef]
- Khairou, K.S.; Hassan, R.M. Pectate Polyelectrolyte Ionotropic Gels. I. Kinetics and Mechanism of Formation of Manganate(VI)-Pectate Intermediate Complex during Oxidation of Pectate Polysaccharide by Alkaline Permanganate. Eur. Polym. J. 2000, 36, 2021–2030. [Google Scholar] [CrossRef]
- Driske, T.B.; Timmer, R. Corrosion of zinc in potassium hydroxide solutions. J. Electrochem. Soc. 1969, 116, 162–165. [Google Scholar] [CrossRef]
- Vorkapic, L.Z.; Drazic, D.M.; Despic, A.R. Corrosion of pure and amalgamated zinc in concentrated alkali hydroxide solutions. J. Electrochem. Soc. 1974, 121, 1385–1392. [Google Scholar] [CrossRef]
- Rezenfeld, I.L. Corrosion Inhibitors; McGraw-Hill Inc.: New York, NY, USA, 1981. [Google Scholar]
- McCafferty, E. Corrosion Control by Coating; Leidheiser, H., Jr., Ed.; Science Press: Princeton, NJ, USA, 1979. [Google Scholar]
- Khairou, K.S.; El-Sayed, A. Inhibition Effect of Some Polymers on The Corrosion of Cadmium in A Hydrochloric Acid Solution. J. Appl. Polym. Sci. 2003, 88, 866–871. [Google Scholar] [CrossRef]
- Chauban, L.R.; Gunsekaran, G. Corrosion Inhibition of Mild Steel by Plant Extract in Dilute HCl Medium. Corros. Sci. 2007, 49, 1143–1161. [Google Scholar] [CrossRef]
- Keles, H.; Keles, M.; Dehri, I.; Serindag, O. Adsorption and Inhibitive Properties of Aminobiphenyl and Its Schiff Base on Mild Steel Corrosion In 0.5 M HCl Medium. Colloids Surf. A Physicochem. Eng. Asp. 2008, 320, 138–145. [Google Scholar] [CrossRef]
- Elewady, G.L.; El-Said, I.A.; Fouda, A.S. Anion Surfactants as Corrosion Inhibitors for Aluminum Dissolution in HCl Solutions. Int. J. Electrochem. Sci. 2008, 3, 177–190. [Google Scholar]
- Mazhar, M.A.; Badawy, W.A.; Abou Romia, R.M. Impedance Studies of Corrosion Resistance of Aluminium in Chloride Media. Surf. Coat. Technol. 1986, 29, 335–345. [Google Scholar] [CrossRef]
- Noor, E.A. Potential of Aqueous Extract of Hibiscus Sabdariffa Leaves for Inhibiting the Corrosion of Aluminum in Alkaline Solutions. J. Appl. Electrochem. 2009, 39, 1465–1475. [Google Scholar] [CrossRef]
- Noor, E.A.; Moubaraki, A.H. Thermodynamic Study of Metal Corrosion and Inhibitor Adsorption Processes in Mild Steel/1-Methyl-4[4′(-X)-Styrylpyridinium Iiodides/Hydrochloric Acid Systems. Mater. Chem. Phys. 2008, 110, 145–154. [Google Scholar] [CrossRef]
- Kliskic, M.; Radosevic, J.; Gudic, S.; Kotalinik, V. Aqueous extract of Rosmarinus Officinalis L. as Inhibitor of Al–Mg Alloy Corrosion in Chloride Solution. J. Appl. Electrochem. 2000, 30, 823–830. [Google Scholar] [CrossRef]
- Yurt, A.; Ulutaas, S.; Dal, H. Electrochemical and Theoretical Investigation on the Corrosion of Aluminium in Acidic Solution Containing Some Schiff Bases. Appl. Sur. Sci. 2006, 253, 919–925. [Google Scholar] [CrossRef]
- Glasstone, S.; Laidler, K.J.; Eyring, H. The Theory of the Rate Processes; McGraw-Hill: New York, NY, USA, 1941; p. 141. [Google Scholar]
- Li, X.; Tang, L. Synergistic Inhibition between OP and NaCl on the Corrosion of Cold-Rolled Steel in Phosphoric Acid. Mater. Chem. Phys. 2005, 90, 286–297. [Google Scholar] [CrossRef]
- Tang, L.; Mu, G.; Liu, G. The Effect of Neutral Red on the Corrosion Inhibition of Cold Rolled Steel in 1.0 M Hydrochloric Acid. Corros. Sci. 2003, 45, 2251–2262. [Google Scholar] [CrossRef]
- Tang, L.; Lie, X.; Si, Y.; Mu, G.; Liu, G. The Synergistic Inhibition between 8-Hydroxyquinoline and Chloride Ion for the Corrosion of Cold Rolled Steel in 0.5 M Sulfuric Acid. Mater. Chem. Phys. 2006, 95, 29–38. [Google Scholar] [CrossRef]
- Li, X.; Mu, G. Tween-40 as Corrosion Inhibitor for Cold Rolled Steel in Sulphuric Acid: Weight Loss Study, Electrochemical Characterization, and AFM. Appl. Sur. Sci. 2005, 252, 1254–1265. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hassan, R.M.; Zaafarany, I.A. Kinetics of Corrosion Inhibition of Aluminum in Acidic Media by Water-Soluble Natural Polymeric Pectates as Anionic Polyelectrolyte Inhibitors. Materials 2013, 6, 2436-2451. https://doi.org/10.3390/ma6062436
Hassan RM, Zaafarany IA. Kinetics of Corrosion Inhibition of Aluminum in Acidic Media by Water-Soluble Natural Polymeric Pectates as Anionic Polyelectrolyte Inhibitors. Materials. 2013; 6(6):2436-2451. https://doi.org/10.3390/ma6062436
Chicago/Turabian StyleHassan, Refat M., and Ishaq A. Zaafarany. 2013. "Kinetics of Corrosion Inhibition of Aluminum in Acidic Media by Water-Soluble Natural Polymeric Pectates as Anionic Polyelectrolyte Inhibitors" Materials 6, no. 6: 2436-2451. https://doi.org/10.3390/ma6062436
APA StyleHassan, R. M., & Zaafarany, I. A. (2013). Kinetics of Corrosion Inhibition of Aluminum in Acidic Media by Water-Soluble Natural Polymeric Pectates as Anionic Polyelectrolyte Inhibitors. Materials, 6(6), 2436-2451. https://doi.org/10.3390/ma6062436




