Cosmetic Analysis Using Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Imaging (MALDI-MSI)
Abstract
:1. Introduction
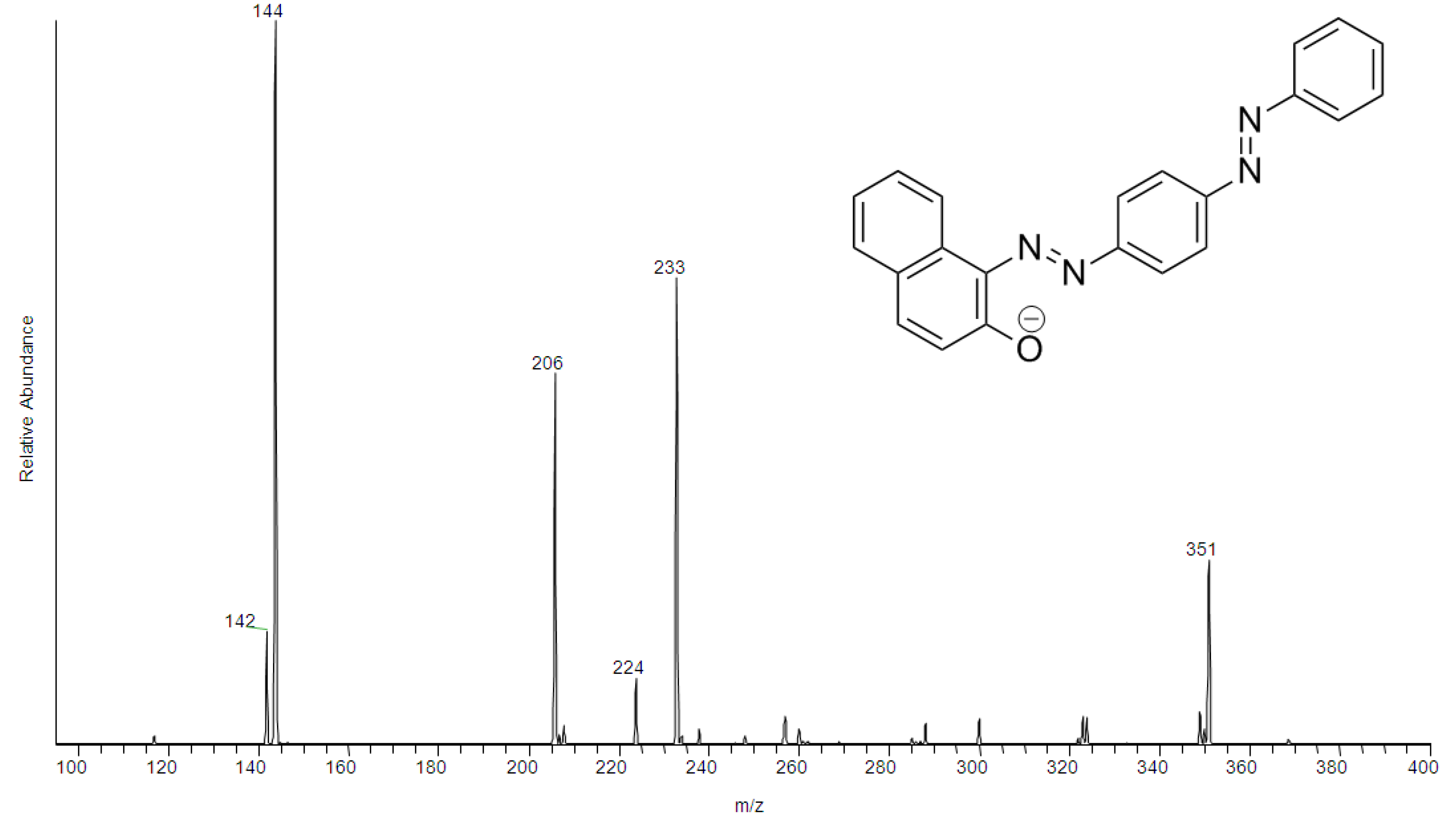
2. Results and Discussion
2.1. Chemometric Analysis
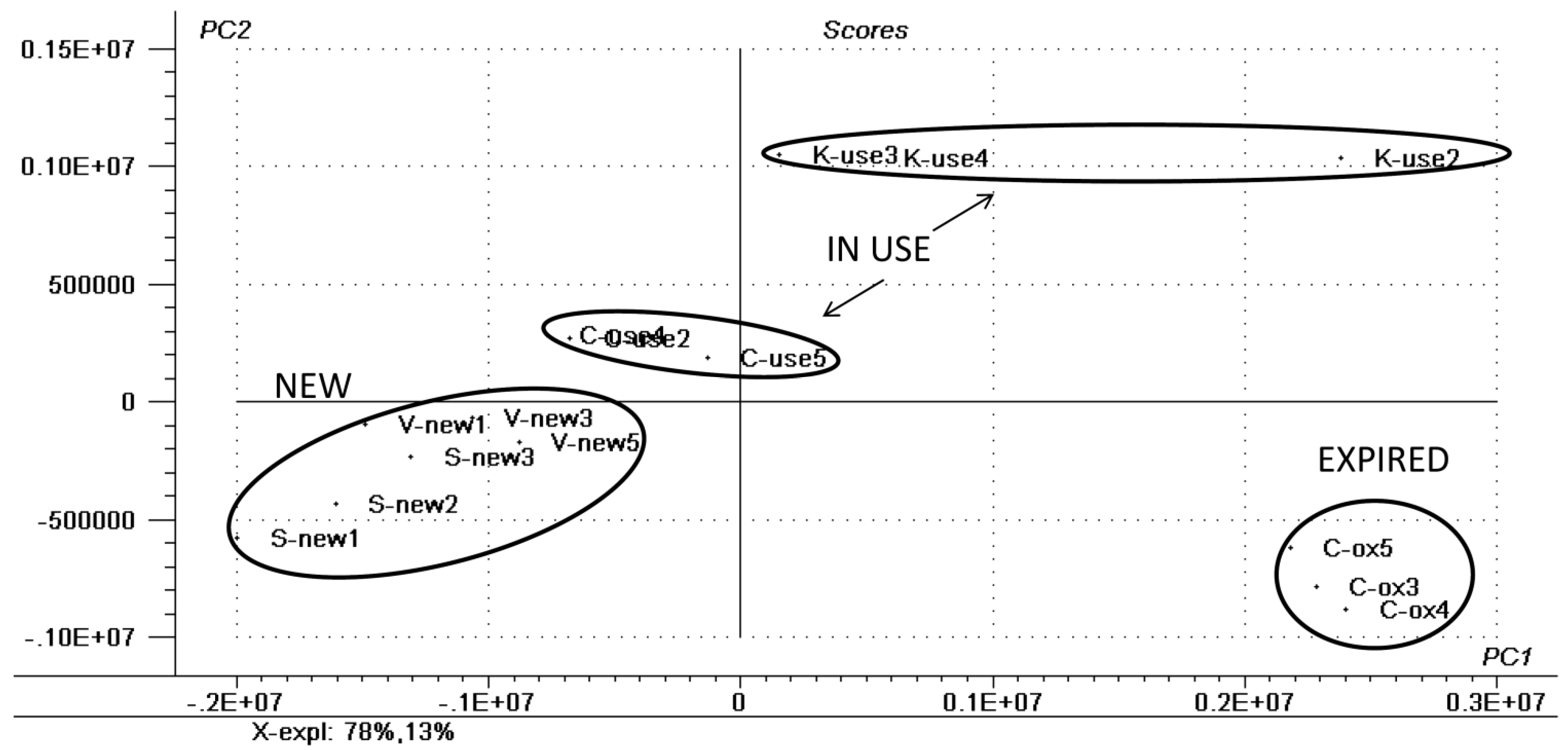
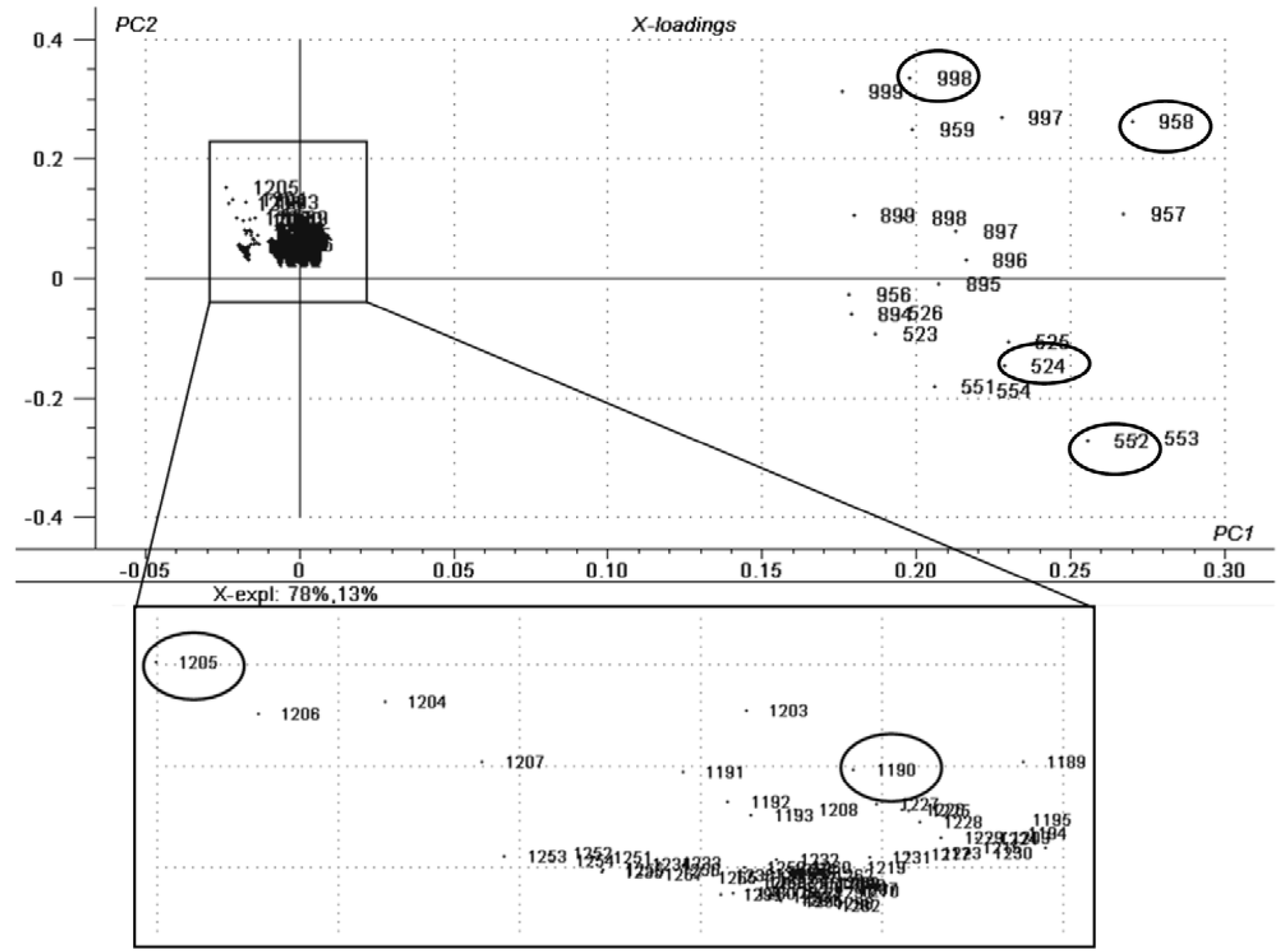
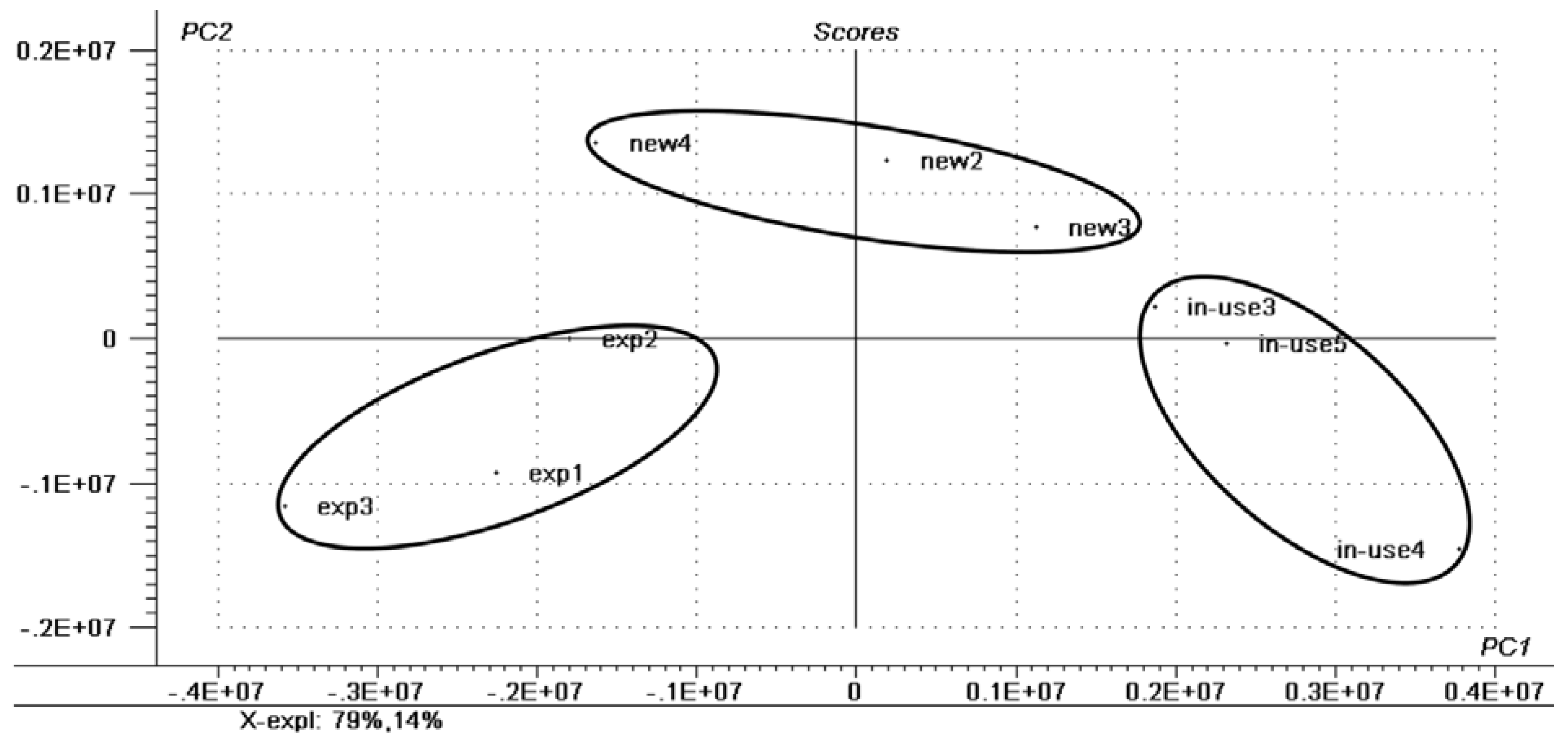
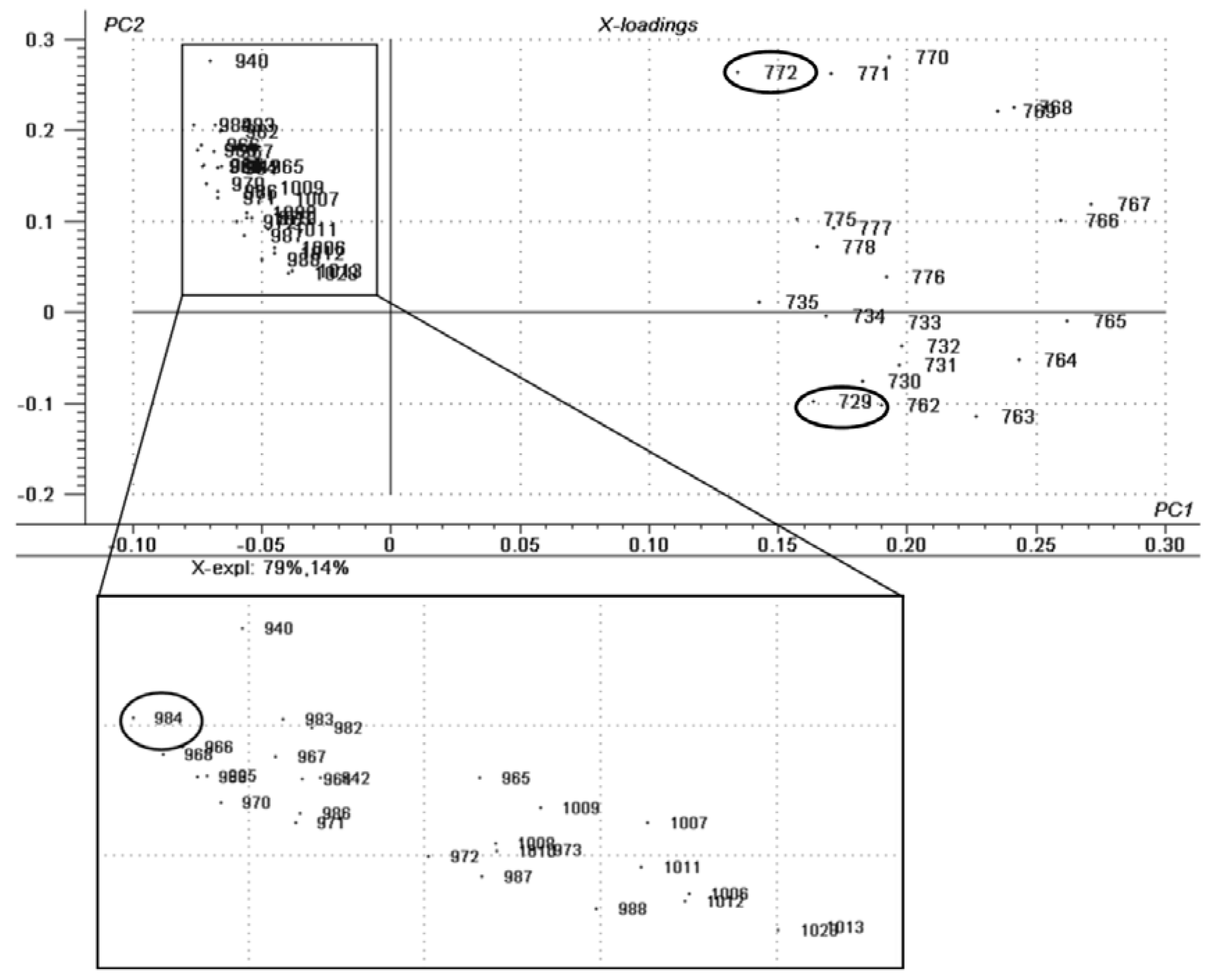
| SAMPLE | LIPID CLASS | CN:DB a | [M + Na]+ | CID Fragments m/z |
|---|---|---|---|---|
| m/z | ||||
| LIPSTICK—NEW | Ceramides | 38:1 | 1190 | 1146, 1045, 1001 |
| Ceramides | 36:1 | 1205 | 1161, 1060, 907 | |
| LIPSTICK—USED | Triacylglycerols | 58:6 | 958 | 914, 844, 660 |
| Triacylglycerols | 60:0 | 998 | 700, 419, 363 | |
| LIPSTICK—EXPIRED | Diacylglycerols | 28:6 | 524 | 480, 299, 270 |
| Diacylglycerols | 30:6 | 552 | 508, 299, 407 | |
| EYELINER—NEW | Triacylglycerols | 44:1 | 772 | 728, 627 |
| EYELINER—USED | Triacylglycerols | 41:2 | 729 | 685, 611, 567 |
| EYELINER—EXPIRED | Triacylglycerols | 59:0 | 984 | 966, 940, 839 |
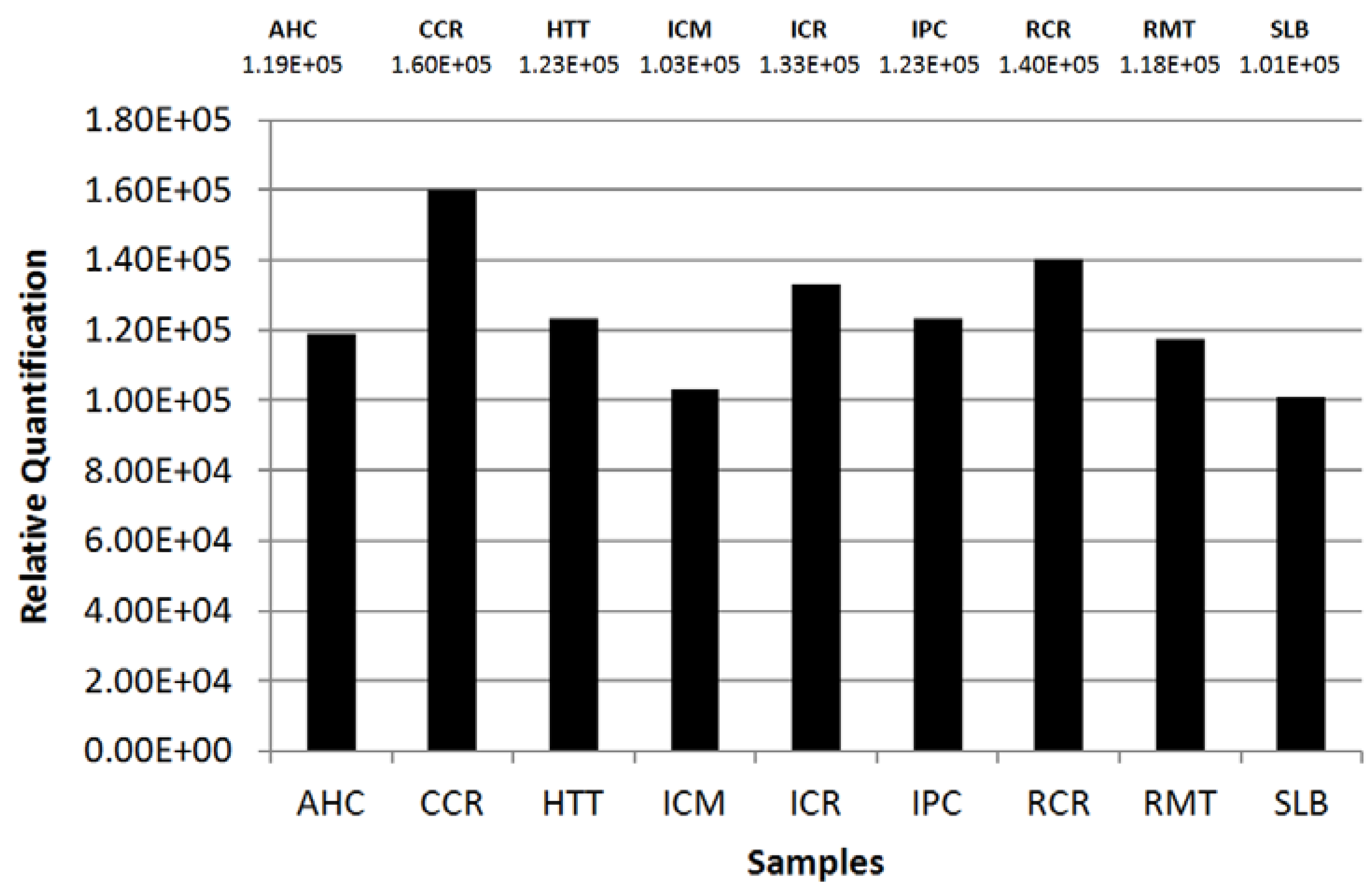
2.2. Quantification by Imaging
| Sample | Color | Aspect |
|---|---|---|
| AHC | Dark Blue | Creamy |
| CCR | Light Blue | Creamy-Glossy |
| HTT | Brown | Plain |
| ICM | Red | Metallic |
| ICR | Orange-Golden | Creamy |
| IPC | Orange | Plain |
| RCR | Red | Creamy |
| RMT | Pink | Metallic |
| SLB | Golden | Plain |

3. Experimental Section
3.1. Sample Preparation
3.2. MALDI Mass Spectrometry Imaging
3.3. Quantification by Mass Spectrometry Imaging (MSI)
3.4. Statistical Analysis of Data
4. Conclusions
Acknowledgments
References
- Brandt, F.S.; Cazzaniga, A.; Hann, M. Cosmeceuticals: Current trends and market analysis. Semin. Cutan. Med. Surg. 2011, 30, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S. Exploratory analysis of global cosmetic industry: Major players, technology and market trends. Technovation 2005, 25, 1263–1272. [Google Scholar] [CrossRef]
- Oh, C.H.; Rugman, A.M. Regional sales of multinationals in the world cosmetics industry. Eur. Manag. J. 2006, 24, 163–173. [Google Scholar] [CrossRef]
- Draelos, Z.D. Cosmeceuticals: Undefined, unclassified, and unregulated. Clin. Dermatol. 2009, 27, 431–434. [Google Scholar] [CrossRef] [PubMed]
- Doi, T.; Takeda, A.; Asada, A.; Kajimura, K. Characterization of the decomposition of compounds derived from imidazolidinyl urea in cosmetics and patch test materials. Contact Dermat. 2012, 67, 284–292. [Google Scholar] [CrossRef]
- Salvador, A.; Pascual-Marti, M.C.; Chisvert, A.; de la Ossa, M.D. News & views green analytical strategies for sunscreen control. Green Chem. 2002, 4, G55–G62. [Google Scholar] [CrossRef]
- Solon, E.G. Autoradiography: High-resolution molecular imaging in pharmaceutical discovery and development. Expert Opin. Drug Discov. 2007, 2, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Earnshaw, C.J.; Carolan, V.A.; Richards, D.S.; Clench, M.R. Direct analysis of pharmaceutical tablet formulations using matrix-assisted laser desorption/ionisation mass spectrometry imaging. Rapid Commun. Mass Spectrom. 2010, 24, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Hoshi, T.; Kudo, M. High resolution static SIMS imaging by time of flight SIMS. Appl. Surf. Sci. 2003, 203–204, 818–824. [Google Scholar] [CrossRef]
- Strick, R.; Strissel, P.L.; Gavrilov, K.; Levi-Setti, R. Cation-chromatin binding as shown by ion microscopy is essential for the structural integrity of chromosomes. J. Cell Biol. 2001, 155, 899–910. [Google Scholar] [CrossRef] [PubMed]
- Ifa, D.R.; Wiseman, J.M.; Song, Q.; Cooks, R.G. Development of capabilities for imaging mass spectrometry under ambient conditions with desorption electrospray ionization (DESI). Int. J. Mass Spectrom. 2007, 259, 8–15. [Google Scholar] [CrossRef]
- Caprioli, R.M.; Farmer, T.B.; Gile, J. Molecular imaging of biological samples—Localization of peptides and proteins using MALDI-TOF MS. Anal. Chem. 1997, 69, 4751–4760. [Google Scholar] [CrossRef] [PubMed]
- Reyzer, M.L.; Hsieh, Y.; Ng, K.; Korfmacher, W.A.; Caprioli, R.M. Direct analysis of drug candidates in tissue by matrix-assisted laser desorption/ionization mass spectrometry. J. Mass Spectrom. 2003, 38, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- Haddad, R.; Catharino, R.R.; Marques, L.A.; Eberlin, M.N. Perfume fingerprinting by easy ambient sonic-spray ionization mass spectrometry: Nearly instantaneous typification and counterfeit detection. Rapid Commun. Mass Spectrom. 2008, 22, 3662–3666. [Google Scholar] [CrossRef] [PubMed]
- Basheer, C.; Tan, S.H.; Lee, H.K. Extraction of lead ions by electromembrane isolation. J. Chromatogr. A 2008, 1213, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Gunduz, S.; Akman, S. Investigation of lead contents in lipsticks by solid sampling high resolution continuum source electrothermal atomic absorption spectrometry. Regul. Toxicol. Pharmacol. 2013, in press. [Google Scholar]
- Challinor, J.M. A rapid simple pyrolysis derivatisation gas chromatography-mass spectrometry method for profiling of fatty acids in trace quantities of lipids. J. Anal. Appl. Pyrolysis 1996, 37, 185–197. [Google Scholar] [CrossRef]
- Ahmed Refat, N.A.G.; Ibrahim, Z.S.; Moustafa, G.G.; Sakamoto, K.Q.; Ishizuka, M.; Fujita, S. The induction of cytochrome P450 1A1 by sudan dyes. J. Biochem. Mol. Toxicol. 2008, 22, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Grey, A.C.; Crouch, R.K.; Koutalos, Y.; Schey, K.L.; Ablonczy, Z. Spatial localization of A2E in the retinal pigment epithelium. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3926–3933. [Google Scholar] [CrossRef]
- Gassmann, M.; Grenacher, B.; Rohde, B.; Vogel, J. Quantifying Western blots: Pitfalls of densitometry. Electrophoresis 2009, 30, 1845–1855. [Google Scholar] [CrossRef] [PubMed]
- Okada, J.; Kanbe, R.; Kuzukawa, M.; Ikeda, Y.; Yoshimura, K.; Hayakawa, R.; Matsunaga, K. Identification of contact allergens in C.I. Solvent Red 23 (commercial Sudan III) by chemical analysis and animal testing. Contact Dermat. 1991, 25, 313–318. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Oliveira, D.N.; De Bona Sartor, S.; Ferreira, M.S.; Catharino, R.R. Cosmetic Analysis Using Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Imaging (MALDI-MSI). Materials 2013, 6, 1000-1010. https://doi.org/10.3390/ma6031000
De Oliveira DN, De Bona Sartor S, Ferreira MS, Catharino RR. Cosmetic Analysis Using Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Imaging (MALDI-MSI). Materials. 2013; 6(3):1000-1010. https://doi.org/10.3390/ma6031000
Chicago/Turabian StyleDe Oliveira, Diogo Noin, Sabrina De Bona Sartor, Mônica Siqueira Ferreira, and Rodrigo Ramos Catharino. 2013. "Cosmetic Analysis Using Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Imaging (MALDI-MSI)" Materials 6, no. 3: 1000-1010. https://doi.org/10.3390/ma6031000
APA StyleDe Oliveira, D. N., De Bona Sartor, S., Ferreira, M. S., & Catharino, R. R. (2013). Cosmetic Analysis Using Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry Imaging (MALDI-MSI). Materials, 6(3), 1000-1010. https://doi.org/10.3390/ma6031000




