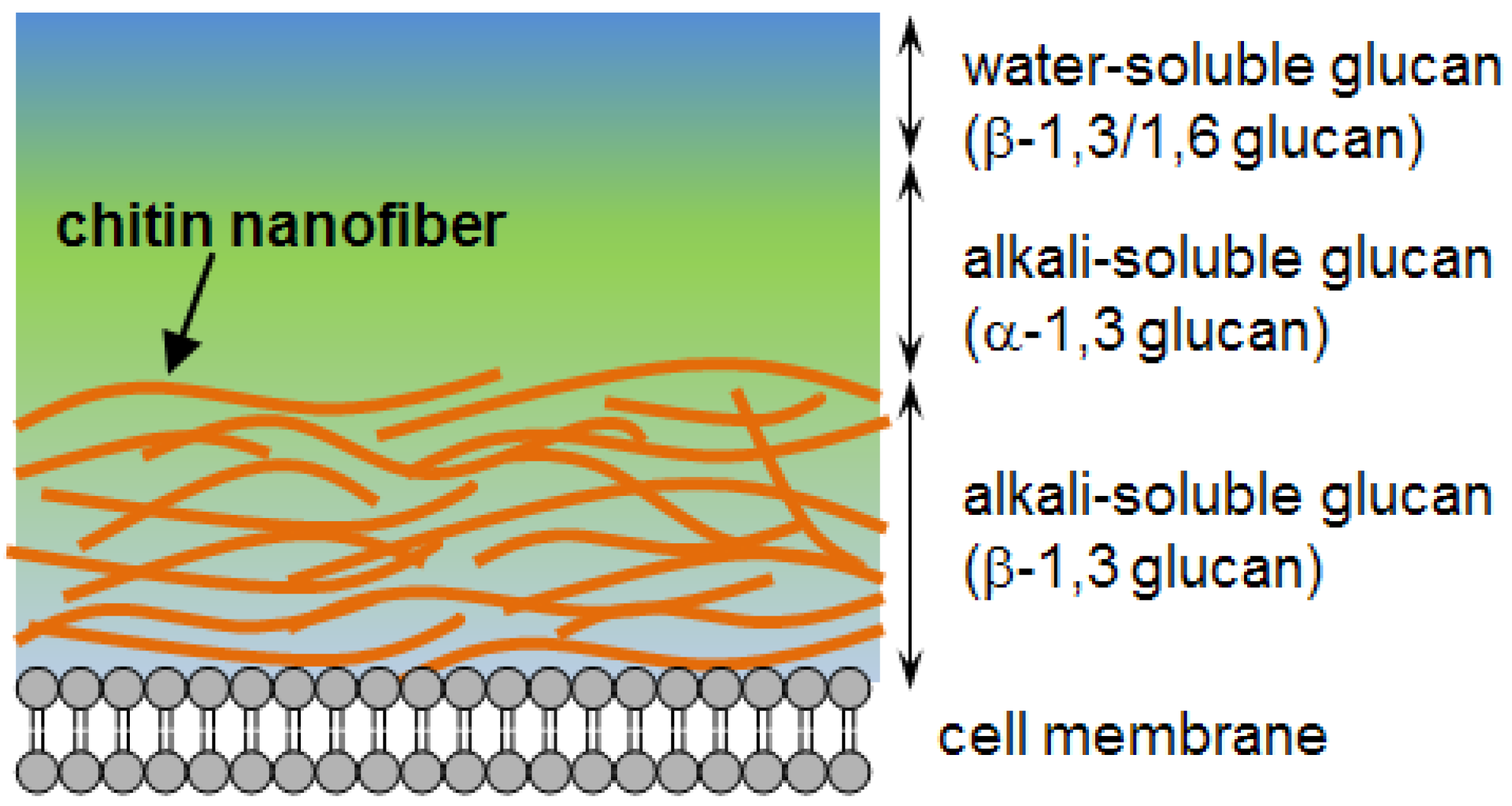
2.1. Preparation of Chitin Nanofibers from Mushrooms
Five different species of mushrooms:
Pleurotus eryngii,
Agaricus bisporus,
Lentinula edodes,
Grifola frondosa, and
Hypsizygus marmoreus widely used for human consumption were selected to isolate chitin nanofibers in this study. The chitin nanofibers were obtained from these mushrooms by a series of purification and chemical treatments described in
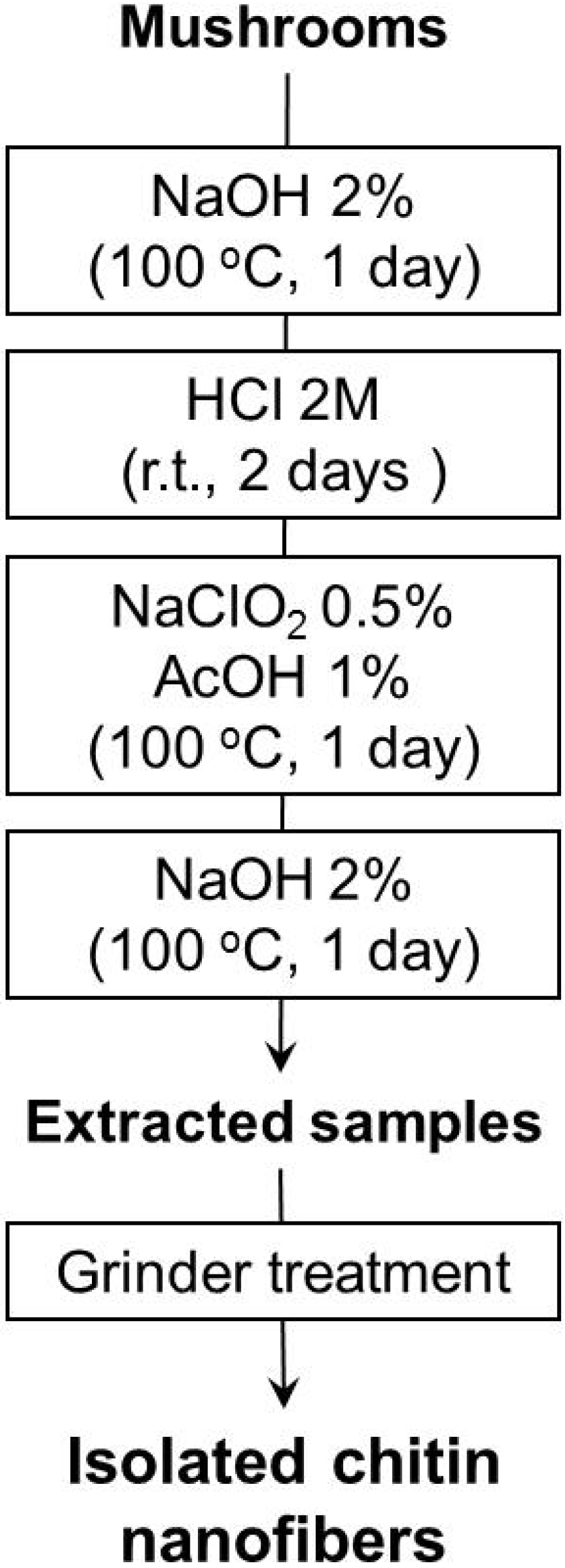
Figure 2 [
9] to remove associated components: proteins, pigments, glucans, and minerals. The procedure and treatments are as follows. At the first stage, sodium hydroxide was used to dissolve, hydrolyze, and remove proteins and alkali-soluble glucans. Hydrochloric acid was added to remove minerals. At this stage, it is known that partial neutral saccharides and acid-soluble protein compounds are also separated.
Figure 2.
Preparation procedure of chitin nanofibers from mushrooms.
Figure 2.
Preparation procedure of chitin nanofibers from mushrooms.
The extraction step with sodium chlorite and acetic acid can bleach the pigments in the sample. For the final stage, the sample was treated with sodium hydroxide again to eliminate and remove the residual glucans, including trace amounts of proteins. The drying process causes strong hydrogen bonding between chitin fibers after removal of matrix substances which makes it difficult to obtain chitin in the form of nanofibers [
10]. Therefore, the sample must be kept wet after removal of the matrices to obtain chitin nanofibers. The extracted sample with 1 %w/w of chitin was passed through a grinder for nano-fibrillation along with acetic acid. Acidic conditions are necessary for nanofiber preparation. The cationization of a small portion of amino groups on the chitin fiber causes electrostatic repulsion, which facilitate nano-fibrillation. Although it is difficult to measure the degree of deacetylation (DDA) of the extracted chitin since the sample contains glucans as mentioned below, the DDA is expected to be less than 5%. See reference to the previous paper [
5]. After grinder treatment, the chitin slurry had a high viscosity with a homogeneous and stable dispersibility indicating that the sample was successfully nano-fibrillated.
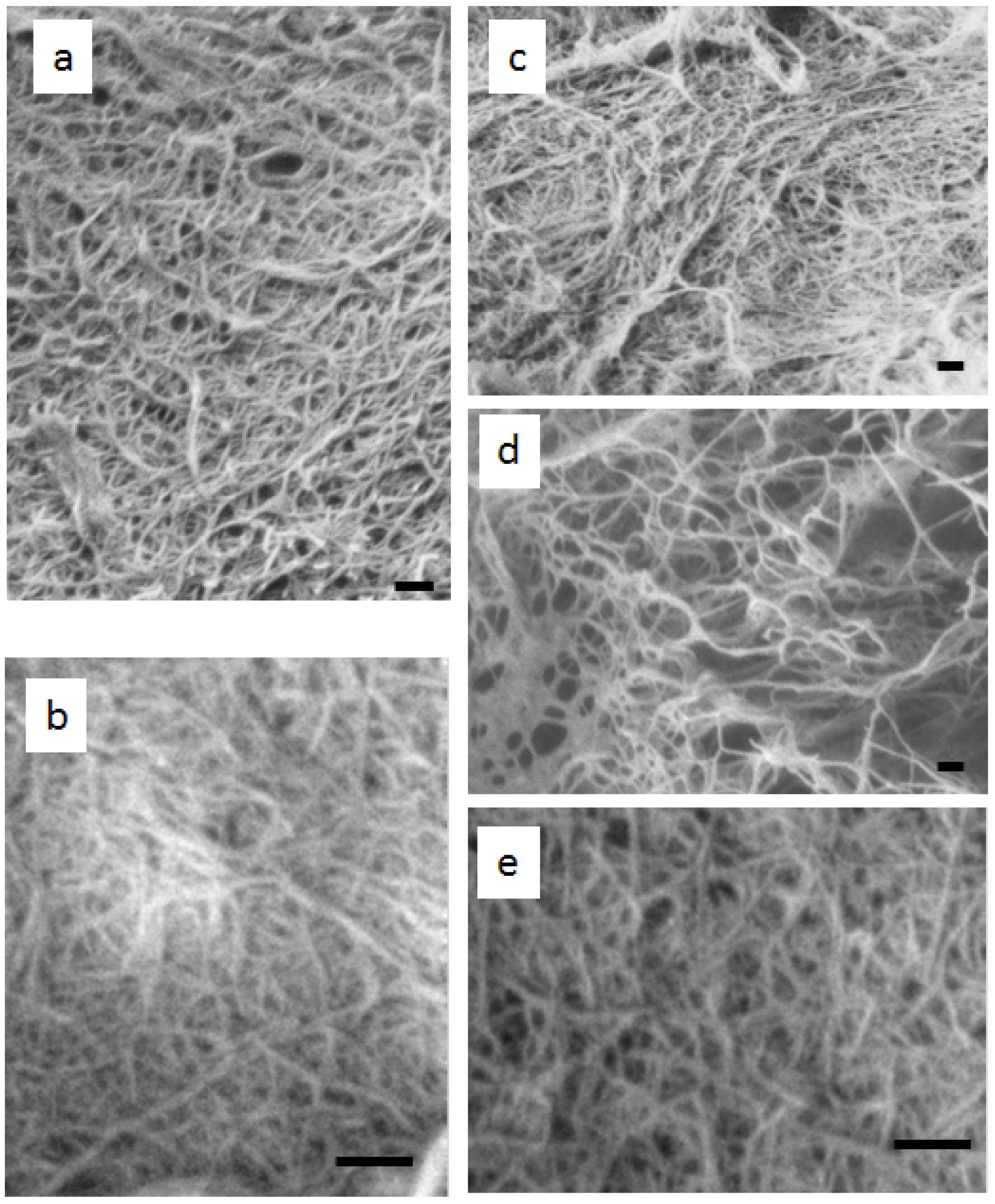
Figure 3 shows SEM images of chitin from five mushrooms after removing matrix components and one cycle of grinding. The isolated chitins are well-fibrillated and images show uniform nanofibers. The appearance of the fibers was similar to that of chitin nanofibers prepared from crab and prawn shells. Damaged fibers were not observed during a series of chemical and mechanical treatment. The lengths of the nanofibers are very long. The average width seems slightly different depending on the type of mushroom. This will be discussed in the latter part of the text. In this study, we have demonstrated that the preparation of chitin nanofibers from several varieties of mushrooms can be done similarly to the method used for crab and prawn shell. The chitin nanofiber slurries were not transparent, which was the same situation as for the chitin nanofibers from crab and prawn shell. This is because the nanofiber surface was hydrophobic compared with cellulose nanofiber, and very long chitin nanofibers in high concentration (1%) interacted with each other.
Figure 3.
FE-SEM micrographs of chitin nanofibers prepared from (a) Pleurotus eryngii (b) Agaricus bisporus (c) Lentinula edodes (d) Grifola frondosa (e) Hypsizygus marmoreus. The scale bars are 200 nm in length.
Figure 3.
FE-SEM micrographs of chitin nanofibers prepared from (a) Pleurotus eryngii (b) Agaricus bisporus (c) Lentinula edodes (d) Grifola frondosa (e) Hypsizygus marmoreus. The scale bars are 200 nm in length.
2.2. Characterization of Chitin Nanofibers
The N and C contents of chitin nanofibers determined by elemental analysis are listed in
Table 1. The N contents of all mushroom samples were smaller than the N content of commercial chitin macromolecules (6.89%). And the mushroom-to-commercial N content ratio varied widely with the type of mushrooms in the range 92.45–42.96%. The reason for low N atom contents in mushroom chitin is that there exists a chitin-glucan complex in mushrooms [
9]. Complete removal of glucan does not occur by chemical treatment of the materials that have decreased the N atomic ratio in mushroom chitin. However in the case of crabs and prawns, the matrix substances such as proteins and minerals were completely removed by the chemical treatment with NaOH and HCl solutions (<0.1%) [
11]. A similar example of incomplete removal of components is remnants of hemicelluloses in wood [
12]. Average fiber widths of chitin nanofibers are listed in
Table 1. The width varied depending on the type of mushroom. It is clear from the table that the lower the N content the wider are the nanofibers: 92.45 and 42.96% N containing nanofibers correspond to 20 and 28 nm width, respectively. This indicates that chitin nanofibers with low N content have a certain amount of glucans on the surface of the nanofiber as mentioned above and thus the thickness of the chitin nanofiber is increased.
Table 1.
Elemental analysis data, crystalline index, fiber width, and yield of chitin nanofibers.
Table 1.
Elemental analysis data, crystalline index, fiber width, and yield of chitin nanofibers.
| samples | Elemental analysis data | crystalline index | fiber width (nm) | yield (%) |
|---|
| N (%) | C (%) | N content ratio (%)a |
|---|
| Chitin | 6.89 | 47.29 | 100 | 88.5 | - | - |
| Cellulose | 0 | 44.44 | 0 | - | - | - |
| Pleurotus eryngii | 6.37 | 45.43 | 92.45 | 80.0 | 20 | 3.5 |
| Agaricus bisporus | 6.19 | 43.69 | 89.84 | 77.1 | 20 | 3.2 |
| Lentinula edodes | 4.89 | 45.30 | 70.97 | 65.0 | 22 | 1.3 |
| Grifola frondosa | 3.67 | 42.72 | 53.27 | 65.0 | 25 | 3.2 |
| Hypsizygus marmoreus | 2.96 | 43.77 | 42.96 | 47.6 | 28 | 1.8 |
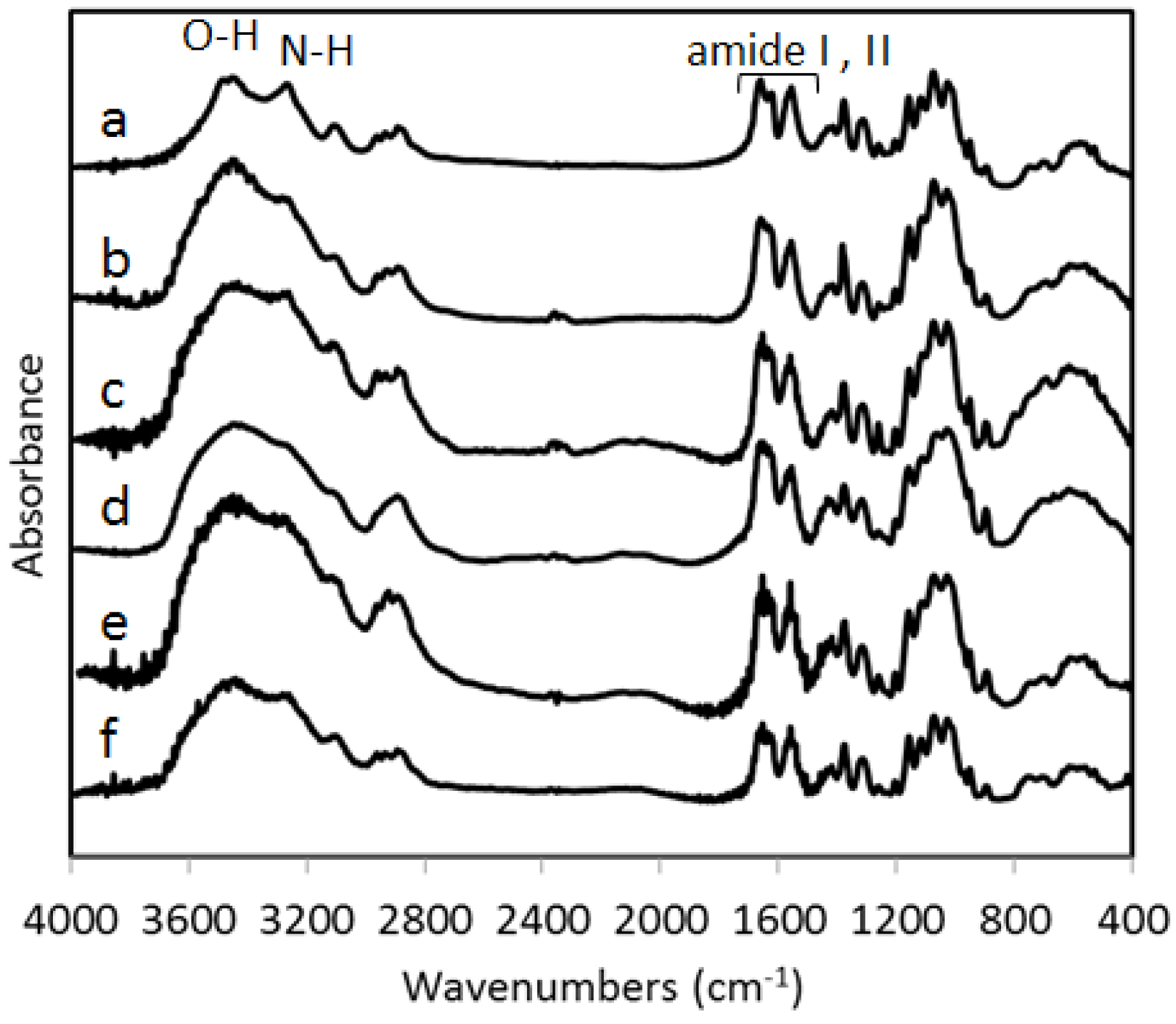
Figure 4 shows the FT-IR spectra (a) of commercial chitin obtained from crab shells and (b–f) are of chitin nanofibers prepared from five species of mushrooms following the method described in this article. The spectral bands (b–f) of chitin nanofibers of mushrooms correspond exactly and overlap with that of commercial pure chitin. The characteristic bands of the chitin molecule: O-H stretching (3450 cm
−1), N-H stretching (3270 cm
−1), amide I (1660 and 1620 cm
−1), and amide II (1560 cm
−1) are prominent bands observed in the FT-IR spectra of the chitin nanofibers and are similar to the commercial chitin.
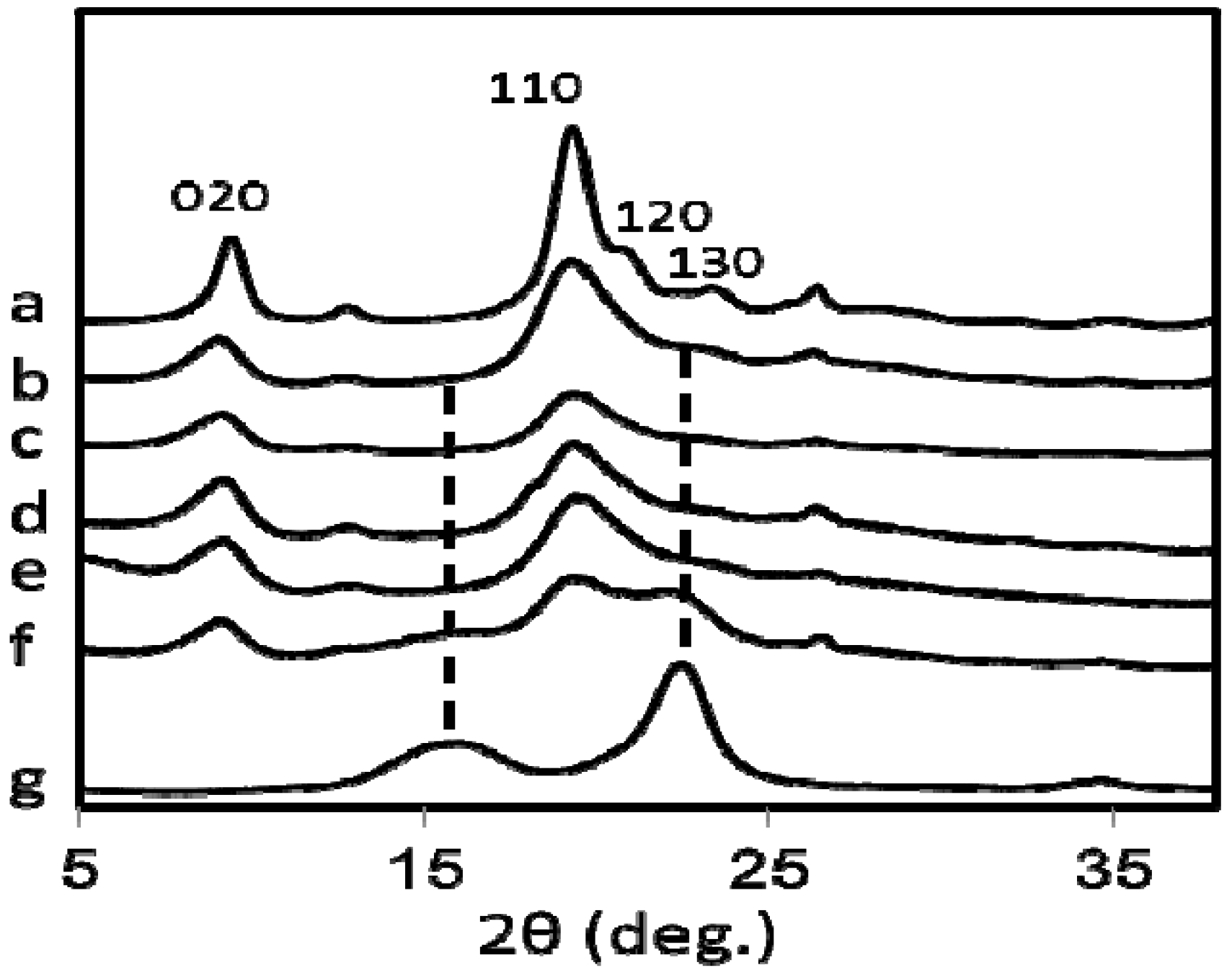
Figure 5 shows the X-ray diffraction pattern of commercially available chitin (a), (b–f) are the chitin bands of nanofibers prepared from all five species of mushrooms while the pattern (g) is of commercial cellulose. The four diffraction peaks of chitin nanofibers noticed at 9.4°, 19.3°, 20.6°, and 22.5° correspond to the 020, 110, 120, and 130 planes, respectively. They are typical crystal patterns of α-chitin. Thus, chitin nanofibers were extracted from several types of mushroom and the α-chitin crystalline structures were maintained after the removal of matrix substances followed by grinder treatment. However, in the case of
Hypsizygus marmoreus, which has the lowest N content (
Table 1), the X-ray diffractogram (
Figure 5f) contains crystal patterns of cellulose (
Figure 5g). The diffraction peaks observed from 15° to 17°, and 22.5°, corresponding to the 110, 1–10, and 200 planes, respectively are typical for the cellulose I crystal. Relative crystalline indices (CI) of chitin nanofibers were determined from the X-ray diffraction profiles and listed in
Table 1.
Figure 4.
FT-IR spectra of (a) commercially available chitin, and chitin nanofibers prepared from (b) Lentinula edodes (c) Pleurotus eryngii (d) Hypsizygus marmoreus (e) Grifola frondosa and (f) Agaricus bisporus.
Figure 4.
FT-IR spectra of (a) commercially available chitin, and chitin nanofibers prepared from (b) Lentinula edodes (c) Pleurotus eryngii (d) Hypsizygus marmoreus (e) Grifola frondosa and (f) Agaricus bisporus.
Figure 5.
X-ray diffraction profiles of (a) commercially available chitin, and chitin nanofibers prepared from (b) Pleurotus eryngii (c) Agaricus bisporus (d) Lentinula edodes (e) Grifola frondosa (f) Hypsizygus marmoreus and (g) commercially available cellulose.
Figure 5.
X-ray diffraction profiles of (a) commercially available chitin, and chitin nanofibers prepared from (b) Pleurotus eryngii (c) Agaricus bisporus (d) Lentinula edodes (e) Grifola frondosa (f) Hypsizygus marmoreus and (g) commercially available cellulose.
Commercial chitin has high crystallinity because the amorphous part is removed by acid hydrolysis in the purification process. Chitin nanofiber from Pleurotus Eryngii has the highest CI of 80.0%, which has the highest N content ratio. The indices decreased from 80.0 to 47.6%, with a reasonable correlation to N content ratios from 92.45 to 42.96%, respectively, indicating that the crystallinity also decreased with the increase of the amount of amorphous glucan on the surface of the nanofibers. The CI value would affect the mechanical properties of the chitin nanofiber such as Young’s modulus, fracture strength, and thermal expansion.