Stability Criteria of Fullerene-like Nanoparticles: Comparing V2O5 to Layered Metal Dichalcogenides and Dihalides
Abstract
:1. Introduction
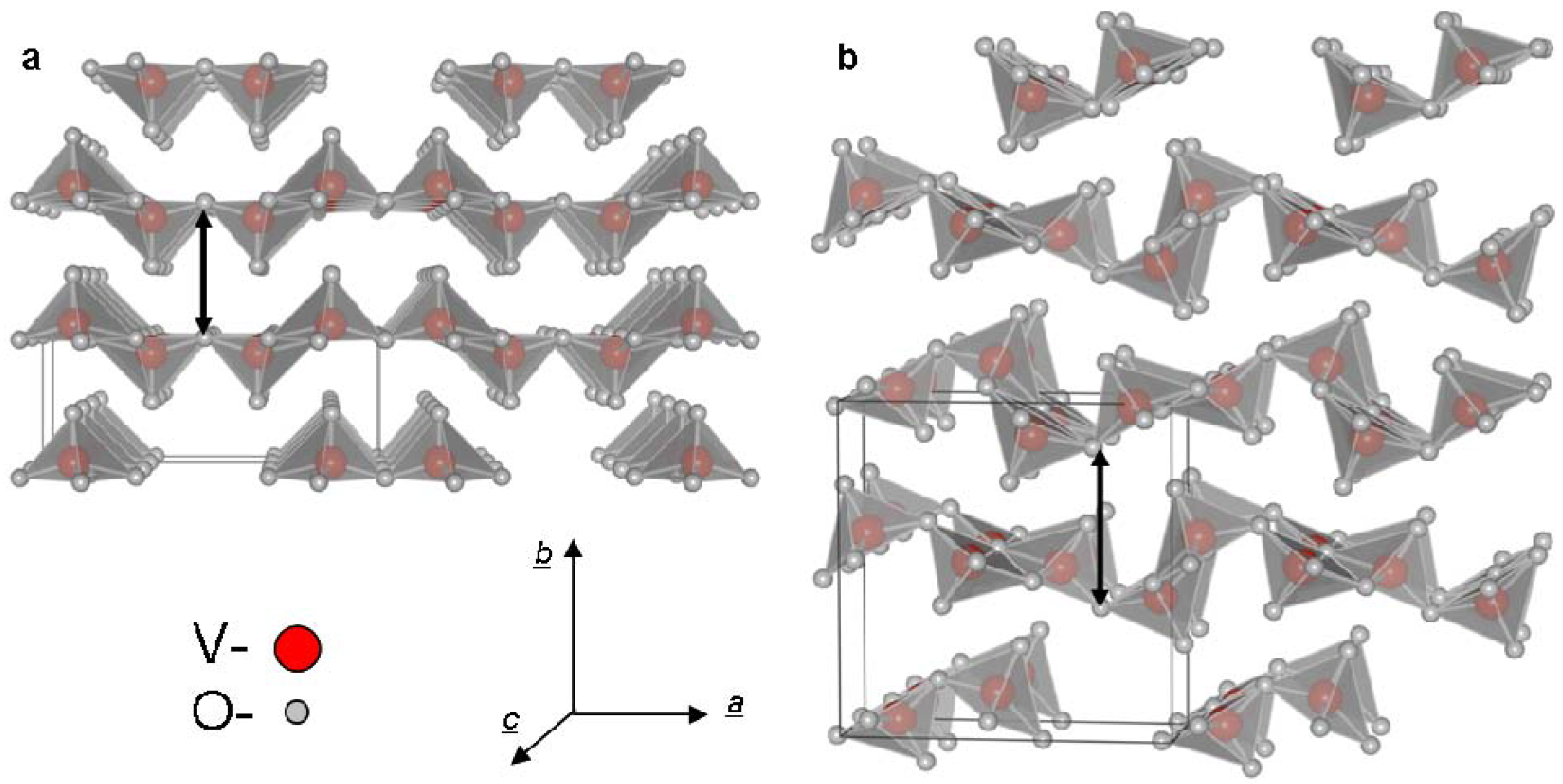
2. Results and Discussion
2.1. NIF-V2O5 characteristics and formation mechanism

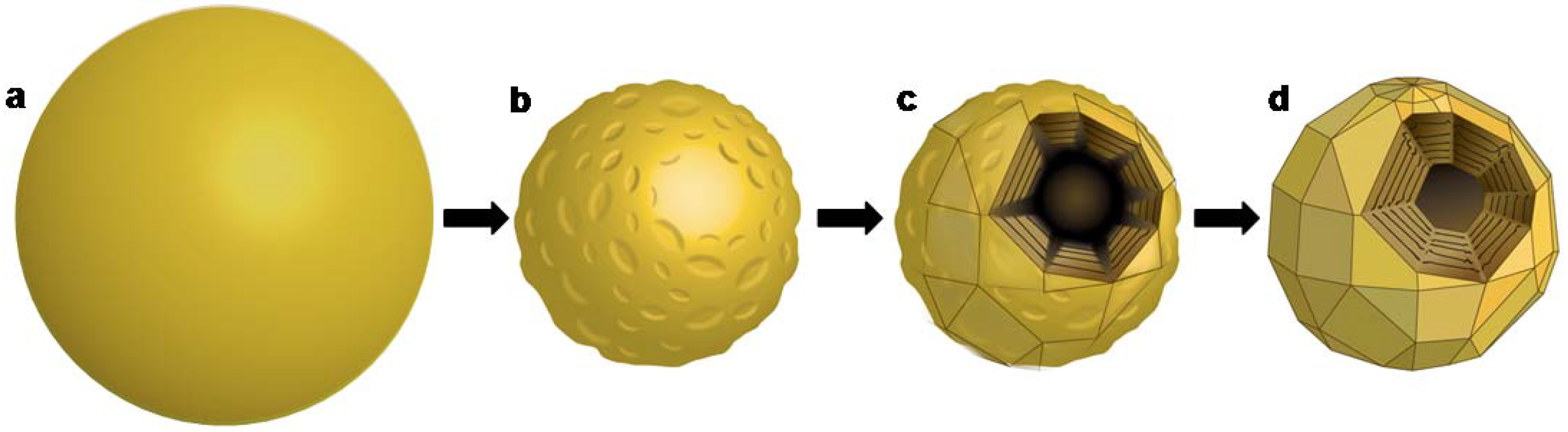
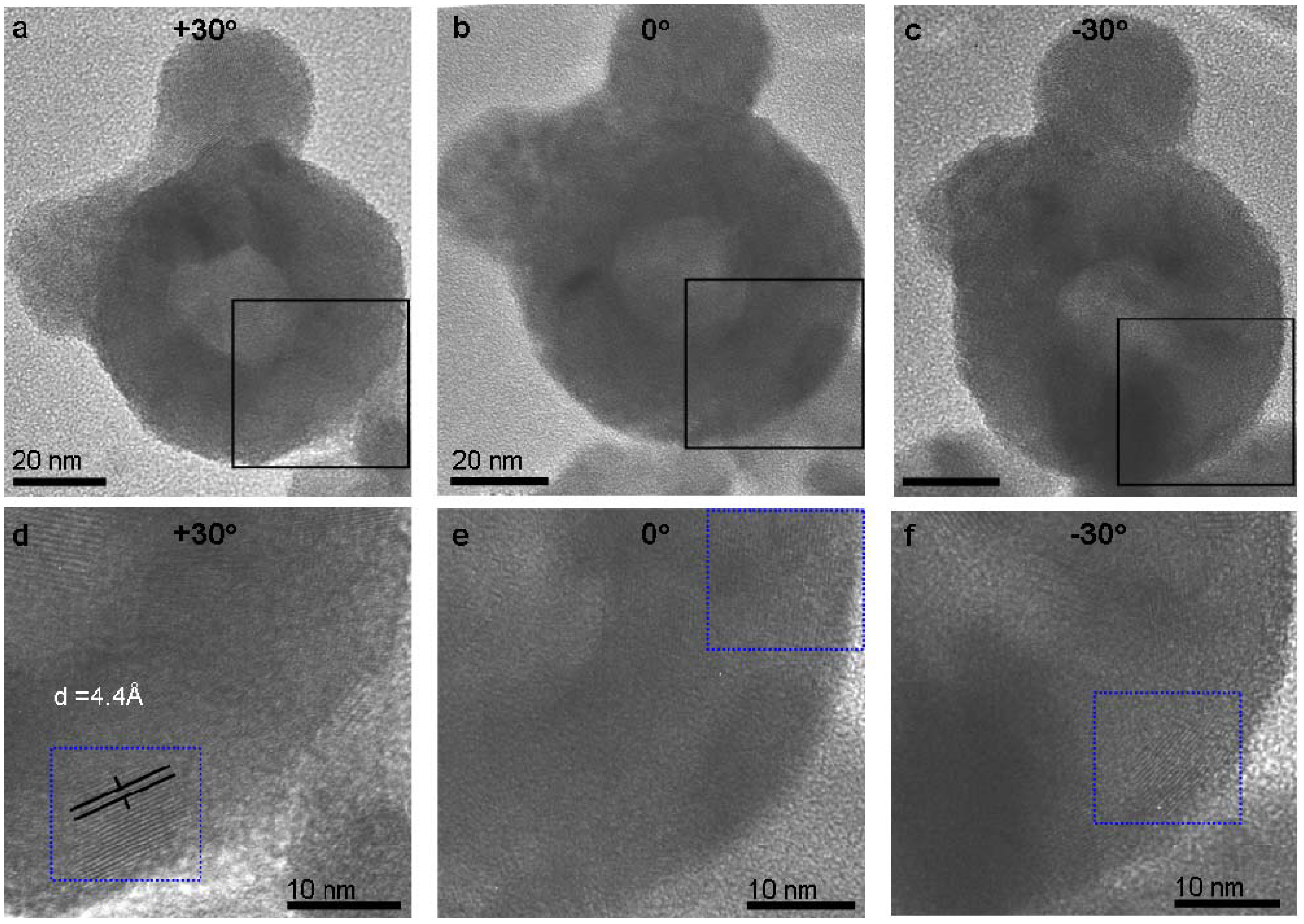

2.2. Post-reaction treatment

2.3. Comparison of seaming in IF and INT
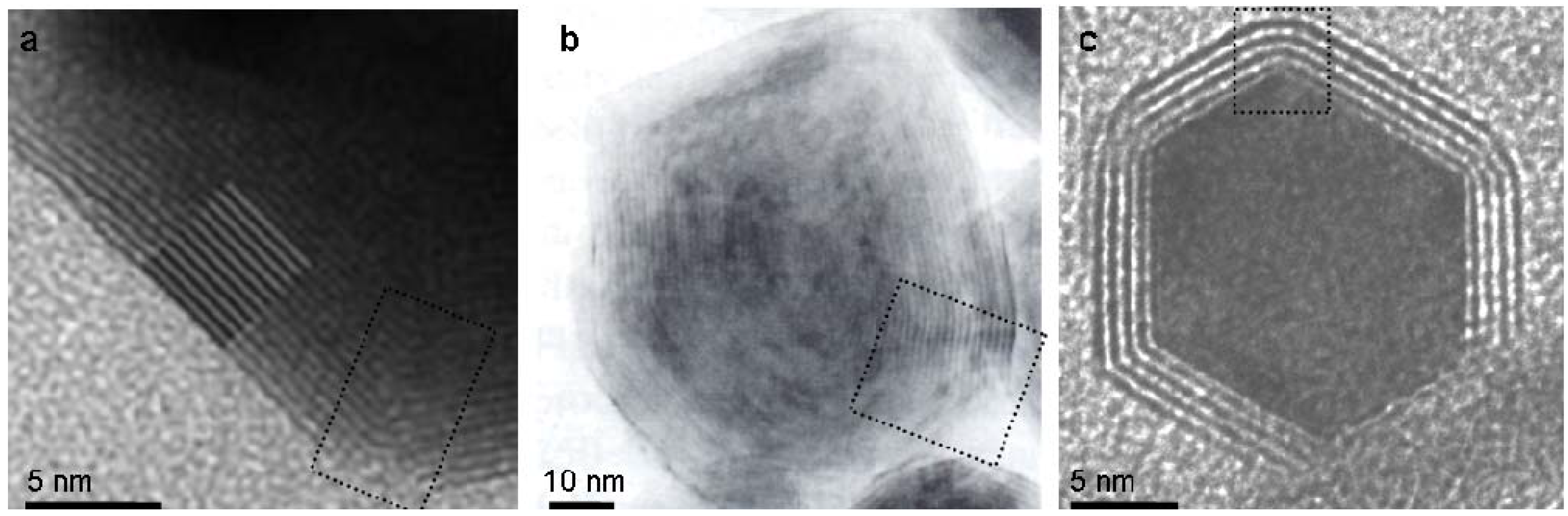
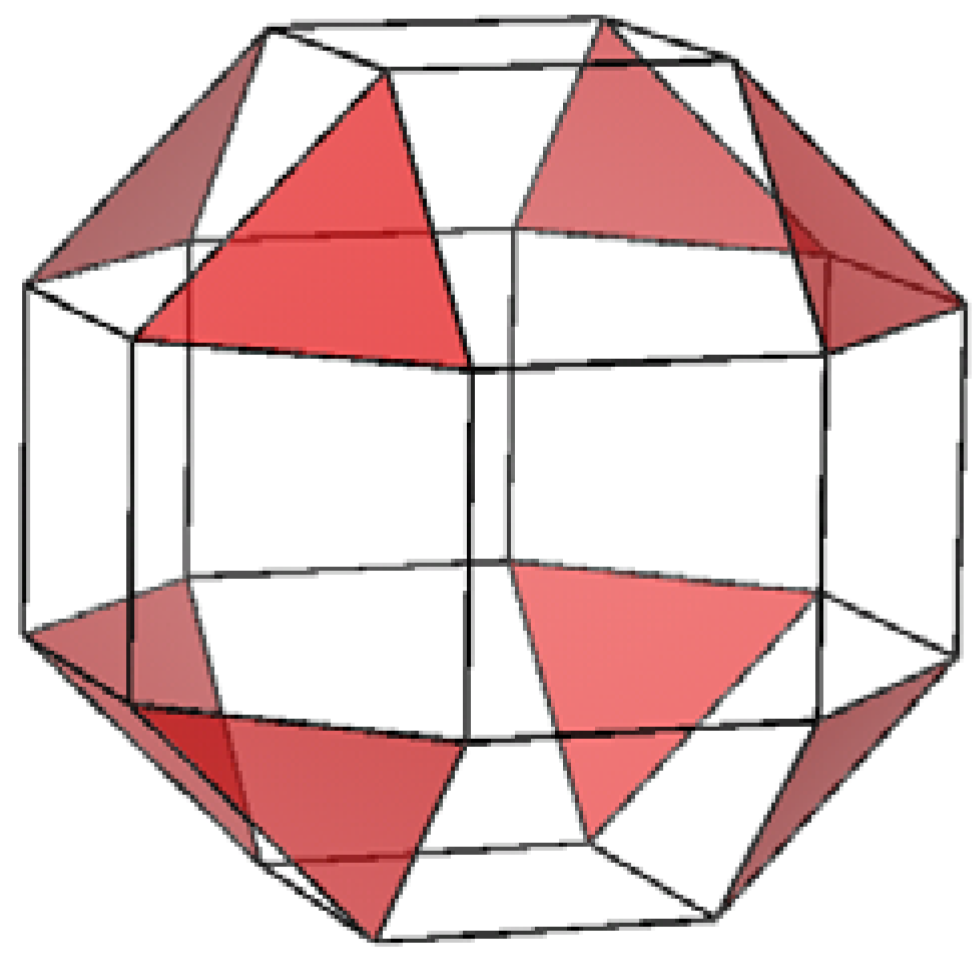
3. Experimental Section
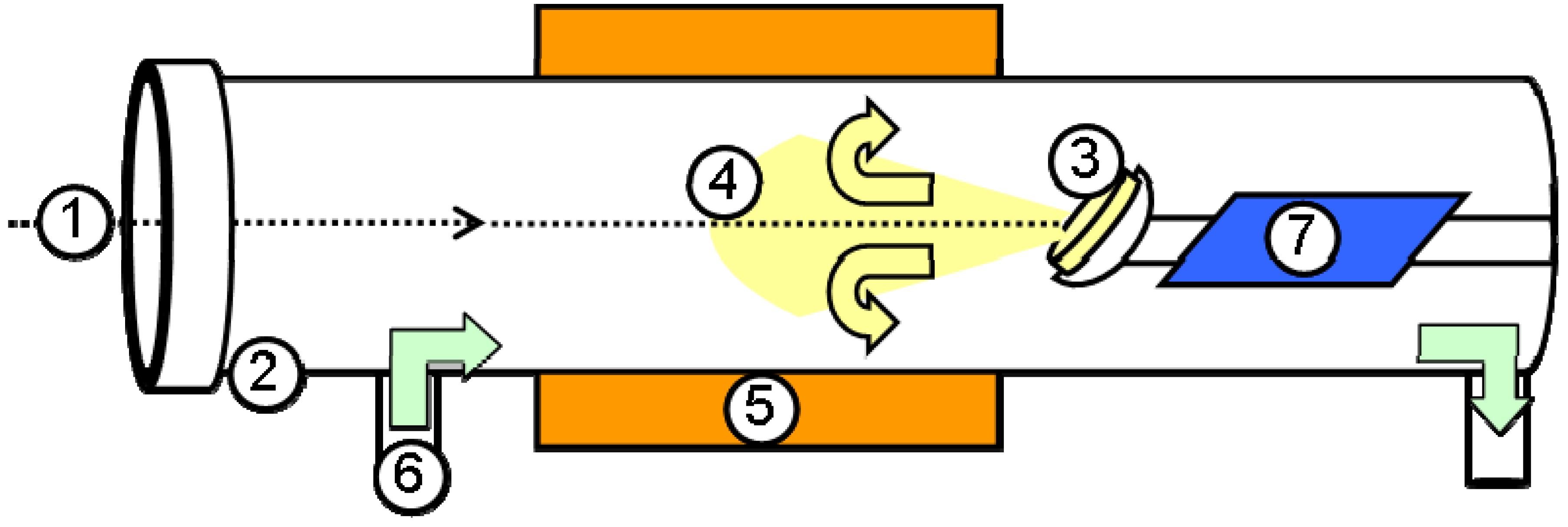
3.1. Laser ablation
3.2. Post-reaction treatment
3.3. Transmission electron microscopy (TEM)
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgements
References and Notes
- Tenne, R.; Seifert, G. Recent Progress in the Study of Inorganic Nanotubes and Fullerene-Like Structures. Ann. Rev. Mater. Res. 2009, 39, 387–413. [Google Scholar] [CrossRef]
- Rao, C.N.R.; Achutharao, G. Synthesis of Inorganic Nanotubes. J. Adv. Mater. 2009, 21, 4208–4233. [Google Scholar] [CrossRef]
- Tenne, R.; Margulis, L.; Genut, M.; Hodes, G. Polyhedral and Cylindrical Structures of Tungsten Disulfide. Nature 1992, 360, 444–446. [Google Scholar] [CrossRef]
- Seifert, G.; Terrones, H.; Terrones, M.; Jungnickel, G.; Frauenheim, T. Structure and Electronic Properties of MoS2 Nanotubes. Phys. Rev. Lett. 2000, 85, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Enyashin, A.N.; Gemming, S.; Bar-Sadan, M.; Popovitz-Biro, R.; Hong, S.Y.; Prior, Y.; Tenne, R.; Seifert, G. Structure and Stability of Molybdenum Sulfide Fullerenes. Angew. Chem-Int. Ed. 2007, 46, 623–627. [Google Scholar] [CrossRef]
- Popovitz-Biro, R.; Sallacan, N.; Tenne, R. CdI2 nanoparticles with closed-cage (fullerene-like) structures. J. Mater. Chem. 2003, 13, 1631–1634. [Google Scholar] [CrossRef]
- Albu-Yaron, A.; Arad, T.; Popovitz-Biro, R.; Bar-Sadan, M.; Prior, Y.; Jansen, M.; Tenne, R. Preparation and structural characterization of stable Cs2O closed-cage structures. Angew. Chem-Int. Ed. 2005, 44, 4169–4172. [Google Scholar] [CrossRef]
- Popovitz-Biro, R.; Twersky, A.; Hacohen, Y.R.; Tenne, R. Nanoparticles of CdCl2 with closed cage structures. Isr. J. Chem. 2000, 41, 7–14. [Google Scholar]
- Singh, P.; Kaur, D. Influence of film thickness on texture and electrical and optical properties of room temperature deposited nanocrystalline V2O5 thin films. J. Appl. Phys. 2008, 103, 043507–043509. [Google Scholar] [CrossRef]
- Yan, B.; Liao, L.; You, Y.M.; Xu, X.J.; Zheng, Z.; Shen, Z.X.; Ma, J.; Tong, L.M.; Yu, T. Single-Crystalline V2O5 Ultralong Nanoribbon Waveguides. J. Adv. Mater. 2009, 21, 2436–2440. [Google Scholar] [CrossRef]
- Ng, S.H.; Patey, T.J.; Buechel, R.; Krumeich, F.; Wang, J.Z.; Liu, H.K.; Pratsinis, S.E.; Novak, P. Flame spray-pyrolyzed vanadium oxide nanoparticles for lithium battery cathodes. Phys. Chem. Chem. Phys. 2009, 11, 3748–3755. [Google Scholar] [CrossRef] [PubMed]
- Asim, N.; Radiman, S.; Yarmo, M.A.; Golriz, M.S.B. Vanadium pentoxide: Synthesis and characterization of nanorod and nanoparticle V2O5 using CTAB micelle solution. Microporous Mesoporous Mat. 2009, 120, 397–401. [Google Scholar] [CrossRef]
- Penner, S.; Klotzer, B.; Jenewein, B. Structural and redox properties of VOx and Pd/VOx thin film model catalysts studied by TEM and SAED. Phys. Chem. Chem. Phys. 2007, 9, 2428–2433. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.M.; Zhang, X.M.; Qiao, Z.P.; Lei, D.M. Preparation and characterization of nanocrystal V2O5. J. Solid State Chem. 2001, 159, 181–185. [Google Scholar] [CrossRef]
- Menezes, W.G.; Reis, D.M.; Benedetti, T.M.; Oliveira, M.M.; Soares, J.F.; Torresi, R.M.; Zarbin, A.J.G. V2O5 nanoparticles obtained from a synthetic bariandite-like vanadium oxide: Synthesis, characterization and electrochemical behavior in an ionic liquid. J. Colloid Interface Sci. 2009, 337, 586–593. [Google Scholar] [CrossRef] [PubMed]
- Ding, N.; Liu, S.H.; Chen, C.H.; Lieberwirth, I. An electron microscopic investigation of structural variation of V2O5 fibers after working as ethanol sensors. Appl. Phys. Lett. 2008, 93, 1735101–1735103. [Google Scholar]
- Fang, G.J.; Liu, Z.L.; Wang, Y.; Liu, Y.H.; Yao, K.L. Synthesis and structural, electrochromic characterization of pulsed laser deposited vanadium oxide thin films. J. Vac. Sci. Technol. A 2001, 19, 887–892. [Google Scholar] [CrossRef]
- Hu, Y.; Li, Z.C.; Zhang, Z.J.; Meng, D.Q. Effect of magnetic field on the visible light emission of V2O5 nanorods. Appl. Phys. Lett. 2009, 94, 1031071–1031073. [Google Scholar]
- Umadevi, P.; Nagendra, C.L.; Thutupalli, G.K.M. Structural, electrical and infrared optical properties of vanadium pentoxide (V2O5) thick-film thermistors. Sensor. Actuator. A-Phys. 1993, 39, 59–69. [Google Scholar] [CrossRef]
- Beke, S.; Giorgio, S.; Korosi, L.; Nanai, L.; Marine, W. Structural and optical properties of pulsed laser deposited V2O5 thin films. Thin Solid Films 2008, 516, 4659–4664. [Google Scholar] [CrossRef]
- Cocciantelli, J.M.; Gravereau, P.; Doumerc, J.P.; Pouchard, M.; Hagenmuller, P. On the Preparation and Characterization of a New Polymorph of V2O5. J. Solid State Chem. 1991, 93, 497–502. [Google Scholar] [CrossRef]
- Ikemiya, N.; Umemoto, J.; Hara, S.; Ogino, K. Surface tensions and densities of molten Al2O3, Ti2O3, V2O5 and Nb2O5. ISIJ Int. 1993, 33, 156–165. [Google Scholar] [CrossRef]
- Rivoalen, L.; Revcolevschi, A.; Livage, J.; Collongues, R. Amorphous Vanadium Pentoxide. J. Non-Cryst. Solids 1976, 21, 171–179. [Google Scholar] [CrossRef]
- Benmoussa, M.; Ibnouelghazi, E.; Bennouna, A.; Ameziane, E.L. Structural, Electrical and Optical-Properties of Sputtered Vanadium Pentoxide Thin-Films. Thin Solid Films 1995, 265, 22–28. [Google Scholar] [CrossRef]
- Teller, R.G.; Antonio, M.R.; Brazdil, J.F.; Grasselli, R.K. New Materials Synthesis—Characterization of Some Metal-Doped Antimony Oxides. J. Solid State Chem. 1986, 64, 249–260. [Google Scholar] [CrossRef]
- Witko, M.; Hermann, K.; Tokarz, R. Adsorption and reactions at the (0 1 0) V2O5 surface: cluster model studies. Catal. Today 1999, 50, 553–565. [Google Scholar] [CrossRef]
- Wang, Y.; Takahashi, K.; Lee, K.; Cao, G.Z. Nanostructured vanadium oxide electrodes for enhanced lithium-ion intercalation. Adv. Funct. Mater. 2006, 16, 1133–1144. [Google Scholar] [CrossRef]
- Petkov, V.; Trikalitis, P.N.; Bozin, E.S.; Billinge, S.J.L.; Vogt, T.; Kanatzidis, M.G. Structure of V2O5.nH2O xerogel solved by the atomic pair distribution function technique. J. Am.Chem. Soc. 2002, 124, 10157–10162. [Google Scholar] [CrossRef] [PubMed]
- Levi, R.; Bar-Sadan, M.; Albu-Yaron, A.; Popovitz-Biro, R.; Houben, L.; Shahar, C.; Enyashin, A.; Seifert, G.; Prior, Y.; Tenne, R. Hollow V2O5 Nanoparticles (Fullerene-Like Analogues) Prepared by Laser Ablation. J. Am. Chem. Soc. Available online: http://pubs.acs.org/doi/abs/10.1021/ja103719x (accessed on 22 July 2010).
- Spahr, M.E.; Bitterli, P.; Nesper, R.; Muller, M.; Krumeich, F.; Nissen, H.U. Redox-active nanotubes of vanadium oxide. Angew. Chem.-Int. Ed. 1998, 37, 1263–1265. [Google Scholar] [CrossRef]
- Zhou, F.; Zhao, X.M.; Liu, Y.Q.; Yuan, C.G.; Li, L. Synthesis of millimeter-range orthorhombic V2O5 nanowires and impact of thermodynamic and kinetic properties of the oxidant on the synthetic process. Eur. J. Inorg. Chem. 2008, 16, 2506–2509. [Google Scholar] [CrossRef]
- Chou, S.L.; Wang, J.Z.; Sun, J.Z.; Wexler, D.; Forsyth, M.; Liu, H.K.; MacFarlane, D.R.; Dou, S.X. High Capacity, Safety, and Enhanced Cyclability of Lithium Metal Battery Using a V2O5 Nanomaterial Cathode and Room Temperature Ionic Liquid Electrolyte. Chem. Mater. 2008, 20, 7044–7051. [Google Scholar] [CrossRef]
- Ajayan, P.M.; Stephan, O.; Redlich, P.; Colliex, C. Carbon Nanotubes as Removable Templates for Metal-Oxide Nanocomposites and Nanostructures. Nature 1995, 375, 564–567. [Google Scholar] [CrossRef]
- Pinna, N.; Wild, U.; Urban, J.; Schlogl, R. Divanadium pentoxide nanorods. J. Adv. Mater . 2003, 15, 329–331. [Google Scholar] [CrossRef]
- Satishkumar, B.C.; Govindaraj, A.; Nath, M.; Rao, C.N.R. Synthesis of metal oxide nanorods using carbon nanotubes as templates. J. Mater. Chem. 2000, 10, 2115–2119. [Google Scholar] [CrossRef]
- Kim, J.H.; Hong, Y.C.; Uhm, H.S. Synthesis of oxide nanoparticles via microwave plasma decomposition of initial materials. Surf. Coat. Tech. 2007, 201, 5114–5120. [Google Scholar] [CrossRef]
- Bi, X.; Gardner, J. T.; Kumar, S.; Kambe, N. Processing of vanadium oxide particles with heat. U.S. Patent 5,589,514, 23 Nov 1999. [Google Scholar]
- Cao, A.M.; Hu, J.S.; Liang, H.P.; Wan, L.J. Self-assembled vanadium pentoxide (V2O5) hollow microspheres from nanorods and their application in lithium-ion batteries. Angew.Chem.-Int. Ed. 2005, 44, 4391–4395. [Google Scholar] [CrossRef]
- Nanai, L.; George, T.F. Laser-assisted formation of metallic oxide microtubes. Int. J. Mater. Res. 1997, 12, 283–284. [Google Scholar] [CrossRef]
- Lakshmi, B.B.; Patrissi, C.J.; Martin, C.R. Sol-Gel Template Synthesis of Semiconductor Oxide Micro- and Nanostructures. Chem. Mater. 1997, 9, 2544–2550. [Google Scholar] [CrossRef]
- Hacohen, Y.R.; Popovitz-Biro, R.; Prior, Y.; Gemming, S.; Seifert, G.; Tenne, R. Synthesis of NiCl2 nanotubes and fullerene-like structures by laser ablation: theoretical considerations and comparison with MoS2 nanotubes. Phys. Chem. Chem. Phys. 2003, 5, 1644–1651. [Google Scholar] [CrossRef]
- Parilla, P.A.; Dillon, A.C.; Jones, K.M.; Riker, G.; Schulz, D.L.; Ginley, D.S.; Heben, M.J. The first true inorganic fullerenes? Nature 1999, 397, 114–114. [Google Scholar] [CrossRef]
- Enyashin, A.N.; Ivanovskaya, V.V.; Makurin, Y.N.; Ivanovskii, A.L. Electronic band structure of scroll-like divanadium pentoxide nanotubes. Phys. Lett. A 2004, 326, 152–156. [Google Scholar] [CrossRef]
- Russo, R.E.; Mao, X.; Mao, S.S. The physics of laser ablation in microchemical analysis. Anal. Chem. 2002, 74, 70–77. [Google Scholar] [CrossRef]
- Russo, R.E. Laser Ablation. Appl. Spectrosc. 1995, 49, 14A–28A. [Google Scholar] [CrossRef]
- Nath, M.; Rao, C.N.R.; Popovitz-Biro, R.; Albu-Yaron, A.; Tenne, R. Nanoparticles Produced by Laser Ablation of HfS3 in Liquid Medium: Inorganic Fullerene-Like Structures of Hf2S. Chem. Mater. 2004, 16, 2238–2243. [Google Scholar] [CrossRef]
- Liu, Z.; Fang, G.; Wang, Y.; Bai, Y.; Yao, K.L. Laser-induced colouration of V2O5. J. Phys. D-Appl. Phys. 2000, 33, 2327–2332. [Google Scholar]
- Michailovits, L.; Bali, K.; Szorenyi, T.; Hevesi, I. Characterization of amorphous vanadium pentoxide thin films prepared by chemical vapour deposition (CVD) and vacuum deposition. Acta phys. Acad. Sci. Hung. 1980, 49, 217–221. [Google Scholar] [CrossRef]
- Mosset, A.; Lecante, P.; Galy, J.; Livage, J. Structural analysis of amorphous V2O5 by large-angle X-ray scattering. Phil. Mag. B 1982, 46, 137–149. [Google Scholar] [CrossRef]
- Feldman, Y.; Frey, G.L.; Homyonfer, M.; Lyakhovitskaya, V.; Margulis, L.; Cohen, H.; Hodes, G.; Hutchison, J.L.; Tenne, R. Bulk synthesis of inorganic fullerene-like MS2 (M=Mo, W) from the respective trioxides and the reaction mechanism. J. Am. Chem. Soc. 1996, 118, 5362–5367. [Google Scholar] [CrossRef]
- Ramana, C.V.; Smith, R.J.; Hussain, O.M.; Julien, C.M. On the growth mechanism of pulsed-laser deposited vanadium oxide thin films. Mater. Sci. Eng. B-Solid State M 2004, 111, 218–225. [Google Scholar] [CrossRef]
- Garcia, A.; Cohen, M.L. First-principles ionicity scales. I. Charge asymmetry in the solid state. Phys. Rev. B 1993, 47, 4215–4220. [Google Scholar] [CrossRef]
- Zak, A.; Sallacan-Ecker, L.; Margolin, A.; Genut, M.; Tenne, R. Insight into the Growth Mechanism of WS2 Nanotubes in the Scaled-Up Fluidized Bed Reactor. Nano 2009, 4, 91–98. [Google Scholar] [CrossRef]
- Patzke, G.R.; Krumeich, F.; Nesper, R. Oxidic nanotubes and nanorods-anisotropic modules for a future nanotechnology. Angew. Chem. Int. Ed. 2002, 41, 2446–2461. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Hermann, K.; Druzinic, R.; Witko, M.; Wagner, F.; Petersen, M. Geometric and electronic structure of vanadium pentoxide: A density functional bulk and surface study. Phys. Rev. B 1999, 59, 10583–10590. [Google Scholar] [CrossRef]
- Parker, J.C.; Lam, D.J.; Xu, Y.N.; Ching, W.Y. Optical-Properties of Vanadium Pentoxide Determined from Ellipsometry and Band-Structure Calculations. Phys. Rev. B 1990, 42, 5289–5293. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Levi, R.; Bar-Sadan, M.; Albu-Yaron, A.; Popovitz-Biro, R.; Houben, L.; Prior, Y.; Tenne, R. Stability Criteria of Fullerene-like Nanoparticles: Comparing V2O5 to Layered Metal Dichalcogenides and Dihalides. Materials 2010, 3, 4428-4445. https://doi.org/10.3390/ma3084428
Levi R, Bar-Sadan M, Albu-Yaron A, Popovitz-Biro R, Houben L, Prior Y, Tenne R. Stability Criteria of Fullerene-like Nanoparticles: Comparing V2O5 to Layered Metal Dichalcogenides and Dihalides. Materials. 2010; 3(8):4428-4445. https://doi.org/10.3390/ma3084428
Chicago/Turabian StyleLevi, Roi, Maya Bar-Sadan, Ana Albu-Yaron, Ronit Popovitz-Biro, Lothar Houben, Yehiam Prior, and Reshef Tenne. 2010. "Stability Criteria of Fullerene-like Nanoparticles: Comparing V2O5 to Layered Metal Dichalcogenides and Dihalides" Materials 3, no. 8: 4428-4445. https://doi.org/10.3390/ma3084428
APA StyleLevi, R., Bar-Sadan, M., Albu-Yaron, A., Popovitz-Biro, R., Houben, L., Prior, Y., & Tenne, R. (2010). Stability Criteria of Fullerene-like Nanoparticles: Comparing V2O5 to Layered Metal Dichalcogenides and Dihalides. Materials, 3(8), 4428-4445. https://doi.org/10.3390/ma3084428






