Nanocomposites Derived from Polymers and Inorganic Nanoparticles
Abstract
:1. Introduction
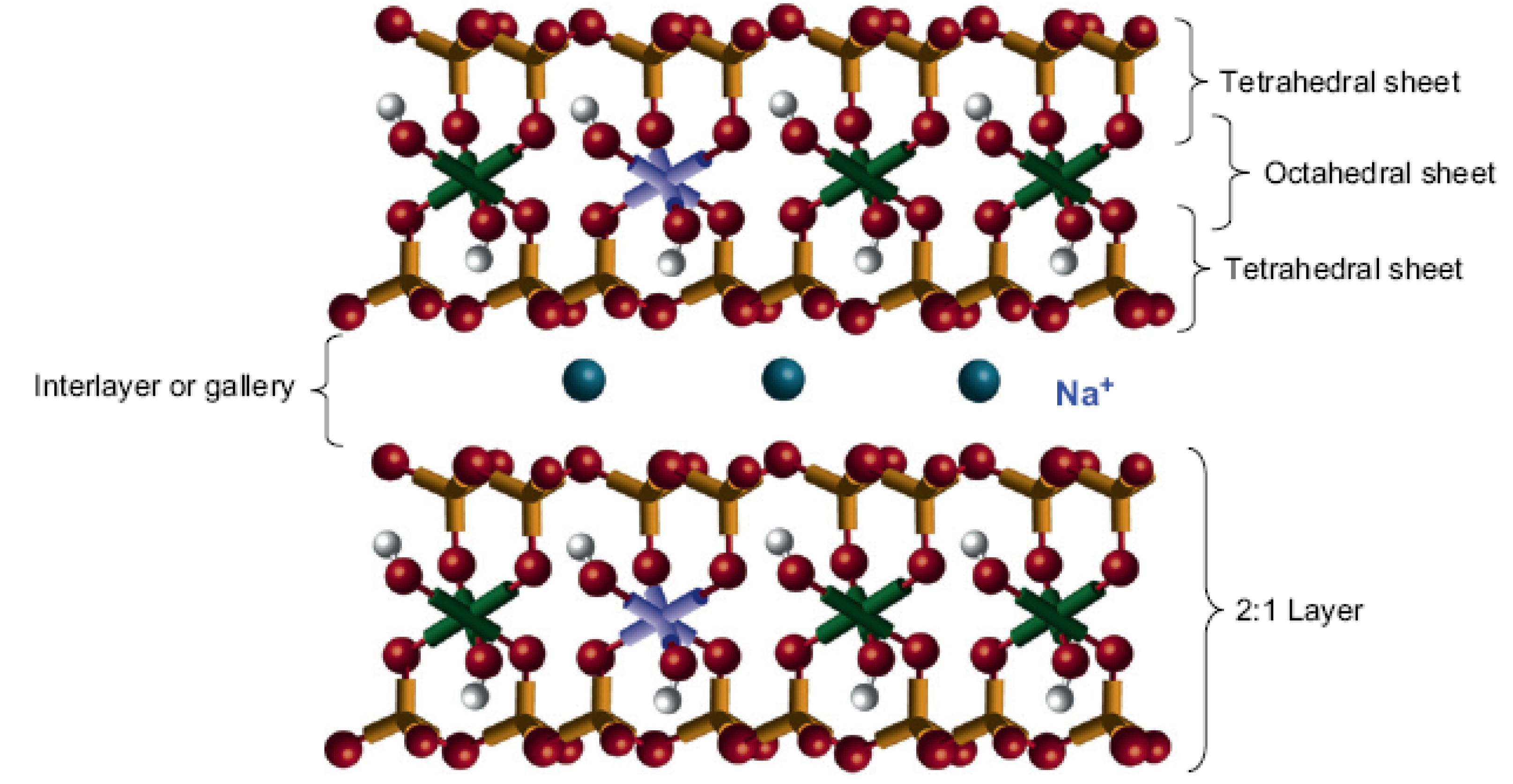
2. Inorganic Nanoparticles
| Metal | Starting Material | Reducing Agent | Stabilizer a | Notes | Avg Diam (nm) |
|---|---|---|---|---|---|
| Co | Co(OAc)2 | N2H4·H2O | none | ~20 | |
| Ni | NiCl2 | N2H4·H2O+NaOH | CTAB | reaction performed at 60 °C | 10-36 |
| Ni | Ni(OAc)2 | N2H4·H2O+NaOH | none | (10-20) × (200-300) rods | |
| Cu | CuSO4 | N2H4·H2O | SDS | ~35 | |
| Ag | AgNO3 | Ascorbic acid | Daxad 19 | 15-26 | |
| Ag | AgNO3 | NaBH4 | TADDD | 3-5 | |
| Pt | H2PtCl6 | potassium bitartrate | TDPC | 60 °C | <1.5 |
| Au | HAuCl4 | trisodium citrate | S3MP | simultaneous addition of reductant and stabilizer | not stated |
| Compd. | Starting Material | Solvent a | Reductant b | Stabilizer c | Conditions | Product Size d (nm) |
|---|---|---|---|---|---|---|
| Fe | Fe(OEt)2 | THF | NaBEt3H | THF | 16 h at 67 °C | 10-100 |
| Fe | Fe(acac)3 | THF | Mg+ | THF | ~8 e | |
| Fe20Ni80 | Fe(OAc)2 Ni(OAc)2 | EG | EG | EG | reflux (150-160 °C) | 6 (A) |
| Co | Co(OH)2 | THF | NaBEt3H | THF | 2h at 23 °C | 10-100 |
| Co | CoCl2 | THF | Mg+ | THF | ~12 | |
| Co20Ni80 | Co(OAc)2 Ni(OAc)2 | EG | EG | EG | reflux (150-160 °C) | 18-22 (A) |
| Ni | Ni(acac)2 | HDA | NaBH4 | HDA | 160 °C | 3.7 (C) |
| Ni | NiCl2 | THF | Mg+ | THF | ~94 e | |
| Ni | Ni(OAc)2 | EG | EG | EG | reflux (150-160 °C) | 25 (A) |
| Ru | RuCl3 | 1,2-PD | 1,2-PD | Na(OAc) and DT | 170 °C | 1-6 (C) |
| Ag | AgNo3 | methanol | NaBH4 | MSA | room temp | 1-6 (C) |
| Ag | AgClO4 | DMF | DMF | 3-APTMS | 20-156 °C | 7-20 (C) |
| Au | AuCl3 | THF | K+(15C)2K- | THF | -50 °C | 6-11 (C) |
| Au | HAuCl3 | formamide | formamide | PVP | 30 °C | 30 (C) |
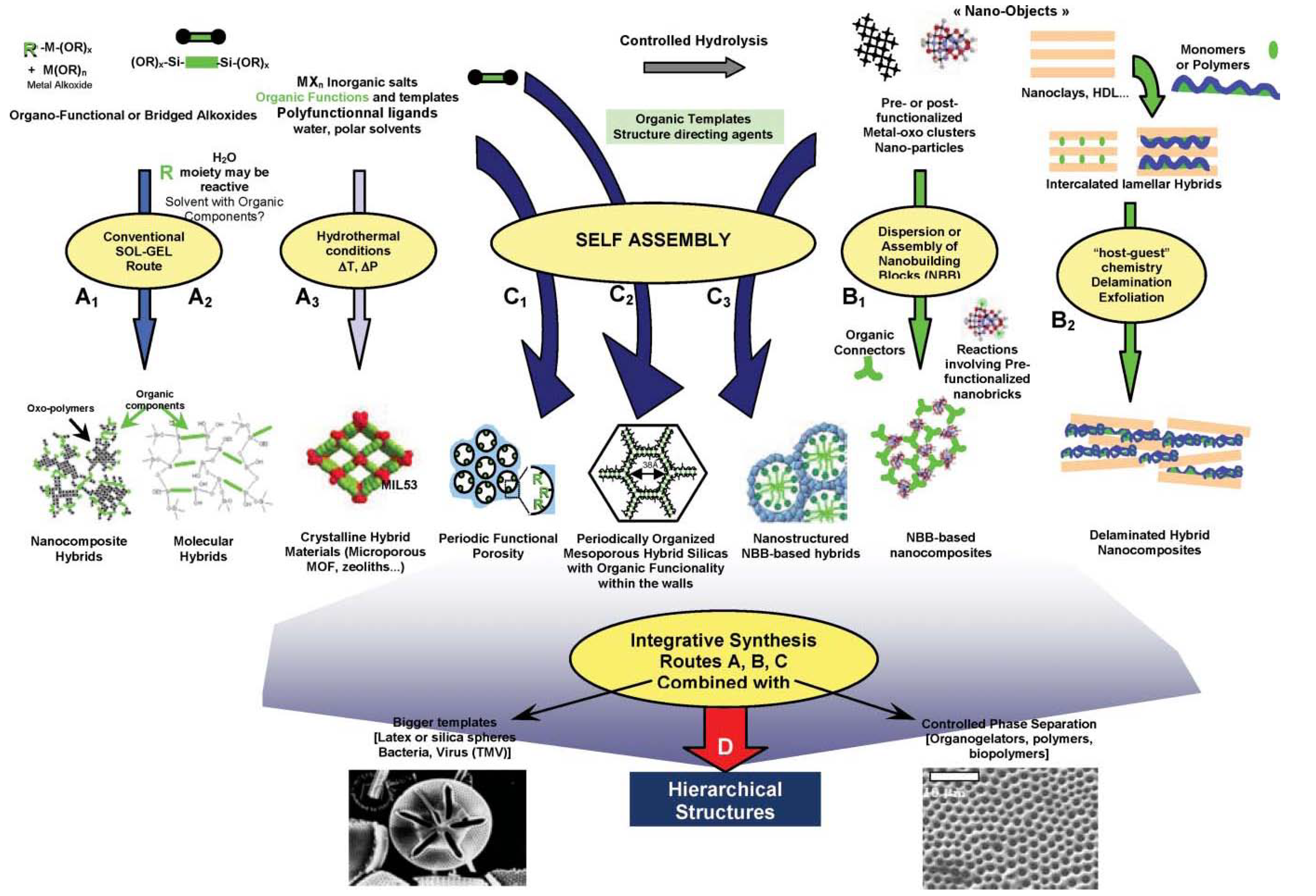
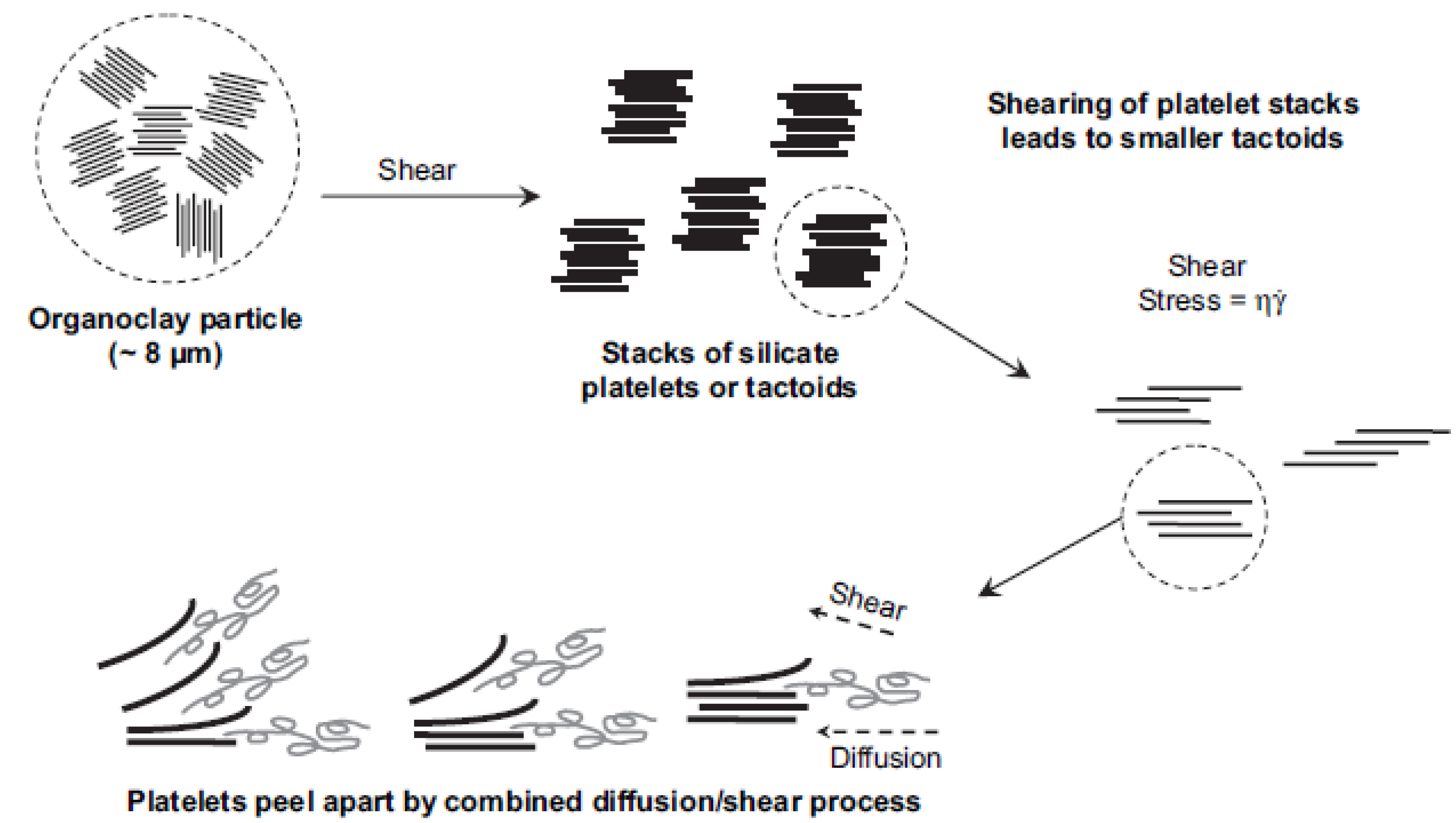
3. Nanocomposite Synthesis
4. Properties of Polymer-based Nanocomposites
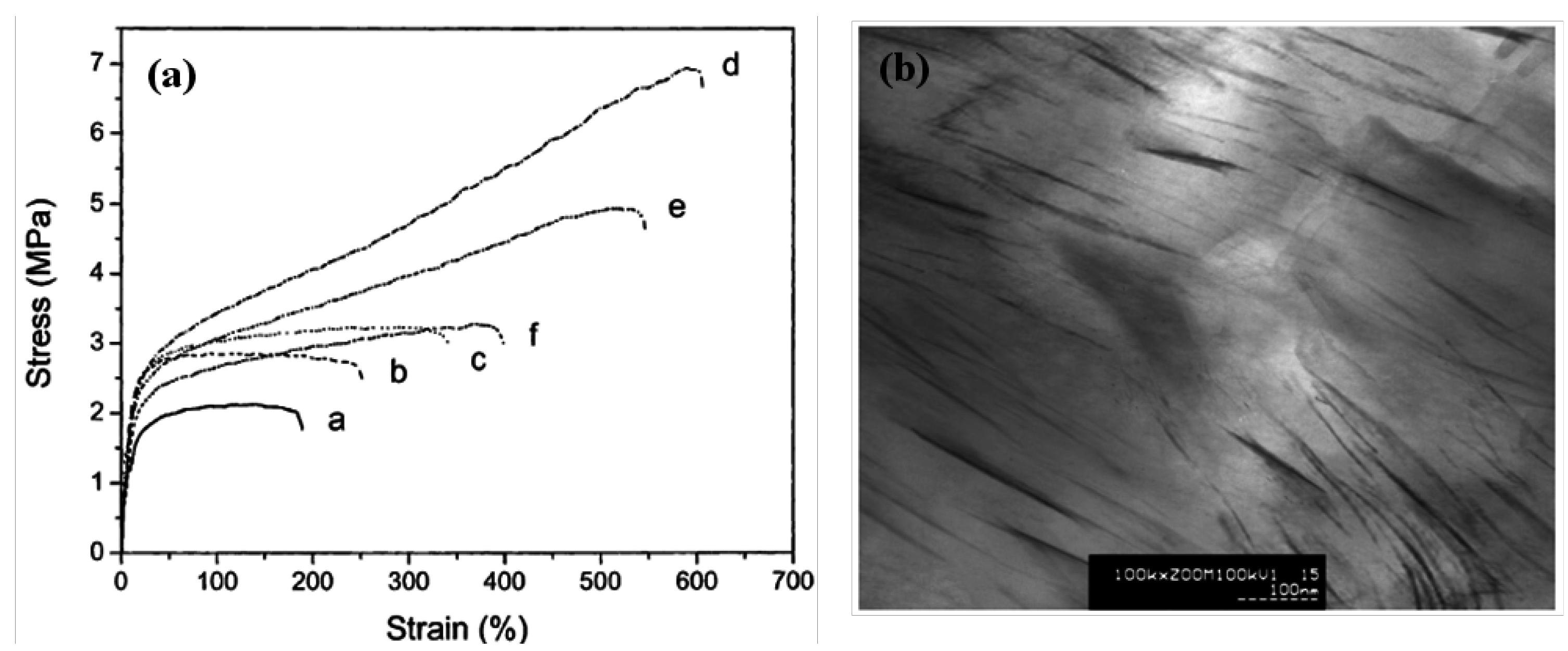
4.1. Mechanical properties

4.2. Thermal properties
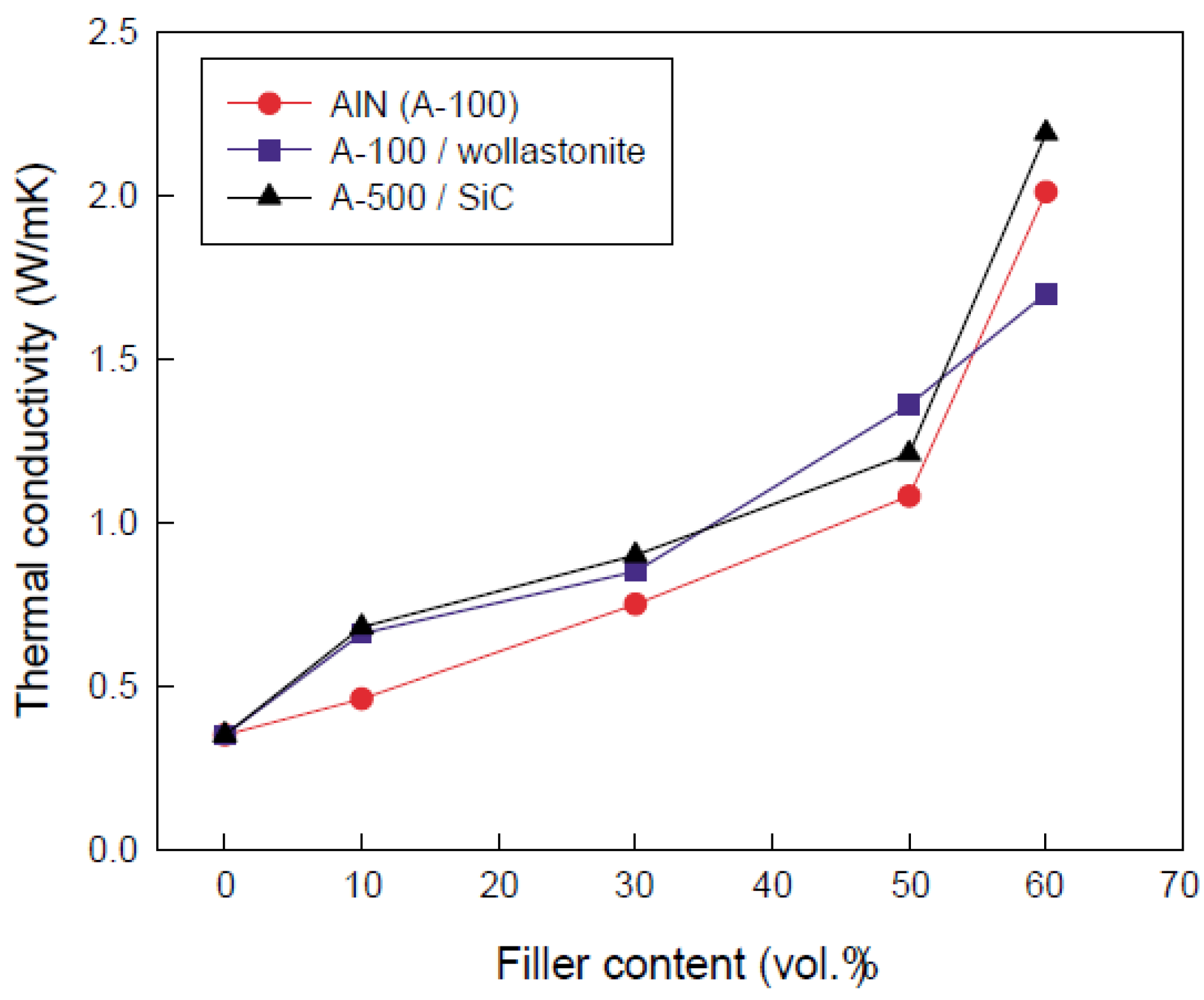
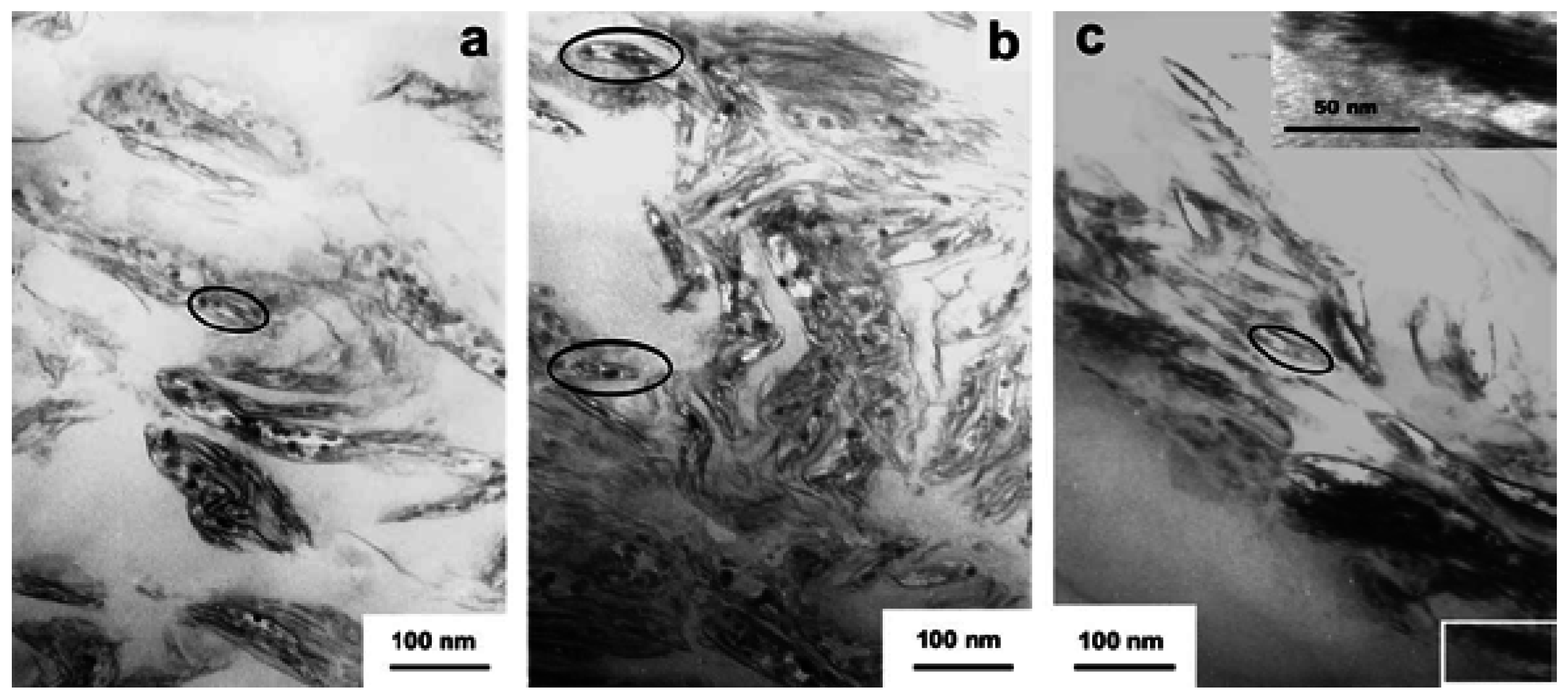

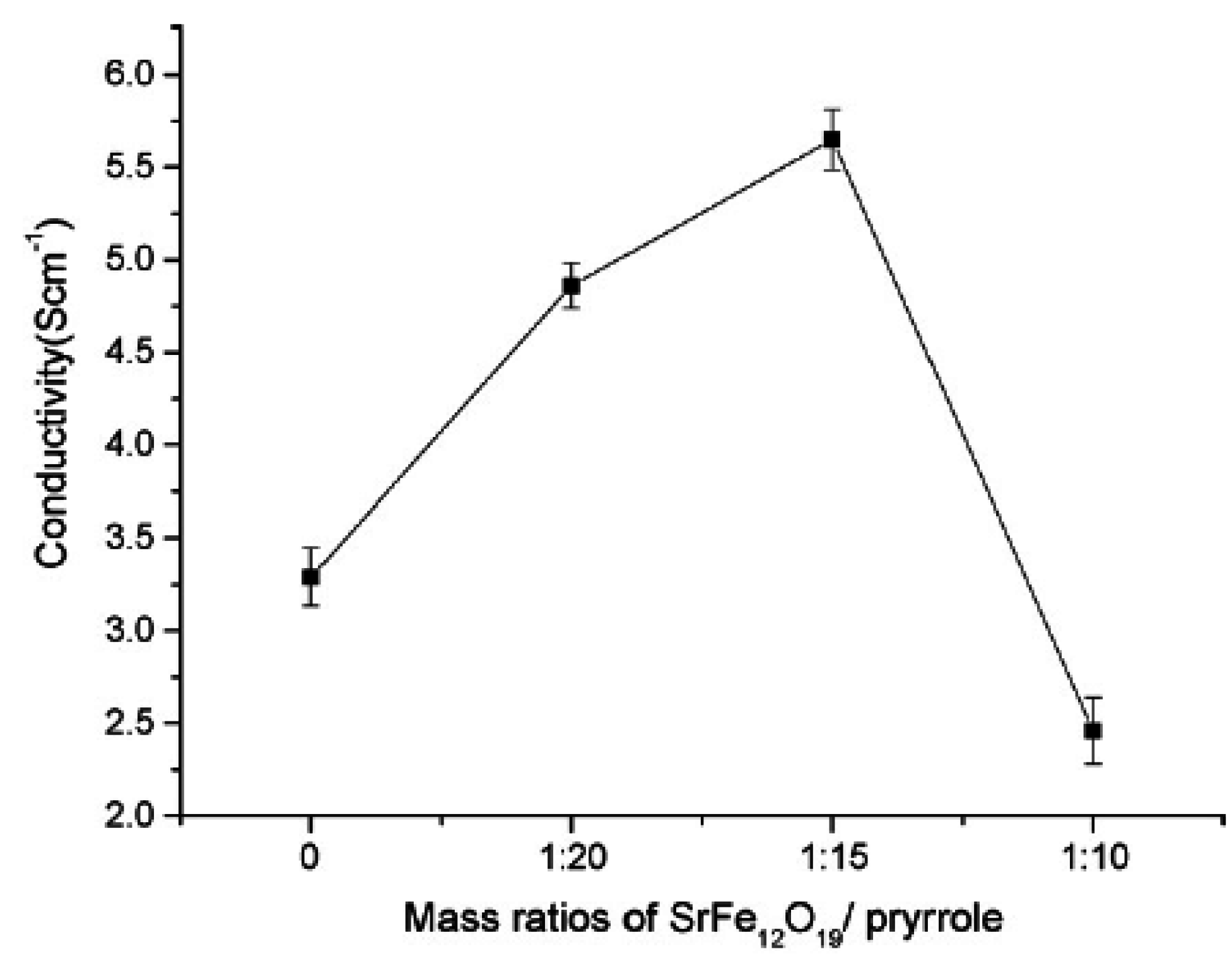
4.3. Electrical properties
4.4. Optical properties

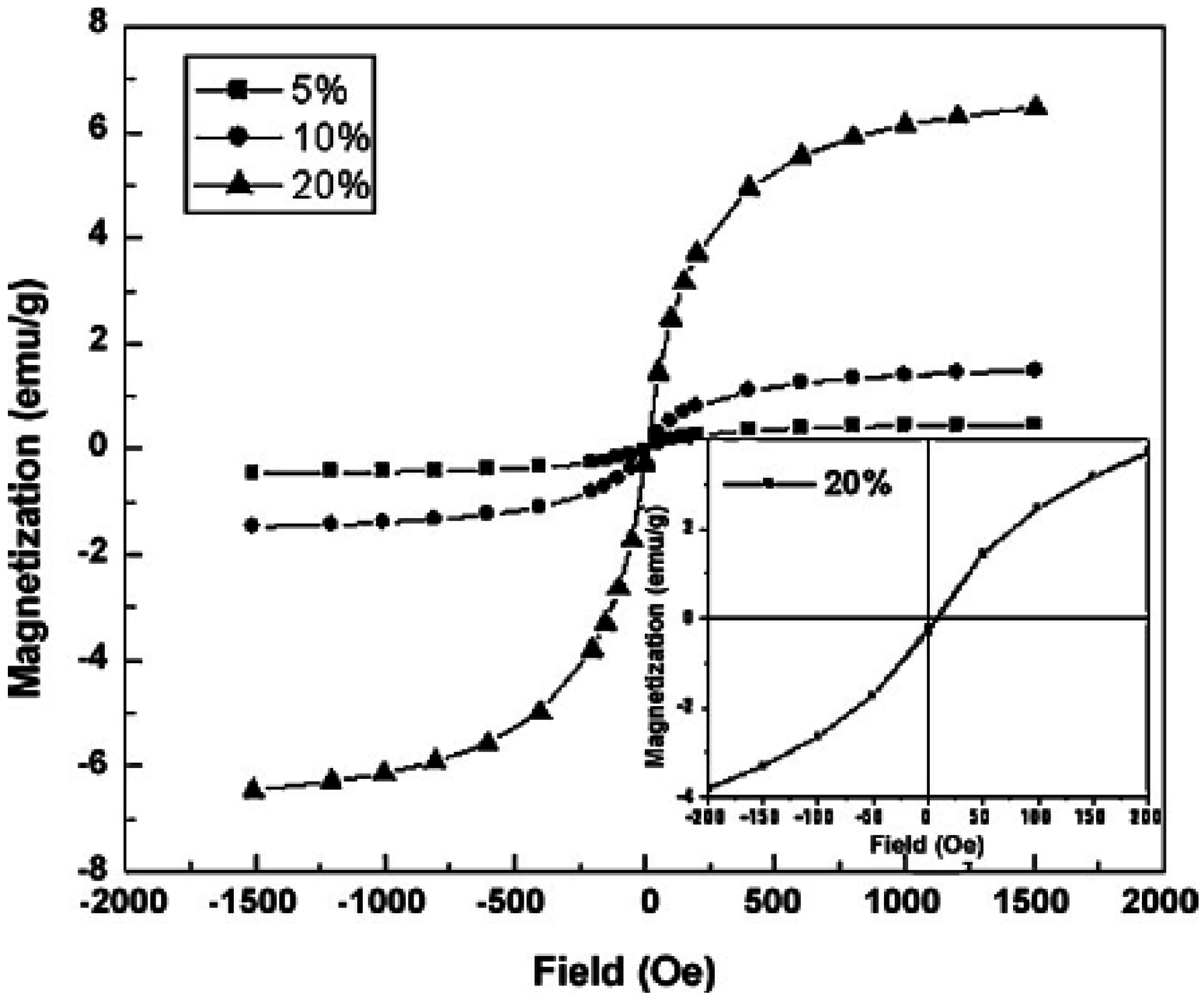
4.5. Magnetic properties

5. Applications of Nanocomposites
| Nanocomposites | Applications |
|---|---|
| Polycarprolactone/SiO2 | Bone-bioerodible for skeletal tissue repair. |
| Polyimide/SiO2 | Microelectronics. |
| PMMA/SiO2 | Dental application, optical devices. |
| Polyethylacrylate/SiO2 | Catalysis support, stationary phase for chromatography. |
| Poly(p-phenylene vinylene)/SiO2 | Non-linear optical material for optical waveguides. |
| Poly(amide-imide)/TiO2 | Composite membranes for gas separation. |
| Poly(3,4-ethylene-dioxythiphene)/ V2O5 | Cathode materials for rechargeable lithium batteries. |
| Polycarbonate/SiO2 | Abrasion resistant coating. |
| Shape memory polymers/SiC | Medical devices for gripping or releasing therapeutics within blood vessels. |
| Nylon-6/LS | Automotive timing-belt-TOYOTA. |
| Nylon-6/clay | Barrier films – Bayer AG |
| Nylon-6/clay | Films and bottles - Honeywell |
| Nylon-6, 12, 66/clay | Auto fuel systems - Ube |
| Nylon-6/PP/clay | Electrically conductive |
| UHMWPE/clay | Earthquake-resistance pipes – Yantai Haili Ind.& Commerce of China |
| Polypropylene/clay | Packaging - Clariant |
| PEO/LS | Airplane interiors, fuel tanks, components in electrical and electronic parts, brakes and tires. |
| PLA/LS | Lithium battery development. |
| PET/clay | Food packaging application. Specific examples include packaging for processed meats, cheese, confectionery, cereals and boil-in-the-bag foods, fruit juice and dairy products, beer and carbonated drinks bottles. |
| Thermoplastic olefin/clay | Beverage containers. |
| Polyimide/clay | Automotive step assists – GM Safari and Astra Vans. |
| Epoxy/MMT | Materials for electronics. |
| SPEEK/laponite | Direct methanol fuel cells. |
| EVA/clay | Wires and cables – Kabelwerk Eupen of Belgium |
| Unsaturated polyester/clay | Marine, transportation – Polymeric Supply |
6. Conclusions
Acknowledgements
References and Notes
- Ajayan, P.M.; Schadler, L.S.; Braun, P.V. Nanocomposite Science and Technology; Wiley: New York, NY, USA, 2003; p. 112. [Google Scholar]
- Jordan, J.; Jacob, K.I.; Tannenbaum, R.; Sharaf, M.A.; Jasiuk, I. Experimental trends in polymer nanocomposites - A review. Mater. Sci. Eng. A 2005, 393, 1–11. [Google Scholar] [CrossRef]
- Berta, M.; Lindsay, C.; Pans, G.; Camino, G. Effect of chemical structure on combustion and thermal behaviour of polyurethane elastomer layered silicate nanocomposites. Polym. Degrad. Stabil. 2006, 91, 1179–1191. [Google Scholar] [CrossRef]
- Sanchez, C.; Julián, B.; Belleville, P.; Popall, M. Applications of hybrid organic-inorganic nanocomposites. J. Mater. Chem. 2005, 15, 3559–3592. [Google Scholar] [CrossRef]
- Hiemenz, P.; Rajagopalan, R. Principles of Colloid and Surface Chemistry; Marcel Dekker Inc.: New York, NY, USA, 1997; pp. 6–10. [Google Scholar]
- Suryanarayana, C.; Froes, F.H. The structure and mechanical properties of metallic nanocrystals. Metall. Trans. A 1992, 23, 1071–1081. [Google Scholar] [CrossRef]
- Chandra, A.; Turng, L.S.; Gopalan, P.; Rowell, R.M.; Gong, S. Study of utilizing thin polymer surface coating on the nanoparticles for melt compounding of polycarbonate/alumina nanocomposites and their optical properties. Comp. Sci. Technol. 2008, 68, 768–776. [Google Scholar] [CrossRef]
- Osman, M.A.; Rupp, J.E.P.; Suter, U.W. Effect of non-ionic surfactants on the exfoliation and properties of polyethylene-layered silicate nanocomposites. Polymer 2005, 46, 8202–8209. [Google Scholar] [CrossRef]
- Cho, J.W.; Paul, D.R. Nylon 6 nanocomposites by melt compounding. Polymer 2001, 42, 1083–1094. [Google Scholar] [CrossRef]
- Chang, J.H.; An, Y.U.; Cho, D.; Giannelis, E.P. Poly(lactic acid) nanocomposites: comparison of their properties with montmorillonite and synthetic mica (II). Polymer 2003, 44, 3715–3720. [Google Scholar] [CrossRef]
- Paul, D.R.; Robeson, L.M. Polymer nanotechnology: nanocomposites. Polymer 2008, 49, 3187–3204. [Google Scholar] [CrossRef]
- Yasmin, A.; Luo, J.J.; Abot, J.L.; Daniel, I.M. Mechanical and thermal behavior of clay/epoxy nanocomposites. Comp. Sci. Technol. 2006, 66, 2415–2422. [Google Scholar] [CrossRef]
- Wang, K.; Chen, L.; Wu, J.; Toh, M.L.; He, C.; Yee, A.F. Epoxy nanocomposites with highly exfoliated clay: Mechanical properties and fracture mechanisms. Macromolecules 2005, 38, 788–800. [Google Scholar] [CrossRef]
- Lippens, P.E.; Lannoo, M. Calculation of the band gap for small CdS and ZnS crystallites. Phys. Rev. B 1989, 39, 10935–10942. [Google Scholar] [CrossRef]
- Palik, E.; Ghosh, G. Handbook of Optical Constants of Solids; Academic press: Orlando, FL, USA, 1985; pp. 597–602. [Google Scholar]
- Prevenslik, T.V. Acoustoluminescence and sonoluminescence. J. Lumin. 2000, 87, 1210–1212. [Google Scholar] [CrossRef]
- Hiemenz, P.; Rajagopalan, R. Principles of Colloid and Surface Science; Marcel Decker press: New York, NY, USA, 1997; pp. 604–616. [Google Scholar]
- Wu, T.; Ke, Y. Melting, crystallization and optical behaviors of poly (ethylene terephthalate)-silica/polystyrene nanocomposite films. Thin Solid Films 2007, 515, 5220–5226. [Google Scholar] [CrossRef]
- Shenhar, R.; Norsten, T.B.; Rotello, V.M. Polymer-mediated nanoparticle assembly: structural control and applications. Adv. Mater. 2005, 17, 657–669. [Google Scholar] [CrossRef]
- Han, K.; Yu, M. Study of the preparation and properties of UV-blocking fabrics of a PET/TiO2 nanocomposite prepared by in situ polycondensation. J. Appl. Polym. Sci. 2006, 100, 1588–1593. [Google Scholar] [CrossRef]
- Guo, Z.; Pereira, T.; Choi, O.; Wang, Y.; Hahn, H.T. Surface functionalized alumina nanoparticle filled polymeric nanocomposites with enhanced mechanical properties. J. Mater. Chem. 2006, 16, 2800–2808. [Google Scholar] [CrossRef]
- Cushing, B.; Kolesnichenko, V.; O'Connor, C. Recent advances in the liquid-phase syntheses of inorganic nanoparticles. Chem. Rev. 2004, 104, 3893–3946. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, properties and uses of a new class of materials. Mater. Sci. Eng. R. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Ogata, N.; Kawakage, S.; Ogihara, T. Structure and thermal/mechanical properties of poly(ethylene oxide)-clay mineral blends. Polymer 1997, 38, 5115–5118. [Google Scholar] [CrossRef]
- Jeon, H.G.; Jung, H.T.; Lee, S.W.; Hudson, S.D. Morphology of polymer/silicate nanocomposites: high density polyethylene and a nitrile copolymer. Polym. Bull. 1998, 41, 107–113. [Google Scholar] [CrossRef]
- Okamoto, M.; Morita, S.; Kim, Y.H.; Kotaka, T.; Tateyama, H. Synthesis and structure of smectic clay/poly(methyl methacrylate) and clay/polystyrene nanocomposites via in situ intercalative polymerization. Polymer 2000, 41, 3887–3890. [Google Scholar] [CrossRef]
- Okamoto, M.; Morita, S.; Kotaka, T. Dispersed structure and ionic conductivity of smectic clay/polymer nanocomposites. Polymer 2001, 42, 2685–2688. [Google Scholar] [CrossRef]
- Yao, K.J.; Song, M.; Hourston, D.J.; Luo, D.Z. Polymer/layered clay nanocomposites 2: polyurethane nanocomposites. Polymer 2002, 43, 1017–1020. [Google Scholar] [CrossRef]
- Vaia, R.A.; Giannelis, E.P. Lattice model of polymer melt intercalation in organically-modified layered silicates. Macromolecules 1997, 30, 7990–7999. [Google Scholar] [CrossRef]
- Kawasumi, M.; Hasegawa, N.; Kato, M.; Usuki, A.; Okada, A. Preparation and mechanical properties of polypropylene-clay hybrids. Macromolecules 1997, 30, 6333–6338. [Google Scholar] [CrossRef]
- Gyoo, P.; Venkataramani, S.; Kim, S. Morphology, thermal, and mechanical properties of polyamide 66/clay nanocomposites with epoxy-modified organoclay. J. Appl. Polym. Sci. 2006, 101, 1711–1722. [Google Scholar] [CrossRef]
- Erdem, N.; Cireli, A.; Erdogan, U. Flame retardancy behaviors and structural properties of polypropylene/nano-SiO2 composite textile filaments. J. Appl. Polym. Sci. 2009, 111, 2085–2091. [Google Scholar] [CrossRef]
- Du, H.; Xu, G.; Chin, W.; Huang, L.; Ji, W. Synthesis, characterization, and nonlinear optical properties of hybridized CdS-polystyrene nanocomposites. Chem. Mater. 2002, 14, 4473–4479. [Google Scholar] [CrossRef]
- Carrado, K.A.; Xu, L. In situ synthesis of polymer-clay nanocomposites from silicate gels. Chem. Mater. 1998, 10, 1440–1445. [Google Scholar] [CrossRef]
- Tomasko, D.L.; Han, X.; Liu, D.; Gao, W. Supercritical fluid applications in polymer nanocomposites. Curr. Opin. Solid St. Mater. Sci. 2003, 7, 407–412. [Google Scholar] [CrossRef]
- Park, S.S.; Bernet, N.; De La Roche, S.; Hahn, H.T. Processing of iron oxide-epoxy vinyl ester nanocomposites. J. Comp. Mater. 2003, 37, 465–476. [Google Scholar] [CrossRef]
- Evora, V.M.F.; Shukla, A. Fabrication, characterization, and dynamic behavior of polyester/TiO2 nanocomposites. Mater. Sci. Eng. A 2003, 361, 358–366. [Google Scholar] [CrossRef]
- Aymonier, C.; Bortzmeyer, D.; Thomann, R.; Mülhaupt, R. Poly(methyl methacrylate)/palladium nanocomposites: synthesis and characterization of the morphological, thermomechanical, and thermal properties. Chem. Mater. 2003, 15, 4874–4878. [Google Scholar] [CrossRef]
- Avadhani, C.V.; Chujo, Y. Polyimide-silica gel hybrids containing metal salts: preparation via the sol-gel reaction. Appl. Organometal. Chem. 1997, 11, 153–161. [Google Scholar] [CrossRef]
- Liu, J.; Gao, Y.; Wang, F.; Li, D.; Xu, J. Preparation and characteristic of a new class of silica/polyimide nanocomposites. J. Mater. Sci. 2002, 37, 3085–3088. [Google Scholar] [CrossRef]
- Kickelbick, G. Concepts for the incorporation of inorganic building blocks into organic polymers on a nanoscale. Prog. Polym. Sci. 2003, 28, 83–114. [Google Scholar] [CrossRef]
- Wang, K.; Choi, M.; Koo, C.; Choi, Y.; Chung, I. Synthesis and characterization of maleated polyethylene/clay nanocomposites. Polymer 2001, 42, 9819–9826. [Google Scholar] [CrossRef]
- Chaichana, E.; Jongsomjit, B.; Praserthdam, P. Effect of nano-SiO2 particle size on the formation of LLDPE/SiO2 nanocomposite synthesized via the in situ polymerization with metallocene catalyst. Chem. Eng. Sci. 2007, 62, 899–905. [Google Scholar] [CrossRef]
- Fornes, T.; Yoon, P.; Keskkula, H.; Paul, D. Nylon 6 nanocomposites: the effect of matrix molecular weight. Polymer 2001, 42, 9929–9940. [Google Scholar] [CrossRef]
- Homminga, D.; Goderis, B.; Mathot, V.; Groeninckx, G. Crystallization behavior of polymer/montmorillonite nanocomposites. Part III. Polyamide-6/montmorillonite nanocomposites, influence of matrix molecular weight, and of montmorillonite type and concentration. Polymer 2006, 47, 1630–1639. [Google Scholar] [CrossRef]
- Zhao, Q.; Samulski, E. In situ polymerization of poly (methyl methacrylate)/clay nanocomposites in supercritical carbon dioxide. Macromolecules 2005, 38, 7967–7971. [Google Scholar] [CrossRef]
- Zerda, A.; Caskey, T.; Lesser, A. Highly concentrated, intercalated silicate nanocomposites: synthesis and characterization. Macromolecules 2003, 36, 1603–1608. [Google Scholar] [CrossRef]
- Pattanayak, A.; Jana, S.C. Properties of bulk-polymerized thermoplastic polyurethane nanocomposites. Polymer 2005, 46, 3394–3406. [Google Scholar] [CrossRef]
- Min, K.D.; Kim, M.Y.; Choi, K.Y.; Lee, J.H.; Lee, S.G. Effect of layered silicates on the crystallinity and mechanical properties of HDPE/MMT nanocomposite blown films. Polym. Bull. 2006, 57, 101–108. [Google Scholar] [CrossRef]
- Lee, H.; Lin, L. Waterborne polyurethane/clay nanocomposites: novel effects of the clay and its interlayer ions on the morphology and physical and electrical properties. Macromolecules 2006, 39, 6133–6141. [Google Scholar] [CrossRef]
- Fornes, T.D.; Paul, D.R. Crystallization behavior of nylon 6 nanocomposites. Polymer 2003, 44, 3945–3961. [Google Scholar] [CrossRef]
- Zhang, X.; Simon, L.C. In situ polymerization of hybrid polyethylene-alumina nanocomposites. Macromol. Mater. Eng. 2005, 290, 573–583. [Google Scholar] [CrossRef]
- Chae, D.W.; Kim, B.C. Characterization on polystyrene/zinc oxide nanocomposites prepared from solution mixing. Polym. Adv. Technol. 2005, 16, 846–850. [Google Scholar] [CrossRef]
- Sarwar, M.I.; Zulfiqar, S.; Ahmad, Z. Polyamide-silica nanocomposites: Mechanical, morphological and thermomechanical investigations. Polym. Int. 2008, 57, 292–296. [Google Scholar] [CrossRef]
- Lu, X.; Lu, N.; Gao, J.; Jin, X.; Lu, C. Synthesis and properties of ZnS polyimide nanocomposite films. Polym. Int. 2007, 56, 601–605. [Google Scholar] [CrossRef]
- Reddy, C.S.; Das, C.K. Polypropylene-nanosilica-filled composites: effects of epoxy-resin-grafted nanosilica on the structural, thermal, and dynamic mechanical properties. J. Appl. Polym. Sci. 2006, 102, 2117–2124. [Google Scholar] [CrossRef]
- Yu, S.; Hing, P.; Hu, X. Thermal conductivity of polystyrene-aluminum nitride composite. Compos. Part A 2002, 33, 289–292. [Google Scholar] [CrossRef]
- Lee, G.; Park, M.; Kim, J.; Lee, J.; Yoon, H. Enhanced thermal conductivity of polymer composites filled with hybrid filler. Compos. Part A 2006, 37, 727–734. [Google Scholar] [CrossRef]
- Wang, H.; Xu, P.; Zhong, W.; Shen, L.; Du, Q. Transparent poly(methyl methacrylate)/silica/zirconia nanocomposites with excellent thermal stabilities. Polym. Degrad. Stabil. 2005, 87, 319–327. [Google Scholar] [CrossRef]
- Du, L.; Qu, B.; Zhang, M. Thermal properties and combustion characterization of nylon 6/MgAl-LDH nanocomposites via organic modification and melt intercalation. Polym. Degrad. Stabil. 2007, 92, 497–502. [Google Scholar] [CrossRef]
- Du, L.; Qu, B. Structural characterization and thermal oxidation properties of LLDPE/MgAl-LDH nanocomposites. J. Mater. Chem. 2006, 16, 1549–1554. [Google Scholar] [CrossRef]
- Lu, H.; Hu, Y.; Li, M.; Chen, Z.; Fan, W. Structure characteristics and thermal properties of silane-grafted-polyethylene/clay nanocomposite prepared by reactive extrusion. Comp. Sci. Technol. 2006, 66, 3035–3039. [Google Scholar] [CrossRef]
- Qiu, L.; Chen, W.; Qu, B. Structural characterisation and thermal properties of exfoliated polystyrene/ZnAl layered double hydroxide nanocomposites prepared via solution intercalation. Polym. Degrad. Stabil. 2005, 87, 433–440. [Google Scholar] [CrossRef]
- Valera-Zaragoza, M.; Ramírez-Vargas, E.; Medellín-Rodríguez, F.J.; Huerta-Martínez, B.M. Thermal stability and flammability properties of heterophasic PP-EP/EVA/organoclay nanocomposites. Polym. Degrad. Stabil. 2006, 91, 1319–1325. [Google Scholar] [CrossRef]
- Su, S.J.; Kuramoto, N. Processable polyaniline-titanium dioxide nanocomposites: effect of titanium dioxide on the conductivity. Synthet. Metal. 2000, 114, 147–153. [Google Scholar] [CrossRef]
- Mo, T.C.; Wang, H.W.; Chen, S.Y.; Yeh, Y.C. Synthesis and dielectric properties of polyaniline/titanium dioxide nanocomposites. Ceram. Int. 2008, 34, 1767–1771. [Google Scholar] [CrossRef]
- Olad, A.; Rashidzadeh, A. Preparation and anticorrosive properties of PANI/Na-MMT and PANI/O-MMT nanocomposites. Prog. Org. Coat. 2008, 62, 293–298. [Google Scholar] [CrossRef]
- Tang, Q.; Sun, X.; Li, Q.; Lin, J.; Wu, J. Preparation and electrical conductivity of SiO2/polypyrrole nanocomposite. J. Mater. Sci. 2009, 44, 849–854. [Google Scholar] [CrossRef]
- Zhang, C.; Li, Q.; Ye, Y. Preparation and characterization of polypyrrole/nano-SrFe12O19 composites by in situ polymerization method. Synthet. Metal. 2009, 159, 1008–1013. [Google Scholar] [CrossRef]
- Yu, Y.H.; Jen, C.C.; Huang, H.Y.; Wu, P.C.; Huang, C.C.; Yeh, J.M. Preparation and properties of heterocyclically conjugated poly(3-hexylthiophene)-clay nanocomposite materials. J. Appl. Polym. Sci. 2004, 91, 3438–3446. [Google Scholar] [CrossRef]
- Ma, C.C.M.; Chen, Y.J.; Kuan, H.C. Polystyrene nanocomposite materials - preparation, mechanical, electrical and thermal properties, and morphology. J. Appl. Polym. Sci. 2006, 100, 508–515. [Google Scholar] [CrossRef]
- Xu, B.H.; Lin, B.Z.; Sun, D.Y.; Ding, C. Preparation and electrical conductivity of polyethers/WS2 layered nanocomposites. Electrochim. Acta 2007, 52, 3028–3034. [Google Scholar] [CrossRef]
- Wang, Y.J.; Kim, D. Crystallinity, morphology, mechanical properties and conductivity study of in situ formed PVdF/LiClO4/TiO2 nanocomposite polymer electrolytes. Electrochim. Acta 2007, 52, 3181–3189. [Google Scholar] [CrossRef]
- Pandey, G.P.; Agrawal, R.C.; Hashmi, S.A. Magnesium ion-conducting gel polymer electrolytes dispersed with nanosized magnesium oxide. J. Power Sources 2009, 190, 563–572. [Google Scholar] [CrossRef]
- Garnett, J. Colours in metal glasses, in metallic films, and in metallic solutions. II. Philos. Trans. R. Soc. London A 1906, 237–288. [Google Scholar] [CrossRef]
- Biswas, A.; Aktas, O.C.; Kanzow, J.; Saeed, U.; Strunskus, T.; Zaporojtchenko, V.; Faupel, F. Polymer-metal optical nanocomposites with tunable particle plasmon resonance prepared by vapor phase co-deposition. Mater. Lett. 2004, 58, 1530–1534. [Google Scholar] [CrossRef]
- Chai, R.; Lian, H.; Yang, P.; Fan, Y.; Hou, Z.; Kang, X.; Lin, J. In situ preparation and luminescent properties of LaPO4:Ce3+, Tb3+ nanoparticles and transparent LaPO4:Ce3+, Tb3+/PMMA nanocomposite. J. Colloid. Interf. Sci. 2009, 336, 46–50. [Google Scholar] [CrossRef]
- Avasthi, D.K.; Mishra, Y.K.; Kabiraj, D.; Lalla, N.P.; Pivin, J.C. Synthesis of metal-polymer nanocomposite for optical applications. Nanotechnology 2007, 18, 125604. [Google Scholar] [CrossRef]
- Chau, J.L.H.; Tung, C.T.; Lin, Y.M.; Li, A.K. Preparation and optical properties of titania/epoxy nanocomposite coatings. Mater. Lett. 2008, 62, 3416–3418. [Google Scholar] [CrossRef]
- Alam, J.; Riaz, U.; Ahmad, S. Effect of ferrofluid concentration on electrical and magnetic properties of the Fe3O4/PANI nanocomposites. J. Magn. Magn. Mater. 2007, 314, 93–99. [Google Scholar] [CrossRef]
- Yu, Q.; Shi, M.; Cheng, Y.; Wang, M.; Chen, H. Fe3O4@ Au/polyaniline multifunctional nanocomposites: their preparation and optical, electrical and magnetic properties. Nanotechnology 2008, 19, 265702. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Ouyang, C.; Jia, R.; Li, Y.; Wang, X. Magnetic and optical properties of poly(vinylidene difluoride)/Fe3O4 nanocomposite prepared by coprecipitation approach. J. Appl. Polym. Sci. 2009, 111, 1763–1768. [Google Scholar] [CrossRef]
- Zhan, J.; Tian, G.; Jiang, L.; Wu, Z.; Wu, D.; Yang, X.; Jin, R. Superparamagnetic polyimide/γ-Fe2O3 nanocomposite films: preparation and characterization. Thin Solid Films 2008, 516, 6315–6320. [Google Scholar] [CrossRef]
- Sun, D.C.; Sun, D.S. The synthesis and characterization of electrical and magnetic nanocomposite: PEDOT/PSS-Fe3O4. Mater. Chem. Phys. 2009, 118, 288–292. [Google Scholar] [CrossRef]
- Camargo, P.; Satyanarayana, K.; Wypych, F. Nanocomposites: synthesis, structure, properties and new application opportunities. Mater. Res. 2009, 12, 1–39. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jeon, I.-Y.; Baek, J.-B. Nanocomposites Derived from Polymers and Inorganic Nanoparticles. Materials 2010, 3, 3654-3674. https://doi.org/10.3390/ma3063654
Jeon I-Y, Baek J-B. Nanocomposites Derived from Polymers and Inorganic Nanoparticles. Materials. 2010; 3(6):3654-3674. https://doi.org/10.3390/ma3063654
Chicago/Turabian StyleJeon, In-Yup, and Jong-Beom Baek. 2010. "Nanocomposites Derived from Polymers and Inorganic Nanoparticles" Materials 3, no. 6: 3654-3674. https://doi.org/10.3390/ma3063654
APA StyleJeon, I.-Y., & Baek, J.-B. (2010). Nanocomposites Derived from Polymers and Inorganic Nanoparticles. Materials, 3(6), 3654-3674. https://doi.org/10.3390/ma3063654




