Polyoxometalates in Oxidative Delignification of Chemical Pulps: Effect on Lignin
Abstract
:1. Lignin
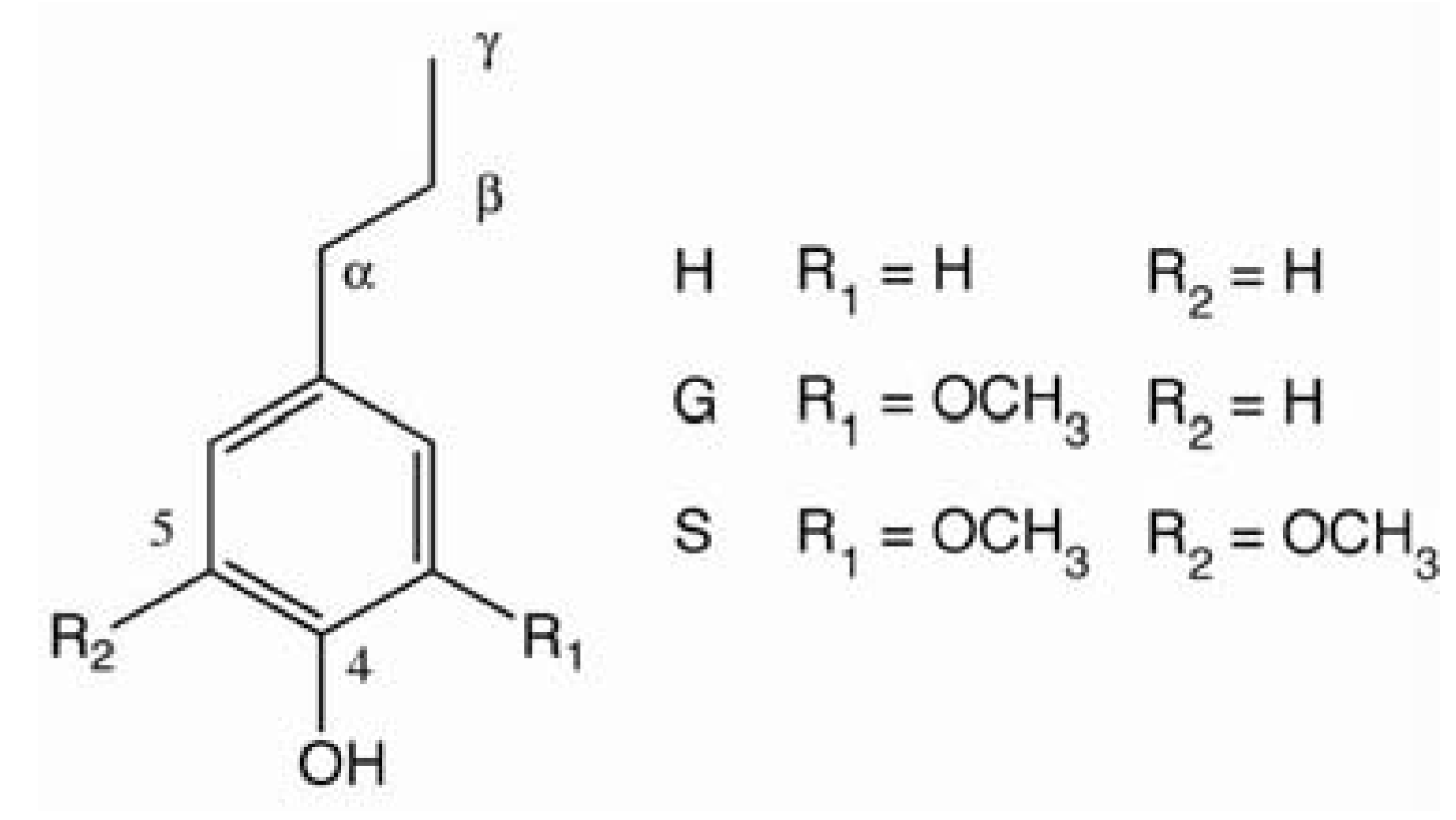
2. Pulping and Bleaching
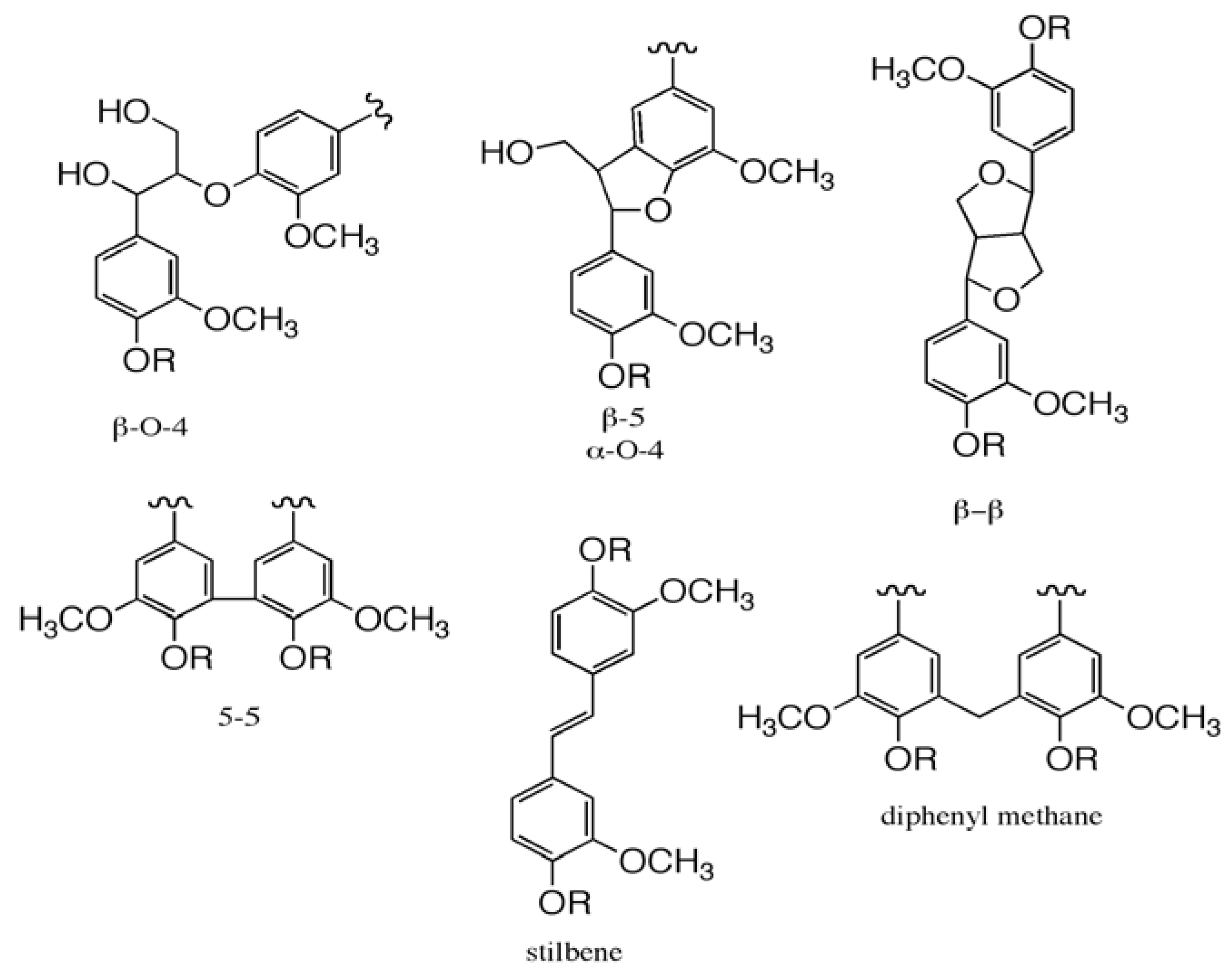
3. Polyoxometalates in Bleaching

3.1. Delignification with POMs: Anaerobic Conditions

Effect on lignin
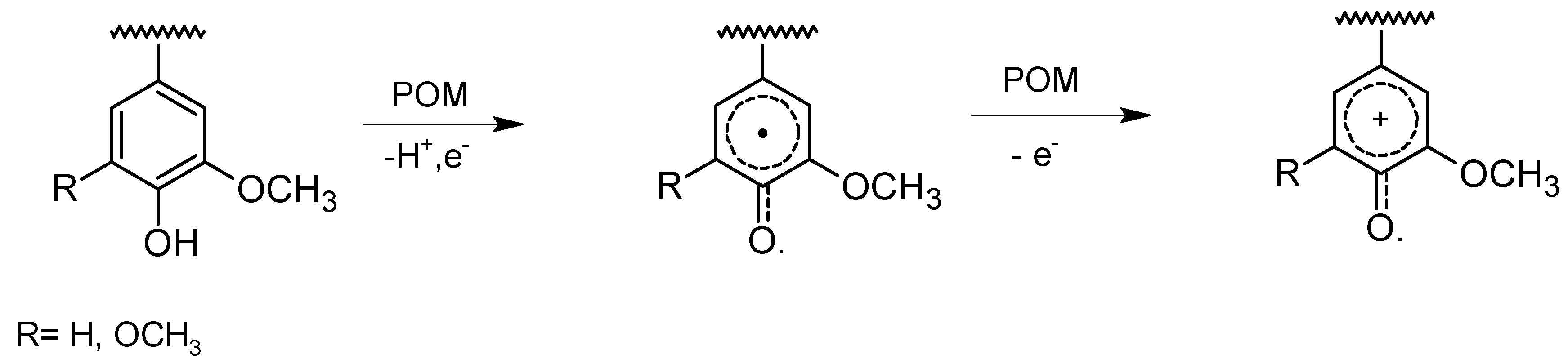
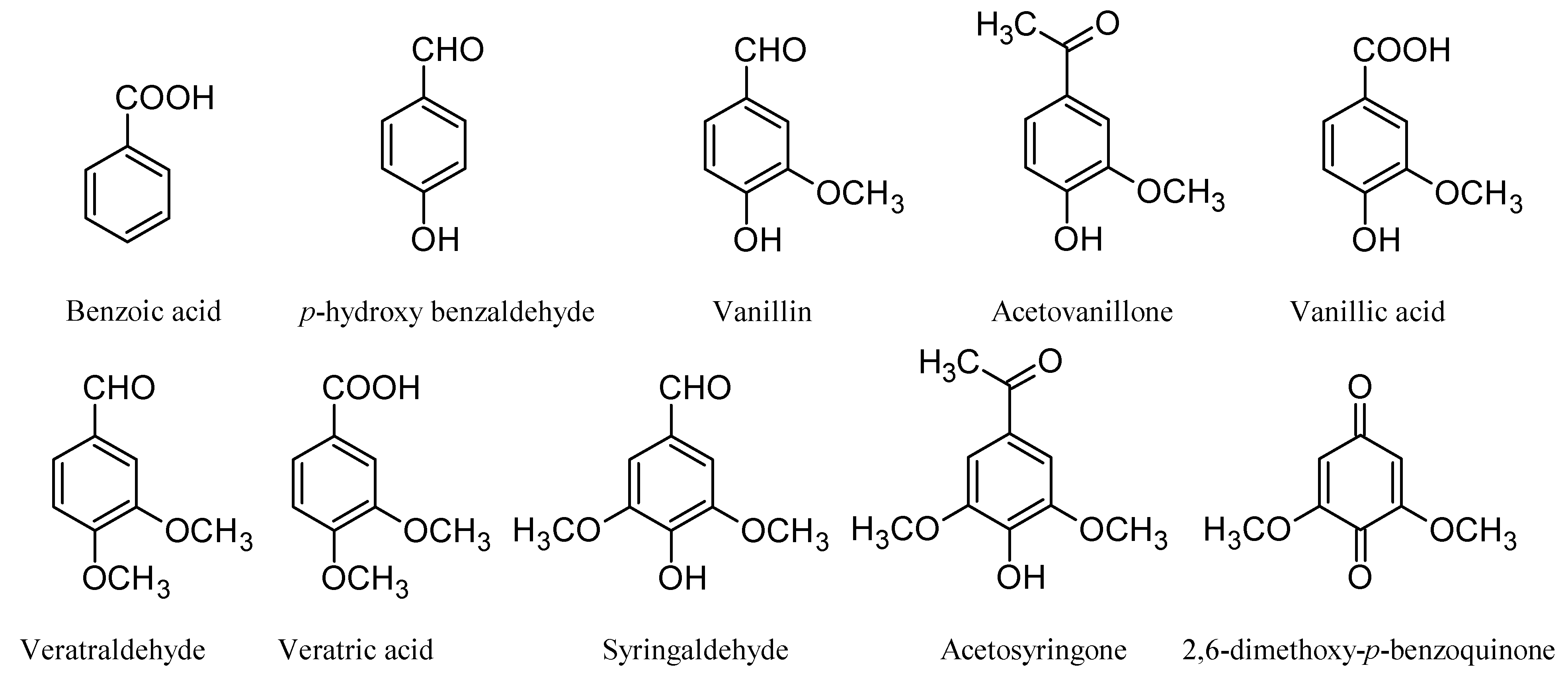
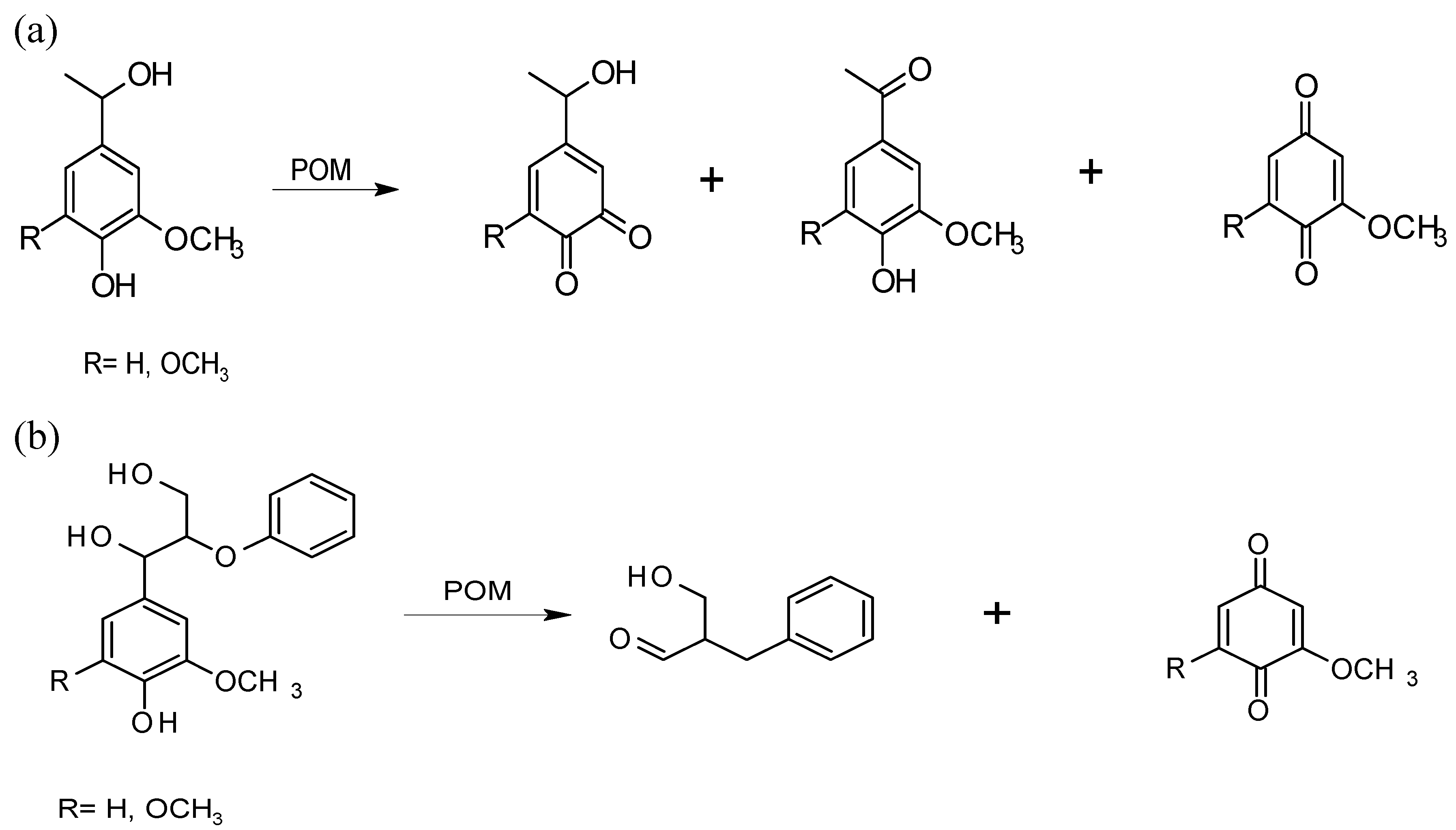
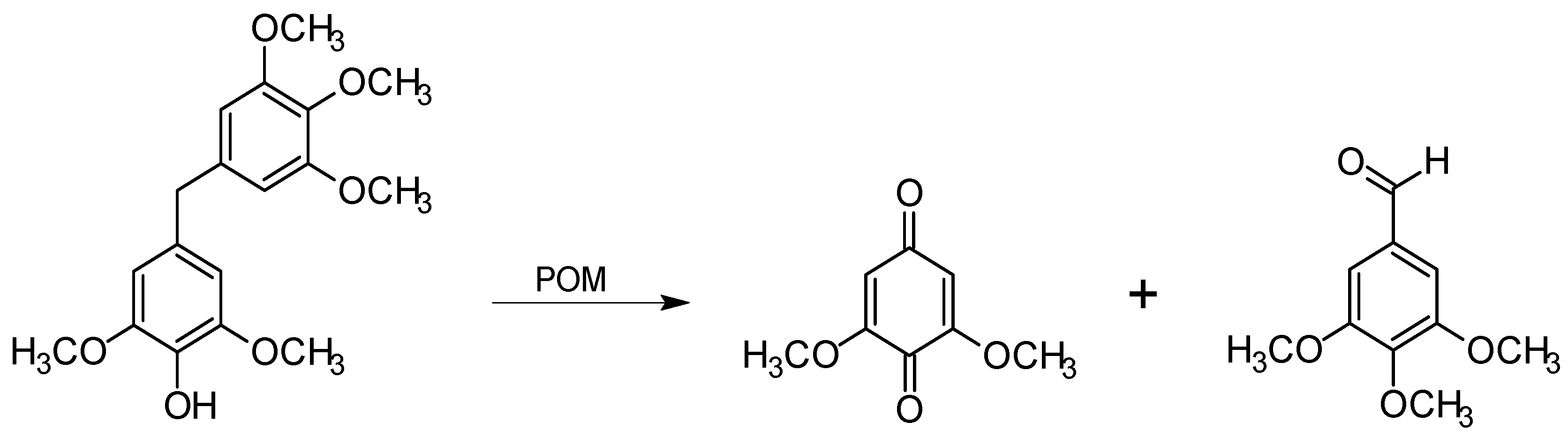
3.2. Delignification with POMs: Aerobic Conditions
3.2.1. Effect on lignin
3.2.2. Laccase as a catalyst of re-oxidation of POMs
4. Conclusion
References and Notes
- Brunow, G.; Lundquist, K.; Gellerstedt, G. Lignin. In Analytical Methods in Wood Chemistry, Pulping, and Papermaking; Sjöström, E., Alén, R., Eds.; Springer Ser. in Wood Sci., Springer‑Verlag: Berlin Heidelberg, Germany, 1999; pp. 77–124. [Google Scholar]
- Brunow, G.; Kilpeläinen, I.; Sipilä, J.; Syrjänen, K.; Karhunen, P.; Setälä, H.; Rumakko, P. Oxidative Coupling of Phenols and Biosynthesis of Lignin. In Lignin and Lignan Biosynthesis; Lewis, N.G., Sarkanen, S., Eds.; ACS Symp. Ser. 697; American Chemical Society: Washington, DC, USA, 1998; pp. 131–147. [Google Scholar]
- Watanabe, T. Analysis of Native Bonds Between Lignin and Carbohydrates by Specific Chemical Reactions. In Association between Lignin and Carbohydrates in Wood and Other Plant Tissues; Koshijima, T., Watanabe, T., Eds.; Springer Ser. in Wood Sci.; Springer-Verlag: Berlin Heidelberg, Germany, 2003; pp. 91–130. [Google Scholar]
- Sjöström, E. Wood Chemistry Fundamentals and Applications; Academic Press, Inc.: Orlando, FL, USA, 1981; pp. 104–168. [Google Scholar]
- Choi, J.W.; Choi, D.-H.; Faix, O. Characterization of lignin-carbohydrate linkages in the residual lignins isolated from chemical pulps of spruce (Picea abies) and beech wood (Fagus sylvatica). J. Wood Sci. 2007, 53, 309–313. [Google Scholar] [CrossRef]
- Majtnerova, A.; Gellerstedt, G. Radical coupling–A major obstacle to delignification in kraft pulping. Nordic Pulp Paper Res. J. 2006, 21, 129–134. [Google Scholar] [CrossRef]
- Koshijima, T.; Watanabe, T. Residual Lignin in Alkaline Pulps. In Association Between Lignin and Carbohydrates in Wood and Other Plant Tissues; Koshijima, T., Watanabe, T., Eds.; Springer Ser. in Wood Sci.; Springer-Verlag: Heidelberg, Berlin, Germany, 2003; pp. 131–215. [Google Scholar]
- Ahvazi, B.C.; Pageau, G.; Argyropoulos, D.S. On the formation of diphenylmethane structures in lignin under kraft, EMCC, and soda kraft pulping conditions. Can. J. Chem. 1998, 76, 506–512. [Google Scholar] [CrossRef]
- Gellerstedt, G.; Zhang, L. Chemistry of TCF-Bleaching with Oxygen and Hydrogen Peroxide. In Oxidative Delignification Chemistry Fundamentals and Catalysis; Argyropoulos, D.S., Ed.; ACS Symp. Ser. 785; American Chemical Society: Washington, DC, USA, 2001; pp. 61–72. [Google Scholar]
- Argyropoulos, D.S.; Suchy, M. Catalysis and Activation of Oxygen and Peroxide Delignification of Chemical Pulps: A Review. In Oxidative Delignification Chemistry Fundamentals and Catalysis; ACS Symp. Ser. 785; American Chemical Society: Washington, DC, USA, 2001; pp. 2–43. [Google Scholar]
- Elegir, G.; Daina, S.; Zoia, L.; Bestetti, G.; Orlandi, M. Laccase mediator system: Oxidation of recalcitrant lignin model structures in residual kraft lignin. Enzyme Microb. Tech. 2005, 37, 340–346. [Google Scholar] [CrossRef]
- Wang, H.; Shi, Y.; Le, L.; Wang, S.-M.; Wei, J.; Chang, S.-G. POZONE technology to bleach pulp. Ind. Eng. Chem. Res. 1997, 36, 3656–3661. [Google Scholar] [CrossRef]
- Weinstock, I.A.; Atalla, R.H.; Reiner, R.S.; Houtman, C.J.; Hill, C.L. Selective transition-metal catalysis of oxygen delignification using water-soluble salts of polyoxometalates (POM) Anions: Part I. Chemical principles and process concepts. Holzforschung 1998, 52, 304–310. [Google Scholar] [CrossRef]
- Atalla, R.H.; Weinstock, I.A.; Bond, J.S.; Reiner, R.S.; Sonnen, D.M.; Houtman, C.J.; Heintz, R.A.; Hill, C.G.; Hill, C.L.; Wemple, M.W.; Geletii, Yu.V.; Barbuzzi, E.M.G. Polyoxometalate‑based Closed Systems for Oxidative Delignification of Wood Pulp Fibers. In Oxidative Delignification Chemistry Fundamentals and Catalysis; Argyropoulos, D.S., Ed.; ACS Symp. Ser. 785; American Chemical Society: Washington, DC, USA, 2001; pp. 313–326. [Google Scholar]
- Evtuguin, D.V.; Pascoal Neto, C. New polyoxometalate promoted method of oxygen delignification. Holzforschung 1997, 51, 338–342. [Google Scholar] [CrossRef]
- Evtuguin, D.V.; Pascoal Neto, C.; de Jesus, J.D. Bleaching of kraft pulp by oxygen in the presence of polyoxometalates. J. Pulp Paper Sci. 1998, 24, 133–140. [Google Scholar]
- Grigoriev, V.A.; Hill, C.L.; Weinstock, I.A. Polyoxometalate Oxidation of Phenolic Lignin Models. In Oxidative Delignification Chemistry Fundamentals and Catalysis; Argyropoulos, D.S., Ed.; ACS Symp. Ser. 785; American Chemical Society: Washington, DC, USA, 2001; pp. 297–312. [Google Scholar]
- Evtuguin, D.V.; Pascoal Neto, C. Catalytic Oxidative Delignification with Keggin-type Molybdovanadophosphate Heteropolyanions. In Oxidative Delignification Chemistry Fundamentals and Catalysis; Argyropoulos, D.S., Ed.; ACS Symp. Ser. 785; American Chemical Society: Washington, DC, USA, 2001; pp. 342–355. [Google Scholar]
- Kozhevnikov, I.V. Catalysis by heteropoly acids and multicomponent polyoxometalates in liquid-phase reactions. Chem. Rev. 1998, 98, 171–198. [Google Scholar] [CrossRef] [PubMed]
- Evtuguin, D.V.; Pascoal Neto, C. Lignin Degradation Reactions in Aerobic Delignification Catalyzed by Heteropolyanion [PMo7V5O40]8-. In Oxidative Delignification Chemistry Fundamentals and Catalysis; Argyropoulos, D.S., Ed.; ACS Symp. Ser. 785; American Chemical Society: Washington, DC, USA, 2001a; pp. 327–341.
- Weinstock, I.A.; Atalla, R.H.; Reiner, R.S.; Moen, M.A.; Hammel, K.E.; Houtman, C.J.; Hill, C.L. A new environmentally benign technology and approach to bleaching kraft pulp. polyoxometalates for selective delignification and waste mineralization. New. J. Chem. 1996, 20, 269–275. [Google Scholar]
- Aguié-Béghin, V.; Baumberger, S.; Monties, B.; Douillard, R. Formation and characterization of spread lignin layers at the air/water interface. Langmuir 2002, 18, 5190–5196. [Google Scholar] [CrossRef]
- Reiner, R.S.; Weinstock, I.A.; Atalla, R.H.; Bond, J.S.; Sonnen, D.M.; Houtman, C.J.; Heintz, R.A.; Springer, E.L.; Wemple, M.; Hill, C.L. Thermodynamically Stable, Self-Buffering Polyoxometalate Delignification System. In Proceedings of the 11th ISWPC, Nice, France, June 2001; Volume III, pp. 349–352.
- Weinstock, I.A.; Barbuzzi, E.M.G.; Wemple, M.; Cowan, J.J.; Reiner, R.S.; Sonnen, D.M.; Heintz, R.A.; Bond, J.S.; Hill, C.L. Equilibrating metal-oxide cluster ensembles for oxidation reactions using oxygen in water. Nature 2001, 414, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, I.A.; Hammel, K.E.; Moen, M.A.; Landucci, L.L.; Ralph, S.; Sullivan, C.E.; Reiner, R.S. Selective transition-metal catalysis of oxygen delignification using water-soluble salts of POM anions. Part. II. Reactions of α-[SiVW11O40]5- with phenolic lignin-model compounds. Holzforschung 1998, 52, 311–318. [Google Scholar] [CrossRef]
- Yokoyama, T.; Chang, H.-M.; Reiner, R.S.; Atalla, R.H.; Weinstock, I.A.; Kadla, J.F. Polyoxometalate oxidation of non-phenolic lignin subunits in water: effect of substrate structure on reaction kinetics. Holzforschung 2004, 58, 116–121. [Google Scholar] [CrossRef]
- Kim, Y.S.; Chang, H.-M.; Kadla, J.F. Polyoxometalate (POM) oxidation of lignin model compounds. Holzforschung 2008, 61, 38–49. [Google Scholar]
- Kim, Y.S.; Chang, H.-M.; Kadla, J.F. Polyoxometalate (POM) oxidation of milled wood lignin (MWL). J. Wood Chem. Technol. 2007, 27, 225–241. [Google Scholar] [CrossRef]
- Bujanovic, B.; Reiner, R.S.; Hirth, K.C.; Ralph, S.A.; Atalla, R.H. Studies of Lignin Transformation in Polyoxometalate (POM) Bleaching of Kraft Pulp. In Proceedings of the 13th ISWFPC, Auckland, New Zealand, May 2005; Volume 3, pp. 49–56.
- Bujanovic, B.; Reiner, R.S.; Agarwal, U.P.; Ralph, S.A.; Atalla, R.H. Structural Changes of Residual Lignin of Softwood and Hardwood Kraft Pulp upon Oxidative Treatment with Polyoxometalates. In Proceedings of the 2005 TAPPI Engineering, Pulping and Environmental Conference, Philadelphia, PA, USA, August 2005; pp. 89–92.
- Bujanovic, B.; Ralph, S.A.; Reiner, R.S.; Atalla, R.H. Lignin modification in the initial phase of softwood kraft pulp delignification with polyoxometalates (POMs). Holzforschung 2007, 61, 492–498. [Google Scholar] [CrossRef]
- Bujanovic, B.; Hirth, K.C.; Ralph, S.A.; Reiner, R.S.; Atalla, R.H. Composition of the Organic Components in Polyoxometalate (POM) Liquors from Kraft pulp Bleaching. In Presented at the 14th ISWFPC, Durban, South Africa, June 2007.
- Li, J.; Gellerstedt, G. Improved lignin properties and reactivity by modifications in the autohydrolysis process of aspen wood. Ind. Crop. Prod. 2008, 175–181. [Google Scholar] [CrossRef]
- Bujanovic, B.; Reiner, R.S.; Ralph, S.A.; Atalla, R.H. Polyoxometalate delignification of birch kraft pulp and effect on residual lignin. J. Wood Chem. Technol. 2010. submitted. [Google Scholar]
- Weinstock, I.A.; Minor, J.L.; Reiner, R.S.; Agarwal, U.P.; Atalla, R.H. Raman and UV Visible Spectroscopic Studies of a Highly Selective Polyoxometalate Bleaching System. In Proceedings of the 1993 Pulping Conference, Atlanta, GA, USA, November 1993; pp. 519–532.
- Jääskeläinen, A.S.; Sun, Y.; Argyropoulos, D.S.; Tamminen, T.; Hortling, B. The effect of isolation method on the chemical structure of residual lignin. Wood Sci. Technol. 2003, 37, 91–102. [Google Scholar] [CrossRef]
- Jiang, Z.-H.; Argyropoulos, D.S. Isolation and characterization of residual lignins in kraft pulps. J. Pulp Paper Sci. 1999, 25, 25–29. [Google Scholar]
- Balakshin, M.Y.; Capanema, E.A; Chen, C.-L.; Gracz, H. Elucidation of the structures of residual and dissolved pine kraft lignin using an HMQC NMR technique. J. Agric. Food. Chem. 2003, 51, 6116–6127. [Google Scholar]
- Gaspar, A.R.; Evtuguin, D.V.; Pascoal Neto, C. New Highly Selective Oxygen Delignification of Kraft Pulp Promoted by Mn-Assisted Polyoxometalate. In Proceedings of the 11th ISWPC, Nice, France, June 2001; Volume I, pp. 227–230.
- Gaspar, A.R.; Evtuguin, D.V.; Pascoal Neto, C. Polyoxometalate-catalyzed oxygen delignification of kraft pulp: A pilot-plant experience. Ind. Eng. Chem. Res. 2004, 43, 7754–7761. [Google Scholar] [CrossRef]
- Gaspar, A.R.; Evtuguin, D.V.; Pascoal Neto, C. Lignin reactions in oxygen delignification catalysed by Mn(II)-substituted molybdovanadophosphate polyanion. Holzforschung 2004, 58, 640–649. [Google Scholar]
- Evtuguin, D.V.; Pascoal Neto, C.; Carapuça, H.; Soares, J. Lignin degradation in oxygen delignification catalysed by [PMo7V5O40]8- Polyanion: Part II. Study on lignin monomeric model compounds. Holzforschung 2000, 54, 511–518. [Google Scholar]
- Evtuguin, D.V.; Daniel, A.I.D.; Silvestre, A.J.D.; Amado, F.M.L.; Pascoal Neto, C. Lignin aerobic oxidation promoted by molybdovanadophosphate polyanion by [PMo7V5O40]8-. Study on the oxidative cleavage of β-O-4 aryl ether structures using model compounds. J. Mol. Catal. A. Chem. 2000, 154, 217–224. [Google Scholar] [CrossRef]
- Evtuguin, D.V.; Pascoal Neto, C.; Rocha, J. Lignin degradation in oxygen delignification catalysed by [PMo7V5O40]8- polyanion. Part I. Study on wood lignin. Holzforschung 2000, 54, 381–389. [Google Scholar]
- Gamelas, J.A.F.; Tavares, A.P.M.; Evtuguin, D.V.; Xavier, A.M.B. Oxygen bleaching of kraft pulp with polyoxometalates and laccase applying a novel multi-stage process. J. Mol. Catal. B: Enzym. 2005, 33, 57–64. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bujanovic, B.; Ralph, S.; Reiner, R.; Hirth, K.; Atalla, R. Polyoxometalates in Oxidative Delignification of Chemical Pulps: Effect on Lignin. Materials 2010, 3, 1888-1903. https://doi.org/10.3390/ma3031888
Bujanovic B, Ralph S, Reiner R, Hirth K, Atalla R. Polyoxometalates in Oxidative Delignification of Chemical Pulps: Effect on Lignin. Materials. 2010; 3(3):1888-1903. https://doi.org/10.3390/ma3031888
Chicago/Turabian StyleBujanovic, Biljana, Sally Ralph, Richard Reiner, Kolby Hirth, and Rajai Atalla. 2010. "Polyoxometalates in Oxidative Delignification of Chemical Pulps: Effect on Lignin" Materials 3, no. 3: 1888-1903. https://doi.org/10.3390/ma3031888
APA StyleBujanovic, B., Ralph, S., Reiner, R., Hirth, K., & Atalla, R. (2010). Polyoxometalates in Oxidative Delignification of Chemical Pulps: Effect on Lignin. Materials, 3(3), 1888-1903. https://doi.org/10.3390/ma3031888




