Injectable, Biodegradable Hydrogels for Tissue Engineering Applications
Abstract
:1. Introduction
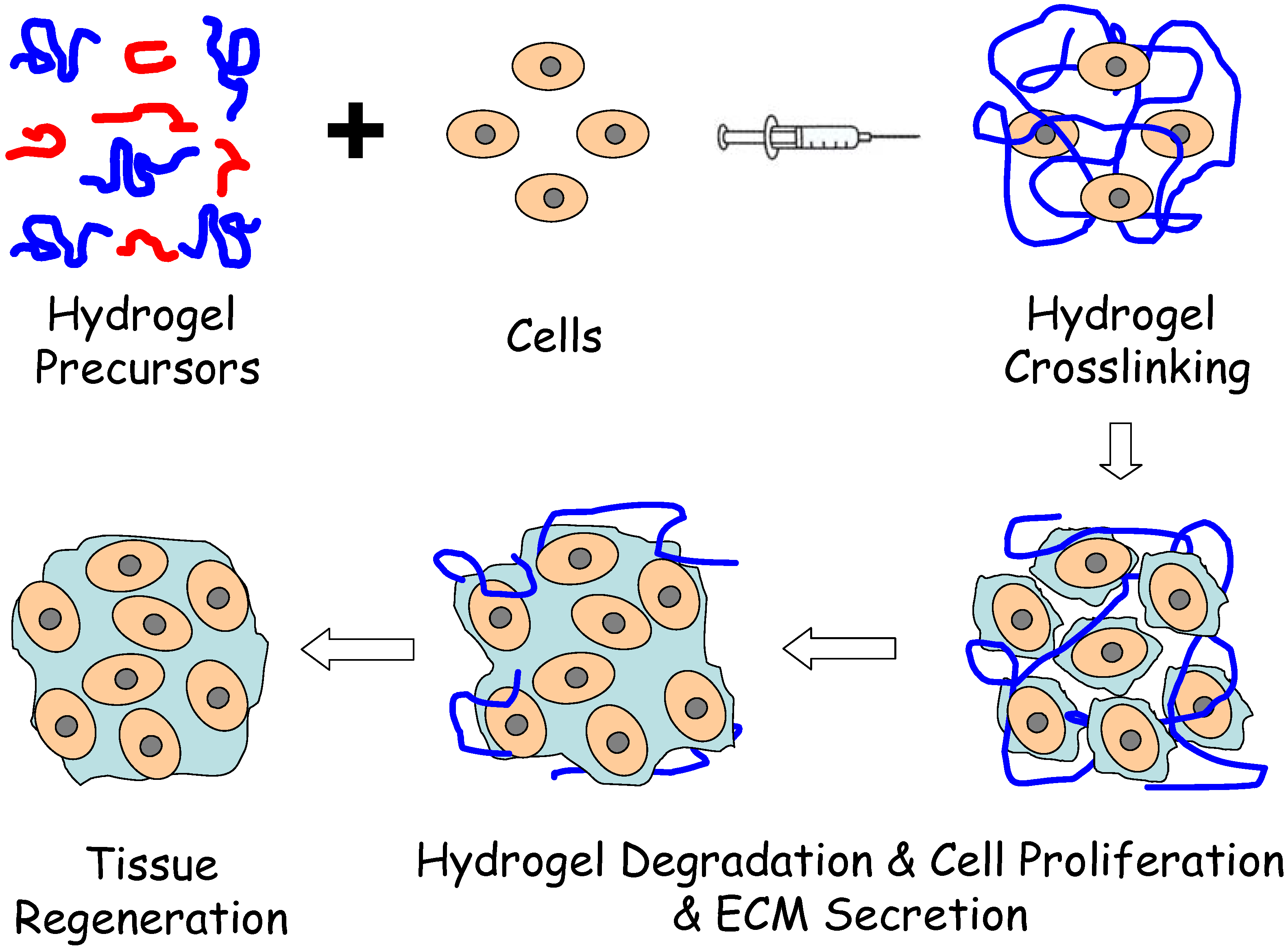
| Hydrogels | Polymers | Gelation Mechanism |
|---|---|---|
| Natural hydrogels | Collagen/Gelatin | Thermal/Chemical crosslinking |
| Chitosan | Thermal/Chemical/Schiff-base reaction/Free radical crosslinking | |
| Hyaluronic acid | Thermal/Chemical/Schiff-base reaction/Michael-type addition/Free radical crosslinking | |
| Chondroitin sulfate | Free radical crosslinking | |
| Alginate | Ionic/Free radical crosslinking | |
| Agar/Agarose | Thermal crosslinking | |
| Fibrin | Thermal crosslinking | |
| Synthetic hydrogels | PEG/PEO | Michael-type addition/Chemical/Free radical crosslinking |
| PVA | Chemical/Free radical crosslinking | |
| PPF/OPF | Free radical crosslinking | |
| PNIPAAm | Thermal crosslinking | |
| PEO-PPO-PEO PLGA-PEG-PLGA PEG-PLLA-PEG | Thermal crosslinking | |
| Poly(aldehyde guluronate) | Chemical crosslinking | |
| Polyanhydrides | Free radical crosslinking |
2. Biodegradable Materials for Injectable Hydrogels
2.1. Natural Materials
2.2. Synthetic Materials
3. Injectable Hydrogel Systems
3.1. Physical Crosslinking of Hydrogels
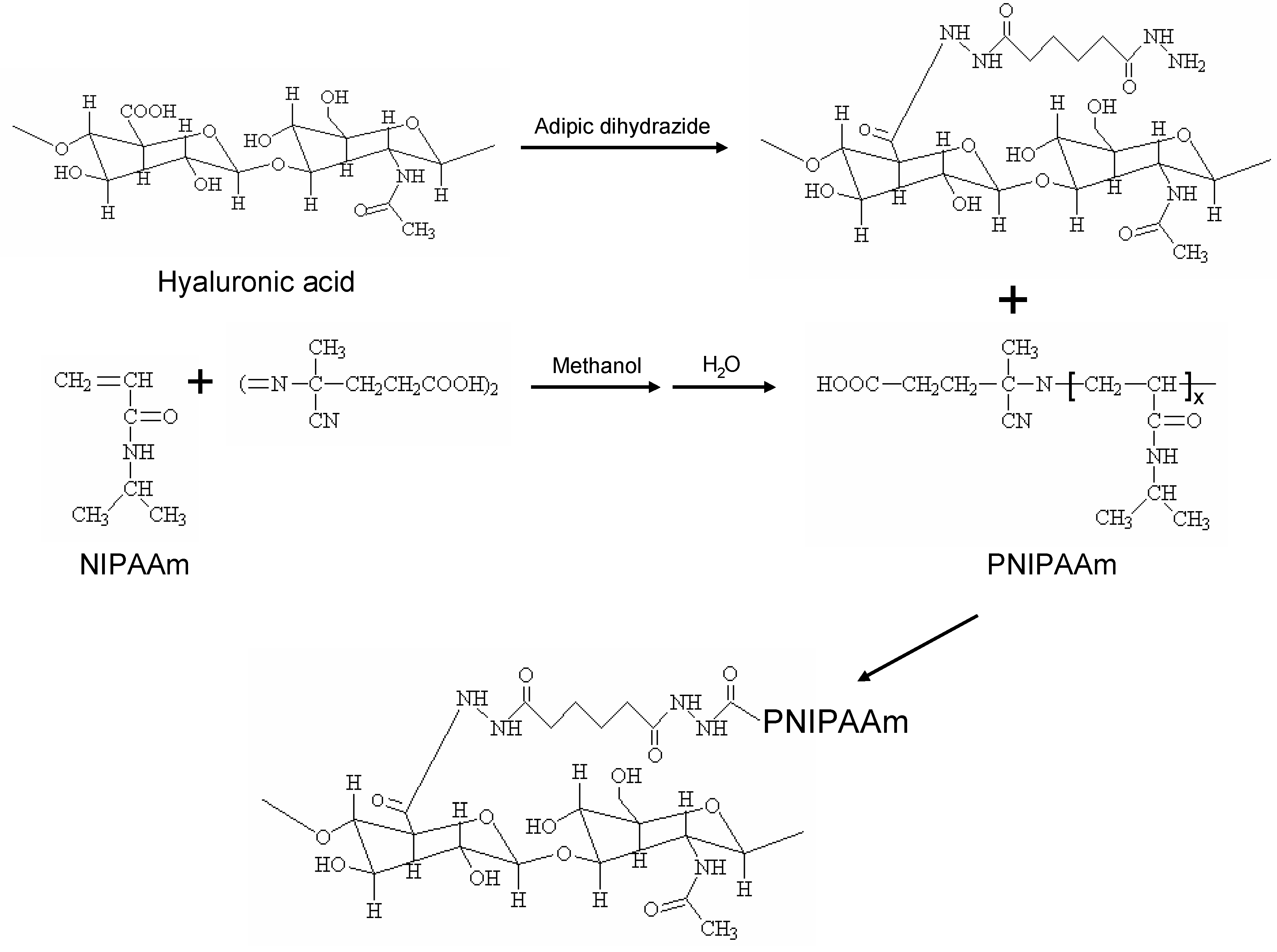
3.2. Chemical Crosslinking of Hydrogels
3.2.1. Free radical polymerization
3.2.2. Michael-type addition reaction hydrogels
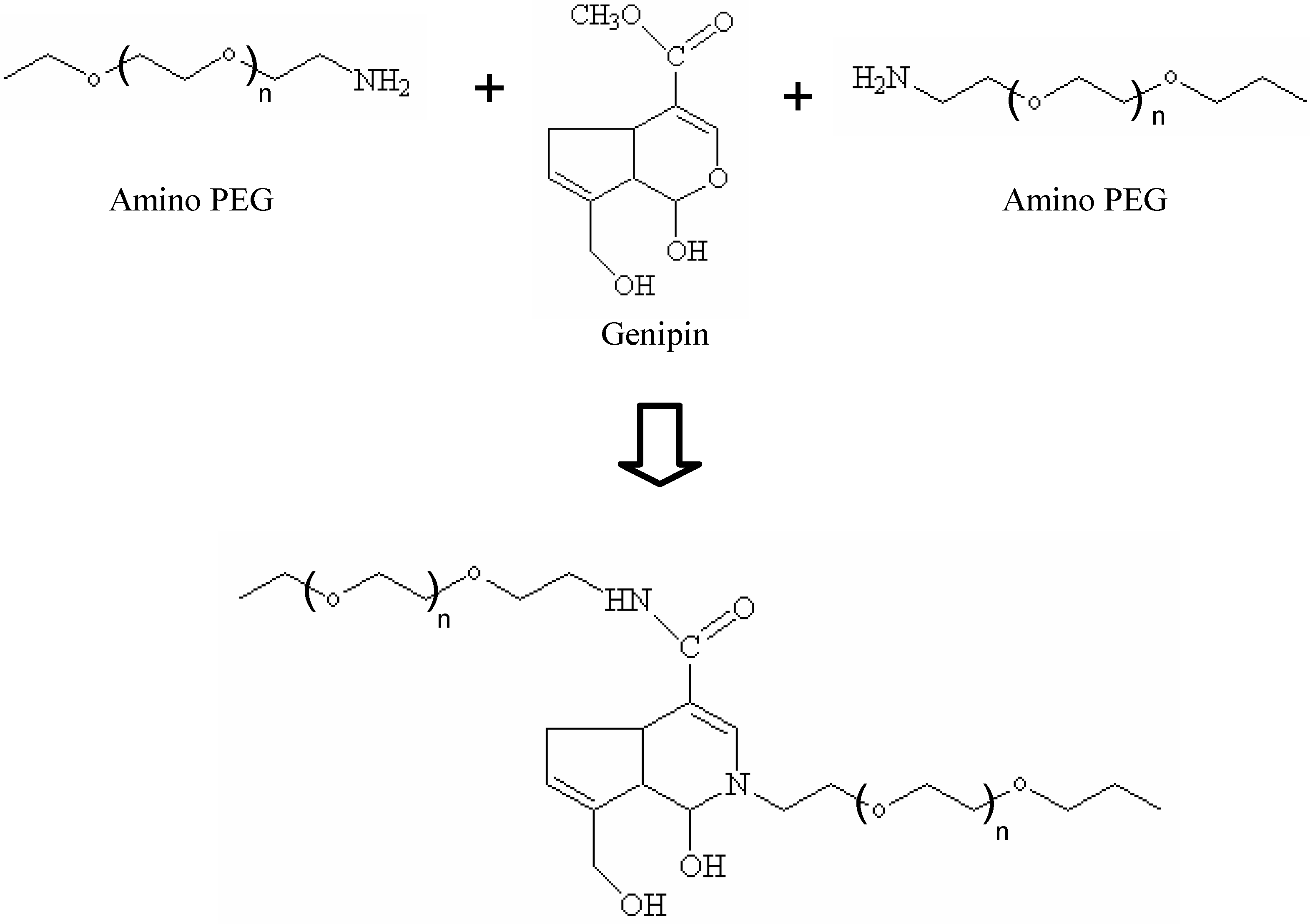
3.2.3. Genipin crosslinked hydrogels
3.2.4. Schiff-base crosslinked hydrogels
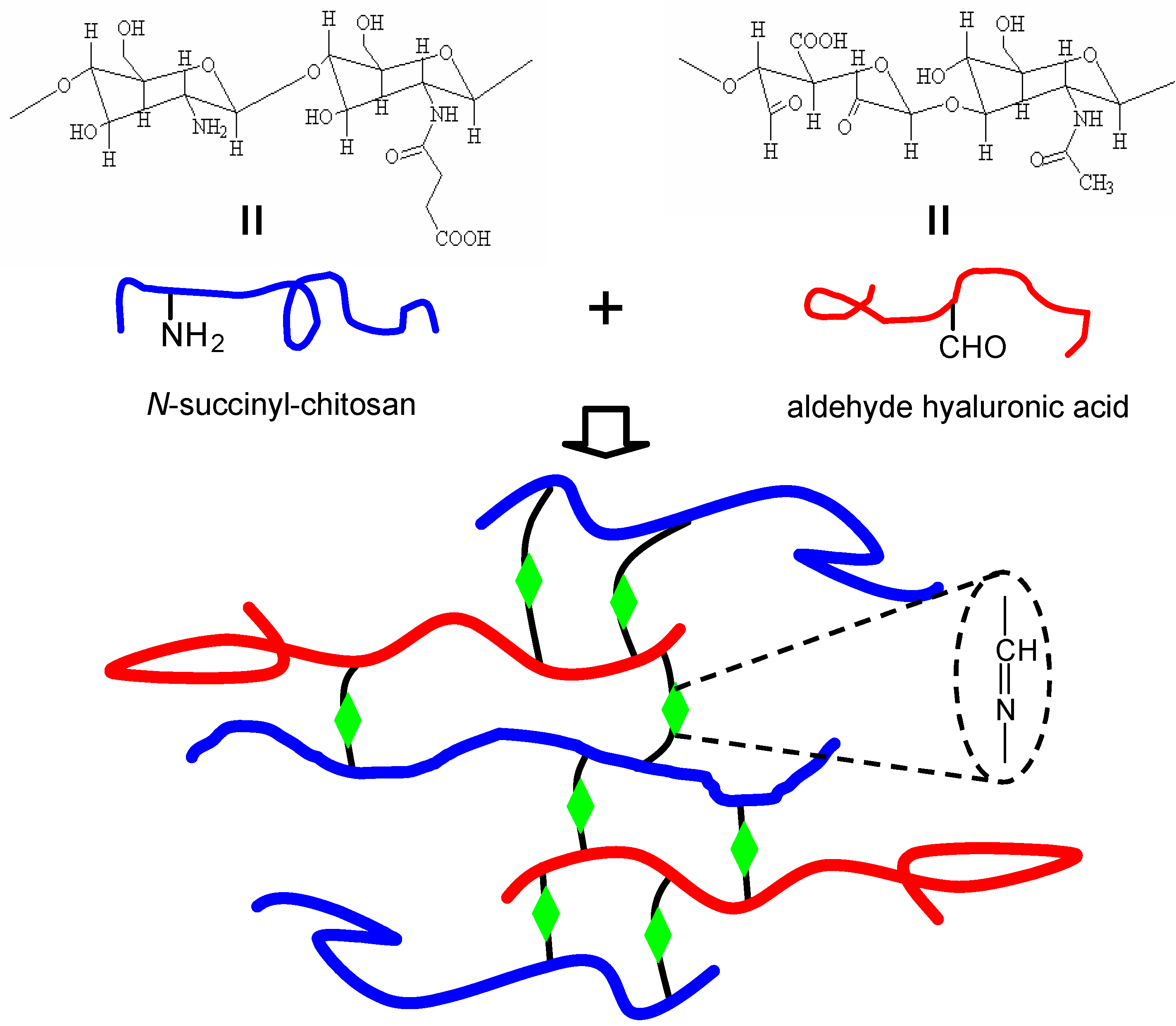
4. Applications of Injectable Hydrogels
4.1. Clinical Applications
4.2. Cartilage Repair
4.3. Soft Tissue Regeneration
5. Conclusions
References and Notes
- Lee, K.Y.; Mooney, D.J. Hydrogels for tissue engineering. Chem. Rev. 2001, 101, 1869–1879. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef] [PubMed]
- Tememoff, J.S.; Mikos, A.G. Injectable biodegradable materials for orthopedic tissue engineering. Biomaterials 2000, 21, 2405–2412. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.P.; De Bank, P.A.; Shakesheff, K.M. Injectable scaffolds for tissue regeneration. J. Mater. Chem. 2004, 14, 1915–1923. [Google Scholar] [CrossRef]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef] [PubMed]
- Nuttelman, C.R.; Rice, M.A.; Rydholm, A.E.; Salinas, C.N.; Shah, D.N.; Anseth, K.S. Macromolecular monomers for the synthesis of hydrogel niches and their application in cell encapsulation and tissue engineering. Prog. Polym. Sci. 2008, 33, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Brandl, F.; Sommer, F.; Goepferich, A. Rational design of hydrogels for tissue engineering: Impact of physical factors on cell behavior. Biomaterials 2007, 28, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Rehfeldt, R.; Engler, A.J.; Eckhardt, A.; Ahmed, F.; Discher, D.E. Cell responses to the mechanochemical microenvironment--implications for regenerative medicine and drug delivery. Adv. Drug Deliv. Rev. 2007, 59, 1329–1339. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, G.D.; Bryant, S.J. Cell encapsulation in biodegradable hydrogels for tissue Engineering applications. Tissue Eng. 2008, 14, 149–165. [Google Scholar] [CrossRef]
- Yu, L.; Ding, J. Injectable hydrogels as unique biomedical materials. Chem. Soc. Rev. 2008, 37, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Gong, Y.; Lao, L.; Mao, Z.; Gao, C. Gelatin/chitosan/hyaluronan ternary complex scaffold containing basic fibroblast growth factor for cartilage tissue engineering. J. Mater. Sci.: Mater. Med. 2007, 18, 1961–1968. [Google Scholar] [CrossRef]
- Awad, H.A.; Wickham, M.Q.; Leddy, H.A.; Gimble, J.M.; Guilak, F. Chondrogenic differentiation of adipose-derived adult stem cells in agarose, alginate, and gelatin scaffolds. Biomaterials 2004, 25, 3211–3222. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Wan, L.; Wu, J.; Gao, C. Microscale control over collagen gradient on poly(L-lactide) membrane surface for manipulating chondrocyte distribution. Colloids Surf. B: Biointerfaces 2008, 67, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Singla, A.; Lee, Y. Biomedical applications of collagen. Int. J. Pharm. 2001, 221, 1–22. [Google Scholar] [CrossRef]
- Furthmayr, H.; Timol, R. Immunochemistry of collagens and procollagens. Int. Rev. Connect. Tiss. Res. 1976, 7, 61–99. [Google Scholar]
- Yannas, I.V.; Burke, J.F. Design of an artificial skin I. Basic design principles. J. Biomed. Mater. Res. 1980, 14, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Stefan, M.M.; Shortkroff, S.; Schneider, T.O.; Breinan, H.A.; Yannas, I.V.; Spector, M. Meniscus cells seeded in type I and type II collagen-GAG matrices in vitro. Biomaterials 1999, 20, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Huang, D.; Lao, L.; Gao, C. RGD modified PLGA/gelatin microspheres as microcarriers for chondrocyte delivery. J. Biomed. Mater. Res. 2009, 91B, 228–238. [Google Scholar] [CrossRef]
- Huang, Y.; Onyeri, S.; Siewe, M.; Moshfeghian, A.; Madihally, S.V. In vitro characterization of chitosan-gelatin scaffolds for tissue engineering. Biomaterials 2005, 26, 7616–7627. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Lao, L.; Wu, J.; Gong, Y.; Gao, C. Biomimetic modification of chitosan with covalently grafted lactose and blended heparin for improvement of in vitro cellular interaction. Polym. Adv. Technol. 2008, 19, 15–23. [Google Scholar] [CrossRef]
- Hsieh, W.C.; Chang, C.P.; Lin, S.M. Morphology and characterization of 3D micro-porous structured chitosan scaffolds for tissue engineering. Colloids Surf. B: Biointerfaces 2007, 57, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Chesnutt, B.M.; Utturkar, G.; Haggard, W.O.; Yang, Y.; Ong, J.L.; Bumgardner, J.D. The effect of cross-linking of chitosan microspheres with genipin on protein release. Carbohydr. Polym. 2007, 68, 561–567. [Google Scholar] [CrossRef]
- Berger, J.; Reist, M.; Mayer, J.M.; Felt, O.; Peppas, N.A.; Gurny, R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Europ. J. Pharm. Biopharm. 2004, 57, 19–34. [Google Scholar] [CrossRef]
- Mao, J.; Zhao, L.; Yao, K.; Shang, Q.; Yang, G.; Cao, Y. Study of novel chitosan-gelatin artificial skin in vitro. J. Biomed. Mater. Res. 2003, 64A, 301–308. [Google Scholar] [CrossRef]
- Ma, J.; Wang, H.; He, B.; Chen, J. A preliminary in vitro study on the fabrication and tissue engineering applications of a novel chitosan bilayer material as a scaffold of human neofetal dermal fibroblasts. Biomaterials 2001, 22, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Chatelet, C.; Damour, O.; Domard, A. Influence of the degree of acetylation on some biological properties of chitosan films. Biomaterials 2001, 22, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Goa, K.L.; Benfield, P. Hyaluronic acid. A review of its pharmacology and use as a surgical aid in ophthalmology, and its therapeutic potential in joint disease and wound healing. Drugs 1994, 47, 536–566. [Google Scholar] [CrossRef] [PubMed]
- Toole, B.P. Hyaluronan and its binding proteins, the hyaladherins. Curr. Opin. Cell Biol. 1990, 2, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Wu, J.; Lao, L.; Gao, C. Gelatin/chitosan/hyaluronan scaffold integrated with PLGA microspheres for cartilage tissue engineering. Acta Biomater. 2009, 5, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Fraser, J.R.; Laurent, T.C.; Laurent, U.B. Hyaluronan: its nature, distribution, functions and turnover. J. Intern. Med. 1997, 242, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Dowthwaite, G.P.; Edwards, J.C.; Pitsillides, A.A. An essential role for the interaction between hyaluronan and hyaluronan binding proteins during joint development. J. Histochem. Cytochem. 1998, 46, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Cheung, W.F.; Crue, T.F.; Turley, E.A. Receptor for hyaluronan mediated motility (RHAMM), a hyaladherin that regulates cell responses to growth factors. Biochem. Soc. Trans. 1999, 27, 135–142. [Google Scholar] [PubMed]
- Entwistle, J.; Hall, C.L.; Turley, E.A. Hyaluronan receptors: regulators of signalling to the cytoskeleton. J. Cell Biochem. 1996, 61, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.S.; Thompson, A.Y.; Heidaran, M.A.; Poser, J.W.; Spiro, R.C. An osteoconductive collagen/hyaluronate matrix for bone regeneration. Biomaterials 1999, 20, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Campoccia, D.; Doherty, P.; Radice, M.; Brun, P.; Abatangelo, G.; Williams, D.F. Semisynthetic resorbable materials from hyaluronan esterification. Biomaterials 1998, 23, 2101–2127. [Google Scholar] [CrossRef]
- Rowley, J.A.; Madlambayan, G.; Mooney, D.J. Alginate hydrogels as synthetic extracellular matrix materials. Biomaterials 1999, 20, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Kong, H.J.; Larson, R.G.; Mooney, D.J. Hydrogel formation via cell crosslinking. Adv. Mater. 2003, 15, 1828–1832. [Google Scholar] [CrossRef]
- Cao, Y.; Shen, X.C.; Chen, Y.; Guo, J.; Chen, Q.; Jiang, X.Q. pH-Induced self-assembly and capsules of sodium alginate. Biomacromolecules 2005, 6, 2189–2196. [Google Scholar] [CrossRef] [PubMed]
- Paige, K.T.; Cima, L.G.; Yaremchuk, M.J.; Vacanti, J.P.; Vacanti, C.A. Injectable cartilage. Plast. Reconstr. Surg. 1995, 96, 1390–1400. [Google Scholar] [CrossRef] [PubMed]
- Paige, K.T.; Cima, L.G.; Yaremchuk, M.J.; Schloo, B.L.; Vacanti, J.P.; Vacanti, CA. De novo cartilage generation using calcium alginate-chondrocyte constructs. Plast. Reconstr. Surg. 1996, 97, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Skjak-Braerk, G.; Grasdalen, H.; Smidsrod, O. Inhomogeneous polysaccharide ionic gels. Carbohydr. Polym. 1989, 10, 31–54. [Google Scholar] [CrossRef]
- Stevens, M.M.; Qanadilo, H.F.; Langer, R.; Shastri, V.P. A rapid-curing alginate gel system: Utility in periosteum-derived cartilage tissue engineering. Biomaterials 2004, 25, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.K.; Ma, P.X. Ionically crosslinked alginate hydrogels as scaffolds for tissue engineering: Part 1. Structure, gelation rate and mechanical properties. Biomaterials 2001, 22, 511–521. [Google Scholar] [CrossRef] [PubMed]
- Bouhadir, K.H.; Lee, K.Y.; Alsberg, E.; Damm, K.L.; Anderson, K.W.; Mooney, D.J. Degradation of partially oxidized alginate and its potential application for tissue engineering. Biotechnol. Prog. 2001, 17, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.J.; Alsberg, E.; Kaigler, D.; Lee, K.Y.; Mooney, D.J. Controlling degradation of hydrogel via the size of cross-linked junctions. Adv. Mater. 2004, 16, 1917–1921. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, B.; Jayakrishnan, A. Self-cross-linking biopolymers as injectable in situ forming biodegradable scaffolds. Biomaterials 2005, 26, 3941–3951. [Google Scholar] [CrossRef] [PubMed]
- Behravesh, E.; Sikavitsas, V.I.; Mikos, A.G. Quantification of ligand surface concentration of bulk-modified biomimetic hydrogels. Biomaterials 2003, 24, 4365–4374. [Google Scholar] [CrossRef] [PubMed]
- Anseth, K.S.; Metters, A.T.; Bryant, S.J.; Martens, P.J.; Elisseeff, J.H.; Bowman, C.N. In situ forming degradable networks and their application in tissue engineering and drug delivery. J. Control. Rel. 2002, 78, 199–209. [Google Scholar] [CrossRef]
- Guillaudeu, S.J.; Fox, M.E.; Haidar, Y.M.; Dy, E.E.; Szoka, F.C.; Fréchet, J.M.J. PEGylated dendrimers with core functionality for biological applications. Bioconjugate. Chem. 2008, 19, 461–469. [Google Scholar] [CrossRef]
- Feng, X.; Taton, D.; Chaikof, E.L.; Gnanou, Y. Bouquet-type dendrimer-like poly(ethylene Oxide)s with a focal aldehyde and peripheral hydroxyls. Biomacromolecules 2007, 8, 2374–2378. [Google Scholar] [CrossRef] [PubMed]
- Hiemstra, C.; Zhong, Z.; Li, L.; Dijkstra, P.J.; Feijen, J. In-situ formation of biodegradable hydrogels by stereocomplexation of PEG-(PLLA)8 and PEG-(PDLA)8 star block copolymers. Biomacromolecules 2006, 7, 2790–2795. [Google Scholar] [CrossRef] [PubMed]
- Hiemstra, C.; Zhong, Z.; van Tomme, S.R.; van Steenbergen, M.J.; Jacobs, J.J.L.; Otter, W.D.; Hennink, W.E.; Feijen, J. In vitro and in vivo protein delivery from in situ forming poly(ethylene glycol)–poly(lactide) hydrogels. J. Control. Rel. 2007, 119, 320–327. [Google Scholar] [CrossRef]
- Holland, T.A.; Tessmar, J.K.; Tabata, Y.; Mikos, A.G. Transforming growth factor-beta1 release from oligo(poly(ethylene glycol) fumarate) hydrogels in conditions that model the cartilage wound healing environment. J. Control. Rel. 2004, 94, 101–114. [Google Scholar] [CrossRef]
- Wieland, J.A.; Houchin-Ray, T.L.; Shea, L.D. Non-viral vector delivery from PEG-hyaluronic acid hydrogels. J. Control. Rel. 2007, 120, 233–241. [Google Scholar] [CrossRef]
- Leach, J.B.; Bivens, K.A.; Collins, C.N.; Schmidt, C.E. Development of photocrosslinkable hyaluronic acidpolyethylene glycol-peptide composite hydrogels for soft tissue engineering. J. Biomed. Mater. Res. 2004, 70A, 74–82. [Google Scholar] [CrossRef]
- Almany, L.; Seliktar, D. Biosynthetic hydrogel scaffolds made from fibrinogen and polyethylene glycol for 3D cell cultures. Biomaterials 2005, 26, 2467–2477. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, N.; Ramay, H.R.; Gunn, J.; Matsen, F.A.; Zhang, M. PEG-grafted chitosan as an injectable thermosensitive hydrogel for sustained protein release. J. Control. Rel. 2005, 103, 609–624. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Chae, B.S.; Zhang, L.; Kiick, K.L.; Furst, E.M. Rheological characterization of polysaccharide-poly(ethylene glycol) star copolymer hydrogels. Biomacromolecules 2005, 6, 1931–1940. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, G.D.; Villanueva, I.; Bryant, S.J. Mechanical stimulation of TMJ condylar chondrocytes encapsulated in PEG hydrogels. J. Biomed. Mater. Res. 2007, 83A, 323–331. [Google Scholar] [CrossRef]
- Hudalla, G.A.; Eng, T.S.; Murphy, W.L. An approach to modulate degradation and mesenchymal stem cell behavior in poly(ethylene glycol) networks. Biomacromolecules 2008, 9, 842–849. [Google Scholar] [CrossRef] [PubMed]
- Weber, L.M.; Cheung, C.Y.; Anseth, K.S. Multifunctional pancreatic islet encapsulation barriers achieved via multilayer PEG hydrogels. Cell Transplant. 2008, 16, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.D.; Stayton, P.S.; Hoffman, A.S. Semi-interpenetrating network of poly(ethylene glycol) and poly(D,L-lactide) for the controlled delivery of protein drugs. J. Biomater. Sci. Polym. Edn. 2005, 16, 189–201. [Google Scholar] [CrossRef]
- Cascone, M.G.; Laus, M.; Ricci, D.; Sbarbati Del Guerra, R. Evaluation of poly(vinyl alcohol) hydrogels as a component of hybrid artificial tissues. J. Mater. Sci.: Mater. Med. 1995, 6, 71–75. [Google Scholar] [CrossRef]
- Nuttelman, C.R.; Mortisen, D.J.; Henry, S.M.; Anseth, K.S. Attachment of fibronectin to poly(vinyl alcohol) hydrogels promotes NIH3T3 cell adhesion, proliferation, and migration. J. Biomed. Mater. Res. 2001, 57, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Chenite, A.; Chaput, C.; Wang, D.; Combes, C.; Buschmann, M.D.; Hoemann, C.D.; Leroux, J.C.; Atkinson, B.L.; Binette, F.; Selmani, A. Novel injectable neutral solutions of chitosan from biodegradable gels in situ. Biomaterials 2000, 21, 2155–2161. [Google Scholar] [CrossRef] [PubMed]
- Chenite, A.; Buschmann, M.; Wang, D.; Chaput, C.; Kandani, N. Rheological characterisation of thermogelling chitosan/glycerol–phosphate solutions. Carbohydr. Polym. 2001, 46, 39–46. [Google Scholar] [CrossRef]
- Crompton, K.E.; Prankerd, R.J.; Paganin, D.M.; Scott, T.F.; Horne, M.K.; Finkelstein, D.I.; Gross, K.A.; Forsythe, J.S. Morphology and gelation of thermosensitive chitosan hydrogels. Biophys. Chem. 2005, 117, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Hoemann, C.D.; Sun, J.; Legare, A.; McKee, M.D.; Buschmann, M.D. Tissue engineering of cartilage using an injectable and adhesive chitosan-based cell-delivery vehicle. Osteoarthritis Cartilage 2005, 13, 318–329. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Yamato, M.; Isoi, Y.; Akutsu, T.; Setomaru, T.; Abe, K.; Kikuchi, A.; Umezu, M.; Okano, T. Fabrication of pulsatile cardiac tissue grafts using a novel 3-dimensional cell sheet manipulation technique and temperature-responsive cell culture surfaces. Circ. Res. 2002, 90, 40–48. [Google Scholar] [CrossRef]
- Gan, T.; Zhang, Y.; Guan, Y. In situ gelation of P(NIPAM-HEMA) microgel dispersion and its applications as injectable 3D cell scaffold. Biomacromolecules 2009, 10, 1410–1415. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, S.S.; Kim, S.J.; Lee, Y.M. Rapid temperature/pH response of porous alginate-g-poly(N-isopropylacrylamide) hydrogels. Polymer 2002, 43, 7549–7558. [Google Scholar] [CrossRef]
- Wang, L.Q.; Tu, K.; Li, Y.; Zhang, J.; Jiang, L.; Zhang, Z. Synthesis and characterization of temperature responsive graft copolymers of dextran with poly(N-isopropylacrylamide). React Funct. Polym. 2002, 53, 19–27. [Google Scholar] [CrossRef]
- Lin, H.H.; Cheng, Y.L. In-situ thermoreversible gelation of block and star copolymers of poly(ethylene glycol) and poly(N-isopropylacrylamide) of varying architectures. Macromolecules 2001, 34, 3710–3715. [Google Scholar] [CrossRef]
- Stile, R.A.; Burghardt, W.R.; Healy, K.E. Synthesis and characterization of injectable poly(N-isopropylacrylamide)-based hydrogels that support tissue formation in vitro. Macromolecules 1999, 32, 7370–7379. [Google Scholar] [CrossRef]
- Lee, S.B.; Ha, D.I.; Cho, S.K.; Kim, S.J.; Lee, Y.M. Temperature/pH-sensitive comb-type graft hydrogels composed of chitosan and poly(N-isopropylacrylamide). J. Appl. Polym. Sci. 2004, 92, 2612–2620. [Google Scholar] [CrossRef]
- Lee, J.W.; Jung, M.C.; Park, H.D.; Park, K.D.; Ryu, G.H. Synthesis and characterization of thermosensitive chitosan copolymer as a novel biomaterial. J. Biomed. Mater. Res. 2004, 15, 1065–1079. [Google Scholar]
- Wang, J.; Chen, L.; Zhao, Y.; Guo, G.; Zhang, R. Cell adhesion and accelerated detachment on the surface of temperature-sensitive chitosan and poly(N-isopropylacrylamide) hydrogels. J. Mater. Sci: Mater. Med. 2009, 20, 583–590. [Google Scholar] [CrossRef]
- Chen, J.P.; Cheng, T.H. Thermo-responsive chitosan-graft-poly(N-isopropylacrylamide) injectable hydrogel for cultivation of chondrocytes and meniscus cells. Macromol. Biosci. 2006, 6, 1026–1039. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Ramirez, C.M.; Miljkovic, N.; Li, H.; Rubin, J.P.; Marra, K.G. Thermosensitive Injectable Hyaluronic Acid Hydrogel for Adipose Tissue Engineering. Biomaterials 2009, 30, 6844–6853. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.H.; Kim, S.H.; Park, K.D.; Jung, M.C.; Yang, W.I.; Han, S.W.; Noh, J.Y.; Jin, J.W.; Lee, W. Chondrogenic differentiation of human mesenchymal stem cells using a thermosensitive poly(N-isopropylacrylamide) and water-soluble chitosan copolymer. Biomaterials 2004, 25, 5743–5751. [Google Scholar] [CrossRef] [PubMed]
- Ha, D.I.; Lee, S.B.; Chong, M.S.; Lee, Y.M.; Kim, S.Y.; Park, Y.H. Preparation of thermo-responsive and injectable hydrogels based on hyaluronic acid and poly(N-isopropylacrylamide) and their drug release behaviors. Macromol. Res. 2006, 14, 87–93. [Google Scholar] [CrossRef]
- Guan, J.; Hong, Y.; Ma, Z.; Wagner, W. Protein-reactive, thermoresponsive copolymers with high flexibility and biodegradability. Biomacromolecules 2008, 9, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Ohya, S.; Nakayama, Y.; Matsuda, T. Thermoresponsive artificial extracellular matrix for tissue engineering: hyaluronic acid bioconjugated with poly(N-isopropylacrylamide) grafts. Biomacromolecules 2001, 2, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Ibusuki, S.; Fujii, Y.; Iwamoto, Y.; Matsuda, T. Tissue-engineered cartilage using an injectable and in situ gelable thermoresponsive gelatin: fabrication and in vitro performance. Tissue Eng. 2003, 9, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, B.; Vidts, A.; Bulcke, A.; Verbeeck, R.; Schacht, E. Synthesis and thermal properties of poly(ethylene glycol)-poly(ε-caprolactone) copolymers. Polymer 1998, 39, 1631–1636. [Google Scholar] [CrossRef]
- Shim, W.S.; Kim, J.-H.; Park, H.; Kim, K.; Kwon, I.C.; Lee, D.S. Biodegradability and biocompatibility of a pH- and thermo-sensitive hydrogel formed from a sulfonamide-modified poly(ε-caprolactone-co-lactide)–poly(ethylene glycol)–poly(ε-caprolactone-co-lactide) block copolymer. Biomaterials 2006, 27, 5178–5185. [Google Scholar] [CrossRef] [PubMed]
- Cellesi, F.; Tirelli, N.; Hubbell, J.A. Materials for cell encapsulation via a new tandem approach combining reverse thermal gelation and covalent crosslinking. Macromol. Chem. Phys. 2002, 203, 1466–1472. [Google Scholar] [CrossRef]
- Jeong, B.; Bae, Y.H.; Kim, S.W. In situ gelation of PEG-PLGA-PEG triblock copolymer aqueous solutions and degradation thereof. J. Biomed. Mater. Res. 2000, 50, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Sieminski, A.L.; Semino, C.E.; Gong, H.; Kamm, R.D. Primary sequence of ionic self-assembling peptide gels affects endothelial cell adhesion and capillary morphogenesis. J. Biomed. Mater. Res. A 2008, 87, 494–504. [Google Scholar] [CrossRef]
- Paramonov, S.E.; Jun, H.W.; Hartgerink, J.D. Modulation of peptide-amphiphile nanofibers via phospholipid inclusions. Biomacromolecules 2006, 7, 24–6. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Maeda, M. DNA-responsive hydrogels that can shrink or swell. Biomacromolecules 2005, 6, 2927–2929. [Google Scholar] [CrossRef] [PubMed]
- Elisseeff, J.; McIntosh, W.; Anseth, K.; Riley, S.; Ragan, P.; Langer, R. Photoencapsulation of chondrocytes in poly(ethylene oxide)-based semi-interpenetrating networks. J. Biomed. Mater. Res. 2000, 51, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Varghese, S.; Hwang, N.S.; Canver, A.C.; Theprungsirikul, P.; Lin, D.W.; Elisseeff, J. Chondroitin sulfate based niches for chondrogenic differentiation of mesenchymal stem cells. Matrix Biology 2008, 27, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.D.; Tirelli, N.; Hubbell, J.A. Photopolymerized hyaluronic acid-based hydrogels and interpenetrating networks. Biomaterials 2003, 24, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Masters, K.S.; Shah, D.N.; Walker, G.; Leinwand, L.A.; Anseth, K.S. Designing scaffolds for valvular interstitial cells: cell adhesion and function on naturally derived materials. J. Biomed. Mater. Res. 2004, 71A, 172–180. [Google Scholar] [CrossRef]
- Leach, J.B.; Bivens, K.A.; Collins, C.N.; Schmidt, C.E. Development of photocrosslinkable hyaluronic acid polyethylene glycol-peptide composite hydrogels for soft tissue engineering. J. Biomed. Mater. Res. 2004, 70A, 74–82. [Google Scholar] [CrossRef]
- Jongpaiboonkit, L.; King, W.J.; Lyons, G.E.; Paguirigan, A.L.; Warrick, J.W.; Beebe, D.J.; Murphy, W.L. An adaptable hydrogel array format for 3-dimensional cell culture and analysis. Biomaterials 2008, 29, 3346–3356. [Google Scholar] [CrossRef] [PubMed]
- DeLong, S.A.; Gobin, A.S.; West, J.L. Covalent immobilization of RGDS on hydrogel surfaces to direct cell alignment and migration. J. Control. Rel. 2005, 109, 139–148. [Google Scholar] [CrossRef]
- Garagorri, N.; Fermanian, S.; Thibault, R.; Ambrose, W.M.; Schein, O.D.; Chakravarti, S.; Elisseeff, J. Keratocyte behavior in three-dimensional photopolymerizable poly(ethylene glycol) hydrogels. Acta Biomater. 2008, 4, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Bryant, S.J.; Anseth, K.S.; Lee, D.A.; Bader, D.L. Crosslinking density influences the morphology of chondrocytes photoencapsulated in PEG hydrogels during the application of compressive strain. J. Orthop. Res. 2004, 22, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.A.; Anseth, K.S. Encapsulating chondrocytes in copolymer gels: Bimodal degradation kinetics influence cell phenotype and extracellular matrix development. J. Biomed. Mater. Res. 2004, 70A, 560–568. [Google Scholar] [CrossRef]
- Bryant, S.J.; Bender, R.; Durand, K.L.; Anseth, K.S. Encapsulating chondrocytes in degrading PEG hydrogels with high modulus: engineering gel structural changes to facilitate cartilaginous tissue production. Biotechnol. Bioeng. 2004, 86, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Gao, C. Photoinitiating polymerization to prepare biocompatible chitosan hydrogels. J. Appl. Polym. Sci. 2008, 110, 1059–1067. [Google Scholar] [CrossRef]
- Hong, Y.; Mao, Z.; Wang, H.; Gao, C.; Shen, J. Covalently crosslinked chitosan hydrogel formed at neutral pH and body Temperature. J. Biomed. Mater. Res. 2006, 79A, 913–922. [Google Scholar] [CrossRef]
- Peter, S.J.; Yaszemski, M.J.; Suggs, L.J.; Payne, R.G.; Hayes, W.C.; Langer, R.; Unroe, M.; Alemany, L.B.; Engel, P.S.; Mikos, A.G. Characterization of partially saturated poly(propylene fumarate) for orthopaedic application. J. Biomater. Sci. Polym. Edn. 1997, 8, 893–904. [Google Scholar] [CrossRef]
- Peter, S.J.; Lu, L.C.; Mikos, A.G. Marrow stormal osteoblast function on a poly(propylene fumarate)/β-tricalcium phosphate biodegradable orthopaedic composite. Biomaterials 2000, 21, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- He, S.L.; Yaszemski, M.J.; Yasko, A.W.; Engel, P.S.; Mikos, A.G. Injectable biodegradable polymer composites based on poly(propylene fumarate) crosslinked with poly(ethylene glycol)-dimethacrylate. Biomaterials 2000, 21, 2389–2394. [Google Scholar] [CrossRef] [PubMed]
- Peter, S.J.; Miller, S.T.; Zhu, G.M.; Yasko, A.W.; Mikos, A.G. In vivo degradation of a poly(propylene fumarate)/β-tricalcium phosphate injectable composite scaffold. J. Biomed. Mater. Res. 1998, 41, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Ruhé, P.Q.; Mikos, A.G.; Jansen, J.A. In vivo bone and soft tissue response to injectable, biodegradable oligo(poly(ethylene glycol) fumarate) hydrogels. Biomaterials 2003, 24, 3201–3211. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.; Shin, H.; Shung, A.K.; Fisher, J.P.; Mikos, A.G. Synthesis and characterization of oligo(poly(ethylene glycol) fumarate) macromer. Macromolecules 2001, 34, 2839–2844. [Google Scholar] [CrossRef]
- Temenoff, J.S.; Park, H.; Jabbari, E.; Sheffield, T.L.; LeBaron, R.G.; Ambrose, C.G.; Mikos, A.G. In vitro osetogenic differentiation of marrow stromal cells encapsulated in biodegradable hydrogels. J. Biomed. Mater. Res. 2004, 70A, 235–244. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Tirelli, N.; Cerritelli, S.; Cavalli, L.; Hubbell, J.A. Systematic modulation of Michael-type reactivity of thiols through the use of charged amino acids. Bioconjugate Chem. 2001, 12, 1051–1056. [Google Scholar] [CrossRef]
- Park, Y.D.; Tirelli, N.; Hubbell, J.A. Photopolymerized hyaluronic acid-based hydrogels and interpenetrating networks. Biomaterials 2003, 24, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Pratt, A.B.; Weber, F.E.; Schmoekel, H.G.; Müller, R.; Hubbell, J.A. Synthetic extracellular matrices for in situ tissue engineering. Biotechnol. Bioeng. 2004, 86, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Vernon, B.; Tirelli, N.; Bächi, T.; Haldimann, D.; Hubbell, J.A. Water-borne, in situ crosslinked biomaterials from phase-segregated precursors. J. Biomed. Mater. Res. 2003, 64A, 447–456. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Hubbell, J.A. Synthesis and physicochemical characterization of end-linked poly(ethylene glycol)-co-peptide hydrogels formed by Michael-type addition. Biomacromolecules 2003, 4, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Lutolf, M.P.; Raeber, G.P.; Zisch, A.H.; Nicola, T.; Hubbell, J.A. Cell-responsive synthetic hydrogels. Adv. Mater. 2003, 15, 888–892. [Google Scholar] [CrossRef]
- Sung, H.W.; Huang, R.N.; Huang, L.L.H.; Tsai, C.C.; Chiu, C.T. Feasibility study of a natural crosslinking reagent for biological tissue fixation. J. Biomed. Mater. Res. 1998, 42, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Tsai, C.C.; Liang, H.C.; Sung, H.W. In vivo evaluation of cellular and acellular bovine pericardia fixed with a naturally occurring crosslinking agent (genipin). Biomaterials 2002, 23, 2447–2457. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.C.; Huang, R.N.; Sung, H.W.; Liang, H.C. In vitro evaluation of the genotoxicity of a naturally occurring crosslinking agent (genipin) for biologic tissue fixation. J. Biomed. Mater. Res. 2000, 52, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.S.; Yao, C.H.; Chen, Y.S.; Hsu, S.H. In vitro evaluation of degradation and cytotoxicity of a novel composite as a bone substitute. J. Biomed. Mater. Res. 2003, 67A, 1163–1169. [Google Scholar] [CrossRef]
- Mi, F.L.; Tan, Y.C.; Liang, H.C.; Huang, R.N.; Sung, H.W. In vitro evaluation of a chitosan membrane cross-linked with genipin. J. Biomater. Sci. Polym. Ed. 2001, 12, 835–850. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.F.; Yiu-Fai, N.G.; Pudney, D.A. Mechanism and kinetics of crosslinking reaction between biopolymers containing primary amine groups and genipin. J. Polym. Sci. A 2003, 41, 3941–3953. [Google Scholar] [CrossRef]
- Moffat, K.L.; Marra, K.G. Biodegradable poly(ethylene glycol) hydrogels crosslinked with genipin for tissue engineering applications. J. Biomed. Mater. Res. 2004, 71B, 181–187. [Google Scholar] [CrossRef]
- Ferretti, M.; Marra, K.G.; Kobayashi, K.; Defail, A.J.; Chu, C.R. Controlled in vivo degradation of genipin crosslinked polyethylene glycol hydrogels within osteochondral defects. Tissue Eng. 2006, 12, 2657–2663. [Google Scholar] [CrossRef] [PubMed]
- DeFail, A.J.; Chu, C.R.; Izzo, N.; Marra, K.G. Controlled release of bioactive TGF-β1 from microspheres embedded within biodegradable hydrogels. Biomaterials 2006, 27, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; DeFail, A.J.; Rubin, J.P.; Chu, C.R.; Marra, K.G. Novel multi-arm PEG-based hydrogels for tissue engineering. J. Biomed. Mater. Res. 2010, 92A, 979–987. [Google Scholar]
- Tan, H.; Chu, C.R.; Payne, K.A.; Marra, K.G. Injectable in situ forming biodegradable chitosan-hyaluronic acid based hydrogels for cartilage tissue engineering. Biomaterials 2009, 30, 2499–2506. [Google Scholar] [CrossRef] [PubMed]
- Maia, J.; Ferreira, L.; Carvalho, R.; Ramos, M.A.; Gil, M.H. Synthesis and characterization of new injectable and degradable dextran-based hydrogels. Polymer 2005, 46, 9604–9614. [Google Scholar] [CrossRef]
- Nishi, K.K.; Jayakrishnan, A. Preparation and in vitro evaluation of primaquine-conjugated gum arabic microspheres. Biomacromolecules 2004, 5, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.A.; Varghese, S.; Sharma, B.; Strehin, I.; Fermanian, S.; Gorham, J.; Fairbrother, D.H.; Cascio, B.; Elisseeff, J.H. Multifunctional chondroitin sulphate for cartilage tissue–biomaterial integration. Nat. Mater. 2007, 6, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Ruhela, D.; Riviere, K.; Szoka, F.C. Efficient synthesis of an aldehyde functionalized hyaluronic acid and its application in the preparation of hyaluronan-lipid conjugates. Bioconjug. Chem. 2006, 17, 1360–1363. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Yeo, Y.; Highley, C.B.; Bellas, E.; Benitez, C.A.; Kohane, D.S. The prevention of peritoneal adhesions by in situ cross-linking hydrogels of hyaluronic acid and cellulose derivatives. Biomaterials 2007, 28, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Yeo, Y.; Clifton, R.J.; Jiao, T.; Kohane, D.S.; Kobler, J.B.; Zeitels, S.M.; Langer, R. Hyaluronic acid-based microgels and microgel networks for vocal fold regeneration. Biomacromolecules 2006, 7, 3336–3344. [Google Scholar] [CrossRef] [PubMed]
- Holland, T.A.; Tessmar, J.K.; Tabata, Y.; Mikos, A.G. Transforming growth factor-beta 1 release from oligo(poly(ethylene glycol) fumarate) hydrogels in conditions that model the cartilage wound healing environment. J. Control. Rel. 2004, 94, 101–114. [Google Scholar] [CrossRef]
- Ferretti, M.; Marra, K.G.; Kobayashi, K.; DeFail, A.J.; Chu, C.R. Controlled in vivo degradation of genipin crosslinked poly(ethylene glycol) hydrogels within osteochondral defects. Tissue Eng. 2006, 12, 2657–2663. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Hollinger, J.O.; Marra, K.G. Controlled release from coated polymer microparticles embedded in tissue-engineered scaffolds. J. Drug Targeting 2001, 9, 431–438. [Google Scholar] [CrossRef]
- Marra, K.G.; Defail, A.J.; Clavijo-Alvarez, J.A.; Badylak, S.F.; Taieb, A.; Schipper, B.; Bennett, J.; Rubin, J.P. FGF-2 enhances vascularization for adipose tissue engineering. Plast. Reconstr. Surg. 2008, 121, 1153–1164. [Google Scholar] [CrossRef] [PubMed]
- Rubin, J.P.; Bennett, J.M.; Doctor, J.S.; Tebbets, B.M.; Marra, K.G. Collagenous microbeads as a scaffold for tissue engineering with adipose-derived stem cells. Plast.Reconstr. Surg. 2007, 120, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.; Snow, S.; McAlindon, T.E.; Kellingray, S.; Stuart, B.; Coggon, D. Risk factors for the incidence and progression of radiographic knee osteoarthritis. Arthritis Rheum 2000, 43, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.; Helmick, C.; Arnett, F.; Deyo, R.; Felson, D.; Giannini, E.; Heyse, S.; Hirsch, R.; Hochberg, M.; Hunder, G.; Liang, M.; Pillemer, S.; Steen, V.; Wolfe, F. Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthritis Rheum 1998, 41, 778–799. [Google Scholar] [CrossRef] [PubMed]
- Finch, G.D.; Dawe, C.J. Hemiatrophy. J. Pediatr. Orthop. 2003, 23, 99–101. [Google Scholar] [PubMed]
- Fokin, A.A.; Robicsek, F. Poland's syndrome revisited. Ann. Thorac. Surg. 2002, 74, 2218–2225. [Google Scholar] [CrossRef] [PubMed]
- Peer, L.A. The neglected free fat graft. Plast.Reconstr. Surg. 1956, 18, 233–250. [Google Scholar] [CrossRef]
- Hemmrich, K.; Van de Sijpe, K.; Rhodes, N.P.; Hunt, J.A.; Di Bartolo, C.; Pallua, N.; Blondeel, P.; von Heimburg, D. Autologous in vivo adipose tissue engineering in hyaluronan—based gels—a pilot study. J. Surg. Res. 2008, 144, 82–88. [Google Scholar] [CrossRef] [PubMed]
- West, D.C.; Kumar, S. Hyaluronan and angiogenesis. Ciba. Found Symp. 1989, 143, 187–201. [Google Scholar] [PubMed]
- Yuksel, E.; Weinfeld, A.B.; Cleek, R.; Wamsley, S.; Jensen, J.; Boutros, S.; Waugh, J.M.; Shenaq, S.M.; Spira, M. Increased free fat-graft survival with the long-term, local delivery of insulin, insulin-like growth factor-i, and basic fibroblast growth factor by plga/peg microspheres. Plast.Reconstr. Surg. 2000, 105, 1712–1720. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Ozeki, M.; Inamoto, T.; Tabata, Y. Adipose tissue engineering based on human preadipocytes combined with gelatin microspheres containing basic fibroblast growth factor. Biomaterials 2003, 2324, 2513–2521. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tan, H.; Marra, K.G. Injectable, Biodegradable Hydrogels for Tissue Engineering Applications. Materials 2010, 3, 1746-1767. https://doi.org/10.3390/ma3031746
Tan H, Marra KG. Injectable, Biodegradable Hydrogels for Tissue Engineering Applications. Materials. 2010; 3(3):1746-1767. https://doi.org/10.3390/ma3031746
Chicago/Turabian StyleTan, Huaping, and Kacey G. Marra. 2010. "Injectable, Biodegradable Hydrogels for Tissue Engineering Applications" Materials 3, no. 3: 1746-1767. https://doi.org/10.3390/ma3031746
APA StyleTan, H., & Marra, K. G. (2010). Injectable, Biodegradable Hydrogels for Tissue Engineering Applications. Materials, 3(3), 1746-1767. https://doi.org/10.3390/ma3031746




